Philips Takes EUR250 Million Hit on US Recall of Sleep, Respiratory-Care Products
14 June 2021 - 4:40PM
Dow Jones News
By Adria Calatayud
Koninklijke Philips NV said Monday that it is starting a recall
notification for the U.S. in relation to certain sleep and
respiratory-care devices, leading it to book 250 million euros
($302.8 million) in additional costs.
The Dutch medical-technology company said it identified
potential health risks related to a sound-abatement foam component
in certain devices. The majority of the affected devices are in the
first-generation DreamStation sleep and respiratory-care product
family, the company said.
Philips issued a recall notification for the U.S. and a
field-safety notice for the rest of the world in consultation with
regulatory agencies.
The company said it will replace the current sound-abatement
foam with a new material and modify the first-generation
DreamStation product families with a different foam.
As a result, Philips said it expects an increase of EUR250
million in costs, in addition to the provision of EUR250 million it
booked in its first-quarter results.
However, the company said its 2021 guidance for adjusted
earnings before interest, taxes and amortization margin and
comparable sales growth remains unchanged as revenue headwinds in
the sleep and respiratory-care business will be offset by strength
in other segments.
Write to Adria Calatayud at adria.calatayud@dowjones.com
(END) Dow Jones Newswires
June 14, 2021 02:33 ET (06:33 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
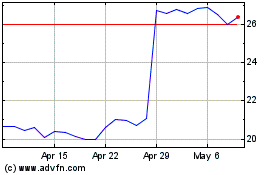
Koninklijke Philips NV (NYSE:PHG)
Historical Stock Chart
From Mar 2024 to Apr 2024
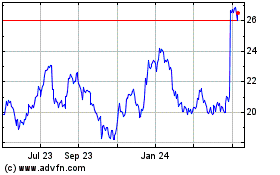
Koninklijke Philips NV (NYSE:PHG)
Historical Stock Chart
From Apr 2023 to Apr 2024