Acadia Pharmaceuticals Shares Tumble Premarket After FDA Panel Vote
21 June 2022 - 10:29PM
Dow Jones News
By Colin Kellaher
Shares of Acadia Pharmaceuticals Inc. plunged more than 30% in
premarket trading Tuesday after a negative U.S. Food and Drug
Administration advisory committee vote on expanded approval of its
antipsychotic Nuplazid.
The San Diego company, which is seeking an FDA green light for
Nuplazid for the treatment of hallucinations and delusions
associated with Alzheimer's disease psychosis, late Friday said the
committee voted 9-to-3 that the evidence presented doesn't support
a conclusion that the drug is effective for the proposed
population.
The FDA, which has set a target action date of Aug. 4 for
Acadia's application, isn't bound to follow the advice of its
advisory committees, but it usually does.
Analysts at Mizuho Securities said in a research note that it is
unlikely the FDA will approve the application, and that they expect
the agency will request another study.
Acadia said it is disappointed with the vote, and that it
believes there is substantial evidence across multiple independent
clinical studies and endpoints that supports the efficacy of
Nuplazid in Alzheimer's disease psychosis, for which there are
currently no FDA-approved treatments.
The FDA in 2016 approved Nuplazid to treat hallucinations and
delusions associated with Parkinson's disease psychosis.
Acadia shares, which closed Friday at $19.51, were recently
changing hands at $13.63, down 30.1%, in premarket trading.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
June 21, 2022 08:14 ET (12:14 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
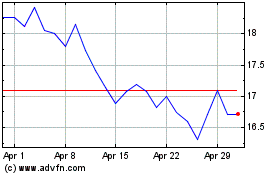
Acadia Pharmaceuticals (NASDAQ:ACAD)
Historical Stock Chart
From Mar 2024 to Apr 2024
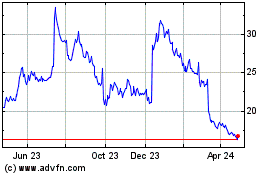
Acadia Pharmaceuticals (NASDAQ:ACAD)
Historical Stock Chart
From Apr 2023 to Apr 2024