Acadia Pharma Shares Touch 52-Week Low After FDA AdCom Vote
22 June 2022 - 1:31AM
Dow Jones News
By Colin Kellaher
Shares of Acadia Pharmaceuticals Inc. lost a third of their
value and hit a new 52-week low after the latest setback in the
pharmaceutical company's efforts to win expanded U.S. Food and Drug
Administration approval for its Nuplazid antipsychotic drug.
Acadia late Friday said an FDA advisory committee voted 9-to-3
that the evidence presented doesn't support a conclusion that
Nuplazid is effective for the treatment of hallucinations and
delusions associated with Alzheimer's disease psychosis.
Nuplazid has been approved in the U.S. since 2016 to treat
hallucinations and delusions associated with Parkinson's disease
psychosis, and the FDA is weighing Acadia's request for expanded
use in patients with Alzheimer's, with a decision expected by Aug.
4.
Wall Street analysts in general believe that the advisory
committee vote means the FDA will once again reject the
application.
Acadia in 2020 filed for FDA approval of Nuplazid to treat
hallucinations and delusions associated with dementia-related
psychosis, but the agency turned the application away in April
2021, citing, among other things, a lack of statistical
significance in some of the subgroups of dementia.
The company resubmitted its application in February of this
year, this time seeking approval in patients with Alzheimer's
disease.
Acadia said it is disappointed with the vote, and that it
believes there is substantial evidence across multiple independent
clinical studies and endpoints that supports the efficacy of
Nuplazid in Alzheimer's disease psychosis.
The FDA isn't required to follow the advice of its advisory
committees, but it usually does, and analysts at Mizuho Securities
said in a research note that they expect the agency will once again
reject Acadia's application and request another study.
The negative advisory committee vote prompted several Wall
Street firms to cut their price targets on Acadia shares, as many
now expect the FDA to issue a so-called complete response letter,
or CRL, by Aug. 4, indicating that the agency won't approve the
application in its current form.
On a potentially positive note, Citi analyst Neena Bitritto-Garg
said a CRL could make Acadia a potential takeover target for
biopharmaceutical companies looking to boost their revenue.
Nuplazid sales exceeded $480 million last year, and the company has
forecast 2022 sales of $510 million to $560 million.
Acadia shares were recently changing hands at $12.64, down
35.2%, after hitting a 52-week low of $12.24 earlier in the
session.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
June 21, 2022 11:16 ET (15:16 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
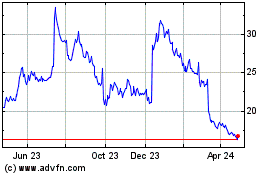
Acadia Pharmaceuticals (NASDAQ:ACAD)
Historical Stock Chart
From Mar 2024 to Apr 2024
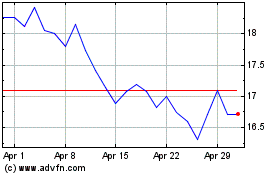
Acadia Pharmaceuticals (NASDAQ:ACAD)
Historical Stock Chart
From Apr 2023 to Apr 2024