Agios Launches “Red Cell Revolution” in Recognition of Rare Disease Day
28 February 2023 - 11:00PM

Agios Pharmaceuticals, Inc. (Nasdaq: AGIO), a leader in the field
of cellular metabolism pioneering therapies for rare diseases,
today announced the launch of a multi-stakeholder advocacy advisory
council for hemolytic anemias including PK deficiency, thalassemia
and sickle cell disease. The council will bring together patients,
caregivers and physicians from these communities, which share
commonalities in underlying disease pathology, difficulties in
transitioning from pediatric to adult care and immense quality of
life concerns, to generate published evidence and meaningful
solutions that are relevant across disease areas and to create a
platform to raise awareness about the needs and experiences of
people touched by these diseases.
“The Red Cell Revolution advisory council is designed to build
connections among PK deficiency, thalassemia and sickle cell
disease advocates and thought leaders, and to generate insights to
raise disease awareness, inform Agios’ mission and contribute to
shaping the broader hemolytic anemia field through evidence-based
patient advocacy,” said Sarah Gheuens, M.D., Ph.D., chief medical
officer and head of R&D at Agios. “On Rare Disease Day, we
celebrate the power of various rare disease communities coming
together, finding common ground and amplifying each other’s voices;
our hope is that the Red Cell Revolution can accomplish similar
goals and have an important impact for rare blood disorder
communities.”
“The Red Cell Revolution is a real opportunity to bring together
great minds and passionate individuals, including people who live
every day with these conditions that have more similarities than
differences,” said Biree Andemariam, M.D., hematologist and
founding director of the New England Sickle Cell Institute (NESCI)
at the University of Connecticut Health Center, and council member
of the Red Cell Revolution. “Together, we can be a powerful,
unified voice to drive change not only in the U.S., but also
throughout the world, and to strive for these underserved
conditions to get the same prioritization, innovation, access and
compassionate care that more common illnesses get. I’m grateful to
Agios for spearheading this initiative and facilitating these
connections which can fuel positive changes for people with sickle
cell disease, thalassemia and PK deficiency.”
To learn more about the Red Cell Revolution, watch this video or
listen to this podcast episode, which debuted yesterday across
three Agios-sponsored podcasts, including Just Listen: Voices of PK
Deficiency, Thal Pals: The Alpha Beta Revolution and Cheat Codes: A
Sickle Cell Podcast.
The Agios team is also supporting rare disease communities
throughout the month of February as a sponsor of the following
initiatives:
- Uplifting Athletes: Health Equity Initiative, which supports
rare disease researchers from underrepresented backgrounds
- MassBio: Rare Disease Day 2023
- National Organization for Rare Disorders: Rare Disease Day
2023
About AgiosAgios is a biopharmaceutical company
that is fueled by connections. The Agios team cultivates strong
bonds with patient communities, healthcare professionals, partners
and colleagues to discover, develop and deliver therapies for rare
diseases. In the U.S., Agios markets a first-in-class pyruvate
kinase (PK) activator for adults with PK deficiency, the first
disease-modifying therapy for this rare, lifelong, debilitating
hemolytic anemia. Building on the company's leadership in the field
of cellular metabolism, Agios is advancing a robust clinical
pipeline of investigational medicines with programs in alpha- and
beta-thalassemia, sickle cell disease, pediatric PK deficiency and
MDS-associated anemia. In addition to its clinical pipeline, Agios
has a PAH stabilizer in preclinical development as a potential
treatment for phenylketonuria (PKU) and deep scientific expertise
in classical hematology. For more information, please visit the
company’s website at www.agios.com.
Cautionary Note Regarding Forward-Looking
StatementsThis press release contains forward-looking
statements within the meaning of The Private Securities Litigation
Reform Act of 1995. Such forward-looking statements include those
regarding the potential benefits of Agios’ strategic plans and
focus. The words “anticipate,” “expect,” “goal,” “hope,”
“milestone,” “plan,” “potential,” “possible,” “strategy,” “will,”
“vision,” and similar expressions are intended to identify
forward-looking statements, although not all forward-looking
statements contain these identifying words. Such statements are
subject to numerous important factors, risks and uncertainties that
may cause actual events or results to differ materially from Agios’
current expectations and beliefs. For example, there can be no
guarantee that any product candidate Agios is developing will
successfully commence or complete necessary preclinical and
clinical development phases, or that development of any of Agios’
product candidates will successfully continue. There can be no
guarantee that any positive developments in Agios’ business will
result in stock price appreciation. Management's expectations and,
therefore, any forward-looking statements in this press release
could also be affected by risks and uncertainties relating to a
number of other important factors, including, without limitation:
risks and uncertainties related to the impact of the COVID-19
pandemic to Agios’ business, operations, strategy, goals and
anticipated milestones, including its ongoing and planned research
activities, ability to conduct ongoing and planned clinical trials,
clinical supply of current or future drug candidates, commercial
supply of current or future approved products, and launching,
marketing and selling current or future approved products; Agios’
results of clinical trials and preclinical studies, including
subsequent analysis of existing data and new data received from
ongoing and future studies; the content and timing of decisions
made by the U.S. FDA, the EMA or other regulatory authorities,
investigational review boards at clinical trial sites and
publication review bodies; Agios’ ability to obtain and maintain
requisite regulatory approvals and to enroll patients in its
planned clinical trials; unplanned cash requirements and
expenditures; competitive factors; Agios' ability to obtain,
maintain and enforce patent and other intellectual property
protection for any product candidates it is developing; Agios’
ability to maintain key collaborations; the failure of Agios to
receive milestone or royalty payments related to the sale of its
oncology business, the uncertainty of the timing of any receipt of
any such payments, and the uncertainty of the results and
effectiveness of the use of proceeds from the transaction with
Servier; and general economic and market conditions. These and
other risks are described in greater detail under the caption "Risk
Factors" included in Agios’ public filings with the Securities and
Exchange Commission. Any forward-looking statements contained in
this press release speak only as of the date hereof, and Agios
expressly disclaims any obligation to update any forward-looking
statements, whether as a result of new information, future events
or otherwise, except as required by law.
Investor & Media Contact
Jessica RennekampSenior Director, Corporate
CommunicationsJessica.Rennekamp@agios.com
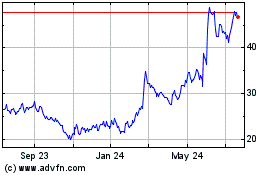
Agios Pharmaceuticals (NASDAQ:AGIO)
Historical Stock Chart
From Mar 2024 to Apr 2024
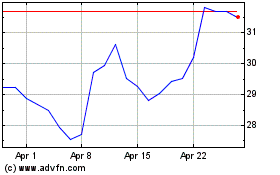
Agios Pharmaceuticals (NASDAQ:AGIO)
Historical Stock Chart
From Apr 2023 to Apr 2024