Alpine Immune End Davoceticept Study Enrollment After 2nd Patient Death
24 October 2022 - 11:21PM
Dow Jones News
By Colin Kellaher
Alpine Immune Sciences Inc. on Monday said it has voluntarily
ended enrollment in a pair of studies of its davoceticept drug
candidate following the death of a second patient in one of the
studies.
The Seattle clinical-stage immunotherapy company said the
patient, who was enrolled in a study of davoceticept in combination
with Merck & Co.'s cancer drug Keytruda, died of cardiogenic
shock after receiving one dose of each drug.
The U.S. Food and Drug Administration earlier this year had
placed a partial clinical hold on the combination study due to a
death attributed to cardiogenic shock, but the hold was lifted in
early May.
Alpine said it is conducting a comprehensive assessment of all
participants in both studies, and that it will now focus on the
pursuit of expedited development plans for ALPN-303 in multiple
autoimmune and inflammatory indications.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
October 24, 2022 08:06 ET (12:06 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
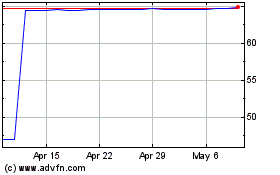
Alpine Immune Sciences (NASDAQ:ALPN)
Historical Stock Chart
From Mar 2024 to Apr 2024
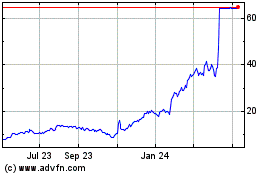
Alpine Immune Sciences (NASDAQ:ALPN)
Historical Stock Chart
From Apr 2023 to Apr 2024