AlloVir Gets FDA Regenerative Medicine Advanced Therapy Designation for Posoleucel
06 January 2022 - 12:21AM
Dow Jones News
By Michael Dabaie
AlloVir Inc. said the U.S. Food and Drug Administration granted
its lead multi-virus specific T cell therapy posoleucel
Regenerative Medicine Advanced Therapy designation.
The designation is for the treatment of adenovirus infection
following allogeneic hematopoietic stem cell transplant and is
based on results from a Phase 2 study.
RMAT designation recognizes the potential for posoleucel to
address the unmet medical need posed by AdV, a potentially
life-threatening condition with no approved treatment options, the
late-clinical stage company said.
This designation enables early interactions with the FDA to
discuss clinical trial design and other actions to expedite
development and review.
The FDA previously granted RMAT designation to posoleucel for
the treatment of hemorrhagic cystitis caused by BK virus in adults
and children following allo-HCT.
A Phase 3 study of posoleucel for the treatment of AdV viremia
is now enrolling pediatric and adult patients following allo-HCT.
This study is the second Phase 3 registrational study of
posoleucel.
The company also said it initiated a Phase 1/2 clinical trial of
ALVR106 for the treatment of infections caused by human
metapneumovirus, influenza, parainfluenza virus and respiratory
syncytial virus.
Write to Michael Dabaie at michael.dabaie@wsj.com
(END) Dow Jones Newswires
January 05, 2022 08:06 ET (13:06 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
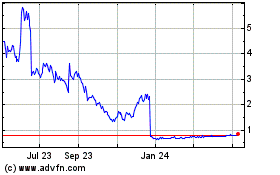
AlloVir (NASDAQ:ALVR)
Historical Stock Chart
From Mar 2024 to Apr 2024
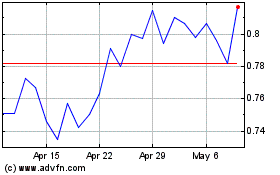
AlloVir (NASDAQ:ALVR)
Historical Stock Chart
From Apr 2023 to Apr 2024