AN2 Therapeutics Provides Update on Epetraborole Ex-U.S. Development Plan in Treatment-Refractory MAC Lung Disease
06 October 2022 - 10:00PM

AN2 Therapeutics, Inc. (Nasdaq: ANTX), a clinical-stage
biopharmaceutical company focused on developing treatments for
rare, chronic, and serious infectious diseases with high unmet
needs, today provided an update on the ex-U.S. development plan for
epetraborole in treatment-refractory Mycobacterium
avium complex (MAC) lung disease, the most common form of
non-tuberculous mycobacterial (NTM) lung disease.
“We believe that epetraborole has the potential to provide
significant benefit to patients with NTM lung disease and are
pleased with the progress we’ve made globally across our
epetraborole development program, including, most recently, the
results from our Phase 1 trial in Japan (EBO-103) that further
validate data from our previous studies and support our recommended
Phase 2/3 dose in the ongoing EBO-301 pivotal trial in
treatment-refractory MAC patients,” said Eric Easom, Co-Founder,
President and Chief Executive Officer of AN2 Therapeutics. “These
data are an important step towards including patients in our
ongoing EBO-301 pivotal Phase 2/3 trial from Japan, a country with
one of the highest prevalence rates of MAC lung disease in the
world.”
Easom added, “In addition, the European Commission’s decision to
adopt the orphan medicinal product designation for epetraborole for
NTM lung disease is an important milestone. After discussion with
the European Medicines Agency, we believe the clearest path to
address the unmet medical need in Europe is through further
development of epetraborole in patients with treatment-naïve MAC
lung disease, which is in scope of this designation. We expect to
provide further updates on our approach to this important patient
population in the future.”
Results from Phase 1 PK/Safety Study of Oral
Epetraborole in Japan (EBO-103)This Phase 1 study was
designed to evaluate the pharmacokinetics, safety, and tolerability
of oral epetraborole administered as a single 500 mg dose to
healthy Japanese volunteers with different alcohol dehydrogenase
(ADH) genotypes. Results of the study showed that no subjects
experienced treatment-emergent adverse events and support use of
AN2’s recommended dose in Japanese patients enrolled in its ongoing
Phase 2/3 study. Epetraborole exposures were similar amongst
patients with different ADH genotypes, and consistent with those
observed in non-Japanese subjects in a previously reported Phase 1
study (EBO-101).
European Union Orphan Medicinal Product
Designation In August, the Company received the orphan
medicinal product designation for epetraborole in NTM lung disease
from the European Commission, which grants orphan medicinal product
designation to medicines intended for the treatment, prevention or
diagnosis of a life-threatening or very serious condition, with a
prevalence in the European Union of not more than five in 10,000
people, and where either no satisfactory method of diagnosis,
prevention or treatment of the condition in question exists, or if
such method exists that the medicinal product will be of
significant benefit to those affected by that condition. The
designation provides various incentives, including ten-year market
exclusivity upon regulatory approval, if received, fee reductions,
and access to protocol assistance.
About AN2 Therapeutics, Inc. AN2 Therapeutics,
Inc. is a clinical-stage biopharmaceutical company developing
treatments for rare, chronic, and serious infectious diseases with
high unmet needs. AN2 is developing epetraborole, a once-daily,
oral treatment with a novel mechanism of action for patients with
NTM lung disease, a rare, chronic, and progressive infectious
disease caused by bacteria, known as mycobacteria, that leads to
irreversible lung damage and can be fatal. For more information,
please visit our website at www.an2therapeutics.com.
Forward-Looking Statements This press release
contains forward-looking statements within the meaning of the
Private Securities Litigation Reform Act of 1995. Forward-looking
statements expressed or implied in this press release include, but
are not limited to, statements regarding: AN2's ability to provide
significant benefits to patients with NTM lung disease, anticipated
progress, business plans, business strategy and planned clinical
trials; AN2's plans to include Japan in its Phase 2/3 pivotal
clinical trial and the timing thereof; the potential clinical
benefits and therapeutic potential of epetraborole; the ability to
receive market exclusivity in the European Union; the ability of
AN2’s clinical trials to support regulatory approval; and other
statements that are not historical fact. These statements are based
on AN2's current plans, objectives, estimates, expectations and
intentions, are not guarantees of future performance and inherently
involve significant risks and uncertainties. Actual results and the
timing of events could differ materially from those anticipated in
such forward-looking statements as a result of these risks and
uncertainties, which include those described under the heading
“Risk Factors” in AN2's Quarterly Report on Form 10-Q for the
quarter ended June 30, 2022 filed with the U.S. Securities and
Exchange Commission (SEC). These filings, when available, are
available on the investor relations section of our website at
investor.an2therapeutics.com and on the SEC's website at
www.sec.gov. Forward-looking statements contained in this press
release are made as of this date, and AN2 undertakes no duty to
update such information except as required under applicable
law.
COMPANY CONTACT:Lucy O. DayChief Financial
Officerl.day@an2therapeutics.com
INVESTOR AND MEDIA CONTACT: Anne
Bowdidge ir@an2therapeutics.com
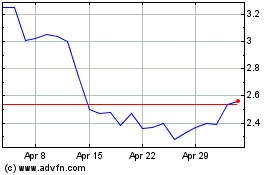
AN2 Therapeutics (NASDAQ:ANTX)
Historical Stock Chart
From Mar 2024 to Apr 2024
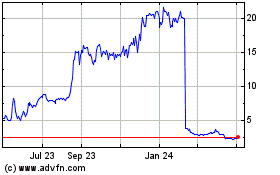
AN2 Therapeutics (NASDAQ:ANTX)
Historical Stock Chart
From Apr 2023 to Apr 2024