Aurinia Announces LUPKYNIS® (voclosporin) Patent Challenge Settlement Reached With Sun Pharmaceuticals
04 January 2023 - 1:12AM
Business Wire
- Sun Pharmaceuticals and Aurinia agreed to
file a joint motion to dismiss Inter Parties Review of Patent No.
10,286,036
- Parties have settled the patent infringement
claim against Sun’s CEQUA® product
- Settlements are Contingent Upon U.S. Patent
Trial and Appeal Board (PTAB) Terminating Inter Partes Review
Aurinia Pharmaceuticals Inc. (NASDAQ: AUPH) (Aurinia or the
Company) announces it has entered into a settlement agreement with
Sun Pharmaceutical Industries, Inc., Sun Pharmaceuticals
Industries, Ltd., and Sun Pharma Global FZE (collectively, Sun
Pharmaceuticals) that involves both Aurinia and Sun Pharmaceuticals
filing a joint motion to terminate the ongoing Inter Partes Review
(IPR) directed at Aurinia’s U.S. Patent No. 10,286,036, as well as
settlement of the ongoing patent infringement litigation in the
United States (U.S.) related to Sun’s CEQUA® product. Under the
settlement agreement, which is contingent upon the U.S. Patent
Trial and Appeal Board approving the joint motion to terminate the
IPR, both parties agreed to dismiss their claims and counterclaims
against each other and cease bringing any future further action
against the other.
About LUPKYNIS LUPKYNIS is the first FDA-approved oral
therapy for lupus nephritis (LN). LN causes irreversible kidney
damage and significantly increases the risk of kidney failure,
cardiac events, and death. It is one of the most serious and common
complications of the autoimmune disease systemic lupus
erythematosus (SLE). LUPKYNIS is in the United States (U.S.) and
across the European Union (E.U).
About Lupus Nephritis LN is a serious manifestation of
SLE, a chronic and complex autoimmune disease. About
200,000-300,000 people live with SLE in the U.S. and about
one-third of these people are diagnosed with lupus nephritis at the
time of their SLE diagnosis. About 50 percent of all people with
SLE may develop lupus nephritis. If poorly controlled, LN can lead
to permanent and irreversible tissue damage within the kidney.
Black and Asian individuals with SLE are four times more likely to
develop LN and individuals of Hispanic ancestry are approximately
twice as likely to develop the disease when compared with Caucasian
individuals. Black and Hispanic individuals with SLE also tend to
develop LN earlier and have poorer outcomes when compared to
Caucasian individuals.
About Aurinia Aurinia Pharmaceuticals is a fully
integrated biopharmaceutical company focused on delivering
therapies to treat targeted patient populations that are impacted
by serious diseases with a high unmet medical need. In January
2021, the Company introduced LUPKYNIS® (voclosporin), the first
FDA-approved oral therapy for the treatment of adult patients with
active lupus nephritis (LN). The Company’s head office is in
Victoria, British Columbia, its U.S. commercial hub is in
Rockville, Maryland, and the Company focuses its development
efforts globally.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230103005512/en/
Investor/Media Contact: Aurinia@westwicke.com
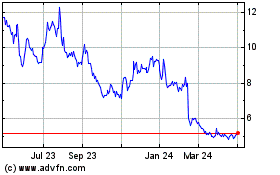
Aurinia Pharmaceuticals (NASDAQ:AUPH)
Historical Stock Chart
From Mar 2024 to Apr 2024
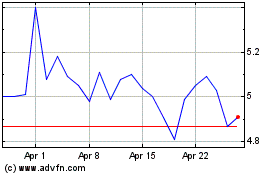
Aurinia Pharmaceuticals (NASDAQ:AUPH)
Historical Stock Chart
From Apr 2023 to Apr 2024