Applied
UV Provides Shareholder Letter
Mount
Vernon, N.Y. // April 7, 2021 – Applied UV, Inc. (NasdaqCM: AUVI)
(“Applied UV” or the “Company”), a pathogen elimination technology company that applies the power of narrow-range
ultraviolet light (“UVC”) for surface areas and catalytic bioconversion technology for air purification to destroy pathogens
safely, thoroughly, and automatically, announces Shareholder letter.
Dear
Applied UV Inc., Shareholders,
I
am writing to reflect on the Company’s 2021 accomplishments and provide to the extent possible, insights into our focus for 2022.
Despite the pandemic that crippled the world, 2021 saw the company complete significant positive milestones that, in my opinion, were
truly historic in nature. Given the herculean effort put forth by our entire Team and Board, what we accomplished as a public company
since the completion of our Initial Public Offering almost 18 months ago, in my opinion, is not currently being recognized in our valuation.
Why
we are we here! – The Center for Disease Control states that 1 in 25 patients have at least one Hospital Associated Infection (HAI)
annually and that 3 million serious infections occur every year in long-term care facilities. Scientists globally have been advocating
improving air quality post pandemic, significantly boosting global adoption to control airborne pathogen transmission. Governments globally
mandating health agencies to address air quality via grants and mechanisms to ease visitation and protect facilities against future pathogens
(Centers for Medicare and Medicaid Services – February 2022 Long- term Care Initiative). Indoor air quality has become an
even more important issue as world economies start the recovery process. In 2021, 39 scientists reiterated
the need for a "paradigm shift" and called for improvements in, "how we view and address the transmission of respiratory
infections to protect against unnecessary suffering and economic losses." Most recently, on March 22, 2022, the Federal Government
through the Environmental Protection Agency (EPA) issued “Clean Air Guidelines” aimed at improving air quality in public
spaces to prevent the transmission and spread of airborne related pathogens. In addition, on March 25, 2022, the White House announced
its “Clean Air in Buildings Challenge” through the Office of Science and Technology Policy. This initiative is seen to emphasize
what scientists globally have been championing. In its launch, the White House, explained “Research shows changing the air in a
room multiple times an hour with filtered or clean outdoor air — using a window fan, by using higher MERV filters in an Heating,
Ventilation, and Air Conditioning (HVAC) system, using portable air cleaning devices, and even just opening a window — can reduce
the risk of COVID-19 transmission — with studies showing five air changes an hour reduce transmission risk by 50 percent. And,
improving indoor air has benefits beyond COVID-19: it will reduce the risk of getting the flu, a common cold, or other diseases spread
by air, and lead to better overall health outcomes.” Lastly, in addition to this, the global
air purifier market size
is set to grow exponentially. It was valued at $9.24 billion in 2021 and is predicted to grow to approximately $22.84 billion by
2030. According to Precedence Research, the immense demand for air purification and sterilization in the US will be driven by the commercial
sector. While these are merely statistics, our mandate and mission is to rapidly build a science-based pathogen elimination platform
that is safe, effective and simple, that protects businesses, facilities and people around the globe.
What
we have accomplished – Since our inception, the Company has completed a $5.8M IPO, a
$7.3M
follow-on offering, a $13.8M Series Preferred and, a $9.2M Secondary offering all of which enabled the completion of three acquisitions
(Akida Holdings, Kes Science and Technologies and Scientific Air Management) which position AUVI as a fully integrated company offering
total pathogen disinfection platforms (Air & Surface). With the acquisitions complete, our wholly owned subsidiary, SteriLumen addresses
the ever-changing challenges of air and surface pathogens, healthcare-acquired infections (“HAIs”), hospital borne infections
(HBI’s), protecting businesses, the facilities they occupy, their employees, and consumers who frequent them. The Company’s
products are targeted for use in facilities that have high customer touch and turnover which, include hospitals, hotels, commercial facilities,
sports arenas, dental offices, schools, food processing, post-harvest, cannabis grow facilities, long-term care and other public spaces.
As we continue to grow and build out our air purification and surface disinfection platform portfolio, we are continually looking for
synergistic, attractive air purification businesses to allow us to gain further market penetration and share. We also will seek out low-cost
opportunities to bolster our legacy hospitality business, Munnworks. The recent Visionmark (www.visionmarknyc.com)
asset acquisition is an example of just that, as it expands our reach further into the luxury hospitality world of hotel construction
and remodeling beyond its core mirror business while also potentially contributing top line revenues to Munnworks.
The
Company’s product portfolio is currently used by globally recognized names including Walmart, Whole Foods, SuperValue, Delmonte,
Esmeralda, Joel Gott Wines, Athena Healthcare, NYC Health and Hospitals, Kaiser Permanente, Advent Health, University Rochester Medical
Center and Baptist Health South Florida. This past year, the Company’s product portfolio expanded its reach and deployed its air
purification products into The Palace Versailles, Uruguayan School Systems, Tennessee Department of Corrections, Armed Forces Research
Institute of Medical Sciences (AFRIMS), US Army Aberdeen Proving Grounds, Schools throughout South Korea, NYU Medical Center, University
of Rochester Medical Center, The Federal VA Hospital System, Baptist Health South, Kaiser Permanente Medical of California, and, New
York Health + Hospital Corp.
The
Company works with a global base of distributors to sell both its air purification and surface disinfection products as well as the Munnworks
product lines. The past year, the Company has signed distribution agreements covering Africa (360BioPharma), US Healthcare (Axis), Lootah
Batta Water and Environment (United Arab Emirates), and Plandent Division, a wholly owned subsidiary of Planmeca Oy (Scandinavia). The
Company plans to continue to expand its global distribution base of significant breadth and scale to introduce the entire air purification
and surface disinfection product lines to new markets. These include Long-Term Care, healthcare, prisons, cannabis grow facilities, wineries,
food preservation, and logistics. By leveraging the synergies of the acquired companies, taking advantage of global cross sales and marketing
initiatives through its international distribution base, the Company plans to launch one of the largest, targeted, multi-faceted marketing
campaigns in our short history led by a Fortune 500 executive with extensive experience in this regard.
The
Company’s product portfolio is one of the only research-backed, clinically proven pure-play air and surface disinfection
technology companies with international distribution and globally recognized end users, with product developed for NASA. In addition
to the numerous recognized research institutions and globally recognized names who published the reports that were completed by the
acquired companies, Airocide was independently proven to kill SARS, MERSA and Anthrax. This past year saw independent testing
verifying that the Company’s air purification (Airocide) and surface disinfection (Lumicide) products were proven to kill both
Candida Auris (Resinnova Laboratories) and SARS CoV-2 (COVID-19) (MRIGlobal).
It
is important to note, we are driven by a single competitive principal, which is that we have a patented, disruptive air purification
and surface disinfection technology platform that is differentiated by the global customers who use it to protect their facilities and
customers and, the independent studies validating its efficacy. Our enthusiasm could not be any higher about the impact that our air
purification and surface disinfection platform could have as the world’s businesses and economies re-open from this three-year
pandemic armed with a technology that can prevent this type of global disruption from re- occurring in the future.
We
have updated our website with the most recent Investor Presentation that covers more of our plans for 2022. You can also find more information
maintained at www.applieduvinc.com
In
closing, as true of rapidly growing companies, we are well along the learning curve, are extremely mindful of the challenges and grateful
for the opportunities. I can assure you that the entire team and board are solely focused towards refining our path forward and the result
will be a stronger company with a board and senior management team possessing the expertise to navigate this exciting next chapter in
our corporate history. On behalf of the entire management team and board, thank you for your continued support. As we continue this journey,
it is our sincere hope and expectation that we will unlock shareholder value and the truest potential through focused execution. Please
feel free to contact me directly with any questions.
Best Regards,
Max Munn
Interim
Chief Executive Officer / Director
About
Applied UV
Applied
UV is focused on the development and acquisition of technology that address infection control in the healthcare, hospitality,
commercial and municipal markets. The Company has two wholly owned subsidiaries - SteriLumen, Inc. (“SteriLumen”) and
Munn Works, LLC (“Munn Works”). SteriLumen’s connected platform for Data Driven Disinfection™ applies the
power of ultraviolet light (UVC) to destroy pathogens safely, thoroughly, and automatically, addressing the challenge of
healthcare-acquired infections ("HAIs"). Targeted for use in facilities that have high customer turnover such as
hospitals, hotels, commercial facilities, and other public spaces, the Company’s Lumicide™ platform uses UVC LEDs in
several patented designs for infection control in and around high-traffic areas, including sinks and restrooms, killing bacteria,
viruses, and other pathogens residing on hard surfaces within devices’ proximity. The Company’s patented in-drain
disinfection device, Lumicide Drain, is the only product on the market that addresses this critical pathogen intensive location.
SteriLumen’s Airocide® air purification devices are research backed, clinically proven, and developed for NASA with
assistance from the University of Wisconsin. Airocide® is listed as an FDA Class II Medical device, utilizes a proprietary
photocatalytic (PCO) bioconversion technology that draws air into a reaction chamber that converts damaging molds, microorganisms,
dangerous airborne pathogens, destructive VOCs, allergens, odors and biological gasses into harmless water vapor and green carbon
dioxide without producing ozone or other harmful byproducts. Airocide® applications include healthcare, hospitality, grocery
chains, wine making facilities, commercial real estate, schools, dental offices, post-harvest, grocery, cannabis facilities and
homes. For more information about Applied UV, Inc., and its subsidiaries, please visit the following websites: https://www.applieduvinc.com/; https://sterilumen.com/; https://www.airocide.com https://kesscience.com; https://scientificairmanagement.com www.airoclean420.com
and, https://munnworks.com/.
Forward-Looking
Statements
The
information contained herein may contain “forward-looking statements.” Forward-looking statements reflect the current view
about future events. When used in this press release, the words “anticipate,” “believe,” “estimate,”
“expect,” “future,” “intend,” “plan,” or the negative of these terms and similar expressions,
as they relate to us or our management, identify forward-looking statements. Such statements include, but are not limited to, statements
contained in this press release relating to the view of management of Applied UV concerning its business strategy, future operating results
and liquidity and capital resources outlook. Forward-looking statements are based on the Company’s current expectations and assumptions
regarding its business, the economy and other future conditions. Because forward–looking statements relate to the future, they
are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict. The Company’s actual results
may differ materially from those contemplated by the forward-looking statements. They are neither statements of historical fact nor guarantees
of assurance of future performance. We caution you therefore against relying on any of these forward-looking statements. Factors or events
that could cause the Company’s actual results to differ may emerge from time to time, and it is not possible for the Company to
predict all of them. The Company cannot guarantee future results, levels of activity, performance, or achievements. Except as required
by applicable law, including the securities laws of the United States, the Company does not intend to update any of the forward-looking
statements to conform these statements to actual results.
For
additional Company Information: Applied UV Inc.
Max
Munn
Applied
UV Interim CEO max.munn@sterilumen.com
Contact:
Brett Maas,
Managing Principal Hayden IR brett@haydenir.com
(646) 536-7331
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
10-K
(Mark
One)
☒
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF
THE SECURITIES EXCHANGE ACT OF 1934
For
the Fiscal Year Ended December 31, 2021
Or
☐
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d)
OF THE SECURITIES EXCHANGE ACT OF 1934
For
the transition period from _____ to _____
Commission
File Number: 001-39480
APPLIED
UV, INC.
(Exact
name of registrant as specified in its charter)
| Delaware |
84-4373308 |
(State
or other jurisdiction of incorporation or organization) |
(I.R.S.
Employer Identification No.) |
150
N. Macquesten Parkway
Mount
Vernon, NY
10550
(914)
665-6100
(Address,
including zip code, of registrant's principal executive offices and
telephone
number, including area code)
Securities
registered pursuant to Section 12(b) of the Act:
| Title
of Each Class |
|
Trading
Symbol |
|
Name
of each exchange on which registered |
| Common
Stock, par value $0.0001 per share |
|
AUVI |
|
The
Nasdaq
Stock Market LLC |
| 10.5%
Series A Cumulative Perpetual Preferred Stock, par value $0.0001 per share |
|
AUVIP |
|
The
Nasdaq
Stock
Market LLC |
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Yes: ☐ No: ☒
Indicate
by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act.
Yes:
☐ No: ☒
Indicate
by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities
Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports),
and (2) has been subject to such filing requirements for the past 90 days.
Yes: ☒ No: ☐
Indicate
by check mark whether the registrant has submitted electronically, every Interactive Data File required to be submitted pursuant to Rule 405
of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant
was required to submit such files).
Yes ☒ No ☐
Indicate
by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting
company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,”
“smaller reporting company,” and “emerging growth company, in Rule 12b-2 of the Exchange Act.
| Large accelerated filer
☐ |
Accelerated filer
☐ |
| Non-accelerated
filer ☒ |
Smaller reporting company
☒ |
| Emerging
Growth Co ☒ |
|
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate
by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness
of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C.7262(b)) by the registered
public accounting firm that prepared or issued its audit report. ☐
Indicate
by check mark whether the registrant is a shell company (as defined in Rule12b-2 of the Act).
Yes: ☐ No: ☒
The
aggregate market value of the voting and non-voting common stock held by non-affiliates of the registrant as of June 30, 2021 was $92,364,937.
At
April 7, 2022, the registrant had 12,888,174
shares of common stock, par value $0.0001 per share, outstanding.
Table
of Contents
SPECIAL
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This
report contains forward-looking statements. The forward-looking statements are contained principally in the sections entitled “Business,”
and “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” These statements involve
known and unknown risks, uncertainties and other factors which may cause our actual results, performance or achievements to be materially
different from any future results, performances or achievements expressed or implied by the forward-looking statements. In some cases,
you can identify forward-looking statements by terms such as “anticipates,” “believes,” “could,”
“estimates,” “expects,” “intends,” “may,” “plans,” “potential,”
“predicts,” “projects,” “should,” “would,” and similar expressions intended to identify
forward-looking statements. Forward-looking statements reflect our current views with respect to future events and are based on assumptions
and subject to risks and uncertainties. You should read these factors and the other cautionary statements made in this report and in
the documents we incorporate by reference into this report as being applicable to all related forward-looking statements wherever they
appear in this report or the documents we incorporate by reference into this report. If one or more of these factors materialize, or
if any underlying assumptions prove incorrect, our actual results, performance or achievements may vary materially from any future results,
performance or achievements expressed or implied by these forward-looking statements.
Given
these uncertainties, you should not place undue reliance on these forward-looking statements. These forward-looking statements include,
among other things, statements relating to:
| • |
|
our
ability to continue as a going concern; |
| • |
|
our
ability to secure sufficient funding and alternative source of funding to support our current and proposed operations, which could
be more difficult in light of the negative impact of the COVID-19 pandemic on investor sentiment and investing ability; |
| • |
|
our
anticipated growth strategies and our ability to manage the expansion of our business operations effectively; |
| • |
|
our
ability to maintain or increase our market share in the competitive markets in which we do business; |
| • |
|
our
ability to grow net revenue and increase our gross profit margin; |
| • |
|
our
ability to keep up with rapidly changing technologies and evolving industry standards, including our ability to achieve technological
advances; |
| • |
|
our
dependence on the growth in demand for our products; |
| • |
|
our
ability to compete with larger companies with far greater resources than we have; |
| • |
|
our
continued ability to obtain raw materials and other supplies for our products at competitive prices and on a timely basis, particularly
in light of the potential impact of the COVID-19 pandemic on our suppliers and supply chain; |
| • |
|
our
ability to diversify our product offerings and capture new market opportunities; |
| • |
|
our
ability to source our needs for skilled labor, machinery, parts, and raw materials economically; |
| • |
|
our
ability to retain key members of our senior management; |
| • |
|
our
ability to continue to operate safely and effectively during the COVID-19 pandemic; and |
| • |
|
our
ability to maintain our listing on The Nasdaq Capital Markets. |
Also,
forward-looking statements represent our estimates and assumptions only as of the date of this report. You should read this report and
the documents that we reference and file as exhibits to this report completely and with the understanding that our actual future results
may be materially different from what we expect. Except as required by law, we assume no obligation to update any forward-looking statements
publicly, or to update the reasons actual results could differ materially from those anticipated in any forward-looking statements, even
if new information becomes available in the future.
Use
of Certain Defined Terms
Except
where the context otherwise requires and for the purposes of this report only:
| • |
|
the
“Company,” “Applied UV,” “we,” “us,” and “our” refer to the combined
business of Applied UV, Inc., a Delaware corporation and its wholly-owned subsidiaries, SteriLumen, Inc., a New York corporation
(“SteriLumen”) and Munn Works, LLC, a New York limited liability company (“MunnWorks”). |
| • |
|
“Exchange
Act” refers the Securities Exchange Act of 1934, as amended; |
| • |
|
“SEC”
refers to the Securities and Exchange Commission; and |
| • |
|
“Securities
Act” refers to the Securities Act of 1933, as amended. |
SUMMARY
OF RISK FACTORS
Our
business is subject to a number of risks. You should be aware of these risks before making an investment decision. These risks are discussed
more fully in Item 1A: Risk Factors in this Annual Report. These risks include, among others, that:
| • | | We
operate through SteriLumen and MunnWorks and our only current source of revenue is distributions
from our subsidiaries. SteriLumen has incurred losses since its inception and we anticipate
it will continue to incur significant losses for the foreseeable future and revenue from
MunnWorks may not be sufficient to offset those loses; |
| • | | We
could need to raise additional capital in the future, and if we are unable to secure adequate
funds on terms acceptable to us, we could be unable to execute our business plan; |
| • | | Our
suppliers may not supply us with a sufficient amount or adequate quality of materials; |
| • | | Our
reliance on third parties requires us to share our trade secrets, which increases the possibility
that a competitor will discover them or that our trade secrets will be misappropriated or
disclosed; |
| • | | We
could be subject to significant warranty obligations if our products are defective; |
| • | | Product
liability claims against us could be costly and could harm our reputation; |
| • | | If
we lose our key management personnel, or are unable to attract or retain qualified personnel,
it could adversely affect our ability to execute our growth strategy; |
| • | | Climate
change initiatives could materially and adversely affect our business, financial condition,
and results of operations; |
| • | | Our
financial controls and procedures may not be sufficient to ensure timely and reliable reporting
of financial information, which, as a public company, could materially harm our stock price; |
| • | | The
air purification market is fragmented and competitive and we may not be able to compete successfully
with our existing competitors or new entrants into the markets we serve; |
| • | | If
we are unable to execute our plan to distribute SteriLumen’s Airocide products, we
may not be able to generate revenues and your investment could be materially adversely affected; |
| • | | We
are subject to significant regulatory oversight and changes in applicable regulatory requirements
could adversely affect our business; |
| • | | SteriLumen’s
Scientific Air business is highly dependent on a sole distributor and any disruption in their
operations could have a material adverse effect on our business, results of operations and
financial condition; |
| • | | SteriLumen’s
business is highly dependent on market perceptions of it and the safety and quality of its
products; |
| • | | Rapidly
changing standards and competing technologies could harm demand for our products; |
| • | | We
could be unable to effectively manage and implement our growth strategies, which could have
a material adverse effect on our business, financial condition, and results of operations; |
| • | | International
sales may comprise a significant portion of SteriLumen’s revenues and will be subject
to risks associated with operating in domestic and international markets; |
| • | | SteriLumen’s
collaborations with outside scientists and consultants may be subject to restriction and
change; |
| • | | The
custom design decorative framed mirror supply market is highly competitive; |
| • | | MunnWorks’
possible failure to develop new products or respond to changing consumer preferences and
purchasing practices could have a material adverse effect; |
| • | | MunnWorks’
business is highly dependent on market perceptions of it and the quality of its products; |
| • | | If
the patents that we own or license, or our other intellectual property rights, do not adequately
protect our technologies, we could lose market share to our competitors and be unable to
operate our business profitably; |
| • | | Changes
in U.S. patent law could diminish the value of patents in general; |
| • | | Our
directors and officers beneficially will own more than 50% of the voting power of our voting
stock and will be able to exert a controlling influence over our business affairs and matters
submitted to stockholders for approval; |
| • | | We
have not paid dividends on our Common Stock in the past and do not expect to pay dividends
on our Common Stock in the future, and any return on investment may be limited to the value
of our stock; and |
| • | | Provisions
of our Amended and Restated Certificate of Incorporation could delay or prevent the acquisition
or sale of our business. |
| • | | We
expect that we will need to raise additional capital, and raising additional funds by issuing
additional equity securities or with additional debt financing may cause dilution to shareholders
or restrict our operations; |
| • | | The
Series A Preferred Stock ranks junior to all of our indebtedness and other liabilities; |
| • | | The
Series A Preferred Stock will be effectively subordinated to the obligations of our subsidiaries; |
| • | | A
holder of Series A Preferred Stock has essentially no voting rights; |
| • | | If
we are not paying full dividends on any future dividend parity stock, we will not be able
to pay full dividends on the Series A Preferred Stock; |
PART
I
Item
1. Business
Corporate
History
Applied
UV is focused on the development, acquisition and commercialization of technology that address air purification and infection control
in the healthcare, hospitality, commercial, municipal and residential markets. The Company offers science-based solutions and products
in air purification under the Airocide brand and label and disinfection of hard surfaces under the Lumicide brand and label. MunnWorks,
our subsidiary focused on the hospitality market, manufactures and supplies fine decorative framed mirrors, framed art, and vanities.
MunnWorks provides us cross-selling opportunities for our Airocide and Lumicide products. Applied UV is a holding company. Our current
operating companies are SteriLumen and MunnWorks.
Air
Purification Solutions: Airocide Air Purification
On
February 8, 2021 we acquired substantially all of the assets of Akida Holdings LLC (“Akida”), which owned the Airocide™
system of air purification technologies for $7.88 million, consisting of $0.76 million in cash and 1.375 million shares of our Common
Stock (the “Acquisition”) with a fair market value of $7.12 million. Akida’s revenue for the full calendar year of
2020 was approximately $4.7 million. Prior to the Acquisition, Akida granted KES Science & Technology, Inc. (“KES”) a
non-exclusive irrevocable royalty free license (the “KES License”) to manufacture and sell products based on Airocide™
technology in the United States and Canada for use in the commercial food preservation and preparation market, and the cannabis/hemp
market. KES also manufactures, distributes and provides technical support for our Airocide™ products pursuant to certain service
agreements (the “KES Service Agreements”). At the closing of the KES Acquisition, the KES Service Agreements were assigned
to us and we assumed all of the obligations under the KES Service Agreements.
On
September 28, 2021, we acquired substantially all of the assets of KES, including the assignment of contracts related to the supply chain
management and sale of the Airocide™ system of air purification technologies, for $4.3 million in cash and 300,000 shares of our
common stock with a fair market value of $1,959,000 (the “KES Acquisition”). KES’s revenue for the twelve months ended
October 31, 2020 was approximately $4.5 million. The KES Acquisition along with the Acquisition provides us with all of the rights, title
and interest to the Airocide™ system of air purification technologies, including all of the rights KES had under the KES Service
Agreements.
The
Airocide™ system of air purification technologies, originally developed for the National Aeronatuics and Space Administration (“NASA”)
with assistance from the University of Wisconsin at Madison, uses a combination of UVC and a proprietary, titanium dioxide based photocatalyst
to eliminate airborne bacteria, mold, fungi, viruses, volatile organic compounds and many odors. We believe Airocide™ can provide
solutions to accelerate the reopening of the global economy with applications in the hospitality, hotel, healthcare, nursing homes, grocer,
wine, commercial buildings and retail sectors. The Airocide™ system has been used by brands such as DelMonte, Kroger, Opus One,
and in March 2021 the Boston Red Sox agreed with SteriLumen to install an Airocide™ system at Fenway Park and JetBlue Park.
The
core Airocide™ technology has been in use on the International Space Station and is based on photo-catalytic oxidation (PCO), a
bioconversion process that continuously converts damaging molds, microorganisms, dangerous pathogens, destructive volatile organic chemicals
(VOCs) and biological gasses into harmless water vapor.
Unlike
other air purification systems that provide “active” air cleaning, ozone producing systems, ionization or “photo-electrochemical
oxidation”, Airocide’s nanocoating technology permanently bonds titanium dioxide to the surface of the catalytic bed. This
permits the perpetual generation of surface-bound (OH-) radicals over the large surface area created by their advanced geometric design
and prevents the generation and release of ozone and other harmful byproducts. The proprietary formulation and methods for creating the
catalyst are the basis of Airocide’s competitive advantage, making it the only consistently robust, highly effective, ozone free
PCO technology on the market.
Airocide™
has been tested over the past 12 years by governmental agencies such as NASA, the National Renewable Energy Laboratory, independent universities
including the University of Wisconsin, Texas Tech University and Texas A&M, and air quality science laboratories. Airocide technology
has been cleared by the FDA as a class II medical device, making it suitable for providing medical grade air purification in critical
hospital use cases. Airocide® product lines include: APS (consumer units) and the GCS and HD lines (commercial units)
and will enable the commercial units with SteriLumen’s Clarity D3™ app to bring connectivity, reporting and asset management
to our air purification products.
The
APS series provides true choice, low maintenance filter-less PCO or a filtered air purification option ideal for restaurants, conference
rooms, residential and small business or home office spaces. The GCS series is suitable for larger public spaces and enclosed rooms that
may have high occupancy such as offices, waiting rooms and hotel lobbies, and airport gate areas. The HD series is the most powerful,
providing two-stage purification for fast sanitization of larger or industrial spaces such as sporting venues and locker rooms, airports,
museums, winery cellars, warehouses and food-processing facilities. All Airocide™ products also extend the life of any perishables
like fruit, produce or flowers.
Scientific
Air
On
October 13, 2021, we acquired substantially all of the assets of Old SAM Partners, LLC F/K/A Scientific Air Management, LLC (“Old
SAM”), which owned a line of air purification technologies (“Scientific Air”) for a purchase price of $9.5 million
in cash and 200,000 fully vested shares of our Common Stock (the “Vested Shares”) and 200,000 shares of our Common Stock
that are subject to vesting (the “Earnout Shares”) (the “’SciAir’ Acquisition”). The number of shares
of Common Stock included in the purchase price was based on a per share value of $10.00. With respect to the Vested Shares and the Earnout
Shares, the Acquisition agreement provided that we would pay additional cash compensation to Scientific Air if the if the 20 day average
price of our common stock was below $10.00 on April 14, 2022, in the case of the Vested Shares or March 31, 2023, in the case of the
Earnout Shares. Such amounts were payable only if Old SAM or its affiliates owned Vested Shares or Earnout Shares on the applicable determination
date. As described below, Old SAM and certain of its affiliates have relinquished their right to all 400,000 Vested Shares and Earnout
Shares.
On
March 31, 2022 we entered into a Settlement Agreement (the “Settlement Agreement”) with Old SAM and the members thereof who
executed the Settlement Agreement (collectively, the “Old SAM Parties”), pursuant to which the Old SAM Parties relinquished
all of their right, title and interest in any of the Vested Shares and the Earnout Shares and no longer have the right to any additional
cash consideration as described above. The Settlement Agreement also contains a mutual release of all claims we and the Old SAM parties
have against each other, other than claims related to certain consulting agreements executed in connection with Acquisition. The Settlement
Agreement was not the result of any litigation, and no claims were filed by us against any of the Old SAM Parties or by any of the Old
SAM Parties against us.
The
Scientific Air product line uses a combination of UVC and a proprietary, patented system to eliminate airborne bacteria, mold, fungi,
viruses, volatile organic compounds and many odors without producing any harmful by- products. Scientific Air’s products are well
suited for larger spaces within a facility due to the higher air flow of these units. The units are also mobile with industrial grade
casters, allowing for movement throughout a facility to address increased bio burden from larger meetings or increased human traffic.
Both of these key items extend our Airocide line, creating a comprehensive air disinfection portfolio that spans from small to large
spaces and mobile applications.
Scientific
Air’s products are currently sold predominantly in North America and into the healthcare market. We see a number of bidirectional
synergies. First, we look to leverage Airocide’s global distribution capabilities to start distributing Scientific Air’s
products internationally. Second, we look to leverage Scientific Air’s strength in healthcare to pull through existing Airocide
units, creating a broad healthcare product line, from small clinics, patient rooms and doctor’s offices to larger spaces such as
nursing stations, waiting rooms and cafeterias. Scientific Air currently leverages one of the largest healthcare distributors in North
America, which is a relationship we intend to further develop to increase our customer base and revenue. Finally, we look to extend our
Data Driven Disinfection platform by integrating all our units via our Clarity D3 web and mobile phone application, creating smart asset
management, reporting, and control across enterprises.
Market
Opportunity
Our
goal is to build a company that successfully designs, develops and markets our platform for Data Driven Disinfection which will enable
US and global economies to re-open and provide safe environments during and following the pandemic. Our platform will also be positioned
to help decrease the national rate of Healthcare Associated Infections (HAIs). We will seek to achieve this goal by having our products
actively involved in the following activities:
•
Focus on large facilities in hospitality and sporting venues: this market segment has strong incentives to invest
in additional disinfection to ensure guests and patrons return and increase sales to pre-pandemic levels. Hotels wishing to make disinfection,
safety, and cleanliness part of their branded experience can benefit from our solutions and products. In addition to existing and developing
Airocide and Lumicide specific sales efforts, we intend to leverage the Company’s hospitality business for cross-selling opportunities
of our air purification and surface disinfectant solutions and products. Our initial research indicates that the key stakeholders in
this market value the asset management and reporting capabilities of our platform and provide key points of differentiation.
•
Secondary focus on healthcare facilities outside of the hospital market: (i) Target infection prevention professionals
in healthcare facilities including assisted living and long-term care, clinics and ambulatory surgical centers.; (ii) Identify and target
facilities that have been fined for high infection rates.
•
Leverage relationships with Environmental Health and Safety organizations who are responsible for employee safety.
•
Continue scientific validation through lab testing and data from real world deployments; publish case studies in
peer reviewed journals.
•
Leverage an outsourced supply chain: Leverage an outsourced supply chain for production and warehousing.
Beyond
healthcare facilities, we are prioritizing our opportunities in additional market segments:
Airplane
and Cruise Line Bathrooms. Because of the close quarters inside most airplane cabins, pathogens can easily be spread amongst
passengers, especially on long flights where airplane bathrooms have more use and may not be cleaned before flight’s end. Our Data
Driven Disinfection platform could provide an important means for disinfecting airplane bathrooms, keeping track of cleanliness during
flights, and decreasing the spread of germs in the airplane cabin.
Schools.
Schools and education authorities pay very close attention to disinfection to prevent the spread of influenza as well as other contagious
threats. The CDC maintains guidelines for disinfection of schools and many other organizations also advise schools on proper disinfection
protocol. Our disinfection systems could find a receptive audience among school facilities managers looking for new solutions that can
assist in safely returning students to classrooms during the pandemic and post Covid-19.
Restaurants.
Given the need to prevent foodborne illness, restaurants are constantly urging staff to wash their hands and are required by public
health authorities to keep their premises as clean and germ-free as possible. There is an opportunity to become part of upscale restaurants’
strategy to demonstrate dedication and commitment to customers and health department to reduce the spread of infections within their
establishment.
Homes.
Our Airocide brands have a strong foothold in the consumer marketplace and we will expand digital marketing activities to gain greater
market share.
Surface
Disinfection Solutions: Lumicide
The
Company’s Lumicide brand of products are unique, patented, and automated disinfecting systems that rely on LEDs in the “C”
range of the ultraviolet spectrum (UVC). Lumicide offers configurable options for placement of the UVC LEDs in a wide variety of fixtures
including but not limited to vanities, restrooms, above desks or along countertops. Lumicide disinfects hard surfaces within 12 to 24
inches of the unit, generating adequate energy required to kill pathogens in a typical disinfection cycle at extremely low power measured
in milliwatts. Lumicide has been tested by ResInnova Laboratories, an International Antimicrobial Council certified BSL-2 testing facility
(“ResInnova”).
Our
product platform includes the following attributes:
•
Focus on high-contamination surfaces Focuses on pathogens that accumulate on the sink area, including handles, faucets,
backsplash and in the drain.
•
Germicidal UVC LEDs. The UVC LEDs in our devices have demonstrated destruction of the most clinically relevant pathogens
causing HAIs as well as destruction of SARS-CoV-2, the virus causing COVID-19.
•
Automatic operation. A built-in programmable controller within the unit ensures operation for the appropriate UVC dosage
required to conduct four logs pathogen destruction and is not dependent on manual operation. Its functionality is expandable and may
become a source for recurring income through additional data reporting, leasing and maintenance of add-on elements.
•
Continuous operation. Works in pre-programmed cycles that can be managed by an on-board programmable controller. The motion
detectors enclosed within each device allow continuous disinfection of high contamination areas as long as room occupants are outside
a safe distance of the device. Research has shown that microbes can rebound to pre-disinfection levels within two hours following manual
cleaning and disinfection. Lumicide products operating continuously mitigate pathogen regeneration.
•
Safety. Built-in redundant motion sensors automatically shut off when movement is detected within range of the UVC light,
or failure of motion sensors on the unit, eliminating safety concerns about UVC exposure. Once there is no movement in the room for programmed
time period, Lumcide comes back on to restart and continue its cycle.
We
currently have five product lines incorporating Lumicide including:
(i)
disinfecting drain device (in market);
(ii)
disinfecting shelf, is a ribbon that can be installed above a sink, beneath an existing bathroom mirror, or above other high-contamination
risk surfaces (in market);
(iii)
disinfecting back-lit mirror (in development);
(iv)
mirrored medicine cabinet for residential use (in development).
The
Company has pursued validation of its platform in both laboratory and real clinical settings. Devices were independently tested in 2017,
before the pandemic, and again in 2020 at ResInnova Laboratories, an International Antimicrobial Council certified BSL-2 testing facility.
The disinfecting mirror and disinfecting drain devices were found to be effective in killing (3-4 log reductions) the most infectious
and clinically important pathogens including C. difficile, methicillin resistant staphylococcus aureus (MRSA), E. Coli, and OC43 human
coronavirus, a strain structurally and genetically similar to SARS-CoV-2 and accepted as a surrogate for that virus. The Company is also
sponsoring a study at Mount Sinai and its Icahn School of Medicine on the effectiveness of Lumicide in patient bathrooms in a New York
hospital in the Mount Sinai system. Mount Sinai has agreed to provide the results of their study in a report to be issued in the third
quarter of 2022 as well as publishing their results in an academic, peer reviewed journal.
Axis
Lighting Licensing & Joint Development Agreement
In
October of 2020, we entered into an Exclusive Licensing & Joint Development Agreement with Axis Lighting to commercialize UVC devices
specifically for the hospital market. Axis Lighting is one of the largest independent architectural lighting companies in North America
and operates a manufacturing facility with on-site design, engineering, and marketing staff to deliver high-performance LED luminaires
for general, ambient and task lighting in offices, as well as in commercial and institutional spaces. BalancedCare™ by Axis provides
healthcare lighting for wellness, offering patent-pending performance lighting for both visual comfort and functionality. The licensed
product from Axis once launched will be offered through the BalancedCare™ platform and brand. BalancedCare™ addresses a number
of requirements of today's complex healthcare environment, including infection control, and is supported and distributed through 98 Axis
agents across North America.
Under
the agreement, we will work with Axis Lighting's BalancedCare™ team to leverage our intellectual property and proprietary know-how
to commercialize a range of new LED-based technologies designed for use in the hospital sector. Axis will pay royalties on sales of the
commercial products developed through the collaboration. Our first jointly developed disinfection lighting fixture has completed the
design phase and will launch later this year.
The
Company has received confirmation from the U.S. Food & Drug Administration that our Lumicide products are not “devices”
under Section 201(h) of the FDA Act and therefore the Company is not required to comply with the requirements of the FDA Act with respect
to Lumicide. However, our Limicide products are in compliance with the FDA’s March 2020 “Enforcement Policy for Sterilizers,
Disinfectant Devices, and Air Purifiers” enacted during the COVID-19 Public Health Emergency. In addition, the Lumicide disinfecting
shelf and drain systems have received certification from Edison Testing Laboratories (ETL), a global leader in product safety testing
and certification.
Integrating
Our Solutions Through Data Driven Disinfection Platform
The
Clarity D3™ application enables Data Driven Disinfection™ with IOT connectivity using Wi-Fi, Bluetooth, and other RF technology
for continuous transmission of use and functionality data for collection and analysis. Clarity D3 provides remote asset management and
full visibility into the operation of our devices as well as efficacy reporting on the cycles that have run and subsequent cleanliness
status. The application also enables Smart Maintenance, with LED cartridge lifecycle alerts, fault detection/reports and other automatic
system updates.
Hospitality
Segment
Through
our subsidiary, MunnWorks, we manufacture, and supply custom designed decorative framed mirrors, framed art, and bathroom vanities primarily
to the hospitality market. We supply the major hotel brands in North America including hotel chains such as Hilton Hotels & Resorts,
the various Hyatt branded hotels, the various Marriott branded hotels, Four Seasons Hotels and Resorts and the subsidiary hotel brands
for each of these major brands. We have a national sales force and an established distribution network for hotels and restaurants in
every major market in the United States and have begun to develop a distribution network for the assisted living market. These distribution
networks will also be a significant asset for cross selling and recommending our Airocide and Lumicide products and solutions, as those
networks will be utilized for marketing and sales.
Recent
Developments
On
July 16, 2021, we completed an underwritten public offering of our Series A Preferred Stock (“Series A Preferred Stock”)
at an offering price of $25.00 per share. We initially sold 480,000 shares of Series A Preferred Stock and, on July 29, 2021, we sold
an additional 72,000 shares of Series A Preferred Stock as a result of the exercise of the underwriters’ over-allotment option
in full. The sale of Series A Preferred Stock generated aggregate gross proceeds to us of approximately $13.8 million before deduction
of underwriter fees and commissions and other offering expenses.
On
December 31, 2021, we we completed an underwritten public offering of 2,666,667 shares of our common stock at an offering price of $3.00
per share. The sale of our common stock generated aggregate gross proceeds to us of approximately $8 million before deduction underwriter
fees and commissions and other offering expenses.
Employees
As of December
31, 2021, we had 61 employees.
Corporate
Information
Our
principal executive offices are located at 150 N. Macquesten Parkway, Mount Vernon, NY 10550. Our website address is www.applieduvinc.com.
Item
1a. Risk Factors.
Investing
in our common stock is highly speculative and involves a significant degree of risk. Before you invest in our securities, you should
give careful consideration to the following risk factors, in addition to the other information included in this Annual Report on Form
10-K, including our financial statements and related notes, before deciding whether to invest in our securities. The occurrence of any
of the adverse developments described in the following risk factors could materially and adversely harm our business, financial condition,
results of operations or prospects. In that case, the trading price of our common stock could decline, and you may lose all or part of
your investment.
Risks
Related to Our Business and Operations
The
Company operates through SteriLumen and MunnWorks and its only material assets are its equity interests in those subsidiaries. As a result,
our only current source of revenue is distributions from our subsidiaries and SteriLumen has incurred losses since its inception and
we anticipate it will continue to incur significant losses for the foreseeable future and revenue from MunnWorks may not be sufficient
to offset those loses.
The
Company is a holding company for SteriLumen and MunnWorks and has no material assets other than its equity interests in those subsidiaries.
Therefore, the only current revenue source is future distributions from its subsidiaries. SteriLumen is an early-stage designer and marketer
of disinfection systems with limited operating history spanning from December 2016. We expect negative cash flows from SteriLumen’s
operations for the foreseeable future which may not be off-set by cash flows from MunnWorks. Our utilization of cash has been and will
continue to be highly dependent on SteriLumen’s product development programs and cash flow from MunnWorks’ operations. Our
cash expenses will be highly dependent on the product development programs that SteriLumen chooses to pursue, the progress of these product
development programs, the results of SteriLumen’s validation/marketing studies, the terms and conditions of SteriLumen’s
contracts with service providers and manufacturing contractors, and the terms of recruitment of facilities in our validation/marketing
studies. In addition, the continuation of SteriLumen’s validation/marketing studies, and quite possibly its entire business, will
depend on results of upcoming clinical data analyses and our financial resources at the time. Failure to raise capital as and when needed,
on favorable terms or at all, would have a negative impact on our financial condition and SteriLumen’s ability to develop its product
candidates.
SteriLumen
has devoted substantially all of its financial resources to develop its product candidates. SteriLumen has financed its operations primarily
through the contributions of its founders. The amount of SteriLumen’s future net losses will depend, in part, on the development
of adequate distribution channels for the Disinfecting System, the demand for the Disinfecting System, the rate of its future expenditures
and our ability to obtain funding through the issuance of our securities, strategic collaborations or grants. Disinfection product development
is a highly speculative undertaking and involves a substantial degree of risk. SteriLumen successfully completed the prototype phase
of testing and development for the Disinfecting System, but has not yet commenced pivotal testing in health care facility settings. Even
if SteriLumen obtains positive results from such testing, its future revenue will depend upon its ability to achieve sufficient market
acceptance, pricing, the performance of its independent sales representatives and its manufacturing and distribution suppliers.
We
expect SteriLumen to continue to incur significant losses until it is able to commercialize the Disinfecting System, which it may not
be successful in achieving. We anticipate that SteriLumen’s expenses will increase substantially if and as SteriLumen:
| |
• |
continues
the research and development of the Disinfecting System and other disinfecting products; |
| |
|
|
| |
• |
expands
the scope of its testing for the Disinfecting System and other disinfecting products; |
| |
|
|
| |
• |
establishes
a sales, marketing, and distribution infrastructure to commercialize its product candidates; |
| |
|
|
| |
• |
seeks
to maintain, protect, and expand its intellectual property portfolio; |
| |
|
|
| |
• |
seeks
to attract and retain skilled personnel; and |
| |
|
|
| |
• |
creates
additional infrastructure to support its product candidate development and planned future commercialization efforts. |
Furthermore,
any additional fundraising efforts may divert our management from our subsidiaries’ day-to-day activities, which may adversely
affect our ability to develop and commercialize their product candidates. In addition, we cannot guarantee that future financing will
be available in sufficient amounts or on terms acceptable to us, if at all. Moreover, the terms of any financing may adversely affect
the holdings or the rights of holders of our securities and the issuance of additional securities, whether equity or debt, by us, or
the possibility of such issuance, may cause the market price of our shares to decline. The incurrence of indebtedness could result in
increased fixed payment obligations, and we may be required to agree to certain restrictive covenants, such as limitations on our ability
to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other operating restrictions
that could adversely affect our ability to conduct our business. We could also be required to seek funds through arrangements with collaborative
partners or otherwise at an earlier stage than otherwise would be desirable, and we may be required to relinquish rights to some of our
technologies or product candidates or otherwise agree to terms unfavorable to us, any of which may have a material adverse effect on
our business, operating results, and prospects. Even if we believe that we have sufficient funds for our current or future operating
plans, we may seek additional capital if market conditions are favorable or if we have specific strategic considerations.
If
we are unable to obtain funding on a timely basis, we may be required to significantly curtail, delay or discontinue one or more of our
research or development programs or the commercialization of any product candidates or be unable to expand our operations or otherwise
capitalize on our business opportunities, as desired, which could materially affect our business, financial condition and results of
operations.
The
pandemic caused by the spread of the Coronavirus could have an adverse impact on our financial condition and results of operations and
other aspects of our business.
In
December 2019, a novel strain of Coronavirus was reported to have surfaced in Wuhan, China. The virus has since spread to over 150 countries
and every state in the United States. On January 30, 2020, the World Health Organization declared the outbreak of Coronavirus a “Public
Health Emergency of International Concern”. On March 11, 2020, the World Health Organization declared the outbreak a pandemic,
and on March 13, 2020, the United States declared a national emergency.
The
spread of the virus in many countries continues to adversely impact global economic activity and has contributed to significant volatility
and negative pressure in financial markets and supply chains. The pandemic has had, and could have a significantly greater, material
adverse effect on the U.S. economy where we conduct a majority of our business. The pandemic has resulted, and may continue to result
for an extended period, in significant disruption of global financial markets, which may reduce our ability to access capital in the
future, which could negatively affect our liquidity.
Most
states and cities have reacted by instituting quarantines, restrictions on travel, “stay at home” and “social distancing”
rules and restrictions on the types of businesses that may continue to operate, as well as guidance in response to the pandemic and the
need to contain it.
We
have taken steps to take care of our employees, including providing the ability for employees to work remotely and implementing strategies
to support appropriate social distancing techniques for those employees who are not able to work remotely. We have also taken precautions
with regard to employee, facility, and office hygiene as well as implementing significant travel restrictions. We are also assessing
our business continuity plans for all business units in the context of the pandemic. This is a rapidly evolving situation, and we will
continue to monitor and mitigate developments affecting our workforce, our suppliers, our customers, and the public at large to the extent
we are able to do so. We have and will continue to carefully review all rules, regulations, and orders and responding accordingly. We
are dependent upon suppliers to provide us with all of the raw materials for products that we manufacture and sell and we currently manufacture
the majority of our products in China. The pandemic has impacted and may continue to impact suppliers of materials and the manufacturing
locations for our products. As a result, we have faced and may continue to face delays or difficulty manufacturing certain products,
which could negatively affect our business and financial results. Even if we are able to find alternate sources for materials and manufacturing,
they may cost more, which could adversely impact our profitability and financial condition.
If
the current pace of the pandemic cannot be slowed and the spread of the virus is not contained, our business operations could be further
delayed or interrupted. We expect that government and health authorities may announce new or extend existing restrictions, which could
require us to make further adjustments to our operations in order to comply with any such restrictions. We may also experience limitations
in employee resources. In addition, our operations could be disrupted if any of our employees were suspected of having the virus, which
could require quarantine of some or all such employees or closure of our facilities for disinfection. The duration of any business disruption
cannot be reasonably estimated at this time but may materially affect our ability to operate our business and result in additional costs.
Although
it is difficult to predict the effect and ultimate impact of the Coronavirus outbreak on our business, it is likely that the impact of
Coronavirus will adversely affect our results of operations, financial condition and cash flows in fiscal year 2022.
We
are vulnerable to continued global economic uncertainty and volatility in financial markets.
Our
business is highly sensitive to changes in general economic conditions as a seller of goods and services. Financial markets inside the
United States and internationally have experienced extreme disruption in recent times, including, among other things, extreme volatility
in security prices, severely diminished liquidity and credit availability, and declining valuations of investments. We believe these
disruptions are likely to have an ongoing adverse effect on the world economy. A continuing economic downturn and financial market disruptions
could have a material adverse effect on our business, financial condition, and results of operations, including by:
| |
• |
reducing
demand for our products and services, increasing order cancellations and resulting in longer sales cycles and slower adoption of
new technologies; |
| |
• |
increasing
the difficulty of collecting accounts receivable and the risk of excess and obsolete inventories; |
| |
• |
increasing
price competition in our served markets; and |
| |
• |
resulting
in supply interruptions, which could disrupt our ability to produce our products. |
We
could need to raise additional capital in the future, and if we are unable to secure adequate funds on terms acceptable to us, we could
be unable to execute our business plan.
To
remain competitive, we must continue to make significant investments in the development of our products, the expansion of our sales and
marketing activities, and the expansion of our operating and management infrastructure as we increase sales domestically and internationally.
If cash generated from our operations is insufficient to fund such growth, we could be required to raise additional funds through the
issuance of equity or debt securities in the public or private markets, or through a collaborative arrangement or sale of assets. Additional
financing opportunities may not be available to us, or if available, may not be on favorable terms. The availability of financing opportunities
will depend, in part, on market conditions, and the outlook for our business. Any future issuance of equity securities or securities
convertible into equity securities could result in substantial dilution to our stockholders, and the securities issued in such a financing
could have rights, preferences or privileges senior to those of our Common Stock. In addition, if we raise additional funds through debt
financing, we could be subject to debt covenants that place limitations on our operations. We could not be able to raise additional capital
on reasonable terms, or at all, or we could use capital more rapidly than anticipated. If we cannot raise the required capital when needed,
we may not be able to satisfy the demands of existing and prospective customers, we could lose revenue and market share and we may have
to curtail our capital expenditures.
The
following factors, among others, could affect our ability to obtain additional financing on favorable terms, or at all:
| |
• |
our
results of operations; |
| |
• |
general
economic conditions and conditions in the sanitation and disinfection industries and performance of sanitation devices as opposed
to sanitation and disinfection substances; |
| |
• |
the
perception of our business in the capital markets; |
| |
• |
making
capital improvements to improve our infrastructure; |
| |
• |
hiring
qualified management and key employees; |
| |
• |
responding
to competitive pressures; |
| |
• |
complying
with regulatory requirements, if any; |
| |
• |
our
ratio of debt to equity; |
| |
• |
our
financial condition; |
| |
• |
our
business prospects; and |
If
we are unable to obtain sufficient capital in the future, we could have to curtail our capital expenditures. Any curtailment of our capital
expenditures could result in a reduction in net revenue, reduced quality of our products, increased manufacturing costs for our products,
harm to our reputation, or reduced manufacturing efficiencies and could have a material adverse effect on our business, financial condition,
and results of operations.
Raising
additional capital may cause dilution to our existing stockholders and restrict our operations or require us to relinquish certain intellectual
property rights.
We
may seek additional capital through a combination of public and private equity offerings, debt financings, strategic partnerships and
alliances, licensing arrangements, and grants. To the extent that we raise additional capital through the sale of equity or convertible
debt securities, the ownership interest of our existing stockholders may be diluted, and the terms may include liquidation or other preferences
that adversely affect the rights of our stockholders. Debt and receivables financings may be coupled with an equity component, such as
warrants to purchase shares, which could also result in dilution of our existing stockholders’ ownership. The incurrence of indebtedness
would result in increased fixed payment obligations and could also result in certain restrictive covenants, such as limitations on our
ability to incur additional debt, limitations on our ability to acquire or license intellectual property rights and other operating restrictions
that could adversely impact our ability to conduct our business. If we raise additional funds through strategic partnerships and alliances
and licensing arrangements with third parties, we may have to relinquish valuable rights to our or our subsidiaries’ products or
grant licenses on terms that are not favorable to us. A failure to obtain adequate funds may cause us to curtail certain operational
activities, including research and development, validation/marketing studies, sales and marketing, and manufacturing operations, in order
to reduce costs and sustain the business, and would have a material adverse effect on our business and financial condition.
Our
success depends, in part, on our relationships with, and the efforts of, third-party manufacturers, distributors, and other third parties
that perform various tasks for us. If these third parties do not successfully carry out their contractual duties or meet expected, we
may not be able to commercialize our product candidates and our business could be substantially harmed.
While
we internally manufacture all of MunnWorks’ products that are manufactured domestically, currently approximately 75% of MunnWorks’
products and all of SteriLumen’s products are manufactured overseas in China by third party manufacturers. We do not currently
have the infrastructure or capability internally to manufacture all of the components of our products and systems, and we lack the resources
and the capability to manufacture and distribute the Disinfecting System on a commercial scale. We plan to rely on third parties for
such operations. There are a limited number of manufacturers who have the ability to produce our products, and there may be a need to
identify alternate manufacturers to prevent a possible disruption of our manufacturing and distribution process. Switching manufacturers
or distributors, if necessary, may involve substantial costs and is likely to result in a delay in our desired commercial timelines,
which could harm our business and results of operations.
Our
suppliers may not supply us with a sufficient amount or adequate quality of materials, which could have a material adverse effect on
our business, financial condition, and results of operations.
Our
business depends on our ability to obtain timely deliveries of materials, components, and subassemblies of acceptable quality and in
acceptable quantities from third-party suppliers. We generally purchase components and subassemblies from a limited group of suppliers
through purchase orders, rather than written supply contracts. Consequently, many of our suppliers have no obligation to continue to
supply us on a long-term basis. In addition, our suppliers manufacture products for a range of customers, and fluctuations in demand
for the products those suppliers manufacture for others could affect their ability to deliver components for us in a timely manner. Moreover,
our suppliers could encounter financial hardships, be acquired, or experience other business events unrelated to our demand for components,
which could inhibit or prevent their ability to fulfill our orders and satisfy our requirements.
If
any of our suppliers cease to provide us with sufficient quantities of our components in a timely manner or on terms acceptable to us,
or ceases to manufacture components of acceptable quality, we could incur manufacturing delays and sales disruptions while we locate
and engage alternative qualified suppliers, and we might be unable to engage acceptable alternative suppliers on favorable terms. In
addition, we could need to reengineer our components, which could significantly delay production. Any interruption or delay in the supply
of components or materials, or our inability to obtain components or materials from alternate sources at acceptable prices in a timely
manner, could impair our ability to meet the demand of our customers and cause them to cancel orders or switch to competitive procedures.
We are continually in the process of identifying and qualifying alternate source suppliers for our key components. There can be no assurance,
however, that we will successfully identify and qualify an alternate source supplier for any of our key components or that we could enter
into an agreement with any such alternate source supplier on terms acceptable to us, or at all.
Our
reliance on third parties requires us to share our trade secrets, which increases the possibility that a competitor will discover them
or that our trade secrets will be misappropriated or disclosed.
Because
we rely on third parties to develop and manufacture our products, we must, at times, share trade secrets with them. We seek to protect
our proprietary technology in part by entering into confidentiality agreements and, if applicable, material transfer agreements, collaborative
research agreements, consulting agreements or other similar agreements with our collaborators, advisors, employees and consultants prior
to beginning research or disclosing proprietary information. These agreements typically limit the rights of the third parties to use
or disclose our confidential information, such as trade secrets. Despite the contractual provisions employed when working with third
parties, the need to share trade secrets and other confidential information increases the risk that such trade secrets become known by
our competitors, are inadvertently incorporated into the technology of others, or are disclosed or used in violation of these agreements.
Given that our proprietary position is based, in part, on our know-how and trade secrets, a competitor’s discovery of our trade
secrets or other unauthorized use or disclosure would impair our competitive position and may have a material adverse effect on our business.
We
may engage in future acquisitions or strategic transactions which may require us to seek additional financing or financial commitments,
increase our expenses and/or present significant distractions to our management.
We
may make acquisitions in the future. In the event we engage in an acquisition or strategic transaction, we may need to acquire additional
financing (particularly, if the acquired entity is not cash flow positive or does not have significant cash on hand). Obtaining financing
through the issuance or sale of additional equity and/or debt securities, if possible, may not be at favorable terms and may result in
additional dilution to our current stockholders. Any future acquisition by us or one of our subsidiaries may require us to incur non-recurring
or other charges, may increase our near and long-term expenditures, and may pose significant integration challenges or disrupt our management
or business, which could adversely affect our operations and financial results. For example, an acquisition or strategic transaction
such as the acquisition may entail numerous operational and financial risks, including the risks outlined above and additionally:
| |
• |
exposure
to unknown liabilities; |
| |
• |
disruption
of our business and diversion of our management’s time and attention in order to develop acquired products or technologies
higher than expected acquisition and integration costs; |
| |
• |
write-downs
of assets or goodwill or impairment charges; |
| |
• |
increased
amortization expenses; |
| |
• |
difficulty
and cost in combining the operations and personnel of any acquired businesses with our operations and personnel; |
| |
• |
impairment
of relationships with key suppliers or customers of any acquired businesses due to changes in management and ownership; and |
| |
• |
inability
to retain key employees of any acquired businesses. |
Accordingly,
although there can be no assurance that we will undertake or successfully complete any transactions of the nature described above, and
any transactions that we do complete could have a material adverse effect on our business, results of operations, financial condition
and prospects.
Recent
U.S. tax legislation may materially affect our financial condition, results of operations and cash flows.
The
recently enacted Tax Cuts and Jobs Act (the “Tax Act”) has significantly changed the U.S. federal income taxation of U.S.
businesses, including by reducing the U.S. corporate income tax rate, limiting interest deductions, permitting immediate expensing of
certain capital expenditures, modifying or repealing many business deductions and credits.
The
Coronavirus Aid, Relief, and Economic Security Act (the “CARES Act”) modifies certain provisions of the Tax Act, including
increasing the amount of interest expense that may be deducted.
The
Tax Act as modified by the CARES Act is unclear in many respects and could be subject to potential amendments and technical corrections,
as well as interpretations and implementing regulations by the Treasury and IRS, any of which could lessen or increase certain adverse
impacts of the legislation. In addition, it is unclear how these U.S. federal income tax changes will affect state and local taxation,
which often uses federal taxable income as a starting point for computing state and local tax liabilities. Our analysis and interpretation
of this legislation is preliminary and ongoing and there may be material adverse effects resulting from the legislation that we have
not yet identified. While some of the changes made by the tax legislation may adversely affect us, other changes may be beneficial. We
continue to work with our tax advisors and auditors to determine the full impact that the recent tax legislation as a whole will have
on us. We urge our investors to consult with their legal and tax advisors with respect to such legislation and its potential effect on
an investment in our common stock.
Our
operating results are affected by many factors and may fluctuate significantly on a quarterly basis.
Our
operating results may vary substantially from quarter to quarter and may be greater or less than those achieved in the immediately preceding
period or in the comparable period of the prior year. Factors that may cause quarterly results to vary include, but are not limited to,
the following:
| |
• |
the
number of new product introductions by our subsidiaries; |
| |
• |
losses
related to inventory write-offs; |
| |
• |
marketing
exclusivity, if any, which may be obtained on certain new products; |
| |
• |
the
level of competition in the marketplace for certain products; |
| |
• |
our
subsidiaries’ ability to create demand in the marketplace for their products; |
| |
• |
availability
of raw materials and finished products from suppliers; |
| |
• |
our
subsidiaries’ ability to contract manufacturers to make their products; |
| |
• |
Our
dependence on a small number of products for a significant portion of net revenue or income; |
| |
• |
price
erosion and customer consolidation; and |
| |
• |
uncertainty
of future import tariffs. |
The
profitability of our subsidiaries’ product sales is also dependent upon the prices they are able to charge for their products,
the costs to purchase products from third parties, and their ability to manufacture their products in a cost-effective manner. If their
revenues decline or do not grow as anticipated, they may not be able to reduce their operating expenses to offset such declines. Failure
to achieve anticipated levels of revenues could, therefore, significantly harm our operating results for a particular fiscal period.
We
could be subject to significant warranty obligations if our products are defective, which could have a material adverse effect on our
business, financial condition, and results of operations.
In
manufacturing our products, we depend upon third parties for the supply of various components. Many of these components require a significant
degree of technical expertise to design and produce. If we fail to adequately design, or if our suppliers fail to produce components
to specification, or if the suppliers, or we, use defective materials or workmanship in the manufacturing process, the reliability and
performance of our products will be compromised. Our products could contain defects that cannot be repaired easily and inexpensively
that can result some or all of the following:
| |
• |
loss
of customer orders and delay in order fulfillment; |
| |
• |
damage
to our brand reputation; |
| |
• |
increased
cost of our warranty program due to product repair or replacement; |
| |
• |
inability
to attract new customers; |
| |
• |
diversion
of resources from our manufacturing and engineering and development departments into our service department; and |
We
are increasingly dependent on information technology, and our systems and infrastructure face certain risks, including cybersecurity
and data leakage risks.
Significant
disruptions to our information technology systems or breaches of information security could adversely affect our business. In the ordinary
course of business, we collect, store and transmit large amounts of confidential information, and it is critical that we do so in a secure
manner to maintain the confidentiality and integrity of such information. We have also outsourced significant elements of our information
technology infrastructure; as a result, we manage independent vendor relationships with third parties who are responsible for maintaining
significant elements of our information technology systems and infrastructure and who may or could have access to our confidential information.
The size and complexity of our information technology systems, and those of our third-party vendors, make such systems potentially vulnerable
to service interruptions and security breaches from inadvertent or intentional actions by our employees, partners or vendors. These systems
are also vulnerable to attacks by malicious third parties and may be susceptible to intentional or accidental physical damage to the
infrastructure maintained by us or by third parties. Maintaining the secrecy of confidential, proprietary, and/or trade secret information
is important to our competitive business position. While we have taken steps to protect such information and have invested in systems
and infrastructures to do so, there can be no guarantee that our efforts will prevent service interruptions or security breaches in our
systems or the unauthorized or inadvertent wrongful use or disclosure of confidential information that could adversely affect our business
operations or result in the loss, dissemination, or misuse of critical or sensitive information. A breach our security measures or the
accidental loss, inadvertent disclosure, unapproved dissemination, misappropriation or misuse of trade secrets, proprietary information,
or other confidential information, whether as a result of theft, hacking, fraud, trickery or other forms of deception, or for any other
cause, could enable others to produce competing products, use our proprietary technology or information, and/or adversely affect our
business position. Further, any such interruption, security breach, loss or disclosure of confidential information could result in financial,
legal, business, and reputational harm to us and could have a material adverse effect on our business, financial position, results of
operations and/or cash flow.
Product
liability claims against us could be costly and could harm our reputation.
The
sale of our products involves the risk of product liability claims against us. Claims could exceed our product liability insurance coverage
limits. Our insurance policies are subject to various standard coverage exclusions, including damage to the product itself, losses from
recall of our product, and losses covered by other forms of insurance such as workers compensation. We cannot be certain that we will
be able to successfully defend any claims against us, nor can we be certain that our insurance will cover all liabilities resulting from
such claims. In addition, we cannot provide assurance that we will be able to obtain such insurance in the future on terms acceptable
to us, or at all. Regardless of merit or eventual outcome, any product liability claim brought against us could result in harm to our
reputation, decreased demand for our products, costs related to litigation, product recalls, loss of revenue, an increase in our product
liability insurance rates, or the inability to secure coverage in the future, and could have a material adverse effect on our business
by reducing cash collections from customers and limiting our ability to meet our operating cash flow requirements.
Litigation
against us could be costly and time-consuming to defend and could materially and adversely affect our business, financial condition,
and results of operations.
We
are from time to time involved in various claims, litigation matters and regulatory proceedings incidental to our business, including
claims for damages arising out of the use of our products or services and claims relating to intellectual property matters, employment
matters, commercial disputes, competition, sales and trading practices, environmental matters, personal injury, and insurance coverage.
Some of these lawsuits include claims for punitive as well as compensatory damages. The defense of these lawsuits could divert our management’s
attention, and we could incur significant expenses in defending these lawsuits. In addition, we could be required to pay damage awards
or settlements or become subject to unfavorable equitable remedies. Moreover, any insurance or indemnification rights that we could have
may be insufficient or unavailable to protect us against potential loss exposures.
If
we lose our key management personnel, or are unable to attract or retain qualified personnel, it could adversely affect our ability to
execute our growth strategy.
Our
success is dependent, in part, upon our ability to hire and retain management, engineers, marketing and sales personnel, and technical,
research and other personnel who are in high demand and are often subject to competing employment opportunities. Our success will depend
on our ability to retain our current personnel and to attract and retain qualified like personnel in the future. Competition for senior
management, engineers, marketing and sales personnel, and other specialized technicians is intense and we may not be able to retain our
personnel. If we lose the services of any executive officers or key employees, our ability to achieve our business objectives could be
harmed or delayed, which could have a material adverse effect on our daily operations, operating cash flows, results of operations, and
ultimately share price. In general, our officers could terminate their employment at any time without notice for any reason.
Climate
change initiatives could materially and adversely affect our business, financial condition, and results of operations.
Both
domestic and international legislation to address climate change by reducing greenhouse gas emissions and establishing a price on carbon
could create increases in energy costs and price volatility. Considerable international attention is now focused on development of an
international policy framework to address climate change. Proposed and existing legislative efforts to control or limit greenhouse gas
emissions could increase the cost of raw materials derived from sources that generate greenhouse gas emissions. If our suppliers are
unable to obtain energy at a reasonable cost in the future, the cost of our raw materials could be negatively impacted which could result
in increased manufacturing costs.
Our
failure to maintain effective internal controls over financial reporting could have an adverse impact on us.
We
are required to establish and maintain appropriate internal controls over financial reporting. Failure to establish those controls, or
any failure of those controls once established, could adversely impact our public disclosures regarding our business, financial condition
or results of operations. In addition, management’s assessment of internal controls over financial reporting may identify weaknesses
and conditions that need to be addressed in our internal controls over financial reporting or other matters that may raise concerns for
investors. Any actual or perceived weaknesses and conditions that need to be addressed in our internal control over financial reporting,
disclosure of management’s assessment of our internal controls over financial reporting or disclosure of our public accounting
firm’s attestation to or report on management’s assessment of our internal controls over financial reporting may have an
adverse impact on the price of our common stock.
A
control system, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of
the control system are met. In addition, the design of a control system must reflect the fact that there are resource constraints and
the benefit of controls must be relative to their costs. Because of the inherent limitations in all control systems, no system of controls
can provide absolute assurance that all control issues and instances of fraud, if any, within our company have been detected. These inherent
limitations include the realities that judgments in decision-making can be faulty and that breakdowns can occur because of simple error
or mistake. Further, controls can be circumvented by individual acts of some persons, by collusion of two or more persons, or by management
override of the controls. The design of any system of controls is also based in part upon certain assumptions about the likelihood of
future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions.
Over time, a control may become inadequate because of changes in conditions or the degree of compliance with policies or procedures may
deteriorate. Because of inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and may
not be detected.
At
present, we believe that we have effective internal controls in place. However, our management, including our Chief Executive Officer,
cannot guarantee that our internal controls and disclosure controls that we have in place will prevent all possible errors, mistakes
or all fraud.
Our
financial controls and procedures may not be sufficient to ensure timely and reliable reporting of financial information, which, as a
public company, could materially harm our stock price.
We
require significant financial resources to maintain our public reporting status. We cannot assure you we will be able to maintain adequate
resources to ensure that we will not have any future material weakness in our system of internal controls. The effectiveness of our controls
and procedures may in the future be limited by a variety of factors including:
| |
• |
faulty
human judgment and simple errors, omissions or mistakes; |
| |
• |
fraudulent
action of an individual or collusion of two or more people; |
| |
• |
inappropriate
management override of procedures; and |
| |
• |
the
possibility that any enhancements to controls and procedures may still not be adequate to assure timely and accurate financial information. |
Our
internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial
reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles
in the United States of America. Our internal control over financial reporting includes those policies and procedures that (i) pertain
to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets
of the Company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements
in accordance with generally accepted accounting principles, and that receipts and expenditures of the Company are being made only in
accordance with authorizations of management and directors of the Company; and (iii) provide reasonable assurance regarding prevention
or timely detection of unauthorized acquisition, use, or disposition of the Company’s assets that could have a material effect
on the financial statements.
Despite
these controls, because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements.
Therefore, even those systems determined to be effective can provide only reasonable assurance of achieving their control objectives.
Furthermore, smaller reporting companies like us face additional limitations. Smaller reporting companies employ fewer individuals and
can find it difficult to employ resources for complicated transactions and effective risk management. Additionally, smaller reporting
companies tend to utilize general accounting software packages that lack a rigorous set of software controls.
If
we fail to have effective controls and procedures for financial reporting in place, we could be unable to provide timely and accurate
financial information and be subject to investigation by the Securities and Exchange Commission and civil or criminal sanctions.
We
are an “emerging growth company” under the JOBS Act of 2012 and we cannot be certain if the reduced disclosure requirements
applicable to emerging growth companies will make our common stock less attractive to investors.
We
are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”),
and we may take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that
are not “emerging growth companies” including, but not limited to, not being required to comply with the auditor attestation
requirements of section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic
reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and
shareholder approval of any golden parachute payments not previously approved. We cannot predict if investors will find our common stock
less attractive because we may rely on these exemptions. If some investors find our common stock less attractive as a result, there may
be a less active trading market for our common stock and our stock price may be more volatile.
In
addition, Section 107 of the JOBS Act also provides that an “emerging growth company” can take advantage of the extended
transition period provided in Section 7(a)(2)(B) of the Securities Act of 1933 (the “Securities Act”) for complying with
new or revised accounting standards. In other words, an “emerging growth company” can delay the adoption of certain accounting
standards until those standards would otherwise apply to private companies. We are choosing to take advantage of the extended transition
period for complying with new or revised accounting standards.
We
will remain an “emerging growth company” until December 31, 2025, the last day of the fiscal year following the fifth anniversary
of August 31, 2020, the date of the first sale of our Common Stock pursuant to an effective registration statement under the Securities
Act, although we will lose that status sooner if our revenues exceed $1.07 billion, if we issue more than $1 billion in non-convertible
debt in a three year period, or if the market value of our Common Stock that is held by non-affiliates exceeds $700 million as of the
last day of our most recently completed second fiscal quarter.
Our
status as an “emerging growth company” under the JOBS Act may make it more difficult to raise capital as and when we need
it.
Because
of the exemptions from various reporting requirements provided to us as an “emerging growth company” and because we will
have an extended transition period for complying with new or revised financial accounting standards, we may be less attractive to investors
and it may be difficult for us to raise additional capital as and when we need it. Investors may be unable to compare our business with
other companies in our industry if they believe that our financial accounting is not as transparent as other companies in our industry.
If we are unable to raise additional capital as and when we need it, our financial condition and results of operations may be materially
and adversely affected.
There
may be limitations on the effectiveness of our internal controls, and a failure of our control systems to prevent error or fraud may
materially harm our company. If we fail to remediate a material weakness, or if we experience material weaknesses in the future
or otherwise fail to maintain an effective system of internal controls in the future, we may not be able to accurately or timely report
our financial condition or results of operations, which may adversely affect investor confidence in us and, as a result, the value of
our common stock and Warrants.
Prior
to the completion of our initial public offering in August 2020, we had been a private company with limited accounting personnel to adequately
execute our accounting processes and limited supervisory resources with which to address our internal control over financial reporting.
As a newly public company, we have designed a control environment as required of public companies under the rules and regulations
of the SEC.
Proper
systems of internal controls over financial accounting and disclosure controls and procedures are critical to the operation of a public
company. We may be unable to effectively establish such systems, especially in light of the fact that we expect to operate as a publicly
reporting company. This would leave us without the ability to reliably assimilate and compile financial information about our company
and significantly impair our ability to prevent error and detect fraud, all of which would have a negative impact on our company from
many perspectives.
Moreover,
we do not expect that disclosure controls or internal control over financial reporting, even if established, will prevent all errors
and all fraud. A control system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that
the control system’s objectives will be met. Further, the design of a control system must reflect the fact that there are resource
constraints and the benefits of controls must be considered relative to their costs. Because of the inherent limitations in all control
systems, no evaluation of controls can provide absolute assurance that all control issues and instances of fraud, if any, have been detected.
Failure of our control systems to prevent error or fraud could materially adversely impact us.
Geopolitical
conditions, including trade disputes and direct or indirect acts of war or terrorism, could have an adverse effect on our operations
and financial results.
Recently,
Russia initiated significant military action against Ukraine. In response, the U.S. and certain other countries imposed significant sanctions
and export controls against Russia, Belarus and certain individuals and entities connected to Russian or Belarusian political, business,
and financial organizations, and the U.S. and certain other countries could impose further sanctions, trade restrictions, and other retaliatory
actions should the conflict continue or worsen. It is not possible to predict the broader consequences of the conflict, including related
geopolitical tensions, and the measures and retaliatory actions taken by the U.S. and other countries in respect thereof as well as any
counter measures or retaliatory actions by Russia or Belarus in response, including, for example, potential cyberattacks or the disruption
of energy exports, is likely to cause regional instability, geopolitical shifts, and could materially adversely affect regional economies
and the global economy. The situation remains uncertain, and while it is difficult to predict the impact of any of the foregoing, the
conflict and actions taken in response to the conflict could increase our costs, disrupt our supply chain, reduce our sales and earnings,
impair our ability to raise additional capital when needed on acceptable terms, if at all, or otherwise adversely affect our business,
financial condition, and results of operations.
If
we sustain a cyber-attack or suffer privacy or data security breaches that disrupt our information systems or operations, or result in
the dissemination of sensitive personal or confidential information, we could suffer increased costs, exposure to significant liability,
reputational harm, loss of business, and other serious negative consequences.
Our
information technology systems and safety control systems are subject to a growing number of threats from computer programmers, hackers,
and other adversaries that may be able to penetrate our network security and misappropriate our confidential information or that of third
parties, create system disruptions, or cause damage, security issues, or shutdowns. They also may be able to develop and deploy viruses,
worms, and other malicious software programs that attack our systems or otherwise exploit security vulnerabilities. Because the techniques
used to circumvent, gain access to, or sabotage security systems, can be highly sophisticated and change frequently, they often are not
recognized until launched against a target, and may originate from less regulated and remote areas around the world. We may be unable
to anticipate these techniques or implement adequate preventive measures, resulting in potential data loss and damage to our systems.
Our systems are also subject to compromise from internal threats such as improper action by employees, including malicious insiders,
or by vendors, counterparties, and other third parties with otherwise legitimate access to our systems. Our policies, employee training
(including phishing prevention training), procedures, and technical safeguards may not prevent all improper access to our network or
proprietary or confidential information by employees, vendors, counterparties, or other third parties. Our facilities may also be vulnerable
to security incidents or security attacks, acts of vandalism or theft, misplaced or lost data, human errors, or other similar events
that could negatively affect our systems, and our and our members’, data. Additionally, our third-party service providers who process
information on our behalf may cause security breaches for which we are responsible.
Moreover,
we face the ongoing challenge of managing access controls in a complex environment. The process of enhancing our protective measures
can itself create a risk of systems disruptions and security issues. Given the breadth of our operations and the increasing sophistication
of cyber-attacks, a particular incident could occur and persist for an extended period of time before being detected. The extent of a
particular cyber-attack and the steps that we may need to take to investigate the attack may take a significant amount of time before
such an investigation could be completed and full and reliable information about the incident is known. During such time, the extent
of any harm or how best to remediate it might not be known, which could further increase the risks, costs, and consequences of a data
security incident. In addition, our systems must be routinely updated, patched, and upgraded to protect against known vulnerabilities.
The volume of new software vulnerabilities has increased substantially, as has the importance of patches and other remedial measures.
In addition to remediating newly identified vulnerabilities, previously identified vulnerabilities must also be updated. We are at risk
that cyber-attackers exploit these known vulnerabilities before they have been addressed. The complexity of our systems and platforms,
the increased frequency at which vendors are issuing security patches to their products, our need to test patches, and, in some instances,
coordinate with third-parties before they can be deployed, all could further increase our risks.
Any
compromise or perceived compromise of the security of our systems or the systems of one or more of our vendors or service providers could
damage our reputation and brand, cause the termination of relationships with our members, result in disruption or interruption to our
business operations, marketing partners and carriers, reduce demand for our services, and subject us to significant liability and expense,
which would harm our business, operating results, and financial condition.
Risks
Related to SteriLumen’s Business
Certain
ResInnova testing limitations.
Our
claims on the effectiveness of the Disinfecting System against certain pathogens are supported by the analysis of the Disinfecting System
performed in ResInnova’s laboratory. The environment in ResInnova’s laboratory is controlled and does not account for all
real-world variables, which may impact the effectiveness of ultraviolet light in killing microorganisms. Such variables include humidity,
air temperature, and the presence of organic soils that differ from those used in the laboratory tests. If any one of these un-accounted
for variables proves to be significant in the evaluation of the effectiveness of the Disinfecting System, the laboratory results obtained
by ResInnova could be significantly more favorable than the results experienced by our customers. Furthermore, in accordance with CDC
guidelines only laboratories with a biosafety level 3 or 4 may test against SARS-CoV-2, the virus that causes COVID-19. ResInnova is
a biosafety level 2 laboratory and cannot test against SARS-CoV-2 and therefore in its place tested against OC43, which, according to
ResInnova is a common surrogate for SARS-CoV-2 as both are of the Beta genre of coronaviruses. If the results from testing the Disinfecting
System against OC43 are different from the results that would have occurred from testing against SARS-CoV-2, ResInnova’s testing
results could be materially more favorable than the results experienced by our customers with respect to SARS-CoV-2. If the Disinfecting
System is not effective against SARS-CoV-2, this could have a material adverse effect on the Company’s, which would have a material
adverse effect on our business prospects and financial condition.
The
complete and final assembly of the Disinfecting System has not yet received safety certification from a nationally recognized testing
laboratory.
All
of the component parts of the Disinfecting System have been certified by ETL. The ETL listings for ETL Listing numbers: SLR-1 = E519669
for the ribbon unit and SLD-1 = E519957 for the drain unit is new for such a UVC device for the UL and due to incomplete standards
does not guarantee successful deployment and installation at scale in the US. If we are unable or significantly delayed in obtaining
confidence for installations within the marketplace with above certifications, our business and financial prospects will be materially
adversely affected.
In
accordance with an August 24, 1993 Interpretation Letter from OSHA, all electrical equipment must be accepted, certified, labeled, listed,
or otherwise determined that such equipment is safe by a NRTL or by another Federal agency or by a State, municipal, or other local authority
responsible for enforcing occupational safety provisions of the National Electric Safety Code.
If
SteriLumen is unable to develop a successful marketing approach, our business will suffer.
If
SteriLumen fails to develop a successful marketing approach or to manage its growth effectively, our business and financial results will
be materially harmed. Healthcare facilities operate in a highly regulated and dynamic market with changing regulations, codes, standards
and guidelines and as a result are very loyal to vendors they trust. Achieving market acceptance will require extensive customer education
and product validation. Some of the ways in which SteriLumen intends on achieving market acceptance is through working with professional
trade organizations, having articles published in industry trade publications, conducting validation/marketing studies at well-known
hospitals and lab testing. However, there is no assurance that SteriLumen will be successful in organizing these efforts or if the results
from them will be positive. SteriLumen has sought and may also seek in the future to expand its business through complementary or strategic
acquisitions of other businesses, products or assets such as its recent acquisition of the Airocide product line, or through joint ventures,
strategic partnerships or other arrangements with established and trusted disinfection companies. Any such acquisitions, joint ventures
or other business combinations may involve significant integration challenges, operational complexities and time consumption and require
substantial resources and effort. It may also disrupt SteriLumen’s ongoing businesses, which may adversely affect its relationships
with customers, employees and others with whom it has business or other dealings. Further, if SteriLumen is unable to realize synergies
or other benefits expected to result from any acquisitions, joint ventures or other business combinations, or to generate additional
revenue to offset any unanticipated inability to realize these expected synergies or benefits, its growth and ability to compete may
be impaired, which would require it and us to focus additional resources on the integration of operations rather than other profitable
areas of its business, and may otherwise cause a material adverse effect on our business, results of operations and financial condition.
However, failure to acquire or partner with established and trusted disinfection companies would prevent SteriLumen’s products
from being part of a bundle of disinfection products that could be sold all at once, which could have a material adverse effect on the
financial results of our business.
The
air purification market is fragmented and competitive and we may not be able to compete successfully with our existing competitors or
new entrants into the markets we serve.
The
air purification market is fragmented and competitive. SteriLumen’s competition varies by product line, customer classification
and geographic market. The principal competitive factors in our industry are quality of product, pricing, service and delivery capabilities
and availability of product. We will compete with many local, regional and national air purification distributors and dealers. In addition,
some air purification suppliers might sell and distribute their products directly to our customers, and the volume of such direct sales
could increase in the future. Additionally, distributors of products similar to those distributed by us may elect to sell and distribute
to our customers in the future or enter into exclusive supplier arrangements with other distributors. Some of our competitors have greater
financial resources and may be able to withstand sales or price decreases more effectively than we can. We also expect to continue to
face competition from new market entrants. We may be unable to continue to compete effectively with these existing or new competitors,
which could have a material adverse effect on our financial condition and results of operations.
If
we are unable to execute our plan to distribute SteriLumen’s Airocide products, we may not be able to generate revenues and your
investment could be materially adversely affected.
We
acquired the rights to manufacture and sell our Airocide products in February of 2021 and have not yet fully scaled to execute our plan
to sell and distribute SteriLumen’s Airocide products. The success of the Airocide business will depend on the execution of our
plan and the acceptance of Airocide products by the consumer and commercial markets. Achieving such acceptance will require significant
marketing investment. Once we execute our plan to sell and distribute the Airocide products, it may not be accepted by consumers at sufficient
levels to support our operations and build our business. If SteriLumen’s Airocide products are not accepted at sufficient levels,
our business could fail.
We
are subject to significant regulatory oversight and changes in applicable regulatory requirements could adversely affect our business.
We
may become subject to significant government regulation, by the EPA and, to a certain extent, by Congress, other federal agencies and
foreign, state and local authorities. Depending upon the circumstances, noncompliance with legislation or regulations promulgated by
these entities could result in the suspension or revocation of our licenses or registrations, the termination or loss of contracts or
the imposition of contractual damages, civil fines or criminal penalties any of which could have a material adverse effect on our business,
financial condition and results of operations. Furthermore, the adoption or modification of laws or regulations relating to UVC or air
purification or other areas of our business could limit or otherwise adversely affect the manner in which we currently conduct our business.
If we are required to comply with new regulations or legislation or new interpretations of existing regulations. This compliance could
cause us to incur additional expenses or alter our business model.
SteriLumen’s
Scientific Air business is highly dependent on a sole distributor and any disruption in their operations could have a material adverse
effect on our business, results of operations and financial condition.
Currently,
a sole distributor distributes Scientific Air products pursuant to an exclusive distribution agreement which, on a proforma basis, accounted
for approximately 19% of Scientific Air’s revenues for the twelve months ended December 31, 2021. Scientific Air’s revenues
were approximately 22% of the combined revenues of Scientifc Air and the Company for the twelve month period ended December 31, 2021.
Any number of factors relating to such distributor, including labor disruptions, catastrophic weather events, financial difficulties,
solvency problems, such distributor prioritizing the distribution of other products over ours or contractual disputes between us and
such distributor could lead to uncertainty in the distribution of a material amount of our Scientific Air products. While we are in the
process of identifying additional distributors, there can be no assurance that we will be able to do so in the near or foreseeable future.
If SteriLumen experiences distribution disruptions with respect to Scientific Air products, it may not be able to develop alternate sourcing
quickly and could cause it to alter production schedules or suspend production entirely. If any such disruptions occur it could have
a material adverse effect on our business, results of operations and financial condition.
SteriLumen’s
Disinfecting System business is highly dependent on its suppliers’ and any disruption in their operations could have a material
adverse effect on our business, results of operations and financial condition.
SteriLumen’s
Disinfecting System business will be dependent upon the continued ability of its suppliers to deliver systems, components, raw materials,
and finished disinfection products. Its UVC LEDs are manufactured in South Korea and then shipped to China or the United States for assembly
with all other components, which if manufactured in China, the finished products are shipped to Long Beach, California for warehousing
and distribution, otherwise the units are shipped from Mount Vernon. Any number of factors, including labor disruptions, catastrophic
weather events, contractual or other disputes with suppliers, and supplier financial difficulties or solvency problems could disrupt
its suppliers’ operations and lead to uncertainty in its supply chain or cause supply disruptions, which could, in turn, disrupt
its operations. If SteriLumen experiences supply disruptions, it may not be able to develop alternate sourcing quickly. Any disruption
of its production schedule caused by an unexpected shortage of systems, components, raw materials or parts even for a relatively short
period of time could cause it to alter production schedules or suspend production entirely. If any such disruptions occur it could have
a material adverse effect on our business, results of operations and financial condition.
SteriLumen’s
business is highly dependent on market perceptions of it and the safety and quality of its products.
Market
perceptions of SteriLumen’s business are very important to us, especially market perceptions of the safety and quality of SteriLumen’s
products. If any of its products or similar products that other companies distribute are subject to market withdrawal or recall or are
proven to be, or are claimed to be, harmful to end users, then this could have a material adverse effect on our business, results of
operations and financial condition. Also, because SteriLumen’s business is dependent on market perceptions, negative publicity
associated or perceived to be associated with its business, products or product pricing could have a material adverse impact on our business,
results of operations and financial condition.
Customers
may be hesitant in adopting UV light-based technologies, and our inability to overcome this hesitation could limit the market acceptance
of our products and our market share.
Our
UV light disinfection systems represent relatively new technologies in the market. Only a small percentage of professional medical institutions
or hospitality providers are immediately willing to conduct sanitation using our systems. Our future success will depend on our ability
to increase demand for our products by demonstrating to a broad spectrum of medical professional, dentists, hospitality industry, their
patients and customers, the potential performance advantages of our UV light systems over traditional methods of disinfection over competitive
UV light systems, and our inability to do so could have a material adverse effect on our business, financial condition, and results of
operations.
Conventional
germicidal UV light was historically considered as a human health hazard if improperly used and can lead to skin cancer and cataracts.
We may experience long sales cycles because healthcare facilities and hotels and other facilities may be slow to adopt new technologies
on a widespread basis and admit that such technologies can sanitize public space without damaging public health. As a result, we generally
are required to invest a significant amount of time and resources to educate general public about the benefits of our products in comparison
to competing products and technologies before completing a sale, if any. Factors that could inhibit adoption of UV technologies by healthcare
facilities or hospitality companies include the initial cost and concerns about the safety, efficacy, and reliability of our UV systems.
In addition, economic pressure, caused, for example, by an economic slowdown as a result of Coronavirus, changes in health care reimbursement
or by competitive factors in a specific market, could make businesses reluctant to purchase substantial capital equipment or invest in
new technologies. Customer acceptance will depend on the recommendations of governmental authorities, as well as other factors, including
the relative effectiveness, safety, reliability, and comfort of our systems as compared to other instruments and methods for performing
disinfecting procedures.
If
future data proves to be inconsistent with our research results or if competitors’ products present more favorable results our
revenues could decline and our business, financial condition, and results of operations could be materially and adversely affected.
Even
though our disinfecting devices are protected with patents, if new studies or comparative studies generate results that are not as favorable
as our research results, our revenues could decline. Additionally, if future studies indicate that our competitors’ products are
more effective or safer than ours, our revenues could decline. Furthermore, hospitals and businesses could choose not to purchase our
UV light sanitation systems until they receive additional published long-term clinical evidence and recommendations from prominent hospitals
and businesses that indicate our UV light sanitation systems are effective for disinfecting applications.
We
may face competition from other companies, many of which have substantially greater resources than we do. If we do not successfully develop
and commercialize enhanced or new products that remain competitive with products or alternative technologies developed by others, we
could lose revenue opportunities and customers and our ability to grow our business would be impaired.
A
number of competitors have substantially greater capital resources, larger customer bases, larger technical, sales and marketing forces
and stronger reputations with target customers than ours. We compete with a number of domestic and foreign companies that market traditional
chemical sanitation products, as well as companies that market UV technologies. The marketplace is highly fragmented and very competitive.
We expect that the rapid technological changes occurring in the health care industry could lead to the entry of new competitors, particularly
if UV disinfecting increases market acceptance. If we do not compete successfully, our revenue and market share could decline, which
would impact our ability to meet our operating cash flow requirements and our business, financial condition, and results of operations
could be adversely affected.
Our
long-term success depends upon our ability to (i) distinguish our products through improving our product performance and pricing, protecting
our intellectual property, improving our customer support, accurately timing the introduction of new products, and developing sustainable
distribution channels worldwide; and (ii) develop and successfully commercialize new products, new or improved technologies, and additional
applications for our UV light sanitation systems. We may not be able to distinguish our products and commercialize any new products,
new or improved technologies, or additional applications for our UV light disinfecting systems.
We
could incur problems in manufacturing our products.
In
order to grow our business, we must expand our manufacturing capabilities to produce the systems and accessories necessary to meet any
demand we may experience. We could encounter difficulties in increasing the production of our products, including problems involving
production capacity and yields, quality control and assurance, component supply, and shortages of qualified personnel. In addition, before
we can begin commercial manufacture of our products, we must ensure our manufacturing facilities, processes, and quality systems, and
the manufacture of our UV light sanitation systems, quality control, and documentation policies and procedures.From time to time, we
could expend significant resources in obtaining, maintaining, and addressing our compliance with various federal and requirements that
may be subject to changes. Our success will depend in part upon our ability to manufacture our products without FDA approval or compliance
with and other regulatory requirements.
We
have not experienced significant quality issues with components of our products supplied by third parties, however, we could in the future.
Our future success depends on our ability to manufacture our products on a timely basis with acceptable manufacturing costs, while at
the same time maintaining good quality control and complying with applicable regulatory requirements, and an inability to do so could
have a material adverse effect on our product sales, cash collections from customers, and our ability to meet operating cash flow requirements,
which could have a material adverse effect on our business, financial condition, and results of operations.
Adverse
publicity regarding our technology or products could negatively impact us.
Adverse
publicity regarding any of our products or similar products marketed or sold by others could negatively affect us. If any studies raise
or substantiate concerns regarding the efficacy or safety of our products or other concerns, our reputation could be harmed and demand
for our products could diminish, which could have a material adverse effect on growth in new customers and sales of our products, leading
to a decline in revenues, cash collections, and ultimately our ability to meet operating cash flow requirements.
Rapidly
changing standards and competing technologies could harm demand for our products, result in significant additional costs, and have a
material adverse effect on our business, financial condition, and results of operations.
The
markets in which our products compete are subject to rapid technological change, evolving industry standards, changes in the regulatory
environment, and frequent introductions of new devices and evolving sanitizing solutions and practices, specifically catalyzed by the
impact of the Coronavirus pandemic. Competing products could emerge that render our products uncompetitive or obsolete. We cannot guarantee
that we will successfully identify new product opportunities, identify new and innovative applications of our technology, or be financially
or otherwise capable of completing the research and development required to bring new products to market in a timely manner. An inability
to expand our product offerings or the application of our technology could limit our growth. In addition, we could incur higher manufacturing
costs if manufacturing processes or standards change, and we could need to replace, modify, design, or build and install equipment, all
of which would require additional capital expenditures.
We
could be unable to effectively manage and implement our growth strategies, which could have a material adverse effect on our business,
financial condition, and results of operations.
Our
growth strategy includes expanding our product line and applications by developing enhancements and transformational innovations, including
new solutions for various fields and industries. Expansion of our existing product line and entry into new applications divert the use
of our resources and systems, require additional resources that might not be available (or available on acceptable terms), may require
regulatory approvals, result in new or increasing competition, could require longer implementation times or greater start-up expenditures
than anticipated, and could otherwise fail to achieve the desired results in a timely fashion, if at all. These efforts could also require
that we successfully commercialize new technologies in a timely manner, price them competitively and cost-effectively, and manufacture
and deliver sufficient volumes of new products of appropriate quality on time. We could be unable to increase our sales and earnings
by expanding our product offerings in a cost-effective manner, and we could fail to accurately predict future customer needs and preferences
or to produce viable technologies. In addition, we could invest heavily in research and development of products that do not lead to significant
revenue. Even if we successfully innovate and develop new products and product enhancements, we could incur substantial costs in doing
so. In addition, promising new products could fail to reach the market or realize only limited commercial success because of efficacy
or safety concerns, failure to achieve positive clinical outcomes, or uncertainty over third-party reimbursement.
International
sales may comprise a significant portion of SteriLumen’s revenues and will be subject to risks associated with operating in domestic
and international markets.
International
sales may comprise a significant portion of SteriLumen’s revenue, and we intend to continue to pursue and expand our international
business activities. Political and economic conditions outside the United States could make it difficult for us to increase our international
revenue or to operate abroad. International operations are subject to many inherent risks, which could have a material adverse effect
on our revenues and operating cash flow, including among others:
| |
• |
adverse
changes in tariffs and trade restrictions; |
| |
• |
political,
social, and economic instability and increased security concerns; |
| |
• |
fluctuations
in foreign currency exchange rates; |
| |
• |
longer
collection periods and difficulties in collecting receivables from foreign entities; |
| |
• |
exposure
to different legal standards; |
| |
• |
transportation
delays and difficulties of managing international distribution channels; |
| |
• |
reduced
protection for our intellectual property in some countries; |
| |
• |
difficulties
in obtaining domestic and foreign export, import, and other governmental approvals, permits, and licenses, and compliance with foreign
laws; |
| |
• |
the
imposition of governmental controls; |
| |
• |
unexpected
changes in regulatory or certification requirements; |
| |
• |
difficulties
in staffing and managing foreign operations; and |
| |
• |
potentially
adverse tax consequences and the complexities of foreign value-added tax systems. |
We
believe that international sales may represent a significant portion of SteriLumen’s revenue, and we intend to expand its international
operations. In international markets where our sales are denominated in U.S. dollars, an increase in the relative value of the dollar
against the currency in such markets could indirectly increase the price of our products in those markets and result in a decrease in
sales. We do not currently engage in any transactions as a hedge against risks of loss due to foreign currency fluctuations. However,
we could do so in the future.
SteriLumen’s
collaborations with outside scientists and consultants may be subject to restriction and change.
SteriLumen
works with scientists at academic and other institutions, and consultants who assist it in its research, development, and design efforts.
These scientists and consultants have provided, and we expect that they will continue to provide, valuable advice on SteriLumen’s
programs. These scientists and consultants are not our or SteriLumen’s employees, may have other commitments that would limit their
future availability to SteriLumen and typically will not enter into non-compete agreements with SteriLumen. If a conflict of interest
arises between their work for SteriLumen and their work for another entity, SteriLumen may lose their services. In addition, SteriLumen
will be unable to prevent them from establishing competing businesses or developing competing products. For example, if a key scientist
acting as a principal investigator in any of SteriLumen’s clinical trials identifies a potential product or compound that is more
scientifically interesting to his or her professional interests, his or her availability to remain involved in its clinical trials could
be restricted or eliminated.
SteriLumen
has entered into or intends to enter into non-competition agreements with certain of SteriLumen’s employees. These agreements prohibit
its employees, if they cease working for it, from competing directly against it or working for its competitors for a limited period.
However, under current law, SteriLumen may be unable to enforce these agreements against certain of its employees and it may be difficult
for it to restrict their competitors from gaining the expertise its former employees gained while working for it. If SteriLumen cannot
enforce its employees’ non-compete agreements, it may be unable to prevent its competitors from benefiting from the expertise of
its former employees.
Risks
Related to MunnWorks’ Business
The
custom design decorative framed mirror supply market is highly competitive, and we may not be able to compete successfully.
MunnWorks
operates within the highly competitive custom design decorative framed mirror supply market, which is characterized by competition from
a number of other manufacturers. Competition is further intensified during economic downturns. MunnWorks competes with numerous large
national and regional companies for, among other things, customers, raw materials and skilled management and labor resources. Purchase
volumes have fluctuated substantially from time to time in the past, and we expect such fluctuations to occur from time to time in the
future. Some of its competitors have greater financial, marketing and other resources than it does and, therefore, may be able to adapt
to changes in customer preferences more quickly, devote more resources to the marketing and sale of their products, generate greater
national brand recognition or adopt more aggressive pricing policies than MunnWorks can.
In
addition, some of our competitors may resort to price competition to sustain or gain market share and manufacturing capacity utilization,
and MunnWorks may have to adjust the prices on some of its products to stay competitive, which could reduce its revenues. MunnWorks may
not ultimately succeed in competing with other manufacturers and distributors in its market, which may have a material adverse effect
on our business, financial condition or results of operations.
MunnWorks’
possible failure to develop new products or respond to changing consumer preferences and purchasing practices could have a material adverse
effect on our business, financial condition or results of operations.
The
custom design decorative framed mirror supply market is subject to changing consumer trends, demands and preferences. The uncertainties
associated with developing and introducing new products, such as gauging changing consumer preferences and successfully developing, manufacturing,
marketing and selling new products, could lead to, among other things, rejection of a new product line, reduced demand and price reductions
for our products. If MunnWorks’ products do not keep up with consumer trends, demands and preference, it could lose market share,
which could have a material adverse effect on our business, financial condition or results of operations.
Changes
to the buying strategies of MunnWorks’ customers could also affect its ability to compete. Further, the volatile and challenging
economic environment of recent years has caused shifts in trends, demands, preferences and purchasing practices and changes in the business
models and strategies of its customers. Shifts in consumer preferences, which may or may not be long-term, have altered the quantity,
type and prices of products demanded by the end-consumer and MunnWorks’ customers. If it does not timely and effectively identify
and respond to these changing consumer preferences and purchasing practices, its relationships with our customers could be harmed, the
demand for its products could be reduced and its market share could be negatively affected.
MunnWorks’
independent sales force may not be effective.
MunnWorks
hires independent sales representatives who have primary responsibility for contacting existing and potential customers and are paid
on a commission basis. While this sales model has proven successful in the past, to continue to be effective, sales representatives will
need to continue to be extremely knowledgeable about MunnWorks’ products. However, these independent sales representatives may
be selling products from different non-competitive sellers, which could prevent them from focusing on MunnWorks’ products and providing
the required information to the customers, which could have a material adverse effect on our business, results of operations and financial
condition.
MunnWorks’
business is highly dependent on its suppliers’ and any disruption in their operations could have a material adverse effect on our
business, results of operations and financial condition.
MunnWorks’
operations will be dependent upon the continued ability of its suppliers to deliver components, raw materials, and finished products.
Although many of its products are manufactured at our corporate headquarters in New York, many of its products are manufactured overseas.
Any number of factors, including labor disruptions, catastrophic weather events, contractual or other disputes with suppliers, and supplier
financial difficulties or solvency problems could disrupt its suppliers’ operations and lead to uncertainty in its supply chain
or cause supply disruptions, which could, in turn, disrupt its operations. If MunnWorks experiences supply disruptions, it may not be
able to develop alternate sourcing quickly. Any disruption of its production schedule caused by an unexpected shortage of systems, components,
raw materials or parts even for a relatively short period of time could cause it to alter production schedules or suspend production
entirely. If any such disruptions occur it could have a material adverse effect on our business, results of operations and financial
condition.
MunnWorks’
business is highly dependent on market perceptions of it and the quality of its products.
Market
perceptions of MunnWorks’ business are very important to us, especially market perceptions of the quality of MunnWorks’ products.
Because MunnWorks’ business is dependent on market perceptions, negative publicity associated or perceived to be associated with
its business, products or product pricing could have a material adverse impact on our business, results of operations and financial condition.
Risks
Related to Our Intellectual Property
If
the patents that we own or license, or our other intellectual property rights, do not adequately protect our technologies, we could lose
market share to our competitors and be unable to operate our business profitably.
Our
future success depends, in part, on our ability to obtain and maintain patent protection for our products and technology, to preserve
our trade secrets and to operate without infringing the intellectual property of others. We rely on patents to establish and maintain
proprietary rights in our technology and products. We currently possess a number of issued patents and patent applications with respect
to our products and technology. However, we cannot ensure that any additional patents will be issued, that the scope of any patent protection
will be effective in helping us address our competition, or that any of our patents will be held valid if subsequently challenged. It
is also possible that our competitors could independently develop similar or more desirable products, duplicate our products, or design
products that circumvent our patents. The laws of foreign countries may not protect our products or intellectual property rights to the
same extent as the laws of the United States. In addition, there have been recent changes in the patent laws and rules of the U.S. Patent
and Trademark Office (“USPTO”), and there could be future proposed changes that, if enacted, have a significant impact on
our ability to protect our technology and enforce our intellectual property rights. If we fail to protect our intellectual property rights
adequately, our competitive position could be adversely affected, and there could be a material adverse effect on sales, cash collections,
and our ability to meet operating cash flow requirements.
If
third parties claim that we infringe their intellectual property rights, we could incur liabilities and costs and have to redesign or
discontinue selling certain products, which could have a material adverse effect on our business, financial condition, and results of
operations.
We
face substantial uncertainty regarding the impact that other parties’ intellectual property positions will have on UV light applications.
From time to time, we expect to continue to receive, notices of claims of infringement, misappropriation, or misuse of other parties’
proprietary rights. Some of these claims could lead to litigation. We may not prevail in any future intellectual property infringement
litigation given the complex technical issues and inherent uncertainties in litigation. Any claims, with or without merit, could be time-consuming
and distracting to management, result in costly litigation, or cause product shipment delays. Adverse determinations in litigation could
subject us to significant liability and could result in the loss of proprietary rights. A successful lawsuit against us could also force
us to cease selling or redesign products that incorporate the infringed intellectual property. Additionally, we could be required to
seek a license from the holder of the intellectual property to use the infringed technology, and we may not be able to obtain a license
on acceptable terms, or at all.
Patent
terms are limited and we may not be able to effectively protect our products and business.
Patents
have a limited lifespan. In the U.S., the natural expiration of a patent is generally 20 years after it is filed. Although various extensions
may be available, the life of a patent, and the protection it affords, is limited. In addition, upon issuance in the U.S., the patent
term may be extended based on certain delays caused by the applicant(s) or the USPTO. Even if we obtain effective patent rights for all
our current patent applications, we may not have sufficient patent terms or regulatory exclusivity to protect our products, and our business
and results of operations would be adversely affected.
We
may be subject to claims challenging the inventorship of our patents and other intellectual property.
We
may be subject to claims that former employees, collaborators or other third parties have an interest in or right to compensation with
respect to our patents or other intellectual property as an inventor or co-inventor. For example, we may have inventorship disputes arise
from conflicting obligations of consultants or others who are involved in developing our product candidates. Litigation may be necessary
to defend against these and other claims challenging inventorship or claiming the right to compensation. If we fail in defending any
such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights, such as exclusive ownership of,
or right to use, valuable intellectual property. Such an outcome could have a material adverse effect on our business. Even if we are
successful in defending against such claims, litigation could result in substantial costs and be a distraction to management and other
employees. To the extent that our employees have not effectively waived the right to compensation with respect to inventions that they
helped create, they may be able to assert claims for compensation with respect to our future revenue may be successful. As a result,
we may receive less revenue from future products if such claims are successful which in turn could impact our future profitability.
Changes
in U.S. patent law could diminish the value of patents in general, thereby impairing our ability to protect our products.
As
is the case with other equipment manufacturing companies, our success is heavily dependent on intellectual property, particularly patents.
Obtaining and enforcing patents in the biotechnology industry involves both technological and legal complexity. Therefore, obtaining
and enforcing patents is costly, time-consuming, and inherently uncertain. In addition, the U.S. has recently enacted and is currently
implementing wide-ranging patent reform legislation. Recent U.S. Supreme Court rulings have narrowed the scope of patent protection available
in certain circumstances and weakened the rights of patent owners in certain situations. In addition to increasing uncertainty with regard
to our ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents,
once obtained. Depending on future actions by the U.S. Congress, the federal courts and the USPTO, the laws and regulations governing
patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce our existing patents and
patents that we might obtain in the future.
We
may not be able to protect our intellectual property rights throughout the world.
Filing,
prosecuting and defending patents on product candidates in all countries throughout the world would be prohibitively expensive, and our
intellectual property rights in some countries outside the U.S. can be less extensive than those in the U.S. In addition, the laws of
some foreign countries do not protect intellectual property rights to the same extent as federal and state laws in the U.S. Competitors
may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and may also export
otherwise infringing products to territories where we have patent protection, but enforcement is not as strong as that in the U.S. These
products may compete with our products and our patents or other intellectual property rights may not be effective or sufficient to prevent
them from competing.
Many
companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The
legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents, trade secrets
and other intellectual property protection, particularly those relating to biotechnology products, which could make it difficult for
us to stop the infringement of our patents or marketing of competing products in violation of our proprietary rights generally. Proceedings
to enforce our patent rights in foreign jurisdictions, whether or not successful, could result in substantial costs and divert our efforts
and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly and our
patent applications at risk of not issuing and could provoke third parties to assert claims against us. We may not prevail in any lawsuits
that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce
our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual
property that we develop or license.
Risks
Relating to Ownership of Our Securities
The
public price of our Common Stock may be volatile, and could, following a sale decline significantly and rapidly.
The
offering price for the shares will be determined by negotiations between us and the underwriters and may not be indicative of prices
that will prevail in the open market following this offering. The market price of our common stock may decline below the initial offering
price, and you may not be able to sell your shares of our common stock at or above the price you paid in the offering, or at all. Following
this Offering, the public price of our common stock in the secondary market will be determined by private buy and sell transaction orders
collected from broker-dealers.
We
may not be able to maintain a listing of our Common Stock.
In
order for our common stock to continue to be listed on Nasdaq, we must meet certain financial and liquidity criteria to maintain such
listing. If we violate the maintenance requirements for continued listing of our common stock, our common stock may be delisted. In addition,
our board may determine that the cost of maintaining our listing on a national securities exchange outweighs the benefits of such listing.
A delisting of our common stock from Nasdaq may materially impair our stockholders’ ability to buy and sell our common stock and
could have an adverse effect on the market price of, and the efficiency of the trading market for, our common stock. In addition, the
delisting of our common stock could significantly impair our ability to raise capital.
Our
directors and officers beneficially own more than 50% of the voting power of our voting stock as of December 31, 2021, and will be able
to exert a controlling influence over our business affairs and matters submitted to stockholders for approval.
Our
directors and officers have voting control over more than 50% of our voting stock (which excludes 158,959 shares underlying options and
warrants issued to the directors and officers, but does include 2,000 shares of the Company’s Super Voting Preferred Stock beneficially
owned by Max Munn, our Interim Chief Executive Officer, President and director , which is entitled to 1,000 votes per share (2,000,000
votes in aggregate) and votes with the common stock as a single class. As a result, our directors and officers will have control over
all matters submitted to our stockholders for approval, including the election of directors, amendments to our certificate of incorporation
and bylaws, the approval of any business combination and any other significant corporate transaction. These actions may be taken even
if they are opposed by other stockholders. This concentration of ownership may also have the effect of delaying or preventing a change
of control of our company or discouraging others from making tender offers for our shares, which could prevent our stockholders from
receiving a premium for their shares.
Our
directors and officers may have interests different from yours.
We
have not paid dividends on our Common Stock in the past and do not expect to pay dividends on our Common Stock in the future, and any
return on investment may be limited to the value of our stock.
We
have never paid cash dividends on our Common Stock and do not anticipate paying cash dividends on our Common Stock in the foreseeable
future. We currently intend to retain any future earnings to support the development of our business and do not anticipate paying cash
dividends in the foreseeable future. Our payment of any future dividends will be at the discretion of our board of directors after taking
into account various factors, including, but not limited to, our financial condition, operating results, cash needs, growth plans and
the terms of any credit agreements that we may be a party to at the time. In addition, our ability to pay dividends on our Common Stock
is limited by the terms of our Series A Perpetual Preferred Stock and may be limited by Delaware state law. Accordingly, investors must
rely on sales of their Common Stock after price appreciation, which may never occur, as the only way to realize a return on their investment.
The
elimination of personal liability against our directors and officers under Delaware law and the existence of indemnification rights held
by our directors, officers and employees may result in substantial expenses.
Our
Amended and Restated Certificate of Incorporation and our Bylaws eliminate the personal liability of our directors and officers to us
and our stockholders for damages for breach of fiduciary duty as a director or officer to the extent permissible under Delaware law.
Further, our amended and restated certificate of incorporation and our Bylaws and individual indemnification agreements we have entered
with each of our directors and executive officers provide that we are obligated to indemnify each of our directors or officers to the
fullest extent authorized by the Delaware law and, subject to certain conditions, advance the expenses incurred by any director or officer
in defending any action, suit or proceeding prior to its final disposition. Those indemnification obligations could expose us to substantial
expenditures to cover the cost of settlement or damage awards against our directors or officers, which we may be unable to afford. Further,
those provisions and resulting costs may discourage us or our stockholders from bringing a lawsuit against any of our current or former
directors or officers for breaches of their fiduciary duties, even if such actions might otherwise benefit our stockholders, indemnification
rights held by our directors, officers and employees may result in substantial expenses.
Our
certificate of incorporation will designate the Court of Chancery of the State of Delaware as the exclusive forum for certain litigation
that may be initiated by our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for
disputes with us.
Our
amended and restated certificate of incorporation specifies that, unless we consent in writing to the selection of an alternative forum,
the Court of Chancery of the State of Delaware shall be the sole and exclusive forum for: (a) any derivative action or proceeding brought
on behalf of the Company, (b) any action asserting a claim of breach of a fiduciary duty owed by any director, officer, employee or agent
of the Company to the Company or the Company’s stockholders, (c) any action asserting a claim arising pursuant to any provision
of the Delaware General Corporation Law, the Company’s Certificate of Incorporation or the Bylaws, or (d) any action asserting
a claim governed by the internal affairs doctrine, in each case subject to said Court of Chancery having personal jurisdiction over the
indispensable parties named as defendants therein. Any person or entity purchasing or otherwise acquiring any interest in shares of our
capital stock shall be deemed to have notice of and to have consented to the provisions of our amended and restated certificate of incorporation
described above.
We
believe these provisions benefit us by providing increased consistency in the application of Delaware law by chancellors particularly
experienced in resolving corporate disputes, efficient administration of cases on a more expedited schedule relative to other forums
and protection against the burdens of multi-forum litigation. However, the provision may have the effect of discouraging lawsuits against
our directors, officers, employees and agents as it may limit any stockholder’s ability to bring a claim in a judicial forum that
such stockholder finds favorable for disputes with us or our directors, officers, employees or agents. The enforceability of similar
choice of forum provisions in other companies’ certificates of incorporation has been challenged in legal proceedings, and it is
possible that, in connection with any applicable action brought against us, a court could find the choice of forum provisions contained
in our restated certificate of incorporation to be inapplicable or unenforceable in such action. If a court were to find the choice of
forum provision contained in our restated certificate of incorporation to be inapplicable or unenforceable in an action, we may incur
additional costs associated with resolving such action in other jurisdictions, which could adversely affect our business, financial condition
or results of operations.
Our
revenues, operating results and cash flows may fluctuate in future periods and we may fail to meet investor expectations, which may cause
the price of our Common Stock to decline.
Variations
in our quarterly and year-end operating results are difficult to predict and our income and cash flows may fluctuate significantly from
period to period, which may impact our board of directors’ willingness or legal ability to declare a monthly dividend. If our operating
results fall below the expectations of investors or securities analysts, the price of our Common Stock could decline substantially. Specific
factors that may cause fluctuations in our operating results include:
| |
• |
demand
and pricing for our products and services; |
| |
• |
introduction
of competing products; |
| |
• |
our
operating expenses which fluctuate due to growth of our business; and |
| |
• |
variable
sales cycle and implementation periods for content and services. |
The
market price of our securities could be substantially affected by various factors.
The
market price of our Common Stock could be subject to wide fluctuations in response to numerous factors. The price of the our Common Stock
that will prevail in the market after this offering may be higher or lower than the offering price depending on many factors, some of
which are beyond our control and may not be directly related to our operating performance.
These
factors include, but are not limited to, the following:
| |
• |
trading
prices of securities generally; |
| |
• |
general
economic and financial market conditions; |
| |
• |
government
action or regulation; |
| |
• |
the
financial condition, performance and prospects of us and our competitors; |
| |
• |
changes
in financial estimates or recommendations by securities analysts with respect to us or our competitors in our industry; |
| |
• |
our
issuance of preferred equity or debt securities; and |
| |
• |
actual
or anticipated variations in quarterly operating results of us and our competitors. |
As
a result of these and other factors, investors who purchase the Common Stock in this offering may experience a decrease, which could
be substantial and rapid, in the market price of the Common Stock, including decreases unrelated to our operating performance or prospects.
You
should consult your own independent tax advisor regarding any tax matters arising with respect to the securities offered in connection
with this offering.
Participation
in this offering could result in various tax-related consequences for investors. All prospective purchasers of the resold securities
are advised to consult their own independent tax advisors regarding the U.S. federal, state, local and non-U.S. tax consequences relevant
to the purchase, ownership and disposition of the resold securities in their particular situations.
We
have not paid dividends on our common stock in the past and do not expect to pay dividends on our common stock in the future, and any
return on investment may be limited to the value of our stock.
We
have never paid cash dividends on our common stock and do not anticipate paying cash dividends on our common stock in the foreseeable
future. We currently intend to use any future earnings to pay dividends on our Series A Preferred Stock and to support the development
of our business and do not anticipate paying cash dividends on our common stock in the foreseeable future. Our payment of any future
dividends will be at the discretion of our board of directors after taking into account various factors, including, but not limited to,
our financial condition, operating results, cash needs, growth plans and the terms of any credit agreements that we may be a party to
at the time. In addition, our ability to pay dividends on our common stock may be limited by Delaware state law. Accordingly, investors
must rely on sales of their common stock after price appreciation, which may never occur, as the only way to realize a return on their
investment. Investors seeking cash dividends should not purchase our common stock.
We
will have broad discretion in using the proceeds of this offering and we may not effectively spend the proceeds.
We
will use the net proceeds of this offering for general corporate purposes, including investments and acquisitions. We have not allocated
any specific portion of the net proceeds to any particular purpose, and our management will have the discretion to allocate the proceeds
as it determines. We will have significant flexibility and broad discretion in applying the net proceeds of this offering, and we may
not apply these proceeds effectively. Our management might not be able to yield a significant return, if any, on any investment of these
net proceeds, and you will not have the opportunity to influence our decisions on how to use our net proceeds from this offering.
Provisions
of our Amended and Restated Certificate of Incorporation could delay or prevent the acquisition or sale of our business.
Our
Amended and Restated Certificate of Incorporation permits our Board of Directors to designate new series of preferred stock and issue
those shares without any vote or action by our stockholders. Such newly authorized and issued shares of preferred stock could contain
terms that grant special voting rights to the holders of such shares that make it more difficult to obtain stockholder approval for an
acquisition of our business or increase the cost of any such acquisition.
We
expect that we will need to raise additional capital, and raising additional funds by issuing additional equity securities or with additional
debt financing may cause dilution to shareholders or restrict our operations.
We
expect that we will need to raise additional capital in the future. We may raise additional funds through public or private equity or
debt offerings or other financings, as well as borrowings from banks or through the issuance of debt securities.
Any
new debt financing we enter into may involve covenants that restrict our operations more than our current outstanding debt. These restrictive
covenants could include limitations on additional borrowings and specific restrictions on the use of our assets, as well as prohibitions
or limitations on our ability to create liens, pay dividends, receive distributions from our subsidiaries, redeem or repurchase our stock
or make investments. These factors could hinder our access to capital markets and limit or delay our ability to carry out our capital
expenditure plan or pursue other opportunities beyond the current capital expenditure plan. Further, we may incur substantial costs in
pursuing any capital-raising transactions, including investment banking, legal and accounting fees. On the other hand, if we are unable
to obtain capital when needed, on reasonable terms and in amounts sufficient to fund our obligations, expenses, capital expenditure plan
and other strategic initiatives, we could be forced to suspend, delay or curtail these plans or initiatives or could default on our contractual
commitments. Any such outcome could negatively affect our business, performance, liquidity and prospects.
The
Series A Preferred Stock ranks junior to all of our indebtedness and other liabilities.
In
the event of our bankruptcy, liquidation, dissolution or winding-up of our affairs, our assets will be available to pay obligations on
the Series A Preferred Stock only after all of our indebtedness and other liabilities have been paid. The rights of holders of the Series
A Preferred Stock to participate in the distribution of our assets will rank junior to the prior claims of our current and future creditors
and any future series or class of preferred stock we may issue that ranks senior to the Series A Preferred Stock. Also, the Series A
Preferred Stock effectively ranks junior to all existing and future indebtedness and to the indebtedness and other liabilities of any
future subsidiaries. Our existing subsidiaries are, and future subsidiaries would be, separate legal entities and have no legal obligation
to pay any amounts to us in respect of dividends due on the Series A Preferred Stock.
We
have incurred and may in the future incur substantial amounts of debt and other obligations that will rank senior to the Series A Preferred
Stock. If we are forced to liquidate our assets to pay our creditors, we may not have sufficient assets to pay amounts due on any or
all of the Series A Preferred Stock then outstanding.
The
Company’s ability to pay dividends and to meet its debt obligations largely depends on the performance of its subsidiaries and
the ability to utilize the cash flows from those subsidiaries.
The
Company is a holding company for SteriLumen and MunnWorks and has no material assets other than its equity interests in those subsidiaries.
Therefore, the only current revenue source for future dividends on the Series A Preferred Stock is from its subsidiaries. SteriLumen
is an early-stage designer and marketer of disinfection systems with limited operating history spanning from December 2016. As a result
of the acquisition of the Akida assets, we expect that SteriLumen will have positive cash flow, but this cash flow combined with cash
flows from MunnWorks may not be sufficient to pay dividends on the Series A Preferred Stock. Our utilization of cash has been and will
continue to be highly dependent on SteriLumen’s product development programs and cash flow from Airocide and MunnWorks’ operations.
Our cash expenses will be highly dependent on the product development programs SteriLumen chooses to pursue, the progress of these product
development programs, the results of SteriLumen’s validation/marketing studies, the terms and conditions of SteriLumen’s
contracts with service providers and manufacturing contractors, and the terms of recruitment of facilities in our validation/marketing
studies.
In
addition, the subsidiaries and any joint ventures or other entities accounted for as equity method investments are separate and distinct
legal entities that are not obligated to pay dividends or make loans or distributions to the Company, whether to enable us to pay principal
and interest on our debt, our other obligations or dividends on our Common Stock or preferred stock (including the Series A Preferred
Stock offered hereby), and could be precluded from paying any such dividends or making any such loans or distributions under certain
circumstances, including, without limitation, as a result of legislation, regulation, court order, contractual restrictions or in times
of financial distress. The inability to access capital from our subsidiaries and entities accounted for as equity method investments
as well from the capital markets could have a material adverse effect on the Company’s cash flows and financial condition and,
on our ability, to pay dividends on the Series A Preferred Stock.
The
Series A Preferred Stock will be effectively subordinated to the obligations of our subsidiaries.
We
are a holding company and conduct substantially all of our operations through our subsidiaries. Our right to receive any assets of any
of our subsidiaries upon their liquidation, reorganization or otherwise, and thus the ability of a holder of our Series A Preferred Stock
to benefit indirectly from such distribution, will be subject to the prior claims of the subsidiaries’ creditors. Even if we were
a creditor of any of our subsidiaries, our rights as a creditor would be subordinate to any security interest in the assets of those
subsidiaries and any indebtedness of those subsidiaries senior to that held by us.
We
must adhere to prescribed legal requirements and we must also have sufficient cash in order to be able to pay dividends on the Preferred
Stock.
In
accordance with Section 170 of the Delaware General Corporation Law (“DGCL”), we may only declare and pay cash dividends
on the Series A Preferred Stock if we have either net profits during the fiscal year in which the dividend is declared and/or the preceding
fiscal year, or a “surplus”, meaning the excess, if any, of our net assets (total assets less total liabilities) over our
capital. If the capital of the Company, computed in accordance with Sections 154 and 244 of the DGCL, shall have been diminished by depreciation
in the value of its property, or by losses, or otherwise, to an amount less than the aggregate amount of the capital represented by the
issued and outstanding stock of all classes having a preference upon the distribution of assets, the directors of the Company cannot
declare and pay out of such net profits any dividends upon any shares of any classes of its capital stock until the deficiency in the
amount of capital represented by the issued and outstanding stock of all classes having a preference upon the distribution of assets
shall have been repaired. We can provide no assurance that we will satisfy such requirements in any given year. Further, even if we have
the legal ability to declare a dividend, we may not have sufficient cash to pay dividends on the Series A Preferred Stock. Our ability
to pay dividends may be impaired if any of the risks described in this prospectus actually occur. Also, payment of our dividends depend
upon our financial condition and other factors as our board of directors may deem relevant from time to time. We cannot assure you that
our businesses will generate sufficient cash flow from operations or that future borrowings will be available to us in an amount sufficient
to enable us to pay dividends on the Series A Preferred Stock.
The
Series A Preferred Stock represents perpetual equity interests in us, and investors should not expect us to redeem the Series A Preferred
Stock on any such date that the Series A Preferred Stock becomes redeemable by us or on any particular date afterwards.
The
Series A Preferred Stock represents perpetual equity interests in us, and it has no maturity or mandatory redemption, is not redeemable
at the option of investors under any circumstances. As a result, unlike our indebtedness, the Series A Preferred Stock will not give
rise to a claim for payment of a principal amount at a particular date. As a result, holders of the Series A Preferred Stock may be required
to bear the financial risks of an investment in the Series A Preferred Stock for an indefinite period of time.
We
may redeem the Series A Preferred Stock on a date or dates determined in our sole discretion and the investor may not find a new investment
with a comparable dividend or interest rate.
The
Series A Preferred Stock will be a perpetual equity security. This means that it will have no maturity or mandatory redemption date and
will not be redeemable at the option of the holders. We may, at our option, after July 16, 2022, redeem the Series A Preferred Stock,
in whole or in part, at any time or from time to time as we may determine in our sole discretion, for cash at a redemption price equal
to $27.50 until July 16, 2026 and $25.00 thereafter. We may have an incentive to redeem the Series A Preferred Stock voluntarily if we
can do so without paying the premium or if market conditions allow us to issue other preferred stock or debt securities at a rate that
is lower than the dividend rate on the Series A Preferred Stock. If we redeem the Series A Preferred Stock, then from and after the redemption
date, dividends will cease to accrue on shares of Series A Preferred Stock, the shares of Series A Preferred Stock shall no longer be
deemed outstanding and all rights as a holder of those shares will terminate, except the right to receive the redemption price plus accumulated
and unpaid dividends, if any, payable upon redemption. If we choose to redeem the Series A Preferred Stock, you may not be able to reinvest
the redemption proceeds in a comparable security at an effective dividend or interest rate as high as the dividend payable on the Series
A Preferred Stock.
The
conversion feature may not adequately compensate you, and the conversion and redemption features of the Series A Preferred Stock may
make it more difficult for a party to take over our company and may discourage a party from taking over our company.
Upon
the occurrence of a Delisting Event or Change of Control, holders of the Series A Preferred Stock will have the right (unless, prior
to the Delisting Event Conversion Date or Change of Control Conversion Date, as applicable, we have provided or provide notice of our
election to redeem the Series A Preferred Stock) to direct the depositary to convert some or all of the Series A Preferred Stock into
our common stock (or equivalent value of alternative consideration), and under these circumstances we will also have a special optional
redemption right to redeem the Series A Preferred Stock. See “Description of Series A Preferred—Conversion Rights”
and “—Special Optional Redemption.” Upon such a conversion, the holders will be limited to a maximum number of
shares of our common stock equal to the Share Cap multiplied by the number of shares of Series A Preferred Stock converted. If the Common
Stock Price is less than $4.67 (which is approximately 50% of the closing sale price per share of our common stock on July 12, 2021),
subject to adjustment, the holders will receive a maximum of 5.353319 shares of our common stock per share of Series A Preferred
Stock, which may result in a holder receiving value that is less than the liquidation preference of the Series A Preferred Stock. In
addition, those features of the Series A Preferred Stock may have the effect of inhibiting a third party from making an acquisition proposal
for our company or of delaying, deferring or preventing a change of control of our company under circumstances that otherwise could provide
the holders of our common stock and Series A Preferred Stock with the opportunity to realize a premium over the then-current market price
or that stockholders may otherwise believe is in their best interests.
The
Series A Preferred Stock has not been rated.
We
have not sought to obtain a rating for the Series A Preferred Stock, and the Series A Preferred Stock may never be rated. It is possible,
however, that one or more rating agencies might independently determine to assign a rating to the Series A Preferred Stock or that we
may elect to obtain a rating of the Series A Preferred Stock in the future. In addition, we may elect to issue other securities for which
we may seek to obtain a rating. If any ratings are assigned to the Series A Preferred Stock in the future or if we issue other securities
with a rating, such ratings, if they are lower than market expectations or are subsequently lowered or withdrawn, could adversely affect
the market for or the market value of the Series A Preferred Stock. Ratings only reflect the views of the issuing rating agency or agencies
and such ratings could at any time be revised downward or withdrawn entirely at the discretion of the issuing rating agency. A rating
is not a recommendation to purchase, sell or hold any particular security, including the Series A Preferred Stock. Ratings do not reflect
market prices or suitability of a security for a particular investor and any future rating of the Series A Preferred Stock may not reflect
all risks related to us and our business, or the structure or market value of the Series A Preferred Stock.
If
Nasdaq delists the Series A Preferred Stock, investors’ ability to make trades in the Series A Preferred Stock could be limited.
Our
Series A Preferred Stock is traded on the Nasdaq Capital Market under the symbol “AUVIP.” In order to continue to be listed
on the Nasdaq Capital Market, we must meet and maintain certain financial, distribution, and share price levels. Generally, this means
having a minimum number of publicly held shares of Series A Preferred Stock (generally 1,000,000 shares), a minimum market value (generally
$5,000,000), a minimum net income from continuing operations (generally $750,000) and a minimum number of holders (generally 300 public
holders). If we are also unable to meet the listing standards for the Nasdaq Capital Market, we may apply to have our Series A Preferred
Stock quoted by OTC Markets. If we are unable to maintain listing for the Series A Preferred Stock on the Nasdaq Capital Market, the
ability to transfer or sell shares of the Series A Preferred Stock will be limited and the market value of the Series A Preferred Stock
will likely be materially adversely affected. Moreover, since the Series A Preferred Stock has no stated maturity date, investors may
be forced to hold shares of the Series A Preferred Stock indefinitely while receiving stated dividends thereon when, as and if authorized
by our board of directors and paid by us with no assurance as to ever receiving the liquidation value thereof.
The
market for our Series A Preferred Stock may not provide investors with adequate liquidity.
Liquidity
of the market for the Series A Preferred Stock depends on a number of factors, including prevailing interest rates, our financial condition
and operating results, the number of holders of the Series A Preferred Stock, the market for similar securities and the interest of securities
dealers in making a market in the Series A Preferred Stock. We cannot predict the extent to which investor interest in our Company will
maintain a trading market in our Series A Preferred Stock, or how liquid that market will be. If an active market is not maintained,
investors may have difficulty selling shares of our Series A Preferred Stock.
We
are allowed to issue shares of other series of preferred stock that rank above or equal to the Series A Preferred Stock as to dividend
payments and rights upon our liquidation, dissolution or winding up of our affairs without first obtaining the approval of the holders
of our Series A Preferred Stock. The issuance of additional shares of Series A Preferred Stock and/or additional series of preferred
stock could have the effect of reducing the amounts available to the Series A Preferred Stock upon our liquidation or dissolution or
the winding up of our affairs. It also may reduce dividend payments on the Series A Preferred Stock if we do not have sufficient funds
to pay dividends on all Series A Preferred Stock outstanding and other classes or series of stock with equal or senior priority with
respect to dividends. Future issuances and sales of senior or pari passu preferred stock, or the perception that such issuances and sales
could occur, may cause prevailing market prices for the Series A Preferred Stock and our Common Stock to decline and may adversely affect
our ability to raise additional capital in the financial markets at times and prices favorable to us.
Market
interest rates may materially and adversely affect the value of the Series A Preferred Stock.
One
of the factors that will influence the price of the Series A Preferred Stock is the dividend yield on the Series A Preferred Stock (as
a percentage of the market price of the Series A Preferred Stock) relative to market interest rates. Continued increase in market interest
rates may lead prospective purchasers of the Series A Preferred Stock to expect a higher dividend yield (and higher interest rates would
likely increase our borrowing costs and potentially decrease funds available for dividend payments). Thus, higher market interest rates
could cause the market price of the Series A Preferred Stock to materially decrease.
Holders
of the Series A Preferred Stock may be unable to use the dividends-received deduction and may not be eligible for the preferential tax
rates applicable to “qualified dividend income.”
Distributions
paid to corporate U.S. holders of the Series A Preferred Stock may be eligible for the dividends-received deduction, and distributions
paid to non-corporate U.S. holders of the Series A Preferred Stock may be subject to tax at the preferential tax rates applicable to
“qualified dividend income,” only if we have current or accumulated earnings and profits, as determined for U.S. federal
income tax purposes. Additionally, we may not have sufficient current earnings and profits during future fiscal years for the distributions
on the Series A Preferred Stock to qualify as dividends for U.S. federal income tax purposes. If the distributions fail to qualify as
dividends, U.S. holders would be unable to use the dividends-received deduction and may not be eligible for the preferential tax rates
applicable to “qualified dividend income.” If any distributions on the Series A Preferred Stock with respect to any
fiscal year are not eligible for the dividends-received deduction or preferential tax rates applicable to “qualified dividend income”
because of insufficient current or accumulated earnings and profits, it is possible that the market value of the Series A Preferred Stock
might decline.
A
holder of Series A Preferred Stock has essentially no voting rights.
The
holders of Series A Preferred Stock will not be entitled to vote on any matter that comes before our stockholders for a vote unless such
vote is required by the DGCL. This means that, unless it is required by Delaware law, you will not have the right to participate in any
decisions regarding or affecting our Company or your investment, including the election of directors or any extraordinary events, such
as a merger, acquisition or other similar transaction. Decisions on those matters could be made in a manner that materially and adversely
affects your interests. Our shares of the Super Voting Preferred Stock and our Common Stock are the only classes of our securities that
carry full voting rights. See “Description of the Securities--Series A Preferred Stock—Voting Rights.”
Future
issuances of preferred stock may reduce the value of the Series A Preferred Stock.
Upon
the completion of the offering described in this prospectus, we may sell additional shares of preferred stock on terms that may differ
from those described in this prospectus. Such shares could rank on parity with or senior to the Series A Preferred Stock offered hereby
as to dividends, voting or rights upon liquidation, winding up or dissolution. The creation and subsequent issuance of additional classes
of preferred stock on parity with the Series A Preferred Stock, could dilute the interests of the holders of Series A Preferred Stock
offered hereby. Any issuance of preferred stock that is senior to the Series A Preferred Stock would not only dilute the interests of
the holders of Series A Preferred Stock offered hereby, but also could affect our ability to pay distributions on, redeem or pay the
liquidation preference on the Series A Preferred Stock.
If
we are not paying full dividends on any future dividend parity stock, we will not be able to pay full dividends on the Series A Preferred
Stock.
When
dividends are not paid in full on outstanding shares of any class or series of our stock that ranks on a parity with the Series A Preferred
Stock in the payment of dividends (“dividend parity stock”) for a dividend period, all dividends declared with respect to
shares of Series A Preferred Stock and all shares of outstanding dividend parity stock for such dividend period shall be declared pro
rata so that the respective amounts of such dividends declared bear the same ratio to each other as all accrued but unpaid dividends
per share on the shares of Series A Preferred Stock and all shares of outstanding dividend parity stock for such dividend period bear
to each other. Therefore, if we are not paying full dividends on any outstanding shares of dividend parity stock, we will not be able
to pay full dividends on the Series A Preferred Stock.
Item 1B.
Unresolved Staff Comments
None.
Item
2. Properties.
We
lease and maintain our primary offices and manufacturing facility at 150 N. Macquesten Parkway, Mount Vernon, NY 10550 and at 3625 Kennesaw
North Industrial Pkwy NW, Kennesaw, GA. 30144. We also lease offices at 8480 East Orchard Road, Greenwood Village, Colorado 80111 and
1301 W. Copans Road, Pompano Beach, Florida, 33064, We do not currently own any real estate.
Item
3. Legal Proceedings
On
February 25, 2022, James Doyle, a former Chief Operating Officer of the Company filed an arbitration claim against the Company for approximately
$1.5 million plus attorneys’ fees and other costs with the American Arbitration Association in the State of New York for severance
pay and other claims after being terminated by the Company for cause. The Company maintains that the claims Mr. Doyle have made are without
merit and is therefore not obligated to pay any amounts to Mr. Doyle. The arbitration proceeding was initiated pursuant to the arbitration
provision in Mr. Doyle’s employment agreement with the Company. The Company is in the process of responding to Mr. Doyle’s
claims and plans on filing one or more counterclaims against Mr. Doyle. While the Company is unable to provide any assurances as to the
ultimate outcome of this matter, it believes that all of Mr. Doyle’s claims are without merit and intends to vigorously defend
against them. The Company is currently unable to estimate the costs and timing of the arbitration, including any potential damages, if
Mr. Doyle were to prevail on any of his claims.
From
time to time, we may be subject to legal proceedings and claims in the ordinary course of business. We may in the future receive claims
from third parties asserting, among other things, infringement of their intellectual property rights. Future litigation may be necessary
to defend ourselves, our partners and our customers by determining the scope, enforceability and validity of third-party proprietary
rights, or to establish our proprietary rights. The results of any future litigation cannot be predicted with certainty, and regardless
of the outcome, litigation can have an adverse impact on us because of defense and settlement costs, diversion of management resources,
and other factors. To date, we have not been made aware of any actual, pending or threatened litigation against the Company.
Item
4. Mine Safety Disclosures
Not
applicable.
PART
II
Item
5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities.
Market
Information
Our
common stock has been traded on the Nasdaq Capital Market market under the symbol AUVI.
Holders
As
of March 31, 2022, there were 30 stockholders of record of our common stock. Because many of our shares of common stock are held by brokers
and other institutions on behalf of stockholders, this number is not representative of the total number of beneficial owners of our stock.
Dividends
We
have never paid cash dividends on our Common Stock and do not anticipate paying cash dividends on our Common Stock in the foreseeable
future. We currently intend to retain any future earnings to support the development of our business and do not anticipate paying cash
dividends in the foreseeable future. Our payment of any future dividends will be at the discretion of our board of directors after taking
into account various factors, including, but not limited to, our financial condition, operating results, cash needs, growth plans and
the terms of any credit agreements that we may be a party to at the time. In addition, our ability to pay dividends on our Common Stock
is limited by the terms of our Series A Perpetual Preferred Stock and may be limited by Delaware state law. Accordingly, investors must
rely on sales of their Common Stock after price appreciation, which may never occur, as the only way to realize a return on their investment.
Recent
Sales of Unregistered Securities
During
the fiscal year ended December 31, 2021 we made the following unregistered sales of our securities:
On
January 1, 2021 the Company issued in the aggregate 62,500 unvested shares of its Common Stock to its independent directors, which vest
on January 1, 2022.
On
January 14, 2021 the Company issued 3,320 shares of its Common Stock pursuant to the exercise of a common stock purchase warrant.
On
January 25, 2021, the Company issued 3,000 shares of its Common Stock to a consultant as payment for services.
On
February 8, 2021, the Company issued 1,375,000 shares of its Common Stock to the members of Akida Holdings LLC and one of its former
employees in connection with the purchase of substantially all of the assets of Akida Holdings LLC.
On
March 5, 2021 the Company issued 185 shares of its Common Stock pursuant to the exercise of a common stock purchase warrant.
On
March 12, 2021 the Company issued 13,630 shares of its Common Stock pursuant to the exercise of a common stock purchase warrant.
On
May 17, 2021 the Company issued 717 shares of its Common Stock pursuant to the exercise of a common stock purchase warrant.
On
May 28, 2021 the Company issued 12,000 shares of its Common Stock to a consultant as payment for services.
On
September 28, 2021, the Company issued 300,000 shares of its Common Stock to the members of KES Science & Technology, Inc. in connection
with the purchase of substantially all of the assets of Kes Science & Technology, Inc.
On
October 13, 2021, the Company issued 400,000 shares of its Common Stock to the members of Old SAM Partners, LLC (SAM), formerly known
as Scientific Air Management, LLC, in connection with the purchase of substantially all of the assets of SAM. 200,000 shares vest immediately,
and the balance of 200,000 shares will vest March 31, 2023 if not previously cancelled due to certain EBITDA targets for 2021 and 2022.
The EBITDA targets for 2021 have not been met.
On
March 4, 2021, the Company granted an unvested option to purchase 309,836 shares of its Common Stock at an exercise price of $7.80 to
Max Munn, its President, pursuant to his employment agreement with the Company. The option vests monthly over a three-year period.
On April
5, 2021 the Company granted an option to purchase 70,000 shares of its Common Stock at an exercise price of $9.66 to Michael Riccio,
its Chief Financial Officer, pursuant to his employment offer from the Company. These options were cancelled and reissued on September
28, 2021 at an exercise price of $6.53.
On
June 22, 2021 the Company granted options to purchase 88,000 shares of Common Stock at an exercise price of $9.79 to certain of its employees
pursuant to the Company’s stock option plan.
On
September 23, 2021 the Company granted options to purchase 10,000 shares of Common Stock at an exercise price of $6.74 to certain of
its employees pursuant to the Company’s stock option plan.
The foregoing
issuances were exempt from registration under Section 4(a)(2) of the Securities Act or Regulation D promulgated thereunder.
Securities
Authorized for Issuance under Equity Compensation Plans
The
following table sets forth information concerning grants of option awards pursuant to employees, directors, consultants and other independent
contractors during the year ended December 31, 2021:
| Plan
category | |
Number
of securities to be
issued upon exercise of
outstanding options,
warrants and rights | |
Weighted-average
exercise price of
outstanding options,
warrants and rights | |
Number
of securities remaining available
for future issuance under equity
compensation plans |
| Equity
compensation plans approved by security holders(1) | |
| 333,000 | | |
$ | 6.49 | | |
| 267,000 | |
| Equity
compensation plans not approved by security holders(2) | |
| 311,314 | | |
| 7.78 | | |
| — | |
| Total | |
| 644,314 | | |
$ | 7.11 | | |
| — | |
| (1) |
The
Applied UV, Inc. 2020 Omnibus Incentive Plan (the “Plan”) permits grants of equity awards to employees, directors, consultants
and other independent contractors. Our board of directors and shareholders have approved a total reserve of 600,000 shares for issuance
under the Plan. |
| |
|
| (2) |
Includes
309,564 options granted to Max Munn pursuant his employment contract. |
Transfer
Agent
The
transfer agent for the common stock is Vstock Transfer LLC, 18 Lafayette Place, Woodmere, New York, telephone (212) 828-8436.
Purchases
of Equity Securities by the Issuer and Affiliated Purchasers
None.
Item
6. [Reserved].
Item
7. Management’s Discussion and Analysis of Financial Condition and Results of Operations.
Overview
Applied
UV is focused on the development and acquisition of technology that addresses infection control in the healthcare, hospitality,
commercial and municipal markets. The Company has two wholly owned subsidiaries - SteriLumen, Inc. (“SteriLumen”) and MunnWorks,
LLC (“MunnWorks”).
SteriLumen’s
connected platform for Data Driven Disinfection™ applies the power of ultraviolet light (UVC) to destroy pathogens safely, thoroughly,
and automatically, addressing the challenge of healthcare-acquired infections ("HAIs"). Targeted for use in facilities that have
high customer turnover such as hospitals, hotels, commercial facilities, and other public spaces, the Company’s Lumicide™
platform uses UVC LEDs in several patented designs for infection control in and around high-traffic areas, including sinks and restrooms,
killing bacteria, viruses, and other pathogens residing on hard surfaces within the devices’ proximity. The Company’s patented
in-drain disinfection device, Lumicide Drain, is the only product on the market that addresses this critical pathogen-intensive location.
SteriLumen’s
Airocide® air purification devices are research backed, clinically proven and developed for NASA with assistance from the University
of Wisconsin. Airocide® is listed as an FDA Class II Medical device, utilizes a proprietary photocatalytic (PCO) bioconversion technology
that draws air into a reaction chamber that converts damaging molds, microorganisms, dangerous airborne pathogens, destructive VOCs,
allergens, odors and biological gasses into harmless water vapor and green carbon dioxide without producing ozone or other harmful byproducts. Airocide®
applications include healthcare, hospitality, grocery chains, wine making facilities, commercial real estate, schools, dental offices,
post-harvest, grocery, food processing, storage and transportation, cannabis facilities, and homes.
SteriLumen’s
Scientific Air product was developed initially for healthcare facilities and is helping hospitals across the country address the growing
need for effective and safe airborne infection prevention. Utilizing Scientific Air systems, hospitals report significant reductions
in viable airborne pathogens as well as significant declines in non-viable particulates including elimination of odor and VOC's. Scientific
Air products produce no harmful by-products, provide rapid, portable, whole-room disinfection via a patented 3-phase design, are safe
and fast-acting in occupied spaces, and have been proven and tested in facilities with EPA and FDA guidance compliance.
According
to Resource and Markets, the UV Disinfection market is expected to reach $8 billion by 2026 as technology continues to improve and the
focus on stopping the spread of contagious diseases increases. The Center for Disease Control states that 1 in 25 patients have at least
one Hospital Associated Infection (HAI) annually and that 3 million serious infections occur every year in long-term care facilities.
Scientists globally have been advocating improving air quality post pandemic, significantly boosting global adoption to control airborne
pathogen transmission. Governments globally mandating health agencies to address air quality via grants and mechanisms to ease visitation
and protect facilities against future pathogens (Centers for Medicare and Medicaid Services – CMS February 2022 Long-term
Care Initiative).
Indoor
air quality has become an even more important issue as world economies start the recovery process. In 2021, 39 scientists reiterated
the need for a "paradigm shift" and called for improvements in, "how we view and address the transmission of respiratory
infections to protect against unnecessary suffering and economic losses."
In
addition to this, the global air purifier market size is set to grow exponentially. It was valued at $9.24 billion in 2021 and is predicted
to grow to approximately $22.84 billion by 2030. According to Precedence Research, the immense demand for air purification and sterilization
in the US will be driven by the commercial sector.
SteriLumen’s
product portfolio is one of the only research-backed, clinically proven pure-play air and surface disinfection technology companies with
international distribution and globally recognized end users, with product developed for NASA. In addition to the numerous recognized
research institutions and globally recognized names who published the reports that were completed by the acquired companies, Airocide
was independently proven to kill SARS, MRSA and Anthrax, in addition to removing damaging molds, microorganisms, destructive VOC’s,
allergens, odors, and biological gases. Also, SteriLumen’s air purification (Airocide) and surface disinfection (Lumicide) were
independently tested and proven to kill both Candida Auris (Resinnova Laboratories) and SARS CoV-2 (COVID-19) (MRIGlobal).
SteriLumen’s
product portfolio is used by globally recognized names including; Walmart, Whole Foods, SuperValue, Delmonte, Esmeralda, Joel Gott Wines,
Opus One, Athena Healthcare, NYC Health and Hospitals, Kaiser Permanente, Advent Health, University Rochester Medical Center and Baptist
Health South Florida. This past year, the SteriLumen product portfolio expanded its reach and deployed its air purification products
into Boston Red Sox Fenway Park and Jet Blue Park, The Palace Versaille , Uruguayan School Systems, Tennessee Department of Corrections,
Armed Forces Research Institute of Medical Sciences (AFRIMS), US Army Aberdeen Proving Grounds and Schools throughout South Korea.
The
Company works with a global base of distributors to sell both SteriLumen air purification and disinfection products and the MunnWorks
product lines. The past year, the Company has signed distribution agreements covering Africa (360BioPharma), US Healthcare (Axis), Lootah
Batta Water and Environment Sign Exclusive Distribution Agreement for Airocide(R) Air Purification Systems for the United Arab Emirates,
and Plandent a wholly owned subsidiary of Planmeca Oy (Scandinavia). SteriLumen plans to continue to expand its global distribution base
of significant breadth and scale to introduce the entire SteriLumen’s air purification product lines to new markets, including
building management, commercial real estate, retail, healthcare, cannabis and environmental health and safety, leveraging the networks
of the recent acquisitions described above.
MunnWorks
is a manufacturer of custom designed fine mirrors specifically for the hospitality industry with one manufacturing facility in Mount
Vernon, New York. Our goal is to contribute to the creation of what our design industry clients seek: manufacturing better framed mirrors
on budget and on time. As part of our long-term strategy, the Company has instituted multi-site production for high-value items, complicated
designs and finishes. Our headquarters in Mount Vernon, NY serves as the center for multi-country manufacturing. The Company works with
a satellite network of artisans and craftsmen, including gilders, carvers, and old-world finishers.
Acquisitions
In
February of 2021, the Company acquired all the assets and assumed certain liabilities of Akida Holdings, LLC (“Akida”). At
the time of the acquisition, Akida owned the Airocide™ system of air purification technologies, originally developed for NASA with
assistance from the University of Wisconsin at Madison, that uses a combination of UVC and a proprietary, titanium dioxide based photocatalyst
that may help to accelerate the reopening of the global economy with applications in the hospitality, hotel, healthcare, nursing homes,
grocer, wine, commercial buildings and retail sectors. The Airocide™ system has been used by brands such as NASA, Whole Foods,
Dole, Chiquita, Opus One, Sub-Zero Refrigerators and Robert Mondavi Wines. Akida had contracted KES Science & Technology, Inc. (“KES”)
to manufacture, warehouse and distribute the Airocide™ system and Akida’s contractual relationship with KES was assigned
to and assumed by the Company as part of the acquisition.
On
September 28, 2021, the Company acquired all the assets and assumed certain liabilities of KES. At the time of the acquisition, KES was
principally engaged in the manufacturing and distribution of the Airocide™ system of air purification technologies and misting
systems. KES also had the exclusive right to the sale and distribution of the Airocide™ system in certain markets. This acquisition
consolidates all of manufacturing, sale and distribution of the Airocide™ system under the SteriLumen brand and expands the Company’s
market presence in food distribution, post-harvest produce, wineries, and retail sectors. The Company sells its products throughout the
United States, Canada, and Europe.
On
October 13, 2021, we acquired substantially all of the assets of Old SAM Partners, LLC F/K/A Scientific Air Management, LLC, which owned
a line of air purification technologies (“Scientific Air’) for a purchase price of $9.5 million in cash and 200,000 fully
vested shares of our Common Stock (the “Vested Shares”) and 200,000 shares of our Common Stock that are subject to vesting
(the “Earnout Shares”) (the “Scientific Air Acquisition”). The number of shares of Common Stock included in the
purchase price was based on a per share value of $10.00. Scientific Air is a provider of whole-room, aerosol chamber and laboratory certified
air disinfection machines that use a combination of UVC and a proprietary, patented system to eliminate airborne bacteria, mold, fungi,
viruses, volatile organic compounds, and many odors without producing any harmful by-products. The units are well suited for larger spaces
within a facility and are mobile with industrial grade casters allowing for movement throughout a facility to address increased bio burdern
from larger meetings or increased human traffic.
Principal
Factors Affecting Our Financial Performance
Our operating
results are primarily affected by the following factors:
| • |
|
our
ability to acquire new customers or retain existing customers; |
| • |
|
our
ability to offer competitive product pricing; |
| • |
|
our
ability to broaden product offerings; |
| • |
|
industry
demand and competition; and |
| • |
|
market
conditions and our market positions |
Results
of Operations
Year
Ended December 31, 2021 Compared to the Year Ended December 31, 2020
| | |
Year
Ended | |
Year
Ended |
| | |
| December
31, 2021 | | |
| December
31, 2020 | |
| | |
| Hospitality | | |
| Disinfection | | |
| Corporate | | |
| Total | | |
| Hospitality | | |
| Disinfection | | |
| Corporate | | |
| Total | |
| Net
Sales | |
$ | 5,943,664 | | |
$ | 5,723,915 | | |
$ | — | | |
$ | 11,667,579 | | |
$ | 5,732,734 | | |
$ | — | | |
$ | — | | |
$ | 5,732,734 | |
| Cost
of Goods Sold | |
| 4,488,652 | | |
| 3,080,541 | | |
| — | | |
| 7,569,193 | | |
| 4,723,398 | | |
| — | | |
| — | | |
| 4,723,398 | |
| Gross
Profit | |
| 1,455,012 | | |
| 2,643,374 | | |
| — | | |
| 4,098,386 | | |
| 1,009,336 | | |
| — | | |
| — | | |
| 1,009,336 | |
| Research
and development | |
| — | | |
| 53,408 | | |
| — | | |
| 53,408 | | |
| — | | |
| 310,672 | | |
| — | | |
| 310,672 | |
| Selling,
General and Administrative | |
| 2,281,460 | | |
| 6,110,206 | | |
| 2,950,046 | | |
| 11,341,712 | | |
| 1,749,962 | | |
| 770,512 | | |
| 1,491,082 | | |
| 4,011,556 | |
| Total
Operating expenses | |
| 2,281,460 | | |
| 6,163,614 | | |
| 2,950,046 | | |
| 11,395,120 | | |
| 1,749,962 | | |
| 1,081,184 | | |
| 1,491,082 | | |
| 4,322,228 | |
| Operating
Loss | |
| (826,448 | ) | |
| (3,520,240 | ) | |
| (2,950,046 | ) | |
| (7,296,734 | ) | |
| (740,626 | ) | |
| (1,081,184 | ) | |
| (1,491,082 | ) | |
| (3,312,892 | ) |
| Other
(Expense) Income | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Change
in Fair Market Value of Warrant Liability | |
| — | | |
| — | | |
| 66,862 | | |
| 66,862 | | |
| — | | |
| — | | |
| — | | |
| — | |
| Forgiveness
of paycheck protection program loan | |
| — | | |
| — | | |
| 296,827 | | |
| 296,827 | | |
| — | | |
| — | | |
| — | | |
| — | |
| Loss
on contingent consideration | |
| — | | |
| — | | |
| (574,000 | ) | |
| (574,000 | ) | |
| — | | |
| — | | |
| — | | |
| — | |
| Other
income | |
| — | | |
| — | | |
| 24,871 | | |
| 24,871 | | |
| — | | |
| — | | |
| 10,936 | | |
| 10,936 | |
| Total
Other (Expense) Income | |
| — | | |
| — | | |
| (185,440 | ) | |
| (185,440 | ) | |
| — | | |
| — | | |
| 10,936 | | |
| 10,936 | |
| Loss
Before Provision for Income Taxes | |
| (826,448 | ) | |
| (3,520,240 | ) | |
| (3,135,486 | ) | |
| (7,482,174 | ) | |
| (740,626 | ) | |
| (1,081,184 | ) | |
| (1,480,146 | ) | |
| (3,301,956 | ) |
| Provision
(Benefit) from Income Taxes | |
| — | | |
| — | | |
| (91,819 | ) | |
| (91,819 | ) | |
| — | | |
| — | | |
| 66,854 | | |
| 66,854 | |
| Net
Loss | |
| (826,448 | ) | |
$ | (3,520,240 | ) | |
$ | (3,043,667 | ) | |
$ | (7,390,355 | ) | |
$ | (740,626 | ) | |
$ | (1,081,184 | ) | |
$ | (1,547,000 | ) | |
$ | (3,368,810 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Adjusted
EBITA | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Operating
Loss | |
$ | (826,448 | | |
$ | (3,520,240 | ) | |
$ | (2,950,046 | ) | |
$ | (7,296,734 | ) | |
$ | (740,626 | ) | |
$ | (1,081,184 | ) | |
$ | (1,491,082 | ) | |
$ | (3,312,892 | ) |
| Depreciation
and Amortization | |
| 31,205 | | |
| 915,539 | | |
| — | | |
| 946,744 | | |
| 17,638 | | |
| — | | |
| — | | |
| 17,638 | |
| Stock
based compensation | |
| 290,706 | | |
| 229,898 | | |
| 1,029,183 | | |
| 1,549,787 | | |
| 79,845 | | |
| 97,867 | | |
| 509,793 | | |
| 687,505 | |
| Adjusted
EBITDA | |
$ | (504,537 | | |
$ | (2,374,803 | ) | |
$ | (1,920,863 | ) | |
$ | (4,800,203 | ) | |
$ | (643,143 | ) | |
$ | (983,317 | ) | |
$ | (981,289 | ) | |
$ | (2,607,749 | ) |
The
Company utilizes Adjusted EBITDA, a non-GAAP financial measure, to assist in anayzing our segment operating performance by removing the
impact of certain key items that management believes do not directly reflect our underlying operations. In addition, we consider certain
non-GAAP (or "adjusted") measures to be useful to management and investors evaluating our operating performance for the periods
presented, and provide a tool for evaluating our ongoing operations, liquidity, and management of assets. This information can assist
investors in assessing our financial performance and measures our ability to generate capital. These adjusted metrics are consistent
with how management views our business and are used to make financial, operating and planning decisions. These metrics, however, are
not measures of financial performance under GAAP and should not be considered a substitute for revenues, operating income, net income
(loss), earnings (loss) per share (basic and diluted) or net cash from operating activities as determined in accordance with GAAP.
Adjusted EBITDA is defined as Operating Profit (Loss), excluding Depreciation and Amortization, and excluding Stock Based Compensation.
Adjusted EBITDA was a loss of ($4,800,203) for the year ended December 31, 2021, which was an increase of ($2,192,454) as compared to
the year ended December 31, 2020. By segment, Hospitality improved $138,606, while Disinfection decreased ($1,391,486) and Corporate
decreased ($939,574).
Segments
The
Company has three reportable segments: the design, manufacture, assembly and distribution of disinfecting systems for use in healthcare,
hospitality, and commercial municipal and residential markets (Disinfection segment); the manufacture of fine mirrors specifically for
the Hospitality industry (hospitality segment); and the Corporate Segment, which includes expenses primarily related to corporate governance,
such as board fees, legal expenses, audit fees, executive management, and listing costs. See NOTE 14 – Segment Reporting.
Net
Sales
Net
sales of $11,667,579 represented an increase of $5,934,845, or 103.5% for the year ended December 31, 2021 as compared to net sales of
$5,732,734 for the year ended December 31, 2020. This increase was primarily attributable to the addition of the Disinfection segment
in 2021 as a result of the strategic acquisitions of Akida, KES, and Scientific Air. The 2021 net sales for Disinfection of $5,723,915
includes close to 11 months of Akida-related sales (acquisition closed February 8, 2021), one quarter of KES-related sales (acquisition
closed September 28, 2021), and just over 2 months of Scientific Air-related sales (acquisition closed October 13, 2021). The Hospitality
segment began to rebound from the slowdown caused by the pandemic and finished the year ended December 31, 2021 with net sales of $5,943,664,
which represented an increase of $210,930, or 3.7% as compared to the year ended December 31, 2020.
Gross
Profit
Gross
profit increased $3,089,050, or 306%, for the year ended December 31, 2021 as compared to the year ended December 31, 2020, driven by
both volume growth and the higher margin contribution from the Disinfection segment. The Disinfection segment’s gross profit for
the year ended December 31, 2021 was $2,643,374, or 46.2% as a percentage of net sales. The Hospitality segment’s gross margin
for the year ended December 31, 2021 was $1,455.012, or 24.4% as a percentage of net sales, as compared to $1,009,336, or 17.6% as a
percentage of net sales for the year ended December 31, 2020. The Hospitality segment’s gross profit was impacted last year by
the sales slowdown caused by the pandemic, as well as higher overhead cost absorption as the company kept many of their direct labor
employees in compliance with the payroll protection program forgiveness requirements. The company is focused on realizing cost synergies
from consolidation and streamlining of manufacturing operations to help offset increases in material and logistics costs.
Operating
Expenses
Selling,
General, and Administrative – S,G&A costs, excluding stock compensation expense of $1,549,787 for the year ended December
31, 2021 and $687,505 for the year ended December 31, 2020, were $9,791,925 for the year ended December 31, 2021, which represented an
increase of $6,467,874 as compared to the year ended December 31, 2020. This increase was driven primarily by the expansion of the Disinfection
segment. The infrastructure to support this segment was initially implemented in the fourth quarter of 2020, and additional investments
were made during 2021 to support the three strategic acquisitions of Akida, KES, and Scientific Air. Payroll costs increased $2.4 million
year over year as headcount increased from 33 at December 31, 2020 to 61 at December 31, 2021. Consulting costs increased $0.7 million
and legal expense increased $0.4 million, mainly due to acquisition-related expenses. Amortization expense, mostly related to the intangible
assets associated with the acquisitions, increased $1.0 million, and depreciation expense increased $0.1 million. Additional increases
were due to advertising $0.3 million, product certification and testing $0.3m, insurance $0.2million, and rent $0.1 million. We anticipate
efficiency gains in the coming year as we fully integrate all 3 acquisitions and leverage synergies where practical. The Corporate segment
includes expenses primarily related to corporate governace, such as board fees, legal expenses, audit fees, executive management, and
listing costs, allowing for a better reflection of operational measurement of each of the two operating segments.
Stock
based compensation – Stock based compensation was $1,549,787 for the year ended December 31, 2021, which represented and increase
of $862,282 as compared to the year ended December 31, 2020, primarily due to the full year impact of awards that were granted in the
foruth quarter of 2020, and additional awards made during 2021, under the Applied UV, Inc. 2020 Omnibus Incentive Plan (the “Plan”)
which authorized 600,000 shares of common stock to be available for issuance under the terms of the Plan. The Plan permits the granting
of Nonqualified Stock Options, Incentive Stock Options, Stock Appreciation Rights, Restricted Stock, Restricted Stock Units, Performance
Shares, Performance Units and Other Awards. The expense increase is also due to a new employment contract for Max Munn, now Interim CEO,
that became effective in Q1 of 2021, whose options were not part of the Plan. Mr.Munn received options to purchase 309,835 shares of
common stock at an exercise price of $7.80 per share. These options vest at the end of each month for 36 months beginning April 1, 2021,
which resulted in an expense of $391,863 for the year ended December 31, 2021.
Other
The
Company recorded income on the change in fair value of warrant liability in the amount of $66,862 for the year ended December 31, 2021.
Warrants that were issued in connection with the November 2020 public offering contained a cash settlement feature which resulted in
a warrant liability of $68,263 as of December 31, 2021. The fair market value of the warrant liability on the date of grant was $135,125
and was recorded as a reduction of Additional Paid in Capital.
The
company recorded a loss on contingent consideration of $574,000 for the year ended December 31, 2021 as a result of the decrease in our
stock price from the date of acquisition of SciAir and the reporting date.
Other
Income includes the payroll protection program loan forgiveness of $296,827.
Net
Benefit from Income Taxes of $91,819 is due to a loss carryback that was generated in the tax year ended December 31, 2020.
Net
Loss
We
recorded a net loss of $7,390,355 for the year ended December 31, 2021, compared to a net loss of $3,368,810 for the year ended December
31, 2020. The increase of $3,807,244 in the net loss was mainly due to the investments made to grow the Disinfection segment and loss
on contingent consideration, offset by the increased revenue and gross margin from that segment, as discussed above, plus an increase
in corporate governance costs.
Liquidity
and Capital Resources
Year
Ended December 31, 2021 Compared to the Year Ended December 31, 2020
| Net
cash provided by (used in) operating activities | |
$ | (6,997,970 | ) | |
$ | (657,231 | ) |
| Net
cash (used in) investing activities | |
| (14,589,362 | ) | |
| (218,633 | ) |
| Net
cash provided by financing activities | |
| 18,597,558 | | |
| 11,603,858 | |
| Net
increase (decrease) in cash and cash equivalents | |
| (2,989,774 | ) | |
| 10,727,994 | |
| Cash
and cash equivalents at beginning of year | |
| 11,757,930 | | |
| 1,029,936 | |
| Cash,
restricted cash, and cash equivalents at end of year | |
| 8,768,156 | | |
| 11,757,930 | |
In
the year ended December 31, 2021, net cash used in operating activities was ($6,997,970), as compared to ($657,231) in the year ended
December 31, 2020. This increase in net cash used was due mainly to the net loss of ($7,390,355) for the year ended December 31, 2021,
offset primarily by non-cash charges of $1,549,787 for stock based compensation, $946,744 for depreciation and amortization, $574,000
for loss on contingent consideration, increases in inventory and vendor deposits of $306,126 and $951,242 respectively, and decreases
in accounts payable and accrued expenses and deferred revenue of $361,569 and $544,563, respectively.
In
the year ended December 31, 2021, net cash used in investing activities increased ($14,370,729) to ($14,589,362) as compared to ($218,633)
in the year ended December 31, 2020, primarily due to net cash paid for acquisitions of ($14,560,193): Akida ($760,293), KES ($4,299,900),
and Scientific Air ($9,500,000).
In
the year ended December 31, 2021, cash provided by financing activities was $18,597,558, as compared to cash used in financing activities
of $11,603,858 in the year ended December 31, 2010. The major financing activities that provided cash for the year ended December 31,
2021 was cash received from the July preferred stock offering of $12,272,440 and cash received from the December common stock offering
of $6,999,928.
The Company
believes our sources of liquidity and capital will be sufficient to finance our continued operations and growth strategy.
Item
7A. Quantitative and Qualitative Disclosures About Market Risk.
The
Company is a smaller reporting company as defined by Rule 12b-2 of the Exchange Act and is not required to provide the information required
under this item.
Item
8. Financial Statements and Supplementary Data.
Applied
UV, Inc. and Subsidiaries
Consolidated
Financial Statements
December
31, 2021 and 2020
Applied
UV, Inc. and Subsidiaries
Index
to the Consolidated Financial Statements
December
31, 2021 and 2020
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To
the Board of Directors and Stockholders of Applied UV, Inc.
Opinion
on the Consolidated Financial Statements
We
have audited the accompanying consolidated balance sheet of Applied UV, Inc. and Subsidiaries (the “Company”) as of December
31, 2021, the related consolidated statements of operations, stockholders’ equity, and cash flows for the year ended December 31,
2021, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated
financial statements present fairly, in all material respects, the consolidated financial position of the Company as of December 31,
2021, and the consolidated results of its operations and its cash flows for the year ended December 31, 2021, in conformity with accounting
principles generally accepted in the United States of America.
Basis
for Opinion
These
consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion
on the Company’s consolidated financial statements based on our audit. We are a public accounting firm registered with the Public
Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company
in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission
and the PCAOB.
We
conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud.
The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part
of our audit, we are required to obtain an understanding of internal control over financial reporting, but not for the purpose of expressing
an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our
audit included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due
to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence
regarding the amounts and disclosures in the consolidated financial statements. Our audit also included evaluating the accounting principles
used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements.
We believe that our audit provides a reasonable basis for our opinion.
/s/
Mazars USA LLP
We
have served as the Company’s auditor since 2021.
Fort
Washington, PA
April
7, 2022
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the
Board of Directors and Stockholders of
Applied
UV, Inc. and Subsidiaries
Opinion
on the Financial Statements
We
have audited the accompanying consolidated balance sheet of Applied UV, Inc. and Subsidiaries (the "Company") as of December
31, 2020, and the related consolidated statement of operations, stockholders' equity (deficit), and cash flows for the year ended December
31, 2020, and the related notes (collectively referred to as the "financial statements"). In our opinion, the financial statements
present fairly, in all material respects, the financial position of the Company as of December 31, 2020, and the results of its operations
and its cash flows for the year ended December 31, 2020, in conformity with accounting principles generally accepted in the United States
of America.
Basis
for Opinion
These
financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company's
financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board
(United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities
laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We
conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain
reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company
is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits,
we are required to obtain an understanding of internal control over financial reporting, but not for the purpose of expressing an opinion
on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our
audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error
or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding
the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant
estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits
provide a reasonable basis for our opinion.
Critical
Audit Matters
Critical
audit matters are matters arising from the current period audit of the financial statements that were communicated or required to be
communicated to the audit committee and that (1) relate to accounts or disclosures that are material to the financial statements and
(2) involved our especially challenging, subjective, or complex judgments. We determined that there were no critical audit matters.
We have
served as the Company's auditor from 2020 to 2021.
/s/
Adeptus Partners LLC
Adeptus partners LLC
Ocean,
New Jersey
March 30,
2021
Applied
UV, Inc. and Subsidiaries
Consolidated
Balance Sheets
As
of December 31, 2021 and 2020
| | |
| | | |
| | |
| | |
2021 | |
2020 |
| Assets | |
| | | |
| | |
| Current
Assets | |
| | | |
| | |
| Cash
and cash equivalents | |
$ | 7,922,906 | | |
$ | 11,757,930 | |
| Restricted
cash | |
| 845,250 | | |
| — | |
| Accounts
receivable, net of allowance for doubtful accounts | |
| 986,253 | | |
| 232,986 | |
| Inventory | |
| 1,646,238 | | |
| 156,290 | |
| Vendor
deposits | |
| 992,042 | | |
| 40,800 | |
| Prepaid
expense and other current assets | |
| 419,710 | | |
| 158,498 | |
| Total
Current Assets | |
| 12,812,399 | | |
| 12,346,504 | |
| | |
| | | |
| | |
| Property
and equipment, net of accumulated depreciation | |
| 196,611 | | |
| 112,804 | |
| Goodwill | |
| 4,809,811 | | |
| — | |
| Other
intangible assets, net of accumulated amortization | |
| 18,976,556 | | |
| 178,088 | |
| Right
of use asset | |
| 1,730,615 | | |
| 481,425 | |
| Total
Assets | |
$ | 38,525,992 | | |
$ | 13,118,821 | |
| | |
| | | |
| | |
| Liabilities
and Stockholders' Equity | |
| | | |
| | |
| | |
| | | |
| | |
| Current
Liabilities | |
| | | |
| | |
| Accounts
payable and accrued expenses | |
$ | 1,642,108 | | |
$ | 1,398,073 | |
| Contingent
Consideration | |
| 1,460,000 | | |
| — | |
| Deferred
revenue | |
| 788,776 | | |
| 841,636 | |
| Income
tax payable | |
| — | | |
| 173,716 | |
| Warrant
liability | |
| 68,263 | | |
| — | |
| Financing
lease obligations | |
| 7,671 | | |
| 6,648 | |
| Lease
liability | |
| 389,486 | | |
| 139,908 | |
| Payroll
protection program loan | |
| — | | |
| 69,927 | |
| Loan
payable | |
| 97,500 | | |
| 67,500 | |
| Total
Current Liabilities | |
| 4,453,804 | | |
| 2,697,408 | |
| Long-term
Liabilities | |
| | | |
| | |
| Financing
lease obligations - less current portion | |
| — | | |
| 8,240 | |
| Note
payable-less current portion | |
| 60,000 | | |
| 90,000 | |
| Lease
liability-less current portion | |
| 1,346,428 | | |
| 341,517 | |
| Payroll
protection program loan-less current portion | |
| — | | |
| 226,900 | |
| Total
Long-Term Liabilities | |
| 1,406,428 | | |
| 666,657 | |
| | |
| | | |
| | |
| Total
Liabilities | |
| 5,860,232 | | |
| 3,364,065 | |
| | |
| | | |
| | |
| Stockholders'
Equity | |
| | | |
| | |
| Preferred
stock, Series A Cumulative Perpetual, $0.0001
par value, 19,990,000
shares authorized, 552,000
issued and outstanding as of December 31,
2021, and 0 shares
issued and outstanding as of December 31, 2020 | |
| 55 | | |
| — | |
| Preferred
stock, Series X, $0.0001
par value, 10,000
shares authorized, 2,000
shares issued and outstanding as of both
December 31, 2021 and 2020 | |
| 1 | | |
| 1 | |
| Common
stock $.0001 par
value, 150,000,000 shares
authorized; 12,775,674 issued
and outstanding as of December 31, 2021, and 7,945,034
shares issued and outstanding as of December 31, 2020 | |
| 1,278 | | |
| 795 | |
| Additional
paid-in capital | |
| 42,877,622 | | |
| 11,973,051 | |
| Accumulated
deficit | |
| (10,213,196 | ) | |
| (2,219,091 | ) |
| Total
Stockholders' Equity | |
| 32,665,760 | | |
| 9,754,756 | |
| | |
| | | |
| | |
| Total
Liabilities and Stockholders' Equity | |
$ | 38,525,992 | | |
$ | 13,118,821 | |
The
accompanying notes are an integral part of these consolidated financial statements.
Applied
UV, Inc. and Subsidiaries
Consolidated
Statements of Operations
For
the Years Ended December 31, 2021 and 2020
| | |
| | | |
| | |
| | |
2021 | |
2020 |
| Net
Sales | |
$ | 11,667,579 | | |
$ | 5,732,734 | |
| | |
| | | |
| | |
| Cost
of Goods Sold | |
| 7,569,193 | | |
| 4,723,398 | |
| | |
| | | |
| | |
| Gross
Profit | |
| 4,098,386 | | |
| 1,009,336 | |
| | |
| | | |
| | |
| Operating
Expenses | |
| | | |
| | |
| Research
and development | |
| 53,408 | | |
| 310,672 | |
| Selling.
General and Administrative Expenses | |
| 11,341,712 | | |
| 4,011,556 | |
| Total
Operating Expenses | |
| 11,395,120 | | |
| 4,322,228 | |
| | |
| | | |
| | |
| Operating
Loss | |
| (7,296,734 | ) | |
| (3,312,892 | ) |
| | |
| | | |
| | |
| Other
Income (Expense) | |
| | | |
| | |
Change
in Fair Market Value
of Warrant Liability | |
| 66,862 | | |
| — | |
| Forgiveness
of paycheck protection program loan | |
| 296,827 | | |
| — | |
| Loss
on contingent consideration | |
| (574,000 | ) | |
| — | |
| Other
Income | |
| 24,871 | | |
| 10,936 | |
| Total
Other Income (Expense) | |
| (185,440 | ) | |
| 10,936 | |
| | |
| | | |
| | |
| Loss
Before Provision (Benefit) for Income Taxes | |
| (7,482,174 | ) | |
| (3,301,956 | ) |
| | |
| | | |
| | |
| Provision
(Benefit) from Income Taxes | |
| (91,819 | ) | |
| 66,854 | |
| | |
| | | |
| | |
| Net
Loss | |
$ | (7,390,355 | ) | |
$ | (3,368,810 | ) |
| | |
| | | |
| | |
| Net
Loss attributable to common stockholders: | |
| | | |
| | |
| Dividends
to preferred shareholders | |
| (603,750 | | |
| — | |
| Net
Loss attributable to common stockholders | |
| (7,994,105 | ) | |
| (3,368,810 | ) |
| Basic
and Diluted Loss Per Common Share | |
$ | (0.86 | ) | |
$ | (0.59 | ) |
| | |
| | | |
| | |
Weighted
Average Shares Outstanding -
basic and diluted | |
| 9,273,257 | | |
| 5,733,591 | |
The
accompanying notes are an integral part of these consolidated financial statements.
Applied
UV, Inc. and Subsidiaries
Consolidated
Statements of Changes in Stockholders' Equity
For
the Years Ended December 31, 2021 and 2020
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| | |
Preferred
Stock | |
Preferred
Stock | |
| |
| |
| |
| |
|
| | |
Series
A Cumalative | |
Series
X Voting | |
Common
Stock | |
| |
| |
|
| | |
| Shares | | |
| Amount | | |
| Shares | | |
| Amount | | |
| Shares | | |
| Amount | | |
| Additional
Paid-In Capital | | |
| Retained
Earnings (Accumulated
Deficit) | | |
| Total
Stockholders Equity | |
| Balance,
January 1, 2020 | |
| — | | |
$ | — | | |
| 2,000 | | |
$ | 1 | | |
| 5,001,252 | | |
| 500 | | |
| — | | |
$ | 1,149,719 | | |
$ | 1,150,220 | |
| Shares
issued to legal counsel | |
| — | | |
| — | | |
| — | | |
| — | | |
| 161,794 | | |
| 16 | | |
| 808,970 | | |
| — | | |
| 808,986 | |
| Stock-based
compensation | |
| — | | |
| — | | |
| — | | |
| — | | |
| 230,083 | | |
| 23 | | |
| 666,060 | | |
| — | | |
| 666,083 | |
| Common
stock issued in public offering, net of costs | |
| — | | |
| — | | |
| — | | |
| — | | |
| 2,551,905 | | |
| 256 | | |
| 10,498,021 | | |
| — | | |
| 10,498,277 | |
| Net
loss | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| (3,368,810 | ) | |
| (3,368,810 | ) |
| Balance,
December 31, 2020 | |
| — | | |
| — | | |
| 2,000 | | |
| 1 | | |
| 7,945,034 | | |
| 795 | | |
| 11,973,051 | | |
| (2,219,091 | ) | |
| 9,754,756 | |
| Shares
granted to settle previously recorded liability | |
| — | | |
| — | | |
| — | | |
| — | | |
| 3,000 | | |
| — | | |
| 21,420 | | |
| — | | |
| 21,420 | |
| Warrant
liability recognized in connection with initial issuance of November offering (See Note 8) | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| (135,125 | ) | |
| — | | |
| (135,125 | ) |
| Exercise
of warrants | |
| — | | |
| — | | |
| — | | |
| — | | |
| 17,852 | | |
| 2 | | |
| 1,155 | | |
| — | | |
| 1,157 | |
| Common
stock issued for acquisitions | |
| — | | |
| — | | |
| — | | |
| — | | |
| 2,075,000 | | |
| 208 | | |
| 10,195,293 | | |
| — | | |
| 10,195,501 | |
| Stock-based
compensation | |
| — | | |
| — | | |
| — | | |
| — | | |
| 74,500 | | |
| 7 | | |
| 1,549,781 | | |
| — | | |
| 1,549,788 | |
| Common
stock issued in public offering, net of costs | |
| — | | |
| — | | |
| — | | |
| — | | |
| 2,666,667 | | |
| 267 | | |
| 6,999,661 | | |
| — | | |
| 6,999,928 | |
| Preferred
stock issued in public offering, net of costs | |
| 552,000 | | |
| 55 | | |
| — | | |
| — | | |
| — | | |
| — | | |
| 12,272,385 | | |
| — | | |
| 12,272,440 | |
| Dividends
paid to preferred shareholders | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| (603,750 | ) | |
| (603,750 | ) |
| Cancellation
of Restricted Stock | |
| — | | |
| — | | |
| — | | |
| — | | |
| (6,379 | ) | |
| (1 | ) | |
| 1 | | |
| — | | |
| — | |
| Net
loss | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| (7,390,355 | ) | |
| (7,390,355 | ) |
| Balance,
December 31, 2021 | |
| 552,000 | | |
$ | 55 | | |
| 2,000 | | |
$ | 1 | | |
| 12,775,674 | | |
$ | 1,278 | | |
$ | 42,877,622 | | |
$ | (10,213,196 | ) | |
$ | 32,665,760 | |
The
accompanying notes are an integral part of these consolidated financial statements.
Applied
UV, Inc. and Subsidiaries
Consolidated
Statements of Cash Flows
For
the Years Ended December 31, 2021 and 2020
| | |
| | | |
| | |
| | |
2021 | |
2020 |
| Cash
flows from Operating Activities | |
| | | |
| | |
| Net
Loss | |
$ | (7,390,355 | ) | |
$ | (3,368,810 | ) |
| Adjustments
to Reconcile Net Loss to Net Cash Used in Operating Activities | |
| | | |
| | |
| Stock
based compensation | |
| 1,549,788 | | |
| 687,505 | |
| Bad
debt expense (recovery) | |
| (71,003 | ) | |
| 50,000 | |
| Change
in fair market value of warrant liability | |
| (66,862 | ) | |
| — | |
| Forgiveness
of paycheck protection program loan | |
| (296,827 | ) | |
| — | |
| Loss
on contingent consideration | |
| 574,000 | | |
| — | |
| Gain
on settlement of loan payable | |
| (20,000 | ) | |
| — | |
| Amortization
of right-of-use asset | |
| 635,540 | | |
| — | |
| Depreciation
and amortization | |
| 946,744 | | |
| 17,638 | |
| Changes
in operating assets and liabilities, net of effects of acquisitions | |
| | | |
| | |
| Accounts
receivable | |
| 73,189 | | |
| 1,944,805 | |
| Inventory | |
| (306,126 | ) | |
| (56,747 | ) |
| Vendor
deposits | |
| (951,242 | ) | |
| 63,717 | |
| Prepaid
expenses | |
| 35,273 | | |
| (134,596 | ) |
| Income
taxes payable | |
| (173,716 | ) | |
| 66,855 | |
| Accounts
payable and accrued expenses | |
| (361,569 | ) | |
| 710,636 | |
| Deferred
revenue | |
| (544,563 | ) | |
| (638,234 | ) |
| Operating
lease payments | |
| (630,241 | ) | |
| — | |
| Total
Adjustments | |
| 392,385 | | |
| 2,711,579 | |
| Net
Cash Used in Operating Activities | |
| (6,997,970 | ) | |
| (657,231 | ) |
| | |
| | | |
| | |
| Cash
Flows From Investing Activities | |
| | | |
| | |
| Cash
paid for patent costs | |
| (14,434 | ) | |
| (122,562 | ) |
| Purchase
of machinery and equipment | |
| (14,735 | ) | |
| (96,071 | ) |
| Acquisitions,
net of cash acquired (Note 2) | |
| (14,560,193 | ) | |
| — | |
| Net
Cash Used in Investing Activities | |
| (14,589,362 | ) | |
| (218,633 | ) |
| | |
| | | |
| | |
| Cash
Flows From Financing Activities | |
| | | |
| | |
| Payments
on financing leases | |
| (7,217 | ) | |
| (6,704 | ) |
| Proceeds
from warrant exercise | |
| 1,157 | | |
| — | |
| Loan
to officer | |
| — | | |
| 4,225 | |
| Dividends
to preferred shareholders | |
| (603,750 | ) | |
| — | |
| Payments
on loans payable | |
| (65,000 | ) | |
| — | |
| Proceeds
from equity raises, net (Note 9) | |
| 19,272,368 | | |
| 11,309,510 | |
| Proceeds
from payroll protection program | |
| — | | |
| 296,827 | |
| Net
Cash Provided by Financing Activities | |
| 18,597,558 | | |
| 11,603,858 | |
| | |
| | | |
| | |
| Net
Increase (Decrease) in Cash and equivalents | |
| (2,989,774 | ) | |
| 10,727,994 | |
| Cash,
restricted cash, and cash equivalents at January 1, | |
| 11,757,930 | | |
| 1,029,936 | |
| Cash,
restricted cash, and cash equivalents at December 31, | |
$ | 8,768,156 | | |
$ | 11,757,930 | |
| | |
| | | |
| | |
| Supplemental
Disclosures of Cash Flow Information: | |
| | | |
| | |
| Cash
paid during the year for: | |
| | | |
| | |
| Interest | |
$ | 1,022 | | |
$ | 2,133 | |
| Income
taxes | |
$ | 16,246 | | |
$ | — | |
| Disclosures
of Non-Cash Investing and Financing Activities: | |
| | | |
| | |
| Initial
recognition of warrant liability | |
$ | 135,125 | | |
$ | — | |
| Common
stock issued to settle liability | |
$ | 21,420 | | |
$ | 808,986 | |
| Common
stock issued in connection with acquisitions | |
$ | 10,195,501 | | |
$ | — | |
| Initial
recognition of contingenct consideration liability | |
$ | 886,000 | | |
$ | — | |
| Recognition
of right of use asset - operating lease | |
$ | 1,884,730 | | |
$ | — | |
The
accompanying notes are an integral part of these consolidated financial statements.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
1 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Nature
of Business
Applied
UV, Inc. (the "Parent") was formed and incorporated in the State of Delaware for the intended purpose of holding the equity of
SteriLumen, Inc. (“SteriLumen”), MunnWorks, LLC (“MunnWorks” and together with SteriLumen, the “Subsidiaries”)
and other companies acquired or created by the Parent in the future. The Parent acquired the Subsidiaries pursuant to three share exchanges
whereby the equity holders of the Subsidiaries exchanged all of their equity interests in the Subsidiaries for shares of voting stock
of the Parent. As a result of the share exchanges, each Subsidiary became a wholly-owned subsidiary of the Parent. The Parent and each
Subsidiary are collectively referred to herein as (the "Company").
SteriLumen
is engaged in the design, manufacture, assembly and distribution of (i) automated disinfecting mirror systems for use in hospitals and
other healthcare facilities and (ii) air purification systems through its purchase of substantially all of the assets and certain liabilities
of Akida Holdings, LLC, KES Science & Technology, and Scientific Air Management LLC, as described below. MunnWorks, LLC is engaged
in the manufacture of fine mirrors specifically for the hospitality industry.
In
February of 2021, the Company acquired all the assets and assumed certain liabilities of Akida Holdings, LLC (“Akida”). At
the time of this acquisition, Akida owned the Airocide™ system of air purification technologies, originally developed for NASA,
with assistance from the University of Wisconsin at Madison, that uses a combination of UVC and a proprietary, titanium dioxide based
photocatalyst that may help to accelerate the reopening of the global economy with applications in the hospitality, hotel, healthcare,
nursing homes, grocer, wine, commercial buildings and retail sectors. The Airocide™ system has been used by brands and organizations
such as NASA, Whole Foods, Dole, Chiquita, Opus One, Sub-Zero Refrigerators and Robert Mondavi Wines. Akida contracted KES Science &
Technology, Inc. (“KES”) to manufacture, warehouse and distribute the Airocide™ system and Akida’s contractual
relationship with KES was assigned to and assumed by the Company as part of the acquisition.
On
September 28, 2021, the Company acquired all the assets and assumed certain liabilities of KES. At the time of the acquisition, KES was
principally engaged in the manufacturing and distribution of the Airocide™ system of air purification technologies and misting
systems. KES also had the exclusive right to the sale and distribution of the Airocide™ system in certain markets. This acquisition
consolidates all of manufacturing, sale and distribution of the Airocide™ system under the SteriLumen brand and expands the Company’s
market presence in food distribution, post-harvest produce, wineries, and retail sectors. The Company sells its products throughout the
United States, Canada, and Europe
On
October 13, 2021, the Company acquired all the assets and assumed certain liabilities of Scientific Air Management LLC, ("SciAir").
SciAir is a provider of whole-room, aerosol chamber and laboratory certified air disinfection machines. SciAir is a provider of whole-room,
aerosol chamber and laboratory certified air disinfection machines that use a combination of UVC and a proprietary, patented system to
eliminate airborne bacteria, mold, fungi, viruses, volatile organic compounds, and many odors without producing any harmful by-products.
The units are well suited for larger spaces within a facility and are mobile with industrial grade casters allowing for movement throughout
a facility to address increased bio burdern from larger meetings or increased human traffic.
Principles
of Consolidation
The
consolidated financial statements include the accounts of Applied UV, Inc., MunnWorks, LLC and SteriLumen, Inc. All significant intercompany
transactions and balances are eliminated in consolidation.
Use
of Estimates
The
preparation of consolidated financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that
affect the reported amounts of assets and liabilities, and disclosure of contingent assets and liabilities, as of the date of the consolidated
financial statements, and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from
those estimates. Significant estimates include the valuation and accounting for equity awards related to warrants and stock-based compensation,
determination of fair value for derivative instruments, the accounting for business combinations and allocating purchase price and estimating
the useful life of intangible assets.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
1 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Concentration
of Credit and Business Risk
At
times throughout the year, the Company maintains cash balances at various institutions, which may exceed the Federal Deposit Insurance
Corporation limit. As of December 31, 2021 and 2020, amounts of $8,518,156
and $11,507,930,
respectively were in excess of FDIC insured limits.
The
Company provides credit in the normal course of business. The Company performs ongoing credit evaluations of its customers and maintains
allowances for doubtful accounts based on factors surrounding the credit risk of specific customers, historical trends and other information.
For
the year ended December 31, 2021, the Company had no major suppliers and for the year ended December 31, 2020, the Company had two major
suppliers that accounted for approximately 25.4%
of supplies and materials used by the Company. The amounts have been recorded as costs of sales in the consolidated statements of operations.
Cash,
Restricted Cash and Cash Equivalents
Cash
and equivalents include highly liquid investments that have original maturities less than 90 days at the time of their purchase. The
company holds funds in money market accounts. These investments are carried at cost, which approximates market value because of their
short maturities. As of December 31, 2021 and 2020, the Company had $1,076,664
and $0,
respectively, in cash equivalents. The Company also maintains a restricted cash balance to satisfy its preferred shareholder redemption
requirements (Refer to Note 9).
Accounts
receivable
An
allowance for uncollectible accounts receivable is recorded when management believes the collectability of the accounts receivable is
doubtful. Subsequent recoveries, if any, are credited to the allowance. The allowance is determined based on management’s review
of the debtor’s ability to repay and repayment history, aging history, and estimated value of collateral, if any. The Company had
an allowance for doubtful accounts approximating $9,000
and $100,000
as of December 31, 2021 and 2020, respectively.
Inventory
Inventories,
which consists of raw materials and finished goods, is valued at the lower of cost or net realizable value, using the first-in, first-out
(“FIFO”) valuation method. Inventory costs are comprised primarily of product, freight and duty. The Company writes down
inventory for estimated obsolescence equal to the difference between the cost of inventory and the estimated market value based upon
assumptions about future demand and market conditions. The company did not
have any reserves for inventory as of December 31, 2021 and 2020.
Property
and Equipment
Property
and equipment are recorded at cost. Depreciation of furniture and fixtures is provided using the straight-line method, generally over
the terms of the lease. Repairs and maintenance expenditures, which do not extend the useful lives of the related assets, are expensed
as incurred. Depreciation of machinery and equipment is based on the estimated useful lives of the assets.
| Schedule
of estimated useful lives |
|
| |
Years |
| Machinery
and equipment |
5-7 |
| Leasehold
improvements |
Lesser
of term of lease or useful life |
| Furniture
and fixtures |
7 |
Business
Acquisition Accounting
The
Company applies the acquisition method of accounting for those that meet the criteria of a business combination. The Company allocates
the purchase price of its business acquisitions based on the fair value of identifiable tangible and intangible assets. The difference
between the total cost of the acquisition and the sum of the fair values of acquired tangible and identifiable intangible assets less
liabilities is recorded as goodwill. Transaction costs are expensed as incurred in general and administrative expenses.
Goodwill
and Intangible Assets
The
Company has recorded intangible assets, including goodwill, in connection with business combinations. Estimated useful lives of amortizable
intangible assets are determined by management based on an assessment of the period over which the asset is expected to contribute to
future cash flows.
In
accordance with U.S. GAAP for goodwill and other indefinite-lived intangibles, the Company tests these assets for impairment annually
and whenever events or circumstances make it more likely than not that impairment may have occurred. For the purposes of that assessment,
the Company has determined to assign assets acquired in business combinations to a single reporting unit including all goodwill and indefinite-lived
intangible assets acquired in business combinations.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
1 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Derivative
Instruments
The
Company evaluates its warrants to determine if those contracts or embedded components of those contracts qualify as derivatives. The
result of this accounting treatment is that the fair value of the embedded derivative is marked-to-market each balance sheet date and
recorded as a liability. In the event that the fair value is recorded as a liability, the change in fair value is recorded in the statements
of operations as other income or expense.
The
classification of derivative instruments, including whether such instruments should be recorded as liabilities or as equity, is re-assessed
at the end of each reporting period. The Company has concluded that there are no such reclassifications required to be made as of and
for the periods ended December 31, 2021 and 2020.
The
Company utilizes the Black-Scholes valuation model to value the derivative warrants as stipulated in the agreement for the warrant holders
to receive cash based on that value.
Fair
Value of Financial Instruments
The
carrying amounts reported in the consolidated balance sheets for loans payable approximate fair value because of the immediate or short-term
maturity of the financial instruments. The Company's financial assets and liabilities are measured using inputs from the three levels
of the fair value hierarchy.
Loss
Per Share
Basic
loss per share is computed by dividing net loss attributable to common shareholders (the numerator) by the weighted-average number of
common shares outstanding (the denominator) for the period. In periods of losses, diluted loss per share is computed on the same basis
as basic loss per share as the inclusion of any other potential shares outstanding would be anti-dilutive.
The
following table sets forth the number of potential shares of common stock that have been excluded from diluted net loss per share because
their effect was anti-dilutive:
| Schedule
of Anti-dilutive Securities Excluded from Computation of Earnings Per Share | |
| | | |
| | |
| Schedule
of Anti-dilutive Securities Excluded from Computation of Earnings Per Share: |
| |
| | |
As
of December 31, |
| | |
2021 | |
2020 |
| Common
stock options | |
| 644,314 | | |
| 136,750 | |
| Common
stock warrants | |
| 192,419 | | |
| 235,095 | |
| Total | |
| 836,733 | | |
| 371,845 | |
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
1 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Stock-
Based Compensation
The
Company accounts for its stock-based compensation awards in accordance with Financial Accounting Standards Board ("FASB") Accounting
Standards Codification Topic 718 ("ASC"), Compensation-Stock Compensation ("ASC 718"). ASC 718 requires all stock-based
payments to employees, including grants of employee stock options and restricted stock and modifications to existing stock options, to
be recognized in the statements of operations based on their fair values over the requisite service period.
Research
and Development
The
Company accounts for research and development costs in accordance with Accounting Standards Codification subtopic 730-10, Research and
Development (“ASC 730-10”). Under ASC 730-10, all research and development costs must be charged to expense as incurred.
Accordingly, research and development costs are expensed as incurred.
Income
Taxes
The
Company files income tax returns using the cash basis of accounting. Income taxes are accounted for under the asset and liability method.
Current income taxes are based on the year's income taxable for federal and state tax reporting purposes. Deferred income tax assets
and liabilities are computed annually for differences between the financial statement and tax bases of assets and liabilities that will
result in taxable or deductible amounts in the future based on enacted tax laws and rates applicable to the periods in which the differences
are expected to affect taxable income. The carrying amount of deferred tax assets is reviewed at each reporting date and reduced to the
extend that it is no longer probable that sufficient taxable income will be available to allow all or part of the asset to be recovered.
Per
FASB ASC 740-10, disclosure is not required of an uncertain tax position unless it is considered probable that a claim will be asserted
and there is a more-likely-than-not possibility that the outcome will be unfavorable. Using this guidance, as of December 31, 2021 and
2020, the Company has no uncertain tax positions that qualify for either recognition or disclosure in the financial statements. The Company's
2020, 2019, and 2018 Federal and State tax returns remain subject to examination by their respective taxing authorities. Neither of the
Company's Federal or State tax returns are currently under examination.
Revenue
Recognition
The
Company recognizes revenue when the performance obligations in the client contract has been achieved. A performance obligation is a contractual
promise to transfer product to the customer. The transaction price of a contract is allocated to each distinct performance obligation
and recognized as revenue when or as, the customer receives the benefit of the performance obligation. Under ASC 606, revenue is recognized
when a customer obtains control of goods in an amount that reflects the consideration the Company expects to receive in exchange for
those goods. To achieve this core principle, the Company applies the following five steps:
1)
Identify the contract with a customer.
2)
Identify the performance obligations in the contract.
3)
Determine the transaction price.
4)
Allocate the transaction price to performance obligations in the contract.
5)
Recognize revenue when or as the Company satisfies a performance obligation.
For
projects, that are completed within the Company’s facility, the company designs, manufactures and sells custom mirrors for hotels
and hospitals through contractual agreements. These sales require the company to deliver the products within three to nine months from
commencement of order acceptance. Deferred revenue represents amounts billed in excess of revenues recognized. Revenues recognized in
excess of amounts billed typically does not occur as the Company will not perform any work in excess of the amount the company bills
to its customers. If work is performed in excess of amounts billed, the Company will record an unbilled receivable.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
1 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Each
product or service delivered to a third-party customer that is manufactured by a third-party vendor is considered to satisfy a performance
obligation. Performance obligations generally occur at a point in time and are satisfied when control of the goods passes to the customer.
These sales are shipped from the manufacturer to the customer without our taking physical inventory possession. The Company reports direct
sales on a gross basis, that is, the amounts billed to our customers are recorded as "Sales," and inventory purchased from manufacturers
are recorded as cost of sales. The Company is the principal of direct sales because the Company has the risk of loss and we control the
inventory before it is transferred to our customers. Our control is evidenced by us being primarily responsible for fulfilling the promise
to our customers, taking on inventory risk of returned product, and having discretion in establishing pricing. Returns have historically
been insignificant to our operations. The Company typically pay our vendors a portion of the total cost up front and the remaining balance
is accrued for and paid within 30 to 60 days of when the products are shipped from the third-party warehouse. Deferred revenue represents
amounts invoiced or deposits received from our customer for which the Company has not yet satisfied our performance obligation.
The
company applied the five-step model to the sales of Akida’s and KES’s Airocide and misting system products, and SciAir’s
whole-room aerosol chamber and laboratory certified air disinfection machines. At contract inception and once the contract is determined
to be within the scope of ASC 606, the Company assesses the goods or services promised within each contract and determines those that
are performance obligations and assesses whether each promised good or service is distinct. The Company sells Airocide air sterilization
units, misting systems, and whole-room aerosol chamber and laboratory certified disinfection machines to both consumer and commercial
customers. These products are sold both domestically and internationally. The cycle from contract inception to shipment of products is
typically one day to three months. The Company’s contracts for both its consumer and commercial customers each contain a single
performance obligation (delivery of Airocide, KES, and SciAir products), as the promise to transfer the individual goods or services
is not separately identifiable from other promises in the contracts and, therefore, not distinct. As a result, the entire transaction
price is allocated to this single performance obligation. The Company recognizes revenues at a point in time when the customer obtains
control of the Company’s product, which typically occurs upon shipment of the product by the Company or upon customer pick-up via
third party common carrier.
Revenue
recognized over time and revenue recognized at a point in time for the years ended:
| Schedule
of revenue | |
| | | |
| | |
| | |
December
31, |
| | |
2021 | |
2020 |
| Recognized
over time | |
$ | 1,606,950 | | |
$ | 2,984,655 | |
| Recognized
at a point in time | |
| 10,060,629 | | |
| 2,748,079 | |
| | |
$ | 11,667,579 | | |
$ | 5,732,734 | |
Deferred
revenue was comprised of the following as of:
| | |
December
31, | |
December
31, |
| | |
2021 | |
2020 |
| Recognized
over time | |
$ | 94,867 | | |
$ | 233,080 | |
| Recognized
at a point in time | |
| 693,908 | | |
| 608,556 | |
| | |
$ | 788,775 | | |
$ | 841,636 | |
All
deferred revenue as of December 31, 2020 was recognized as revenue during the year ended December 31, 2021.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
1 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Shipping
and Handling Charges
The
Company reports shipping and handling fees charged to customers as part of net sales and the associated expense as part of cost of sales.
Shipping charges amounted to $963,385 and
$1,225,752 for
the years ended December 31, 2021 and 2020, respectively.
Advertising
Advertising
costs consist primarily of online search advertising and placement, trade shows, advertising fees, and other promotional expenses. Advertising
costs are expensed as incurred and are included in sales and marketing on the consolidated statements of operations. Advertising expense
for the years ended December 31, 2021 and 2020 was $799,799
and $317,560.
Vendor
deposits
Vendor
payments to third manufactures are capitalized until completion of the project and are recorded as vendor deposits. As of December 31,
2021 and 2020, the vendor deposit balance was $992,042
and $40,800,
respectively.
Patent
Costs
The
Company capitalizes costs consisting principally of outside legal costs and filing fees related to obtaining and maintaining patents.
The Company amortizes patent costs over the useful life of the patent which is typically 20
years, beginning with the date the patent is
filed with the U.S. Patent and Trademark Office, or foreign equivalent. As of December 31, 2021 and 2020, capitalized patent costs net
of accumulated amortization was $1,693,124 and
$178,088,
respectively. For the years ended December 31, 2021 and 2020, the Company recorded $32,398
and $4,566,
respectively, of amortization expense for these patents.
Recent
Accounting Pronouncements
In
December 2019, the FASB issued ASU No. 2019-12, Income Taxes (Topic 740) (“ASU 2019-12”): Simplifying the Accounting for
Income Taxes. The new standard eliminates certain exceptions related to the approach for intraperiod tax allocation, the methodology
for calculating income taxes in an interim period, and the recognition of deferred tax liabilities for outside basis differences related
to changes in ownership of equity method investments and foreign subsidiaries. The guidance also simplifies aspects of accounting for
franchise taxes and enacted changes in tax laws or rates and clarifies the accounting for transactions that result in a step-up in the
tax basis of goodwill. For public business entities, it is effective for fiscal years beginning after December 15, 2020, including interim
periods within those fiscal years. The adoption of this guidance did not have a material impact on the accompanying consolidated financial
statements.
In
June 2020, the FASB issued ASU No. 2020-06, Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging - Contracts
in Entity’s Own Equity (Subtopic 815-40). This standard eliminates the beneficial conversion and cash conversion accounting models
for convertible instruments. It also amends the accounting for certain contracts in an entity’s own equity that are currently accounted
for as derivatives because of specific settlement provisions. In addition, the new guidance modifies how particular convertible instruments
and certain contracts that may be settled in cash or shares impact the diluted EPS computation. For public business entities, it is effective
for fiscal years beginning after December 15, 2021, including interim periods within those fiscal years using the fully retrospective
or modified retrospective method. Early adoption is permitted but no earlier than fiscal years beginning after December 15, 2020, including
interim periods within those fiscal years. The Company has not adopted this accounting pronouncement and is currently evaluating the
potential impact of this standard on our consolidated financial statements.
From
time to time, new accounting pronouncements are issued by the FASB or other standard setting bodies that the Company adopts as of the
specified effective date. The Company does not believe that the impact of recently issued standards that are not yet effective will have
a material impact on the Company’s financial position or results of operations upon adoption.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
2 – BUSINESS ACQUISITION
The
Company accounted for the acquisitions as a business combinations using the acquisition method of accounting as prescribed in Accounting
Standards Codification 805, Business Combinations (“ASC 805”) and ASC 820 – Fair Value Measurements and Disclosures
(“ASC 820”). In accordance with ASC 805 and ASC 820, the Company used its best estimates and assumptions to accurately assign
fair value to the tangible assets acquired, identifiable intangible assets and liabilities assumed as of the acquisition dates. Goodwill
as of the acquisition date is measured as the excess of purchase consideration over the fair value of tangible and identifiable intangible
assets acquired and liabilities assumed. The results of operations of the acquired businesses since the date of acquisition are included
in the consolidated financial statements of the Company for the year ended December 31, 2021. The total purchase consideration was allocated
to the assets acquired and liabilities assumed at their estimated fair values as of the date of acquisition, as determined by management.
The excess of the purchase price over the amounts allocated to assets acquired and liabilities assumed has been recorded as goodwill.
The value of the goodwill from the acquisitions described below can be attributed to a number of business factors including, but not
limited to, cost synergies expected to be realized and a trained technical workforce.
In
conjunction with acquisitions noted below, we used various valuation techniques to determine fair value of the assets acquired, with
the primary techniques being discounted cash flow analysis, relief-from-royalty, a form of the multi-period excess earnings and the with-and-without
valuation approaches, which use significant unobservable inputs, or Level 3 inputs, as defined by the fair value hierarchy. Inputs to
these valuation approaches require significant judgment including: (i) forecasted sales, growth rates and customer attrition rates, (ii)
forecasted operating margins, (iii) royalty rates and discount rates used to present value future cash flows, (iv) the amount of synergies
expected from the acquisition, (v) the economic useful life of assets and (vi) the evaluation of historical tax positions. In certain
acquisitions, historical data is limited, therefore, we base our estimates and assumptions on budgets, business plans, economic projections,
anticipated future cash flows and marketplace data.
On
February 8, 2021 Applied UV, Inc. (the “Company”), entered into an asset purchase agreement (the “APA”) by and
among the Company, SteriLumen, Inc., a New York corporation and wholly-owned subsidiary of the Company (the “Purchaser”)
and Akida Holdings LLC, a Florida limited liability company (the “Seller”) pursuant to which the Purchaser acquired substantially
all of the assets of the Seller and assumed certain of its current liabilities and contract obligations, as set forth in the APA (the
“Acquisition”). In the Acquisition, the Purchaser acquired all the Seller’s assets and was assigned its contracts related
to the manufacturer and sale of the Airocide™ system, originally developed for NASA with assistance from the University of Wisconsin
at Madison, that uses a combination of UV-C and a proprietary, titanium dioxide-based photocatalyst that has applications in the hospitality,
hotel, healthcare, nursing homes, grocer, wine, commercial buildings, and retail sectors.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
2 – BUSINESS ACQUISITION (CONTINUED)
The
purchase price and purchase price allocation as of the acquisition completion date follows.
| Schedule
of Recognized Identified Assets Acquired and Liabilities Assumed | |
| | |
| Purchase
Price: | |
|
| Cash | |
$ | 760,293 | |
| Fair
market value of common stock issued (1,375,000 shares) | |
| 7,122,500 | |
| Total
Purchase Price, Net of Cash Acquired | |
| 7,882,793 | |
| | |
| | |
| Assets
Acquired: | |
| | |
| Accounts
receivable | |
| 233,241 | |
| Inventory | |
| 211,105 | |
| Prepaid
expenses | |
| 285,490 | |
| Machinery
and equipment | |
| 168,721 | |
| Customer
relationships | |
| 539,000 | |
| Trade
names | |
| 1,156,000 | |
| Intellectual
property | |
| 3,468,000 | |
| Total
Assets Acquired: | |
| 6,061,557 | |
| | |
| | |
| Liabilities
Assumed: | |
| | |
| Accounts
payable | |
| (415,341 | ) |
| Deferred
revenue | |
| (491,702 | ) |
| Total
Liabilities Assumed | |
| (907,043 | ) |
| Net
Assets Acquired | |
| 5,154,514 | |
| Excess
Purchase Price "Goodwill" | |
$ | 2,728,279 | |
The
excess purchase price has been recorded as goodwill in the amount of approximately $2,728,279.
The estimated useful life of the identifiable intangible assets (see note 5) is seven to ten years. The goodwill is amortizable for tax
purposes.
On
September 28, 2021, SteriLumen, Inc. completed an Asset Purchase Agreement with KES Science & Technology, Inc. (“KES”),
a Georgia corporation.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
2 – BUSINESS ACQUISITION (CONTINUED)
The
purchase price and purchase price allocation as of the acquisition completion date follows.
| Purchase
Price: | |
|
| Cash | |
$ | 4,299,900 | |
| Fair
market value of common stock issued (300,000 shares) | |
| 1,959,001 | |
| Total
Purchase Price, Net of Cash Acquired | |
| 6,258,901 | |
| | |
| | |
| Assets
Acquired: | |
| | |
| Accounts
receivable | |
| 392,367 | |
| Inventory | |
| 602,746 | |
| Prepaid
expenses | |
| 10,995 | |
| Machinery
and equipment | |
| 36,146 | |
| Customer
relationships | |
| — | |
| Trade
names | |
| 914,000 | |
| Intellectual
property | |
| 3,656,000 | |
| Total
Assets Acquired: | |
| 5,612,254 | |
| | |
| | |
| Liabilities
Assumed: | |
| | |
| Accounts
payable | |
| (296,681 | ) |
| Net
Assets Acquired | |
| 5,315,573 | |
| Excess
Purchase Price "Goodwill" | |
$ | 943,328 | |
The
excess purchase price has been recorded as goodwill in the amount of $943,328.
The estimated useful life of the identifiable intangible assets is ten years (see note 5). The goodwill is amortizable for tax purposes.
On
October 13, 2021, the Company entered into an asset purchase agreement by and among the Company, SteriLumen, Inc., a New York corporation
and wholly-owned subsidiary of the Company (the “Purchaser”) and Old SAM Partners, LLC, a Florida limited liability company
(the “Seller”), pursuant to which the Purchaser acquired substantially all of the assets of the Seller, including the assignment
of an exclusive distribution agreement. On October 13, 2021 the Seller received, as consideration for the Acquisition (i) $9,500,000
in cash; and (ii) 200,000 shares of the Company’s common stock and (iii) 200,000 unvested shares of the Company’s common
stock, which are subject to cancellation if the earnout is not met. On the date of acquisiton, the fair market value of the 200,000 vested
shares was $5.57 for a total value of $1,114,000. An additional liability was recorded for $886,000 as a result of the agreement calling
for additional cash consideration to the extent the share price is below $10 on the free trading date, as defined in the agreement. On
December 31, 2021, the share price of our common stock was $2.70 per share and a loss on contingent consideration of $574,000 was recorded
in the consolidated statements of operations and increased the liability to $1,460,000.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
The
preliminary purchase price and purchase price allocation as of the acquisition completion date follows.
| Purchase
Price: | |
|
| Cash | |
$ | 9,500,000 | |
| Fair
market value of common stock issued | |
| 1,114,000 | |
| Contingent
consideration based on stock price | |
| 886,000 | |
| Total
Purchase Price, net of cash acquired | |
| 11,500,000 | |
| | |
| | |
| Assets
Acquired: | |
| | |
| Accounts
receivable | |
| 129,845 | |
| Inventory | |
| 369,970 | |
| Machinery
and equipment | |
| 1,982 | |
| Customer
relationships | |
| 6,784,000 | |
| Patents | |
| 1,533,000 | |
| Intellectual
property | |
| 1,217,000 | |
| Trade
names | |
| 326,000 | |
| Total
Assets Acquired: | |
| 10,361,797 | |
| | |
| | |
| Assets
Acquired | |
| 10,361,797 | |
| Excess
Purchase Price "Goodwill" | |
$ | 1,138,203 | |
The
excess purchase price has been recorded as goodwill in the amount of approximately $1,138,203.
The estimated useful life of the identifiable intangible assets (see note 5) is five to seventeen years. The goodwill is amortizable
for tax purposes.
The
purchase price allocation presented above related to SciAir is preliminary. We are in the process of evaluating additional information
necessary to finalize the valuation of assets acquired and liabilities assumed as of the acquisition date including, but not limited
to, post-closing adjustments to the working capital acquired including certain holdbacks, as well as the valuation and step-up on property,
plant and equipment. The final fair value determination could result in material adjustments to the values presented in the preliminary
purchase price allocation, including other intangible assets, goodwill and the related tax impact of such adjustments. We expect to finalize
the purchase price allocation by mid-2022.
See
Note 15 for the required pro forma information related to these business combinations.
NOTE
3 – INVENTORY
Inventory
consists of the following:
| Schedule
of Inventory | |
| | | |
| | |
| | |
December
31, | |
December
31, |
| | |
2021 | |
2020 |
| Raw
materials | |
$ | 356,759 | | |
$ | 156,290 | |
| Finished
goods | |
| 1,429,482 | | |
| — | |
| Inventory
at cost | |
$ | 1,786,241 | | |
$ | 156,209 | |
NOTE
4 – PROPERTY AND EQUIPMENT
Property
and equipment (including machinery and equipment under financing leases) are summarized by major classifications as follows:
| Schedule
of property and equipment | |
| | | |
| | |
| | |
December
31, | |
December
31, |
| | |
2021 | |
2020 |
| Machinery
and Equipment | |
$ | 254,685 | | |
$ | 61,083 | |
| Leasehold
improvements | |
| 67,549 | | |
| 60,223 | |
| Furniture
and Fixtures | |
| 54,041 | | |
| 33,385 | |
| | |
| 376,275 | | |
| 154,691 | |
| Less:
Accumulated Depreciation | |
| (179,664 | ) | |
| (41,887 | ) |
| | |
$ | 196,611 | | |
$ | 112,804 | |
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
4 – PROPERTY AND EQUIPMENT (continued)
Depreciation
expense, including depreciation of assets under financing leases, for the year ended December 31, 2021 and 2020 was $137,777
and $17,638,
respectively.
Machinery
and equipment under financing leases are summarized by as follows:
| Machinery
and equipment | |
| | | |
| | |
| | |
December
31, | |
December
31, |
| | |
2021 | |
2020 |
| Machinery
and Equipment | |
$ | 61,083 | | |
$ | 61,083 | |
| Less:
Accumulated Depreciation | |
| (37,420 | ) | |
| (28,023 | ) |
| Total | |
$ | 23,663 | | |
$ | 33,060 | |
The
cost of assets acquired under financing leases was $61,083.
Depreciation of assets under financing leases, for the years ended December 31, 2021 and 2020 was $9,397,
respectively.
NOTE
5 – INTANGIBLE ASSETS
Intangible
assets as of December 31, 2021 and 2020 consist of the following:
| Schedule
of Intangible Assets | |
| | | |
| | |
| | |
December
31, | |
December
31, |
| | |
2021 | |
2020 |
| Intangible
assets subject to amortization | |
| | | |
| | |
| Customer
Relationship | |
$ | 7,323,000 | | |
$ | — | |
| Trade
Names | |
| 2,396,000 | | |
| — | |
| Patents | |
| 1,730,089 | | |
| 182,654 | |
| Intellectual
Property | |
| 8,341,000 | | |
| — | |
| | |
| 19,790,089 | | |
| 182,654 | |
| Less:
Accumulated Amortization | |
| (813,533 | ) | |
| (4,566 | ) |
| | |
$ | 18,976,556 | | |
$ | 178,088 | |
During
the years ended December 31, 2021 and 2020, the Company recorded total amortization expense related to intangible assets of $808,967
and $4,566,
respectively. The useful lives of tradenames ranges from 5 to 10 years, technology is 10 years, customer relationships ranges from 7
to 14 years, and patents range from 17 to 20 years.
Future
amortization of intangible assets is as follows:
| Future
amortization of intangible assets | | |
| | |
| For
the year ending December 31, | |
|
| 2022 | | |
$ | 1,767,181 | |
| 2023 | | |
| 1,767,181 | |
| 2024 | | |
| 1,767,181 | |
| 2025 | | |
| 1,767,181 | |
| 2026 | | |
| 1,750,881 | |
| Thereafter | | |
| 10,156,951 | |
| Total | | |
$ | 18,976,556 | |
NOTE
6 – FINANCING LEASE OBLIGATION
The
Company's future minimum principal and interest payments under a capital lease for machinery and equipment are as follows as of December
31, 2021:
For the
Year Ended December 31,
| Schedule
of future minimum principal and interest payments under capital lease arrangements | |
| | |
| 2022 | |
$ | 8,240 | |
| Less:
Amount representing interest | |
| (569 | ) |
| Present
value of future minimum lease payments | |
| 7,671 | |
| Less:
current portion | |
| (7,671 | ) |
| Financing
lease obligations | |
$ | — | |
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
7 – LOANS PAYABLE
In
April of 2021, the company settled a previously issued loan payable of $85,000
which was included in accounts payable and accrued
expenses as of December 31, 2020, for $65,000
and recorded a $20,000
gain on settlement.
The Company
entered into a loan agreement in April of 2019 where the company was required to pay $157,500
in five payments in the amount of $30,000
per year, with an additional $7,500,
representing interest, in year two to a loan holder. As of December 31, 2021, the company has an outstanding balance of $157,500,
respectively.
Minimum
obligations under this loan agreement are as follows:
| Schedule
of minimum obligations under loan agreement | |
| | |
| For
the year ending December 31, | |
|
| 2022 | |
$ | 97,500 | |
| 2023 | |
| 30,000 | |
| 2024 | |
| 30,000 | |
| | |
$ | 157,500 | |
NOTE
8– FAIR VALUE MEASUREMENTS
Accounting
guidance on fair value measurements requires that financial assets and liabilities be classified and disclosed in one of the following
categories of the fair value hierarchy:
Level
1 – Based on unadjusted quoted prices for identical assets or liabilities in an active market.
Level
2 – Based on observable market-based inputs or unobservable inputs that are corroborated by market data.
Level
3– Based on unobservable inputs that reflect the entity’s own assumptions about the assumptions that a market participant
would use in pricing the asset or liability.
We
did not have any transfers between levels during the periods presented.
The
following table presents assets and liabilities that were measured at fair value in the Consolidated Balance Sheets on a recurring basis
as of December 31, 2021:
| Fair
Value, Assets Measured on Recurring Basis | |
| | | |
| | | |
| | | |
| | | |
| | |
| | |
As
of December 31, 2021 |
| | |
Carrying
Amount | |
Fair
Value | |
Quoted
Priced in active markets (Level 1) | |
Significant
other observable inputs (Level 2) | |
Significant
unobservable inputs (Level 3) |
| Assets | |
| |
| |
| |
| |
|
| Money
market funds | |
$ | 1,076,664 | | |
$ | 1,076,664 | | |
$ | 1,076,664 | | |
$ | — | | |
$ | — | |
| Total
assets | |
$ | 1,076,664 | | |
$ | 1,076,664 | | |
$ | 1,076,664 | | |
$ | — | | |
$ | — | |
| Liabilities | |
| | | |
| | | |
| | | |
| | | |
| | |
| Contingent
consideration | |
$ | 1,460,000 | | |
$ | 1,460,000 | | |
$ | 1,460,000 | | |
| | | |
| $ | |
| Warrant
liability | |
$ | 68,263 | | |
$ | 68,263 | | |
$ | — | | |
$ | — | | |
$ | 68,263 | |
| Total
liabilities | |
$ | 1,528,263 | | |
$ | 1,528,263 | | |
$ | 1,460,000 | | |
$ | — | | |
$ | 68,263 | |
The
carrying amounts of accounts receivable, accounts payable and short-term debt approximated fair values as of December 31, 2021 because
of the relatively short maturity of these instruments. There were no other level 3, level 2, or level 1 assets or liabilities as
of December 31, 2021, and there were no level 3, level 2, or level 1 assets or liabilities as of December 31, 2020.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
Money
Market Funds – Cash equivalents of $1,076,664 and $0 as of December 31, 2021 and 2020, respectively, consisted of money market
funds. Money market funds are classified as Level 1 of the fair value hierarchy because they are valued using unadjusted quoted market
prices in active markets.
Contingent
consideration – The fair value of the contingent consideration is derived through the quoted market price of our common stock,
which represents a Level 1 measurement within the fair value hierarchy.
Warrant
liability – The fair value of the warrant liability is derived through the Black scholes method and is based on significant inputs
not observable in the market, which represents a Level 3 measurement within the fair value hierarchy. See note 9 for significant assumptions
used.
Other
Fair Value Measurements
In addition
to assets and liabilities that are recorded at fair value on a recurring basis, GAAP requires that, under certain circumstances, we also
record assets and liabilities at fair value on a nonrecurring basis.
In
connection with the acquisitions of SciAir, KES, and Akida during 2021, as discussed in Note 2, we used various valuation techniques
to determine fair value, with the primary techniques being discounted cash flow analysis and the relief-from-royalty, a form of the multi-period
excess earnings, which use significant unobservable inputs, or Level 3 inputs, as defined by the fair value hierarchy.
NOTE
9 – STOCKHOLDERS' EQUITY
Amendment
of the Certificate of Designation
On
June 17, 2021, the Company filed an amendment of the certificate of designation of Series A Preferred Stock. The Board of Directors,
by unanimous written consent, duly adopted resolutions to amend the Series A Preferred Stock Certificate of Designations and changed
the name from “Series A Preferred Stock” to “Series X Preferred Stock”. All dividend, liquidation preference,
voting, conversion, and redemption rights, did not change from the originally filed Certificate of Designation of Series A Preferred
Stock. There are 2,000 Series X Preferred Shares issued and outstanding as of December 31, 2021.
Pursuant
to the Company’s amended and restated certificate of incorporation, as amended, the Company is authorized to designate and issue
up to 20,000,000
shares of preferred stock, par value $0.0001
per share, in one or more classes or series.
During the year ended December 31, 2021, the Company had 10,000
preferred shares designated as Series X Preferred
Stock and 19,990,000 shares
of preferred stock designated as 10.5% Series A Cumulative Perpetual Preferred Stock (the “Series A Preferred Stock”). There
are 552,000 shares of Series A Preferred Stock issued and outstanding as of December 31, 2021. Upon certain events, the Company may,
subject to certain conditions, at the Company’s option, redeem the Series A Preferred Stock. See below for a further description
of the Series A Preferred Stock:
Dividends:
Holders are entitled to receive, cumulative cash dividends at the annual rate of 10.5% on $25.00 liquidation preference per share
of the Series A Perpetual Preferred Stock. Dividends accrue and are payable in arrears beginning August 15, 2021, regardless of whether
declared or there are sufficient earnings or funds available for payment. Sufficient net proceeds from the offering must be set aside
to pay dividends for the first twelve months from issuance. The company has classified $845,250 as restricted cash as of December 31,
2021 as a reserve to pay the remaining required dividends for the first year.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
9 – STOCKHOLDERS' EQUITY (continued)
Redemption:
Company has an optional redemption right beginning July 16, 2022, which redemption price declines annually. The initial redemption
price after year 1 is $30 and decreases annually over 5 years to $25 per share. The Company also has a special optional redemption right
upon the occurrence of a Delisting Event or Change of Control, as defined, at $25 per share plus accrued and unpaid dividends.
Voting
Rights: The holders have no voting rights, except for voting on certain corporate decisions, or upon default in payment of dividends
for any twelve periods, in which case the holders would have voting rights to elect two additional directors to serve on the Board of
Directors.
Conversion
Rights: Such shares are not convertible unless and until the occurrence of a Delisting Event or Change of Control and when the
Company has not exercised its special optional redemption right. The conversion price would be the lesser of the amount converted based
on the $25.00 liquidation preference plus accrued dividends divided by the common stock price of the Delisting Event or Change of Control
(as defined) or $5.353319 (Share Cap). Effectively, the Share Cap limits the common stock price to no lower than $4.67.
Reverse
Stock Split
In
June of 2020, the Company effected
a 5:1 reverse stock split (the “Reverse
Stock Split”) by filing an amendment to the Company’s Amended and Restated Certificate Incorporation with the Delaware Secretary
of State. The Reverse Stock Split combined every five shares of Common Stock issued and outstanding immediately prior to effecting the
Reverse Stock Split into one share of Common Stock. As a result, the number of issued and outstanding shares of Common Stock have been
retroactively adjusted in the consolidated financial statements.
2020
Incentive Plan
On
March 31, 2020, the Company adopted the Applied UV, Inc. 2020 Omnibus Incentive Plan (the “Plan”) with 600,000
(post-split adjusted) shares of common stock
available for issuance under the terms of the Plan. The Plan permits the granting of Nonqualified Stock Options, Incentive Stock Options,
Stock Appreciation Rights, Restricted Stock, Restricted Stock Units, Performance Shares, Performance Units and Other Awards. The objectives
of the Plan are to optimize the profitability and growth of the Company through incentives that are consistent with the Company’s
goals and that link the personal interests of Participants to those of the Company’s stockholders. The Plan is further intended
to provide flexibility to the Company in its ability to motivate, attract and retain the services of Participants who make or are expected
to make significant contributions to the Company’s success and to allow Participants to share in the success of the Company. From
time to time, the Company may issue Incentive Awards pursuant to the Plan. Each of the awards will be evidenced by and issued under a
written agreement.
If
an incentive award granted under the Plan expires, terminates, is unexercised or is forfeited, or if any shares are surrendered to the
company in connection with an incentive award, the shares subject to such award and the surrendered shares will become available for
future awards under the Plan. The number of shares subject to the Plan, and the number of shares and terms of any Incentive Award may
be adjusted in the event of any change in our outstanding common stock by reason of any stock dividend, spin-off, stock split, reverse
stock split, recapitalization, reclassification, merger, consolidation, liquidation, business combination or exchange of shares, or similar
transaction. There are 267,000
shares available for future grants under the
plan. The Company also granted an additional 309,835
options outside of the plan during the year ended
December 31, 2021.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
9 – STOCKHOLDERS' EQUITY (continued)
A
summary of the Company’s option activity and related information follows:
| Schedule
of the Company's option activity | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| | |
Shares
Available for Grant | |
Number
of
Options | |
Weighted-Average
Exercise Price | |
Weighted-Average
Grant Date Fair Value | |
Weighted-Average
Remaining Contractual Life (in years) | |
Aggregate
intrinsic value |
| Balances,
January 1, 2020 | |
| — | | |
| — | | |
$ | — | | |
$ | — | | |
| | | |
$ | — | |
| Options
granted | |
| 462,500 | | |
| 137,500 | | |
| 4.96 | | |
| 2.27 | | |
| 10 | | |
| — | |
| Options
forfeited/cancelled | |
| 750 | | |
| (750 | ) | |
| 5.00 | | |
| — | | |
| | | |
| — | |
| Options
exercised | |
| — | | |
| — | | |
| — | | |
| — | | |
| | | |
| — | |
| Balances,
December 31, 2020 | |
| 463,250 | | |
| 136,750 | | |
$ | 4.96 | | |
$ | 2.27 | | |
| 9.95 | | |
$ | — | |
| Options
granted outside of the plan | |
| — | | |
| 309,564 | | |
| 7.80 | | |
| 5.06 | | |
| 10 | | |
| — | |
| Options
granted | |
| (293,000 | ) | |
| 293,000 | | |
| 7.82 | | |
| 5.29 | | |
| 10 | | |
| — | |
| Options
forfeited/cancelled | |
| 95,000 | | |
| (95,000 | ) | |
| 4.96 | | |
| 3.73 | | |
| | | |
| — | |
| Options
exercised | |
| — | | |
| — | | |
| — | | |
| — | | |
| | | |
| — | |
| Balances,
December 31, 2021 | |
| 265,250 | | |
| 644,314 | | |
$ | 7.11 | | |
$ | 5.03 | | |
| 8.47 | | |
$ | — | |
| Vested
and exercisable | |
| | | |
| 135,836 | | |
$ | 6.92 | | |
| | | |
| | | |
$ | — | |
Share-based
compensation expense for options totaling $721,783
and $35,565
was recognized for the years ended December 31,
2021 and 2020, respectively, based on requisite service periods.
The
valuation methodology used to determine the fair value of the options issued during the year was the Black-Scholes option-pricing model.
The Black-Scholes model requires the use of a number of assumptions including volatility of the stock price, the average risk-free interest
rate, and the weighted average expected life of the options.
The
risk-free interest rate assumption is based upon observed interest rates on zero coupon U.S. Treasury bonds whose maturity period is
appropriate for the term of the options.
Estimated
volatility is a measure of the amount by which the Company’s stock price is expected to fluctuate each year during the expected
life of the award. The Company’s calculation of estimated volatility is based on historical stock prices of peer entities over
a period equal to the expected life of the awards. The Company uses the historical volatility of peer entities due to the lack of sufficient
historical data of its stock price.
As
of December 31, 2021, there was $2,262,765
of total unrecognized compensation expense related
to unvested employee options granted under the Company’s share-based compensation plans that is expected to be recognized over
a weighted average period of approximately 3.1 years.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
9– STOCKHOLDERS' EQUITY (continued)
The
weighted average fair value of options granted, and the assumptions used in the Black-Scholes model during the years ended December 31,
2021 and 2020 are set forth in the table below.
| Schedule
of Share-based Payment Award, Stock Options, Valuation Assumptions | |
| | | |
| | |
| | |
2021 | |
2020 |
| Risk-free
interest rate | |
| 1.02%
to 1.54% | | |
| 0.31%
to 0.37% | |
| Volatility | |
| 50.13%
to 89.96% | | |
| 41.40%
to 51.45% | |
| Expected
life (years) | |
| 5.36-6.25 | | |
| 5.5 | |
| Dividend
yield | |
| 0.00 | % | |
| 0.00 | % |
Common
Stock Warrants
A summary
of the Company’s warrant activity and related information follows:
| Schedule
of the Company's warrant activity | |
| | | |
| | |
| | |
Number
of
Shares | |
Weighted-Average
Exercise Price |
| Warrants
Outstanding at January 1, 2020 | |
| — | | |
| | |
| Granted
(See note below) | |
| 235,095 | | |
$ | 5.89 | |
| Warrants
Outstanding at December 31, 2020 | |
| 235,095 | | |
$ | 5.89 | |
| Granted | |
| | | |
$ | — | |
| Exercised | |
| (42,676) | | |
$ | (6.10 | ) |
| Warrants
Outstanding, December 31, 2021 | |
| 192,419 | | |
$ | 5.84 | |
On
August 31, 2020, the Company closed its offering (the “August Offering”) in which it issued 1,150,000
common shares at a public offering price of $5.00
per share. In connection with the Offering, the
Company (i) received $5,750,000
less underwriting fees of $517,500
and write-off of capitalized IPO Costs in the
amount of $341,145,
resulting in net proceeds of $4,891,355.
Additionally, the Company issued an additional 161,794
shares to Ross Carmel of Carmel, Milazzo &
Feil LLP related to the offering for compensation. The shares were offered and sold to the public pursuant to the Company’s registration
statement on Form S-1, filed by the Company with the SEC on August 26, 2020, as amended, which became effective on August 28, 2020. In
connection with the August Offering, the company granted 80,000
warrants to the underwriters for compensation.
The fair market value of the warrants were recorded as a reduction of the proceeds and netted with additional paid in capital.
On
November 13, 2020, the Company closed its second offering (the “November Offering”) in which it issued 1,401,905
common shares at a public offering price of $5.25
per share. In connection with the Offering, the
Company (i) received $7,360,000
less underwriting fees of $625,600
and write-off of previously related capitalized
IPO Costs in the amount of $316,246,
resulting in net proceeds of $6,418,155.
In connection with the November Offering, the company granted 70,095
warrants to the underwriters for compensation.
The fair market value of the warrants were recorded as a reduction of the proceeds and netted with additional paid in capital.
Share-based
compensation expense of $0
and $100,896
for warrants granted was recognized for the years
ended December 31, 2021 and 2020, respectively, based on requisite service period. The warrants granted in 2020 were issued in connection
with the August and November offerings (150,095 warrants) and were not previously disclosed in the 2020 financial statements. The warrants
issued in connection with the November offering contained a cash settlement feature which resulted in a warrant liability of $68,263
as of December 31, 2021. In connection with the
preparation of the consolidated financial statements for the year ended December 31, 2021, the Company recognized an error relating to
the recognition of the initial warrant liability in November of 2020. The error caused additional paid in capital to be overstated by
approximately $135,000, warrant liability to be understated by approximately $110,000, and net loss to be overstated by approximately
$25,000 as of and for the year ended December 31, 2020. The Company concluded the impact on the year ended December 31, 2020 financial
statements was immaterial and corrected the balances as of December 31, 2021. The Company valued the warrant using the Black-Scholes
option pricing model with the following terms on date of grant of: (a) exercise price of $6.5625, (b) volatility rate of 50.39%, (c)
risk free rate of 0.26%, (d) term of five years, and (e) dividend rate of 0%. The Company valued the warrant using the Black-Scholes
option pricing model with the following terms on December 31, 2021: (a) exercise price of $6.5625, (b) volatility rate of 77.34%, (c)
risk free rate of 0.98%, (d) term of 3.86 years, and (e) dividend rate of 0%.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
9 – STOCKHOLDERS' EQUITY (continued)
The
valuation methodology used to determine the fair value of the warrants issued during the periods was the Black-Scholes option-pricing
model. The Black-Scholes model requires the use of a number of assumptions including volatility of the stock price, the average risk-free
interest rate, and the weighted average expected life of the warrants.
The
risk-free interest rate assumption is based upon observed interest rates on zero coupon U.S. Treasury bonds whose maturity period is
appropriate for the term of the warrants.
Estimated
volatility is a measure of the amount by which the Company’s stock price is expected to fluctuate each year during the expected
life of the award. The Company’s calculation of estimated volatility is based on historical stock prices of peer entities over
a period equal to the expected life of the awards. The Company uses the historical volatility of peer entities due to the lack of sufficient
historical data of its stock price.
The
weighted average fair value of warrants granted, and the assumptions used in the Black-Scholes model during the year ended December 31,
2021 are set forth in the table below.
| Defined
Benefit Plan, Assumptions | |
| | | |
| | |
| | |
2021 | |
2020 |
| Risk-free
interest rate | |
| 0.26 | % | |
| 0.31%-37% | |
| Volatility | |
| 50.13%-89.96% | | |
| 41.40%
to 51.45% | |
| Expected
life (years) | |
| 5 | | |
| 5 | |
| Dividend
yield | |
| 0.00 | % | |
| 0.00 | % |
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
9 – STOCKHOLDERS' EQUITY (continued)
Preferred
Stock Offering
On
July 13, 2021, Applied UV, Inc. (the “Company”) entered into an underwriting agreement (the “Underwriting Agreement”)
with Ladenburg Thalmann & Co. Inc. as representative (“Representative”) of the underwriters (“Underwriters”),
related to the offering of 480,000 shares (the “Shares”) of the Company’s 10.5% Series A Cumulative Perpetual Preferred
Stock [non-convertible], par value $0.0001 per share (“Series A Preferred Stock”), at a public offering price of $25.00 per
share, which excludes 72,000 shares of Series A Cumulative Perpetual Preferred Stock that may be purchased by the Underwriters pursuant
to their overallotment option granted to the Underwriters under the terms of the Underwriting Agreement. The Shares were offered and
sold by the Company pursuant to the terms of the Underwriting Agreement and registered pursuant to the Company’s registration statement
on (i) Form S-1 (File No. 333-257197), as amended, which was filed with the SEC and declared effective by the Commission on July 12,
2021 and (ii) the Company’s registration statement on Form S-1MEF (File No. 333-257862), which was filed with the Commission on
July 13, 2021 and declared effective upon filing. The closing of the offering for the Shares took place on July 16, 2021 and were approved
for listing on Nasdaq under the trading symbol “AUVIP”. On July 29, 2021, in connection with its offering of its 10.5% Series
A Cumulative Perpetual Preferred Stock, par value $0.0001 per share, the Company closed the exercise of the underwriter’s overallotment
option of 72,000 shares at $25.00 per share. Aggregate gross proceeds including the exercise of the underwriter's overallotment option
was $12,272,440 after deducting underwriting discounts and commissions and fees and other offering expenses. As of December 31, 2021,
the Company paid $603,750
to its cumulative perpetual preferred shareholders in a form
of a dividend. There are no dividends accrued for as of December 31, 2021.
Common
Stock Offering
On
December 28, 2021, the Company closed a common stock offering in which it issued 2,666,667
common shares at a public offering price of $3.00
per share. In connection with the Offering, the
Company (i) received $8,000,000
less underwriting fees of $560,000
and offering Costs in the amount of $440,073,
resulting in net proceeds of $6,999,928.
On
January 5, 2022, the underwriters fully exercised their over-allotment option to purchase an additional 400,000
shares of common stock at the public offering
price of $3.00
per share. The Company received gross proceeds
of $1,200,000
for the over-allotment, which resulted in net
proceeds to us of $1,092,000,
after deducting underwriting discounts and commissions of $108,000.
Restricted
Stock Awards
The
Company records compensation expense for restricted stock awards based on the quoted market price of our stock at the grant date and
the expense is amortized over the vesting period. These restricted stock awards are subject to time-based vesting conditions based on
the continued service of the restricted stock award holder. Restricted stock awards granted typically have an initial annual cliff vest
and then vest quarterly over the remaining service period, which is generally one to four years.
The
following table presents the restricted stock units activity for the years ended December 31, 2021 and 2020
| Schedule
of Unvested Restricted Stock Units Activity | |
| | | |
| | |
| | |
Number
of
Shares | |
Weighted-Average
Fair Market Value |
| Unvested
shares at January 1, 2020 | |
| — | | |
$ | — | |
| Granted
and unvested | |
| 230,083 | | |
| 5.00 | |
| Vested | |
| (42,528 | ) | |
| 5.00 | |
| Forfeited/Cancelled | |
| — | | |
| — | |
| Unvested
shares, December 31, 2020 | |
| 187,555 | | |
$ | 5.00 | |
| Granted
and unvested | |
| 274,500 | | |
| 5.16 | |
| Vested | |
| (163,176 | ) | |
| 5.24 | |
| Forfeited/Cancelled | |
| (6,379 | ) | |
| 5.00 | |
| Unvested
shares, December 31, 2021 | |
| 292,500 | | |
$ | 4.71 | |
| Vested
as of December 31, 2021 | |
| 205,704 | | |
$ | 5.19 | |
Upon
vesting, the restricted stock units are converted to common shares. Based on the terms of the restricted share and restricted stock
unit grants, all forfeited shares revert back to the Company.
In
connection with the grant of restricted shares, the Company recognized $550,138
and $828,006
of compensation expense within its statements
of operations for the years ended December 31, 2021 and 2020, respectively.
The
unvested shares as of December 31, 2021 represent $125,000
in unrecognized stock based compensation which
will be recognized over a weighted average period of 2.5 years.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
10 - LEASING ARRANGEMENTS
The
Company determines whether an arrangement qualifies as a lease under ASC 842 at inception. The Company has operating leases for office
space and office equipment. The Company’s leases have remaining lease terms of one year to seven years, some of which include options
to extend the lease term for up to five years. The Company considered these options to extend in determining the lease term used to establish
the Company’s right-of use assets and lease liabilities once reasonably certain of exercise. The Company’s lease agreements
do not contain any material residual value guarantees or material restrictive covenants.
ROU
assets represent the Company’s right to use an underlying asset for the lease term and lease liabilities represent the Company’s
obligation to make lease payments arising from the lease. Operating lease ROU assets and operating lease liabilities are recognized at
the lease commencement date based on the present value of the future lease payments over the lease term. The operating lease ROU asset
also includes any lease payments made in advance of lease commencement and excludes lease incentives. The lease terms used in the calculations
of the operating ROU assets and operating lease liabilities include options to extend or terminate the lease when the Company is reasonably
certain that it will exercise those options. Lease expense for lease payments is recognized on a straight-line basis over the lease term.
As
the Company’s leases do not provide an implicit rate, the Company uses an incremental borrowing rate of 7.6%
based on the information available at commencement date in determining the present value of lease payments.
MunnWorks,
LLC entered into a lease agreement in Mount Vernon, New York for a term that commenced on April 1, 2019 and will expire on the 31st day
of March 2024 at a monthly rate of $13,400. In March of 2021, the Company obtained additional lease space and the agreement was amended
to increase rent expense to $15,000 per month. On July 1, 2021, the Company again obtained additional lease space and rent expense was
increased to $27,500 per month through July 1, 2024 and $29,150 per month from July 1, 2024 through July 1, 2026.
On
September 28, 2021, the Company entered into a lease agreement in Kennesaw, Georgia for office and production space for a term that commenced
on September 29, 2021 and will expire on October 1, 2024, with a rate ranging from $14,729 to $15,626 per month.
Rent
expense for the years ended December 31, 2021 and 2020 was $249,100
and $171,600,
respectively.
Schedule
maturities of operating lease liabilities outstanding as of December 31, 2021 are as follows:
| Schedule
of maturities of operating lease liabilities | |
| | |
| 2022 | |
$ | 508,071 | |
| 2023 | |
| 513,413 | |
| 2024 | |
| 470,532 | |
| 2025 | |
| 349,800 | |
| 2026 | |
| 174,900 | |
| Total
lease payments | |
| 2,016,716 | |
| Less:
Imputed Interest | |
| (280,802 | ) |
| Present
value of future minimum lease payments | |
$ | 1,735,914 | |
Consistent
with ASC 842-20-50-4, the Company calculated its total lease cost based solely on its monthly rent obligation. The Company had no cash
flows arising from its lease, no finance lease cost, short term lease cost, or variable lease costs. The Company’s lease does not
produce any sublease income, or any net gain or loss recognized from sale and leaseback transactions. As a result, the Company did not
need to segregate amounts between finance and operating leases for cash paid for amounts included in the measurement of lease liabilities,
segregated between operating and financing cash flows; supplemental non-cash information on lease liabilities arising from obtaining
right-of-use assets; weighted-average calculations for the remaining lease term; or the weighted-average discount rate.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
11 - PAYROLL PROTECTION PROGRAM
In
April of 2020, the Company submitted a Paycheck Protection Program (“PPP”) application to Chase Bank for a loan amount equal
to $296,827.
The amount was approved, and the Company received the funds. The PPP Loan, which is in the form of a PPP promissory note and agreement,
matures in April of 2025 and bears interest at a rate of 1.00% per annum. The Lender will have 90 days to review borrower’s forgiveness
application and the SBA will have an additional 60 days to review the Lender’s decision as to whether the borrower’s loan
may be forgiven. Under the CARES Act, loan forgiveness is available for the sum of documented payroll costs, covered rent payments, covered
utilities, and certain covered mortgage interest payments during the twenty-four-week period beginning on the date of first disbursement
of the PPP Loan. For purposes of the CARES Act, payroll costs exclude compensation of an individual employee earning more than $100,000,
prorated annually. Not
more than 40% of the forgiven amount may be for non-payroll costs. Forgiveness is reduced if full-time headcount declines, or if salaries
and wages for employees with salaries of $100,000 or less annually are reduced by more than 25%. The loan was forgiven in July of 2021
and in accordance with ASC 470, the amount was recorded as other income.
NOTE
12 - INCOME TAXES
The provision
for federal and state income taxes for the years ended is as follows:
| Schedule
of federal and state income | |
| | | |
| | |
| | |
2021 | |
2020 |
| Current
provision: | |
| | | |
| | |
| Federal | |
$ | (110,234 | ) | |
$ | 53,265 | |
| State | |
| 18,415 | | |
| 993 | |
| Deferred
provision (benefit): | |
| | | |
| | |
| Federal | |
$ | — | | |
$ | 12,110 | |
| State | |
| — | | |
| 486 | |
| Total
Deferred | |
| — | | |
| 12,596 | |
| Total
provision for income taxes | |
$ | 91,819 | | |
$ | 66,854 | |
Realization
of the future tax benefits related to the deferred tax assets is dependent on many factors, including the Company’s ability to
generate taxable income within the net operating loss carryforward period. Management has considered these factors in reaching its conclusion
as to the valuation allowance for financial reporting purposes and has recorded a full valuation allowance against the deferred tax asset.
The
income tax effect of temporary differences comprising the deferred tax assets and deferred tax liabilities is a result of the following
at December 31:
| Schedule
of deferred tax assets and deferred tax liabilities | |
| | | |
| | |
| | |
2021 | |
2020 |
| Deferred
tax assets: | |
| | | |
| | |
| Net
operating loss | |
$ | 1,732,422 | | |
$ | 299,088 | |
| Fixed
assets | |
| 14,493 | | |
| — | |
| Lease | |
| 1,201 | | |
| — | |
| Loss
on contingency | |
| 130,765 | | |
| — | |
| Intangible
assets | |
| 1,707 | | |
| — | |
| Stock
based compensation | |
| 284,468 | | |
| 189,883 | |
| Accrual
to cash conversion | |
| 216,068 | | |
| 367,289 | |
| Total | |
| 2,381,124 | | |
| 856,260 | |
| Valuation
allowance | |
| (2,381,124 | ) | |
| (856,260 | ) |
| Net | |
$ | — | | |
$ | — | |
The
income tax provision differs from the expense that would result from applying federal statutory rates to income before income taxes because
the Company is subject to state income taxes, deferred income taxes are based on average tax rates and a portion of gifts and meals and
entertainment are not tax deductible. In addition, the foregiveness of PPP loan of $296,826
is not taxable income.
Prior
to the share exchange, Munn Works, LLC was taxed as a single member Limited Liability Company for federal and state income tax purposes.
As such, the Company will not pay income taxes for earnings prior to the share exchange, as any income or loss will be included in the
tax returns of the individual member. Accordingly, no provision is made for income taxes in the financial statements prior to the share
exchange.
Effective
tax rates differ from the federal statutory rate of 21%
applied to income before provision for income taxes due to the following:
| Schedule
of income before provision for income taxes | |
| | | |
| | |
| | |
2021 | |
2020 |
| Federal
statutory rate times financial statement income | |
$ | (1,552,673 | ) | |
$ | (677,930 | ) |
| Permanent
tax basis differences | |
| (72,997 | ) | |
| 613 | |
| Deferred
true up | |
| 13,911 | | |
| 38,048 | |
| State
taxable income, net | |
| (86,700 | ) | |
| (205,098 | ) |
| Change
in Valuation allowance | |
| 1,524,637 | | |
| 856,520 | |
| Rate
change | |
| 154,179 | | |
| | |
| True
up | |
| (91,819 | ) | |
| 54,258 | |
| Other | |
| 19,643 | | |
| 443 | |
| Total
provision for income taxes | |
$ | (91,819 | ) | |
$ | 66,854 | |
The
Company has available net operating loss approximately $9,054,000
for tax purposes to offset future taxable income
. Pursuant to the Tax Reform Act of 1986, annual utilization of the Company’s net operating loss and contribution carryforwards
may be limited if a cumulative change in ownership of more than 50%
is deemed to occur within any three-year period. The tax years 2018 through 2020 remain open to examination by federal agencies and other
jurisdictions in which it operates.
NOTE
13- NOTE RECEIVABLE- RELATED PARTY
The
company contemplated an acquisition with an entity where certain board members of the Company were also board members of the potential
acquiree. In February of 2021, the Company entered into a non-interest bearing note receivable agreement whereby the Company loaned $500,000 to
the entity. The note receivable was recorded at cost basis which approximates fair value because of the short-term maturity of the instrument.
The loan matures on the earlier of (i) 180 days from the issuance date or (ii) the closing of the transactions set forth in a definitive
acquisition entered into between the lender and the borrower. In the event the loan is paid in full on or before the maturity date, there
shall be no interest accrued or payable on the outstanding principal amount. If an acquisition occurs, the $500,000 will
be applied against the total acquisition price. If the company decides not to execute a definitive agreement within 180 days from the
issuance date, the maturity date shall be the one-year anniversary of the issuance date. The maturity date has since been extended to
November 30, 2021. The acquisition did not occur and the full amount of $500,000
was repaid on November 30, 2021.
NOTE
14 - SEGMENT REPORTING
FASB
Codification Topic 280, Segment Reporting, establishes standards for reporting financial and descriptive information about an enterprise’s
reportable segments. The Company has two reportable segments: the design, manufacture, assembly and distribution of disinfecting systems
for use in healthcare, hospitality, and commercial municipal and residential markets (disinfectant segment) and the manufacture of fine
mirrors specifically for the hospitality industry (hospitality segment). The segments are determined based on several factors, including
the nature of products and services, the nature of production processes, customer base, delivery channels and similar economic characteristics.
An
operating segment’s performance is evaluated based on its pre-tax operating contribution, or segment income. Segment income is
defined as net sales less cost of sales, segment selling, general and administrative expenses, research and development costs and stock-based
compensation. It does not include other charges (income), net and interest and other, net.
Applied
UV, Inc. and Subsidiaries
Notes
to the Consolidated Financial Statements
NOTE
14 - SEGMENT REPORTING (continued)
| SEGMENT
REPORTING | |
| | | |
| | | |
| | | |
| | |
| | |
Hospitality | |
Disinfectant | |
Corporate | |
Total |
| Balance
sheet at December 31, 2021 | |
| | | |
| | | |
| | | |
| | |
| Assets | |
$ | 2,158,789 | | |
$ | 27,851,691 | | |
$ | 8,515,512 | | |
$ | 38,525,992 | |
| Liabilities | |
$ | 2,481,186 | | |
$ | 1,528,706 | | |
$ | 1,850,366 | | |
$ | 5,860,233 | |
Balance
sheet at December 31, 2020 | |
| | | |
| | | |
| | | |
| | |
| Assets | |
$ | 12,655,779 | | |
$ | 463,042 | | |
$ | — | | |
$ | 13,118,821 | |
| Liabilities | |
$ | 2,721,396 | | |
$ | 642,669 | | |
$ | — | | |
$ | 3,364,065 | |
| | |
Hospitality | |
Disinfectant | |
Corporate | |
Total |
| Income
Statement for the year ended December 31, 2021 | |
| | | |
| | | |
| | | |
| | |
| Net
Sales | |
$ | 5,943,664 | | |
$ | 5,723,915 | | |
$ | — | | |
$ | 11,667,579 | |
| Cost
of Goods Sold | |
$ | 4,488,652 | | |
$ | 3,080,541 | | |
$ | — | | |
$ | 7,569,193 | |
| Research
and development | |
$ | — | | |
$ | 53,408 | | |
$ | — | | |
$ | 53,408 | |
| Selling,
General and Administrative expenses | |
$ | 2,281,460 | | |
$ | 6,110,206 | | |
$ | 2,950,046 | | |
$ | 11,341,712 | |
| | |
Hospitality | |
Disinfectant | |
Corporate | |
Total |
| Income
Statement for the year ended December 31, 2020 | |
| | | |
| | | |
| | | |
| | |
| Net
Sales | |
$ | 5,732,734 | | |
$ | — | | |
$ | — | | |
$ | 5,732,734 | |
| Cost
of Goods Sold | |
$ | 4,723,398 | | |
$ | — | | |
$ | — | | |
$ | 4,723,398 | |
| Research
and development | |
$ | — | | |
$ | 310,672 | | |
$ | — | | |
$ | 310,672 | |
| Selling,
General and Administrative expenses | |
$ | 1,749,962 | | |
$ | 770,512 | | |
$ | 1,491,082 | | |
$ | 4,011,556 | |
NOTE
15 – PROFORMA FINANCIAL STATEMENTS (UNAUDITED)
Unaudited
Supplemental Pro Forma Data
Unaudited
pro forma results of operations for the years ended December 31, 2021 and 2020 as though the company acquired Akida, KES and SciAir (the
“Acquired Companies”) on January 1, 2020 is set forth below.
| Business
Acquisition, Pro Forma Information | |
| | | |
| | |
| | |
Year
Ended December 31, 2021 | |
Year
Ended December 31, 2020 |
| Net
Sales | |
$ | 19,169,612 | | |
$ | 23,297,450 | |
| Cost
of Goods Sold | |
| 10,619,220 | | |
| 12,425,826 | |
| Gross
Profit | |
| 8,550,392 | | |
| 10,871,624 | |
| | |
| | | |
| | |
| Research
and development | |
| 53,408 | | |
| 310,672 | |
| Selling,
General and Administrative Expenses | |
| 17,198,118 | | |
| 10,527,740 | |
| Total
Operating expenses | |
| 17,251,526 | | |
| 10,838,412 | |
| Operating
Loss | |
| (8,701,134 | ) | |
| 33,212 | |
| Other
(Expense) Income | |
| | | |
| | |
| Change
in Fair Market Value of Warrant Liability | |
| 66,862 | | |
| — | |
| Foregiveness
of paycheck protection program loan | |
| 296,827 | | |
| — | |
| Other
income | |
| (74,317 | ) | |
| 3,719 | |
| Total
Other Income | |
| 289,372 | | |
| 3,719 | |
| Loss
Before Provision for Income Taxes | |
| (8,411,762 | ) | |
| 36,961 | |
| Benefit
from Income Taxes | |
| (91,819 | ) | |
| 83,601 | |
| Net
Loss | |
$ | (8,319,943 | ) | |
$ | (46,670 | ) |
| | |
| | | |
| | |
| Net
Loss attributable to common stockholders: | |
| | | |
| | |
| Dividends
to preferred shareholders | |
| (603,750 | ) | |
| — | |
| Net
Loss attributable to common stockholders | |
| (8,923,693 | ) | |
| (46,670 | ) |
| Basic
and Diluted Loss Per Common Share | |
$ | (0.91 | ) | |
$ | (0.01 | ) |
| | |
| | | |
| | |
| Weighted
Average Shares Outstanding- basic and diluted | |
| 9,799,627 | | |
| 7,511,385 | |
NOTE
16 – SUBSEQUENT EVENT
The
Acquisition
On
March 25, 2022, MunnWorks, LLC (“Purchaser”), a New York limited liability company and a wholly-owned subsidiary of Applied
UV, Inc. (the “Company”) entered into an asset purchase agreement (the “APA”) with VisionMark, LLC (“Seller”),
Maya Systems, LLC d/b/a Benchmark Furniture MFG (“Maya”), Mega Vision, Inc. (“MV” and, together with Maya, the
“Members”), Sandy Marks (“Marks”) and Michael Chiriac, Sr. (“Chiriac” and, together with Marks, the
“Key Persons”). Pursuant to the APA, Purchaser acquired substantially all of the assets of Seller (the “Business”),
including raw materials, inventory and work-in-progress, and assumed certain limited obligations of Seller, as set forth in the APA (the
“Acquisition”).
On
March 25, 2022 (the “Closing Date”), the Acquisition was completed.
On
the Closing Date, Seller received, as consideration for the Acquisition, the purchase price consisting of: (i) $10 in cash; and (ii)
the agreement by Purchaser to assume approximately $1.2 million in past due rent owing by Seller (the “Past Due Rent”) under
that certain lease agreement by and between MV and the landlord thereunder (the “Prime Lease”), payable in equal monthly
installments spread over thirty six (36) months, as set forth in the Sublease, dated as of March 29, 2022 among the Purchaser, the Seller
and Randolph Associates (the “Sublease”).
Sales
Commission Payments
Pursuant
to the APA, Purchaser agreed to make certain additional payments to Seller in the form of a sales commission during the four (4) year
period following the Closing Date (the “Sales Commission Payments”), as follows: (i) during the first two (2) years following
the Closing Date (the “Initial Period”), Purchaser will pay to Seller, on a quarterly basis, an amount equal to 2.5% of certain
net sales generated by the Business; and (ii) during the two (2) years following the Initial Period, Purchaser will pay to Seller, on
a quarterly basis, an amount equal to 5% of certain net sales generated the Business. Sales that qualify for the Sales Commission Payments
will be limited to (i) sales made to new clients generated by the Members or the Key Persons, and (ii) sales to certain existing clients
of Seller specified under the APA; provided that Sales Commission Payments related to such existing clients are conditioned on the Members
and the Key Persons providing support to Purchaser in the transition and maintenance of those existing clients as described in the APA.
Sublease
and Sublease Guaranty
Purchaser
has agreed to sublease all of Seller’s right, title and interest in the Prime Lease, subject to the terms of the Sublease, which
include: (i) an initial term expiring June 30, 2023, with two (2) successive 1-year renewal options to be exercised at Purchaser’s
discretion; (ii) Purchaser’s obligation to pay base rent, as defined in the Prime Lease, directly to the landlord each month; (iii)
Purchaser’s obligation to pay Past Due Rent in equal monthly payments spread over thirty six (36) months. However, if the Purchaser
does not renew the Sublease, its aggregate obligation to pay Past Due Rent shall be decreased to $600,000.
Additionally,
the Company has agreed to guarantee the obligations of the Purchaser under the Sublease pursuant to a Guaranty of Sublease dated as of
March 29, 2022 made by the Company in favor of the Seller and Randolph Associates (the “Sublease Guaranty”). Under the terms
of the Sublease Guaranty, the Company will guarantee the Purchaser’s obligations under the Sublease, including base rent payable
during the term of the Sublease and the Purchaser’s obligations to pay Past Due Rent.
Settlement
Agreement
On
March 31, 2022 we entered into a Settlement Agreement (the “Settlement Agreement”) with Old SAM, LLC and the members thereof
who exected the Settlement Agreement (collectively, the “Old SAM Parties”), pursuant to which we settled a dispute with Old
SAM Parties. Under the terms of the Settlement Agreement the Old SAM Parties relinquished all of their right, title and interest in any
of the 200,000 vested shares and 200,000 unvested shares issued to them in connection with the acquisition of Sci Air and no longer have
the right to any additional cash consideration if the avegage price of our stock is below $10.00 on certain measurement dates described
in the Sci Air acquisition agreement. The Settlement Agreement also contains a mutual release of all claims we and the Old SAM parties
have against each other, other than claims related to certain consulting agreements executed in connection with acquisition of Sci Air.
We will review for potential impairment impact as of and for the period ending March 31, 2022.
Item
9. Changes in and Disagreements With Accountants on Accounting and Financial Disclosures
None.
Item
9A. Controls and Procedures
Disclosure
Controls and Procedures
The
Company’s management, with the participation of the Company’s Chief Executive Officer and Chief Financial Officer, have evaluated
the effectiveness of the Company’s disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Securities
Exchange Act of 1934, as amended (the “Exchange Act”)) as of December 31, 2021. Based on that evaluation, the Company’s
Chief Executive Officer and the Company’s Chief Financial Officer have concluded that as of December 31, 2021, due to the existence
of the material weakness in the Company’s internal control over financial reporting described below, the Company’s disclosure
controls and procedures were not effective.
Evaluation
of Disclosure Controls and Procedures
Our
Chief Financial Officer is responsible for establishing and maintaining adequate internal control over financial reporting. Internal
control over financial reporting is defined in Rules 13a-15(f) and 15d-15(f) promulgated under the Exchange Act as a process designed
by, or under the supervision of, our principal executive and principal financial officers, or persons performing similar functions, and
effected by our Board, senior management and other personnel, to provide reasonable assurance regarding the reliability of financial
reporting and the preparation of financial statements for external purposes in accordance with U.S. GAAP.
Because
of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of
any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions,
or that the degree of compliance with the policies or procedures may deteriorate. We continue to review our internal control over financial
reporting and may from time to time make changes aimed at enhancing their effectiveness and to ensure that our systems evolve with our
business.
Under
the supervision and with the participation of management, including the interim Chief Executive Officer and Chief Financial Officer,
we conducted an evaluation of the effectiveness of our internal control over financial reporting based on the framework in “Internal
Control — Integrated Framework (2013)” issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO).
Based on the control deficiencies identified during this evaluation and set forth below, our senior management has concluded that we
did not maintain effective internal control over financial reporting as of December 31, 2021 due to the existence of a material weakness
in internal control over financial reporting as described below.
As
set forth below, management will continue to take steps to remediate the control deficiencies identified below. Notwithstanding the control
deficiencies described below, we have performed additional analyses and other procedures to enable management to conclude that our consolidated
financial statements included in this Form 10-K fairly present, in all material respects, our financial condition and results of operations
as of and for the year ended December 31, 2021.
A
material weakness is a deficiency, or a combination of deficiencies, in internal controls over financial reporting, such that there is
a reasonable possibility that a material misstatement of the Company’s annual or interim financial statements will not be prevented
or detected on a timely basis.
While
preparing its annual report for the year ended December 31, 2020 the Company identified an error in the timing of recognizing certain
revenues that effected the previously filed consolidated financial statements as of and for the years ended December 31, 2019 and 2018.
The error also impacted the previously reported financial statements for the quarters ended September 30, 2020, June 30, 2020 and March
31, 2020. The impact in those periods has been disclosed in the September 30, 2021, June 30, 2021 and March 31, 2021 10-Q filings.
In
connection with the preparation of the consolidated financial statements for the three months ended March 31, 2021, the Company recognized
an error relating to the recognition of the initial warrant liability in November of 2020. The error caused additional paid in capital
to be overstated by approximately $135,000, warrant liability to be understated by approximately $110,000, and net loss to be overstated
by approximately $25,000 as of and for the year ended December 31, 2020. The Company concluded the impact on the interim and year ended
December 31, 2020 financial statements was immaterial and corrected the balances as of March 31, 2021,
As
part of such process, our management and audit committee determined that our disclosure controls and procedures for the Affected Periods
were not effective and that the foregoing arose as a result of a material weakness in our internal control over financial reporting.
The
Company’s management has developed a remediation plan to address the material weakness and as of January 1, 2021 began using a
new cloud-based software which tracks the progress of jobs and more accurately reflects the percentage of job completeness ensure such
revenue is recognized in the appropriate period. In addition, the Company intends to further remediate the deficiency by performing the
following:
| • |
|
design
and implement additional internal controls and policies to ensure that we routinely review and document our application of established
significant accounting policies; and |
| • |
|
implement
additional systems and technologies to enhance the timeliness and reliability of financial data within the organization. |
| • |
|
continue
to engage third-party subject matter experts to aid in identifying and applying US GAAP rules related to complex financial instruments
as well as to enhance the financial reporting function. |
Limitations
on Effectiveness of Controls and Procedures
In
designing and evaluating the disclosure controls and procedures and internal control over financial reporting, management recognizes
that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired
control objectives. In addition, the design of disclosure controls and procedures and internal control over financial reporting must
reflect the fact that there are resource constraints and that management is required to apply judgment in evaluating the benefits of
possible controls and procedures relative to their costs.
Changes
in Internal Controls over Financial Reporting
No
change in the Company’s internal controls over financial reporting (as defined in Rule 13a-15(f) and 15d-15(f) of the Exchange
Act) occurred during the Company’s most recent fiscal quarter that has materially affected, or is reasonably likely to materially
affect, our internal control over financial reporting.
Item
9B. Other Information.
None.
PART
III
Item
10. Directors, Executive Officers and Corporate Governance
The following
are our executive officers and directors and their respective ages and positions as of April 7, 2022.
| Name |
|
Age |
|
Position |
| Max
Munn |
|
78 |
|
President,
Acting Chief Executive Officer and Director |
| John
J Hayman |
|
51 |
|
Senior
Vice President, Interim Chief Operations Officer |
| Michael
Riccio |
|
63 |
|
Chief
Financial Officer |
| Joel
Kanter |
|
65 |
|
Chairman
of the Board |
| Dr.
Eugene A. Bauer, M.D. |
|
79 |
|
Director |
| Dr.
Alastair J. Clemow, Ph.D. |
|
70 |
|
Director |
| Dallas
C. Hack, M.D |
|
69 |
|
Director |
| Eugene
E. Burleson |
|
81 |
|
Director |
| John
F. Andrews1 |
|
68 |
|
Director |
1
Mr. Andrews executed an employment agreement with the Company that appoints him as Chief Executive Officer, effective April 11,
2022
Max
Munn is the President, Acting Chief Executive Officer and a director of the Company and is also the Chief Executive Officer of Munn
Works, LLC, our subsidiary focused on the hospitality market. Mr. Munn has held this position at Munn Works for over 20 years. Mr. Munn
is also Co-chairman of Dieu Donne Inc., a not-for-profit and a leading, world recognized atelier wherein dimensional, handmade paper
is utilized in the making of art. Mr. Munn attended MIT from 1961-1966, majored in chemistry and architecture; and received a Bachelor’s
of Architecture degree. Mr. Munn also attended Columbia University for post graduate studies from 1966-1968, working toward a Ph.D. in
architectural history.
John
J. Hayman III, is our Senior Vice President and Interim Chief Operations Officer. Mr. Hayman has over 28 years’ experience
in general management, operations, marketing, sales and consulting. From January 2003 to September 2021, Mr. Hayman was the President
and Chief Executive Officer of KES Science & Technology, Inc. From September 2021 to December 2021, he was the Senior Vice President
of Product Development for SteriLumen, Inc., a wholly-owned subsidiary of the Company. Mr. Hayman received a BBA from the University
of Georgia in 1992. Mr. Hayman was a Certified Public Accountant and worked for KPMG Peat Marwick as a Staff/Senior Accountant for two
years.
Michael
Riccio is our Chief Financial Officer. Mr. Riccio joins Applied UV with more than 25 years of broad experience in leadership
roles in corporate and operational finance, including corporate merger and acquisition planning and integration. From September 1986
to March 2021 Mr. Riccio was Chief Financial Officer and Treasurer of Panasonic Corporation of North America. At Panasonic Mr. Riccio
lead the finance organization that supports the principal North American subsidiary of Osaka, Japan-based Panasonic Corporation. Prior
to Panasonic, Mr. Riccio was the Corporate Accounting Manager for Sealed Air Corporation (SEE on NYSE), and prior to Sealed Air he began
his career at CohnReznick, where he was a Senior Auditor. Mr. Riccio is a Certified Public Accountant (CPA) and holds a B.A. in accounting
from Rutgers University and an MBA in finance from Rutgers Business School. Mr. Riccio is a member of the American Institute of Certified
Public Accountants and the New Jersey Society of Certified Public Accountants.
Joel
Kanter is the Chairman of the Board of the Company. Mr. Kanter has served as President of Windy City, Inc., a privately held investment
firm, since July 1988. From 1989 to November 1999, Mr. Kanter served as the President, and subsequently as the President and Chief Executive
Officer of Walnut Financial Services, Inc., a publicly traded company (Nasdaq: WNUT). Walnut Financial’s primary business focus
was the provision of various forms of financing to small businesses including equity financing to start-up and early stage development
companies, bridge financing to small and medium-sized companies, and later stage institutional financing to mature enterprises. Tower
Hill Capital Group bought the Company in 1999 in a transaction valued at approximately $400 million. From 1978 - 1980, Mr. Kanter served
as a Legislative Assistant to former Congressman Abner J. Mikva (D-Ill.). In that position, Mr. Kanter provided support to Congressman
Mikva with respect to activities related to his position on the House Judiciary Committee. In particular, Mr. Kanter was intimately involved
in efforts to reform the Federal Criminal Code. Mikva subsequently became the Chief Judge of the U.S. Court of Appeals for the District
of Columbia Circuit, and then served as White House Counsel to President Clinton. From 1980 - 1983, Mr. Kanter served as Special Assistant
to the National Association of Attorneys General. In that position, he represented the interests of the State Attorneys General in Washington,
D.C. in the criminal justice and environmental arenas. Mr. Kanter serves on the Board of Directors of one currently public company, Magna-Labs,
Inc., which was formerly involved in the development of a cardiac MRI device. Mr. Kanter also serves on the boards of several private
life sciences and medical device companies. Mr. Kanter is a National Association of Corporate Directors “Governance Fellow.”
Mr. Kanter is a current Trustee Emeritus and past President of the Board of Trustees of The Langley School in McLean, Virginia, a former
Trustee at the Georgetown Day School in Washington, D.C., and a former Trustee of the Union Institute & University, the Country’s
first Online University, and currently serves on the Board of the School of Science and Engineering at Tulane University.
Eugene
A. Bauer, M.D. is a Director of the Company. Dr. Bauer is currently a director of Evommune, Inc., a private research and development
company and innovation engine in chronic inflammation, First Wave Technologies, Inc., a private company focusing on a next-generation
anesthesia system for the use of inhaled anesthetics, and Biolojic Design, Ltd., a private biotechnology company that computationally
designs functional antibodies. In 2010, Dr. Bauer co-founded Dermira, a publicly traded specialty biopharmaceutical company acquired
by Eli Lilly and Company in 2020. Dr. Bauer served as Chief Medical Officer of Dermira, a wholly-owned subsidiary of Lilly, through March
2020. Prior to founding Dermira, Dr. Bauer served as Director, President and Chief Medical Officer of Pelpin, Inc., a publicly traded
specialty pharmaceutical company, from 2008 to 2010. Dr. Bauer served as Chief Executive Officer of Neosil, Inc., a specialty pharmaceutical
company, from 2004 to 2008, and he co-founded and served as a member of the board of directors at Connetics, a publicly traded specialty
pharmaceutical company, from 1990 to 2006. Prior to initiating his career in industry, Dr. Bauer served as Dean of Stanford University
School of Medicine from 1995 to 2001 and as Chair of the Department of Dermatology at Stanford University School of Medicine from 1988
to 1995. Dr. Bauer is Professor Emeritus at Stanford University School of Medicine, a position he has held since 2002. Dr. Bauer was
a U.S. National Institutes of Health (“NIH”)-funded investigator for 25 years and has served on review groups and Councils
for the NIH. Dr. Bauer has served on the boards of directors of a number of public and private companies, including Aevi Genomic Medicine,
Inc. (formerly Medgenics, Inc.), First Wave Technologies, Inc., Cerecor, Inc. and Kadmon Holdings, Inc. He is member of numerous honorific
societies, including the National Academy of Medicine. Dr. Bauer received his B.S. in medicine from Northwestern University and his M.D.
from Northwestern University Medical School.
Alastair
J. Clemow, Ph.D. is a Director of the Company. Dr. Clemow currently serves as Chairman of Ensemble Orthopedics, an early-stage company
selling an innovative pyro carbon finger joint for the treatment for early state osteoarthritis. He is also currently an independent
director and Chairman of the Audit Committee of First Wave Technologies, Inc., a private company focusing on a next-generation anesthesia
system for the use of inhaled anesthetics, and an independent director and Chair of the Strategic Committee of GreenBone Spa, a private
Italian company focusing on bone regeneration using natural materials. From 2011 to 2019, Dr. Clemow served as President and Chief Executive
Officer of Regentis Biomaterials, a private company developing an innovative material for cartilage repair. He also held positions of
president and chief executive officer in a number of companies that he helped found, including Nexgen Spine, Inc., which developed an
artificial spinal disc; Gelifex, Inc., which developed an innovative spinal nucleus replacement implant and which was acquired in 2004
by Synthes Spine, Inc.; and Minimally Invasive Surgical Technologies, Inc., which developed a novel series of implants for minimally
invasive total knee replacement and which was acquired in 2005 by MAKO Surgical Corp. From 2000 to 2004, Dr. Clemow served as Principal
of Tanton Technologies, Inc., an organization that provided strategic and technical assessment of new medical device opportunities for
large, mid-cap, and early-stage development companies. From 1981 to 2000, Dr. Clemow held numerous positions with Johnson & Johnson,
including Vice President of Worldwide Business Development for Ethicon Endo-Surgery, Inc.; Vice President of New Business Development
for Johnson & Johnson Professional, Inc.; and Director of Research and Development of Johnson & Johnson Orthopedics. In those
capacities, Dr. Clemow was responsible for acquiring or developing what today represents billions of dollars of Johnson & Johnson
revenue. Dr. Clemow serves or has served on the boards of numerous private and public companies including Aevi Genomics; Encore Medical;
Echo Healthcare Acquisition Corp.; BioMedical Enterprises, Inc.; and Kinetic Muscles, Inc. He graduated from the University of Surrey
with a Bachelor of Science Degree in metallurgy and Ph.D. in metallurgy and earned his MBA degree from Columbia University in New York.
Dallas
C. Hack, M.D., M.P.H. is a Director of the Company. He is board certified in General Preventive Medicine and Public Health and a
Fellow in the American College of Military Public Health. Since July 2018, he has been the Chief Medical Officer of Virtech Bio, Inc.,
a private company that sells a ready-to-use oxygen-carrying plasma expander to restore circulatory system parameters and since 2015 has
been a consultant to numerous biotech and non-profit companies to advance medical research and transition the progress to improved clinical
practice. From June 2018 to March 2020, he was a director, member of the Audit and Product Development Committees and the interim Chief
Executive Officer and Chief Financial Officer of CVR Medical Corp. (OTC: CRRVF), a publicly traded med-tech company dedicated to the
development and advancement of revolutionary technology at work within the healthcare sector. He directed trauma research for the military
from 2008 to 2014, with responsibility for more than $2 billion in grant funding. He held numerous military medical leadership positions
including Commander of the NATO Headquarters Healthcare Facility, and Command Surgeon at the strategic level during Operations Enduring
Freedom and Iraqi Freedom. COL.(R) Hack, who served for 28 years, received numerous military awards including the Bronze Star, two Legion
of Merit awards, and was inducted as a Distinguished Member of the Military Order of Medical Merit. He has a BA from Andrews University
(1972), an MPH from Johns Hopkins University (1995), a MD from Loma Linda University (1976), an MSS from the US Army War College (2004),
and a CPE from the Certifying Commission in Medical Management (1997). He was recognized as the Distinguished Alumnus of the Year by
Loma Linda University in May 2015. Dr. Hack has an appointment from the School of Medicine, University of Pittsburgh as Adjunct Professor
of Neurosurgery and from Virginia Commonwealth University as an Associate Clinical Professor, Department of Physical Medicine and Rehabilitation.
Eugene
E. Burleson is a Director of the Company. He is a private investor, currently a director and Chairman of the Compensation Committee
of Sunlink Health Systems, Inc. (NYSE American: SSY), an investor in healthcare facilities and related businesses located in the Southeast,
and a director and the Chairman of the Compensation Committee of HealthNow New York, a private health insurance company. He was Chairman
of PET DRx Corporation from June 2005 to July 1, 2010, and its Chief Executive Officer from October 2008 until its acquisition by VCA
Antech in July 2010. Mr. Burleson was a director of HealthMont Inc. from September 2000 until its acquisition by SunLink in October 2003.
Mr. Burleson served as Chairman of the Board of Directors of Mariner Post-Acute Network, Inc. from January 2000 to June 2002. Mr. Burleson
also served as Chairman of the Board of Directors of Alterra Healthcare Inc. a developer and operator of assisted living facilities and
is on the Board of Deckers Outdoor Corporation Inc. Mr. Burleson served as Chairman of the Board and Chief Executive Officer of GranCare
Inc. from October 1988 to November 2000. Additionally, Mr. Burleson served as President and Chief Executive Officer of GranCare Inc.
from December 1990 to February 1997. Upon completion of the merger of GranCare’s pharmacy operations with Vitalink Pharmacy Services
Inc. in February 1997, he became Chief Executive Officer and a Director of Vitalink Pharmacy Services Inc. Mr. Burleson resigned as Chief
Executive Officer and Director of Vitalink Pharmacy Services in August 1997. From June 1986 to March 1989, Mr. Burleson served as President,
Chief Operating Officer and a Director of American Medical International Inc. (“AMI”), an owner and operator of acute
care hospitals. Based in London from May 1981 to June 1986, Mr. Burleson served as Managing Director of AMI’s international operations.
Mr. Burleson received his early management training at Eastman Kodak from 1963 to 1974. He graduated from East Tennessee State University
with a Bachelor of Science Degree in accounting and earned an MBA degree in 1972.
John F. Andrews has over 30 years of senior
leadership experience in telecom and technology companies, both public and private. From 2021 to April of 2022 Mr. Andrews served as Chief
Executive Officer of Trinity IT Services LLC, which is an IT services company in Jacksonville Florida. At Trinity IT Mr. Andrews successfully
launched the company, signed contracts with the State of Florida, Fiserv, Mastercard, Point72, Credit Agricole and Tech Mahindra. From
2020 to April 2022 Mr. Andrews was the Chief Executive Officer and Director of TrekSecure LLC, a SAAS based contagion response platform
firm where he finalized development of V1 of the Contagion Response Platform, established go to market plans for Government and Travel
and developed a sales pipeline exceeding $100 million. From Mr. Andrews 2018 to 2020 Mr. Andrews was the Chief Technology Officer and
Co-founder of KyckGlobal Inc., an IT firm based in Atlanta, Georgia. At KyckGlobal Mr. Andrews was responsible for all technology matters
and developed the on-demand payment processing product plan and developed the SAAS based multi-tenant platform within schedule and budget.
Mr. Andrews also functioned as a key principal in securing $5 million in venture capital and $3.5 million in private capital. From 2017
to 2019 Mr. Andrews was the Chief Operating Officer for RiseIT Inc., an $80 million IT software and services provider in Jacksonville,
Florida. At RiseIT Mr. Andrews had P&L responsibility, functioned as a key principal in securing $4 million in private capital, assimilated
three acquisitions totaling $75 million in revenue and developed integrated operating plan. Mr. Andrews has an MBA from the University
of Puget-Sound, Seattle, Washington, and a BA, Business Administration and Finance from Whitworth University, Spokane, Washington.
We believe that Mr. Andrews’ extensive business
experience will contribute to the Board’s business strategy and planning on a going-forward basis.
Board
of Directors
Our
business and affairs are managed under the direction of our Board. Our Board consists of six directors, five of whom qualify as “independent”
under the listing standards of Nasdaq.
Directors
serve until the next annual meeting and until their successors are elected and qualified. Officers are appointed to serve for one year
until the meeting of the Board following the annual meeting of shareholders and until their successors have been elected and qualified.
Board
Leadership Structure and Risk Oversight
The
Board oversees our business and considers the risks associated with our business strategy and decisions. The Board currently implements
its risk oversight function as a whole. Each of the Board committees, as set forth below, will also provide risk oversight in respect
of its areas of concentration and reports material risks to the board for further consideration.
Director
Independence
Our
board of directors are composed of a majority of “independent directors” as defined under the rules of Nasdaq. Nasdaq Listing
Rule 5605(a)(2) provides that an “independent director” is a person other than an officer or employee of the company
or any other individual having a relationship which, in the opinion of the Company’s Board, would interfere with the exercise of
independent judgment in carrying out the responsibilities of a director.
Under
such definition, our Board has undertaken a review of the independence of each director. Based on information provided by each director
concerning his or her background, employment and affiliations, our Board has determined that Joel Kanter, Dr. Eugene A. Bauer, Dr. Alastair
Clemow, Dr. Dallas Hack and Eugene Burleson are all independent directors of the Company.
Board
Committees
Our
board of directors has established three standing committees: audit committee, compensation committee and nominating and corporate governance
committee, each of which operate under a charter that has been approved by our board of directors. We have appointed persons to the board
of directors and committees of the board of directors as required meeting the corporate governance requirements of the Nasdaq Listing
Rules.
Audit
Committee
We
have established an audit committee consisting of Eugene Burleson, Alastair Clemow and Dallas Hack. Eugene Burleson is the Chairman of
the audit committee. In addition, our Board has determined that Eugene Burleson is an audit committee financial expert within the meaning
of Item 407(d) of Regulation S-K under the Securities Act of 1933, as amended, or the Securities Act. The audit committee’s
duties, which are specified in our Audit Committee Charter, include, but are not limited to:
| • |
|
reviewing
and discussing with management and the independent auditor the annual audited financial statements, and recommending to the board
whether the audited financial statements should be included in our annual disclosure report; |
| • |
|
discussing
with management and the independent auditor significant financial reporting issues and judgments made in connection with the preparation
of our financial statements; |
| • |
|
discussing
with management major risk assessment and risk management policies; |
| • |
|
monitoring
the independence of the independent auditor; |
| • |
|
verifying
the rotation of the lead (or coordinating) audit partner having primary responsibility for the audit and the audit partner responsible
for reviewing the audit as required by law; |
| • |
|
reviewing
and approving all related-party transactions; |
| • |
|
inquiring
and discussing with management our compliance with applicable laws and regulations; |
| • |
|
pre-approving
all audit services and permitted non-audit services to be performed by our independent auditor, including the fees and terms of the
services to be performed; |
| • |
|
appointing
or replacing the independent auditor; |
| • |
|
determining
the compensation and oversight of the work of the independent auditor (including resolution of disagreements between management and
the independent auditor regarding financial reporting) for the purpose of preparing or issuing an audit report or related work; |
| • |
|
establishing
procedures for the receipt, retention and treatment of complaints received by us regarding accounting, internal accounting controls
or reports which raise material issues regarding our financial statements or accounting policies; and |
| • |
|
approving
reimbursement of expenses incurred by our management team in identifying potential target businesses. |
The
audit committee is composed exclusively of “independent directors” who are “financially literate” as defined
under the Nasdaq listing standards. The Nasdaq listing standards define “financially literate” as being able to read and
understand fundamental financial statements, including a company’s balance sheet, income statement and cash flow statement.
Compensation
Committee
We
have established a compensation committee of the board of directors to consist of Alastair Clemow, Eugene Bauer and Eugene Burleson,
each of whom is an independent director. Each member of our compensation committee is also a non-employee director, as defined
under Rule 16b-3 promulgated under the Exchange Act, and an outside director, as defined pursuant to Section 162(m) of
the Code. Alastair Clemow is the chairman of the compensation committee. The compensation committee’s duties, which are specified
in our Compensation Committee Charter, include, but are not limited to:
| • | | reviews,
approves and determines, or makes recommendations to our board of directors regarding, the
compensation of our executive officers; |
| • | | administers
our equity compensation plans; |
| • | | reviews
and approves, or makes recommendations to our board of directors, regarding incentive compensation
and equity compensation plans; and |
| • | | establishes
and reviews general policies relating to compensation and benefits of our employees. |
Nominating
and Corporate Governance Committee
We
have established a nominating and corporate governance committee consisting of Eugene Bauer, Eugene Burleson, Alastair Clemow, Dallas
Hack and Joel Kanter. Eugene Bauer is the Chairman of the audit Committee. The audit committee’s duties, which are specified in
our Nominating and Corporate Governance Audit Committee Charter, include, but are not limited to:
| • |
|
identifying,
reviewing and evaluating candidates to serve on our board of directors consistent with criteria approved by our board of directors; |
| • |
|
evaluating
director performance on our board of directors and applicable committees of our board of directors and determining whether continued
service on our board of directors is appropriate |
| • |
|
evaluating
nominations by stockholders of candidates for election to our board of directors; and |
| • |
|
corporate
governance matters |
Code
of Ethics
Our
Board has adopted a written code of business conduct and ethics (“Code”) that applies to our directors, officers and
employees, including our principal executive officer, principal financial officer and principal accounting officer or controller, or
persons performing similar functions. The Code is applicable to all of our directors, officers and employees and is available on our
corporate website, www.applieduvinc.com. We intend to disclose any amendments to our code of business conduct and ethics, or waivers
of its requirements, on our website or in filings under the Exchange Act to the extent required by applicable rules and exchange requirements.
Family
Relationships
There
are no family relationships among the officers and directors, nor are there any arrangements or understanding between any of the Directors
or Officers of our Company or any other person pursuant to which any Officer or Director was or is to be selected as an officer or director.
Involvement
in Certain Legal Proceedings
On
June 25, 2018, MunnWorks filed a voluntary petition for reorganization pursuant to Chapter 11 of the Bankruptcy Code in order to (i)
pursue its appeal of a $1.4 million New York State court judgment arising out of an employment and business dispute with a former employer
of Mr. Munn (of which Mr. Munn’s family-owned 50% of the equity) and (ii) preserve its ongoing operations from collection efforts
with respect to such judgment. MunnWorks settled the dispute by making a $400,000 cash payment and on June 28, 2019, concluded its Chapter
11 proceedings.
None
of our other directors, executive officers, significant employees or control persons have been involved in any legal proceeding listed
in Item 401(f) of Regulation S-K in the past 10 years.
Item
11. Executive Compensation
Summary
Compensation Table
The
following summary compensation table provides information regarding the compensation paid during our fiscal years ended December 31,
2021 and 2020 for our Chief Executive Officer (principal executive officer), our President, our Chief Financial Officer and our Chief
Operating Officer. We refer to these individuals as our “named executive officers.”:
| Name
and Principal Position |
|
Fiscal
Year Ended |
|
Salary
($) |
|
Stock
Awards ($) |
|
Option
Awards ($)(1) |
|
All
Other Compensation ($) |
|
Total
($) |
| Max
Munn |
|
|
2021 |
|
|
$ |
310,357 |
|
|
|
— |
|
|
$ |
1,567,449 |
(3) |
|
|
$25,510
|
(4) |
|
$ |
1,903,316 |
|
| President
and Interim Chief Executive Officer, President and Director(2) |
|
|
2020 |
|
|
|
--- |
|
|
|
— |
|
|
$ |
2,000 |
(5) |
|
$ |
94,396 |
(6) |
|
$ |
96,396 |
|
| Michael
Riccio |
|
|
2021 |
|
|
$ |
146,154 |
|
|
|
— |
|
|
$ |
297,711 |
(8) |
|
|
— |
|
|
$ |
443,865 |
|
| Senior
Vice President and Chief Financial Officer(7) |
|
|
2020 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Keyoumars
Saeed |
|
|
2021 |
|
|
$ |
331,154 |
|
|
|
— |
|
|
|
— |
|
|
$ |
353,846 |
|
|
$ |
685,000 |
|
| Former
Chief Executive Officer(9)(10) |
|
|
2020 |
|
|
$ |
110,577 |
|
|
$ |
446,540 |
|
|
|
— |
|
|
|
— |
|
|
$ |
557,117 |
|
| James
Doyle |
|
|
2021 |
|
|
$ |
272,885 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
$ |
272,885 |
|
| Former
Chief Operations Officer(11) |
|
|
2020 |
|
|
$ |
86,882 |
|
|
$ |
159,480 |
|
|
|
— |
|
|
|
— |
|
|
$ |
246,362 |
|
(1)
See Footnote 9 in the Notes to the Consolidated Financial Statements for a description of all of the assumptions made in the valuation
of these options.
(2)
Mr. Munn became the acting Chief Executive Officer upon the resignation of Mr. James Alexich on December 23, 2021.
(3)
Mr.Munn received options to purchase 309,835 shares of common stock at an exercise price of $7.80 per share. These options vest
at the end of each month for 36 months beginning April 1, 2021. These options were approved as a component of his March 4, 2021 employment
agreement.
(4)
Includes $25,510 in auto lease and auto insurance payments the Company provided to Mr. Munn.
(5)
Mr. Munn’s received options to purchase 1,000 shares of our common stock at an exercise price of $5.00 per share. The options
were granted and approved by the board during the second and third quarter of year ended 2020.
(6)
Mr.Munn also received warrants to purchase 80,000 share of restricted common stock at an exercise price of $5.00 per share in the
first quarter of 2020. See Footnote 9 in the Notes to the Consolidated Financial Statements for a description of all of the assumptions
made in the valuation of this warrant.
(7)
Mr. Riccio was hired as the Chief Financial Officer as of April 5, 2021.
(8)
Mr. Riccio received options to purchase 70,000 shares of common stock at an exercise price of $6.53.
(9)
Mr. Saeed’s compensation began on September 2, 2020 and ended upon the date of his resignation on December 7, 2021.
(10)
Mr. Saeed’s common stock award is valued at $5.00, which represents the aggregate grant date fair value computed in accordance
with FASB ASC Topic 718. Mr. Saeed resigned as of December 7, 2021.
(11)
Mr. Doyle’s common stock award is valued at $5.00, which represents the aggregate grant date fair value computed in accordance
with FASB ASC Topic 718. Mr. Doyle’s employment was terminated on December 22, 2021.
Employment
Agreements
Max
Munn. On March 4, 2021, we entered into an employment agreement with Mr. Munn regarding his employment as President of the Company,
reporting to the Board of Directors. The agreement was effective on that date and was for a three year term which automatically renews
for one year periods unless earlier terminated pursuant to the terms thereof. As base salary for the entire term, the Company paid Mr.
Munn a one-time payment $3.00 and granted to him Base Salary Options which have an exercise price equal to the closing price of our common
stock on the effective date and that vest monthly over the term at an even rate (1/36th per month), commencing on April 1,
2021. The Base Salary Options means options to purchase a number of shares of common stock equal to $944,997 divided by the Board Approved
Option Value which was determined to be $3.05. For each full fiscal year of the Company that begins and ends during the term of
the agreement, Mr. Munn is eligible to earn an annual cash bonus in such amount that will be determined by the Compensation Committee
of the Board based on the achievement by the Company reasonable performance goals established by the Compensation Committee and agreed
to by Mr. Munn for each such fiscal year; provided, that the annual bonus will be no greater than $500,000; and provided however in no
event will his annual bonus be less than the annual bonus received by the Chief Executive Officer of the Company. Mr. Munn is provided
an an allowance for an automobile and expenses to operate an automobile for business and personal use, is provided an allowance for insurance,
is reimbursed for Company travel and entertainment expenses, is entitled to four weeks of paid vacation per year and to participate in
all Company welfare benefits plans maintained for senior executive officers. The Company has agreed to indemnify Mr. Munn and cover him
under its directors’ and officers’ liability insurance policy. Mr. Munn has agreed not to compete with the Company for a
period of two years after termination of his employment in the areas where the Company has been operating, and not to interfere with
the Company’s suppliers or attempt to hire the Company’s employees.
Michael
Riccio. On February 19, 2021, we entered into an offer letter with Mr. Riccio regarding his employment as our Senior Vice
President and Chief Financial Officer. The offer letter has an effective date of April 5, 2021 and is for no specific period of time
permitting the Company may terminate his employment at any time and for any reason whatsoever, with or without “cause.” Mr.
Riccio’s annual base salary is $200,000.00 less applicable payroll deductions and withholdings, payable on the Company’s
regular payroll dates and he was awarded 70,000 “Stock Options” as defined in, and subject to, the Company’s 2020 Omnibus
Incentive Option Plan. We entered into an employment agreement with Mr. Riccio as of January 1, 2022 for a two year term, automatically
renewable for two years on each two year anniversary, pursuant to which we will pay him an annual salary of $300,000, subject to yearly
review by the Board of Directors. Mr. Riccio was granted 50,000 shares of restricted common stock and a ten year option to purchase 70,000
shares of our common stock at the fair market value on the date of grant, which restricted stock and options vest over a three-year period
and terminate 90 days after his separation from service to the Company. Mr. Riccio is also eligible for an annual discretionary performance
bonus of up to 100% of his base salary based on targets agreed by him and the Company’s Chief Executive Officer, which bonus must
be approved by the Audit and Compensation Committees provided that he is employed by the Company as of December 31 of each year. The
Company will reimburse him for Company business travel and he is entitled to paid holidays and leave as set forth in the Company’s
policies and to participate in all Company welfare benefits plans maintained for senior executive officers. Mr. Riccio has agreed to
maintain the confidentiality of Company information and for one year after the termination of his employment, he has agreed not to disparage
the Company, not to compete with the Company or to solicit the business of any of the Company’s customers.
Severance
Agreement
Effective
December 7, 2021, we executed a Separation Agreement and General Release with Mr. Saeed, our former Chief Executive Officer, pursuant
to which, upon his resignation, we agreed (i) pay him $300,000; (ii) permit the remaining unvested 14,883 shares of restricted stock
to vest immediately; and (iii) pay him $53,846 in unused vacation pay. The parties signed mutual releases and agreed not to disparage
the other. The Company agreed to have restrictive legends removed from his restricted shares. Mr. Saeed agreed to maintain the confidentiality
of Company confidential information and acknowledged his non-competition and non-solicitation obligations. On that date, the Company
engaged Mr. Saeed as an independent consultant for a specific project and terminated that agreement on January 2, 2022 with a one-time
payment of $10,000.
Outstanding
Equity Awards at Fiscal Year-End
The
following table sets forth information concerning outstanding equity awards held by the certain executives of the Company as of December
31, 2021:
| | |
Option Awards | |
Stock Awards | |
Equity
Incentive Plan Awards |
| | |
Number
of Securities Underlying Unexercised Options | |
| |
| |
| |
| |
| |
| |
|
| Name | |
Exercisable | |
Unexercisable | |
Equity
Incentive Plan Awards: Number of Securities Underlying Unexercised Options | |
Option
Exercise Price | |
Option
Expiration Date | |
Number
of Shares or Units of Stock That Have Not Vested | |
Market
Value of Shares or Units of Stock That Have Not Vested | |
Number
of Unearned Shares, Units of Stock, or Other Rights That Have Not Vested | |
Market
Value of Unearned Shares, Units of Stock, or Other Rights That HaveNot Vested |
| Keyoumars
Saeed (1) | |
| — | | |
| — | | |
| — | | |
$ | — | | |
| — | | |
| — | | |
| | | |
| — | | |
$ | — | |
| Max
Munn | |
| 77,459 | | |
| 232,376 | | |
| — | | |
$ | 7.80 | | |
| 3/4/2031 | | |
| — | | |
| — | | |
| — | | |
$ | — | |
| James
Doyle (2) | |
| — | | |
| — | | |
| — | | |
$ | — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | |
| Michael
Riccio(4) | |
| 28,962 | | |
| 41,038 | | |
| | | |
| 6.53 | | |
| 9/28/2031 | | |
| — | | |
| — | | |
| — | | |
| — | |
(1)
Mr. Saeed resigned as of December 7, 2021.
(2)
Mr. Doyle’s employment was terminated as of December 22, 2021.
(3)
Mr. Hayman was hired as the acting Chief Operations Officer as of December 22, 2021.
(4)
Mr. Riccio was hired as the Chief Financial Officer as of April 5, 2021.
Option
Exercises: No stock options were execised by the above named individuals in 2021.
Director
Compensation
The
annual compensation for each non-employee Director is summarized in the table below.
2021
Director Compensation Table
| Name | |
Fees
earned or paid in cash
($)(1) | |
Stock
Awards
($) | |
Option
Awards
($) | |
Total
($) |
| Joel
Kanter (2) | |
$ | 37,500 | | |
$ | 79,975 | | |
$ | 0 | | |
$ | 117,475 | |
| Alastair
J. Clemow (3) | |
$ | 37,500 | | |
$ | 57,125 | | |
$ | 0 | | |
$ | 94,625 | |
| Eugene
A. Bauer (4) | |
$ | 37,500 | | |
$ | 57,125 | | |
$ | 0 | | |
$ | 94,625 | |
| Eugene
Burleson (5) | |
$ | 37,500 | | |
$ | 57,125 | | |
$ | 0 | | |
$ | 94,625 | |
| Dallas
C Hack (6) | |
$ | 37,500 | | |
$ | 34,275 | | |
$ | 0 | | |
$ | 71,775 | |
(1)
Directors are paid $6,250 per quarter. There were two payments of $6,250 in 2021 for two quarters in 2020 that should have been
paid in 2020.
(2)
In 2021, Mr. Kanter was awarded 7,500 shares of restricted common stock that vest one year from the date of grant for his service
as a director and 10,000 shares of restricted common stock that vest one year from the date of grant for his service as the Chairman
of the Board.
(3)
In 2021, Mr. Clemow was awarded 7,500 shares of restricted common stock that vest one year from the date of grant for his service
as a director and 5,000 shares of restricted common stock that vest one year from the date of grant for his service as the Chairman of
the Compensation Committee.
(4)
In 2021, Mr. Bauer was awarded 7,500 shares of restricted common stock that vest one year from the date of grant for his service
as a director and 5,000 shares of restricted common stock that vest one year from the date of grant for his service as the Chairman of
the Nominating and Corporate Governance Committee.
(5)
In 2021, Mr. Burleson was awarded 7,500 shares of restricted common stock that vest one year from the date of grant for his service
as a director and 5,000 shares of restricted common stock that vest one year from the date of grant for his service as the Chairman of
the Audit Committee.
(6)
In 2021, Dr. Hack was awarded 7,500 shares of restricted common stock that vest one year from the date of grant for his service
as a director.
(6)
Mr. Munn was an employee director and not separately compensated for his services on the Board.
Item
12. Security ownership Certain Beneficial Owners and Management and Related Stockholder Matters
The
table below sets forth information regarding the beneficial ownership of the common stock by (i) our directors and named executive officers;
(ii) all the named executives and directors as a group and (iii) any other person or group that to our knowledge beneficially owns more
than five percent of our outstanding shares of common stock.
We
have determined beneficial ownership in accordance with the rules and regulations of the SEC. These rules generally provide that a person
is the beneficial owner of securities if such person has or shares the power to vote or direct the voting thereof, or to dispose or direct
the disposition thereof or has the right to acquire such powers within 60 days. Shares of common stock subject to options that are currently
exercisable or exercisable within 60 days of April 7, 2022 are deemed to be outstanding and beneficially owned by the person holding
the options. Shares issuable pursuant to stock options or warrants are deemed outstanding for computing the percentage ownership of the
person holding such options or warrants, but are not deemed outstanding for computing the percentage ownership of any other person. Except
as indicated by the footnotes below, we believe, based on the information furnished to us, that the persons and entities named in the
table below will have sole voting and investment power with respect to all shares of common stock that they will beneficially own, subject
to applicable community property laws. The percentage of beneficial ownership is based on 12,888,174 shares of common stock outstanding
on April 7, 2022.
| |
|
Number
of Shares
Beneficially Owned |
|
Beneficial
Ownership Percentages |
| Name
and Address of Beneficial Owner(1) |
|
Common
Stock |
|
Series
X Super Voting Preferred
Stock(2) |
|
Percent
of
Common
Stock |
|
Percent
of Series X Super Voting Preferred
Stock |
|
Percent
of
Voting
Stock(3) |
| Officers
and Directors |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Max
Munn, Interim Chief Executive Officer , President and Director |
|
|
5,221,492 |
(4) |
|
|
2,000 |
(5) |
|
|
40.5 |
% |
|
|
100 |
% |
|
|
48.5 |
% |
| Michael
Riccio, Chief Financial Officer |
|
|
92,611 |
|
|
|
— |
|
|
|
* |
|
|
|
— |
|
|
|
* |
|
| John
J. Hayman III, Interim Chief Operating Officer |
|
|
203,907 |
|
|
|
— |
|
|
|
1.6 |
% |
|
|
— |
|
|
|
1.4 |
% |
| Joel
Kanter, Chairman |
|
|
53,000 |
(6) |
|
|
— |
|
|
|
* |
|
|
|
— |
|
|
|
* |
|
| Alastair
Clemow, Director |
|
|
47,500 |
|
|
|
— |
|
|
|
* |
|
|
|
— |
|
|
|
* |
|
| Eugene
Bauer, Director |
|
|
47,500 |
|
|
|
— |
|
|
|
* |
|
|
|
— |
|
|
|
* |
|
| Eugene
Burleson, Director |
|
|
47,500 |
|
|
|
|
|
|
|
* |
|
|
|
— |
|
|
|
* |
|
| Dallas
Hack, Director |
|
|
32,500 |
|
|
|
|
|
|
|
* |
|
|
|
— |
|
|
|
* |
|
| Officers
and Directors as a Group (total of 2 persons) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
| |
|
|
5,746,010 |
|
|
|
2,000 |
|
|
|
44.6 |
% |
|
|
100 |
% |
|
|
52.0 |
% |
| 5%
Stockholders |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| The
Munn Family 2020 Irrevocable Trust |
|
|
5,000,000 |
|
|
|
2,000 |
|
|
|
38.8 |
% |
|
|
100 |
% |
|
|
47.0 |
% |
| Fakhruddin
Holdings FZC |
|
|
785,714 |
|
|
|
— |
|
|
|
6.1 |
% |
|
|
N/A |
|
|
|
5.3 |
% |
(1)
The principal address of the named officers, directors and 5% stockholders of the Company is c/o Applied UV, Inc. 150 N. Macquesten
Parkway Mount Vernon, New York 10550..
(2)
Entitles the holder to 1,000 votes per share and votes with the common as a single class.
(3)
Represents total ownership percentage with respect to all shares of common stock and Series X Super Voting Preferred Stock, as
a single class.
(4)
Includes (i) 5,000,000 shares which are held in the name of The Munn Family 2020 Irrevocable Trust, for which the spouse of Max
Munn is the trustee; (ii) 20,000 shares owned by Mr. Munn directly (iii) 80,000 shares underlying a warrant issued to Mr. Munn, which
is exercisable at $5.00 per share; (iv) 1,000 vested shares underlying an option granted to Mr. Munn as director compensation, which
are exerciseable at $4.33 per share and (v) 120,492 vested shares underlying an option granted to Mr. Munn pursuant to his employment
agreement, which are exercisable at $7.80 per share. Mr. Munn is a guarantor of a $1,500,000 loan to The Munn Family Trust pursuant to
which 5,000,000 shares of Common Stock held by the Munn Family Trust are pledged as collateral.
(5)
Held by The Munn Family 2020 Irevoccable Trust..
(6)
Includes 500 vested options exercisacble at $4.33 per share.
Item
13. Certain Relationships and Related party Transactions, and Director Independence
In
February of 2019, the Company engaged Carmel, Milazzo & Feil LLP (the "Firm") to represent and assist the company with all
general corporate legal matters including the preparation and filing with the Securities and Exchange Commission of a registration statement
on Form S-1 in connection with the Company's initial public offering. Ross Carmel, a former member of the Company’s Board of Directors,
who resigned on May 1, 2020, is a partner at the Firm. The Firm performed legal services in exchange for 161,794 shares of the Company's
common stock
Item
14. Principal Accounting Fees and Services
Fees
For Audit Services: The following table sets forth the aggregate fees for services we have paid to Adeptus Partners LLC (“Adeptus”)
our independent registered public accounting firms for the fiscal years ended December 31, 2021 and 2020:
| Years | |
Audit
Fees | |
Audit
Related Fees | |
Tax
Fees | |
All
other Fees |
| 2021(1) | |
$ | 95,000 | | |
| — | | |
| — | | |
| — | |
| 2020(1) | |
$ | 146,000 | | |
| — | | |
| — | | |
| — | |
The following
table sets forth the aggregate fees for services we have paid to Mazars USA LLP (“Mazars”) our independent registered public
accounting firm for the fiscal year ended December 31, 2021:
| Years | |
Audit
Fees | |
Audit
Related Fees | |
Tax
Fees | |
All
other Fees |
| 2021(1) | |
$ | 99,225 | | |
| — | | |
| — | | |
| — | |
(1)
Audit fees consist of fees billed for professional services rendered for the audit of our consolidated annual financial statements,
review of the interim consolidated financial statements, the issuance of consent and comfort letters in connection with registration
statement filings with the SEC and all services that are normally provided by the accounting firm in connection with statutory and regulatory
filings or engagements.
PART
IV
Item
15. Exhibits, Financial Statement Schedules.
(a)
The following documents are filed as part of this Annual Report:
(1)
The financial statements are filed as part of this Annual Report under “Item 8. Financial Statements and Supplementary Data.”
(2) The
financial statement schedules are omitted because they are either not applicable or the information required is presented in the financial
statements and notes thereto under “Item 8. Financial Statements and Supplementary Data.”
(3) The
exhibits listed in the following Exhibit Index are filed, furnished or incorporated by reference as part of this Annual Report.
(b) Exhibits
See the
Exhibit Index immediately preceding the signature page of this Annual Report.
Item
16. Form 10-K Summary
None.
EXHIBIT
INDEX
| No. |
|
Exhibit No. |
| 3.1 |
Certificate
of Incorporation of the Registrant (incorporated by reference to Exhibit 3.1 of the Registrant’s Registration Statement on
Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 3.2 |
Amended
and Restated Certificate of Incorporation of the Registrant (incorporated by reference to Exhibit 3.2 of the Registrant’s Registration
Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 3.3 |
Bylaws
of the Registrant (incorporated by reference to Exhibit 3.3 of the Registrant’s Registration Statement on Form S-1 (File No.
333-239892) filed with the SEC as of July 16, 2020). |
| 3.4 |
Certificate
of Designation, Preferences and Rights of Series A Preferred Stock (incorporated by reference to Exhibit 3.4 of the Registrant’s
Registration Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 3.5 |
Certificate
of Amendment of Certificate of Incorporation filed on June 17, 2020 (incorporated by reference to Exhibit 3.5 of the Registrant’s
Registration Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 3.6 |
Certificate
of Amendment of Certificate of Incorporation filed on June 23, 2020 (incorporated by reference to Exhibit 3.6 of the Registrant’s
Registration Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 3.7 |
Certificate
of Amendment of Certificate of Incorporation filed July 14, 2020 (incorporated by reference to Exhibit 3.7 of the Registrant’s
Registration Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 3.8 |
Certificate
of Amendment to Certificate of Designation of Series A Preferred Stock, filed on June 17, 2021 (incorporated by reference to Exhibit
3.1 of the Registrant’s Current Report on Form 8-K, filed on July 19, 2021). |
| 3.9 |
Certificate
of Designation, Preferences and Rights of 10.5% Series A Cumulative Perpetual Preferred Stock (incorporated by reference to Exhibit
3.9 of the Registrant’s Registration Statement on Form S-1 (File No. 333-257197) filed with the SEC as of June 25, 2021). |
| 3.10 |
Certificate
of Amendment to the Amended and Restated Certificate of Incorporation, filed on October 7, 2021 |
| 3.11 |
Certificate
of Amendment to the Certificate of Designation of Series A Preferred Stock, filed on December 8, 2021 |
| 10.1 |
Exchange
Agreement, dated March 26, 2019 among the Registrant, SteriLumen, Inc. and each of the stockholders of SteriLumen, Inc. (incorporated
by reference to Exhibit 10.1 of the Registrant’s Registration Statement on Form S-1 (File No. 333-239892) filed with the SEC
as of July 16, 2020). |
| 10.2 |
Exchange
Agreement, dated March 27, 2019 among the Registrant, SteriLumen, Inc. and Laurie Munn (incorporated by reference to Exhibit 10.2
of the Registrant’s Registration Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 10.3 |
Exchange
Agreement, dated July 1, 2019 among the Registrant, Munn Works, LLC and Laurie Munn (incorporated by reference to Exhibit 10.3 of
the Registrant’s Registration Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 10.4 |
Warrant,
dated April 1, 2020 issued to Max Munn (incorporated by reference to Exhibit 10.4 of the Registrant’s Registration Statement
on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 10.5 |
The
Registrant’s 2020 Omnibus Incentive Plan (incorporated by reference to Exhibit 10.5 of the Registrant’s Registration
Statement on Form S-1 (333-239892) filed with the SEC as of July 16, 2020). |
| 10.6 |
Form
of Option Agreement and Grant issued under February 18, 2020 Board Approval (incorporated by reference to Exhibit 10.6 of the Registrant’s
Registration Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 10.7 |
Agreement,
dated April 20, 2020 between Icahn School of Medicine at Mount Sinai and SteriLumen, Inc. (incorporated by reference to Exhibit 10.7
of the Registrant’s Registration Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 10.8 |
Employment
Agreement, dated June 30, 2020 between the Registrant and James L. Doyle III (incorporated by reference to Exhibit 10.9 of the Registrant’s
Registration Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 10.9 |
Common
Stock Purchase Warrant, dated July 1, 2020 (incorporated by reference to Exhibit 10.10 of the Registrant’s Registration Statement
on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 10.10 |
Common
Stock Purchase Warrant, dated July 1, 2020 (incorporated by reference to Exhibit 10.11 of the Registrant’s Registration Statement
on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 10.11 |
Form
of Option issued to Medical Advisory Board members (incorporated by reference to Exhibit 10.12 of the Registrant’s Registration
Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 10.12 |
Asset
Purchase Agreement, dated as of February 8, 2021, by and among Applied UV, Inc., SteriLumen, Inc., Akida Holdings LLC, and members
of Akida Holdings, LLC. (incorporated by reference to Exhibit 2.1 of the Registrant’s Current Report on Form 8-K filed with
the SEC as of February 11, 2021). |
| 10.13 |
Contract
Manufacturing Agreement, dated as of January 1, 2021, by and between KES Science & Technology, Inc. and Akida Holdings LLC. |
| 10.14 |
Intellectual
Property Assignment and License Agreement, dated as of January 1, 2021, by and among KES Science & Technology, Inc., KES Air
Technologies, LLC and Akida Holdings LLC. |
| 10.15 |
Management
Services Agreement, dated as of January 1, 2021, by and between KES Science & Technology, Inc. and Akida Holdings LLC. |
| 10.16 |
Employment
Offer to Michael Riccio (incorporated by reference to Exhibit 10.1 of the Registrant’s Current Report on Form 8-K filed with
the SEC as of April 20, 2021). |
| 10.17 |
Employment
Agreement, dated June 30, 2020 between the Registrant and Max Munn (incorporated by reference to Exhibit 10.9 of the Registrant’s
Registration Statement on Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020). |
| 21.1 |
|
List
of Subsidiaries of the Registrant (incorporated by reference to Exhibit 21.1 of the Registrant’s Registration Statement on
Form S-1 (File No. 333-239892) filed with the SEC as of July 16, 2020) |
| 31.1** |
|
Certification
of the Chief Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
| 31.2** |
|
Certification
of the Principal Financial Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 . |
| 32.1** |
|
Certification
by the Chief Executive Officer pursuant to 18 U.S.C. 1350 as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
| 32.2** |
|
Certification
of Principal Financial Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of
2002 |
**
Filed herewith
SIGNATURES
Pursuant
to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report
to be signed on its behalf by the undersigned, thereunto duly authorized.
| Date:
April 07, 2022 |
APPLIED
UV, INC. |
| |
|
| |
By: |
/s/
Max Munn |
| |
|
Max
Munn |
| |
|
Interim
Chief Executive Officer |
POWER
OF ATTORNEY
Each
individual person whose signature appears below hereby appoints Keyoumars Saeed and Max Munn as attorneys-in-fact with full power of
substitution, severally, to execute in the name and on behalf of each such person, individually and in each capacity stated below, one
or more amendments to this annual report which amendments may make such changes in the report as the attorney-in-fact acting in the premises
deems appropriate, to file any such amendment to the report with the SEC, and to take all other actions either of them deem necessary
or advisable to enable the Company to comply with the rules, regulations and requirements of the SEC.
Pursuant
to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the
registrant and in the capacities and on the dates indicated.
| Signature |
|
Title |
|
Date |
| |
|
|
|
|
| |
|
|
|
|
| /s/
Max Munn |
|
Interim
Chief Executive Officer, President and Director |
|
April
07, 2022 |
| Max
Munn |
|
|
|
|
| |
|
|
|
|
| /s/
Michael Riccio |
|
Chief
Financial Officer |
|
April
07, 2022 |
| Michael
Riccio |
|
(principal
financial and accounting officer) |
|
|
| |
|
|
|
|
| /s/
Joel Kanter |
|
Director |
|
April
07, 2022 |
| Joel
Kanter |
|
|
|
|
| |
|
|
|
|
| /s/
Dr. Alastair Clemow |
|
Director |
|
April
07, 2022 |
| Dr.
Alastair Clemow |
|
|
|
|
| |
|
|
|
|
| /s/
Eugene Burleson |
|
Director |
|
April
07, 2022 |
| Eugene
Burleson |
|
|
|
|
| |
|
|
|
|
| /s/
Dr. Eugene Bauer |
|
Director |
|
April
07, 2022 |
| Dr.
Gene Bauer |
|
|
|
|
| |
|
|
|
|
| /s/
Dr. Dallas C. Hack |
|
Director |
|
April
07, 2022 |
| Dr.
Dallas C. Hack |
|
|
|
|
| |
|
|
|
|
| /s/
John F. Andrews |
|
Director |
|
April
07, 2022 |
| John
F. Andrews |
|
|
|
|
| BOARD
OF DIRECTORS |
|
SENIOR
EXECUTIVE OFFICERS |
|
INVESTOR
INFORMATION |
•
Joel Kanter,
Chairman
of the Board. President of Windy City, Inc., a privately held investment firm. |
|
•
Max Munn – Interim Chief Executive Officer. |
|
COMPANY
ADDRESS: |
| |
|
|
|
|
•
Max Munn.
Interim
Chief Executive Officer and President |
|
•
Michael Riccio – Chief Financial Officer. |
|
•
Applied UV, Inc.
150
N. Macquesten Parkway
Mount
Vernon, New York 10550
Telephone:
(914) 665-6100
Website:
www.applieduvinc.com
|
| |
|
|
|
|
•
Dr. Eugene A. Bauer,
M.D.
Co-founder of Dermira, a publicly traded specialty biopharmaceutical company acquired by Eli Lilly and Company in 2020. |
|
•
John J. Hayman III - Interim Chief Operations Officer. |
|
|
| |
|
|
|
|
•
Dr. Alastair J. Clemow, Ph.D.
Chairman
of Ensemble Orthopedics, an early-stage company developing an innovative pyrocarbon finger joint for the treatment for early
state osteoarthritis. |
|
INDEPENDENT
AUDITORS |
|
STOCK
EXCHANGE: |
| |
|
|
|
|
•
Dallas C. Hack, M.D., M.P.H.
Board
certified in General Preventive Medicine and Public Health and a Fellow in the American College of Military Public Health. |
|
•
Mazars USA LLP
135
West 50th Street New York, New York 10020-1299 |
|
•
The Common Stock, par value $0.0001 per share, of Applied UV, Inc. trades on the Nasdaq Capital Market under the symbol “AUVI.” |
| |
|
|
|
|
•
Eugene E. Burleson.
Private
investor and Chairman of PET DRx Corporation. |
|
|
|
•
The 10.5% Series A Cumulative Perpetual Preferred Stock, $0.0001 par value per share, of Applied UV, Inc. trades on the Nasdaq Capital
Market under the symbol “AUVIP.” |
| |
|
|
|
|
| |
|
|
|
|
| |
|
FORM
10-K |
|
REGISTRAR
AND TRANSFER AGENT |
| |
|
|
|
|
| |
|
Our
2021 Form 10-K is included in this Annual Report in its entirety with the exception of certain
exhibits. All of the exhibits may be obtained on our Investor Relations homepage at https://irdirect.net/AUVI
or by accessing our filings with the U.S. Securities and Exchange Commission at www.sec.gov.
In addition, stockholders may obtain a paper copy of any exhibit by writing to:
Michael
Riccio,Chief Financial Officer, Applied UV, Inc., 150 N. Macquesten Parkway, Mount Vernon,
New York 10550.
|
|
The
Registrar and Transfer Agent for the Common Stock and the 10.5% Series A Cumulative Preferred
Stock of Applied UV, Inc. is :
•
VStock Transfer, LLC 18 Lafayette Place Woodmere, New York 11598
Telephone:
(212) 828-8436
Internet:
www.vstocktransfer.com
|
| |
|
|
|
|
| |
|
INVESTOR
RELATIONS CONTACT |
|
|
| |
|
|
|
|
| |
|
Brett
Maas, Managing Partner Hayden IR
1441
Broadway Fifth Floor New York, NY 10018
Tel.
(646) 536-7331
Email:
brett@haydenir.com
|
|
|
Applied UV (NASDAQ:AUVI)
Historical Stock Chart
From Mar 2024 to Apr 2024
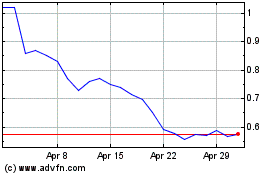
Applied UV (NASDAQ:AUVI)
Historical Stock Chart
From Apr 2023 to Apr 2024