false
0001604821
0001604821
2024-05-09
2024-05-09
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of
the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
May 9, 2024
Natera, Inc.
(Exact name of registrant as specified
in its charter)
| Delaware |
|
001-37478 |
|
01-0894487 |
|
(State or other jurisdiction
of incorporation) |
|
(Commission
File Number) |
|
(IRS Employer
Identification No.) |
13011 McCallen Pass
Building A Suite 100
Austin, TX 78753
(Address of principal executive offices,
including zip code)
(650)
980-9190
(Registrant’s telephone number,
including area code)
N/A
(Former name or
former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under
any of the following provisions:
| ¨ | Written communications pursuant to Rule 425 under the
Securities Act (17 CFR 230.425) |
| ¨ | Soliciting material pursuant to Rule 14a-12 under the
Exchange Act (17 CFR 240.14a-12) |
| ¨ | Pre-commencement communications pursuant to Rule 14d-2(b)
under the Exchange Act (17 CFR 240.14d-2(b)) |
| ¨ | Pre-commencement communications pursuant to Rule 13e-4(c)
under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the
Act:
| Title of each class |
|
Trading Symbol(s) |
|
Name of each exchange
on which registered |
| Common Stock, par value $0.0001 per share |
|
NTRA |
|
Nasdaq Stock Market LLC
(Nasdaq Global Select Market) |
Indicate by check mark whether the registrant is an emerging
growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of
the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ¨
If an emerging growth company, indicate by check mark if the
registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards
provided pursuant to Section 13(a) of the Exchange Act. ¨
| Item 2.02. |
Results of Operations and Financial Condition. |
On May 9, 2024, Natera, Inc. issued a press release announcing
the results for its first quarter ended March 31, 2024 and provided a related investor presentation. A copy of the press release
and a copy of the investor presentation are furnished herewith as Exhibit 99.1 and Exhibit 99.2, respectively, to this Current
Report on Form 8-K and are incorporated herein by reference.
The information in this Current Report on Form 8-K and the accompanying
Exhibit 99.1 and Exhibit 99.2 shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange
Act of 1934, as amended (the “Exchange Act”) or otherwise subject to the liabilities of that section, nor shall it be deemed
incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, regardless of any general incorporation
language in such filing, unless expressly incorporated by reference in such filing.
| Item 9.01. |
Financial Statements and Exhibits. |
(d) Exhibits.
SIGNATURES
Pursuant to the requirements of the Securities
Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly
authorized.
| |
Natera, Inc. |
| |
|
|
| |
By: |
/s/ Michael Brophy |
| |
|
Michael Brophy |
| |
|
Chief Financial Officer (Principal Financial and Accounting Officer) |
Dated: May 9, 2024
Exhibit 99.1
Natera Reports First Quarter 2024
Financial Results
AUSTIN, Texas, May 9, 2024 /PRNewswire/ — Natera, Inc.
(NASDAQ: NTRA), a global leader in cell-free DNA and genetic testing, today reported
its financial results for the first quarter ended March 31, 2024.
Recent Strategic and Financial Highlights
| · | Achieved positive cash flow of approximately $3.0 million1 in the first quarter of 2024. |
| · | Generated total revenues of $367.7 million in the first quarter of 2024, compared to $241.8 million in
the first quarter of 2023, an increase of 52.1%. Product revenues grew 53.4% over the same period. |
| · | Generated gross margins of 56.7% in the first quarter of 2024, compared to gross margins of 38.7% in the
first quarter of 2023. |
| · | Processed approximately 735,800 tests in the first quarter of 2024, compared to approximately 626,200
tests in the first quarter of 2023, an increase of 17.5%. |
| · | Performed approximately 114,800 oncology tests in the first quarter of 2024, compared to approximately
71,000 in the first quarter of 2023, an increase of 61.7%. |
| · | Launched fetal RhD NIPT test backed by excellent performance in clinical validation study. |
| · | Announced updated guidelines from a leading medical society in nephrology that support genetic testing
in patients with chronic kidney disease to establish cause of disease. |
| · | Published largest, prospective, donor-derived cfDNA study in kidney transplantation. |
| · | Released analysis from randomized, phase III IMvigor011 trial in bladder cancer demonstrating that patients
with undetectable circulating tumor DNA on serial Signatera testing may be able to forgo treatment. |
| · | Published key data in breast cancer and uterine cancer. |
“We had an excellent start to the year,
driving robust volume, revenue and margin growth and achieving cash flow breakeven earlier than expected,” said Steve Chapman, chief
executive officer of Natera. “We continued to deliver on our mission to transform disease management with the launch of a highly
differentiated product in women’s health and the publication of practice-changing data in oncology and organ health.”
First Quarter Ended March 31, 2024 Financial
Results
Total revenues were $367.7 million in the first
quarter of 2024 compared to $241.8 million in the first quarter of 2023, an increase of 52.1%. The increase in total revenues was driven
primarily by a 53.4% increase in product revenues, which were $364.7 million in the first quarter of 2024 compared to $237.8 million in
the first quarter of 2023. The increase in product revenues was primarily driven by an increase in volume, as well as average selling
price improvements.
Natera processed approximately 735,800 tests in
the first quarter of 2024, including approximately 718,700 tests accessioned in its laboratory, compared to approximately 626,200 tests
processed, including approximately 607,700 tests accessioned in its laboratory, in the first quarter of 2023.
In the first quarter of 2024, Natera recognized
revenue on approximately 679,400 tests for which results were reported to customers in the period (tests reported), including approximately
663,500 tests reported from its laboratory, compared to approximately 583,400 tests reported, including approximately 566,000 tests reported
from its laboratory, in the first quarter of 2023, an overall increase of 16.5% from the prior period.
Gross profit2 for the three months
ended March 31, 2024 and 2023 was $208.6 million and $93.6 million, respectively, representing a gross margin of 56.7% and 38.7%,
respectively. Natera had higher gross margin in the first quarter of 2024 primarily as a result of higher revenues and continuous progress
in reducing cost of goods sold associated with tests processed. Total operating expenses, representing research and development expenses
and selling, general and administrative expenses, for the first quarter of 2024 were $282.9 million, compared to $231.9 million in the
same period of the prior year, an increase of 22.0%. The increase was primarily driven by headcount growth to support new product offerings
as well as increases in consulting and legal expenses.
Loss from operations for the first quarter of
2024 was $74.3 million compared to $138.3 million for the same period of the prior year.
Natera reported a net loss for the first quarter
of 2024 of $67.6 million, or ($0.56) per diluted share, compared to a net loss of $136.9 million, or ($1.23) per diluted share, for the
same period in 2023. Weighted average shares outstanding were approximately 120.8 million in the first quarter of 2024 compared to 111.8
million in the first quarter of the prior year.
At March 31, 2024, Natera held approximately
$882.9 million in cash, cash equivalents, short-term investments and restricted cash, compared to $879.0 million as of December 31,
2023. As of March 31, 2024, Natera had a total outstanding debt balance of $363.7 million, comprised of $80.4 million including accrued
interest under its line of credit with UBS at a variable interest rate of 30-day SOFR plus 50 bps and a net carrying amount of $283.3
million under its seven-year convertible senior notes issued in April 2020. The gross principal balance outstanding for the convertible
senior notes was $287.5 million as of March 31, 2024.
Financial Outlook
Natera anticipates 2024 total revenue of $1.42
billion to $1.45 billion; 2024 gross margin to be approximately 53% to 55% of revenues; selling, general and administrative costs to be
approximately $700 million to $750 million; research and development costs to be $350 million to $375 million, and net cash inflow (consumption)
to be ($25) million to $25 million3.
Test Volume Summary
| Unit | |
Q1 2024 | | |
Q1 2023 | | |
Definition |
|
| Tests processed | |
| 735,800 | | |
| 626,200 | | |
Tests accessioned in our laboratory plus units processed outside of our laboratory |
|
| Tests accessioned | |
| 718,700 | | |
| 607,700 | | |
Test accessioned in our laboratory |
|
| Tests reported | |
| 679,400 | | |
| 583,400 | | |
Total tests reported |
|
| Tests reported in our laboratory | |
| 663,500 | | |
| 566,000 | | |
Total tests reported in our laboratory less units reported outside of our laboratory |
|
About Natera
Natera™
is a global leader in cell-free DNA and genetic testing, dedicated to oncology, women’s health, and organ health. We aim to make
personalized genetic testing and diagnostics part of the standard of care to protect health and enable earlier, more targeted interventions
that help lead to longer, healthier lives. Natera’s tests are validated by more than 200 peer-reviewed publications that demonstrate
high accuracy. Natera operates ISO 13485-certified and CAP-accredited laboratories certified under the Clinical Laboratory Improvement
Amendments (CLIA) in Austin, Texas and San Carlos, California. For more information, visit www.natera.com.
Conference Call Information
| Event: |
Natera’s First Quarter 2024 Financial Results Conference Call |
|
| Date: |
Thursday, May 9, 2024 |
|
| Time: |
1:30 p.m. PT (4:30 p.m. ET) |
|
| Live Dial-In: |
(888) 770-7321, Domestic |
| |
(929) 201-7107, International |
| Conference ID: |
7684785 |
| Webcast Link: |
https://events.q4inc.com/attendee/575350879 |
Forward-Looking Statements
This press release contains forward-looking statements
under the meaning of the Private Securities Litigation Reform Act of 1995. All statements other than statements of historical facts, including
the company’s financial guidance for fiscal 2024, its market opportunity, anticipated products and launch schedules, reimbursement
coverage and product costs, commercial and strategic partnerships and acquisitions, user experience, clinical trials and studies, and
its strategies, goals and general business and market conditions, are forward-looking statements. Any forward-looking statements contained
in this press release are based upon Natera’s current plans, estimates, and expectations, as of the date of this release, and are
not a representation that such plans, estimates, or expectations will be achieved.
These forward-looking statements are subject to
known and unknown risks and uncertainties that may cause actual results to differ materially, including: we face numerous uncertainties
and challenges in achieving our financial projections and goals; we may be unable to further increase the use and adoption of our products
through our direct sales efforts or through our laboratory partners; we have incurred losses since our inception and we anticipate that
we will continue to incur losses for the foreseeable future; our quarterly results may fluctuate from period to period; our estimates
of market opportunity and forecasts of market growth may prove to be inaccurate; we may be unable to compete successfully with existing
or future products or services offered by our competitors; we may engage in acquisitions, dispositions or other strategic transactions
that may not achieve our anticipated benefits and could otherwise disrupt our business, cause dilution to our stockholders or reduce our
financial resources; we may not be successful in commercializing our cloud-based distribution model; our products may not perform as expected;
the results of our clinical studies, including our SNP-based Microdeletion and Aneuploidy Registry, or SMART, Study, may not be compelling
to professional societies or payors as supporting the use of our tests, particularly for microdeletions screening, or may not be able
to be replicated in later studies required for regulatory approvals or clearances; if either of our primary CLIA-certified laboratories
becomes inoperable, we will be unable to perform our tests and our business will be harmed; we rely on a limited number of suppliers or,
in some cases, single suppliers, for some of our laboratory instruments and materials and may not be able to find replacements or immediately
transition to alternative suppliers; if we are unable to successfully scale our operations, our business could suffer; the marketing,
sale, and use of Panorama and our other products could result in substantial damages arising from product liability or professional liability
claims that exceed our resources; we may be unable to expand, obtain or maintain third-party payer coverage and reimbursement for our
tests, and we may be required to refund reimbursements already received; third-party payers may withdraw coverage or provide lower levels
of reimbursement due to changing policies, billing complexities or other factors; we could incur substantial costs and delays associated
with trying to obtain premarket clearance or approval, and incur costs associated with complying with post-market controls, if and when
the FDA begins actively regulating our tests pursuant to recently enacted FDA regulations; litigation or other proceedings, resulting
from either third party claims of intellectual property infringement or third party infringement of our technology, is costly, time-consuming
and could limit our ability to commercialize our products or services; any inability to effectively protect our proprietary technology
could harm our competitive position or our brand; and we cannot guarantee that we will be able to service and comply with our outstanding
debt obligations or achieve our expectations regarding the conversion of our outstanding convertible notes.
Additional risks and uncertainties that could
affect our financial results are included under the captions, "Risk Factors" and "Management’s Discussion and Analysis
of Financial Condition and Results of Operations" in our most recent filings on Forms 10-K and 10-Q and in other filings that we
make with the SEC from time to time. These documents are available on our website at www.natera.com under the Investor Relations section
and on the SEC’s website at www.sec.gov.
In light of the significant uncertainties in these
forward-looking statements, you should not regard these statements as a representation or warranty by us or any other person that we will
achieve our objectives and plans in any specified time frame, or at all. Natera assumes no obligation to, and does not currently intend
to, update any such forward-looking statements after the date of this release.
References
| 1. | Positive cash flow for the quarter ended March 31, 2024, is derived from the GAAP Statement of Cash
Flows as follows: net cash provided by operating activities of $27.0 million, net cash provided by financing activities of $6.5 million,
offset by net cash used in investing activities for purchases of property and equipment and acquisition of an asset of $30.8 million. |
| 2. | Gross profit is calculated as GAAP total revenues less GAAP cost of revenues. Gross margin is calculated
as gross profit divided by GAAP total revenues. |
| 3. | Cash inflow (consumption) is calculated as the sum of GAAP net cash provided by (used in) operating activities,
GAAP net cash provided by (used in) financing activities, and GAAP net cash provided by (used in) investing activities for purchases of
property and equipment and acquisition of assets. |
Contacts
Investor Relations
Mike Brophy, CFO, Natera, Inc., 510-826-2350
Media
Lesley Bogdanow, VP of Corporate Communications, Natera, Inc., pr@natera.com
Natera, Inc.
Consolidated Balance Sheets
(Unaudited)
(in thousands, except shares)
| | |
March 31, | | |
December 31, | |
| | |
2024 | | |
2023 | |
| | |
| | |
(1) | |
| Assets | |
| | | |
| | |
| Current assets: | |
| | | |
| | |
| Cash, cash equivalents and restricted cash | |
$ | 813,817 | | |
$ | 642,095 | |
| Short-term investments | |
| 69,121 | | |
| 236,882 | |
| Accounts receivable, net of allowance of $7,252 and $6,481 at March 31, 2024 and December 31, 2023, respectively | |
| 288,748 | | |
| 278,289 | |
| Inventory | |
| 43,024 | | |
| 40,759 | |
| Prepaid expenses and other current assets, net | |
| 46,734 | | |
| 60,524 | |
| Total current assets | |
| 1,261,444 | | |
| 1,258,549 | |
| Property and equipment, net | |
| 125,791 | | |
| 111,210 | |
| Operating lease right-of-use assets | |
| 54,553 | | |
| 56,537 | |
| Other assets | |
| 26,417 | | |
| 15,403 | |
| Total assets | |
$ | 1,468,205 | | |
$ | 1,441,699 | |
| Liabilities and Stockholders’ Equity | |
| | | |
| | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable | |
$ | 26,038 | | |
$ | 14,998 | |
| Accrued compensation | |
| 39,577 | | |
| 45,857 | |
| Other accrued liabilities | |
| 142,228 | | |
| 149,405 | |
| Deferred revenue, current portion | |
| 17,705 | | |
| 16,612 | |
| Short-term debt financing | |
| 80,401 | | |
| 80,402 | |
| Total current liabilities | |
| 305,949 | | |
| 307,274 | |
| Long-term debt financing | |
| 283,273 | | |
| 282,945 | |
| Deferred revenue, long-term portion and other liabilities | |
| 20,712 | | |
| 19,128 | |
| Operating lease liabilities, long-term portion | |
| 64,160 | | |
| 67,025 | |
| Total liabilities | |
| 674,094 | | |
| 676,372 | |
| | |
| | | |
| | |
| Commitments and contingencies | |
| | | |
| | |
| | |
| | | |
| | |
| Stockholders’ equity: | |
| | | |
| | |
| Common stock (2) | |
| 12 | | |
| 11 | |
| Additional paid-in capital | |
| 3,241,326 | | |
| 3,145,837 | |
| Accumulated deficit | |
| (2,445,035 | ) | |
| (2,377,436 | ) |
| Accumulated other comprehensive loss | |
| (2,192 | ) | |
| (3,085 | ) |
| Total stockholders’ equity | |
| 794,111 | | |
| 765,327 | |
| Total liabilities and stockholders’ equity | |
$ | 1,468,205 | | |
$ | 1,441,699 | |
| (1) | The consolidated balance sheet at December 31, 2023 has been derived from the audited consolidated
financial statements at that date included in the Company’s Annual Report on Form 10-K for the year ended December 31,
2023. |
| (2) | As of March 31, 2024 and December 31, 2023, there were approximately 122,234,000 and 119,581,000
shares of common stock issued and outstanding, respectively. |
Natera, Inc.
Consolidated Statements of Operations and Comprehensive
Loss
(Unaudited)
(in thousands, except per share data)
| | |
Three months ended | |
| | |
March 31, | |
| | |
2024 | | |
2023 | |
| Revenues | |
| | | |
| | |
| Product revenues | |
$ | 364,672 | | |
$ | 237,797 | |
| Licensing and other revenues | |
| 3,069 | | |
| 3,959 | |
| Total revenues | |
| 367,741 | | |
| 241,756 | |
| Cost and expenses | |
| | | |
| | |
| Cost of product revenues | |
| 158,833 | | |
| 147,754 | |
| Cost of licensing and other revenues | |
| 307 | | |
| 370 | |
| Research and development | |
| 88,637 | | |
| 82,306 | |
| Selling, general and administrative | |
| 194,278 | | |
| 149,627 | |
| Total cost and expenses | |
| 442,055 | | |
| 380,057 | |
| Loss from operations | |
| (74,314 | ) | |
| (138,301 | ) |
| Interest expense | |
| (3,124 | ) | |
| (3,061 | ) |
| Interest and other income, net | |
| 10,267 | | |
| 4,585 | |
| Loss before income taxes | |
| (67,171 | ) | |
| (136,777 | ) |
| Income tax expense | |
| (428 | ) | |
| (160 | ) |
| Net loss | |
$ | (67,599 | ) | |
$ | (136,937 | ) |
| Unrealized gain on available-for-sale securities, net of tax | |
| 893 | | |
| 4,564 | |
| Comprehensive loss | |
$ | (66,706 | ) | |
$ | (132,373 | ) |
| | |
| | | |
| | |
| Net loss per share: | |
| | | |
| | |
| Basic and diluted | |
$ | (0.56 | ) | |
$ | (1.23 | ) |
| Weighted-average number of shares used in computing basic and diluted net loss per share: | |
| | | |
| | |
| Basic and diluted | |
| 120,814 | | |
| 111,767 | |
| | |
| | | |
| | |
Exhibit 99.2

Natera, Inc. Investor presentation Q1 2024 Earnings Call May 9, 2024

Not for reproduction or further distribution. This presentation contains forward - looking statements under the meaning of the Private Securities Litigation Reform Act of 1995 . All statements other than statements of historical facts contained in this presentation, including statements regarding our market opportunity, our anticipated products and launch schedules, our reimbursement coverage and our product costs, our commercial and strategic partnerships and potential acquisitions, our user experience, our clinical trials and studies, and our strategies, goals and general business and market conditions are forward - looking statements . These forward - looking statements are subject to known and unknown risks and uncertainties that may cause actual results to differ materially, including : we face numerous uncertainties and challenges in achieving our financial projections and goals ; we may be unable to further increase the use and adoption of our products through our direct sales efforts or through our laboratory partners ; we have incurred losses since our inception and we anticipate that we will continue to incur losses for the foreseeable future ; our quarterly results may fluctuate from period to period ; our estimates of market opportunity and forecasts of market growth may prove to be inaccurate ; we may be unable to compete successfully with existing or future products or services offered by our competitors ; we may engage in acquisitions, dispositions or other strategic transactions that may not achieve our anticipated benefits and could otherwise disrupt our business, cause dilution to our stockholders or reduce our financial resources ; we may not be successful in commercializing our cloud - based distribution model ; our products may not perform as expected ; the results of our clinical studies, including our SNP - based Microdeletion and Aneuploidy Registry, or SMART, Study, may not be compelling to professional societies or payors as supporting the use of our tests, particularly for microdeletions screening, or may not be able to be replicated in later studies required for regulatory approvals or clearances ; if either of our primary CLIA - certified laboratories becomes inoperable, we will be unable to perform our tests and our business will be harmed ; we rely on a limited number of suppliers or, in some cases, single suppliers, for some of our laboratory instruments and materials and may not be able to find replacements or immediately transition to alternative suppliers ; if we are unable to successfully scale our operations, our business could suffer ; the marketing, sale, and use of Panorama and our other products could result in substantial damages arising from product liability or professional liability claims that exceed our resources ; we may be unable to expand, obtain or maintain third - party payer coverage and reimbursement for our tests, and we may be required to refund reimbursements already received ; third - party payers may withdraw coverage or provide lower levels of reimbursement due to changing policies, billing complexities or other factors ; we could incur substantial costs and delays associated with trying to obtain premarket clearance or approval and incur costs associated with complying with post - market controls, if and when the FDA begins actively regulating our tests pursuant to recently enacted FDA regulations ; litigation or other proceedings, resulting from either third party claims of intellectual property infringement or third party infringement of our technology, is costly, time - consuming and could limit our ability to commercialize our products or services ; any inability to effectively protect our proprietary technology could harm our competitive position or our brand ; and we cannot guarantee that we will be able to service and comply with our outstanding debt obligations or achieve our expectations regarding the conversion of our outstanding convertible notes . We discuss these and other risks and uncertainties in greater detail in the sections entitled “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in our periodic reports on Forms 10 - K and 10 - Q and in other filings we make with the SEC from time to time . Moreover, we operate in a very competitive and rapidly changing environment . New risks emerge from time to time . It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward - looking statement . In light of these risks, uncertainties and assumptions, the forward - looking events and circumstances discussed in this presentation may not occur and our actual results could differ materially and adversely from those anticipated or implied . As a result, you should not place undue reliance on our forward - looking statements . Except as required by law, we undertake no obligation to update publicly any forward - looking statements for any reason after the date of this presentation to conform these statements to actual results or to changes in our expectations . We file reports, proxy statements, and other information with the SEC . Such reports, proxy statements, and other information concerning us is available at http : //www . sec . gov . Requests for copies of such documents should be directed to our Investor Relations department at Natera, Inc . , 13011 McCallen Pass, Building A Suite 100 , Austin, TX 78753 . Our telephone number is ( 650 ) 980 - 9190 . 2 Safe harbor statement

Not for reproduction or further distribution. 3 Record performance in Q1 2024 • Achieved cash flow 1 breakeven quarter earlier than expected . • Revenue of $367.7M in Q1 2024 vs $241.8M in Q1 2023; year - over - year growth of 52%. • 736K total tests processed in Q1 2024 vs 627K in Q4 2023; sequential growth of 17%. • Performed ~115K oncology tests in Q1 2024; year - over - year growth of 62% and up 17K units versus Q4 2023. • Gross margin 2 of 57% in Q1 2024 vs 39% in Q1 2023. • Raising 2024 guidance: revenue of $1.42B to $1.45B, gross margin of 53% - 55%, cash flow breakeven for 2024. • Launched fetal RhD NIPT test. • Announced positive updated KDIGO guidelines. • Published ProActive , the largest prospective dd - cfDNA study in kidney transplant. • Released positive analysis from IMvigor011 trial in bladder cancer. • Published new data in breast cancer & uterine cancer. 1. Non - GAAP cash inflows for the quarter ended March 31, 2024, is derived from the GAAP Statement of Cash Flows as follows: net cas h provided by operating activities of $27.0 million, net cash provided by financing activities of $6.5 million, offset by net cash used in investing activities for purchases of property and equipment and acquisition of an asset of $30.8 million . 2. Gross margin is calculated as gross profit divided by GAAP total revenues. Gross profit is calculated as GAAP total revenues les s GAAP cost of revenues.
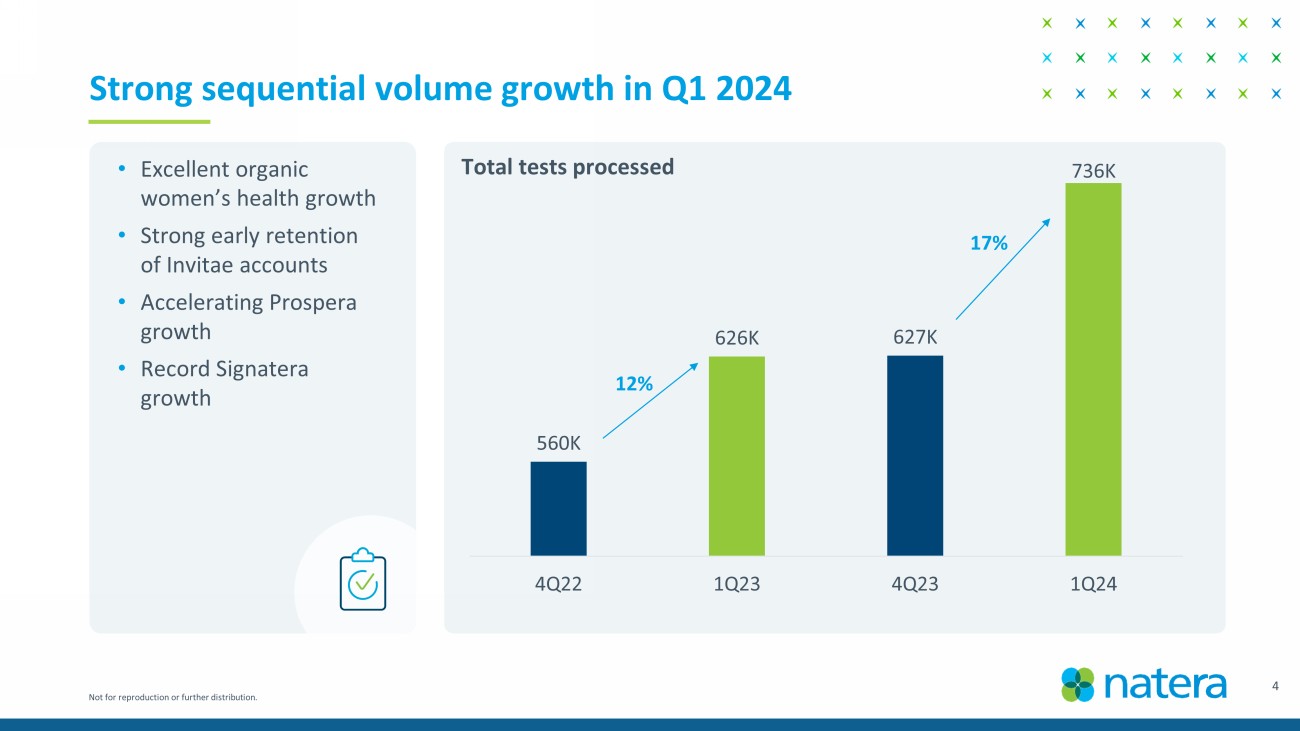
Not for reproduction or further distribution. 560K 626K 627K 736K 4Q22 1Q23 4Q23 1Q24 4 Strong sequential volume growth in Q1 2024 12% 17% • Excellent organic women’s health growth • Strong early retention of Invitae accounts • Accelerating Prospera growth • Record Signatera growth Total tests processed
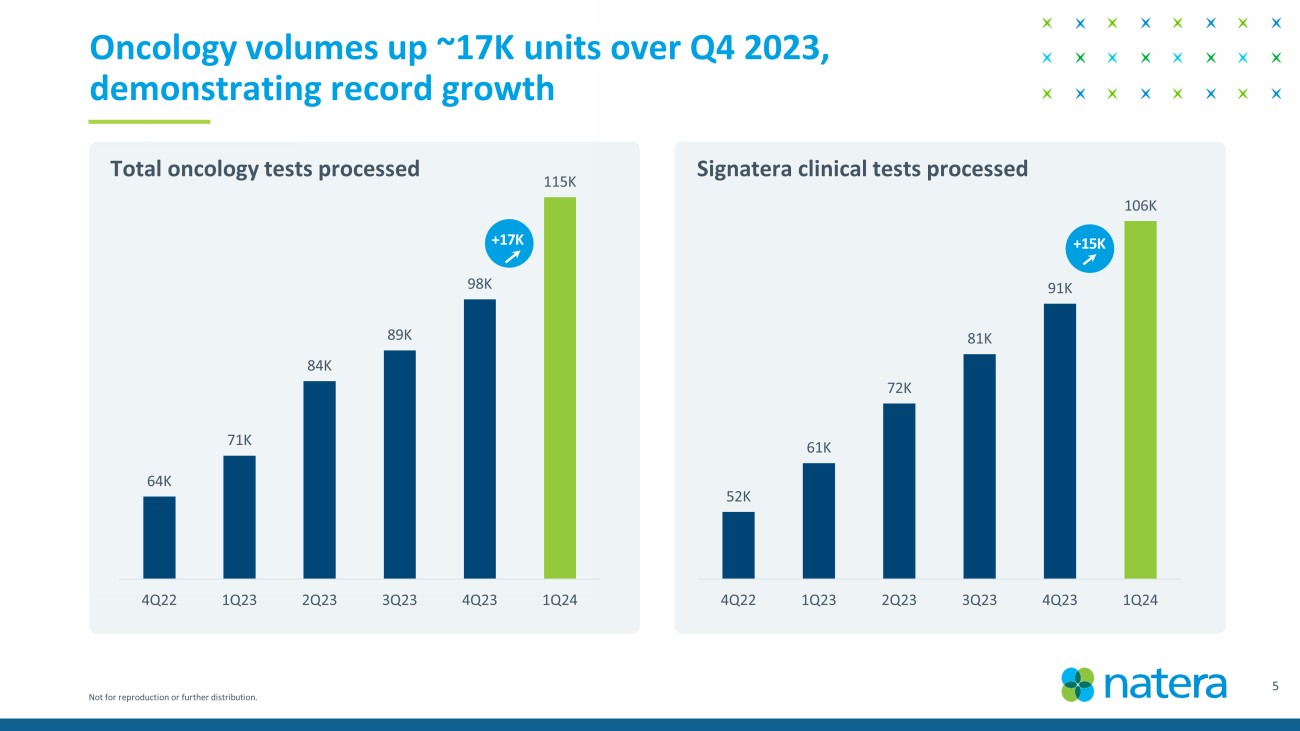
Not for reproduction or further distribution. 5 Oncology volumes up ~17K units over Q4 2023, demonstrating record growth Total oncology tests processed Signatera clinical tests processed 64K 71K 84K 89K 98K 115K 4Q22 1Q23 2Q23 3Q23 4Q23 1Q24 52K 61K 72K 81K 91K 106K 4Q22 1Q23 2Q23 3Q23 4Q23 1Q24 +15K +17K
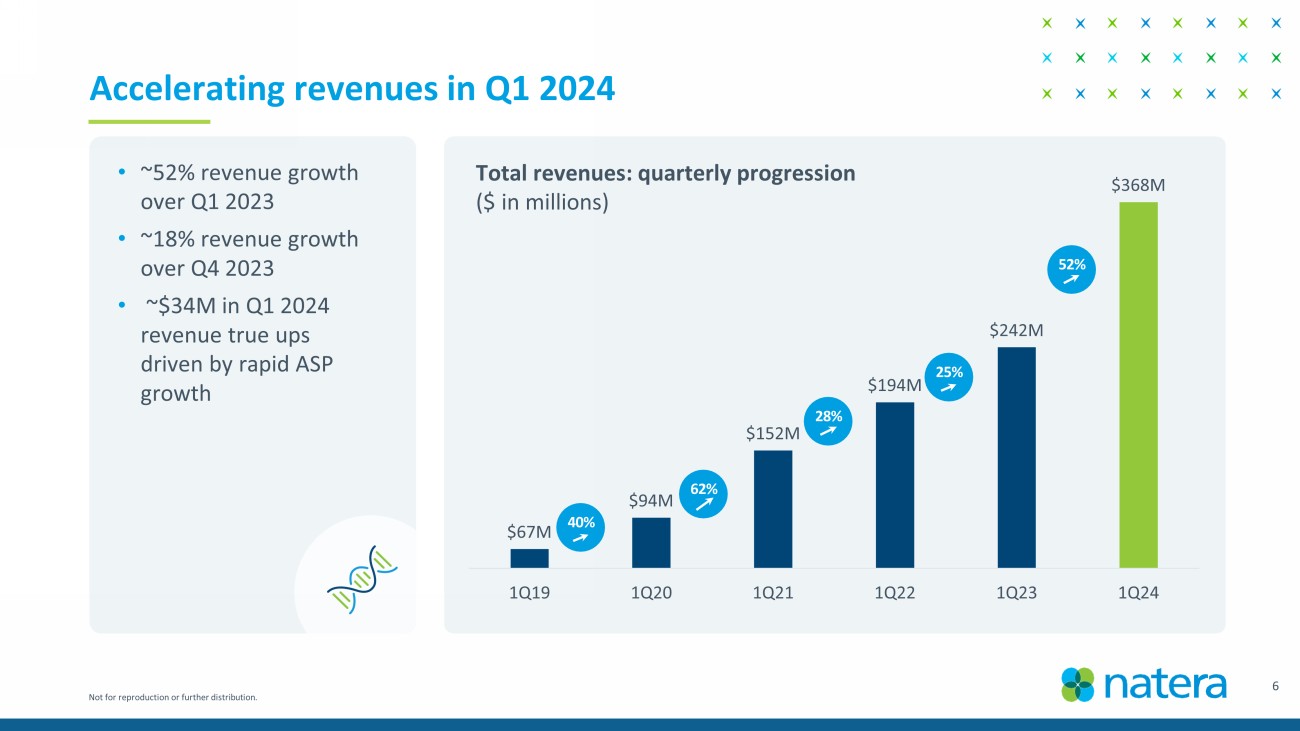
Not for reproduction or further distribution. 6 Accelerating revenues in Q1 2024 $67M $94M $152M $194M $242M $368M 1Q19 1Q20 1Q21 1Q22 1Q23 1Q24 62 % 28 % 25 % 52 % 40 % • ~ 52% revenue growth over Q1 2023 • ~18% revenue growth over Q4 2023 • ~$34M in Q1 2024 revenue true ups driven by rapid ASP growth Total revenues: quarterly progression ($ in millions)
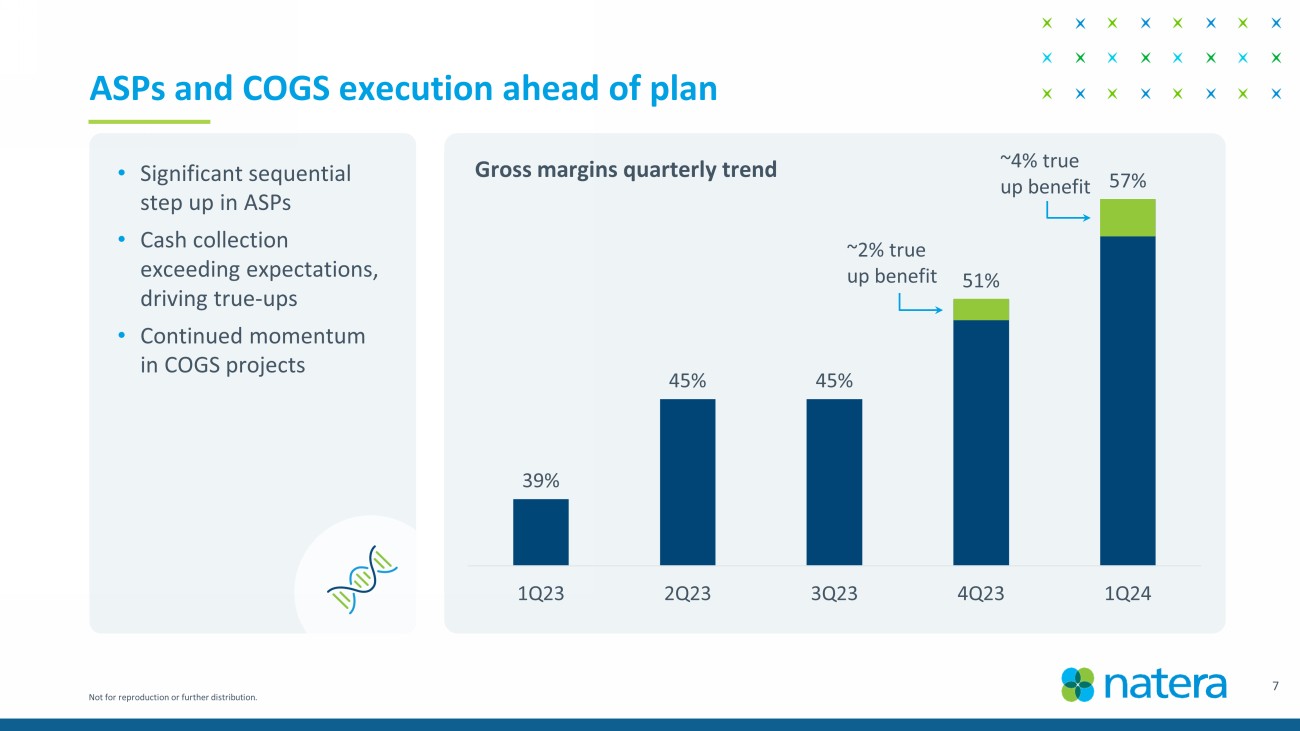
Not for reproduction or further distribution. 7 ASPs and COGS execution ahead of plan • Significant sequential step up in ASPs • Cash collection exceeding expectations, driving true - ups • Continued momentum in COGS projects 39% 45% 45% 51% 57% 1Q23 2Q23 3Q23 4Q23 1Q24 ~2% true up benefit ~4% true up benefit Gross margins quarterly trend
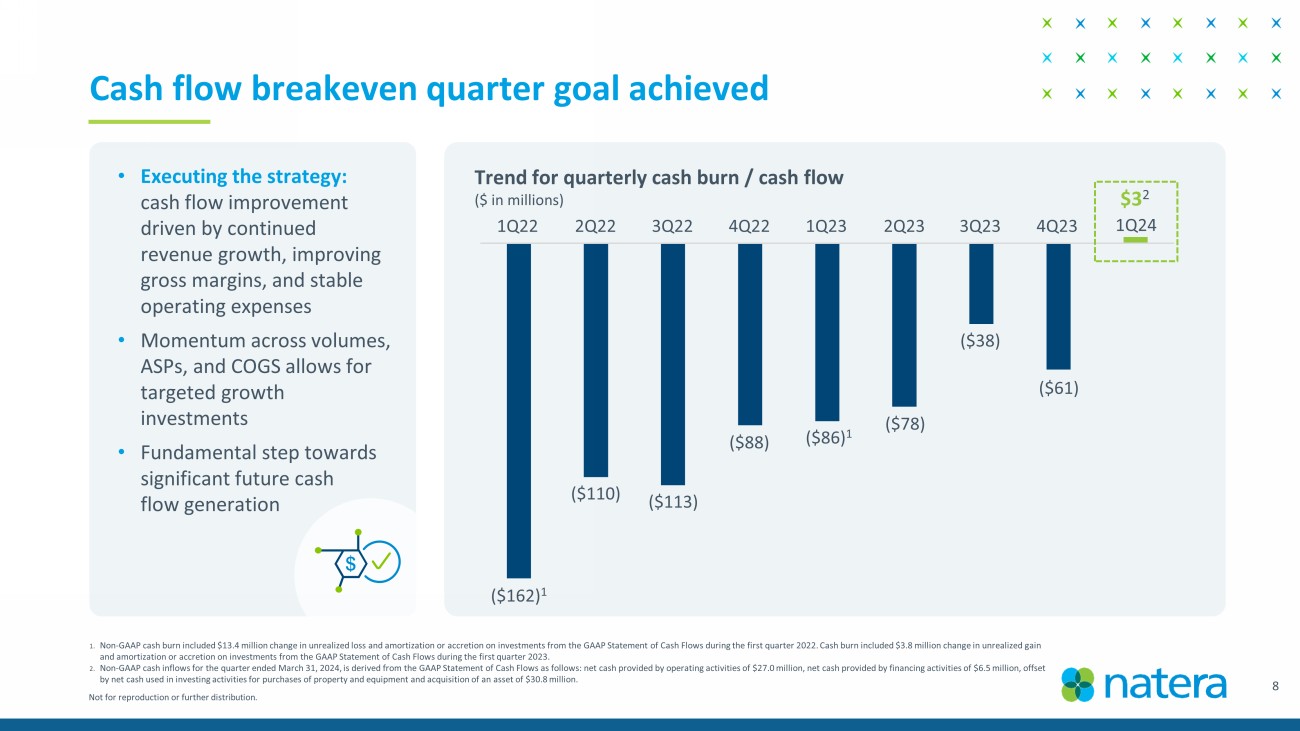
Not for reproduction or further distribution. 8 1. Non - GAAP cash burn included $13.4 million change in unrealized loss and amortization or accretion on investments from the GAAP S tatement of Cash Flows during the first quarter 2022. Cash burn included $3.8 million change in unrealized gain and amortization or accretion on investments from the GAAP Statement of Cash Flows during the first quarter 2023. 2. Non - GAAP cash inflows for the quarter ended March 31, 2024, is derived from the GAAP Statement of Cash Flows as follows: net cas h provided by operating activities of $27.0 million, net cash provided by financing activities of $6.5 million, offset by net cash used in investing activities for purchases of property and equipment and acquisition of an asset of $30.8 million . Cash flow breakeven quarter goal achieved • Executing the strategy: cash flow improvement driven by continued revenue growth, improving gross margins, and stable operating expenses • Momentum across volumes, ASPs, and COGS allows for targeted growth investments • Fundamental step towards significant future cash flow generation ($162) 1 ($110) 1Q22 2Q22 3Q22 4Q22 1Q23 2Q23 3Q23 4Q23 1Q24 ($113) ($88) ($86) 1 ($78) ($38) ($61) $3 2 Trend for quarterly cash burn / cash flow ($ in millions)
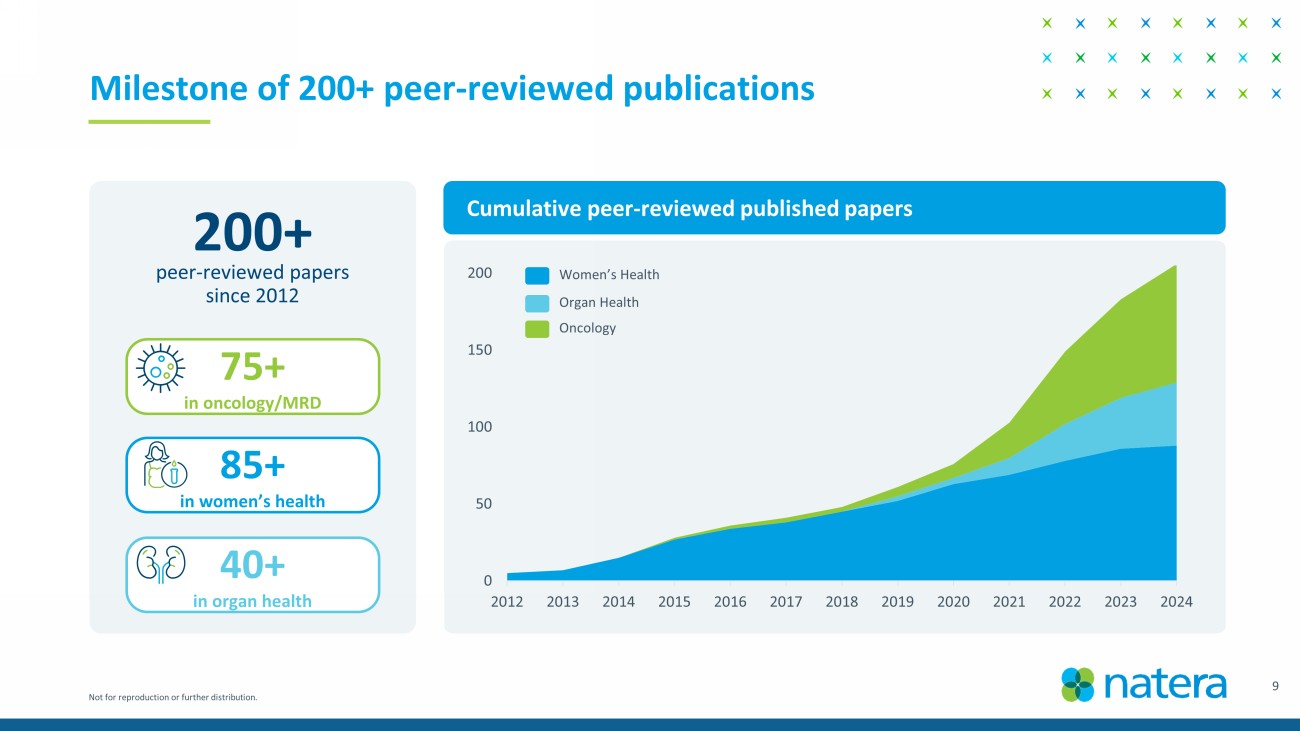
Not for reproduction or further distribution. 9 Milestone of 200+ peer - reviewed publications Cumulative peer - reviewed published papers 200+ peer - reviewed papers since 2012 75+ in oncology/MRD 0 50 100 150 200 2012 2013 2014 2015 2016 2017 2018 2019 2020 2021 2022 2023 2024 Women’s Health Organ Health Oncology 85+ in women’s health 40+ in organ health

Not for reproduction or further distribution. 10 New fetal RhD test supports physicians & patients during RhIg shortage Confidential Draft “… the use of NIPT to prioritize use of RhIg and conserve RhIg supply is a reasonable consideration ...” Leverages Natera’s core technology Backed by large, clinical validation study Further differentiates Natera >650 patients 100% sensitivity >99% specificity -- The American College of Obstetricians and Gynecologists (ACOG) in a Practice Advisory updated on April 24, 2024

Not for reproduction or further distribution. 11 KDIGO guidelines support genetic testing in majority of patients with CKD to establish cause of disease Confidential Draft Supports use of genetic testing Clinical indications impacting vast majority of CKD patients • KDIGO states that genetic tests should be used, among other factors, as a diagnostic tool to establish cause of CKD. • KDIGO notes that genetic testing can impact clinical management of people with CKD. • KDIGO recommends clinical indications where genetic testing can be “particularly informative” - Conditions with high prevalence of monogenic subtypes within the clinical category; - Early age of onset of CKD; - Syndromic/multisystem features; - Consanguinity; - Possibility of identifying a condition amenable to targeted treatment; and - CKD/ESRD of unknown etiology when kidney biopsy would not be informative due to advanced disease 1. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 Clinical Practice Guideline for the Evaluation a nd Management of Chronic Kidney Disease. Kidney Int. 2024;105(4S): S117 – S314 .

Not for reproduction or further distribution. 12 1. Sigdel TK, Archila FA, Constantin T, et al. Optimizing Detection of Kidney Transplant Injury by Assessment of Donor - Derived Cell - Free DNA via Mass ively Multiplex PCR. J Clin Med. 2018;8(1):19. 2. Halloran PF, Reeve J, Madill - Thomsen KS, et al. Combining Donor - derived Cell - free DNA Fraction and Quantity to Detect Kidney Tra nsplant Rejection Using Molecular Diagnoses and Histology as Confirmation. Transplantation. 2022;106(12): 2435 - 2442. 3. Bunnapradist S, Homkrailas P, Ahmed E, Fehringer G, Billings P, Tabriziani H. Using both the Fraction and Quantity of Donor - Derived Cell - Free DNA to Detect Kidney Allograft Rejection. J. Am. Soc. Nephrol. 2021;32(10), 2439 - 2441. 4. Bromberg J, Bunnapradist S, Samaniego - Picota Milagros, et al. Elevation of Donor - derived Cell - free DNA Before Biopsy - proven Rejection in Kidney Transplant. Transplantation. 2024. Publication of ProActive , the largest prospective dd - cfDNA study in kidney transplantation Trial background • 1,631 kidney transplant patients | ≥ 18 months follow up |54 participating centers Key findings: Prospera TM is a leading indicator of rejection • Predicted ABMR up to 5 months and TCMR up to 2 months before biopsy - proven rejection 1 . • Serum creatinine levels were not significantly elevated at any time point before biopsy - proven TCMR or ABMR compared to non rejection. • Demonstrated an area under the curve of 0.88 and NPV of 98% , showcasing Prospera performance consistent with prior validations. 1 - 4
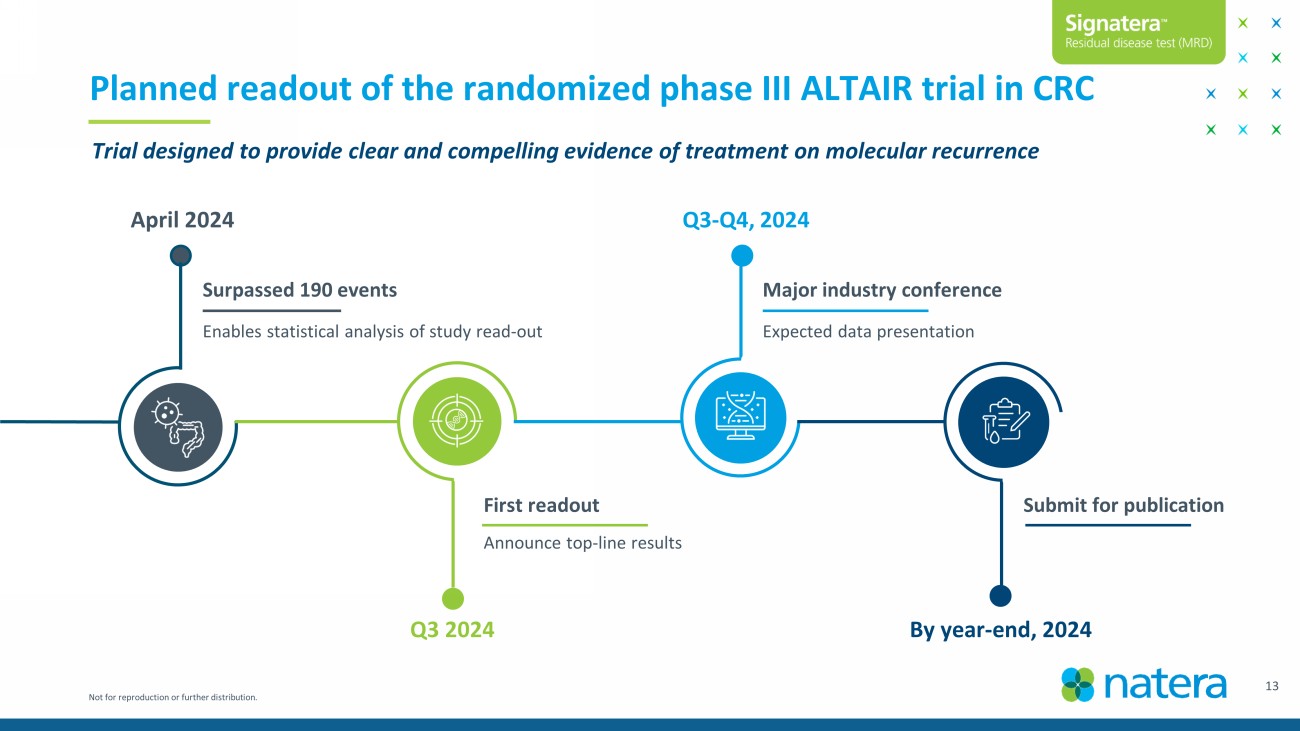
Not for reproduction or further distribution. 13 Planned readout of the randomized phase III ALTAIR trial in CRC Surpassed 190 events Major industry conference First readout Submit for publication Announce top - line results Expected data presentation Enables statistical analysis of study read - out April 2024 Q3 2024 Q3 - Q4, 2024 By year - end, 2024 Trial designed to provide clear and compelling evidence of treatment on molecular recurrence
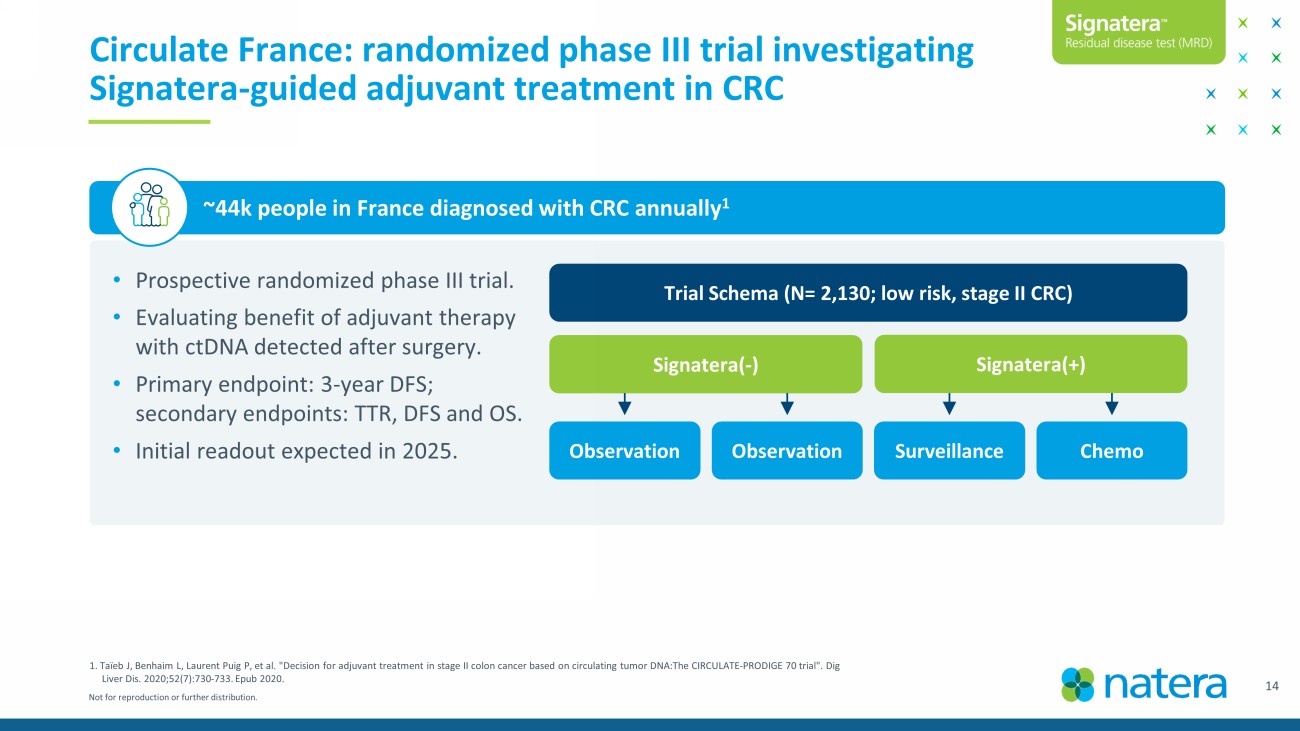
Not for reproduction or further distribution. 14 Circulate France: randomized phase III trial investigating Signatera - guided adjuvant treatment in CRC • Prospective randomized phase III trial. • Evaluating benefit of adjuvant therapy with ctDNA detected after surgery. • Primary endpoint: 3 - year DFS; secondary endpoints: TTR, DFS and OS. • Initial readout expected in 2025. ~44k people in France diagnosed with CRC annually 1 Trial Schema (N= 2,130; low risk, stage II CRC) Signatera ( - ) Signatera (+) Surveillance Chemo Observation Observation 1. Taïeb J, Benhaim L, Laurent Puig P, et al. "Decision for adjuvant treatment in stage II colon cancer based on circulating tumor DNA:The CIRCULATE - PRODIGE 70 trial". Dig Liver Dis. 2020;52(7):730 - 733. Epub 2020.
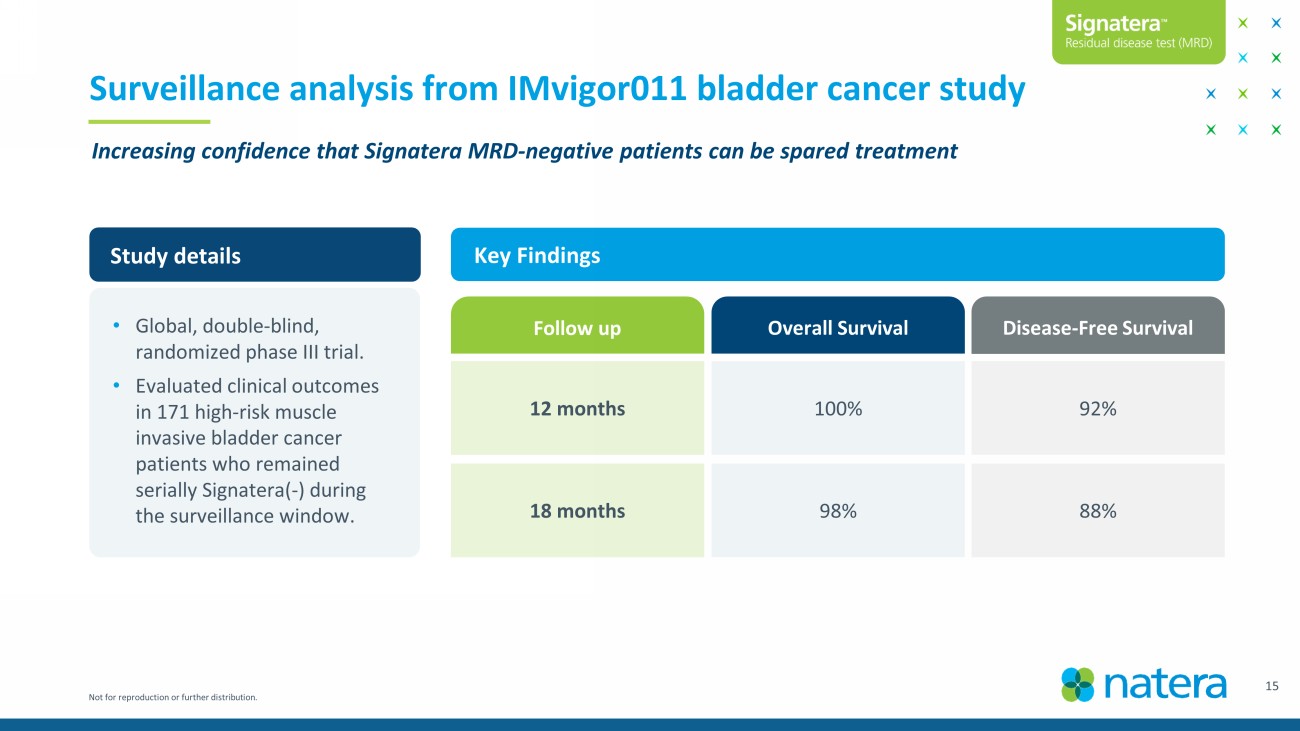
Not for reproduction or further distribution. 15 Surveillance analysis from IMvigor011 bladder cancer study Increasing confidence that Signatera MRD - negative patients can be spared treatment Study details Key Findings • Global, double - blind, randomized phase III trial. • Evaluated clinical outcomes in 171 high - risk muscle invasive bladder cancer patients who remained serially Signatera ( - ) during the surveillance window. Disease - Free Survival 92% 88% 100% 98% 12 months 18 months Follow up Overall Survival
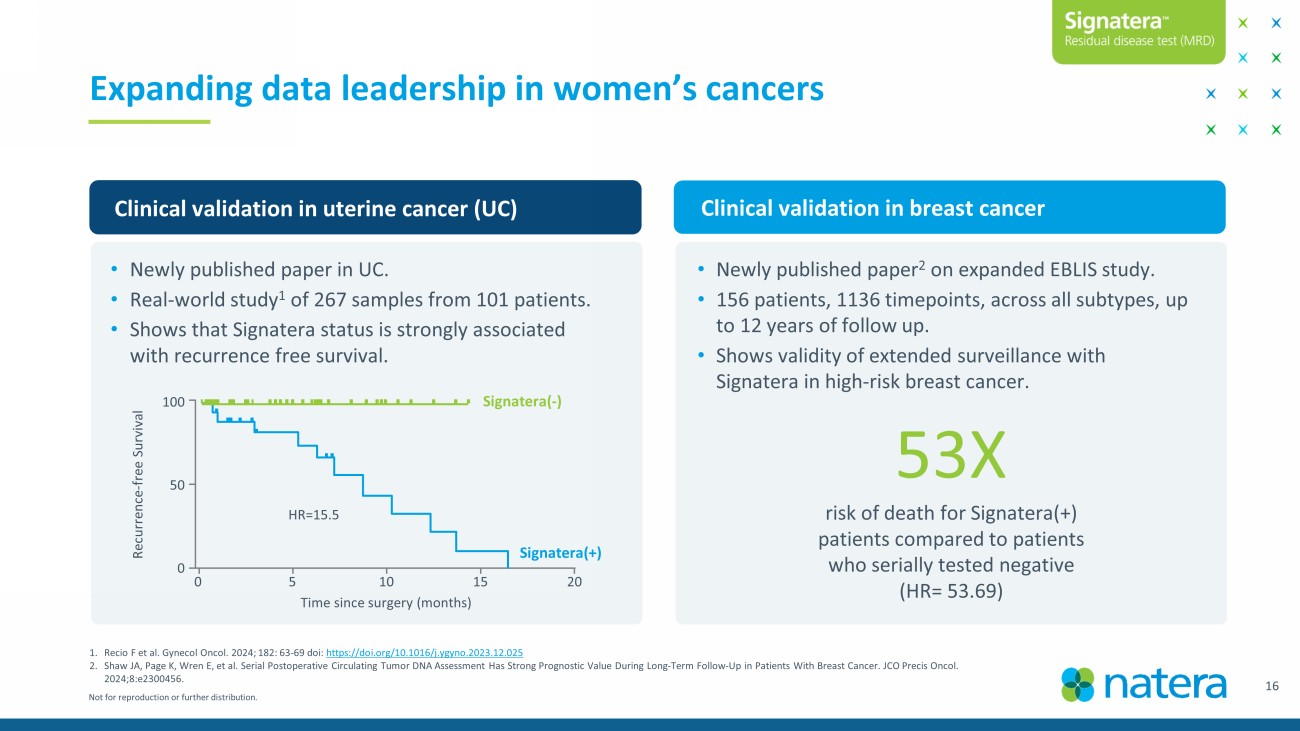
Not for reproduction or further distribution. 1. Recio F et al. Gynecol Oncol. 2024; 182: 63 - 69 doi : https://doi.org/10.1016/j.ygyno.2023.12.025 2. Shaw JA, Page K, Wren E, et al. Serial Postoperative Circulating Tumor DNA Assessment Has Strong Prognostic Value During Long - Term Follow - Up in Patients With Breast Cancer. JCO Precis Oncol. 2024;8:e2300456. Expanding data leadership in women’s cancers Clinical validation in uterine cancer (UC) Clinical validation in breast cancer • Newly published paper in UC. • Real - world study 1 of 267 samples from 101 patients. • Shows that Signatera status is strongly associated with recurrence free survival. • Newly published paper 2 on expanded EBLIS study. • 156 patients, 1136 timepoints, across all subtypes, up to 12 years of follow up. • Shows validity of extended surveillance with Signatera in high - risk breast cancer. 16 Recurrence - free Survival 100 50 0 HR=15.5 Time since surgery (months) 0 5 15 20 10 Signatera (+) Signatera ( - ) 53X risk of death for Signatera (+) patients compared to patients who serially tested negative (HR= 53.69)
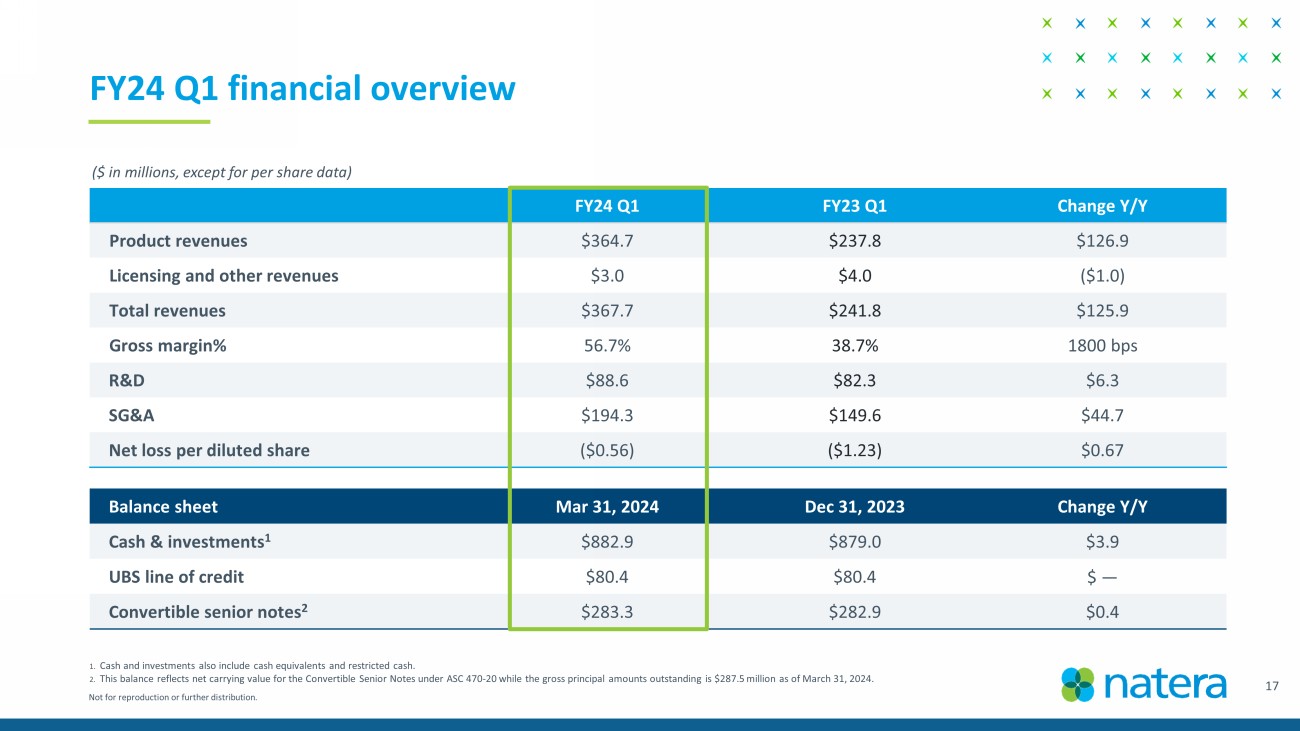
Not for reproduction or further distribution. FY24 Q1 financial overview 17 1. Cash and investments also include cash equivalents and restricted cash. 2. This balance reflects net carrying value for the Convertible Senior Notes under ASC 470 - 20 while the gross principal amounts out standing is $287.5 million as of March 31, 2024. ($ in millions, except for per share data) Balance sheet Mar 31, 2024 Dec 31, 2023 Change Y/Y Cash & investments 1 $882.9 $879.0 $3.9 UBS line of credit $80.4 $80.4 $ — Convertible senior notes 2 $283.3 $282.9 $0.4 FY24 Q1 FY23 Q1 Change Y/Y Product revenues $364.7 $237.8 $126.9 Licensing and other revenues $3.0 $4.0 ($1.0) Total revenues $367.7 $241.8 $125.9 Gross margin% 56.7% 38.7% 1800 bps R&D $88.6 $82.3 $6.3 SG&A $194.3 $149.6 $44.7 Net loss per diluted share ($0.56) ($1.23) $0.67
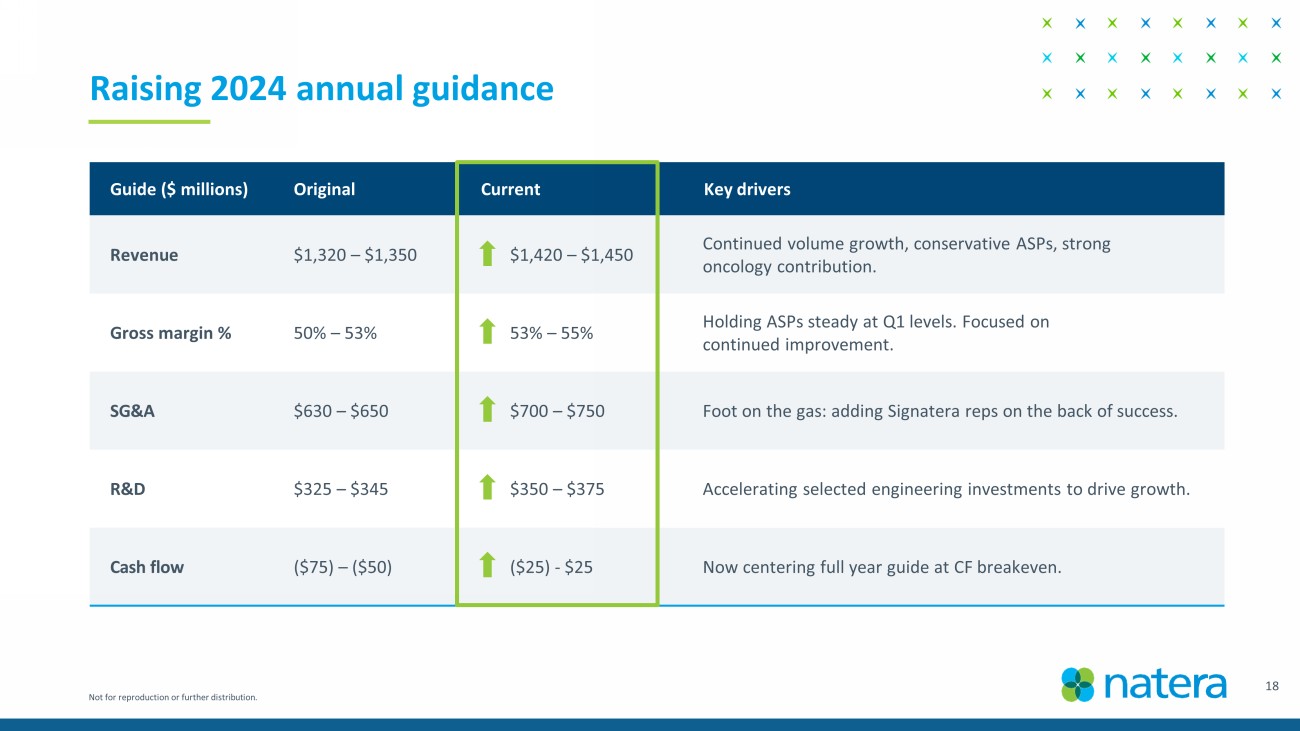
Not for reproduction or further distribution. Guide ($ millions) Original Current Key drivers Revenue $ 1,320 – $1, 350 $ 1,420 – $1, 450 Continued volume growth, conservative ASPs, strong oncology contribution. Gross margin % 50 % – 53 % 53 % – 55 % Holding ASPs steady at Q1 levels. Focused on continued improvement. SG&A $ 630 – $ 650 $ 700 – $ 750 Foot on the gas: adding Signatera reps on the back of success. R&D $325 – $345 $ 350 – $ 375 Accelerating selected engineering investments to drive growth. Cash flow ($ 75) – ($ 50) ( $25) - $25 Now centering full year guide at CF breakeven. 18 Raising 2024 annual guidance

Not for reproduction or further distribution. 19 Executing on plan: flashback to Q1’22 earnings call Exact slide from Natera’s Q1’22 earnings presentation

Not for reproduction or further distribution. 20 Upside potential with future catalysts in 2024 Potential Catalysts Continued execution on ASPs, COGS, volumes Guideline inclusion of women’s health products ALTAIR top - line results in Q3 Additional MolDx coverage for Signatera Potential uplift from biomarker states Product launches in women’s health and oncology

©202 4 Natera, Inc. All Rights Reserved. Not for reproduction or further distribution. ® 21
v3.24.1.u1
Cover
|
May 09, 2024 |
| Cover [Abstract] |
|
| Document Type |
8-K
|
| Amendment Flag |
false
|
| Document Period End Date |
May 09, 2024
|
| Entity File Number |
001-37478
|
| Entity Registrant Name |
Natera, Inc.
|
| Entity Central Index Key |
0001604821
|
| Entity Tax Identification Number |
01-0894487
|
| Entity Incorporation, State or Country Code |
DE
|
| Entity Address, Address Line One |
13011 McCallen Pass
|
| Entity Address, Address Line Two |
Building A Suite 100
|
| Entity Address, City or Town |
Austin
|
| Entity Address, State or Province |
TX
|
| Entity Address, Postal Zip Code |
78753
|
| City Area Code |
650
|
| Local Phone Number |
980-9190
|
| Written Communications |
false
|
| Soliciting Material |
false
|
| Pre-commencement Tender Offer |
false
|
| Pre-commencement Issuer Tender Offer |
false
|
| Title of 12(b) Security |
Common Stock, par value $0.0001 per share
|
| Trading Symbol |
NTRA
|
| Security Exchange Name |
NASDAQ
|
| Entity Emerging Growth Company |
false
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Natera (NASDAQ:NTRA)
Historical Stock Chart
From Aug 2024 to Sep 2024
Natera (NASDAQ:NTRA)
Historical Stock Chart
From Sep 2023 to Sep 2024