AbbVie Gets FDA Approval for Rinvoq for Ages 12 and Older
15 January 2022 - 6:04AM
Dow Jones News
By Chris Wack
AbbVie Inc. said the U.S. Food and Drug Administration has
approved Rinvoq upadacitinib for the treatment of moderate to
severe atopic dermatitis in adults and children 12 years of age and
older.
The company said Rinvoq 15 mg once daily can now be initiated in
adults and children 12 years of age and older weighing at least 40
kg. In those children and adults less than 65 years of age who
don't achieve an adequate response, the dose may be increased to 30
mg once daily.
AbbVie said the FDA approval is supported by efficacy and safety
data from one of the largest registrational Phase 3 programs for
atopic dermatitis, with more than 2,500 patients evaluated across
three studies. About 52% of the patients had prior exposure to
systemic atopic dermatitis treatment.
These studies evaluated the efficacy and safety of Rinvoq
monotherapy and with topical corticosteroids, compared to placebo,
in adults and children 12 years of age and older with moderate to
severe atopic dermatitis.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
January 14, 2022 13:49 ET (18:49 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
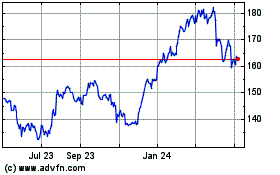
AbbVie (NYSE:ABBV)
Historical Stock Chart
From Mar 2024 to Apr 2024
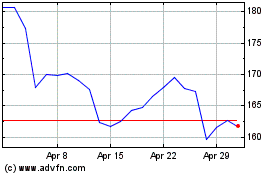
AbbVie (NYSE:ABBV)
Historical Stock Chart
From Apr 2023 to Apr 2024