Aurora Spine Corporation Announces FDA 510(k) Clearance for its SiLO TFX™ MIS Sacroiliac Joint Fixation System
06 October 2022 - 10:15PM

Aurora Spine Corporation ("Aurora Spine" or the "Company") (TSXV:
ASG) (OTCQB: ASAPF), a designer and manufacturer of innovative
medical devices that improve spinal surgery outcomes, today
announced it has received 510(k) clearance from the United States
Food and Drug Administration (FDA) for the patented minimally
invasive SiLO TFX MIS Sacroiliac Joint Fixation System. The Aurora
Spine SiLO TFX MIS Sacroiliac Joint Fixation System, is intended
for sacroiliac joint fusion for conditions including sacroiliac
joint disruptions and degenerative sacroiliitis. The SiLO TFX MIS
Sacroiliac Joint Fixation System includes a Transfixing-Cone, an
ilium screw, a sacrum screw and associated instrumentation. The
SiLO TFX implants are designed to transfix the sacrum and ilium,
providing stability for bony fusion.
“We are pleased with our continued commercial momentum as we
expand our footprint for providing differentiated surgical devices
in the sacropelvic/sacroiliac space,” said Trent Northcutt,
President, and Chief Executive Officer. “We will make meaningful
investments in our sales force and physician training, which we
believe will enable us to further capitalize on the growth in
outpatient surgery centers across the United States.”
Mr. Laszlo Garamszegi, Chief Technology Officer of Aurora Spine,
added, "We are excited about this patented game-changing
technology. Increased awareness of SI joint disruption as a
significant contributor to back pain led us to develop a minimally
invasive solution to treat the condition. It is a breakthrough for
Aurora to offer a titanium version of our SiLO family of products,
which offers additional fixation options to transfix the
joint.”
"This is a significant milestone in the evolution of the
treatment of SI joint dysfunction and clearly sets SiLO TFX apart
from any other surgical options. This patented system transfixes
the sacrum and ilium to provide superior clinical outcomes," said
Dr. Michael Stoffman, Neurosurgeon at the University at Buffalo
Neurosurgery. "The SiLO TFX clearance is an essential milestone for
Aurora Spine as they are committed to helping patients experiencing
chronic SI Joint pain."
About Aurora Spine
Aurora Spine is focused on bringing new solutions to the spinal
implant market through a series of innovative, minimally invasive,
regenerative spinal implant technologies. Additional information
can be accessed at www.aurora-spine.com or
www.aurorapaincare.com.
Neither TSX Venture Exchange nor its Regulation Services
Provider (as that term is defined in the policies of the TSX
Venture Exchange) accepts responsibility for the adequacy or
accuracy of this release.
Forward-Looking Statements
This news release contains forward-looking information that
involves substantial known and unknown risks and uncertainties,
most of which are beyond the control of Aurora Spine, including,
without limitation, those listed under "Risk Factors" and
"Cautionary Statement Regarding Forward-Looking Information" in
Aurora Spine's final prospectus (collectively, "forward-looking
information"). Forward-looking information in this news release
includes information concerning the proposed use and success of the
company’s products in surgical procedures. Aurora Spine cautions
investors of Aurora Spine's securities about important factors that
could cause Aurora Spine's actual results to differ materially from
those projected in any forward-looking statements included in this
news release. Any statements that express, or involve discussions
as to, expectations, beliefs, plans, objectives, assumptions or
future events or performance are not historical facts and may be
forward-looking and may involve estimates, assumptions and
uncertainties which could cause actual results or outcomes to
differ unilaterally from those expressed in such forward-looking
statements. No assurance can be given that the expectations set out
herein will prove to be correct and, accordingly, prospective
investors should not place undue reliance on these forward-looking
statements. These statements speak only as of the date of this
press release and Aurora Spine does not assume any obligation to
update or revise them to reflect new events or circumstances.
Contact:
Aurora Spine Corporation
Trent Northcutt
President and Chief Executive Officer
(760) 424-2004
Chad Clouse
Chief Financial Officer
(760) 424-2004
www.aurora-spine.com
Adam Lowensteiner
LYTHAM PARTNERS, LLC
Phoenix | New York
Telephone: 646-829-9700
asapf@lythampartners.com
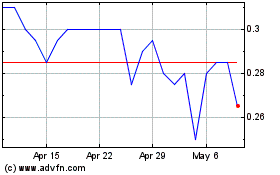
Aurora Spine (TSXV:ASG)
Historical Stock Chart
From Mar 2024 to Apr 2024
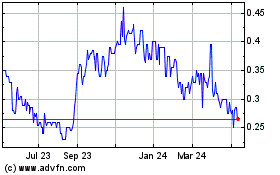
Aurora Spine (TSXV:ASG)
Historical Stock Chart
From Apr 2023 to Apr 2024