- Five new launches reinforce Baxter’s focus on differentiated
products and enhance the company’s Pharmaceuticals portfolio in
critical therapeutic areas
- Products feature ready-to-use formulations to help support
patient safety and offer added convenience for healthcare
professionals
Baxter International Inc. (NYSE:BAX), a global leader in
injectables, anesthesia and drug compounding, announced the
continued expansion of its Pharmaceuticals portfolio with five
injectable product launches in the U.S.
“These launches demonstrate Baxter’s focus on differentiated
products that address unmet patient needs in key therapeutic areas,
including anti-infective and anti-hypotensive medications,” said
Alok Sonig, executive vice president and group president,
Pharmaceuticals, at Baxter. “We are proud to offer important new
options for our customers and look forward to continuing to bring
new innovations to the market.”
Product launches within Baxter’s Pharmaceuticals portfolio in
the U.S. include the following. For all products, please see full
Indications, Important Risk Information and links to full
Prescribing Information below.
- Norepinephrine Bitartrate in 5% Dextrose Injection in a new 16
mg/250 mL strength, which is indicated to raise blood pressure in
adult patients with severe, acute hypotension. Baxter offers the
first and only FDA-approved ready-to-use Norepinephrine in
Dextrose, and now provides Norepinephrine in 4 mg/250 mL, 8 mg/250
mL and 16 mg/250 mL strengths.
- Vasopressin in 0.9% Sodium Chloride Injection, the first and
only FDA-approved ready-to-use Vasopressin in a flexible container.
Vasopressin uses Baxter’s proprietary sterile, closed system
container, which is collapsible and does not require a vented
intravenous set.1 Manufactured closed drug-delivery systems
decrease the need for manual admixture and reduce the possibility
of contamination.2 Vasopressin is indicated to increase blood
pressure in adults with vasodilatory shock who remain hypotensive
despite fluids and catecholamines. Baxter offers Vasopressin in 20
units/100 mL and 40 units/100 mL strengths.
- Vancomycin Injection, USP in 5% Dextrose in new 1.25 g/250 mL
and 1.5 g/300 mL strengths. These launches are Baxter’s first
frozen ready-to-use offerings in 250 mL and 300 mL volumes.
Vancomycin is indicated for serious or severe infections caused by
susceptible strains of methicillin-resistant (β-lactam-resistant)
staphylococci. Baxter now offers Vancomycin in 500 mg/100 mL, 750
mg/150 mL, 1 g/200 mL, 1.25 g/250 mL and 1.5 g/300 mL
strengths.
- Ropivacaine Hydrochloride Injection, USP in a ready-to-use,
single-dose infusion bag. Ropivacaine is indicated in adults to
produce local or regional anesthesia for surgery and for acute pain
management. Baxter offers Ropivacaine in 200 mg/100 mL and 400
mg/200 mL strengths.
- Regadenoson Injection pre-filled syringe, a coronary
vasodilator that is commonly used in pharmacologic stress testing.
Baxter offers Regadenoson in a 0.4 mg/5 mL strength.
Ready-to-use formats of standard concentrations of commonly
prescribed drugs may offer operational efficiencies for healthcare
providers. Compounding a drug for patient use is a multi-step,
manual process that requires oversight by pharmacy staff. A
ready-to-use product can simplify the preparation process and
support patient safety by reducing the chance of contamination2 and
avoiding potential errors that may occur when medications are
compounded.3
These newly launched products are now available for use in the
U.S.
About Baxter
Every day, millions of patients, caregivers and healthcare
providers rely on Baxter’s leading portfolio of diagnostic,
critical care, kidney care, nutrition, hospital and surgical
products used across patient homes, hospitals, physician offices
and other sites of care. For more than 90 years, we’ve been
operating at the critical intersection where innovations that save
and sustain lives meet the healthcare providers who make it happen.
With products, digital health solutions and therapies available in
more than 100 countries, Baxter’s employees worldwide are now
building upon the company’s rich heritage of medical breakthroughs
to advance the next generation of transformative healthcare
innovations. To learn more, visit www.baxter.com and follow us on
X/Twitter, LinkedIn and Facebook.
Norepinephrine Bitartrate in 5% Dextrose Injection
Indications
Norepinephrine Bitartrate in Dextrose Injection is indicated to
raise blood pressure in adult patients with severe, acute
hypotension.
Important Risk Information
- Contraindications: None.
- Tissue Ischemia: Administration of Norepinephrine Bitartrate in
Dextrose Injection to patients who are hypotensive from hypovolemia
can result in severe peripheral and visceral vasoconstriction,
decreased renal perfusion and reduced urine output, tissue hypoxia,
lactic acidosis, and reduced systemic blood flow despite “normal”
blood pressure. Address hypovolemia prior to initiating
Norepinephrine Bitartrate in Dextrose Injection. Avoid use in
patients with mesenteric or peripheral vascular thrombosis, as this
may increase ischemia and extend the area of infarction. Gangrene
of the extremities has occurred in patients with occlusive or
thrombotic vascular disease or who received prolonged or high dose
infusions. Monitor for changes to the skin of the extremities in
susceptible patients. Extravasation of Norepinephrine Bitartrate in
Dextrose Injection may cause necrosis and sloughing of surrounding
tissue. To reduce the risk of extravasation, infuse into a large
vein, check the infusion site frequently for free flow, and monitor
for signs of extravasation. Avoid administration into the veins in
the leg in elderly patients. Emergency
Treatment of Extravasation: Infiltrate the ischemic area as
soon as possible, using a syringe with a fine hypodermic needle
with 5 to 10 mg of phentolamine mesylate in 10 to 15 mL of 0.9%
Sodium Chloride Injection in adults.
- Hypotension after Abrupt Discontinuation: Sudden cessation of
the infusion rate may result in marked hypotension. When
discontinuing the infusion, gradually reduce the infusion rate
while expanding blood volume with intravenous fluids.
- Cardiac Arrhythmias: Norepinephrine Bitartrate in Dextrose
Injection elevates intracellular calcium concentrations and may
cause arrhythmias, particularly in the setting of hypoxia or
hypercarbia. Perform continuous cardiac monitoring of patients with
arrhythmias.
- Elderly Patients: May be at a greater risk of developing
adverse reactions.
- Adverse Reactions: Most common adverse reactions are
hypertension and bradycardia.
- Drug Interactions:
- Co-administration of Norepinephrine Bitartrate in Dextrose
Injection with monoamine oxidase (MAO) inhibitors or other drugs
with MAO-inhibiting properties (e.g., linezolid) or with tricyclic
antidepressants can cause severe, prolonged hypertension.
- Anti-diabetics: Norepinephrine Bitartrate in Dextrose Injection
can decrease insulin sensitivity and raise blood glucose.
- Concomitant use of Norepinephrine Bitartrate in Dextrose
Injection with halogenated anesthetics may lead to ventricular
tachycardia or ventricular fibrillation. Monitor cardiac rhythm in
patients receiving concomitant halogenated anesthetics.
Please see accompanying full Prescribing Information for
Norepinephrine Bitartrate in 5% Dextrose Injection.
Vasopressin in 0.9% Sodium Chloride Injection
Indications
Vasopressin in Sodium Chloride Injection is indicated to
increase blood pressure in adults with vasodilatory shock who
remain hypotensive despite fluids and catecholamines.
Important Risk Information
- Contraindications: Vasopressin in Sodium Chloride Injection is
contraindicated in patients with a known allergy or
hypersensitivity to 8-L-arginine vasopressin.
- Worsening Cardiac Function: A decrease in cardiac index may be
observed with the use of vasopressin.
- Reversible Diabetes Insipidus: Patients may experience
reversible diabetes insipidus, manifested by the development of
polyuria, a dilute urine, and hypernatremia, after cessation of
treatment with vasopressin. Monitor serum electrolytes, fluid
status and urine output after vasopressin discontinuation. Some
patients may require readministration of vasopressin or
administration of desmopressin to correct fluid and electrolyte
shifts.
- Adverse Reactions:
- The most common adverse reactions include decreased cardiac
output, bradycardia, tachyarrhythmias, hyponatremia and ischemia
(coronary, mesenteric, skin, digital).
- Drug Interactions:
- Pressor effects of catecholamines and Vasopressin in Sodium
Chloride Injection are expected to be additive.
- Indomethacin may prolong effects of Vasopressin in Sodium
Chloride Injection.
- Co-administration of ganglionic blockers or drugs causing SIADH
(syndrome of inappropriate antidiuretic hormone secretion) may
increase the pressor response.
- Co-administration of drugs causing diabetes insipidus may
decrease the pressor response.
- Pregnancy: May induce tonic uterine contractions that could
threaten the continuation of pregnancy.
Please see accompanying full Prescribing Information for
Vasopressin in 0.9% Sodium Chloride Injection.
Vancomycin Injection, USP in 5% Dextrose
Indications
Vancomycin is indicated for the treatment of serious or severe
infections caused by susceptible strains of methicillin-resistant
(beta-lactam-resistant) staphylococci. It is indicated for
penicillin-allergic patients, for patients who cannot receive or
who have failed to respond to other drugs, and for infections
caused by vancomycin-susceptible organisms that are resistant to
other antimicrobial drugs.
Vancomycin is effective in the treatment of:
- Infective Endocarditis (staphylococcal endocarditis;
endocarditis caused by Streptococcus viridans or S. bovis, alone or
in combination with an aminoglycoside; endocarditis caused by
enterococci (e.g., E. faecalis), only in combination with an
aminoglycoside; diphtheroid endocarditis; early-onset prosthetic
valve endocarditis caused by S. epidermidis or diphtheroids in
combination with either rifampin, an aminoglycoside, or both)
- Septicemia
- Skin and Skin Structure Infections
- Bone Infections
- Lower Respiratory Tract Infections
To reduce the development of drug-resistant bacteria and
maintain the effectiveness of vancomycin and other antibacterial
drugs, vancomycin should be used only to treat or prevent
infections that are proven or strongly suspected to be caused by
susceptible bacteria.
Important Risk Information
- Contraindications: Vancomycin is contraindicated in patients
with known hypersensitivity to this antibiotic. Solutions
containing dextrose may be contraindicated in patients with known
allergy to corn or corn products.
- Infusion Reactions: Rapid bolus administration (e.g., over
several minutes) may be associated with exaggerated hypotension,
including shock, and, rarely, cardiac arrest. During or soon after
rapid infusion of vancomycin, patients may develop anaphylactoid
reactions, including hypotension, wheezing, dyspnea, urticaria, or
pruritus. Rapid infusion may also cause flushing of the upper body
(“vancomycin infusion reaction”) or pain and muscle spasm of the
chest and back. Vancomycin should be administered over a period of
not less than 60 minutes. Stopping the infusion usually results in
prompt cessation of these reactions.
- Nephrotoxicity: Systemic vancomycin exposure may result in
acute kidney injury (AKI). The risk of AKI increases as systemic
exposure/serum levels increase. Monitor renal function in all
patients; especially with underlying renal impairment, with
co-morbidities, and receiving concomitant therapy with a known
nephrotoxic drug.
- Ototoxicity: It may be transient or permanent. It has been
reported mostly in patients who have been given excessive doses,
who have an underlying hearing loss, or who are receiving
concomitant therapy with another ototoxic agent, such as an
aminoglycoside. Vancomycin should be used with caution in patients
with renal insufficiency.
- Severe Dermatologic Reactions: Toxic epidermal necrolysis
(TEN), Stevens-Johnson syndrome (SJS), drug reaction with
eosinophilia and systemic symptoms (DRESS), acute generalized
exanthematous pustulosis (AGEP), and linear IgA bullous dermatosis
(LABD) have been reported. Cutaneous signs or symptoms reported
include skin rashes, mucosal lesions, and blisters. Discontinue
vancomycin Injection at the first appearance of signs and symptoms
of TEN, SJS, DRESS, AGEP, or LABD. Dosage of vancomycin must be
adjusted for patients with renal dysfunction.
- Clostridioides difficile associated diarrhea (CDAD): May range
in severity from mild diarrhea to fatal colitis. CDAD must be
considered in all patients who present with diarrhea following
antibiotic use. If CDAD is suspected or confirmed, ongoing
antibiotic use not directed against C. difficile may need to be
discontinued. Appropriate fluid and electrolyte management, protein
supplementation, antibiotic treatment of C. difficile, and surgical
evaluation should be instituted as clinically indicated.
- Hemorrhagic Occlusive Retinal Vasculitis: Including permanent
loss of vision, occurred in patients receiving intracameral or
intravitreal administration of vancomycin during or after cataract
surgery. The safety and efficacy of vancomycin administered by the
intracameral or the intravitreal route have not been
established.
- Adverse Reactions: Not already mentioned above, patients have
been reported to have neutropenia, phlebitis, drug fever, nausea,
chills, and vasculitis in association with administration of
vancomycin.
- Drug Interactions:
- Anesthetic Agents: Concomitant administration of vancomycin and
anesthetic agents has been associated with erythema and
histamine-like flushing and anaphylactoid reactions.
- Monitor renal function in patients receiving vancomycin and
concurrent and/or sequential systemic or topical use of other
potentially neurotoxic and/or nephrotoxic drugs, such as
amphotericin B, aminoglycosides, bacitracin, polymyxin B, colistin,
viomycin, or cisplatin.
Please see accompanying full Prescribing Information for
Vancomycin Injection, USP.
Ropivacaine Hydrochloride Injection, USP
Indications
Ropivacaine Hydrochloride Injection is an amide local anesthetic
indicated in adults for the production of local or regional
anesthesia for surgery and for acute pain management.
- Surgical Anesthesia: epidural
block for surgery including cesarean section; major nerve block;
local infiltration.
- Acute Pain Management: epidural
continuous infusion or intermittent bolus, e.g., postoperative or
labor; local infiltration.
Important Risk Information
- Contraindications: Ropivacaine Hydrochloride Injection is
contraindicated in patients with a known hypersensitivity to
ropivacaine or to any local anesthetic agent of the amide
type.
- General Warning: Delay in proper management of dose-related
toxicity, underventilation, and/or altered sensitivity may lead to
the development of acidosis, cardiac arrest and, possibly, death.
The safe and effective use of local anesthetics depends on proper
dosage, correct technique, adequate precautions, and readiness for
emergencies. It is essential that aspiration for blood, or
cerebrospinal fluid (where applicable), be done prior to injecting
any local anesthetic, both the original dose and all subsequent
doses, to avoid intravascular or subarachnoid injection.
- Unintended Intravenous Injection: In performing Ropivacaine
Hydrochloride Injection blocks, unintended intravenous injection is
possible and may result in cardiac arrhythmia or cardiac arrest.
Ropivacaine hydrochloride injection should be administered in
incremental doses. It is not recommended for emergency situations,
where a fast onset of surgical anesthesia is necessary.
- Intra-Articular Infusions and Risk of Chondrolysis:
Intra-articular infusions of local anesthetics may cause
chondrolysis. Ropivacaine Hydrochloride Injection is not approved
for this use.
- Risk of Methemoglobinemia: Patients with glucose-6-phosphate
dehydrogenase deficiency, congenital or idiopathic
methemoglobinemia, cardiac or pulmonary compromise, infants under 6
months of age, and concurrent exposure to oxidizing agents or their
metabolites are more susceptible to developing clinical
manifestations of the condition. If local anesthetics must be used
in these patients, close monitoring for symptoms and signs of
methemoglobinemia is recommended. Signs of methemoglobinemia may
occur immediately or may be delayed some hours after exposure.
Immediate treatment is required to avert more serious central
nervous system and cardiovascular adverse effects, including
seizures, coma, arrhythmias, and death. Discontinue ropivacaine
hydrochloride injection and any other oxidizing agents.
- Central Nervous System Toxicity: Careful and constant
monitoring of cardiovascular and respiratory vital signs and the
patient's state of consciousness should be performed after each
local anesthetic injection.
- Hepatic Disease: Because amide-type local anesthetics such as
ropivacaine are metabolized by the liver, these drugs, especially
repeat doses, should be used cautiously in patients with hepatic
disease. Patients with severe hepatic disease are at a greater risk
of developing toxic plasma concentrations.
- Adverse Reactions: Most common adverse reactions (incidence ≥
5%) are hypotension, nausea, vomiting, bradycardia, fever, pain,
postoperative complications, anemia, paresthesia, headache,
pruritus, and back pain.
- Drug Interactions:
- Agents structurally related to amide-type local anesthetics:
Concurrent use may cause additive effects.
Please see accompanying full Prescribing Information for
Ropivacaine Hydrochloride Injection, USP.
Regadenoson Injection
Indications
Regadenoson injection is a pharmacologic stress agent indicated
for radionuclide myocardial perfusion imaging (MPI) in patients
unable to undergo adequate exercise stress.
Important Risk Information
- Contraindications: Do not administer regadenoson injection to
patients with: Second- or third-degree AV block, or Sinus node
dysfunction, unless these patients have a functioning artificial
pacemaker.
- Myocardial Ischemia: Fatal cardiac events have occurred. Avoid
use in patients with symptoms or signs of acute myocardial
ischemia, for example unstable angina or cardiovascular
instability, who may be at greater risk. Cardiac resuscitation
equipment and trained staff should be available before
administration.
- Sinoatrial (SA) and Atrioventricular (AV) Nodal Block:
Adenosine receptor agonists, including regadenoson injection, can
depress the SA and AV nodes and may cause first-, second- or
third-degree AV block, or sinus bradycardia.
- Atrial Fibrillation/Atrial Flutter: New-onset or recurrent
atrial fibrillation with rapid ventricular response and atrial
flutter have been reported.
- Hypersensitivity, Including Anaphylaxis: Anaphylaxis,
angioedema, cardiac or respiratory arrest, respiratory distress,
decreased oxygen saturation, hypotension, throat tightness,
urticaria and rashes have occurred. Have personnel and
resuscitative equipment immediately available.
- Hypotension: Adenosine receptor agonists, including regadenoson
injection, induce vasodilation and hypotension. The risk of serious
hypotension may be higher in patients with autonomic dysfunction,
stenotic valvular heart disease, pericarditis or pericardial
effusions, stenotic carotid artery disease with cerebrovascular
insufficiency, or hypovolemia.
- Hypertension: Adenosine receptor agonists, including
regadenoson injection, may induce clinically significant increases
in blood pressure particularly in patients with a history of
hypertension and when the MPI includes low level exercise.
- Bronchoconstriction: Adenosine receptor agonists, including
regadenoson injection, may induce dyspnea, bronchoconstriction and
respiratory compromise, especially in patients with chronic
obstructive pulmonary disease (COPD) or asthma. Resuscitative
measures should be available.
- Seizure: Regadenoson injection may lower the seizure threshold.
New onset or recurrence of convulsive seizures has occurred. Some
seizures are prolonged and require urgent anticonvulsive
management. Methylxanthine use is not recommended in patients who
experience a seizure in association with regadenoson
injection.
- Cerebrovascular Accident (Stroke): Hemorrhagic and ischemic
cerebrovascular accidents have occurred.
- Adverse Reactions: The most common (incidence ≥ 5%) adverse
reactions to regadenoson injection are dyspnea, headache, flushing,
chest discomfort, dizziness, angina pectoris, chest pain, and
nausea.
- Drug Interactions:
- Methylxanthines, e.g., caffeine, aminophylline and
theophylline, interfere with the activity of regadenoson
injection.
- Aminophylline may be used to attenuate severe and/or persistent
adverse reactions to regadenoson injection.
- Dipyridamole may increase the activity of regadenoson
injection. When possible, withhold dipyridamole for at least two
days prior to regadenoson injection administration.
Please see accompanying full Prescribing Information for
Regadenoson Injection.
This release includes forward-looking statements concerning
Norepinephrine Bitartrate in 5% Dextrose Injection, Vasopressin in
0.9% Sodium Chloride Injection, Vancomycin Injection, USP in 5%
Dextrose, Ropivacaine Hydrochloride Injection, USP and Regadenoson
Injection pre-filled syringe, including potential benefits
associated with the use of these products. The statements are based
on assumptions about many important factors, including the
following, which could cause actual results to differ materially
from those in the forward-looking statements: demand for and market
acceptance for new and existing products; product development
risks; inability to create additional production capacity in a
timely manner or the occurrence of other manufacturing or supply
difficulties (including as a result of natural disasters, public
health crises and epidemics/pandemics, regulatory actions or
otherwise); satisfaction of regulatory and other requirements;
actions of regulatory bodies and other governmental authorities;
product quality, manufacturing or supply, or patient safety issues;
changes in law and regulations; and other risks identified in
Baxter's most recent filing on Form 10-K and Form 10-Q and other
SEC filings, all of which are available on Baxter's website. Baxter
does not undertake to update its forward-looking statements.
Baxter is a registered trademark of Baxter International
Inc.
1 Baxter data on file
2 Mercaldi CJ, Lanes S, Bradt J. Comparative risk of bloodstream
infection in hospitalized patients receiving intravenous medication
by open, point-of-care, or closed delivery systems. Am J
Health-Syst Pharm. 2013;70:957-965.
3 Billstein-Leber M, Carrillo CJD, Cassano AT, Moline K,
Robertson JJ. ASHP Guidelines on Preventing Medication Errors in
Hospitals. Am J Health Syst Pharm. 2018;75(19):1493-1517.
US-PH57-240005 (v1.0) 04/2024
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240411065937/en/
Media Contact Andrea Johnson, (224) 948-5353
media@baxter.com
Investor Contact Clare Trachtman, (224) 948-3020
Baxter (NYSE:BAX)
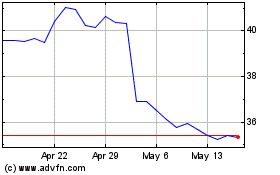
Historical Stock Chart
From May 2024 to Jun 2024
Baxter (NYSE:BAX)
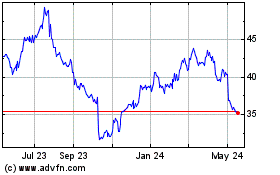
Historical Stock Chart
From Jun 2023 to Jun 2024