0001280776
false
0001280776
2023-11-14
2023-11-14
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of The Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
November 14, 2023
IMMUNIC, INC.
(Exact name of registrant as specified in its
charter)
| Delaware |
001-36201 |
56-2358443 |
(State or other jurisdiction
of incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
1200 Avenue of the Americas, Suite 200
New York, NY 10036
USA
(Address of principal executive offices)
Registrant’s telephone number, including
area code: (332) 255-9818
Check the appropriate box below if the Form 8-K filing is intended
to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ☐ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
Trading Symbol(s) |
Name of exchange on which registered |
| Common Stock, par value $0.0001 |
IMUX |
The Nasdaq Stock Market LLC |
Indicate by check mark whether the registrant is an emerging growth
company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange
Act of 1934 (§ 240.12b2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant
has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant
to Section 13(a) of the Exchange Act. Yes ☐ No ☐
Item 2.02. Results of Operations and Financial
Condition
On November 14, 2023, Immunic, Inc. (the “Company”)
issued a press release, a copy of which is furnished herewith as Exhibit 99.1, announcing the Company’s financial results for the
quarter ended September 30, 2023 and providing a corporate update (the “Press Release”).
The information contained in Item 2.02 of this Current
Report on Form 8-K, including the Press Release, shall not be deemed “filed” for the purposes of Section 18 of the Securities
Exchange Act of 1934, as amended, or otherwise subject to the liability of that section or Sections 11 and 12(a)(2) of the Securities
Act of 1933, as amended. In addition, this information shall not be deemed incorporated by reference into any of the Company’s filings
with the Securities and Exchange Commission, except as shall be expressly set forth by specific reference in any such filing.
Item 8.01. Other Events
On November 14, 2023, the Company posted an updated
presentation on its website. A copy of the presentation is filed herewith as Exhibit 99.2 and is incorporated herein by reference.
Item 9.01. Financial Statements and Exhibits
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934,
as amended, the Registrant has duly caused this report to be signed on its behalf by the undersigned, hereunto duly authorized.
| Dated: November 14, 2023 |
Immunic, Inc. |
| |
|
|
| |
By: |
/s/ Daniel Vitt |
| |
|
Daniel Vitt |
| |
|
Chief Executive Officer |

Immunic, Inc. Reports Third
Quarter 2023 Financial Results
and Provides Corporate Update
–
Positive Interim Analysis from Phase 2 CALLIPER Trial Showed Improvements in Serum Neurofilament Light Chain for Vidofludimus Calcium,
Consistent Throughout the Overall Progressive Multiple Sclerosis Population and All Subtypes –
–
Notice of Allowance for United States Patent Protecting the Treatment of Relapsing Multiple Sclerosis with Vidofludimus and Its Salts
Bolsters Multilayered Intellectual Property Position –
–
$59.7 Million in Cash and Cash Equivalents as of September 30, 2023 Expected to Fund Immunic Into September of 2024 –
–
Webcast to be Held Today, November 14, 2023, at 8:00 am ET –
NEW YORK,
November 14, 2023 – Immunic, Inc. (Nasdaq:
IMUX), a biotechnology company developing a clinical pipeline of orally administered, small molecule therapies for chronic
inflammatory and autoimmune diseases, today announced financial results for the third quarter ended September 30, 2023, and provided a
corporate update.
“We
have continued to make tangible progress on the clinical development of our lead asset, nuclear receptor related 1 (Nurr1) activator,
vidofludimus calcium (IMU-838). Of particular importance, we completed enrollment of our phase 2 CALLIPER trial in patients with progressive
multiple sclerosis (PMS) and most recently reported a stronger than expected, positive interim biomarker analysis of this trial.
Notably, the clear separation observed in serum neurofilament light chain (NfL) over placebo in
this patient population represents another key milestone for what could potentially be a
first-in-class Nurr1 activator for PMS,” stated Daniel Vitt, Ph.D., Chief Executive Officer and President of Immunic. “Serum
NfL responses were consistently observed for vidofludimus calcium across progressive MS disease as well as all subpopulations and even
in non-active SPMS, a population where the medical need for new therapies is particularly high. We believe that the data set provides
biomarker evidence that vidofludimus calcium’s activity extends beyond the previously observed anti-inflammatory effects, further
reinforcing its neuroprotective potential and bolstering our view that it may be associated with the reduced disability-worsening events
we already observed in multiple sclerosis (MS) patients.”
“Assuming that the top-line CALLIPER data,
which we plan to report in April 2025, continues to show a neuroprotective effect, we may be able to position vidofludimus calcium as
the first oral treatment for non-active secondary progressive MS. We also think that the drug’s potential first-in-class ability
to activate Nurr1 may meaningfully benefit our ongoing phase 3 ENSURE program in relapsing MS (RMS). To that end, we continue to enroll
patients in our twin phase 3 ENSURE trials and currently expect to report an interim futility analysis in late 2024, with the read-out
of the first of the ENSURE trials expected at the end of 2025. If ultimately approved, we continue to believe that vidofludimus calcium,
with combined neuroprotective, anti-inflammatory, and antiviral effects, could potentially offer a unique treatment option targeted to
the complex pathophysiology of MS.”

Dr.
Vitt continued, “In October and November, respectively, we presented the previously reported positive results from our phase 1b
clinical trial of our second key program, IMU-856, an orally available and systemically acting small molecule modulator that targets SIRT6
(Sirtuin 6), in patients with celiac disease, at two prestigious medical conferences. The results, gathered during periods of gluten-free
diet and gluten challenge, demonstrated positive effects for IMU-856 over placebo in four key dimensions of celiac disease pathophysiology:
protection of the gut architecture, improvement of patients’ symptoms, biomarker response and enhancement of nutrient absorption.
IMU-856 was also observed to be safe and well-tolerated in this trial. We believe that this highly encouraging data provides initial clinical
proof-of-concept for a potential new therapeutic approach to gastrointestinal disorders by promoting regeneration of bowel architecture.
That said, we are currently preparing for clinical phase 2 testing of IMU-856 in ongoing active celiac disease (OACD), while also considering
further potential clinical applications in other gastrointestinal disorders.”
Third Quarter 2023 and Subsequent Highlights
| · | November 2023: Received a Notice of Allowance from the United States
Patent and Trademark Office for patent application 17/391,442, entitled, “Treatment of Multiple Sclerosis Comprising DHODH
Inhibitors,” covering a daily dose of about 10 mg to 45 mg of vidofludimus calcium and other salt as well as free acid forms for
the treatment of RMS. The claims are expected to provide protection into 2041, unless extended further. |
| · | November 2023: Presented data from the company’s phase 1b
clinical trial of IMU-856 in patients with celiac disease in a poster presentation at the Association of European Coeliac Societies (AOECS)
35th General Assembly Conference 2023. |
| · | October 2023: Presented data from the phase 1b clinical trial of
IMU-856 in patients with celiac disease in a moderated poster session; along with data from the company’s phase 2 CALDOSE-1 trial
of vidofludimus calcium in moderate-to-severe ulcerative colitis (UC) in an oral presentation, both at the United European Gastroenterology
Week (UEGW) 2023. |
| · | October 2023: Presented data from the company’s phase 2 EMPhASIS
trial of vidofludimus calcium in relapsing-remitting MS in an ePoster at MSMilan2023: The 9th Joint ECTRIMS-ACTRIMS Meeting. |
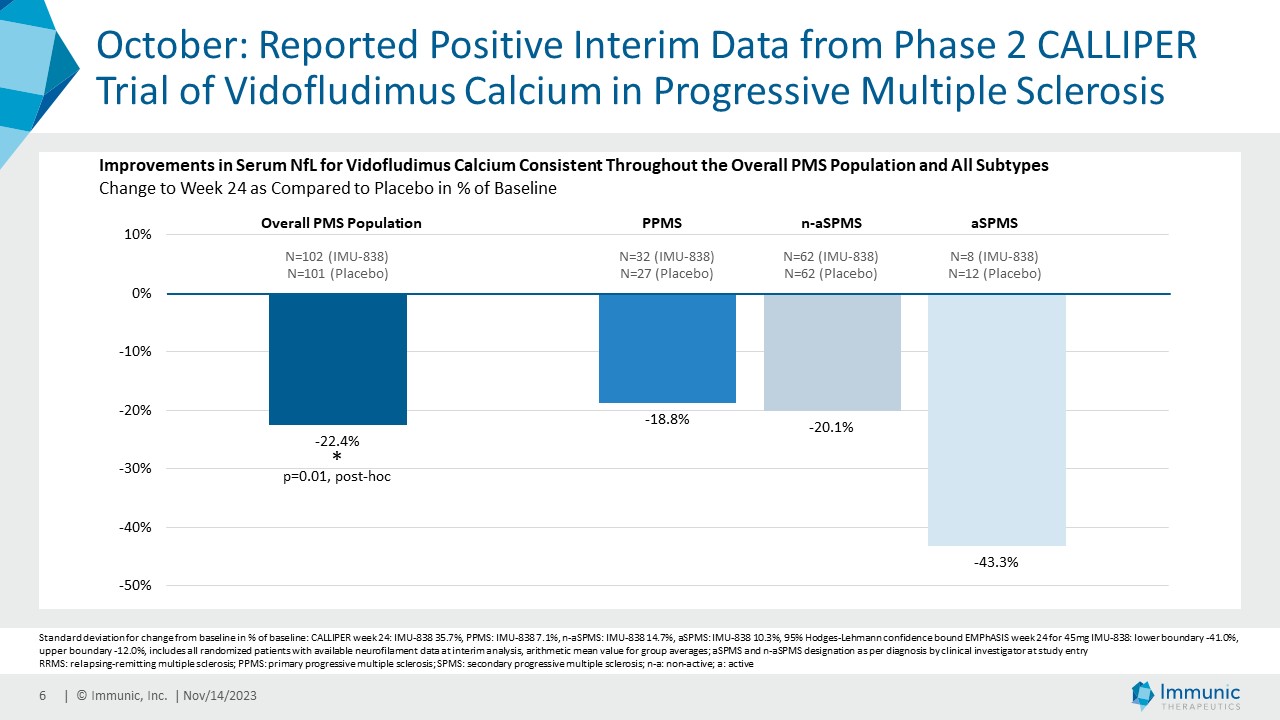
| · | October 2023: Reported positive interim data from the phase 2 CALLIPER
trial of vidofludimus calcium in PMS. Serum NfL responses were consistently observed for vidofludimus calcium across PMS disease and all
subtypes, as well as in patients that show or do not show disease and/or magnetic resonance imaging (MRI) activity. The Company believes
that this data showed biomarker evidence that vidofludimus calcium’s activity extends beyond the previously observed anti-inflammatory
effects, thereby further reinforcing its neuroprotective potential. |
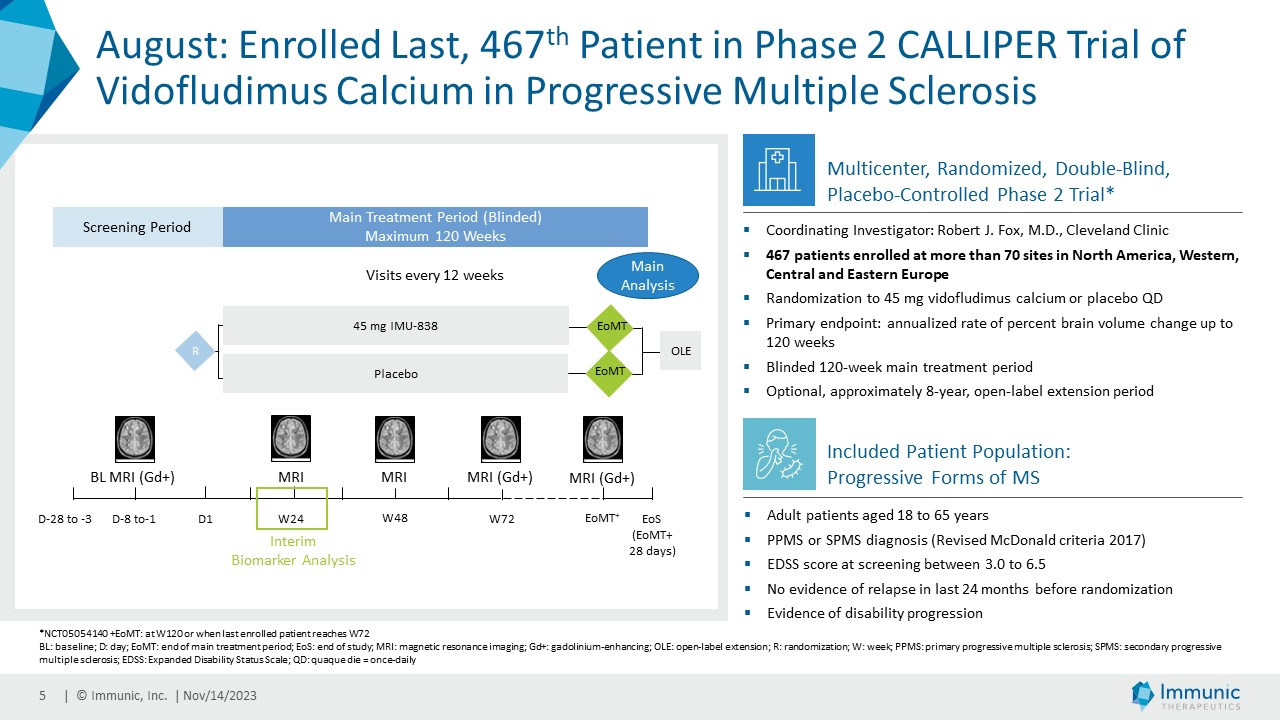
| · | August 2023: Announced the completion of enrollment of the phase
2 CALLIPER trial of vidofludimus calcium in PMS. In total, 467 patients with primary PMS, or active or non-active secondary PMS were randomized
to either 45 mg of vidofludimus calcium or placebo. |
| · | July 2023: Hosted a virtual celiac disease expert roundtable to
discuss the substantial unmet medical need for new therapeutic solutions. Immunic’s management also provided an overview of the
company’s IMU-856 program, including the positive results from the phase 1b clinical trial in patients with celiac disease. |

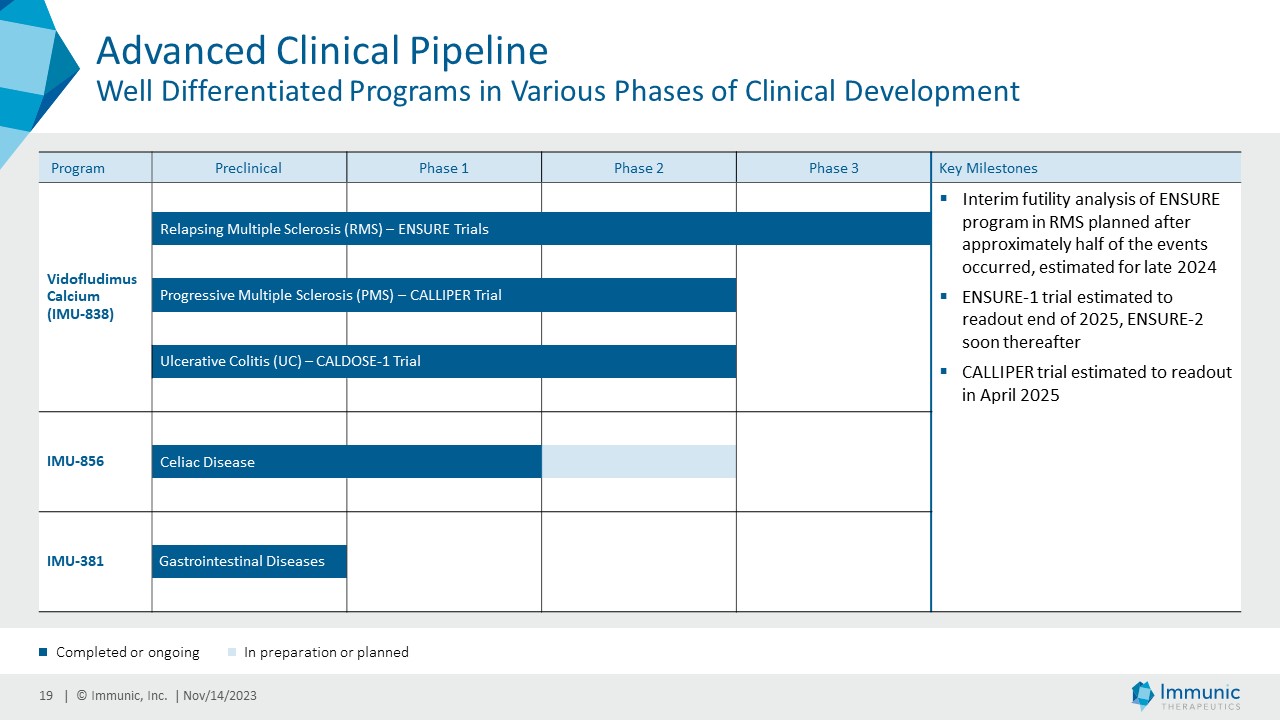
Anticipated Clinical Milestones
| · | Vidofludimus calcium in MS: Top-line data from the phase 2 CALLIPER trial of vidofludimus calcium
in PMS is expected in April of 2025. An interim futility analysis of the ENSURE program is expected in late 2024, with the read-out of
the first of the ENSURE trials at the end of 2025. |
| · | IMU-856 in celiac disease: Based on the positive data from the phase 1b clinical trial, testing
IMU-856 in celiac disease patients during periods of gluten-free diet and gluten challenge, the company is actively preparing for clinical
phase 2 testing of IMU-856 in OACD patients. |
Financial and Operating Results
| · | Research and Development (R&D) Expenses were $19.8 million for the three months ended September
30, 2023, as compared to $16.5 million for the three months ended September 30, 2022. The $3.3 million increase reflects (i) a $2.8 million
increase in external development costs related to the phase 3 clinical program of vidofludimus calcium in RMS, (ii) a $1.2 million increase
in drug supply costs for vidofludimus calcium to support our ongoing trials, (iii) a $1.0 million increase in external development costs
related to the phase 2 clinical trial of vidofludimus calcium in PMS, (iv) a $0.5 million increase in personnel expense in R&D related
to an increase in headcount and (v) a $0.9 million increase in related costs across numerous categories. The increases were partially
offset by (i) a decrease of $2.5 million resulting from deprioritizing the izumerogant program in psoriasis and castration-resistant prostate
cancer and (ii) a $0.6 million decrease in external development costs related to the phase 1 clinical trial of IMU-856. |
For the nine months ended September
30, 2023, R&D expenses were $63.9 million, as compared to $50.5 million for the same period ended September 30, 2022. The $13.4 million
increase reflects (i) a $10.1 million increase in external development costs related to the phase 3 clinical program of vidofludimus calcium
in RMS, (ii) a $2.9 million increase in external development costs related to the phase 2 clinical trial of vidofludimus calcium in PMS,
(iii) a $2.3 million increase in drug supply costs for vidofludimus calcium to support our ongoing trials, (iv) a $1.8 million increase
in external development costs related to the phase 1 clinical trial of IMU-856, (v) a $1.6 million increase in personnel expense in R&D
related to an increase in headcount, $0.2 million of which was due to non-cash stock based compensation and (vi) a $0.5 million increase
in related costs across numerous categories. The increases were partially offset by (i) a decrease of $4.0 million resulting from deprioritizing
the izumerogant program in psoriasis and castration-resistant prostate cancer and (ii) a decrease of $1.8 million in external development
costs related to the phase 2 clinical trial of vidofludimus calcium in UC.
| · | General and Administrative (G&A) Expenses were $3.8 million for the three months ended September
30, 2023, as compared to $3.6 million for the same period ended September 30, 2022. The $0.2 million increase was spread across numerous
categories. |
For the nine months ended September
30, 2023, G&A expenses were $11.9 million, as compared to $11.6 million for the same period ended September 30, 2022. The $0.3 million
increase was primarily due to (i) a $0.2 million increase in travel expense, (ii) a $0.2 million increase in legal and consultancy expense
and (iii) a $0.5 million increase across numerous categories. The increases were partially offset by a decrease of $0.6 million in personnel
expenses in G&A, which was primarily due to non-cash stock-based compensation decrease.

| · | Other Income (Expense) was $0.8 million for the three months ended September 30, 2023, as compared
to ($1.1 million) for the same period ended September 30, 2022. The $1.9 million increase was primarily attributable to (i) a $1.8 million
decrease in foreign exchange losses and (ii) a $0.5 million increase in interest income as a result of higher interest rates. The increase
was partially offset by a $0.4 million decrease in R&D tax incentives for clinical trials in Australia as a result of decreased spending
on clinical trials in Australia primarily for IMU-856. |
For the nine months ended September
30, 2023, other income was $3.8 million, as compared to ($1.8 million) for the same period ended September 30, 2022. The $5.6 million
increase was primarily attributable to (i) a $2.2 million increase in interest income as a result of higher interest rates, (ii) a $3.2
million decrease in foreign exchange losses and (iii) a $1.1 million research allowance attributable to tax year 2021 from the German
Federal Ministry of Finance. The increase was partially offset by a $0.9 million decrease in R&D tax incentives for clinical trials
in Australia as a result of decreased spending on clinical trials in Australia.
| · | Net Loss for the three months ended September 30, 2023, was approximately $22.8 million, or
$0.51 per basic and diluted share, based on 44,574,377 weighted average common shares outstanding, compared to a net loss of approximately
$21.2 million, or $0.69 per basic and diluted share, based on 30,564,995 weighted average common shares outstanding for the same period
ended September 30, 2022. |
Net loss for the nine months ended
September 30, 2023, was approximately $72.0 million, or $1.63 per basic and diluted share, based on 44,227,264 weighted average common
shares outstanding, compared to a net loss of approximately $63.9 million, or $2.16 per basic and diluted share, based on 29,655,946 weighted
average common shares outstanding for the same period ended September 30, 2022.
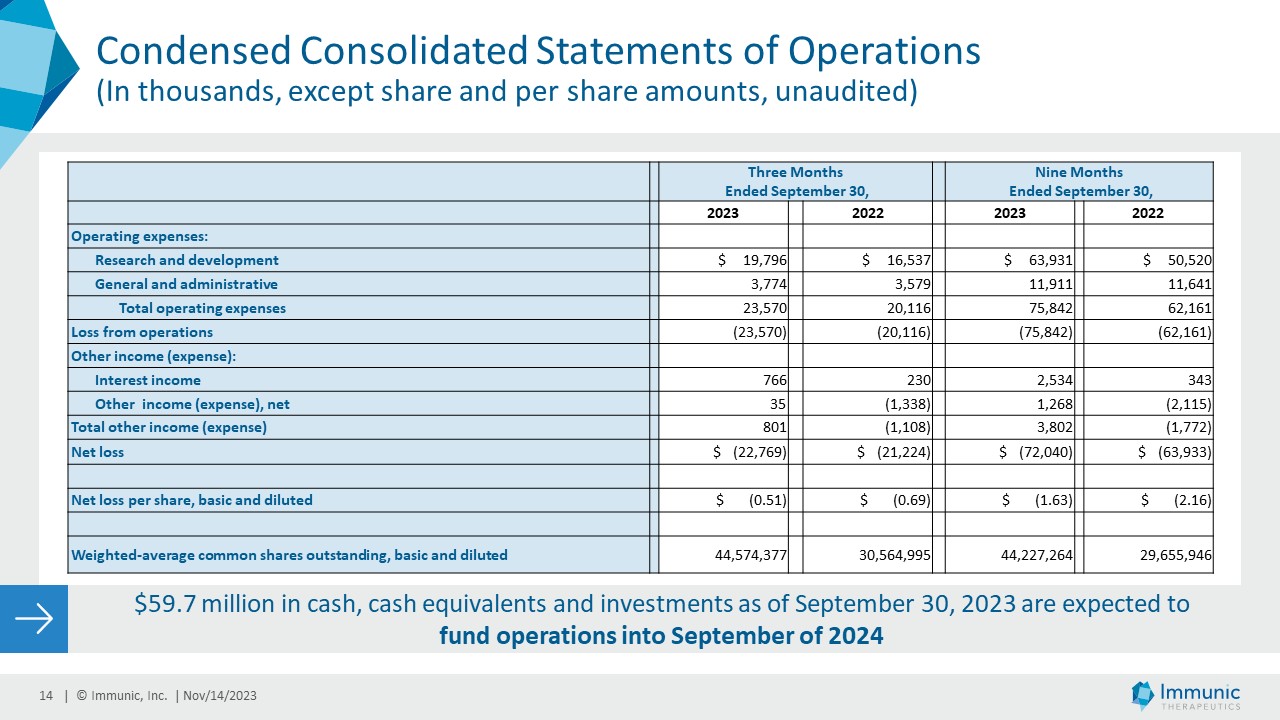
| · | Cash, Cash Equivalents and Investments as of September 30, 2023 were $59.7 million. With these
funds Immunic expects to be able to fund its operations into September of 2024. |
Webcast Information
Immunic will host a webcast today at 8:00 am ET.
To participate in the webcast, please register in advance at: https://imux.zoom.us/webinar/register/WN_AxIAE0pCQ12oMZIiehNKDg
or on the “Events and Presentations” section of Immunic’s website at: ir.imux.com/events-and-presentations.
Registrants will receive a confirmation email containing a link for online participation or a telephone number for dial in access.
An archived replay of the webcast will be available
approximately one hour after completion on Immunic’s website at: ir.imux.com/events-and-presentations.

About Immunic,
Inc.
Immunic, Inc. (Nasdaq:
IMUX) is a biotechnology company developing a clinical pipeline of orally administered, small molecule therapies for chronic inflammatory
and autoimmune diseases. The company’s lead development program, vidofludimus calcium (IMU-838), is currently in phase 3 and phase
2 clinical trials for the treatment of relapsing and progressive multiple sclerosis, respectively, and has shown therapeutic activity
in phase 2 clinical trials in patients suffering from relapsing-remitting multiple sclerosis, progressive multiple sclerosis and moderate-to-severe
ulcerative colitis. Vidofludimus calcium combines neuroprotective effects, through its mechanism as a first-in-class nuclear receptor
related 1 (Nurr1) activator, with additional anti-inflammatory and anti-viral effects, by selectively inhibiting the enzyme dihydroorotate
dehydrogenase (DHODH). IMU-856, which targets the protein Sirtuin 6 (SIRT6), is intended to restore intestinal barrier function and regenerate
bowel epithelium, which could potentially be applicable in numerous gastrointestinal diseases, such as celiac disease, where it is currently
in preparations for a phase 2 clinical trial. IMU-381, which currently is in preclinical testing, is a next generation molecule being
developed to specifically address the needs of gastrointestinal diseases. For further information, please visit: www.imux.com.
Cautionary
Statement Regarding Forward-Looking Statements
This press
release contains “forward-looking statements” that involve substantial risks and uncertainties for purposes of the safe harbor
provided by the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, included
in this press release regarding strategy, future operations, future financial position, future revenue, projected expenses, sufficiency
of cash, expected timing, development and results of clinical trials, prospects, plans and objectives of management are forward-looking
statements. Examples of such statements include, but are not limited to, statements relating to Immunic’s development programs and
the targeted diseases; the potential for Immunic’s development programs to safely and effectively target diseases; preclinical and
clinical data for Immunic’s development programs; the timing of current and future clinical trials and anticipated clinical milestones;
the nature, strategy and focus of the company and further updates with respect thereto; the development and commercial potential of any
product candidates of the company; and the company’s expected cash runway. Immunic may not actually achieve the plans, carry out
the intentions or meet the expectations or projections disclosed in the forward-looking statements and you should not place undue reliance
on these forward-looking statements. Such statements are based on management’s current expectations and involve substantial risks
and uncertainties. Actual results and performance could differ materially from those projected in the forward-looking statements as a
result of many factors, including, without limitation, the COVID-19 pandemic, increasing inflation, impacts of the Ukraine – Russia
conflict and the conflict in the Middle East on planned and ongoing clinical trials, risks and uncertainties associated with the ability
to project future cash utilization and reserves needed for contingent future liabilities and business operations, the availability of
sufficient financial and other resources to meet business objectives and operational requirements, the fact that the results of earlier
preclinical studies and clinical trials may not be predictive of future clinical trial results, the protection and market exclusivity
provided by Immunic’s intellectual property, risks related to the drug development and the regulatory approval process and the impact
of competitive products and technological changes. A further list and descriptions of these risks, uncertainties and other factors can
be found in the section captioned “Risk Factors,” in the company’s Annual Report on Form 10-K for the fiscal year ended
December 31, 2022, filed with the SEC on February 23, 2023, and in the company’s subsequent filings with the Securities and Exchange
Commission. Copies of these filings are available online at www.sec.gov or ir.imux.com/sec-filings. Any forward-looking statement made
in this release speaks only as of the date of this release. Immunic disclaims any intent or obligation to update these forward-looking
statements to reflect events or circumstances that exist after the date on which they were made. Immunic expressly disclaims all liability
in respect to actions taken or not taken based on any or all the contents of this press release.

Contact Information
Immunic, Inc.
Jessica Breu
Head of Investor Relations and Communications
+49 89 2080 477 09
jessica.breu@imux.com
US IR Contact
Rx Communications Group
Paula Schwartz
+1 917 633 7790
immunic@rxir.com
US Media Contact
KOGS Communication
Edna Kaplan
+1 617 974 8659
kaplan@kogspr.com

Financials
Immunic, Inc.
Condensed Consolidated Statements of Operations
(In thousands, except share and per share amounts)
(Unaudited)
| | |
Three Months Ended September 30, | |
Nine Months Ended September 30, |
| | |
2023 | |
2022 | |
2023 | |
2022 |
| Operating expenses: | |
| | | |
| | | |
| | | |
| | |
| Research and development | |
$ | 19,796 | | |
$ | 16,537 | | |
$ | 63,931 | | |
$ | 50,520 | |
| General and administrative | |
| 3,774 | | |
| 3,579 | | |
| 11,911 | | |
| 11,641 | |
| Total operating expenses | |
| 23,570 | | |
| 20,116 | | |
| 75,842 | | |
| 62,161 | |
| Loss from operations | |
| (23,570 | ) | |
| (20,116 | ) | |
| (75,842 | ) | |
| (62,161 | ) |
| Other income (expense): | |
| | | |
| | | |
| | | |
| | |
| Interest income | |
| 766 | | |
| 230 | | |
| 2,534 | | |
| 343 | |
| Other income (expense), net | |
| 35 | | |
| (1,338 | ) | |
| 1,268 | | |
| (2,115 | ) |
| Total other income (expense) | |
| 801 | | |
| (1,108 | ) | |
| 3,802 | | |
| (1,772 | ) |
| Net loss | |
$ | (22,769 | ) | |
$ | (21,224 | ) | |
$ | (72,040 | ) | |
$ | (63,933 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Net loss per share, basic and diluted | |
$ | (0.51 | ) | |
$ | (0.69 | ) | |
$ | (1.63 | ) | |
$ | (2.16 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Weighted-average common shares outstanding, basic and diluted | |
| 44,574,377 | | |
| 30,564,995 | | |
| 44,227,264 | | |
| 29,655,946 | |

Immunic, Inc.
Condensed Consolidated Balance Sheets
(In thousands, except share and per share amounts)
(Unaudited)
| | |
September 30, 2023 | |
December 31, 2022 |
| | |
(Unaudited) | |
|
| Assets | |
| | | |
| | |
| Current assets: | |
| | | |
| | |
| Cash and cash equivalents | |
$ | 59,689 | | |
$ | 106,745 | |
| Investments - other | |
| — | | |
| 9,629 | |
| Other current assets and prepaid expenses | |
| 5,545 | | |
| 9,490 | |
| Total current assets | |
| 65,234 | | |
| 125,864 | |
| Property and equipment, net | |
| 288 | | |
| 294 | |
| Right-of-use assets, net | |
| 1,412 | | |
| 1,552 | |
| Other long-term assets | |
| 43 | | |
| 43 | |
| Total assets | |
$ | 66,977 | | |
$ | 127,753 | |
| Liabilities and Stockholders’ Equity | |
| | | |
| | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable | |
$ | 3,199 | | |
$ | 4,281 | |
| Accrued expenses | |
| 13,659 | | |
| 7,986 | |
| Other current liabilities | |
| 923 | | |
| 810 | |
| Total current liabilities | |
| 17,781 | | |
| 13,077 | |
| Long term liabilities | |
| | | |
| | |
| Operating lease liabilities | |
| 789 | | |
| 992 | |
| Total long-term liabilities | |
| 789 | | |
| 992 | |
| Total liabilities | |
| 18,570 | | |
| 14,069 | |
| Commitments and contingencies | |
| | | |
| | |
| Stockholders’ equity: | |
| | | |
| | |
| Preferred stock, $0.0001 par value; 20,000,000 authorized and no shares issued or outstanding at September 30, 2023 and December 31, 2022 | |
| — | | |
| — | |
| Common stock, $0.0001 par value; 130,000,000 shares authorized and 44,595,383 and 39,307,286 shares issued and outstanding as of September 30, 2023 and December 31, 2022, respectively | |
| 4 | | |
| 4 | |
| Additional paid-in capital | |
| 433,818 | | |
| 427,925 | |
| Accumulated other comprehensive income | |
| 3,905 | | |
| 3,035 | |
| Accumulated deficit | |
| (389,320 | ) | |
| (317,280 | ) |
| Total stockholders’ equity | |
| 48,407 | | |
| 113,684 | |
| Total liabilities and stockholders’ equity | |
$ | 66,977 | | |
$ | 127,753 | |















v3.23.3
Cover
|
Nov. 14, 2023 |
| Cover [Abstract] |
|
| Document Type |
8-K
|
| Amendment Flag |
false
|
| Document Period End Date |
Nov. 14, 2023
|
| Entity File Number |
001-36201
|
| Entity Registrant Name |
IMMUNIC, INC.
|
| Entity Central Index Key |
0001280776
|
| Entity Tax Identification Number |
56-2358443
|
| Entity Incorporation, State or Country Code |
DE
|
| Entity Address, Address Line One |
1200 Avenue of the Americas
|
| Entity Address, Address Line Two |
Suite 200
|
| Entity Address, City or Town |
New York
|
| Entity Address, State or Province |
NY
|
| Entity Address, Country |
US
|
| Entity Address, Postal Zip Code |
10036
|
| City Area Code |
(332)
|
| Local Phone Number |
255-9818
|
| Written Communications |
false
|
| Soliciting Material |
false
|
| Pre-commencement Tender Offer |
false
|
| Pre-commencement Issuer Tender Offer |
false
|
| Title of 12(b) Security |
Common Stock, par value $0.0001
|
| Trading Symbol |
IMUX
|
| Security Exchange Name |
NASDAQ
|
| Entity Emerging Growth Company |
false
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionISO 3166-1 alpha-2 country code.
| Name: |
dei_EntityAddressCountry |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:countryCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Immunic (NASDAQ:IMUX)
Historical Stock Chart
From Apr 2024 to May 2024
Immunic (NASDAQ:IMUX)
Historical Stock Chart
From May 2023 to May 2024