Takeda Gets FDA Approval for Ulcerative Colitis Therapy
28 September 2023 - 7:13AM
Dow Jones News
By Ben Glickman
Takeda has received approval from the U.S. Food and Drug
Administration for ENTYVIO as a therapy for adults with moderate to
severe ulcerative colitis.
The Osaka, Japan-based pharmaceutical company said it expected
ENTYVIO to be available in the U.S. as a single-dose pen by the end
of October.
The company currently is waiting for a response from the FDA on
a biologics license application for administering ENTYVIO in the
treatment of adults with Crohn's disease.
Takeda does not expect the approval to affect full-year
forecasts for the company's results.
Write to Ben Glickman at ben.glickman@wsj.com
(END) Dow Jones Newswires
September 27, 2023 16:58 ET (20:58 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
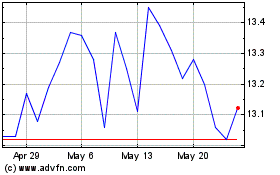
Takeda Pharmaceutical (NYSE:TAK)
Historical Stock Chart
From Apr 2024 to May 2024
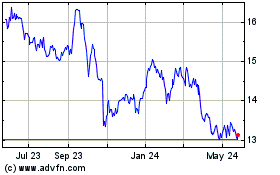
Takeda Pharmaceutical (NYSE:TAK)
Historical Stock Chart
From May 2023 to May 2024