BD Diagnostics, a segment of BD (Becton, Dickinson and Company)
(NYSE: BDX), and Bruker Daltonics Inc., a subsidiary of Bruker
Corporation (NASDAQ: BRKR), announced today an international
co-development and co-marketing collaboration that will promote an
emerging, integrated approach to bacterial and fungal
identification and antimicrobial susceptibility testing. This new
approach has the potential to transform how traditional
microbiology has been performed for decades.
The MALDI Biotyper, a compact, benchtop
mass spectrometry based system for rapid microbial identification.
(Photo: Business Wire)
Through this collaboration, identification of microorganisms
will be performed with the Bruker MALDI Biotyper, a mass
spectrometry-based proteomic “fingerprinting” system specifically
configured for rapid identification of bacteria, yeast and fungi.
The MALDI Biotyper is available in a clinical version with IVD-CE
mark in Europe and is for research use only (RUO) in the United
States, where Bruker intends to seek FDA clearance.
BD and Bruker intend to combine the MALDI Biotyper microbial ID
system with automated antimicrobial susceptibility testing on the
BD Phoenix™ Microbiology System. The BD EpiCenter™ Microbiology
Data Management System, which will manage patient data from both
the identification and the susceptibility test systems, will
facilitate this industry-first integrated approach.
The MALDI Biotyper – BD EpiCenter software integration will be
further developed to optimize workflows for rapid MALDI Biotyper
pathogen identification directly on positive blood cultures from
the leading BD BACTECTM blood culture system. This rapid blood
culture-to-ID workflow is supported by Bruker’s new MALDI
Sepsityper™ consumables kit, which today is for RUO. It is expected
to become clinically important in the future, as time-to-result for
pathogen identification is critical to management of patients
having potentially serious bloodstream infections or sepsis.
“This collaboration is ideal because the competencies of the two
organizations will complement each other by providing excellent
microbiology solutions for the customer,” said Jamie Condie, Vice
President and General Manager, Infectious Disease, BD Diagnostics –
Diagnostic Systems. “We have chosen to collaborate with Bruker
because we believe that its mass spectrometry technologies are the
future of microbial identification, and that the MALDI Biotyper is
the first and best solution to the market. We expect this
collaboration will result in enhanced clinical responsiveness while
improving lab efficiency.”
“At Bruker, we are excited about this new strategic
collaboration with BD because of the potential it holds to
significantly advance clinical microbiology,” said Frank Laukien,
Ph.D., President and CEO of Bruker Corporation. “Working with a
worldwide leader in microbiology and infectious disease will enable
us to further expand the groundbreaking MALDI Biotyper molecular
method for microbial ID to its full extent.”
Microbial identification and antimicrobial susceptibility
testing is the end-point of the major work a microbiology lab
performs on a daily basis. After culturing and isolating bacteria
and fungi from patient specimens, organisms need to be identified
and tested to determine which drugs will inhibit or stop their
growth. The Bruker MALDI Biotyper allows highly accurate, rapid and
cost-effective identification through a process in which organisms
are identified by the unique spectrum of the major proteins and
peptides that constitute their makeup. The accuracy and benefits of
the MALDI Biotyper have been well documented in over 30
peer-reviewed articles.
Antimicrobial susceptibility testing is conducted via
traditional automated systems such as the BD Phoenix System. The
combination of the two leading technologies, the BD Phoenix System
and the MALDI Biotyper, and the data management through the BD
EpiCenter System, will provide laboratorians with a groundbreaking
new approach to identification and susceptibility testing, which
will reduce the turnaround time for critical diagnostic results,
while also improving laboratory efficiency and costs.
About BD
BD is a leading global medical technology company that develops,
manufactures and sells medical devices, instrument systems and
reagents. The Company is dedicated to improving people's health
throughout the world. BD is focused on improving drug delivery,
enhancing the quality and speed of diagnosing infectious diseases
and cancers, and advancing research, discovery and production of
new drugs and vaccines. BD's capabilities are instrumental in
combating many of the world's most pressing diseases. Founded in
1897 and headquartered in Franklin Lakes, New Jersey, BD employs
approximately 29,000 associates in more than 50 countries
throughout the world. The Company serves healthcare institutions,
life science researchers, clinical laboratories, the pharmaceutical
industry and the general public. For more information, please visit
www.bd.com.
About Bruker Daltonics
Bruker Daltonics is a leading manufacturer of mass spectrometry
(MS) instruments and accessories for life science, pharmaceutical,
biochemical and chemical research as well as for more routine
analytical tasks in forensics and food safety. Technical solutions
are based on a comprehensive range of MALDI-TOF/TOF, ESI-Qq-TOF,
ESI-ITMS, ESI/MALDI-FTMS mass spectrometry systems, as well as
automated sample handling systems and productivity enhancing
software designed to answer customers’ needs. For more information
about Bruker Daltonics and Bruker Corporation, please visit
www.bdal.com and www.bruker.com.
CAUTIONARY STATEMENT OF BD
This press release contains certain estimates and other
forward-looking statements (as defined under Federal securities
laws) regarding BD’s performance, including future revenues,
earnings per share and income, or events or developments that BD
expects to occur or anticipates occurring in the future. All such
statements are based upon current expectations of BD and involve a
number of business risks and uncertainties. Actual results could
vary materially from anticipated results described, implied or
projected in any forward-looking statement. With respect to
forward-looking statements contained herein, a number of factors
could cause actual results to vary materially from any
forward-looking statement. For instance, various healthcare reform
proposals, if enacted, would impose an excise tax applicable to
medical device manufacturers, including BD, and these may be
effective in calendar year 2010. Other factors include, but are not
limited to: adverse changes in regional, national or foreign
economic conditions, including any impact that may result from the
current global economic downturn on our ability to access credit
markets and finance our operations, the demand for our products and
services, or our suppliers’ ability to provide products needed for
our operations; changes in interest or foreign currency exchange
rates, particularly in light of increased volatility in currency
exchange rates; potential healthcare reform, including changes in
government pricing and reimbursement policies or other cost
containment reforms; competitive factors; pricing and market share
pressures; difficulties inherent in product development and delays
in product introductions; increases in energy costs and their
effect on, among other things, the cost of producing BD’s products;
fluctuations in costs and availability of raw materials and in BD’s
ability to maintain favorable supplier arrangements and
relationships; uncertainties of litigation (as described in BD’s
filings with the Securities and Exchange Commission); the effects
of potential pandemic diseases; our ability to successfully
integrate any businesses we acquire; and issuance of new or revised
accounting standards, as well as other factors discussed in BD’s
filings with the Securities and Exchange Commission. We do not
intend to update any forward-looking statements to reflect events
or circumstances after the date hereof except as required by
applicable laws or regulations.
CAUTIONARY STATEMENT OF BRUKER CORPORATION
Any statements contained in this press release that do not
describe historical facts may constitute forward-looking statements
as that term is defined in the Private Securities Litigation Reform
Act of 1995. Any forward-looking statements contained herein are
based on current expectations, but are subject to risks and
uncertainties that could cause actual results to differ materially
from those projected, including, but not limited to, risks and
uncertainties relating to adverse changes in conditions in the
global economy and volatility in the capital markets, the
integration of businesses we have acquired or may acquire in the
future, changing technologies, product development and market
acceptance of our products, the cost and pricing of our products,
manufacturing, competition, dependence on collaborative partners
and key suppliers, capital spending and government funding
policies, changes in governmental regulations, realization of
anticipated benefits from economic stimulus programs, intellectual
property rights, litigation, and exposure to foreign currency
fluctuations and other risk factors discussed from time to time in
our filings with the Securities and Exchange Commission. These and
other factors are identified and described in more detail in our
filings with the SEC, including, without limitation, our annual
report on Form 10-K for the year ended December 31, 2009, our most
recent quarterly reports on Form 10-Q and our current reports on
Form 8-K. We expressly disclaim any intent or obligation to update
these forward-looking statements other than as required by law.
Photos/Multimedia Gallery Available:
http://www.businesswire.com/cgi-bin/mmg.cgi?eid=6446518&lang=en
Bruker (NASDAQ:BRKR)
Historical Stock Chart
From Jun 2024 to Jul 2024
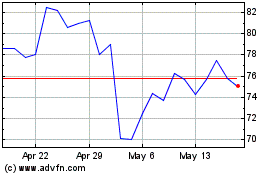
Bruker (NASDAQ:BRKR)
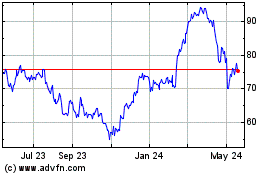
Historical Stock Chart
From Jul 2023 to Jul 2024