Imanis Life Sciences to Present IMMUNO-COV, a First-in-Class Neutralizing Antibody Test for COVID-19, at American Society of ...
14 September 2020 - 11:30PM

Imanis Life Sciences, LLC announced today upcoming presentations
about the development and validation of IMMUNO-COV™, the first
scalable, quantitative neutralizing antibody test for COVID-19. The
company will present a technical overview of the assay and host a
luncheon presentation at the American Society of Gene and Cell
Therapy (ASGCT) COVID-19 Symposium, a virtual event that will be
held September 15-16.
In contrast to other antibody tests, the IMMUNO-COV assay
specifically measures antibodies in the blood that can block
infection by SARS-Co-V-2, the virus that causes COVID19. One of the
key ways the immune system fights a SARS-CoV-2 infection is to make
a huge number of antibodies that recognize and bind to the surface
of the virus in many different ways and at many different sites.
Only a small subset of those antibodies — called neutralizing
antibodies — can actually prevent the virus from infecting new
cells.
The IMMUNO-COV test uses an innocuous virus that mimics
SARS-CoV-2 and detects neutralizing antibodies in the blood that
can recognize and stop the virus from infecting new cells.
Neutralizing antibodies are detectable in the blood typically 14
days after the onset of infection and their levels vary widely from
person to person.
Imanis Life Sciences, under a commercial arrangement with
Vyriad, is performing the IMMUNO-COV assay in its CLIA-certified
laboratory. Mayo Clinic has licensed IMMUNO-COV for evaluating
donors in the national COVID-19 convalescing plasma study.
Applications for the IMMUNO-COV assay include:
- Assessing neutralizing antibody levels in potential donors of
COVID-19 convalescent plasma for treatment of patients infected
with SARS-CoV-2 (plasma with high neutralizing antibody titers is
preferred).
- Evaluating the relative effectiveness of SARS-COV-2 vaccine
candidates in clinical trials.
- Testing healthcare workers and other at-risk professionals with
prior SARS-CoV-2 infections to assess the strength and durability
of their immunity to COVID-19 over time.
Rianna Vandergaast, Ph.D., the lead IMMUNO-COV scientist at
Imanis Life Sciences, will give both presentations at the ASGCT
COVID-19 Symposium.
12:00pm-1:00pm central time, Tuesday, September
15IMMUNO-COVTM: An Antibody Test for
COVID-19-Neutralizing AntibodiesThis luncheon presentation
will provide technical information about the assay and focus on its
significance, potential applications and ongoing assay research and
improvements.
3:48pm-4:00pm central time, Wednesday, September
16Development and Validation of a High-Throughput
Clinical Assay for Detecting SARS-CoV-2-Neutralizing
Antibodies. This is 10-minute technical overview of the
development and validation of IMMUNO-COV, as well as further assay
development
To attend the free virtual conference, register
here.[https://www.webcastregister.live/asgct_covid19_symposium/register.php]
A recording of Dr. Vandergaast’s presentations will be available
on the ASGCT website for 30 days following the symposium.
IMMUNO-COV is a product of Vyriad, Inc., developed in
collaboration with its affiliate Imanis Life Sciences, Regeneron
and Mayo Clinic.
About Imanis Life Sciences Imanis Life Sciences
is a leading provider of products and services to accelerate
development of viro-immuno-oncology drugs. Our mission is to
advance pharmacokinetics and imaging, promote widespread adoption
of noninvasive in vivo imaging in preclinical and clinical
research. Our team of dedicated and passionate scientists
(PhD, MSc, BSc) works closely with clients to facilitate their
research, from study design, custom products, and consultation. .
For more information, visit www.imanislife.com.
About Vyriad, Inc.Vyriad is a clinical-stage
company developing virus-based therapeutics, focusing initially on
proprietary oncolytic virus therapies for the treatment of cancers
with significant unmet needs. Founded by scientists at Mayo Clinic
and the University of Miami, Vyriad programs viruses to selectively
attack cancer cells, thereby igniting antitumor immune responses
that can complete the process of tumor destruction and prevent
disease recurrence. Our lead platforms, derived from either
vesicular stomatitis virus or measles virus (VSV), are being
evaluated in ongoing Phase 2 clinical trials addressing multiple
cancer types. Vyriad and Regeneron Pharmaceuticals, Inc. (NASDAQ:
REGN) have entered into a broad strategic agreement for the
discovery and development of new oncolytic virus treatments for
cancer leveraging Vyriad’s VSV platform and Regeneron’s PD-1
inhibitor Libtayo® (cemiplimab) as well as its unmatched antibody
discovery capabilities. Vyriad is a privately held company based in
Rochester, Minnesota. For more information, visit
www.vyriad.com.
Contacts:
|
MediaDavid Walshdwalsh@vyriad.com651.503.8248 |
Company inquiriesMichael
HerbertHerbert.michael@imanislife.com612.309.7801 |
Regeneron Pharmaceuticals (NASDAQ:REGN)
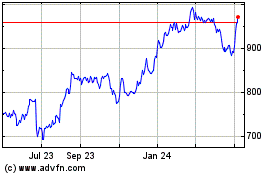
Historical Stock Chart
From Jun 2024 to Jul 2024
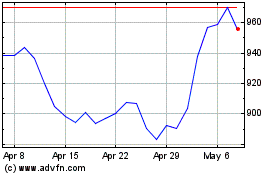
Regeneron Pharmaceuticals (NASDAQ:REGN)
Historical Stock Chart
From Jul 2023 to Jul 2024