Regeneron, Sanofi Pull FDA Application for Libtayo in Advanced Cervical Cancer
28 January 2022 - 11:54PM
Dow Jones News
By Colin Kellaher
Regeneron Pharmaceuticals Inc. and Sanofi SA on Friday said they
have voluntarily withdrawn their application seeking expanded U.S.
approval of the cancer drug Libtayo as a second-line treatment for
patients with advanced cervical cancer.
The companies said they made the decision after they weren't
able to align with the U.S. Food and Drug Administration on certain
post-marketing studies, adding that they are continuing discussions
with regulatory authorities outside the U.S.
The FDA in September had granted priority review to the
companies' application for expanded use of Libtayo to treat
patients with recurrent or metastatic cervical cancer whose disease
progressed on or after chemotherapy and had set a target action
date of Jan. 30.
Tarrytown, N.Y., biotechnology company Regeneron is developing
and commercializing Libtayo with France's Sanofi under a global
collaboration agreement. The drug is approved in several cancer
indications around the world.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
January 28, 2022 07:39 ET (12:39 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
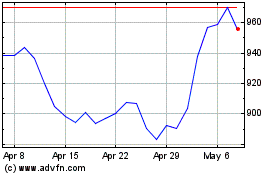
Regeneron Pharmaceuticals (NASDAQ:REGN)
Historical Stock Chart
From Jun 2024 to Jul 2024
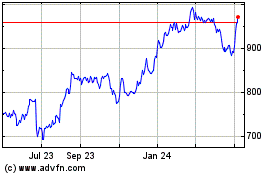
Regeneron Pharmaceuticals (NASDAQ:REGN)
Historical Stock Chart
From Jul 2023 to Jul 2024