Theravance Says FDA Grants Orphan Drug Designation Status to Ampreloxetine
10 May 2023 - 8:58AM
Dow Jones News
By Josh Beckerman
Theravance Biopharma said Tuesday that the Food and Drug
Administration granted Orphan Drug Designation status to
ampreloxetine for treatment of symptomatic neurogenic orthostatic
hypotension in patients with multiple system atrophy.
The company said it is "diligently progressing" its
registrational Phase 3 study of ampreloxetine, a once-daily
norepinephrine reuptake inhibitor.
Theravance shares rose 5 cents during the regular session to
$11.38 and were flat after hours.
Write to Josh Beckerman at josh.beckerman@wsj.com
(END) Dow Jones Newswires
May 09, 2023 18:43 ET (22:43 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
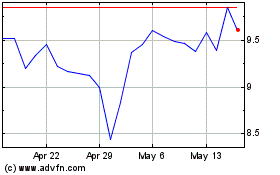
Theravance Biopharma (NASDAQ:TBPH)
Historical Stock Chart
From May 2024 to Jun 2024
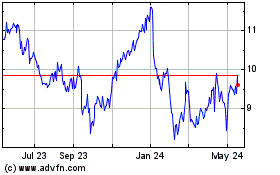
Theravance Biopharma (NASDAQ:TBPH)
Historical Stock Chart
From Jun 2023 to Jun 2024