Abbott Laboratories to Submit Limb-Threatening Ischemia Drug to FDA Review
26 October 2023 - 6:17AM
Dow Jones News
By Sabela Ojea
Abbott Laboratories said it has received positive results from
its chronic limb-threatening ischemia trial and it expects to
submit the drug for review by the U.S. Food and Drug
Administration.
The medical devices and health care company on Wednesday said
the trial met both primary safety and effectiveness endpoints and
showed that its Esprit BTK drug reduces disease progression.
Chronic limb-threatening ischemia is a form peripheral arterial
disease, which causes an accumulation of plaque in the arteries in
legs or arms.
About 22 million people worldwide have been affected by chronic
limb-threatening ischemia.
Write to Sabela Ojea at sabela.ojea@wsj.com
(END) Dow Jones Newswires
October 25, 2023 15:02 ET (19:02 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
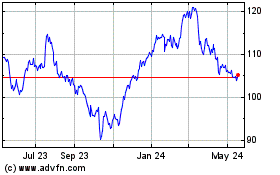
Abbott Laboratories (NYSE:ABT)
Historical Stock Chart
From Apr 2024 to May 2024
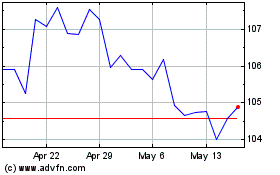
Abbott Laboratories (NYSE:ABT)
Historical Stock Chart
From May 2023 to May 2024