Galapagos reports positive Phase 1 results for GLPG1205
04 December 2013 - 5:34PM

Galapagos NV (Euronext: GLPG) announced today that
GLPG1205, a first-in-class molecule for inflammatory disorders, has
demonstrated target engagement, a good safety profile, and
favorable drug properties in a Phase 1 study. Galapagos is
developing GLPG1205 within its alliance with Janssen Pharmaceutica
NV.
The aim of the Phase 1 study was to evaluate the
safety, tolerability, pharmacokinetics, and pharmacodynamics of
oral single and multiple ascending doses of GLPG1205. The
randomized, double-blind, placebo-controlled, single center study
was conducted in 40 healthy volunteers in Belgium. In the
first part of the study, single ascending doses were evaluated.
In the second part, the new compound was administered daily
for 14 days.
GLPG1205 was safe and well-tolerated over a wide
dose range in healthy volunteers. Engagement of the thus far
undisclosed novel target was confirmed using a relevant biomarker.
GLPG1205 displayed a favorable pharmacokinetic and pharmacodynamic
profile, potentially supporting once-daily dosing. The data
shown in Phase 1 encourage Galapagos to progress GLPG1205 into a
Phase 2A study in Inflammatory Bowel Disease (IBD).
"GLPG1205 is the first molecule against this
target ever to be evaluated clinically, and we are pleased with the
outcome of the Phase 1 study," said Piet Wigerinck, CSO of
Galapagos. "Galapagos continues to deliver novel therapeutics
from its unique target and drug discovery engine. GLPG1205 will be
the fourth compound to enter Phase 2A in our portfolio of
proprietary and partnered programs based on novel targets."
In 2007, Galapagos announced an alliance
agreement with Janssen Pharmaceutica NV providing the option to
worldwide, commercial licenses to certain Galapagos internal
inflammatory disease programs. These programs include a novel
target for inflammatory disorders that was identified and validated
by Galapagos using its proprietary target discovery engine.
Subsequent Galapagos research led to the discovery of GLPG1205, a
first-in-class molecule that entered the clinic for inflammatory
disorders. Galapagos is responsible for execution of Phase 1
and Phase 2A studies with GLPG1205.
About Galapagos Galapagos (Euronext: GLPG; OTC:
GLPYY) is specialized in novel modes-of-action, with a large
pipeline comprising of six Phase 2 studies (three led by GSK), one
Phase 1 study, five pre-clinical, and 20 discovery small-molecule
and antibody programs in cystic fibrosis, inflammation,
antibiotics, metabolic disease, and other indications. AbbVie and
Galapagos signed an agreement in CF where they work collaboratively
to develop and commercialize oral drugs that address two mutations
in the CFTR gene, the G551D and F508del mutation. In the
field of inflammation, AbbVie and Galapagos signed a worldwide
license agreement whereby AbbVie will be responsible for further
development and commercialization of GLPG0634 after Phase 2B.
GLPG0634 is an orally-available, selective inhibitor of JAK1 for
the treatment of rheumatoid arthritis and potentially other
inflammatory diseases, currently in Phase 2B studies in RA and
about to enter Phase 2 studies in Crohn's disease. Galapagos
has another selective JAK1 inhibitor in Phase 2 in ulcerative
colitis, psoriasis, and lupus, GSK2586184 (formerly GLPG0778,
in-licensed by GlaxoSmithKline in 2012). GLPG0974 is the
first inhibitor of FFA2 to be evaluated clinically for the
treatment of IBD; this program is currently in a Proof-of-Concept
Phase 2 study. GLPG1205, a first-in-class molecule that
targets inflammatory disorders, has completed Phase 1 studies. The
Galapagos Group, including fee-for-service companies BioFocus,
Argenta and Fidelta, has around 800 employees and operates
facilities in five countries, with global headquarters in Mechelen,
Belgium. Further information at: www.glpg.com
CONTACT
Galapagos NV Piet Wigerinck, CSO Tel: +32 477 62
7103
Elizabeth Goodwin, Director Investor Relations
Tel: +31 6 2291 6240 ir@glpg.com
Galapagos forward-looking statements This
release may contain forward-looking statements, including, without
limitation, statements containing the words "believes,"
"anticipates," "expects," "intends," "plans," "seeks," "estimates,"
"may," "will," "could," "stands to," and "continues," as well as
similar expressions. Such forward-looking statements may involve
known and unknown risks, uncertainties and other factors which
might cause the actual results, financial condition, performance or
achievements of Galapagos, or industry results, to be materially
different from any historic or future results, financial
conditions, performance or achievements expressed or implied by
such forward-looking statements. Given these uncertainties, the
reader is advised not to place any undue reliance on such
forward-looking statements. These forward-looking statements speak
only as of the date of publication of this document. Galapagos
expressly disclaims any obligation to update any such
forward-looking statements in this document to reflect any change
in its expectations with regard thereto or any change in events,
conditions or circumstances on which any such statement is based,
unless required by law or regulation.
HUG#1747508
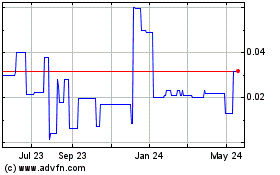
Aristocrat (PK) (USOTC:ASCC)
Historical Stock Chart
From Jun 2024 to Jul 2024
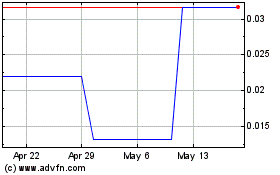
Aristocrat (PK) (USOTC:ASCC)
Historical Stock Chart
From Jul 2023 to Jul 2024