PROSPECTUS
Filed
Pursuant to Rule 424(b)3
Registration
No. 333-195603
43,646,992
Shares of Common Stock
of
Guided Therapeutics, Inc.
________________________
This prospectus relates to up to 43,646,992 shares of our common
stock, consisting of:
|
|
·
|
8,841,836 shares issued or issuable upon conversion of an aggregate principal amount of $3 million
of our senior convertible notes;
|
|
|
·
|
321,820 shares issued as “commitment shares” in connection with the senior convertible
notes;
|
|
|
·
|
6,317,502 shares issued or issuable upon conversion of an aggregate of 2,527 shares of
our Series B convertible preferred stock, of which 3,716,177 were originally registered for resale under a prior
registration
statement but remain unsold;
|
|
|
·
|
1,172,913 shares issued or issuable as payment for dividends on the Series B convertible
preferred stock, payable through December 31, 2015, of which 836,610 were originally registered for resale under a prior
registration statement but remain unsold; and
|
|
|
·
|
26,992,921 shares issued or issuable upon exercise of warrants at exercise prices ranging
from $0.40 per share to $1.08 per share, subject to adjustment as provided in certain of the warrants; of which 20,260,479
were originally registered for resale under a prior registration statement but remain unsold.
|
The shares offered by this prospectus
may be sold from time to time by the selling stockholders at prevailing market prices or prices negotiated at the time of sale.
See “Plan of Distribution” and “Selling Stockholders.” The shares offered by this prospectus were issued
or are issuable upon conversion of securities issued to the selling stockholders in transactions exempt from registration under
the Securities Act of 1933, or Securities Act.
We will not receive any cash proceeds
from the sale of shares by the selling stockholders, but to the extent that the warrants were or are exercised in whole or in part
for cash, we have received or will receive payment for the exercise price. We will pay the expenses of registering the shares.
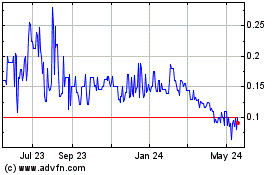
Our common stock is listed on the OTCQB
marketplace under the symbol “GTHP.” The last reported sale price of our common stock on the OTCQB on April 28, 2014
was $0.467 per share. The selling stockholders will sell at prevailing market prices per share (as quoted on the OTCQB), at the
time of sale, at fixed prices, at varying prices determined at the time of sale, or at negotiated prices.
Investing in our common stock involves
a high degree of risk. These risks are described under the caption “Risk Factors” that begins on page
4
of this prospectus.
Neither the Securities and Exchange Commission,
or SEC, nor any state securities commission has approved or disapproved of the common stock that may be offered under this prospectus,
nor have any of these organizations determined if this prospectus is truthful or complete. Any representation to the contrary is
a criminal offense.
The date of this prospectus
is May 12, 2014.
TABLE OF CONTENTS
|
SUMMARY
|
1
|
|
|
|
|
RISK FACTORS
|
4
|
|
|
|
|
USE OF PROCEEDS
|
11
|
|
|
|
|
SELLING STOCKHOLDERS
|
11
|
|
|
|
|
PLAN OF DISTRIBUTION
|
16
|
|
|
|
|
DESCRIPTION OF SECURITIES
|
17
|
|
|
|
|
OUR BUSINESS
|
22
|
|
|
|
|
PROPERTIES
|
29
|
|
|
|
|
LEGAL PROCEEDINGS
|
29
|
|
|
|
|
MARKET FOR OUR COMMON STOCK AND RELATED STOCKHOLDER MATTERS
|
30
|
|
|
|
|
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
|
31
|
|
|
|
|
DIRECTORS AND EXECUTIVE OFFICERS
|
34
|
|
|
|
|
EXECUTIVE COMPENSATION
|
37
|
|
|
|
|
SHARE OWNERSHIP OF DIRECTORS, OFFICERS AND CERTAIN BENEFICIAL OWNERS
|
38
|
|
|
|
|
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS AND DIRECTOR INDEPENDENCE
|
39
|
|
|
|
|
LEGAL MATTERS
|
39
|
|
|
|
|
EXPERTS
|
39
|
|
|
|
|
WHERE YOU CAN GET MORE INFORMATION
|
40
|
|
|
|
ABOUT THIS PROSPECTUS
You should rely
only on the information contained in this prospectus or a prospectus supplement. We have not authorized any other person to provide
you with different information. If anyone provides you with different or inconsistent information, you should not rely on it. This
prospectus is an offer to sell only the common stock offered hereby, but only under circumstances and in jurisdictions where it
is lawful to do so. You should assume that the information appearing in this prospectus is accurate only as of the date hereof.
Our business, financial condition, results of operations and prospects may have changed.
The terms “Guided
Therapeutics,” “Company,” “our,” “we,” and “us,” as used in this prospectus,
refer to Guided Therapeutics, Inc. and its wholly owned subsidiary.
FORWARD-LOOKING STATEMENTS
Statements in this prospectus,
which express “belief,” “anticipation” or “expectation,” as well as other statements that are
not historical facts, are forward-looking statements. These forward-looking statements are subject to risks and uncertainties that
could cause actual results to differ materially from historical results or anticipated results, including those identified in the
foregoing “Risk Factors” and elsewhere in this prospectus. Examples of these uncertainties and risks include, but are
not limited to:
|
|
·
|
access to sufficient debt or equity capital to meet our operating
and financial needs;
|
|
|
·
|
the extent of dilution of the holdings of our existing stockholders
upon conversion or exercise of securities issued as part of our capital raising efforts;
|
|
|
·
|
the effectiveness and ultimate market acceptance of our products;
|
|
|
·
|
whether our products in development will prove safe, feasible and
effective;
|
|
|
·
|
whether and when we or any potential strategic partners will obtain
approval from the FDA and corresponding foreign agencies;
|
|
|
·
|
our need to achieve manufacturing scale-up in a timely manner, and
our need to provide for the efficient manufacturing of sufficient quantities of our products;
|
|
|
·
|
the lack of immediate alternate sources of supply for some critical
components of our products;
|
|
|
·
|
our patent and intellectual property position;
|
|
|
·
|
the need to fully develop the marketing, distribution, customer service
and technical support and other functions critical to the success of our product lines;
|
|
|
·
|
the dependence on potential strategic partners or outside investors
for funding, development assistance, clinical trials, distribution and marketing of some of our products; and
|
|
|
·
|
other risks and uncertainties described from time to time in our
reports filed with the SEC.
|
Forward-looking statements
should not be read as a guarantee of future performance or results, and will not necessarily be accurate indications of the times
at, or by which, such performance or results will be achieved. Forward-looking information is based on information available at
the time and/or management’s good faith belief with respect to future events, and is subject to risks and uncertainties that
could cause actual performance or results to differ materially from those expressed in the statements.
Forward-looking
statements speak only as of the date the statements are made. We assume no obligation to update forward-looking statements to reflect
actual results, changes in assumptions or changes in other factors affecting forward-looking information except to the extent required
by applicable securities laws. If we update one or more forward-looking statements, no inference should be drawn that we will make
additional updates with respect thereto or with respect to other forward-looking statements.
SUMMARY
This summary
highlights information contained elsewhere in this prospectus. This summary is not complete and may not contain all of the information
that may be important to you. We urge you to read the entire prospectus carefully, including the ‘‘Risk Factors’’
section, before making an investment decision.
Our Company
We are a medical
technology company focused on developing innovative medical devices that have the potential to improve healthcare. Our primary
focus is the development of our LuViva™ non-invasive cervical cancer detection device and extension of our cancer detection
technology into other cancers, including lung and esophageal. Our technology, including products in research and development, primarily
relates to biophotonics technology for the non-invasive detection of cancers.
We are a Delaware
corporation, originally incorporated in 1992 under the name “SpectRx, Inc.,” and, on February 22, 2008, changed
our name to Guided Therapeutics, Inc. At the same time, we renamed our wholly owned subsidiary, InterScan, which originally had
been incorporated as “Guided Therapeutics.”
For the years ended
December 31, 2013 and 2012, we reported net losses of $7.2 million and $4.4 million, respectively.
Non-Invasive Cervical Cancer Detection
We believe LuViva will
provide a less invasive and painless alternative to conventional tests for cervical cancer detection. We also believe LuViva can
improve patient well-being and reduce healthcare costs, since it reduces or eliminates pain, is convenient to use and provides
rapid results at the point-of-care. We completed enrollment in our U.S. Food and Drug Administration (“FDA”) pivotal
trial of LuViva in 2008 and on November 18, 2010, the FDA accepted our completed premarket approval (“PMA”) application,
effective September 23, 2010, for substantive review. On March 7, 2011, we announced that the FDA had inspected two clinical trial
sites as part of its review process and raised no formal compliance issues. On January 12, 2012, we announced our intent to seek
an independent panel review of our PMA application after receiving a “not-approvable” letter from the FDA. On November
14, 2012 we filed an amended PMA with FDA. On September 6, 2013 we received a letter from the FDA with additional questions, and
we are scheduled to meet with the FDA on May 8, 2014 to discuss our response. The FDA has granted us a 180-day extension on the
PMA filing to allow time for the meeting to occur. Assuming we can respond to FDA’s most recent questions effectively and
in a timely manner, and then receive FDA approval in 2014, we currently anticipate a 2015 product launch in the United States,
but cannot be assured we will be able to launch on that timetable, or at all. Internationally, we have regulatory approval to sell
LuViva in Europe upon receipt of our Edition 3CE Mark in January 2014. LuViva has marketing approval from Health Canada and the
Singapore Health Sciences Authority, and we have applied for approval in Mexico.
Other Cancers
We believe our
non-invasive cervical cancer detection technology can be applied to other cancers as well. To that end, from 2008 until early 2013
we had worked exclusively with Konica Minolta Opto, Inc., a subsidiary of Konica Minolta, Inc., a Japanese corporation based in
Tokyo (“Konica Minolta”), to adapt our cervical cancer detection technology primarily for the detection of esophageal
cancer. On February 6, 2013, we announced that we had terminated and replaced our existing agreements with Konica Minolta with
a new license agreement allowing us to manufacture and to develop a non-invasive esophageal cancer detection product from Konica
Minolta and based on our biophotonic technology platform (see “Our Business—Lung and Esophageal Cancer Detection—Konica
Minolta”).
Recent Developments
On
April 23, 2014, we entered into a securities purchase agreement with Hanover Holdings I, LLC, an affiliate of Magna
Group, referred to as Hanover. Pursuant to the purchase agreement, we sold Hanover a 6% senior convertible note with a
principal amount of $1.5 million and an 18-month term, for a purchase price of $1.0 million (an approximately 33.3% original
issue discount). Additionally, Hanover is irrevocably bound to purchase, on the tenth trading day after the effective date
of the resale registration statement, an additional 6% senior convertible note with a principal amount of $2.0 million and
an 18-month term, for a fixed purchase price of $2.0 million, subject only to conditions outside of Hanover’s control
or that Hanover cannot cause not to be satisfied, none of which are related to the market price of our common stock. Subject
to certain limitations, the senior convertible notes are convertible at any time after the earlier of October 23, 2014 or
the effectiveness of a resale registration statement, in whole or in part, at Hanover’s option, into shares of our
common stock, at a conversion price equal to the lesser of $0.55 per share and a discount from the lowest daily
volume-weighted average price of our common stock in the five trading days prior to conversion. The discount is 20% if the
conversion takes place prior to December 19, 2014, and 25% if after that date. We paid Hanover a commitment fee for entering
into the purchase agreement in the form of 321,820 shares of common stock.
See “Description of Securities—Senior Convertible Notes”. The resale by Hanover of the shares issued in
connection with, or issuable upon conversion of, the senior convertible notes are covered by this prospectus.
The Offering
|
Common stock that may be offered by the selling stockholders
|
43,646,992 shares of our common stock. See “Selling Stockholders” on page 11.
|
|
Use of proceeds
|
We will not receive any proceeds from the resale of the shares of common stock. However, at the time the senior convertible notes, the Series B convertible preferred stock, or other securities were originally issued to the selling stockholders, as applicable, we did receive payment for the purchase price for those securities. In addition, to the extent the warrants were or are exercised in whole or in part for cash, we have received or will receive payment for the exercise price. The terms of the warrants are described under “Description of Securities—Warrants and Options.” We intend to apply any proceeds received in connection with the exercise of the warrants to increase inventory of our LuViva advanced cervical device to meet current demand for the product, expand our international marketing and sales efforts and continue to seek FDA approval for the LuViva device. However, we will retain broad discretion over the use of the net proceeds and may use the money for other corporate purposes. See “Use of Proceeds” on page 11.
|
|
Market for the common stock
|
Our common stock is listed on the OTCQB marketplace under the symbol “GTHP.” See “Market for Our Common Stock and Related Stockholder Matters” on page 30.
|
|
Risk factors
|
You should read “Risk Factors” beginning on page 4 for an explanation of the risks of investing in our common stock.
|
In addition to
the summary of our offering of the senior convertible notes set forth in “Recent Developments” above, the following
is a summary of the other transactions relating to the securities offered in this prospectus by the selling stockholders.
|
|
·
|
From 2007 to 2010, in connection with various capital-raising transactions,
we issued warrants exercisable for up to 29,832,949 shares of our common stock at exercise prices ranging from $0.65 to $1.01 per
share. The resale by the selling stockholders of the underlying shares issued upon exercise of these warrants were originally covered
by a separate prospectus under a different registration statement, but the remaining unsold shares are now covered by this prospectus.
|
|
|
·
|
On August 30, 2011, we entered into an agreement and release with
certain of our stockholders by which those stockholders agreed to terminate all of their rights under a prior agreement and release
all claims, in exchange for warrants to purchase an aggregate of 2.6 million shares of our common stock, certain royalties related
to the sale of disposables in conjunction with our cervical cancer detection technology and certain additional payments related
to non-ordinary course asset sales or our sale of by merger, with such royalties and related payments subject to certain “caps”
limiting their amounts. The resale by those stockholders of the underlying shares issued upon exercise of these warrants were originally
covered by a separate prospectus under a different registration statement, but the remaining unsold shares are now covered by this
prospectus.
|
|
|
·
|
In July 2012, we completed a warrant exchange program, pursuant to
which we exchanged warrants exercisable for a total of 15,941,640 shares of common stock, or 56.29% of the warrants eligible to
participate, for three classes of new warrants. The first class of new warrants expired on September 17, 2012 and carried an exercise
price of $0.40, $0.45 or $0.50, depending on the date exercised. The second classes of new warrants expired on either July 26,
2013 or March 1, 2014, and were exercisable at $0.65. The third class of new warrants are exercisable for approximately 472,000
shares and 3.6 million shares at $0.80 per share and expire on July 26, 2014 and March 1, 2015, respectively. The resale by participating
warrant holders of the underlying shares issued or issuable upon exercise of these were originally covered by a separate prospectus
under a different registration statement, but the remaining unsold shares are now covered by this prospectus.
|
|
|
·
|
On May 23, 2013, we completed a private placement of our
Series B
convertible preferred stock and warrants to purchase shares of our common stock. We issued an aggregate of 2,527 shares of
Series B convertible preferred stock at a purchase price of $1,000 per share. The initial conversion price of the Series B
convertible preferred stock was $0.68 per share, such that each share would convert into 1,471 shares of our common stock,
subject to customary adjustments, including for any accrued but unpaid dividends and pursuant to certain anti-dilution
provisions. We also issued warrants, on a pro rata basis to the investors, exercisable to purchase an aggregate of 3,716,177
shares of our common stock. The warrants, which carry a five-year term, were split evenly into two tranches, one of which is
subject to a mandatory exercise provision. The warrants are exercisable at any time and had an initial exercise price of
$1.08 per share, subject to certain customary adjustments contained in the respective warrants. As a result of the November
2013 warrant exchange program described below, the conversion price of the Series B convertible preferred stock has been
lowered to $0.40 per share, such that each share is now convertible into 2,500 shares of common stock, and one tranche of
the warrants,
previously exercisable for 1,858,089 shares of common stock at $1.08 per share, is now exercisable for
5,016,840 shares at $0.40 per share. The resale by Series B investors of the shares issued or issuable upon conversion of
the Series B
convertible preferred stock (including shares issuable as dividends) and exercise of the warrants were originally
covered by a separate prospectus under a different registration statement, but the remaining unsold shares are now covered
by this
prospectus, as are the additional shares issuable as a result of the lower conversion price and exercise price.
|
|
|
·
|
In November 2013, we completed another warrant exchange program pursuant
to which we exchanged warrants exercisable for a total of 3,573,691 shares of common stock, or 99.5% of the warrants eligible to
participate, for new warrants exercisable for the same number of shares of common stock, but with a reduced exercise price of $0.40
per share and a shortened exercise period ending on November 27, 2013. The resale by participating warrant holders of the underlying
shares issued upon exercise of these warrants are covered by this prospectus.
|
As of April
24, 2014, we had 59,589,812 shares outstanding and held by stockholders other than affiliates (our “public float”).
As detailed above, this prospectus covers both (1) shares of common stock newly registered for resale, and (2) previously registered
on prior registration statements, but remaining unsold. The following table categorizes the shares offered by this prospectus
by whether they are being newly registered or whether they were originally registered under a prior registration statement but
remain, and lists each category’s percentage of our public float:
|
|
# of Shares
|
% of Public
Float
|
Shares newly registered by this prospectus in connection with the
issuance of the senior convertible notes
|
9,163,656
|
15.4%
|
Shares newly registered by this prospectus in connection with the
November 2013 warrant exchange program
|
3,573,691
|
6.0%
|
Shares newly registered by this prospectus as a result of the operation
of anti-dilution provisions applicable to our Series B convertible
preferred stock and related warrants, originally issued in May 2013
|
6,096,379
|
10.2%
|
|
Total newly registered shares
|
18,833,726
|
31.6%
|
|
|
|
|
Shares originally registered on prior registration statements but
remaining unsold
|
24,813,266
|
41.6%
|
Our principal
executive and operations facility is located at 5835 Peachtree Corners East, Suite D, Norcross, Georgia 30092, and our telephone
number is (770) 242-8723.
RISK FACTORS
Your investment in shares of our common
stock involves substantial risks. In consultation with your own advisers, you should carefully consider, among other matters, the
factors set forth below before deciding whether an investment in shares of our common stock is suitable for you. If any of the
risks contained in this prospectus develop into actual events, our business, financial condition, liquidity, results of operations
and prospects could be materially and adversely affected, the market price of our common stock could decline and you may lose all
or part of your investment. Some statements in this prospectus, including statements in the following risk factors, constitute
forward-looking statements. See “Forward-Looking Statements” in this prospectus.
Although we will be required to raise additional funds in
the third quarter of 2014, there is no assurance that such funds can be raised on terms that we would find acceptable, or at all.
Additional debt or equity
financing will be required for us to continue as a going concern. Management may seek to obtain additional funds for the financing
of our cervical cancer detection business, through additional debt or equity financings and/or new collaborative arrangements.
Management believes that additional financing, if obtainable, will be sufficient to support planned operations only for a limited
period. Management has implemented operating actions to reduce cash requirements. Any required additional funding may not be available
on terms attractive to us or at all.
If we cannot obtain additional funds or achieve profitability,
we may not be able to continue as a going concern.
Because we must obtain
additional funds through further financing transactions or through new collaborative arrangements in order to execute our plans
to launch our cervical cancer detection product line and to generate revenue from operations, there exists substantial doubt about
our ability to continue as a going concern. Therefore, it will be necessary to raise additional funds. There can be no assurance
that we will be able to raise these additional funds. If we do not secure additional funding when needed, we will be unable to
conduct all of our product development efforts as planned, which may cause us to alter our business plan in relation to the development
of our products. Even if we obtain additional funding, we will need to achieve profitability thereafter.
Our independent registered
public accountants’ report on our consolidated financial statements as of and for the year ended December 31, 2013, indicated
that there was substantial doubt about our ability to continue as a going concern because we had suffered recurring losses from
operations and had an accumulated deficit of $103.0 million at December 31, 2013,
summarized as
follows:
|
|
Accumulated deficit from inception to fiscal year ended 2011:
|
|
$85.0 million
|
|
|
|
|
|
|
|
Net Loss for fiscal year 2012, ended 12/31/2011:
|
|
$4.4 million
|
|
|
|
|
|
|
|
Deemed dividends for fiscal year 2012, ended 12/31/2012:
|
|
$2.7 million
|
|
|
|
|
|
|
|
Accumulated deficit at fiscal year ended 12/31/2012:
|
|
$92.1 million
|
|
|
|
|
|
|
|
Net Loss for fiscal year 2013, ended 12/31/2013:
|
|
$7.2 million
|
|
|
|
|
|
|
|
Deemed dividends for fiscal year 2013, ended 12/31/2013:
|
|
$3.7 million
|
|
|
|
|
|
|
|
Accumulated deficit, from inception to 12/31/2013:
|
|
$103.0 million
|
Our management has implemented
reductions in operating expenditures and reductions in some development activities. We have determined to make cervical cancer
detection the focus of our business. We are managing the development of our other programs only when funds are made available to
us via grants or contracts with government entities or strategic partners. However, there can be no assurance that we will be able
to successfully implement or continue these plans.
If we cannot obtain additional funds when needed, we will
not be able to implement our business plan.
We will require
substantial additional capital to develop our products, including completing product testing and clinical trials, obtaining
all required regulatory approvals and clearances, beginning and scaling up manufacturing, and marketing our products. We have
historically financed our operations though the private sale of preferred stock and debt securities, public and private sales
of common stock, funding from collaborative arrangements, and grants. We believe funds on hand as of the date of this
prospectus, along with funds from government contracts and grants, will be sufficient to support planned operations through
the end of the second quarter of 2014, but will not be sufficient to fund our planned operations to the point of commercial introduction of our LuViva cervical
cancer detection device. Any failure to achieve adequate funding in a timely fashion would delay our development programs and could
lead to abandonment of one or more of our development initiatives. To the extent we cannot obtain additional funding, our ability
to continue to develop and introduce products to market will be limited. Further, financing our operations through the public or
private sale of debt or equity may involve restrictive covenants or other provisions that could limit how we conduct our business
or finance our operations. Financing our operations through collaborative arrangements generally means that the obligations of
the collaborative partner to fund our expenditures are largely discretionary and depend on a number of factors, including our ability
to meet specified milestones in the development and testing of the relevant product. We may not be able to obtain an acceptable
collaboration partner, and even if we do, we may not be able to meet these milestones, or the collaborative partner may not continue
to fund our expenditures.
We do not have a long operating history, especially in the
cancer detection field, which makes it difficult to evaluate our business.
Although we have been
in existence since 1992, we have only just begun the process of commercializing our cervical cancer detection technology. Because
limited historical information is available on our revenue trends and operations for our cancer detection programs it is difficult
to evaluate our business. Our prospects must be considered in light of the substantial risks, expenses, uncertainties and difficulties
encountered by entrants into the medical device industry, which is characterized by increasing intense competition and a high failure
rate.
We have a history of losses, and we expect losses to continue.
We have never been profitable
and we have had operating losses since our inception. We expect our operating losses to continue as we continue to expend substantial
resources to complete development of our products, obtain regulatory clearances or approvals, and build our marketing, sales, manufacturing
and finance organizations, and conduct further research and development. To date, we have engaged primarily in research and development
efforts. The further development and commercialization of our products will require substantial development, regulatory, sales
and marketing, manufacturing and other expenditures. We have only generated limited revenues from product sales. Our accumulated
deficit was approximately $103.0 million at December 31, 2013.
Our ability to sell our products is controlled by government
regulations, and we may not be able to obtain any necessary clearances or approvals.
The design, manufacturing,
labeling, distribution and marketing of medical device products are subject to extensive and rigorous government regulation, which
can be expensive and uncertain and can cause lengthy delays before we can begin selling our products.
In the United States, the FDA’s
actions could delay or prevent our ability to sell our products, which would adversely affect our growth and strategy plans.
In order for us to market
our products in the United States, we must obtain clearance or approval from the FDA. We cannot be sure that:
|
|
·
|
we, or any collaborative partner, will make timely filings with the
FDA;
|
|
|
·
|
the FDA will act favorably or quickly on these submissions;
|
|
|
·
|
we will not be required to submit additional information or perform
additional clinical studies; or
|
|
|
·
|
other significant difficulties and costs will not be encountered
to obtain FDA clearance or approval.
|
It can take several years
from initial filing of a PMA application and require the submission of extensive supporting data and clinical information. The
FDA may impose strict labeling or other requirements as a condition of its clearance or approval, any of which could limit our
ability to market our products. Further, if we wish to modify a product after FDA approval of a PMA application, including changes
in indications or other modifications that could affect safety and efficacy, additional clearances or approvals will be required
from the FDA. Any request by the FDA for additional data, or any requirement by the FDA that we conduct additional clinical studies,
could result in a significant delay in bringing our products to market and substantial additional research and other expenditures.
Similarly, any labeling or other conditions or restrictions imposed by the FDA could hinder our ability to effectively market our
products. Any of the above actions by the FDA could delay or prevent altogether our ability to market and distribute our products.
Further, there may be new FDA policies or changes in FDA policies that could be adverse to us.
In foreign countries, including European countries, we are
also subject to government regulation, which could delay or prevent our ability to sell our products in those jurisdictions.
In order for us to market
our products in Europe and some other international jurisdictions, we and our distributors and agents must obtain required regulatory
registrations or approvals. We must also comply with extensive regulations regarding safety, efficacy and quality in those jurisdictions.
We may not be able to obtain the required regulatory registrations or approvals, or we may be required to incur significant costs
in obtaining or maintaining any regulatory registrations or approvals we receive. Delays in obtaining any registrations or approvals
required for marketing our products, failure to receive these registrations or approvals, or future loss of previously obtained
registrations or approvals would limit our ability to sell our products internationally. For example, international regulatory
bodies have adopted various regulations governing product standards, packaging requirements, labeling requirements, import restrictions,
tariff regulations, duties and tax requirements. These regulations vary from country to country. In order to sell our products
in Europe, we must maintain ISO 13485:2003 certification and CE mark certification, which is an international symbol of quality
and compliance with applicable European medical device directives. Failure to maintain ISO 13485:2003 certification or CE mark
certification or other international regulatory approvals would prevent us from selling in some countries in the European Union
and elsewhere.
Even if we obtain clearance or approval
to sell our products, we are subject to ongoing requirements and inspections that could lead to the restriction, suspension or
revocation of our clearance.
We, as well as any potential
collaborative partners, will be required to adhere to applicable FDA regulations regarding good manufacturing practice, which include
testing, control, and documentation requirements. We are subject to similar regulations in foreign countries. Ongoing compliance
with good manufacturing practice and other applicable regulatory requirements is strictly enforced in the United States through
periodic inspections by state and federal agencies, including the FDA, and in international jurisdictions by comparable agencies.
Failure to comply with these regulatory requirements could result in, among other things, warning letters, fines, injunctions,
civil penalties, recall or seizure of products, total or partial suspension of production, failure to obtain premarket clearance
or premarket approval for devices, withdrawal of approvals previously obtained, and criminal prosecution. The restriction, suspension
or revocation of regulatory approvals or any other failure to comply with regulatory requirements would limit our ability to operate
and could increase our costs.
Our success largely depends on our ability
to obtain and protect the proprietary information on which we base our products.
Our success depends in
large part upon our ability to establish and maintain the proprietary nature of our technology through the patent process, as well
as our ability to license from others patents and patent applications necessary to develop our products. If any of our patents
are successfully challenged, invalidated or circumvented, or our right or ability to manufacture our products was to be limited,
our ability to continue to manufacture and market our products could be adversely affected. In addition to patents, we rely on
trade secrets and proprietary know-how, which we seek to protect, in part, through confidentiality and proprietary information
agreements. The other parties to these agreements may breach these provisions, and we may not have adequate remedies for any breach.
Additionally, our trade secrets could otherwise become known to or be independently developed by competitors.
As of December 31, 2013,
we have been issued, or have rights to, 19 U.S. patents (including those under license). In addition, we have filed for, or have
rights to, four U.S. patents
(including those under license) that are still pending. There are additional international
patents and pending applications. One or more of the patents we hold directly or license from third parties, including those for
our cervical cancer detection products, may be successfully challenged, invalidated or circumvented, or we may otherwise be unable
to rely on these patents. These risks are also present for the process we use or will use for manufacturing our products. In addition,
our competitors, many of whom have substantial resources and have made substantial investments in competing technologies, may apply
for and obtain patents that prevent, limit or interfere with our ability to make, use and sell our products, either in the United
States or in international markets.
The medical device industry
has been characterized by extensive litigation regarding patents and other intellectual property rights. In addition, the U.S.
Patent and Trademark Office, or USPTO, may institute interference proceedings. The defense and prosecution of intellectual property
suits, USPTO proceedings and related legal and administrative proceedings are both costly and time consuming. Moreover, we may
need to litigate to enforce our patents, to protect our trade secrets or know-how, or to determine the enforceability, scope and
validity of the proprietary rights of others. Any litigation or interference proceedings involving us may require us to incur substantial
legal and other fees and expenses and may require some of our employees to devote all or a substantial portion of their time to
the proceedings. An adverse determination in the proceedings could subject us to significant liabilities to third parties, require
us to seek licenses from third parties or prevent us from selling our products in some or all markets. We may not be able to reach
a satisfactory settlement of any dispute by licensing necessary patents or other intellectual property. Even if we reached a settlement,
the settlement process may be expensive and time consuming, and the terms of the settlement may require us to pay substantial royalties.
An adverse determination in a judicial or administrative
proceeding or the failure to obtain a necessary license could prevent us from manufacturing and selling our products.
We may not be able to generate sufficient
sales revenues to sustain our growth and strategy plans.
Our cervical cancer diagnostic
activities have been financed to date through a combination of government grants, strategic partners and direct investment. Bringing
this product to market is the main focus of our business. In order to complete product development and prepare for marketing of
the cervical cancer detection product, additional capital will be needed. We need to complete the FDA filing process for our cervical
cancer diagnostic product and obtain capital investment for product development and launch.
Additional product lines
involve the modification of the cervical cancer detection technology for use in other cancers. These product lines are only in
the earliest stages of research and development and are currently not projected to reach market for several years. Our goal is
to receive enough funding from government grants and contracts, as well as payments from strategic partners, to fund development
of these product lines without diverting funds or other necessary resources from the cervical cancer program.
Because our products, which use different
technology or apply technology in different ways than other medical devices, are or will be new to the market, we may not be successful
in launching our products and our operations and growth would be adversely affected.
Our products are based
on new methods of cancer detection. If our products do not achieve significant market acceptance, our sales will be limited and
our financial condition may suffer. Physicians and individuals may not recommend or use our products unless they determine that
these products are an attractive alternative to current tests that have a long history of safe and effective use. To date, our
products have been used by only a limited number of people, and few independent studies regarding our products have been published.
The lack of independent studies limits the ability of doctors or consumers to compare our products to conventional products.
If we are unable to compete effectively
in the highly competitive medical device industry, our future growth and operating results will suffer.
The medical device industry
in general and the markets in which we expect to offer products in particular, are intensely competitive. Many of our competitors
have substantially greater financial, research, technical, manufacturing, marketing and distribution resources than we do and have
greater name recognition and lengthier operating histories in the health care industry. We may not be able to effectively compete
against these and other competitors. A number of competitors are currently marketing traditional laboratory-based tests for cervical
cancer screening and diagnosis. These tests are widely accepted in the health care industry and have a long history of accurate
and effective use. Further, if our products are not available at competitive prices, health care administrators who are subject
to increasing pressures to reduce costs may not elect to purchase them. Also, a number of companies have announced that they are
developing, or have introduced, products that permit non-invasive and less invasive cancer detection. Accordingly, competition
in this area is expected to increase.
Furthermore, our competitors
may succeed in developing, either before or after the development and commercialization of our products, devices and technologies
that permit more efficient, less expensive non-invasive and less invasive cancer detection. It is also possible that one or more
pharmaceutical or other health care companies will develop therapeutic drugs, treatments or other products that will substantially
reduce the prevalence of cancers or otherwise render our products obsolete.
We have little manufacturing experience,
which could limit our growth.
We do not have manufacturing
experience that would enable us to make products in the volumes that would be necessary for us to achieve significant commercial
sales, and we rely upon our suppliers. In addition, we may not be able to establish and maintain reliable, efficient, full scale
manufacturing at commercially reasonable costs in a timely fashion. Difficulties we encounter in manufacturing scale-up, or our
failure to implement and maintain our manufacturing facilities in accordance with good manufacturing practice regulations, international
quality standards or other regulatory requirements, could result in a delay or termination of production. To date, our manufacturing
activities have included since-discontinued products. We had substantial difficulties in establishing and maintaining manufacturing
for these products and those difficulties impacted our ability to increase sales. Companies often encounter difficulties in scaling
up production, including problems involving production yield, quality control and assurance, and shortages of qualified personnel.
Since we rely on sole source suppliers
for our products, any failure of those suppliers to perform would hurt our operations.
Several of the components
used in our current or planned products are available from only one supplier, and substitutes for these components cannot be obtained
easily or would require substantial design or manufacturing modifications. Any significant problem experienced by one of our sole
source suppliers may result in a delay or interruption in the supply of components to us until that supplier cures the problem
or an alternative source of the component is located and qualified. Any delay or interruption would likely lead to a delay or interruption
in our manufacturing operations. The inclusion of substitute components must meet our product specifications and could require
us to qualify the new supplier with the appropriate government regulatory authorities.
Because we operate in an industry with
significant product liability risk, and we have not specifically insured against this risk, we may be subject to substantial claims
against our products.
The development, manufacture
and sale of medical products entail significant risks of product liability claims. We currently have no product liability insurance
coverage beyond that provided by our general liability insurance. Accordingly, we may not be adequately protected from any liabilities,
including any adverse judgments or settlements, we might incur in connection with the development, clinical testing, manufacture
and sale of our products. A successful product liability claim or series of claims brought against us that result in an adverse
judgment against or settlement by us in excess of any insurance coverage could seriously harm our financial condition or reputation.
In addition, product liability insurance is expensive and may not be available to us on acceptable terms, if at all.
The availability of third party reimbursement
for our products is uncertain, which may limit consumer use and the market for our products.
In the United States and
elsewhere, sales of medical products are dependent, in part, on the ability of consumers of these products to obtain reimbursement
for all or a portion of their cost from third-party payors, such as government and private insurance plans. Any inability of patients,
hospitals, physicians and other users of our products to obtain sufficient reimbursement from third-party payors for our products,
or adverse changes in relevant governmental policies or the policies of private third-party payors regarding reimbursement for
these products, could limit our ability to sell our products on a competitive basis. We are unable to predict what changes will
be made in the reimbursement methods used by third-party health care payors. Moreover, third-party payors are increasingly challenging
the prices charged for medical products and services, and some health care providers are gradually adopting a managed care system
in which the providers contract to provide comprehensive health care services for a fixed cost per person. Patients, hospitals
and physicians may not be able to justify the use of our products by the attendant cost savings and clinical benefits that we believe
will be derived from the use of our products, and therefore may not be able to obtain third-party reimbursement.
Reimbursement and health
care payment systems in international markets vary significantly by country and include both government-sponsored health care and
private insurance. We may not be able to obtain approvals for reimbursement from these international third-party payors in a timely
manner, if at all. Any failure to receive international reimbursement approvals could have an adverse effect on market acceptance
of our products in the international markets in which approvals are sought.
Our success depends on our ability to
attract and retain scientific, technical, managerial and finance personnel.
Our ability to operate
successfully and manage our future growth depends in significant part upon the continued service of key scientific, technical,
managerial and finance personnel, as well as our ability to attract and retain additional highly qualified personnel in these fields.
We may not be able to attract and retain key employees when necessary, which would limit our operations and growth. Only our Chief
Executive Officer, our Chief Scientific Officer and our Senior Vice President of Engineering have employment contracts with us,
and none of our employees are covered by key person or similar insurance. In addition, if we are able to successfully develop and
commercialize our products, we will need to hire additional scientific, technical, marketing, managerial and finance personnel.
We face intense competition for qualified personnel in these areas, many of whom are often subject to competing employment offers.
We are significantly influenced by our
directors, executive officers and their affiliated entities.
Our directors, executive
officers and entities affiliated with them beneficially owned an aggregate of about 23.39% of our outstanding common stock as of
April 24, 2014. These stockholders, acting together, would be able to exert significant influence on substantially all matters
requiring approval by our stockholders, including the election of directors and the approval of mergers and other business combination
transactions.
Our stock is thinly traded, so you may be unable to sell at
or near ask prices or at all.
The shares of our common
stock are listed on the OTCQB marketplace. Shares of our common stock are thinly traded, meaning that the number of persons interested
in purchasing our common shares at or near ask prices at any given time may be relatively small or non-existent. This situation
is attributable to a number of factors, including:
|
|
·
|
we are a small company that is relatively unknown to stock analysts,
stock brokers, institutional investors and others in the investment community that generate or influence sales volume; and
|
|
|
·
|
stock analysts, stock brokers and institutional investors may be
risk-averse and be reluctant to follow a company such as ours that faces substantial doubt about its ability to continue as a going
concern or to purchase or recommend the purchase of our shares until such time as we became more viable.
|
As a consequence, our stock price may not reflect
an actual or perceived value. Also, there may be periods of several days or more when trading activity in our shares is minimal
or non-existent, as compared to a seasoned issuer that has a large and steady volume of trading activity that will generally support
continuous sales without an adverse effect on share price. A broader or more active public trading market for our common shares
may not develop or if developed, may not be sustained. Due to these conditions, you may not be able to sell your shares at or near
ask prices or at all if you need money or otherwise desire to liquidate your shares.
Trading in our common stock is subject to special sales practices
and may be difficult to sell.
Our common stock is subject
to the SEC’s “penny stock” rule, which imposes special sales practice requirements upon broker-dealers who sell
such securities to persons other than established customers or accredited investors. Penny stocks are generally defined to be an
equity security that has a market price of less than $5.00 per share. For transactions covered by the rule, the broker-dealer must
make a special suitability determination for the purchaser and receive the purchaser’s written agreement to the transaction
prior to the sale. Consequently, the rule may affect the ability of broker-dealers to sell our securities and also may affect the
ability of our stockholders to sell their securities in any market that might develop.
Stockholders should be aware
that, according to SEC Release No. 34-29093, the market for penny stocks has suffered from patterns of fraud and abuse. Such patterns
include:
|
|
·
|
control of the market for the security by one or a few broker-dealers
that are often related to the promoter or issuer;
|
|
|
·
|
manipulation of prices through prearranged matching of purchases
and sales and false and misleading press releases;
|
|
|
·
|
“boiler room” practices involving high-pressure sales
tactics and unrealistic price projections by inexperienced sales persons;
|
|
|
·
|
excessive and undisclosed bid-ask differentials and markups by selling
broker-dealers; and
|
|
|
·
|
the wholesale dumping of the same securities by promoters and broker-dealers
after prices have been manipulated to a desired level, along with the resulting inevitable collapse of those prices and with consequent
investor losses.
|
Our management is aware
of the abuses that have occurred historically in the penny stock market. Although we do not expect to be in a position to dictate
the behavior of the market or of broker-dealers who participate in the market, management will strive within the confines of practical
limitations to prevent the described patterns from being established with respect to our common stock.
The number of shares of our common stock
issuable upon the conversion of our outstanding senior convertible notes and Series B convertible preferred stock or exercise
of outstanding warrants and options is substantial.
The outstanding
and to-be issued senior convertible notes, if converted immediate upon issuance, would have been convertible into an aggregate
of 8,341,656 shares of our common stock. The outstanding shares of our Series B convertible preferred stock are currently convertible
into an aggregate of 4,342,500 shares of our common stock. In addition, we currently have warrants outstanding and issuable that
are exercisable for an aggregate of 11,969,166 shares and outstanding options for 8,420,703 shares. Together, the shares of common
stock issuable upon conversion or exercise of these securities constitute approximately 42.7% of the total number of shares of
common stock currently issued and outstanding.
Substantial future sales of shares of
our common stock in the public market could cause our stock price to fall.
If our common stockholders
(including those persons who may become common stockholders upon conversion of our senior convertible notes or Series B convertible
preferred stock or exercise of our warrants) sell substantial amounts of our common stock, or the public market perceives
that stockholders might sell substantial amounts of our common stock, the market price of our common stock could decline significantly.
Such sales also might make it more difficult for us to sell equity or equity-related securities in the future at a time and price
that our management deems appropriate.
In addition, our Series
B convertible preferred stock and certain of our outstanding warrants contain anti-dilution provisions that may, under certain
circumstances, reduce the conversion or exercise price or increase the number of shares issuable, or both.
Our need to raise additional capital
in the near future or to use our equity securities for payments could have a dilutive effect on your investment.
In order to continue
operations, we will need to raise additional capital. We may attempt to raise capital through the public or private sale of our
common stock or securities convertible into or exercisable for our common stock. In addition, from time to time we have issued
our common stock or warrants in lieu of cash payments. If we sell additional shares of our common stock or other equity securities,
or issue such securities in respect of other claims or indebtedness, such sales or issuances will further dilute the percentage
of our equity that you own. Depending upon the price per share of securities that we sell or issue in the future, if any, your
interest in us could be further diluted by any adjustments to the number of shares and the applicable exercise price required pursuant
to the terms of the agreements under which we previously issued securities.
Adjustments to the conversion price for
our senior convertible notes or our Series B convertible preferred stock, and the exercise price for certain of our warrants, will
dilute the ownership interests of our existing stockholders.
Under the terms of our
senior convertible notes, the conversion price fluctuates with the market price of our common stock. Accordingly, if the market
price of our common stock decreases, the number of shares issuable upon conversion of the senior convertible notes will increase,
and may result in the issuance of a significant number of additional shares of our common stock upon conversion.
Under the terms of our
Series B convertible preferred stock and certain warrants issued with the Series B convertible preferred stock, subject to certain
exceptions, the conversion price for the Series B convertible preferred stock and the exercise price for the warrants will be lowered
if we issue common stock at a per share price below the then conversion price for the Series B convertible preferred stock or the
then exercise price for the warrants, respectively. Reductions in the conversion price for the Series B convertible preferred stock
and the exercise price for the warrants may result in the issuance of a significant number of additional shares of our common stock
upon conversion or exercise of these securities, which could result in dilution in the value of the shares of our outstanding common
stock and the voting power represented thereby. Due to a warrant exchange program completed in November 2013, the conversion price
of the Series B convertible preferred stock has been lowered from $0.68 per share to $0.40 per share, such that each share is now
convertible into 2,500 shares of common stock, and one tranche of the warrants, previously exercisable for 1,858,089 shares of
common stock at $1.08 per share, is now exercisable for 5,016,840 shares at $0.40 per share. Depending on the market price of our
common stock at the time our senior convertible notes become convertible by the holder, there may be a further reduction in the
conversion price of the Series B convertible preferred stock and the exercise price of those warrants, as well as an increase in
the number of shares underlying those warrants.
Certain provisions of our certificate
of incorporation that authorize the issuance of additional shares of preferred stock may make it more difficult for a third party
to effect a change in control.
Our
certificate of incorporation authorizes our board of directors to issue up to 5 million shares of preferred stock. We have
issued 2,527 shares of Series B convertible preferred stock. We believe the terms of our Series B convertible preferred stock
would not have a substantial impact on the ability of a third party to effect a change in control. The remaining shares of
preferred stock may be issued in one or more series, the terms of which may be determined by the board without further
stockholder action. These terms may include, among other terms, voting rights, including the right to vote as a series on
particular matters, preferences as to liquidation and dividends, repurchase rights, conversion rights, redemption rights and
sinking fund provisions. The issuance of any preferred stock could diminish the rights of holders of our common stock, and
therefore could reduce the value of our common stock. In addition, specific rights granted to future holders of preferred
stock could be used to restrict our ability to merge with or sell assets to a third party. The ability of our board to issue
preferred stock could make it more difficult, delay, discourage, prevent or make it more costly to acquire or effect a change
in control, which in turn could prevent our stockholders from recognizing a gain in the event that a favorable offer is
extended and could materially and negatively affect the market price of our common stock.
USE OF PROCEEDS
We will not receive
any proceeds from the resale of the shares of common stock offered by the selling stockholders listed in this prospectus under
“Selling Stockholders”. However, at the time the senior convertible notes, the Series B convertible preferred stock,
or other securities were originally issued to the selling stockholders, as applicable, we did receive payment for the purchase
price for those securities. To date, warrants exercisable for 11,998,822 shares of our common stock to which this prospectus relates
have been exercised by the selling stockholders. We may receive the proceeds from the exercise of the remaining warrants entitling
the selling stockholders to purchase shares of our common stock. If all such remaining warrants were exercised for cash on April
24, 2014, we would have received a weighted average of $0.67 per underlying share as to 11,998,822 shares, or an aggregate of about
$8.1 million, in cash proceeds.
We intend to apply
any proceeds received in connection with the exercise of the warrants to increase inventory of our LuViva advanced cervical device
to meet current demand for the product, expand our international marketing and sales efforts and continue to seek FDA approval
for the LuViva device. However, we will retain broad discretion over the use of the net proceeds and may use the money for other
corporate purposes.
SELLING STOCKHOLDERS
The shares of our
common stock to which this prospectus relates consist of 9,163,656 shares of common stock issued in connection with, or issuable
upon conversion of, our senior convertible notes, 14,365,343 shares of common stock issued in connection with, or issuable upon
conversion of, our Series B convertible preferred stock (including shares paid or that may be paid as dividends on the Series B
convertible preferred stock pursuant to its terms, payable through December 31, 2015, and shares issued upon exercise of warrants
issued in connection with the Series B convertible preferred stock), and 20,117,993 shares of common stock issued or issuable upon
exercise of certain warrants (other than those issued in connection with the Series B convertible preferred stock). We issued the
senior convertible notes, shares of our Series B convertible preferred stock, and warrants to the selling stockholders in various
exchange offers and private placements and exchange offers exempt from registration under the Securities Act in reliance upon Sections
3(a)(9) and 4(a)(2) of the Securities Act.
In connection with
the private placement of our senior convertible notes, pursuant to a registration rights agreement dated April 23, 2014 that we
entered into with one of the selling stockholders, we granted certain customary registration rights covering the shares of our
common stock issued in connection with, or issuable upon conversion of, the senior convertible notes to that selling stockholder.
This prospectus is intended to satisfy our obligations under that registration rights agreement.
In addition, in
connection with the private placement of our Series B convertible preferred stock, pursuant to a registration rights agreement
dated May 21, 2013 that we entered into with certain of the selling stockholders, we granted certain customary registration rights
covering the shares of our common stock issuable upon conversion or exercise of the Series B convertible preferred stock and warrants,
respectively, that they received. This prospectus is intended to satisfy our obligations under that registration rights agreement,
as well.
Finally, the holders
of certain of the other warrants covered by this prospectus are entitled to certain registration rights with respect to the registration
of the shares issuable upon exercise of those warrants. These rights are provided under the terms of loan agreement, first executed
on March 1, 2007. This prospectus is intended to satisfy our obligations under the loan agreement, as well.
We may require
the selling stockholders to suspend the sales of the common stock covered by this prospectus if we determine in good faith that
the disclosure of any material event that has occurred and is continuing would be materially detrimental to us or our business.
Under the May 21, 2013 registration rights agreement, we will be permitted to suspend the rights of the selling stockholders to
make sales pursuant to the registration statement for periods not to exceed 30 days in any 90-day period and 60 days in any 360-day
period, provided that, in the event the disclosure relates to a previously undisclosed proposed or pending material business transaction,
we may extend a suspension period from 30 days to 45 days during any 90-day period.
The table below
sets forth:
|
|
·
|
the names of the selling stockholders;
|
|
|
·
|
the number of shares of common stock, and the percentages of outstanding
common stock, beneficially owned by the selling stockholders as of March 28, 2014 (except as otherwise indicated), prior to the
selling stockholders’ offering of the shares of common stock pursuant to this prospectus;
|
|
|
·
|
the maximum number of shares of common stock that may be offered
by the selling stockholders pursuant to this prospectus; and
|
|
|
·
|
the number of shares of common stock, and the percentages of outstanding
common stock, to be beneficially owned by the selling stockholders after the sale of the shares of common stock offered pursuant
to this prospectus, assuming all such offered shares are sold by the selling stockholders and that the selling stockholders do
not acquire any additional shares of common stock.
|
The number of shares
disclosed in the table below as ‘‘beneficially owned’’ are those beneficially owned as determined under
the rules of the SEC. Such information is not necessarily indicative of ownership for any other purpose.
We obtained the
information in the table below from the selling stockholders (other than the information regarding the percentages of outstanding
common stock beneficially owned by each selling stockholder). Except as may be noted below, none of the selling stockholders has,
or within the past three years has had, any material relationship with us or any of our affiliates.
We cannot advise
you as to whether the selling stockholders will in fact sell any or all of such shares of common stock. In addition, the selling
stockholders may have sold, transferred or otherwise disposed of, or may sell, transfer or otherwise dispose of, at any time and
from time to time, the shares in transactions exempt from the registration requirements of the Securities Act after the date on
which they provided the information set forth in the table below. Only the selling stockholders referenced in the table below may
sell the securities offered hereby, except as described under “Plan of Distribution” and otherwise permitted by law.
Changed information regarding the selling stockholders will be presented in a prospectus supplement or post-effective amendment
to the registration statement of which this prospectus forms a part if and when required. Except as may be indicated below, no
selling stockholder is a registered broker-dealer or an affiliate of a broker-dealer.
The number of shares
of common stock underlying shares of Series B convertible preferred stock and warrants assumes no adjustment in the number of shares
issuable upon conversion or exercise thereof as a result of stock splits and stock dividends, and conversion price or exercise
price adjustments pursuant to the terms of the certificate of designations governing the Series B convertible preferred stock and
the terms of certain of the warrants, respectively. The number of shares of common stock underlying Series B convertible preferred
stock includes 836,610 shares of common stock that were issued, or may be issuable, at our election and in accordance with the
terms of the Series B convertible preferred stock, as payment for dividends on the Series B convertible preferred stock, payable
through December 31, 2015.
|
Name of Selling Stockholder
|
|
Beneficial Ownership of
Common Stock Prior
to
Offering
|
|
Common
Stock
Being
Offered
Pursuant to
this
Prospectus
(maximum
number
that may be
sold) (1)
|
|
Beneficial Ownership of
Common Stock After
Offering
|
|
|
Shares
|
|
Percentage
|
|
|
Shares
|
|
Percentage
|
|
|
|
|
|
|
|
|
|
|
|
|
|
John Edwin Imhoff (2)
|
|
13,913,499
|
|
19.28%
|
|
7,626,293
|
|
6,287,206
|
|
9%
|
|
Dolores Maloof (3)
|
|
5,666,3
11
|
|
7.85%
|
|
5,666,311
|
|
-
|
|
0%
|
|
David B. Musket (4)
|
|
2,958,430
|
|
4.10%
|
|
2,958,430
|
|
-
|
|
0%
|
|
Alpha Capital Anstalt
|
|
2,842,370
|
|
3.94%
|
|
2,842,370
|
|
-
|
|
0%
|
|
Capital Ventures International (5)
|
|
1,705,421
|
|
2.36%
|
|
1,705,421
|
|
-
|
|
0
%
|
|
Hanover Holdings I, LLC (6)
|
|
3,102,432(7)
|
|
4.30%
|
|
9,163,565(8)
|
|
-
|
|
*
|
|
Ronald Wilson Hart (9)
|
|
1,882,717
|
|
2.6%
|
|
206,683
|
|
1,676,034
|
|
2.3%
|
|
Cranshire Capital Master Fund, Ltd. (10)
|
|
1,136,950
|
|
1.58%
|
|
1,136,950
|
|
-
|
|
0%
|
|
Easton Hunt Capital Partners, LP
|
|
1,061,663
|
|
1.47%
|
|
1,061,663
|
|
-
|
|
*
|
|
Ronald W. Allen
|
|
902,537
|
|
1.3%
|
|
242,535
|
|
660,002
|
|
*
|
|
ProMed Partners, L.P. (11)
|
|
874,910
|
|
1.2%
|
|
874,910
|
|
-
|
|
-
|
|
Kuekenhof Equity Fund, LP (12)
|
|
788,471
|
|
1.1%
|
|
372,134
|
|
416,337
|
|
*
|
|
David Salomon
|
|
750,000
|
|
1.0%
|
|
750,000
|
|
-
|
|
-
|
|
Lynne Imhoff (13)
|
|
725,689
|
|
1.0%
|
|
725,689
|
|
-
|
|
-
|
|
The Whittemore Collection, Ltd. (14)
|
|
568,475
|
|
*
|
|
568,475
|
|
-
|
|
-
|
|
Michael Paul Moore
|
|
547,459
|
|
*
|
|
411,957
|
|
135,502
|
|
*
|
|
Hart Management, LLC (15)
|
|
470,147
|
|
*
|
|
153,846
|
|
316,301
|
|
*
|
|
The Sternfeld Family Trust
|
|
453,986
|
|
*
|
|
363,189
|
|
90,797
|
|
*
|
|
Equitec Specialists, LLC (16)
|
|
284,240
|
|
*
|
|
284,240
|
|
-
|
|
0%
|
|
Webster Mrak & Blumberg Profit Sharing Plan, FBO Richard Blumberg
|
|
401,413
|
|
*
|
|
401,413
|
|
-
|
|
-
|
|
International Developers Group #1, LLC
|
|
322,822
|
|
*
|
|
322,822
|
|
-
|
|
-
|
|
James
E. Funderburke (3)
|
|
300,000
|
|
*
|
|
300,000
|
|
-
|
|
-
|
|
Webster Mrak & Blumberg Profit Sharing Plan, FBO Christine Mrak
|
|
292,116
|
|
*
|
|
292,116
|
|
-
|
|
-
|
|
Simon Halegoua
|
|
255,554
|
|
*
|
|
255,554
|
|
-
|
|
-
|
|
Benny H. Screws
|
|
253,160
|
|
*
|
|
189,870
|
|
63,290
|
|
*
|
|
Ressler & Tesh, PLLC
|
|
250,000
|
|
*
|
|
250,000
|
|
-
|
|
-
|
|
Sherman C. Wade
|
|
225,973
|
|
*
|
|
169,480
|
|
56,493
|
|
*
|
|
Chestnut Ridge
|
|
209,615
|
|
*
|
|
209,615
|
|
-
|
|
-
|
|
Pam Maloof
|
|
192,445
|
|
*
|
|
192,445
|
|
-
|
|
-
|
|
Judy Winstel
|
|
192,444
|
|
*
|
|
192,444
|
|
-
|
|
-
|
|
Laura M. Grunow
|
|
192,444
|
|
*
|
|
192,444
|
|
-
|
|
-
|
|
Rita Maloof
|
|
192,444
|
|
*
|
|
192,444
|
|
-
|
|
-
|
|
Steve Maloof
|
|
192,444
|
|
*
|
|
192,444
|
|
-
|
|
-
|
|
Mark Samuels
|
|
172,976
|
|
*
|
|
133,059
|
|
39,917
|
|
*
|
|
John Conway Imhoff (11)
|
|
170,000
|
|
*
|
|
50,000
|
|
120,000
|
|
*
|
|
TABAS, LLLP
|
|
163,043
|
|
*
|
|
163,043
|
|
-
|
|
-
|
|
Name of Selling Stockholder
|
|
Beneficial Ownership of
Common Stock Prior
to
Offering
|
|
Common
Stock
Being
Offered
Pursuant to
this
Prospectus
(maximum
number
that may be
sold) (1)
|
|
Beneficial Ownership of
Common Stock After
Offering
|
|
|
Shares
|
|
Percentage
|
|
|
Shares
|
|
Percentage
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Peter M. Mondalek
|
|
161,411
|
|
*
|
|
161,411
|
|
-
|
|
-
|
|
Evan Fishel
|
|
160,000
|
|
*
|
|
160,000
|
|
-
|
|
-
|
|
Guided Medical Solutions, LLC
|
|
153,848
|
|
*
|
|
153,848
|
|
-
|
|
-
|
|
Hana Smouha
|
|
153,846
|
|
*
|
|
153,846
|
|
-
|
|
-
|
|
Claude Mosseri-Marlio
|
|
153,808
|
|
*
|
|
153,808
|
|
-
|
|
-
|
|
Richard Blumberg
|
|
114,384
|
|
*
|
|
114,384
|
|
-
|
|
-
|
|
Bristol Investment Fund
|
|
112,804
|
|
*
|
|
112,804
|
|
-
|
|
-
|
|
Kensington Partners, LP
|
|
109,953
|
|
*
|
|
109,953
|
|
-
|
|
-
|
|
L. Peter Reiniger
|
|
109,449
|
|
*
|
|
109,449
|
|
-
|
|
-
|
|
Mark E. & Maureen C. Brennan JT Tenants
|
|
109,001
|
|
*
|
|
109,001
|
|
-
|
|
-
|
|
Joseph Stravato
|
|
95,000
|
|
*
|
|
95,000
|
|
-
|
|
-
|
|
Germain Halegoua Annuity Trust UTA 6/16/95 FBO Germain R. Halegoua
|
|
85,185
|
|
*
|
|
85,185
|
|
-
|
|
-
|
|
Germain Halegoua Annuity Trust UTA 6/16/95 FBO Jason Halegoua
|
|
85,185
|
|
*
|
|
85,185
|
|
-
|
|
-
|
|
Germain Halegoua Annuity Trust UTA 6/16/95 FBO Rachel E. Halegoua
|
|
85,185
|
|
*
|
|
85,185
|
|
-
|
|
-
|
|
William Zachary, Jr.
|
|
81,229
|
|
*
|
|
59,709
|
|
21,520
|
|
*
|
|
Keith D. Ignotz
|
|
76,937
|
|
*
|
|
76,937
|
|
-
|
|
-
|
|
OTAPE Investments, LLC
|
|
76,937
|
|
*
|
|
76,937
|
|
-
|
|
-
|
|
Alan M. Hoberman
|
|
76,923
|
|
*
|
|
76,923
|
|
-
|
|
-
|
|
Jeffrey Belmont
|
|
76,923
|
|
*
|
|
76,923
|
|
-
|
|
-
|
|
Richard Smouha
|
|
76,896
|
|
*
|
|
76,896
|
|
-
|
|
-
|
|
Gregory S. Petrie
|
|
72,668
|
|
*
|
|
72,668
|
|
-
|
|
-
|
|
Christopher Jordan
|
|
63,931
|
|
*
|
|
63,931
|
|
-
|
|
-
|
|
Richard Keim
|
|
49,230
|
|
*
|
|
5,000
|
|
44,230
|
|
*
|
|
Gary S. Kaplan
|
|
47,000
|
|
*
|
|
47,000
|
|
-
|
|
-
|
|
Joseph Mermelstein
|
|
42,547
|
|
*
|
|
42,547
|
|
-
|
|
-
|
|
Douglas Schmidt
|
|
38,474
|
|
*
|
|
38,474
|
|
-
|
|
-
|
|
Andrew Gluck
|
|
38,462
|
|
*
|
|
38,462
|
|
-
|
|
-
|
|
David Naggar
|
|
38,462
|
|
*
|
|
38,462
|
|
-
|
|
-
|
|
Lorianne O'Connor
|
|
38,462
|
|
*
|
|
38,462
|
|
-
|
|
-
|
|
Marshall I. Etra IRA
|
|
38,462
|
|
*
|
|
38,462
|
|
-
|
|
-
|
|
Maryse Hops
|
|
38,462
|
|
*
|
|
38,462
|
|
-
|
|
-
|
|
Rhoda Intervivos Trust
|
|
38,462
|
|
*
|
|
38,462
|
|
-
|
|
-
|
|
Robert P. Brubaker
|
|
38,462
|
|
*
|
|
38,462
|
|
-
|
|
-
|
|
William Bryce Combs
|
|
38,462
|
|
*
|
|
38,462
|
|
-
|
|
-
|
|
Richard Steiner Rev. Trust UTD 8/25/92
|
|
30,769
|
|
*
|
|
30,769
|
|
-
|
|
-
|
|
Ronald Moorhead
|
|
27,000
|
|
*
|
|
27,000
|
|
-
|
|
-
|
|
Michael Maiello
|
|
18,462
|
|
*
|
|
18,462
|
|
-
|
|
-
|
|
Name of Selling Stockholder
|
|
Beneficial Ownership of
Common Stock Prior
to
Offering
|
|
Common
Stock
Being
Offered
Pursuant to
this
Prospectus
(maximum
number
that may be
sold) (1)
|
|
Beneficial Ownership of
Common Stock After
Offering
|
|
|
Shares
|
|
Percentage
|
|
|
Shares
|
|
Percentage
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Carol C. Brubaker
|
|
15,385
|
|
*
|
|
15,385
|
|
-
|
|
-
|
|
Gloria Mosseri
|
|
15,385
|
|
*
|
|
15,385
|
|
-
|
|
-
|
|
Bald Eagle Fund, Ltd.
|
|
5,447
|
|
*
|
|
5,447
|
|
-
|
|
-
|
|
Roy Buster Reaves
|
|
4,400
|
|
*
|
|
4,400
|
|
-
|
|
-
|
|
James Conti
|
|
3,000
|
|
*
|
|
3,000
|
|
-
|
|
-
|
|
Joseph Vellino
|
|
2,500
|
|
*
|
|
2,500
|
|
-
|
|
-
|
__________
|
|
(*)
|
Denotes less than 1%.
|
|
|
(1)
|
Includes shares issued in connection with, or issuable upon conversion
of, senior convertible notes, shares issued or issuable upon conversion of Series B convertible preferred stock or issued or issuable,
at our election, as payment for dividends on the Series B convertible preferred stock, payable through December 31, 2015, or shares
issued or issuable upon exercise of warrants.
|
|
|
(2)
|
Serves on our board of directors.
|
|
|
(3)
|
As part of a dispute settlement with certain of our stockholders,
including the selling stockholder, on August 30, 2011, we entered into a release agreement pursuant to which the stockholders agreed
to cancel all rights under a disputed agreement and we agreed to issue them warrants to purchase an aggregate of 2.6 million shares
of our common stock (all of which have been exercised), to pay them a 2% royalty on gross revenues generated from the sale of LuViva
disposables (capped at $7.2 million), and to pay them up to an additional $4.8 million in connection with a non-ordinary course
asset sale or a sale of Guided Therapeutics by merger. The royalties are payable until the earlier of the sale of Guided Therapeutics
by merger and the sale or exclusive license of all or substantially all of our cervical cancer detection technology.
|
|
|
(4)
|
Mr. Musket acquired these shares in his individual capacity. Mr. Musket
is also a general partner of ProMed Partners, L.P. (“ProMed”), a selling stockholder listed in the table above, but
disclaims beneficial ownership in the shares held by ProMed except to the extent of his pecuniary interest therein. Mr. Musket
is a broker-dealer. He acquired the shares offered pursuant to this prospectus in the ordinary course of business and, at the time
of such acquisition, did not have any agreements or understandings, directly or indirectly, with any person to distribute such
securities.
|
|
|
(5)
|
Heights Capital Management, Inc., the authorized agent of Capital
Ventures International (“CVI”), has discretionary authority to vote and dispose of the shares held by CVI and may be
deemed to be the beneficial owner of these shares. Martin Kobinger, in his capacity as investment manager of Heights Capital Management,
Inc., may also be deemed to have investment discretion and voting power over the shares held by CVI. Mr. Kobinger disclaims any
such beneficial ownership of the shares. CVI is an affiliate of a broker-dealer. CVI acquired the shares offered pursuant to this
prospectus in the ordinary course of business and, at the time of such acquisition, did not have any agreements or understandings,
directly or indirectly, with any person to distribute such securities.
|
|
|
(6)
|
The business address of Hanover is c/o Magna Group, 5 Hanover Square,
New York, New York 10004. Hanover’s principal business is that of a private investment firm. We have been advised that Hanover
is not a member of the Financial Industry Regulatory Authority, or FINRA, or an independent broker-dealer, and that neither Hanover
nor any of its affiliates is an affiliate or an associated person of any FINRA member or independent broker-dealer. We have been
further advised that Joshua Sason is the Chief Executive Officer and Managing Member of Hanover and owns all of the membership
interests in Hanover, and that Mr. Sason has sole power to vote or to direct the vote and sole power to dispose or to direct the
disposition of all securities owned directly by Hanover.
|
|
|
(7)
|
The number of shares reported in this column represent the shares
of common stock that are issuable upon conversion of the senior convertible note issued to the selling stockholder on April 23,
2014, assuming a conversion in full on April 28, 2014, without regard to any limitations on conversion set forth therein, plus
an additional 321,820 shares issued to the selling stockholder in connection with the sale of the senior convertible note. The
terms of the senior convertible notes restrict the selling stockholder from converting the notes to the extent (but only to the
extent) that after giving effect to such conversion the selling stockholder (together with its affiliates) would beneficially own
in excess of 9.99% of our outstanding common stock. As of the date of this filing, Hanover beneficially owns 3,102,432 shares of
our common stock (approximately 4.3% of our outstanding common stock).
|
|
|
(8)
|
The number of shares reported in this column represents approximately
110% of the shares that would have been issuable upon conversion of a senior convertible note issued on April 23, 2014 and an additional
senior convertible note to be issued within ten business days after the effectiveness of a resale registration statement (subject
only to conditions outside of the note holder’s control or that the note holder cannot cause not to be satisfied, none of
which are related to the market price of our common stock), had the notes been converted in full on April 28, 2014 without regard
to any limitations on conversion set forth therein, plus an additional 321,820 shares issued to the selling stockholder in connection
with the sale of the initial senior convertible note. Because the conversion price of the senior convertible notes may be adjusted,
the number of shares that will actually be issued may be more or less than the number of shares being offered by this prospectus.
|
|
|
(9)
|
Serves on our board of directors.
|
|
|
(10)
|
Cranshire Capital Advisors, LLC (“CCA”) is the investment
manager of Cranshire Capital Master Fund, Ltd. (“Cranshire Master Fund”) and has voting and investment discretion over
securities held by Cranshire Master Fund. Mitchell P. Kopin, the president, the sole member and the sole member of the Board of
Managers of CCA, has voting control over CCA. As a result, each of Mr. Kopin and CCA may be deemed to have beneficial ownership
(as determined under Section 13(d) of the Exchange Act of 1934, as amended, or the Exchange Act) of the securities held by Cranshire
Master Fund. CCA is also the investment manager for managed accounts for Equitec Specialists, LLC (“Equitec”) and CCA
has voting control and investment discretion over securities held in the managed accounts for Equitec. As a result, each of Mr.
Kopin and CCA also may be deemed to have beneficial ownership of the securities held in the managed accounts by Equitec. The shares
reported in the table above as beneficially owned by Cranshire Master Fund and Equitec reflect only those shares beneficially owned
by each entity, respectively. Including the shares beneficially owned by each of Cranshire Master Fund and Equitec, these entities
would together beneficially own 818,064 shares of common stock, or 1.24% of the total number of shares of our common stock issued
and outstanding, all of which may be offered pursuant to this prospectus.
|
|
|
(11)
|
See
footnote 4, above. ProMed is an affiliate of a broker-dealer. ProMed acquired
the shares offered pursuant to this prospectus in the ordinary course of business and,
at the time of such acquisition, did not have any agreements or understandings, directly
or indirectly, with any person to distribute such securities.
|
|
|
(12)
|
Shares held of record by Kuekenhof Equity Fund, LP. Michael James,
one of our directors, is a managing partner of Kuekenhof Equity Fund, LP.
|
|
|
(13)
|
Family member of one of our directors.
|
|
|
(14)
|
Parsons & Whittemore Enterprises Corp., a Delaware corporation
(“PWE”), is the sole shareholder of The Whittemore Collection, Ltd., a New York corporation (“TWC”), and,
in such capacity, may be deemed to have beneficial ownership over the shares held by TWC. George F. Landegger is the Chairman and
President of TWC and owns the majority of voting shares of PWE, and, in such capacities, may be deemed to have beneficial ownership
over the shares deemed beneficially owned by PWE and TWC.
|
|
|
(15)
|
Owned or managed by a member of our board of directors.
|
|
|
(16)
|
See footnote 10, above. Equitec is an affiliate of a broker-dealer.
Equitec acquired the shares offered pursuant to this prospectus in the ordinary course of business and, at the time of such acquisition,
did not have any agreements or understandings, directly or indirectly, with any person to distribute such securities.
|
PLAN OF DISTRIBUTION
We are registering
the shares covered by this prospectus on behalf of the selling stockholders. All costs, expenses and fees connected with the registration
of these shares will be borne by us. Any brokerage commissions and similar expenses connected with selling the shares will be borne
by the selling stockholders. The selling stockholders may offer and sell the shares covered by this prospectus from time to time
in one or more transactions. The term “selling stockholders” includes pledgees, donees, transferees and other successors-in-interest
who may acquire shares through a pledge, gift, partnership distribution or other non-sale related transfer from the selling stockholders.
The selling stockholders will act independently of the Company in making decisions with respect to the timing, manner and size
of each sale and they may sell the shares on one or more exchanges, in the over-the-counter market or in privately negotiated transactions
at prevailing market prices at the time of sale, at fixed prices, at varying prices determined at the time of the sale or at negotiated
prices. These transactions include:
|
|
·
|
ordinary brokerage transactions and transactions in which the broker
solicits purchasers;
|
|
|
·
|
purchases by a broker-dealer as principal and resale by the broker-dealer
for its own account pursuant to this prospectus;
|
|
|
·
|
exchange or over-the-counter distributions in accordance with the
rules of the exchange or other market;
|
|
|
·
|
block trades in which the broker-dealer attempts to sell the shares
as agent but may position and resell a portion of the block as principal to facilitate the transaction;
|
|
|
·
|
a combination of any such method of sale; and
|
|
|
·
|
any other method permitted pursuant to applicable law.
|
In
connection with distributions of the shares or otherwise, the selling stockholders may:
|
|
·
|
sell the shares short and redeliver the shares to close out short
positions;
|
|
|
·
|
enter into option or other transactions with broker-dealers or other
financial institutions which require the delivery to them of the shares covered by this prospectus, which they may in turn resell;
and
|
|
|
·
|
pledge the shares to broker-dealers or other financial institutions,
which, upon a default, they may in turn resell.
|
The selling
stockholders may also sell any of the shares under Rule 144 rather than with this prospectus if the sale meets the requirements
of that rule.
In effecting sales,
the selling stockholders may engage broker-dealers or agents, who may in turn arrange for other broker-dealers to participate.
Broker-dealers or agents may receive commissions, discounts or concessions from the selling stockholders and/or from the purchasers
of the shares for whom the broker-dealers may act as agents or to whom they sell as principal, or both. The compensation to a particular
broker-dealer may be in excess of customary commissions. To our knowledge, there is currently no plan, arrangement or understanding
between any selling stockholders and any broker-dealer or agent regarding the sale of any of the shares by the selling stockholders.
The selling stockholders,
any broker-dealers or agents and any participating broker-dealers that act in connection with the sale of the shares covered by
this prospectus may be “underwriters” under the Securities Act with respect to those shares and will be subject to
the prospectus delivery requirements of the Securities Act. Any profit that the selling stockholders realize, and any compensation
that any broker-dealer or agent may receive in connection with any sale, including any profit realized on resale of the shares
acquired as principal, may constitute underwriting discounts and commissions. If the selling stockholders are deemed to be underwriters,
the selling stockholders may be subject to certain liabilities under statutes including, but not limited to, Section 11, 12 and
17 of the Securities Act and Section 10(b) and Rule 10b-5 under the Exchange Act.
The securities
laws of some states may require the selling stockholders to sell the shares in those states only through registered or licensed
brokers or dealers. These laws may also require that we register or qualify the shares for sale in those states unless an exemption
from registration and qualification is available and the selling stockholders and we comply with that exemption. In addition, the
anti-manipulation rules of Regulation M under the Exchange Act may apply to sales of the shares in the market and to the activities
of the selling stockholders and their affiliates. Regulation M may restrict the ability of any person engaged in the distribution
of the shares to engage in market-making activities with respect to the shares. All of the foregoing may affect the marketability
of the shares and the ability of any person to engage in market-making activities with respect to the shares.
If any selling
stockholder notifies us that he has entered into any material arrangement with a broker-dealer for the sale of the shares through
a block trade, special offering, exchange distribution, over-the-counter distribution or secondary distribution, or a purchase
by a broker or dealer, we will file any necessary supplement to this prospectus to disclose:
|
|
·
|
the number of shares involved in the arrangement;
|
|
|
·
|
the terms of the arrangement, including the names of any underwriters,
dealers or agents who purchase the shares, as required;
|
|
|
·
|
the proposed selling price to the public;
|
|
|
·
|
any discount, commission or other underwriting compensation;
|
|
|
·
|
the place and time of delivery for the shares being sold;
|
|
|
·
|
any discount, commission or concession allowed, reallowed or paid
to any dealers; and
|
|
|
·
|
any other material terms of the distribution of the shares.
|
In addition, if
the selling stockholder notifies us that a donee, pledgee, transferee or other successor-in-interest of the selling stockholder
intends to sell more than 500 shares, we will file a supplement to this prospectus.
DESCRIPTION OF SECURITIES
We are authorized
to issue 150 million shares of stock, in two classes: 145 million shares of common stock, par value $.001 per share, and 5 million
shares of preferred stock, including 3,000 shares of Series B convertible preferred stock, par value $.001 per share. As of April
24, 2014, there were 72,172,331 shares of common stock outstanding, which were held of record by 359 stockholders, and 1,737 shares
of preferred stock outstanding, consisting entirely of shares of Series B convertible preferred stock, which were held of record
by 8 stockholders.
Common Stock
The holders of
common stock are entitled to one vote for each share held of record on all matters submitted to a vote of stockholders. Subject
to preferences that may be applicable to any outstanding preferred stock, holders of common stock are entitled to receive ratably
such dividends as may be declared by the board out of funds legally available therefor and in liquidation proceedings. Holders
of common stock have no preemptive or subscription rights and there are no redemption rights with respect to such shares.
Preferred Stock
Our board is authorized,
without further stockholder action, to issue preferred stock in one or more series and to fix the voting rights, liquidation preferences,
dividend rights, repurchase rights, conversion rights, redemption rights and terms, including sinking fund provisions, and certain
other rights and preferences, of the preferred stock.
Although there
is no current intention to do so, our board may, without stockholder approval, issue additional shares of series B convertible
preferred stock or shares of another class or series of preferred stock with voting and conversion rights that could adversely
affect the voting power or dividend rights of the holders of common stock and may have the effect of delaying, deferring or preventing
a change in control.
Series B Convertible Preferred Stock
On May 23, 2013,
we issued and sold 2,527 shares of Series B convertible preferred stock at a price per share of $1,000, which, subject to adjustment
for stock splits, stock dividends or other similar occurrences, we refer to in this prospectus as the invested amount.
Dividends
.
The holders of Series B convertible preferred stock are entitled to receive quarterly, at the end of each calendar quarter, out
of funds legally available therefor, dividends per share at the per annum rate of ten percent of the invested amount, prior and
in preference to any declaration or payment of any dividend on any stock ranking junior to the Series B convertible preferred stock.
Such dividends are cumulative and are compounded annually, and accrue whether or not declared by our board of directors. At our
election, dividends on the Series B convertible preferred stock may be paid by the issuance and delivery of whole shares of common
stock having an aggregate current market price at the time of issuance equal to the amount of dividends so paid, as long as such
shares of common stock are registered for resale under an effective registration statement or such shares are then eligible to
be sold without restriction under Rule 144 of the Securities Act. The shares of any class of our capital stock ranking equal to
the Series B convertible preferred stock as to dividends and the distribution of assets upon liquidation are referred to in
this prospectus as
pari passu
stock. If any dividend becomes due and payable to the holders of Series B convertible preferred
stock and there is also due and payable a dividend to the holders of
pari passu
stock, and we have insufficient funds to
make payment in full to all such holders of such respective dividends, then such funds as are available will be distributed among
the holders, ratably in proportion to the full amounts to which they would otherwise respectively be entitled.
Conversion
.
Each share of Series B convertible preferred stock is convertible into the number of shares of common stock equal to the quotient
obtained by dividing (i) the sum of the invested amount plus all declared or accrued but unpaid dividends on such shares of Series
B convertible preferred stock, by (ii) the conversion price per share. The current per share conversion price is $0.40. The conversion
price is subject to adjustment under certain circumstances to protect the holders of Series B convertible preferred stock from
dilution relative to certain issuances of common stock, or securities convertible into or exercisable for shares of common stock.
Subject to certain exceptions, if we issue shares of common stock, or such other securities, at a price per share less than the
then-effective conversion price, the conversion price will be adjusted to equal such lower per share consideration.
The Series B convertible
preferred stock is convertible at any time, at the option of the holder. In addition, on any “automatic conversion date,”
each share of Series B convertible preferred stock then outstanding automatically will be converted into common stock at the then
effective conversion rate. An automatic conversion date, subject to certain additional limitations and requirements, will occur
upon the earlier of (a) the date that is the 30th day after the later of our receipt of an approvable letter from the FDA for LuViva
and the date on which the common stock achieves an average closing price for 20 consecutive trading days of at least $0.98 with
an average daily trading volume during such 20 consecutive trading days of at least 25,000 shares, (b) the date on which the common
stock achieves an average closing price for 20 consecutive trading days of at least $1.16 with an average daily trading volume
during such 20 consecutive trading days of at least 25,000 shares, or (c) the date after May 23, 2015 on which the common stock
achieves an average closing price for 20 consecutive trading days of at least $0.82 with an average daily trading volume during
such 20 consecutive trading days of at least 25,000 shares.
Voting
.
Each holder of a share of Series B convertible preferred stock is entitled to the number of votes equal to the number of shares
of common stock into which such share of Series B convertible preferred stock would be convertible under the circumstances described
above on the record date for the vote or consent of stockholders, and will otherwise have voting rights and powers equal to the
voting rights and powers of the common stock.
Holders of the
Series B convertible preferred stock have the right to vote on those matters which, under the General Corporation Law of the State
of Delaware, voting by classes of stock is required and, so long as at least 917 shares (such number subject to adjustment) of
Series B convertible preferred stock are outstanding, we may not, without the consent (given by vote in person or by proxy at a
meeting called for the purpose, or by written consent) of the holders of a majority of the shares of Series B convertible preferred
stock then outstanding:
-
create or authorize any shares of any class or series of capital stock
having a preference or priority as to either dividends or distribution of assets upon liquidation equal or superior to any such
preference or priority of the shares of Series B convertible preferred stock, reclassify any existing securities into shares of
such equal or superior stock or amend the terms of any existing securities in a manner inconsistent with the foregoing restriction;
-
amend or repeal any provision of, or add any provision to, our certificate
of incorporation or bylaws, if such action would adversely alter or change the preferences, rights, privileges, or powers of, or
restrictions provided for the benefit of, the Series B convertible preferred stock;
-
declare, pay or set aside any dividends on any stock ranking junior
to the Series B convertible preferred stock, or redeem or repurchase any such junior ranking stock;
-
increase or decrease (other than in connection with a redemption or
conversion) the authorized number of shares of Series B convertible preferred stock; or
-
alter or change the rights, preferences or privileges of the Series
B convertible preferred stock in a manner different from each other class of
pari passu
stock.
Further, and in
addition to the approval rights described above, we may not, without the consent of the holders of all of the shares of Series
B convertible preferred stock then outstanding, adversely amend or repeal any provision of, or add any provision to, the preferences,
rights, privileges or powers of the Series B convertible preferred stock, in respect of:
|
|
·
|
the amount of dividends, or the timing of the required payment thereof;
|
|
|
·
|
the liquidation amount, or the timing of the required payment thereof;
|
|
|
·
|
the automatic conversion date; or
|
|
|
·
|
the conversion rights, including the conversion price.
|
In addition, prior
to the date that is the 30th day after the later of our receipt of an approvable letter from the FDA for LuViva and the date on
which the common stock achieves an average closing price for 20 consecutive trading days of at least $0.98 with an average daily
trading volume during such 20 consecutive trading days of at least 25,000 shares, we may not, without the consent of the holders
of 66 2/3% of the shares of Series B convertible preferred stock then outstanding, incur or cause any of our subsidiaries to incur
indebtedness for borrowed money, or guarantee indebtedness for borrowed money, that is (i) secured by our intellectual property;
or (ii) in excess of $2,000,000.
Redemption
.
Subject to certain conditions, we have the right to redeem, to the fullest extent permitted by law, all or any portion of the outstanding
Series B convertible preferred stock at the then-current redemption price, at any time after May 23, 2015. The redemption price
per share of Series B convertible preferred stock will be equal to the liquidation amount, including unpaid dividends up to and
including the date of redemption.
Liquidation
.
In the event of our voluntary or involuntary liquidation, dissolution or winding up, referred to in this prospectus as a liquidation,
or a “sale or merger” (as described below), the holders of the outstanding shares of Series B convertible preferred
stock, at their election, will be entitled to receive in exchange for and in redemption of each share of their Series B convertible
preferred stock, prior and in preference to the holders of stock ranking junior to the Series B convertible preferred stock, (x)
in the case of a liquidation, from any funds legally available for distribution to stockholders, and (y) in the case of a sale
or merger, from the net proceeds therefrom, an amount equal to the greater of (i) the invested amount per share, plus the aggregate
amount of all declared or accrued, but unpaid, dividends per share, or (ii) the amounts to which such holders would have been entitled
if the shares were converted to shares of common stock immediately before the liquidation, or sale or merger as the case may be.
For purpose of
the Series B convertible preferred stock, a “sale or merger” includes, subject to exclusion by the vote of holders
of Series B convertible preferred stock constituting at least 66 2/3% of the total number of shares of such series outstanding,
voting separately as a class, (a) our merger, reorganization, or consolidation into or with another corporation in which our stockholders
immediately preceding such transaction own less than 50% of the voting securities of the surviving corporation, or (b) the sale,
transfer, or lease (other than a transfer or lease by pledge or mortgage to a
bona fide
lender) of all or substantially
all of our assets to any entity 50% or more of the voting securities of which are not beneficially owned by the beneficial owners
of our voting securities prior to such transaction.
Senior Convertible Notes
On
April 23, 2014, we entered into a securities purchase agreement with Hanover. Pursuant to the purchase agreement, we
sold Hanover a 6% senior convertible note with a principal amount of $1.5 million, for a purchase price of $1.0 million (an
approximately 33.3% original issue discount). Additionally, Hanover is irrevocably bound to purchase, on the tenth trading
day after the effective date of a resale registration statement, an additional 6% senior convertible note with a principal
amount of $2.0 million, for a fixed purchase price of $2.0 million, subject only to conditions outside of Hanover’s
control or that Hanover cannot cause not to be satisfied, none of which are related to the market price of our common
stock.
With
respect to the initial senior convertible note, $200,000 of the outstanding principal amount (together with any accrued and
unpaid interest with respect to such portion of the principal amount) will be automatically extinguished (without any cash
payment by us) if (1) we have properly filed a registration statement with the SEC on or prior to May 23, 2014 covering the
resale by Hanover of shares of our common stock issued or issuable upon conversion of the senior convertible notes and (2) no
event of default, or an event that with the passage of time or giving of notice would constitute an event of default, has
occurred on or prior to such date. Moreover, $300,000 of the outstanding principal amount of the initial senior convertible
note (together with any accrued and unpaid interest with respect to such portion of the principal amount) will be
automatically extinguished (without any cash payment by us) if (1) the resale registration statement is declared effective by
the SEC on or prior to July 22, 2014 and the prospectus contained therein is available for use by Hanover for its resale of
the shares of common stock issued or issuable upon conversion of the senior convertible notes and (2) no event of default, or
an event that with the passage of time or giving of notice would constitute an event of default, has occurred on or prior to
such date.
The
initial senior convertible note matures on October 23, 2015 (subject to extension as provided in the Initial Convertible Note)
and, in addition to the approximately 33.3% original issue discount, accrues interest at an annual rate of 6.0%. If issued, the
additional senior convertible note will mature 18 months from the date of issuance and also will accrue interest at an annual rate
of 6.0%. Subject to certain limitations, the senior convertible notes are convertible at any time after the earlier of October
23, 2014 or the effectiveness of a resale registration statement, in whole or in part, at Hanover’s option, into shares of
our common stock, at a conversion price equal to the lesser of $0.55 per share and a discount from the lowest daily volume-weighted
average price of our common stock in the five trading days prior to conversion. The discount is 20% if the conversion takes place
prior to December 19, 2014, and 25% if after that date. At no time will Hanover be entitled to convert any portion of the
senior convertible notes to the extent that after such conversion, Hanover (together with its affiliates) would beneficially
own more than 9.99% of the outstanding shares of our common stock as of such date.
The
initial senior convertible note includes and, if issued, the additional senior convertible note will include, customary event
of default provisions. Each provides or if issued will provide, as applicable, a default interest rate of 16%. Upon the
occurrence of an event of default, Hanover may require us to pay in cash the “Event of Default Redemption Price”
which is defined in the senior convertible notes to mean the greater of (i) the product of (A) the amount to be redeemed
multiplied by (B) 135% (or 100% if an insolvency related event of default) and (ii) the product of (X) the conversion price
in effect at that time multiplied by (Y) the product of (1) 135% (or 100% if an insolvency related event of default)
multiplied by (2) the greatest closing sale price of the common stock on any trading day during the period commencing on
the date immediately preceding such event of default and ending on the date we make the entire payment required to be made
under this provision.
We
have the right at any time to redeem all or a portion of the total outstanding amount then remaining under the senior convertible
notes in cash at a 25% premium.
We
paid Hanover a commitment fee for entering into the purchase agreement in the form of 321,820 shares of common stock. We
also agreed to pay $50,000 of reasonable attorneys’ fees and expenses incurred by Hanover in connection with the
transaction.
Warrants and Options
We have issued warrants
to purchase our common stock from time to time in connection with certain financing arrangements. Currently, there are warrants
exercisable for an aggregate of 11,998,822 shares of common stock outstanding, as follows:
|
Warrants
(Underlying Shares)
|
|
|
Exercise
Price
|
|
Expiration
Date
|
|
|
471,856
|
(1)
|
|
$0.80 per share
|
|
July 26, 2014
|
|
|
3,590,522
|
(1)
|
|
$0.80 per share
|
|
March 1, 2015
|
|
|
6,790
|
(2)
|
|
$1.01 per share
|
|
September 10, 2015
|
|
|
439,883
|
(3)
|
|
$0.68 per share
|
|
March 31, 2016
|
|
|
285,186
|
(4)
|
|
$1.05 per share
|
|
November 20, 2016
|
|
|
1,858,089
|
(5)
|
|
$1.08 per share
|
|
May 23, 2018
|
|
|
5,016,840
|
(5)(6)
|
|
$0.40 per share
|
|
May 23, 2018
|
|
|
300,000
|
(7)
|
|
$0.65 per share
|
|
April 30, 2019
|
|
|
|
|
|
|
|
|
|
|
__________
|
|
(1)
|
Consists of outstanding warrants issued in conjunction with a June 2012 warrant exchange program.
|
|
(2)
|
Consists of outstanding warrants issued in conjunction with a September 2010 private placement.
|
|
(3)
|
Consists of outstanding warrants issued in conjunction with a buy-back of a minority interest in Interscan in December 2012, which were issued in February 2014. The sale of the shares underlying these warrants is not covered by this prospectus.
|
|
(4)
|
Consists of outstanding warrants issued in conjunction with a November 2011 private placement.
|
|
(5)
|
Consists of outstanding warrants issued in conjunction with a May 2013 private placement.
|
|
(6)
|
Underlying shares increased from 1,858,089 to 5,016,840, and per share exercise price decreased from $1.08 to $0.40, pursuant to the anti-dilution provisions in the warrants, as a result of a November 2013 warrant exchange program.
|
|
(7)
|
Consists of warrants issued in conjunction with the April 2014 private placement.
|
|
|
|
|
|
|
|
|
|
All outstanding
warrant agreements provide for anti-dilution adjustments in the event of certain mergers, consolidations, reorganizations, recapitalizations,
stock dividends, stock splits or other changes in our corporate structure.
The warrants identified
in the table above as issued in conjunction with a May 2013 private placement and having an exercise price of $0.40 per share
are subject to a mandatory exercise provision. This provision permits us, subject to certain limitations, to require exercise
of such warrants at any time following (a) the date that is the 30th day after the later of our receipt of an approvable letter
from the FDA for LuViva and the date on which the common stock achieves an average market price for 20 consecutive trading days
of at least $1.30 with an average daily trading volume during such 20 consecutive trading days of at least 25,000 shares, or (b)
the date on which the average market price of the common stock for 20 consecutive trading days immediately prior to the date we
deliver a notice demanding exercise is at least $1.62 and the average daily trading volume of the common stock exceeds 25,000
shares for such 20 consecutive trading days. If these warrants arenot timely exercised upon demand, they will expire. Upon the
occurrence of certain events, we also may be required to repurchase these warrants, as well as the other warrants issued in conjunction
with the May 2013 private placement.
As of April 24
2014, we have issued options to purchase a total of 8,420,703 shares of our common stock pursuant to various equity incentive plans,
at a weighted average exercise price of $0.63 per share. Recommendations for option grants under our equity incentive plans are
made by the compensation committee of our board, subject to ratification by the full board. The compensation committee may issue
options with varying vesting schedules, but all options granted pursuant to our equity incentive plans must be exercised within
ten years from the date of grant.
OUR BUSINESS
Overview
We are a medical technology
company focused on developing innovative medical devices that have the potential to improve healthcare. Our primary focus is the
development of our LuViva™ non-invasive cervical cancer detection device and extension of our cancer detection technology
into other cancers, including lung and esophageal. Our technology, including products in research and development, primarily relates
to biophotonics technology for the non-invasive detection of cancers.
We are a Delaware corporation,
originally incorporated in 1992 under the name “SpectRx, Inc.,” and, on February 22, 2008, changed our name to Guided
Therapeutics, Inc. At the same time, we renamed our wholly owned subsidiary, InterScan, which originally had been incorporated
as “Guided Therapeutics.”
Non-Invasive Cervical Cancer Detection
We believe LuViva will
provide a less invasive and painless alternative to conventional tests for cervical cancer detection. We also believe LuViva can
improve patient well-being and reduce healthcare costs, since it reduces or eliminates pain, is convenient to use and provides
rapid results at the point-of-care. We completed enrollment in our FDA pivotal trial of LuViva in 2008 and on November 18, 2010,
the FDA accepted our completed PMA application, effective September 23, 2010, for substantive review. On March 7, 2011, we announced
that the FDA had inspected two clinical trial sites as part of its review process and raised no formal compliance issues. On January
12, 2012, we announced our intent to seek an independent panel review of our PMA application after receiving a “not-approvable”
letter from the FDA. On November 14, 2012 we filed an amended PMA with FDA. On September 6, 2013 we received a letter from the
FDA with additional questions, and we are scheduled to meet with the FDA on May 8, 2014 to discuss our response. . The FDA has
granted us a 180-day extension on the PMA filing to allow time for the meeting to occur. Assuming we can respond to FDA’s
most recent questions effectively and in a timely manner, and then receive FDA approval in 2014, we currently anticipate a 2015
product launch in the United States, but cannot be assured we will be able to launch on that timetable, or at all. Internationally,
we have regulatory approval to sell LuViva in Europe upon receipt of our Edition 3CE Mark in January 2014. LuViva has marketing
approval from Health Canada and the Singapore Health Sciences Authority, and we have applied for approval in Mexico.
Other Cancers
We believe our non-invasive
cervical cancer detection technology can be applied to other cancers as well. To that end, from 2008 until early 2013 we had worked
exclusively with Konica Minolta Opto, Inc., a subsidiary of Konica Minolta, Inc., a Japanese corporation based in Tokyo (“Konica
Minolta”), to adapt our cervical cancer detection technology primarily for the detection of esophageal cancer. On February
6, 2013, we announced that we had terminated and replaced our existing agreements with Konica Minolta with a new license agreement
allowing us to manufacture and to develop a non-invasive esophageal cancer detection product from Konica Minolta and based on our
biophotonic technology platform (see “—Lung and Esophageal Cancer Detection—Konica Minolta”).
Our Business Strategy
Our mission is to build
a profitable business that develops and commercializes medical products that improve people’s lives and increases stockholder
value. To achieve this mission, we have completed the FDA pivotal trial for our first product, called LuViva, filed our PMA application
with the FDA, and have raised capital for the development and launch of the LuViva device system. Development of our cancer diagnostic
technology has been financed to date through a combination of government grants, strategic partners and direct investment. Bringing
LuViva to market is the main focus of our business. In order to adequately finance the completion of the FDA review process, complete
product development, and prepare for marketing of LuViva, additional capital will be needed; however, we cannot be assured of the
availability of adequate capital (see “Risk Factors”).
We believe that our technology,
as developed for cervical cancer detection, can be modified and then applied to other cancers. Because development of our technology
for additional cancers is costly and resource intensive, we sought a strategic partner to help defray costs and otherwise assist
in the expansion of our cancer detection technology into other cancers. This resulted in our various collaborative agreements with
Konica Minolta, including past agreements related to the development of a prototype device specifically for esophageal cancer detection
and our current license agreement with Konica Minolta (see “—Lung and Esophageal Cancer Detection—Konica Minolta”).
Industry Overview
Cervical Cancer Detection
Background
According to the American
Cancer Society, cancer is a group of many related diseases. All forms of cancer involve the out-of-control growth and spread of
abnormal cells. Normal body cells grow, divide, and die in an orderly fashion. Cancer cells, however, continue to grow and divide
and can spread to other parts of the body. In America, half of all men and one-third of all women will develop cancer during their
lifetimes. According to the American Cancer Society, the sooner a cancer is found and treatment begins, the better a patient’s
chances are of being cured. We began investigating the applications of our technologies to cancer detection before 1997, when we
initiated a market analysis for these uses. We concluded that our biophotonic technologies had applications for the detection of
a variety of cancers through the exposure of tissue to light. We selected cervical cancer and skin cancer from a list of the ten
most attractive applications as categories of cancer to pursue initially, and currently are focused primarily on the development
of our non-invasive cervical cancer detection product.
Cervical Cancer
Cervical cancer is a cancer
that begins in the lining of the cervix (which is located in the lower part of the uterus). Cervical cancer forms over time and
may spread to other parts of the body if left untreated. There is generally a gradual change from a normal cervix to a cervix with
precancerous cells to cervical cancer. For some women, precancerous changes may go away without any treatment. While the majority
of precancerous changes in the cervix do not advance to cancer, if precancers are treated, the risk that they will become cancers
can be greatly reduced. The Pap smear screening test, or Pap test, which involves a sample of cervical tissue being placed on a
slide and observed in a laboratory, is currently the most common form of cervical cancer screening.
Cervical Cancer Market
The National Cancer Institute
(“NCI”) estimated that in 2013, about 12,340 cases of invasive cervical cancer would be diagnosed and about 4,030 women
would die from cervical cancer in the United States. According to published data, cervical cancer results in about 200,000 deaths
annually worldwide, with 470,000 new cases reported each year.
We believe that our major
market opportunities related to cervical cancer are in diagnosis and screening. Since the introduction of better screening and
diagnostic methods, the number of cervical cancer deaths in the United States has declined dramatically, due mainly to the increased
use of the Pap test. However, over the last five years, the incidences have been increasing. Moreover, the Pap test has a wide
variation in sensitivity, which is the ability to detect the disease, and specificity, which is the ability to exclude false positives.
A study by Duke University for the U.S. Agency for Health Care Policy and Research published in 1999 showed Pap test performance
ranging from a sensitivity of 22% and specificity of 78% to sensitivity of 95% and specificity of 10%. About 60 million Pap tests
are given annually in the United States. The average price of a Pap test in the United States is about $26. New technologies improving
the sensitivity and specificity of the Pap test have recently been introduced and are finding acceptance in the marketplace.
After screening for cervical
cancer by use of a Pap test, if necessary, a visual examination of the cervix using a colposcope is usually followed by a biopsy,
or tissue sampling at one or more locations. This method looks for visual changes attributable to cancer. There are about two million
colposcope examinations annually in the United States and Europe. In 2003, the average cost of a stand-alone colposcope examination
in the United States was $185 and the average cost of a colposcopy with biopsy was $277.
In 2006, a new vaccine
for certain strains of the human papilloma virus, or HPV, was approved by the FDA. Most cervical cancers are associated with certain
strains of HPV. The vaccine is administered in three doses, and according to guidelines, preferably to girls before they become
sexually active. The approved vaccine is effective against 70% of the strains of HPV thought to be responsible for cervical cancer.
Due to the limited availability and lack of 100% protection against all potentially cancer-causing strains of HPV, we believe that
the vaccine will have a limited impact on the cervical cancer screening and diagnostic market for many years.
Our Non-invasive Cervical
Cancer Product
LuViva is a non-invasive
cervical cancer detection product, based on our proprietary biophotonic technology. The device is designed to identify cervical
cancers and precancers painlessly, non-invasively and at the point-of-care by scanning the cervix with light, then analyzing the
light reflected or emanating from the cervix. The information presented by the light would be used to indicate the likelihood of
cervical cancer or precancers and/or to produce a map or image of diseased tissue. This test, unlike the Pap test or biopsy, has
the potential to preserve the perspective and positional information of disease on the cervix, allowing for more accurate diagnosis.
Our system also could allow doctors to make intelligent choices in triaging patients for biopsy or treatment and potentially for
selecting biopsy sites that could be expanded for use in assisting in the detection of cancerous margins for cancer removal. Our
product, in addition to detecting the structural changes attributed to cervical cancer, is also designed to detect the biochemical
changes that precede the development of visual lesions. In this way, cervical cancer may be detected earlier in its development,
which should increase the chances of effective treatment. The product is expected to incorporate a single-use, disposable calibration
and alignment component. FDA approval of the intended use of our device is required and initial approval may be for a limited set
of the above potential capabilities. Our strategy is to continue our launch of LuViva in Canada and Turkey, which we began in the
third quarter of 2013, while also continuing the launch in certain developed countries of Europe, which began in the last quarter
of 2013. In parallel with these international efforts we are continuing steps to procure FDA approval in the United States.
To date, more than 4,000
women have been tested with various LuViva prototype and commercial devices in multiple clinical settings. During 2000, we conducted
human clinical feasibility studies of laboratory prototypes at two U.S. research centers, detecting 31% more cervical precancerous
lesions than conventional Pap tests. The results were presented at the World Health Organization/European Research Organization
on Genital Infection and Neoplasia Joint Experts Conference in Paris in April 2000. The study population included 133 women scheduled
for colposcopy and biopsy, if indicated. A total of 318 tissue-specific comparisons were made between our device and colposcopy/biopsy
results. Of the 318 patients included in this study, 20 had high-grade precancers, 36 had low-grade precancers, 146 had benign
lesions and 116 had normal tissues. Compared to the Pap test, our product detected 31% more precancers and 25% more high-grade
precancers without increasing the false positive rate.
In 2005, we continued
to conduct our pivotal clinical trial, which had collected data on over 900 women by the end of the year. In 2005, we also completed
work on our commercial prototype. In 2006 and 2007, we continued to enroll subjects in our pivotal clinical trial and, by the end
of 2007, had enrolled 1,400 subjects.
In September 2006, we
announced that the National Cancer Institute (“NCI”) awarded a grant of approximately $690,000 for development of our
non-invasive cervical cancer detection technology. This grant was used to further the ongoing FDA pivotal clinical trial. In 2006
and 2007, we received approximately $523,000 and $398,000, respectively, of NCI grant funds. On October 5, 2009, we were awarded
a $2.5 million matching grant by the NCI to bring to market and expand the array features for LuViva. The award provided resources
to complete the regulatory process and begin manufacturing ramp up for LuViva and a single-patient-use disposable patient interface
for the device and will be received over a period of three years. Under the award, we recorded revenue of approximately $150,000
in 2013, $68,000 in 2012 and $912,000 in 2011.
We completed enrollment
in our FDA pivotal trial in 2008 and collection of FDA-recommended follow up data in the third quarter of 2010. On November 18,
2010, the FDA accepted our completed PMA application, effective September 23, 2010, for substantive review. On March 7, 2011, we
announced that the FDA had inspected two clinical trial sites as part of its review process and raised no formal compliance issues.
On January 12, 2012, we announced our intent to seek an independent panel review of our PMA application after receiving a “not-approvable”
letter from the FDA. Assuming we receive FDA approval in 2014, we currently anticipate a 2015 U.S. product launch, but cannot be
assured we will be able to launch on that timetable, or at all.
Internationally, on October
4, 2011, we announced that LuViva was selected for inclusion in a review of new technologies by the United Kingdom’s NICE
program. On January 10, 2014, we announced that we had successfully completed an audit of our quality system and were recertified
under ISO 13485:2003. As a result, we now have regulatory approval to sell LuViva in Europe upon receipt of our Edition 3CE Mark
in January 2014. LuViva has marketing approval from Health Canada and the Singapore Health Sciences Authority, and we have applied
for approval in Mexico.
Sales or leases of LuViva
are expected to include a single-patient-use disposable patient interface. We expect the device itself to be priced at approximately
$20,000, with the disposable interface priced around $30 to $40. Profit margins on the disposable are expected to be approximately
90%. In the United States, we plan on establishing and training a 10-person sales force during the first year after launch, which
will initially focus on early adopters in the larger population centers. Internationally, we plan on contracting with country-specific
or regional distributors. We believe that the international market will be larger than the U.S. market. We have been in contact
with more than 100 potential distributors, have formal distribution agreements in place covering 21 countries and expect to announce
additional agreements over the next several months.
The market for cervical
cancer screening is currently dominated by lab-based cytological screening of samples obtained from patients. The market for primary
screening is dominated by Hologic, Inc., which markets the Thin Prep Pap test and Qiagen, Inc., which markets another method of
cervical cancer screening, HPV detection. Qiagen is attempting to gain permission to use its device for primary screening.
The Qiagen HPV test is already approved for use as a follow-up to ambiguous Pap test results and as an adjunct to the Pap test
for screening women aged 30 and over. We have conducted marketing research related to the cervical cancer market and the impact
of the growth of the lab-based cytological screening products. We are reviewing the impact of the changing competitive landscape
related to our product development pace and our initial and potential positioning. We will have to demonstrate clinical and commercial
effectiveness to be able to change current medical practice behavior and capture market share and cannot be sure that we will be
able to do so.
Lung and Esophageal Cancer
Detection
According to the World
Health Organization, there are 1.2 million cases of lung cancer diagnosed each year worldwide, with at least half of these resulting
in death. In the United States, lung cancer is the leading cause of death due to cancer, with 228,190 new cases and more than 159,480
deaths annually, according to the NCI’s 2013 estimates. Lung cancer is also a serious health issue in other parts of the
world where cigarette smoking is endemic (Japan, for example, with more than 63,000 deaths annually). Despite this enormous and
tragic toll, no effective method of early screening has been able to improve upon these rates. Historically, chest x-rays have
been employed, but typically these identify later stage cancers, which are difficult to cure. Sputum tests to identify cancer markers
in at-risk individuals have not been widely adopted and CT or other scanning technology is likely to be too expensive in the foreseeable
future for screening or widespread use. Once a mass has been identified, usually by chest x-ray or physical symptoms such as bloody
sputum, a bronchoscopy with biopsy and histopathological diagnosis of the mass is performed.
Worldwide, new cases of
esophageal cancer are estimated at 410,000, with more than 17,990 new cases and 15,210 deaths in the United States alone, according
to the NCI’s 2013 estimates. A precursor to esophageal cancer is a condition known as Barrett’s esophagus, which is
caused by excessive acid reflux. Patients with this condition may be subjected to repeated and sometimes poorly directed biopsies
of areas of the esophagus thought to contain cancerous or preceancerous (neoplastic) cells. Because there may be several areas
of suspicion, the clinical challenge is to try to identify those areas of the esophagus with greatest likelihood of neoplastic
change. Endoscopic techniques, using regular white light, have only limited ability to accomplish this and defensively-minded practitioners
often resort to multiple biopsies that are expensive and painful in order to increase the odds of finding disease.
Since the processes associated
with cancer development show similarities between cervical cancer and other cancers, we believe our technology, if integrated with
an endoscopic system, may have the potential to more accurately, or in an earlier state, detect lung and esophageal cancers and
precancers. To that end, we have worked with Konica Minolta to adapt our cervical cancer detection technology for detection of
lung cancer and esophageal cancer (see “—Konica Minolta”). However, we are only in the early stages of clinical
trials to evaluate this potential. We recently announced that we had received Institutional Review Board approval for testing the
technology in humans and were granted a non-significant risk designation for the device. We have two clinics in the Atlanta, Georgia
metropolitan area where we have been conducting a small scale study. The goal of the study, completed in 2012, was to establish
feasibility of the product design and clinical implementation. As part of our feasibility study, qualified subjects underwent a
standard EGD (Esophago Gastro Duodenoscopy) procedure and measurements with our device. Biopsy samples were taken in accordance
with the standard of care.
Konica Minolta
From 2008 to early 2013,
we worked with Konica Minolta to explore the feasibility of adapting our microporation and biophotonic cancer detection technologies
to other areas of medicine and to determine potential markets for these products in anticipation of a development agreement.
On April 28, 2009, we
signed a one-year exclusive negotiation and development agreement of optimization of our microporation system for manufacturing,
regulatory approval, commercialization and clinical utility with Konica Minolta. We renewed the agreement in 2010, 2011 and 2012
for additional one-year terms and changed the licensed technology to our biophotonic cancer detection technology. We received approximately
$750,000 in 2011 from Konica Minolta under this option to license agreements and received a total of $400,000 in 2012.
On
January 28, 2010, we entered into another agreement with Konica Minolta for development of our biophotonic platform specific to
the detection of esophageal cancer. In this agreement, we provided Konica Minolta with technical, regulatory and clinical development
of our biophotonic platform device
for esophageal cancer detection. In March 2011, we extended
this agreement for an additional year, effective May 1, 2011. We received approximately $1.72 million in 2011 from Konica Minolta
under these development agreements and received a total of $1.3 million for the third year of development (original period of
May 1, 2012 to April 30, 2013). In February 2013, we replaced our existing agreements with Konica Minolta with a new agreement,
pursuant to which, subject to the payment of a nominal license fee due upon FDA approval, Konica Minolta has granted us a five-year,
world-wide, non-transferable and non-exclusive right and license to manufacture and to develop a non-invasive esophageal cancer
detection product from Konica Minolta and based on our biophotonic technology platform. The license permits us to use certain
related intellectual property of Konica Minolta. In return for the license, we have agreed to pay Konica Minolta a royalty for
each licensed product we sell. We continue to have the right to seek new collaborative partners to further develop our technology.
Research, Development
and Engineering
To date, we have been
engaged primarily in the research, development and testing of our LuViva non-invasive cervical cancer detection product and our
core biophotonic technologies, as well as our since-discontinued glucose monitoring, diabetes detection and infant jaundice products.
From inception in 1992 to December 31, 2013, we have incurred about $58.4 million in research and development expenses, net of
about $24.6 million reimbursed through collaborative arrangements and government grants. Research and development costs were about
$2.7 million and $3.2 million in 2013 and 2012, respectively.
Since 2008, we have focused
our research and development and our engineering resources almost exclusively on development of our biophotonic cancer detection
technology, with only limited support of other programs funded through government contracts or third party funding. Because we
have not yet launched commercial versions of our technology, only prototypes of our cervical cancer detection product have been
tested. Because our research and clinical development programs for other cancers are at a very early stage, substantial additional
research and development and clinical trials will be necessary before commercial prototypes of our cancer detection products can
be produced.
Several of the components
used in our product or planned products are available from only one supplier, and substitutes for these components could not be
obtained easily or would require substantial modifications to our products.
Manufacturing, Sales Marketing and Distribution
We have only limited experience
in the production planning, quality system management, facility development, and production scaling that will be needed to bring
production to commercial levels. We will need to develop additional expertise in order to successfully manufacture market and distribute
any future products.
Patents
We have pursued a course
of developing and acquiring patents and patent rights and licensing technology. Our success depends in large part on our ability
to establish and maintain the proprietary nature of our technology through the patent process and to license from others patents
and patent applications necessary to develop our products. As of March 31, 2013, we have 18 granted U.S. patents relating to our
biophotonic cancer detection technology and four pending U.S. patent applications. We also have three granted patents that apply
to our interstitial fluid analysis system.
Any of the patents held
directly by us or licensed by us from third parties, or any of the processes used in the manufacture of our products, may be successfully
challenged, invalidated or circumvented. Additionally, we may not otherwise be able to rely on these patents. In addition, we cannot
be sure that competitors, many of whom have substantial resources and have made substantial investments in competing technologies,
will not seek to apply for and obtain patents that prevent, limit or interfere with our ability to make, use and sell our products
either in the United States or in foreign markets. If any of our patents are successfully challenged, invalidated or circumvented
or our rights or ability to manufacture our products were to be proscribed or limited, our ability to continue to manufacture and
market our products could be adversely affected, which would likely have a material adverse effect upon our business, financial
condition and results of operations.
Competition
The medical device industry
in general and the markets for cervical cancer detection in particular, are intensely competitive. If successful in our product
development, we will compete with other providers of cervical cancer detection and prevention products.
Current cervical cancer
screening tests, primarily the Pap test and colposcopy, are well established and pervasive. Improvements and new technologies for
cervical cancer detection and prevention, such as Thin-Prep from Hologic and HPV testing from Qiagen, have led to other new competitors.
In addition, there are other companies attempting to develop products using forms of biophotonic technologies in cervical cancer
detection, such as MediSpectra, Inc. (since acquired by Spectrascience, Inc.). MediSpectra was granted a very limited FDA approval
in March 2006 to market its device for detection of cervical cancers. The limited approval limits use of the MediSpectra device
only after a colposcopy, as an adjunct. We will be required to develop devices that are more accurate, easier to use or less costly
to administer to create devices that have a competitive advantage.
In June 2006, the FDA
approved the HPV vaccine Gardasil from drug maker Merck & Co., Inc. Gardasil is a prophylactic HPV vaccine, meaning that it
is designed to prevent the initial establishment of HPV infections. For maximum efficacy, it is recommended that girls receive
the vaccine prior to becoming sexually active. Since Gardasil will not block infection with all of the HPV types that can cause
cervical cancer, the vaccine should not be considered a substitute for routine Pap tests. On October 16, 2009, GlaxoSmithKline
PLC was granted approval in the United States for a similar preventive HPV vaccine, known as Cervarix.
Government Regulation
All of our products are,
or will be, regulated as medical devices. Medical device products are subject to rigorous FDA and other governmental agency regulations
in the United States and may be subject to regulations of relevant foreign agencies. Noncompliance with applicable requirements
can result in import detentions, fines, civil penalties, injunctions, suspensions or losses of regulatory approvals or clearances,
recall or seizure of products, operating restrictions, denial of export applications, governmental prohibitions on entering into
supply contracts, and criminal prosecution. Failure to obtain regulatory approvals or the restriction, suspension or revocation
of regulatory approvals or clearances, as well as any other failure to comply with regulatory requirements, would have a material
adverse effect on our business, financial condition and results of operations.
The FDA regulates the
clinical testing, design manufacture, labeling, packaging, marketing, distribution and record-keeping for these products to ensure
that medical products distributed in the United States are safe and effective for their intended uses.
In the United States,
medical devices are classified into one of three classes on the basis of the controls deemed necessary by the FDA to reasonably
assure the devices’ safety and effectiveness. Under FDA regulations, Class I devices are subject to general controls, such
as labeling requirements, notification to the FDA before beginning marketing activities and adherence to specified good manufacturing
practices. Class II devices are subject to general and special controls, such as performance standards, surveillance after beginning
market activities, patient registries, and FDA guidelines. Generally, Class III devices are those which must receive premarket
approval from the FDA to ensure their safety and effectiveness. Examples of Class III devices include life-sustaining, life-supporting
and implantable devices, as well as new devices that have not been found substantially equivalent to legally marketed Class I or
II devices.
A medical device manufacturer
may seek clearance to market a medical device by filing a 510(k) premarket notification with the FDA if the manufacturer establishes
that a newly developed device is substantially equivalent to either a device that was legally marketed before May 28, 1976, the
date upon which the Medical Device Amendments of 1976 were enacted, or to a device that is currently legally marketed and has received
510(k) premarket clearance from the FDA. The 510(k) premarket notification must be supported by appropriate information, which
may include data from clinical trials to establish the claim of substantial equivalence. Commercial distribution of a device for
which a 510(k) premarket notification is required can begin only after the FDA determines the device to be substantially equivalent
to a legally marketed device. The FDA has recently been requiring a more rigorous demonstration of substantial equivalence than
in the past. It generally takes from three to 12 months from the date of submission to obtain clearance of a 510(k) submission,
but it may take substantially longer. The FDA may determine that a proposed device is not substantially equivalent to a legally
marketed device, or may require additional information.
An adverse determination
or a request for additional information could delay the market introduction of new products that fall into this category, such
as LuViva, which could have a material adverse effect on our business, financial condition and results of operations. For LuViva,
any of our future products that have to be cleared through the PMA or 510(k) process, including modifications or enhancements that
could significantly affect the safety or effectiveness of the device or that constitute a major change to the intended use of the
device will require new PMA application and approval or a 510(k) premarket notification. Any modified device for which a new PMA
or 510(k) premarket notification is required cannot be distributed until the PMA is approved or 510(k) clearance is obtained. We
may not be able to obtain PMA approval or 510(k) clearance in a timely manner, if at all, for LuViva or any future devices or modifications
to LuViva or such devices for which we may submit a PMA 510(k) application.
A PMA application must
be submitted if a proposed device is not substantially equivalent to a legally marketed Class I or Class II device or for specified
Class III devices. The application must contain valid scientific evidence to support the safety and effectiveness of the device,
which includes the results of clinical trials, all relevant bench tests, and laboratory and animal studies. The application must
also contain a complete description of the device and its components, as well as a detailed description of the methods, facilities
and controls used for its manufacture, including, where appropriate, the method of sterilization and its assurance. In addition,
the application must include proposed labeling, advertising literature and any required training methods. If human clinical trials
of a device are required in connection with an application and the device presents a significant risk, the sponsor of the trial
is required to file an application for an investigational device exemption before beginning human clinical trials. Usually, the
manufacturer or distributor of the device is the sponsor of the trial. The application must be supported by data, typically
including the results of animal and laboratory testing, and a description of how the device will be manufactured. If the application
is reviewed and approved by the FDA and one or more appropriate institutional review boards, human clinical trials may begin at
a specified number of investigational sites with a specified number of patients. If the device presents a non-significant risk
to the patient, a sponsor may begin clinical trials after obtaining approval for the study by one or more appropriate institutional
review boards, but FDA approval for the commencement of the study is not required. Sponsors of clinical trials are permitted to
sell those devices distributed in the course of the study if the compensation received does not exceed the costs of manufacture,
research, development and handling. A supplement for an investigational device exemption must be submitted to and approved by the
FDA before a sponsor or an investigator may make a significant change to the investigational plan that may affect the plan’s
scientific soundness or the rights, safety or welfare of human subjects.
Upon receipt of a PMA
application, the FDA makes a threshold determination as to whether the application is sufficiently complete to permit a substantive
review. If the FDA makes this determination, it will accept the application for filing. Once the submission is accepted for filing,
the FDA begins an in-depth review of the application. An FDA review of a PMA application generally takes one to two years from
the date the application is accepted for filing. However, this review period is often significantly extended by requests for more
information or clarification of information already provided in the submission. During the review period, the submission may be
sent to an FDA-selected scientific advisory panel composed of physicians and scientists with expertise in the particular field.
The FDA scientific advisory panel issues a recommendation to the FDA that may include conditions for approval. The FDA is not bound
by the recommendations of the advisory panel. Toward the end of the PMA application review process, the FDA will conduct an inspection
of the manufacturer’s facilities to ensure that the facilities are in compliance with applicable good manufacturing practice.
If the FDA evaluations of both the PMA application and the manufacturing facilities are favorable, the FDA will issue a letter.
This letter usually contains a number of conditions, which must be met in order to secure final approval of the application. When
those conditions have been fulfilled to the satisfaction of the FDA, the agency will issue an approval letter authorizing commercial
marketing of the device for specified indications and intended uses.
The PMA application review
process can be expensive, uncertain and lengthy. A number of devices for which a premarket approval has been sought have never
been approved for marketing. The FDA may also determine that additional clinical trials are necessary, in which case the premarket
approval may be significantly delayed while trials are conducted and data is submitted in an amendment to the PMA application.
Modifications to the design, labeling or manufacturing process of a device that has received premarket approval may require the
FDA to approve supplements or new applications. Supplements to a PMA application often require the submission of additional information
of the same type required for an initial premarket approval, to support the proposed change from the product covered by the original
application. The FDA generally does not call for an advisory panel review for PMA supplements, though applicants may request one.
If any PMAs are required for our products, we may not be able to meet the FDA’s requirements or we may not receive any necessary
approvals. Failure to comply with regulatory requirements or to receive any necessary approvals would have a material adverse effect
on our business, financial condition and results of operations.
Regulatory approvals and
clearances, if granted, may include significant labeling limitations and limitations on the indicated uses for which the product
may be marketed. In addition, to obtain regulatory approvals and clearances, the FDA and some foreign regulatory authorities impose
numerous other requirements with which medical device manufacturers must comply. FDA enforcement policy strictly prohibits the
marketing of approved medical devices for unapproved uses. Any products we manufacture or distribute under FDA clearances or approvals
are subject to pervasive and continuing regulation by the FDA. The FDA also requires us to provide it with information on death
and serious injuries alleged to have been associated with the use of our products, as well as any malfunctions that would likely
cause or contribute to death or serious injury.
The FDA requires us to
register as a medical device manufacturer and list our products. We are also subject to inspections by the FDA and state agencies
acting under contract with the FDA to confirm compliance with good manufacturing practice. These regulations require that we manufacture
our products and maintain documents in a prescribed manner with respect to manufacturing, testing, quality assurance and quality
control activities. The FDA also has promulgated final regulatory changes to these regulations that require, among other things,
design controls and maintenance of service records. These changes will increase the cost of complying with good manufacturing practice
requirements.
We are also subject to
a variety of other controls that affect our business. Labeling and promotional activities are subject to scrutiny by the FDA and,
in some instances, by the Federal Trade Commission. The FDA actively enforces regulations prohibiting marketing of products for
unapproved users. We are also subject, as are our products, to a variety of state and local laws and regulations in those states
and localities where our products are or will be marketed. Any applicable state or local regulations may hinder our ability to
market our products in those regions. Manufacturers are also subject to numerous federal, state and local laws relating to matters
such as safe working conditions, manufacturing practices, environmental protection, fire hazard control and disposal of hazardous
or potentially hazardous substances. We may be required to incur significant costs to comply with these laws and regulations
now or in the future. These laws or regulations may have a material adverse effect on our ability to do business.
International sales of
our products are subject to the regulatory requirements of each country in which we market our products. The regulatory review
process varies from country to country. The European Union has promulgated rules that require medical products to affix the CE
mark, an international symbol of adherence to quality assurance standards and compliance with applicable European medical directives.
The appropriate ISO certification is one of the CE mark requirements. We maintain ISO 13485:2003 certification, which allows us
to issue a CE mark for our non-invasive cervical cancer detection device once development is complete and sell the device in the
European Union and other markets. Losing the right to affix the CE mark to our cervical cancer detection device or any future products
could have a material adverse effect on our business, financial condition and results of operations.
We will be responsible
for obtaining and maintaining regulatory approvals for our products. The inability or failure to comply with the varying regulations
or the imposition of new regulations would materially adversely affect our business, financial condition and results of operations.
Employees and Consultants
As of December 31, 2013,
we had 30 regular employees and consulting or other contract arrangements with 4 additional persons to provide services to us on
a full- or part-time basis. Of the 34 people employed or engaged by us, 12 are engaged in research and development activities,
5 are engaged in sales and marketing activities, 1 is engaged in clinical testing and regulatory affairs, 5 are engaged in manufacturing
and development, and 6 are engaged in administration and accounting. No employees are covered by collective bargaining agreements,
and we believe we maintain good relations with our employees.
Our ability to operate
successfully and manage our potential future growth depends in significant part upon the continued service of key scientific, technical,
managerial and finance personnel, and our ability to attract and retain additional highly qualified personnel in these fields.
Three of these key employees have an employment contract with us; none are covered by key person or similar insurance. In addition,
if we are able to successfully develop and commercialize our products, we likely will need to hire additional scientific, technical,
marketing, managerial and finance personnel. We face intense competition for qualified personnel in these areas, many of whom are
often subject to competing employment offers. The loss of key personnel or our inability to hire and retain additional qualified
personnel in the future could have a material adverse effect on our business, financial condition and results of operations.
PROPERTIES
Our corporate offices,
which also comprise our administrative, research and development, marketing and production facilities, are located at 5835 Peachtree
Corners East, Suite D, Norcross, Georgia 30092, where we lease approximately 23,000 square feet under a lease that expires in June
2017.
LEGAL PROCEEDINGS
We are subject
to claims and legal actions that arise in the ordinary course of business. However, we are not currently subject to any claims
or actions that we believe would have a material adverse effect on our financial position or results of operations.
MARKET FOR OUR COMMON STOCK AND RELATED
STOCKHOLDER MATTERS
Our common stock
is listed on the OTCQB marketplace under the ticker symbol “GTHP.” The number of record holders of our common stock
at April 24 2014 was $0.49.
The high and low
sales prices for the first quarter of 2014 and calendar years 2013 and 2012, as reported by the OTCQB, are as follows:
|
|
2014
|
|
2013
|
|
2012
|
|
|
High
|
|
Low
|
|
High
|
|
Low
|
|
High
|
|
Low
|
|
First Quarter
|
$ 0.60
|
|
$ 0.46
|
|
$
|
0.80
|
|
$
|
0.66
|
|
$
|
1.74
|
|
$
|
0.69
|
|
Second Quarter
|
|
|
|
$
|
0.94
|
|
$
|
0.68
|
|
$
|
0.90
|
|
$
|
0.64
|
|
Third Quarter
|
|
|
|
$
|
0.73
|
|
$
|
0.52
|
|
$
|
0.94
|
|
$
|
0.68
|
|
Fourth Quarter
|
|
|
|
$
|
0.68
|
|
$
|
0.46
|
|
$
|
0.76
|
|
$
|
0.52
|
Dividend Policy
We have not paid
any dividends since our inception and do not intend to pay any dividends in the foreseeable future. The certificate of designations
pertaining to our Series B convertible preferred stock imposes certain restrictions on our ability to pay dividends on our common
stock. For information about these restrictions and the dividends to which holders of Series B convertible preferred stock are
entitled, see “Description of Securities—Preferred Stock.”
Securities Authorized for Issuance Under Equity
Compensation Plans
All the securities we have provided
our employees, directors and consultants have been issued under our stock option plans, which are approved by our stockholders.
We have issued common stock to other individuals that are not employees or directors, in lieu of cash payments, that are not part
of any plan approved by our stockholders.
Securities authorized for issuance
under equity compensation plans:
|
Plan category
|
|
Number of
securities to be
issued upon
exercise
of
outstanding
options, warrants
and rights
|
|
Weighted-average
exercise price of
outstanding
options, warrants
and rights
|
|
Number of securities
remaining available for future
issuance under equity
compensation plans
(excluding securities reflected in column
(a))
|
|
|
|
(a)
|
|
(b)
|
|
(c)
|
|
Equity compensation plans approved by security holders
|
|
13,259,219
|
|
$0.69
|
|
4,838,516
|
|
Equity compensation plans not approved by security holders
|
|
-
|
|
-
|
|
-
|
|
TOTAL
|
|
13,259,219
|
|
$0.69
|
|
4,838,516
|
MANAGEMENT’s DISCUSSION AND ANALYSIS
OF FINANCIAL CONDITION
AND RESULTS OF OPERATIONS
The following
discussion should be read in conjunction with our financial statements and notes thereto accompanying this prospectus.
Overview
We are a medical technology
company focused on developing innovative medical devices that have the potential to improve healthcare. Our primary focus is the
development of our LuViva non-invasive cervical cancer detection device and extension of our cancer detection technology into other
cancers, including esophageal. Our technology, including products in research and development, primarily relates to biophotonics
technology for the non-invasive detection of cancers.
We are a Delaware corporation,
originally incorporated in 1992 under the name “SpectRx, Inc.,” and, on February 22, 2008, changed our name to Guided
Therapeutics, Inc. At the same time, we renamed our wholly owned subsidiary, InterScan, which originally had been incorporated
as “Guided Therapeutics.”
Since our inception, we
have raised capital through the private sale of preferred stock and debt securities, public and private sales of common stock,
funding from collaborative arrangements, and grants.
Our prospects must be
considered in light of the substantial risks, expenses and difficulties encountered by entrants into the medical device industry.
This industry is characterized by an increasing number of participants, intense competition and a high failure rate. We have experienced
operating losses since our inception and, as of December 31, 2013, we have an accumulated deficit of about $103.0 million. To date,
we have engaged primarily in research and development efforts. We do not have significant experience in manufacturing, marketing
or selling our products. Our development efforts may not result in commercially viable products and we may not be successful in
introducing our products. Moreover, required regulatory clearances or approvals may not be obtained in a timely manner, or at all.
Our products may not ever gain market acceptance and we may not ever generate significant revenues or achieve profitability. The
development and commercialization of our products requires substantial development, regulatory, sales and marketing, manufacturing
and other expenditures. We expect our operating losses to continue through at least the end of 2014 as we continue to expend substantial
resources to introduce LuViva, further the development of our other products, obtain regulatory clearances or approvals, build
our marketing, sales, manufacturing and finance organizations and conduct further research and development.
Our product revenues to
date have been limited. In 2012, the majority of our revenues were from grants from the NCI and NHI and our collaborative arrangements
with Konica Minolta. In 2013, the majority of our revenues were from grants from the NCI and NHI and revenue from the sale of LuViva
devices. We expect that the majority of our revenue in 2014 will be derived from similar sources.
Recent Developments
On October 15, 2013, Michael
C. James was elected Chairman of the board of directors. He replaced Ronald W. Allen, who retired from the board effective January
31, 2014.
On January 7, 2014, we
announced the appointment of Gene Cartwright as Chief Executive Officer of the Company, effective January 6, 2014. He was named
to the board of directors effective January 30, 2014. Mr. Cartwright replaced Mark L. Faupel, who was named Chief Scientific Officer
of the Company.
On
April 23, 2014, we entered into a securities purchase agreement with Hanover. Pursuant to the purchase agreement, we sold
Hanover a 6% senior convertible note with a principal amount of $1.5 million and an 18-month term, for a purchase price
of $1.0 million (an approximately 33.3% original issue discount). Additionally, Hanover is irrevocably bound to purchase,
on the tenth trading day after the effective date of the resale registration statement, an additional 6% senior
convertible note with a principal amount of $2.0 million and an 18-month term, for a fixed purchase price of $2.0 million,
subject only to conditions outside of Hanover’s control or that Hanover cannot cause not to be satisfied, none of which
are related to the market price of our common stock. Subject to certain limitations, the senior convertible notes are
convertible at any time after the earlier of October 23, 2014 or the effectiveness of a resale registration statement, in
whole or in part, at Hanover’s option, into shares of our common stock, at a conversion price equal to the lesser of
$0.55 per share and a discount from the lowest daily volume-weighted average price of our common stock in the five trading
days prior to conversion. The discount is 20% if the conversion takes place prior to December 19, 2014, and 25% if after that
date. We paid Hanover a commitment fee for entering into the purchase agreement in the form of 321,820 shares of common
stock. See “Description of Securities—Senior Convertible Notes”.
Critical Accounting Policies
Our material accounting
policies, which we believe are the most critical to an investors understanding of our financial results and condition, are discussed
below. Because we are still early in our enterprise development, the number of these policies requiring explanation is limited.
As we begin to generate increased revenue from different sources, we expect that the number of applicable policies and complexity
of the judgments required will increase.
Revenue Recognition:
We recognize revenue from contracts on a straight line basis, over the terms of the contract. We recognize revenue from grants
based on the grant agreement, at the time the expenses are incurred. Revenue from the sale of the Company’s products is recognized
upon shipment of such products to its customers.
Valuation of Deferred
Taxes:
We account for income taxes in accordance with the liability method. Under the liability method, we recognize deferred
assets and liabilities based upon anticipated future tax consequences attributable to differences between financial statement carrying
amounts of assets and liabilities and their respective tax bases. We establish a valuation allowance to the extent that it is more
likely than not that deferred tax assets will not be utilized against future taxable income.
Valuation of Equity Instruments
Granted to Employee, Service Providers and Investors:
On the date of issuance, the instruments are recorded at their fair value
as determined using either the Black-Scholes valuation model or Monte Carlo Simulation model. See Note 4 to the consolidated financial
statements accompanying this prospectus for the assumptions used in the Black-Scholes valuation.
Allowance for Accounts
Receivable:
We estimate losses from the inability of our customers to make required payments and periodically review the payment
history of each of our customers, as well as their financial condition, and revise our reserves as a result.
Inventory Valuation:
All
inventories are stated at lower of cost or market, with cost determined substantially on a “first-in, first-out” basis.
Selling, general, and administrative expenses are not inventoried, but are charged to expense when purchased.
Results of Operations
Comparison of 2013 and 2012
General: Net loss attributable
to common stockholders increased to approximately $10.4 million or $0.16 per share in 2013, from $4.4 million or $0.08 per share
in 2012.
Revenue from Grants and
other Agreements: Total revenues decreased to approximately $820,000 in 2013, from $3.3 million in 2012, primarily due to the decrease
in revenue associated with our prior collaborative agreements with Konica Minolta (terminated as of February 2013) to zero in 2013
from approximately $2.5 million in 2012, partially offset by an increase in revenue from NCI and NHI grants to approximately $688,000
in 2013 from $68,000 in 2012. There were no costs of sales associated with this revenue in 2013 and 2012.
Sales Revenue, Cost of
Sales and Gross Loss from Devices and Disposables: Revenues from the sale of LuViva devices for the year ended December 31, 2013
and 2012 were approximately $359,000 and $72,000, respectively. Related costs of sales and valuation allowances on the Net Realizable
Values were approximately $611,000 and $117,000, respectively, which resulted in gross losses on the device of approximately $252,000
and $45,000, respectively.
Research and Development
Expenses: Research and development expenses decreased to approximately $2.7 million in 2013, compared to approximately $3.2 million
in 2012, due to a decrease in expenses associated with our esophageal cancer technology and LuViva devices in production mode.
Sales and Marketing Expenses:
Sales and marketing expenses increased to approximately $901,000 in 2013, compared to approximately $424,000 in 2012, due to an
increase in expenses associated with marketing efforts for LuViva.
General and Administrative
Expense: General and administrative expense decreased to approximately $3.5 million in 2013, from about $3.9 million in 2012. The
decrease was primarily related to a decrease in attorney and consulting expenses for the year ended December 31, 2013.
Other Income: Other income
was approximately $110,000 in 2013, compared to zero in 2012. The increase was primarily
related to approximately $78,000 received from
our insurance provider as a distribution, as well as a refund from one of our distributors of approximately $18,000.
Interest Expense: Interest
expense decreased to approximately $45,000 for the year ended December 31, 2013, as compared to expenses of approximately $72,000
for the same period in 2012. The decrease was primarily due to a reduction in past due notes payable.
Fair Value of Warrants
Expense: Fair value of warrants expensed were approximately $674,000 for the year ended December 31, 2013, as compared to none
for the same period in 2012.
There was no income tax
benefit recorded for the years ended December 31, 2013 and 2012, due to recurring net operating losses.
Liquidity and Capital Resources
Since our inception, we
have raised capital through the private sale of preferred stock and debt securities, public and private sales of common stock,
funding from collaborative arrangements, and grants. At December 31, 2013, we had cash of approximately $613,000 and a working
capital of approximately $268,000.
Our major cash flows in
the year ended December 31, 2013 consisted of cash out-flows of $5.6 million from operations, including approximately $7.2 million
of net loss, cash outflows of $107,000 from investing activities and a net change from financing activities of $5.3 million, which
primarily represented the proceeds received from issuance of common and preferred stock, as well as exercise of outstanding warrants
and options.
In July 2012, we
completed a warrant exchange program, pursuant to which we exchanged warrants exercisable for a total of 15,941,640 shares of
common stock, or 56.29% of the warrants eligible to participate, for three classes of new warrants. The first class of new
warrants expired on September 17, 2012 and carried an exercise price of $0.40, $0.45 or $0.50, depending on the date
exercised. The second class of new warrants expired on either July 26, 2013 or March 1, 2014, and were exercisable at $0.65.
The third class of new warrants are exercisable for approximately 472,000 shares and 3.6 million shares at $0.80 per share
and expire on July 26, 2014 and March 1, 2015, respectively. As of December 31, 2012, we had issued 5,825,957 shares of
common stock and received approximately $2.9 million in cash, in connection with the exercise of these new warrants.
On May 23, 2013, we completed
a private placement of our Series B convertible preferred stock and warrants to purchase shares of our common stock. We issued
an aggregate of 2,527 shares of Series B convertible preferred stock at a purchase price of $1,000 per share. The initial conversion
price of the Series B convertible preferred stock was $0.68 per share, such that each share would convert into 1,471 shares of
our common stock, subject to customary adjustments, including for any accrued but unpaid dividends and pursuant to certain anti-dilution
provisions. We also issued warrants, on a pro rata basis to the investors, exercisable to purchase an aggregate of 3,716,177 shares
of our common stock. The warrants, which carry a five-year term, were split evenly into two tranches, one of which is subject to
a mandatory exercise provision. The warrants are exercisable at any time and had an initial exercise price of $1.08 per share,
subject to certain customary adjustments contained in the respective warrants. As a result of the November 2013 warrant exchange
program described below, the conversion price of the Series B convertible preferred stock has been lowered to $0.40 per share,
such that each share is now convertible into 2,500 shares of common stock, and one tranche of the warrants, previously exercisable
for 1,858,089 shares of common stock at $1.08 per share, is now exercisable for 5,016,840 shares at $0.40 per share.
In November 2013, we completed
another warrant exchange program pursuant to which we exchanged warrants exercisable for a total of 3,573,691 shares of common
stock, or 99.5% of the warrants eligible to participate, for new warrants exercisable for the same number of shares of common stock,
but with a reduced exercise price of $0.40 per share and a shortened exercise period ending on November 27, 2013. As of December
31, 2013, we had issued 3,399,965 shares of common stock and received approximately $1.4 million in cash in connection with the
exercise of these new warrants.
We will be required to
raise additional funds through public or private financing, additional collaborative relationships or other arrangements. We believe
our existing and available capital resources will be sufficient to satisfy our funding requirements through the first quarter of
2014 We are evaluating various options to further reduce our cash requirements to operate at a reduced rate, as well as options
to raise additional funds, including loans.
Substantial capital will
be required to develop our products, including completing product testing and clinical trials, obtaining all required U.S. and
foreign regulatory approvals and clearances, and commencing and scaling up manufacturing and marketing our products. Any failure
to obtain capital would have a material adverse effect on our business, financial condition and results of operations.
Our financial statements
have been prepared and presented on a basis assuming we will continue as a going concern. The above factors raise substantial doubt
about our ability to continue as a going concern, as more fully discussed in Note 1 to the consolidated financial statements contained
herein and in the report of our independent registered public accounting firm accompanying our financial statements.
Off-Balance Sheet Arrangements
We have no material off-balance sheet arrangements;
no special purpose entities; nor do activities that include non-exchange-traded contracts account for at fair value.
DIRECTORS AND EXECUTIVE OFFICERS
Our executive officers are elected by and
serve at the discretion of our board of directors. The following table lists information about our directors and executive
officers as of April 24, 2014:
|
Name
|
Age
|
Position with Guided Therapeutics
|
|
Gene S. Cartwright, Ph.D.
|
60
|
Chief Executive Officer, Acting Chief Financial Officer, President and Director
|
|
Mark L. Faupel, Ph.D.
|
58
|
Chief Scientific Officer and Director
|
|
Richard L. Fowler
|
57
|
Senior Vice President of Engineering
|
|
Ronald W. Hart, Ph.D.
|
71
|
Vice Chairman and Director
|
|
John E. Imhoff, M.D.
|
64
|
Director
|
|
Michael C. James
|
55
|
Chairman of the Board and Director
|
|
Jonathan M. Niloff, M.D.
|
60
|
Director
|
|
Linda Rosenstock, M.D.
|
63
|
Director
|
Except as set forth below,
all of the executive officers have been associated with us in their present or other capacities for more than the past five years.
Officers are elected annually by the board of directors and serve at the discretion of the board. There are no family relationships
among any of our executive officers and directors.
Gene S. Cartwright,
Ph.D.
joined us in January 2014 as the President, Chief Executive Officer and Acting Chief Financial Officer. He was elected
as a director on January 31, 2014. His most recent position was with Omnyx, LLC, a Joint Venture between GE Healthcare and the
University of Pittsburgh Medical Center, where, as CEO for over four years he founded and managed the successful development of
products for the field of Digital Pathology. Prior to his work with Omnyx, LLC, he was President of Molecular Diagnostics for GE
Healthcare. Prior to GE, Dr. Cartwright was Divisional Vice President/General Manager for Abbott Diagnostics’ Molecular Diagnostics
business. In his 24 year career at Abbott, he also served as Divisional Vice President for U.S. Marketing for five years. He received
a Masters of Management degree from Northwestern’s Kellogg School of Management and also holds a Ph.D. in chemistry from
Stanford University and an AB from Dartmouth College.
Dr. Cartwright brings
over 30 years of experience working in the IVD diagnostics industry. He has great experience in the diagnostics market both in
the development and introduction of new diagnostics technologies, as well as extensive successful commercial experience with global
businesses. With his background and experience, Dr. Cartwright, as President, CEO and Director will work with and advise the board
as to how we can successfully market and build the LuViva international sales.
Mark L. Faupel, Ph.D.
was appointed Chief Scientific Officer effective January 7, 2014. He has been a director since 2007 and has more than 26 years
of experience in developing non-invasive alternatives to surgical biopsies and blood tests, especially in the area of cancer screening
and diagnostics. Dr. Faupel also served as our Chief Executive Officer from May 2007 to January 2014 and prior thereto was our
Chief Technical Officer from April 2001 to May 2007. Prior to coming to us in 1998, Dr. Faupel was the co-founder and Vice President
of Research and Development at Biofield Corp. His work in early stage cancer detection has won two international awards and he
is a former member of the European School of Oncology Task Force. Dr. Faupel has served as a National Institutes of Health reviewer,
is the inventor on 15 U.S. patents and has authored numerous scientific publications and presentations, appearing in such peer-reviewed
journals as The Lancet. Dr. Faupel earned his Ph.D. in neuroanatomy and physiology from the University of Georgia.
Dr. Faupel’s extensive
experience in founding and managing point of care cancer detection companies includes the basic scientific applications, clinical
trials, regulatory affairs and financing. As such, Dr. Faupel advises the board on all aspects of our business.
Rick Fowler
, Mr.
Fowler, Sr. VP of Engineering is an accomplished Executive with significant experience in the management of businesses that sell,
market, produce and develop sophisticated medical devices and instrumentation. Mr. Fowler’s 25 plus years of experience includes
assembling and managing teams, leading businesses and negotiating contracts, conducting litigation, and developing ISO, CE, FDA
QSR, GMP and GCP compliant processes and products. He is adept at providing product life cycle management through effective process
definition and communication - from requirements gathering, R&D feasibility, product development, product launch, production
startup and support. Mr. Fowler combines outstanding analytical, out-of-the-box, and strategic thinking with strong leadership,
technical, and communication skills and he excels in dynamic, demanding environments while remaining pragmatic and focused. He
is able to deliver high risk projects on time and under budget as well as enhance operational effectiveness through outstanding
cross-functional team leadership (R&D, marketing, product development, operations, QA, sales, service, and finance). In addition,
Mr. Fowler is well versed in global medical device regulatory and product compliance requirements.
Ronald W. Hart, Ph.D.
has served as a member of our board since March 2007 and was elected Vice Chairman of the Board in 2011. He has published over
600 peer-reviewed publications, has been appointed to a number of academic positions and is credited with developing the first
direct proof that DNA is causal in certain forms of cancer. He chaired a number of federal committees and task forces, including
the development and implementation of the Technology Transfer Act of 1986 and the White House Task Force on Chemical Carcinogenesis.
In 1980, Dr. Hart was appointed Director of the National Center for Toxicological Research, the research arm of the FDA, a position
he held until 1992. In 1992, Dr. Hart was the first ever Presidential Appointee to the position of Distinguished Scientist in Residence
for the US Public Health Service/FDA, a position he held until his retirement in 2000. Dr. Hart received his Ph.D. in physiology
and biophysics from the University of Illinois. Dr. Hart has helped in the development of business strategy for a number of start-up
companies.
Dr. Hart adds considerable
value to the board in at least four critical areas:
(1) As
a former FDA bureau chief, he advises the Board and management on our FDA relationship and strategy.
(2) As
an active participant in the venture community, he advises the Board on financing and other opportunities.
(3) As
an expert in organizational matters, he advises the Board and management regarding company strategy and potential strategic partnerships.
(4) As
an expert in international trade, he advises the Board and management on international partnering and distribution agreements.
John E. Imhoff, M.D.
has served as a member of our board since April 2006. Dr. Imhoff is an ophthalmic surgeon who specializes in cataract and refractive
surgery. He is one of our principal stockholders and invests in many other private and public companies. He has a B.S. in Industrial
Engineering from Oklahoma State University, an M.D. from the University of Oklahoma and completed his ophthalmic residency at the
Dean A. McGee Eye Institute. He has worked as an ophthalmic surgeon and owner of Southeast Eye Center since 1983.
Dr. Imhoff has experience
in clinical trials and in other technical aspects of a medical device company. His background in industrial engineering is especially
helpful to our company, especially as Dr. Imhoff can combine this knowledge with clinical applications. His experience in the investment
community also lends itself as invaluable to a public company that participates in equity transactions.
Michael C. James
has
served as a member of our board since March 2007 and as Chairman of the Board since October 15, 2013. Mr. James is also the Managing
Partner of Kuekenhof Capital Management, LLC, a private investment management company, Chief Executive Officer and the Chief Financial
Officer of Inergetics, Inc., a nutraceutical supplements company and also the Chief Financial Officer of Terra Tech Corporation,
which is a hydroponic and agricultural company. He also holds the position of Managing Director of Kuekenhof Equity Fund, L.P.
and Kuekenhof Partners, L.P. Mr. James currently sits on the board of directors of Inergetics, Inc. Mr. James was Chief Executive
Officer of Nestor, Inc. from January 2009 to September 2009 and served on their board of directors from July 2006 to June
2009. He was employed by Moore Capital Management, Inc., a private investment management company from 1995 to 1999 and held position
of Partner. He was employed by Buffalo Partners, L.P., a private investment management company from 1991 to 1994 and held the position
of Chief Financial and Administrative Officer. He began his career in 1980 as a staff accountant with Eisner LLP. Mr. James received
a B.S. degree in Accounting from Farleigh Dickinson University in 1980.
Mr. James has experience
both in the areas of company finance and accounting, which is invaluable to us during financial audits and offerings. Mr. James
has extensive experience in the management of both small and large companies and his entrepreneurial background is relevant as
we develop as a company.
Jonathan M. Niloff,
M.D.
was elected as a director in April 2010. Dr. Niloff is Vice President and Executive Medical Director Population Health
of McKesson Technology Solutions, a medical software company. Prior to that, Dr. Niloff was the Founder, Chairman of the Board
and Chief Medical Officer of MedVentive Inc. Prior to joining MedVentive, Dr. Niloff served as President of the Beth Israel Deaconess
Physicians Organization, Medical Director for Obstetrics and Gynecology for its Affiliated Physicians Group, and Chief of Gynecology
at New England Deaconess Hospital. He served as an Associate Professor of Obstetrics, Gynecology, and Reproductive Biology at Harvard
Medical School. He has deep expertise in all aspects of medical cost and quality improvement, and has published extensively on
the topic of gynecologic oncology including the development of the CA125 test for ovarian cancer. Dr. Niloff received his undergraduate
education at The Johns Hopkins University, an MD degree from McGill University, and an MBA degree from Boston University.
Dr. Niloff is uniquely
qualified to assist the board and management because he combines his clinical background as a Harvard Ob-Gyn with his business
acumen developed through an MBA degree and as CMO of MedVentive. Dr. Niloff has specific experience in evaluating new medical technology
(e.g., CA125) and its implications to cost containment and reimbursement. Furthermore, Dr. Niloff has numerous professional contacts
in the Ob-Gyn community that can aid in our development and marketing of our cervical cancer detection technology.
Linda Rosenstock,
M.D.
was appointed to the board in April 2012.
Dr. Linda Rosenstock is a Dean Emeritus and Professor of the University
of California, Los Angeles (UCLA) Fielding School of Public Health, a position she has held since 2000. She holds appointments
as Professor of Medicine and Environmental Health Sciences and is a recognized authority in broad areas of public health and science
policy. Internationally, Dr. Rosenstock has been active in teaching and research in many developing countries and has served as
an advisor to the World Health Organization. Dr. Rosenstock also chaired the United Auto Workers/General Motors Occupational Health
Advisory Board. She is an Honorary Fellow of the Royal College of Physicians and an elected member of the National Academy of Sciences'
Institute of Medicine where she has served as a member of their board on Health Sciences Policy and Chair of the Committee for
Preventive Services for Women. In January 2011, she was appointed by President Obama to the Advisory Group on Prevention, Health
Promotion and Integrative and Public Health. She has served on the board of directors for Skilled Health Care since 2009.
Before coming to UCLA
in 2000, Dr. Rosenstock served as Director of the National Institute for Occupational Safety and Health (NIOSH) for nearly seven
years. As Director of NIOSH, Dr. Rosenstock led the only federal agency with a mandate to undertake research and prevention activities
in occupational safety and health. During her tenure, she was instrumental in creating the National Occupational Research Agenda,
a framework for guiding occupational safety and health research, and in expanding the agency's responsibilities. In recognition
of her efforts, Dr. Rosenstock received the Presidential Distinguished Executive Rank Award, the highest executive service award
in the government and was also the James P. Keogh Award Winner for 2011 in appreciation of a lifetime of extraordinary leadership
in occupational health and safety. Dr. Rosenstock received her M.D. and M.P.H. from The Johns Hopkins University. She conducted
her advanced training at the University of Washington, where she was Chief Resident in Primary Care Internal Medicine and a Robert
Wood Johnson Clinical Scholar.
Dr. Rosenstock
is uniquely qualified as a board member for guided therapeutics. First, as a trained physician who also chairs the preventive services
for women committee of the institute of national academy of sciences institute of medicine, she has been directly involved in setting
institutional and government policy for breast and cervical cancer screening, which is directly relevant to our LuViva cervical
cancer detection device. Secondly, she brings a wealth of international experience in developing countries, which is a focus of
our product distribution effort in cancer detection. Thirdly, she has demonstrated a lifetime of extraordinary leadership and her
international recognition as an expert in health policy will provide outstanding credibility to guided therapeutics as a leading
innovator in women’s healthcare.
EXECUTIVE COMPENSATION
Summary Compensation Table
The following table lists specified compensation we paid during
each of the fiscal years ended December 31, 2013 and 2012 to the chief executive officer and our two other most highly compensated
executive officers, collectively referred to as the named executive officers, in 2013:
2013 and 2012 Summary Compensation Table
|
Name and Principal Position
|
Year
|
Salary
($)
|
Bonus
($)
|
Option
Awards
($)(1)
|
Total
($)
|
|
Mark Faupel, Ph.D.
President, CEO, Acting CFO and Director (2)
|
2013
|
243,000
|
-
|
-
|
243,000
|
|
2012
|
243,000
|
-
|
214,500
|
457,000
|
|
Richard Fowler,
Senior Vice President of Engineering
|
2013
2012
|
197,000
195,000
|
-
-
|
-
6,250
|
197,000
195,000
|
|
Shabbir Bambot, Ph.D. (3)
Vice President of Research and Development
|
2013
|
80,222
|
-
|
-
|
80,222
|
|
2012
|
193,000
|
-
|
6,000
|
193,000
|
|
|
(1)
|
See Note 3 to the consolidated financial statements that accompany this prospectus.
|
|
|
(2)
|
Dr. Faupel currently serves as the Company’s Chief Scientific Officer.
|
|
|
(3)
|
Dr. Bambot resigned from the Company on May 10, 2013.
|
Dr. Faupel’s 2013 and 2012 compensation
consisted of a base salary of $243,000, and usual and customary company benefits. As of December 31, 2013, Dr. Faupel’s remaining
deferred salary was approximately $225,861. On July 2, 2012, Dr. Faupel was issued 153,846 shares of common stock at $0.65, in
partial repayment of debt.
Mr. Fowler’s 2013 and 2012 compensation
consisted of a base salary of $197,000 and $195,000, respectively, and usual and customary company benefits. He received no bonus
and no stock options in 2013 and received 6,250 stock options in 2012. As of December 31, 2013, Mr. Fowler’s total deferred
salary was approximately $98,858.
Dr. Bambot’s 2013 and 2012 compensation
consisted of a base salary of $193,000, and $193,000, respectively, and usual and customary company benefits.
Outstanding Equity Awards
to Officers at December 31, 2013
|
|
Option Awards
|
|
Name and Principal Position
|
Number of
Securities
Underlying
Options
Exercisable
(#)(1)
|
Number of
Securities
Underlying
Options Un-
exercisable
(#)
|
Equity Incentive
Plan
Awards: Number of
Securities Under-
lying Un-exercised
Unearned Options
(#)
|
Option
Exercise
Price
($)(2)
|
Option
Expiration
Date
|
|
Mark Faupel, Ph.D.
President, CEO & Acting CFO
|
1,878,244
|
-
|
400,105
|
0.63
|
12/16/2021
|
|
Richard Fowler
Senior Vice President of Engineering
|
405,062
|
-
|
90,938
|
0.48
|
12/16/2021
|
|
|
(1)
|
Represents fully vested options.
|
|
|
(2)
|
Based on all outstanding options
|
Outstanding Equity Awards to Directors at
December 31, 2013
|
|
Option Awards
|
|
Name and Principal Position
|
Option Awards
(#)
|
Exercise Price
($)
|
|
Ronald W. Allen
Former Chairman and Director
|
636,250
|
0.40
|
|
Ronald W. Hart, Ph.D.
Director
|
517,500
|
0.37
|
|
John E. Imhoff, M.D.
Director
|
303,750
|
0.78
|
|
Michael C. James
Current Chairman and Director
|
107,500
|
0.78
|
|
Jonathan Niloff, M.D.
Director
|
142,917
|
0.74
|
|
Linda Rosenstock
Director
|
125,000
|
0.80
|
The following Board members also serve as consultants
to the company:
-
Ronald W. Hart, Ph.D. – Dr. Hart, as part of his board duties, provides
advice on regulatory and clinical issues, especially with advice for the Company with regard to its application to the FDA.
-
Ronald W. Allen – Mr. Allen advised the company with regard to personnel
and financing. As such, he played an important role in identifying potential funding sources.
SHARE OWNERSHIP OF DIRECTORS, OFFICERS
AND CERTAIN BENEFICIAL OWNERS
The following table lists information regarding
the beneficial ownership of our common stock as of April 24, 2014 by (i) each person whom we know to beneficially own more than
5% of the outstanding shares of our common stock (a “5% stockholder”), (ii) each director, (iii) each officer named
in the summary compensation table elsewhere in this prospectus, and (iv) all directors and executive officers as a group. Unless
otherwise indicated, the address of each officer and director is 5835 Peachtree Corners East, Suite D. Norcross, Georgia 30092.
|
Name and Address of Beneficial Owner
|
|
Amount and
Nature of
Beneficial
Ownership (1)
|
|
Percent
of
Class (2)
|
|
John E. Imhoff (3)
|
|
|
13,813,669
|
|
|
|
17.10
|
%
|
The Whittemore Collection, Ltd. / George Landegger (4)
4 International Drive, Rye Brook, NY 10573
|
|
|
7,198,653
|
|
|
|
9.22
|
%
|
|
Michael C. James / Kuekenhof Equity Fund, LLP (5)
|
|
|
361,337
|
|
|
|
*
|
|
|
Ronald Hart (6)
|
|
|
1,987,344
|
|
|
|
2.54
|
%
|
|
Mark L. Faupel (7)
|
|
|
2,580,158
|
|
|
|
3.23
|
%
|
|
Richard L. Fowler (8)
|
|
|
499,114
|
|
|
|
*
|
|
|
Linda Rosenstock (9)
|
|
|
260,000
|
|
|
|
*
|
|
|
Jonathan Niloff (10)
|
|
|
330,209
|
|
|
|
*
|
|
|
|
|
|
|
|
|
|
|
|
|
All directors and executive officers as a group (7 persons) (11)
|
|
|
19,831,830
|
|
|
|
23.39
|
%
|
|
____________
|
|
*
|
Less than 1%
|
|
(1)
|
Except as otherwise indicated in the footnotes to this table and pursuant to applicable community property laws, the persons named in the table have sole voting and investment power with respect to all shares of common stock.
|
|
(2)
|
Percentage ownership is based on 70,503,961 shares of common stock outstanding as of March 15, 2014. Beneficial ownership is determined in accordance with the rules of the SEC, based on factors that include voting and investment power with respect to shares. Shares of common stock subject to currently exercisable options, warrants, convertible preferred stock or convertible notes, or any such securities exercisable within 60 days after March 16, 2014, are deemed outstanding for purposes of computing the percentage ownership of the person holding those options, but are not deemed outstanding for purposes of computing the percentage ownership of any other person.
|
|
(3)
|
Consists of 10,554,984 shares of common stock and common equivalent, 2,954,935 warrants to purchase common stock at an average price of $0.70 per share and 303,750 shares subject to stock options. Dr. Imhoff is on the board of directors.
|
|
(4)
|
Consists of 6,641,409 shares of common stock and 557,244 warrants to purchase common stock at an average price of $0.82 per share.
|
|
(5)
|
Consists of 148,126 warrants to purchase common stock at an average price of $0.80 per share and 107,500 shares subject to stock options held by Michael James; and 105,711 warrants to purchase common stock at an average price of $0.80 per share held by Kuekenhof Equity Fund, LP, Michael James, managing partners. Mr. James is on the board of directors.
|
|
(6)
|
Consists of 926,508 shares of common stock and common equivalent, 89,535 warrants to purchase common stock at an average price of $0.64 per share and 655,000 shares subject to stock options
held by Ronald Hart; and
265,019 shares of common stock and 51,282 warrants to purchase common stock at an average price of $0.80 per share held by Hart Management, LLC
. Dr. Hart is on the board of directors.
|
|
(7)
|
Consists of 267,476 shares of common stock and 2,312,682 shares subject to stock options.
|
|
(8)
|
Consists of 98,115 shares
of common stock and
400,999 shares subject to stock options.
|
|
(9)
|
Consists of 135,000 shares of common stock and 125,000
shares subject to stock options held by Linda Rosenstock. Dr. Rosenstock is on the board of directors.
|
|
(10)
|
Consists of 317,292 shares
of common stock
and 142,917 shares subject to stock options held by Jonathan M. Niloff. Dr. Niloff is on the board of directors.
|
|
(11)
|
Consists of 12,582,519 shares of common stock, 3,201,463 warrants to purchase common stock at $0.40 to $.80 per share and 4,047,848 shares subject to stock options.
|
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS
AND DIRECTOR INDEPENDENCE
Our board of directors
recognizes that related person transactions present a heightened risk of conflicts of interest. The audit committee of the board
has the authority to review and approve all related party transactions involving directors or executive officers of the Company.
Under the policy,
when management becomes aware of a related person transaction, management reports the transaction to the audit committee and requests
approval or ratification of the transaction. Generally, the audit committee will approve only related party transactions that are
on terms comparable to those that could be obtained in arm’s length dealings with an unrelated third person. The audit committee
will report to the full board all related person transactions presented to it.
Based on the definition
of independence of the NASDAQ Stock Market, the board has determined that Messrs. Allen and James, and Drs. Hart, Niloff, Rosenstock
and Imhoff are independent directors.
Director John Imhoff
invested a total of $586,568 to convert 1,466,420 warrants at $0.40 in November 2013. He also participated in our Series B preferred
issuance during the fiscal year ended December 31, 2013 for a total of $500,000.
Between February 20, and April 24, 2014,
we have received cash advances with a 10% interest rate form our Affiliates, as follows:
|
|
|
Director
|
Officer
|
|
Affiliate
|
|
John Imhoff
|
Linda Rosenstock
|
Michael James
|
Gene Cartwright
|
Richard Fowler
|
|
Principal Amount
|
|
$ 25,000
|
$ 50,000
|
$ 50,000
|
$ 150,000
|
$ 50,000
|
LEGAL MATTERS
Jones Day, Atlanta, Georgia, passed
upon the validity of the shares of common stock that may be offered by this prospectus.
EXPERTS
Our consolidated
financial statements as of December 31, 2013 and 2012, and for the years then ended have been audited by UHY LLP, an independent
registered public accounting firm, as set forth in its report, included in this prospectus. Our financial statements and the related
independent registered public accounting firm report thereon have been included herein in reliance upon such report given on the
authority of such firm as experts in accounting and auditing.
The audit report covering the December 31, 2013 consolidated financial statements contains an explanatory
paragraph that states that the Company has suffered recurring losses and has an accumulated deficit that raise substantial doubt
about its ability to continue as a going concern. The consolidated financial statements do not include any adjustments that
might result from the outcome of this uncertainty.
WHERE
YOU CAN GET MORE INFORMATION
We have filed with
the SEC under the Securities Act a registration statement on Form S-1 of which this prospectus forms a part. This prospectus does
not contain all of the information contained in the registration statement and its exhibits. We strongly encourage you to read
carefully the registration statement and its exhibits.
Any statement made
in this prospectus concerning the contents of any contract, agreement or other document is only a summary of the actual contract,
agreement or other document. If we have filed any contract, agreement or other document as an exhibit to the registration statement,
you should read the exhibit for a more complete understanding of the document or matter involved.
We file annual,
quarterly and current reports; proxy statements and other information with the SEC. You may read and copy any of this information
at the SEC’s Public Reference Room at 100 F Street, N.E., Washington, D.C. 20549. Please call the SEC at 1-800-SEC-0330 for
information on the operation of the Public Reference Room. The SEC also maintains an Internet website that contains reports, proxy
statements and other information regarding issuers, including us, who file electronically with the SEC. The address of that site
is http://www.sec.gov. The information contained on the SEC’s website is expressly not incorporated by reference into this
prospectus.
INDEX
TO FINANCIAL STATEMENTS
Annual Consolidated Financial Statements
|
Report of Independent Registered Public Accounting Firm
|
F-2
|
|
Consolidated Balance Sheets as of December 31, 2013 and 2012
|
F-3
|
|
Consolidated Statements of Operations for the Years Ended December 31, 2013 and 2012
|
F-4
|
Consolidated Statements of Changes in Stockholders Equity (Deficit) for the Years Ended
December 31, 2013 and 2012
|
F-5
|
|
Consolidated Statements of Cash Flows for the Years Ended December 31, 2012 and 2011
|
F-6
|
|
Notes to Consolidated Financial Statements
|
F-7
|
|
|
|
FINANCIAL
STATEMENTS AND SUPPLEMENTARY DATA FOR
THE FISCAL YEAR ENDED DECEMBER 31, 2013
Report of Independent Registered Public
Accounting Firm
To the Board of Directors and
Stockholders of Guided Therapeutics,
Inc.
We have audited the accompanying consolidated
balance sheets of Guided Therapeutics, Inc. and Subsidiary (the “Company”) as of December 31, 2013 and 2012, and the
related consolidated statements of operations, stockholders’ equity (deficit), and cash flows for the years then ended. The
Company’s management is responsible for these consolidated financial statements. Our responsibility is to express an opinion
on these consolidated financial statements based on our audits.
We conducted our audits in accordance with
the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform
the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement.
The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting.
Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures that are
appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s
internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test
basis, evidence supporting the amounts and disclosures in the consolidated financial statements, assessing the accounting principles
used and significant estimates made by management, as well as evaluating the overall consolidated financial statement presentation.
We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial
statements referred to above present fairly, in all material respects, the financial position of Guided Therapeutics, Inc. and
Subsidiary as of December 31, 2013 and 2012, and the results of their operations and their cash flows for the years then ended
in conformity with accounting principles generally accepted in the United States of America.
As described in Note 1 to the consolidated
financial statements, the accompanying consolidated financial statements have been prepared assuming that the Company will continue
as a going concern. The Company’s recurring losses from operations and accumulated deficit raise substantial doubt about
its ability to continue as a going concern. Management’s plans concerning these matters are also discussed in Note 1 to
the consolidated financial statements. The consolidated financial statements do not include any adjustments that might result
from the outcome of this uncertainty.
/s/ UHY LLP
UHY LLP
Sterling Heights, Michigan
March 26, 2014
|
GUIDED THERAPEUTICS, INC. AND SUBSIDIARY
|
|
CONSOLIDATED BALANCE SHEETS
|
|
AS OF DECEMBER 31, 2013 AND 2012
|
|
(In Thousands)
|
|
|
|
|
|
|
|
ASSETS
|
|
|
2013
|
|
|
|
2012
|
|
|
CURRENT ASSETS:
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
613
|
|
|
$
|
1,044
|
|
Accounts receivable, net of allowance for doubtful accounts of $18 and $12 at
December 31, 2013 and 2012, respectively
|
|
|
133
|
|
|
|
107
|
|
|
Inventory, net of reserves of $184 and $52 at December 31, 2013 and 2012, respectively
|
|
|
1,193
|
|
|
|
524
|
|
|
Other current assets
|
|
|
101
|
|
|
|
198
|
|
|
Total current assets
|
|
|
2,040
|
|
|
|
1,873
|
|
|
|
|
|
|
|
|
|
|
|
|
Property and equipment, net
|
|
|
920
|
|
|
|
1,274
|
|
|
Other assets
|
|
|
356
|
|
|
|
331
|
|
|
Total noncurrent assets
|
|
|
1,276
|
|
|
|
1,605
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL ASSETS
|
|
$
|
3,316
|
|
|
$
|
3,478
|
|
|
|
|
|
|
|
|
|
|
|
|
LIABILITIES AND STOCKHOLDERS’ (DEFICIT) EQUITY
|
|
|
|
|
|
|
|
|
|
CURRENT LIABILITIES:
|
|
|
|
|
|
|
|
|
|
Short-term notes payable
|
|
$
|
35
|
|
|
$
|
79
|
|
|
Current portion of long term debt
|
|
|
109
|
|
|
|
4
|
|
|
Notes payable – past due
|
|
|
—
|
|
|
|
419
|
|
|
Accounts payable
|
|
|
891
|
|
|
|
765
|
|
|
Accrued liabilities
|
|
|
723
|
|
|
|
1,038
|
|
|
Deferred revenue
|
|
|
14
|
|
|
|
40
|
|
|
Total current liabilities
|
|
|
1,772
|
|
|
|
2,345
|
|
|
|
|
|
|
|
|
|
|
|
|
Warrants, at fair value
|
|
|
1,548
|
|
|
|
—
|
|
|
Long-term debt, net
|
|
|
103
|
|
|
|
—
|
|
|
Total long-term liabilities
|
|
|
1,651
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL LIABILITIES
|
|
|
3,423
|
|
|
|
2,345
|
|
|
|
|
|
|
|
|
|
|
|
|
COMMITMENTS & CONTINGENCIES (Note 5)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
STOCKHOLDERS’ (DEFICIT) EQUITY:
|
|
|
|
|
|
|
|
|
Series B convertible preferred stock, $.001 par value; 3 shares authorized,
2 and zero shares
issued and outstanding as of December 31, 2013 and 2012, respectively (liquidation preference of $2.1
million and $0 at December 31, 2013 and 2012, respectively)
|
|
|
1,139
|
|
|
|
—
|
|
|
Common stock, $.001 par value; 145,000 shares authorized, 70,479 and 62,282 shares issued and outstanding as of December 31, 2013 and 2012, respectively
|
|
|
71
|
|
|
|
62
|
|
|
Additional paid-in capital
|
|
|
101,840
|
|
|
|
93,273
|
|
|
Treasury stock, at cost
|
|
|
(132
|
)
|
|
|
(104
|
)
|
|
Accumulated deficit
|
|
|
(103,025
|
)
|
|
|
(92,098
|
)
|
|
TOTAL GUIDED THERAPEUTICS STOCKHOLDERS’ (DEFICIT) EQUITY
|
|
|
(107
|
)
|
|
|
1,133
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL STOCKHOLDERS’ (DEFICIT) EQUITY
|
|
|
(107
|
)
|
|
|
1,133
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL LIABILITIES AND STOCKHOLDERS’ (DEFICIT) EQUITY
|
|
$
|
3,316
|
|
|
$
|
3,478
|
|
|
|
|
|
|
|
|
The accompanying notes are an integral part of these consolidated statements.
|
|
GUIDED THERAPEUTICS, INC. AND SUBSIDIARY
|
|
CONSOLIDATED STATEMENTS OF OPERATIONS
|
|
FOR THE YEARS ENDED DECEMBER 31, 2013 AND 2012
|
|
(In Thousands Except Per Share Data)
|
|
|
|
|
|
|
|
|
|
|
2013
|
|
|
|
2012
|
|
|
REVENUE:
|
|
|
|
|
|
|
|
|
|
Contract and grant revenue
|
|
$
|
820
|
|
|
$
|
3,338
|
|
|
|
|
|
|
|
|
|
|
|
|
Sales – devices and disposables
|
|
|
359
|
|
|
|
72
|
|
|
Cost of goods sold
|
|
|
611
|
|
|
|
117
|
|
|
Gross loss
|
|
|
(252
|
)
|
|
|
(45
|
)
|
|
|
|
|
|
|
|
|
|
|
|
OPERATING EXPENSES:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
2,742
|
|
|
|
3,227
|
|
|
Sales and marketing
|
|
|
901
|
|
|
|
424
|
|
|
General and administrative
|
|
|
3,533
|
|
|
|
3,923
|
|
|
Total operating expenses
|
|
|
7,174
|
|
|
|
7,574
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating loss
|
|
|
(6,606
|
)
|
|
|
(4,281
|
)
|
|
|
|
|
|
|
|
|
|
|
|
OTHER INCOME (EXPENSES):
|
|
|
|
|
|
|
|
|
|
Other income
|
|
|
110
|
|
|
|
—
|
|
|
Interest expense
|
|
|
(45
|
)
|
|
|
(72
|
)
|
|
Change in fair value of warrants
|
|
|
(674
|
)
|
|
|
—
|
|
|
Total other income
|
|
|
(609
|
)
|
|
|
(72
|
)
|
|
|
|
|
|
|
|
|
|
|
|
LOSS FROM OPERATIONS
|
|
|
(7,215
|
)
|
|
|
(4,353
|
)
|
|
|
|
|
|
|
|
|
|
|
|
PROVISION FOR INCOME TAXES
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
NET LOSS
|
|
|
(7,215
|
)
|
|
|
(4,353
|
)
|
|
|
|
|
|
|
|
|
|
|
|
PREFERRED STOCK DIVIDENDS
|
|
|
(3,175
|
)
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
NET LOSS ATTRIBUTABLE TO COMMON STOCKHOLDERS
|
|
$
|
(10,390
|
)
|
|
$
|
(4,353
|
)
|
|
|
|
|
|
|
|
|
|
|
BASIC AND DILUTED NET LOSS PER SHARE ATTRIBUTABLE
TO COMMON STOCKHOLDERS
|
|
$
|
(0.16
|
)
|
|
$
|
(0.08
|
)
|
|
|
|
|
|
|
|
|
|
|
|
WEIGHTED AVERAGE SHARES OUTSTANDING
|
|
|
65,884
|
|
|
|
57,429
|
|
|
|
|
|
|
|
|
|
|
|
The accompanying notes are an integral part of these consolidated statements.
|
|
|
|
|
|
|
|
|
|
|
GUIDED THERAPEUTICS, INC. AND SUBSIDIARY
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’
EQUITY (DEFICIT)
FOR THE YEARS ENDED DECEMBER 31, 2013 AND
2012
(In Thousands)
|
|
|
Preferred Stock
Series B
|
Common Stock
|
|
Additional
Paid-In
|
|
Treasury
|
|
Accumulated
|
|
Non-
Controlling
|
|
|
|
|
|
Shares
|
|
Amount
|
|
Shares
|
|
Amount
|
|
Capital
|
|
Stock
|
|
Deficit
|
|
Interest
|
|
TOTAL
|
|
BALANCE, January 1, 2012
|
|
|
—
|
|
|
$
|
—
|
|
|
|
52,211
|
|
|
$
|
52
|
|
|
$
|
86,614
|
|
|
$
|
(104
|
)
|
|
$
|
(85,089
|
)
|
|
$
|
104
|
|
|
$
|
1,577
|
|
|
Issuance of stock
|
|
|
—
|
|
|
|
—
|
|
|
|
195
|
|
|
|
—
|
|
|
|
162
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
162
|
|
|
Exercise of warrants/options
|
|
|
—
|
|
|
|
—
|
|
|
|
9,876
|
|
|
|
10
|
|
|
|
3,092
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,102
|
|
|
Stock-based compensation expense
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
645
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
645
|
|
|
Deemed dividends
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
2,656
|
|
|
|
—
|
|
|
|
(2,656
|
)
|
|
|
—
|
|
|
|
—
|
|
|
Acquisition of minority interest
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
104
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(104
|
)
|
|
|
—
|
|
|
Net Loss
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(4,353
|
)
|
|
|
—
|
|
|
|
(4,353
|
)
|
|
BALANCE, December 31, 2012
|
|
|
|
|
|
$
|
—
|
|
|
|
62,282
|
|
|
$
|
62
|
|
|
$
|
93,273
|
|
|
$
|
(104
|
)
|
|
$
|
(92,098
|
)
|
|
$
|
—
|
|
|
$
|
1,133
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuance of Series B preferred stock
|
|
|
3
|
|
|
|
1,341
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
1,341
|
|
|
Deemed dividends on beneficial conversion feature of preferred stock
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
3,148
|
|
|
|
—
|
|
|
|
(3,148
|
)
|
|
|
—
|
|
|
|
—
|
|
|
Preferred dividends
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(27
|
)
|
|
|
—
|
|
|
|
(27
|
)
|
|
Conversion of preferred stock
|
|
|
(1
|
)
|
|
|
(202
|
)
|
|
|
878
|
|
|
|
1
|
|
|
|
201
|
|
|
|
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Issuance of common stock
|
|
|
—
|
|
|
|
—
|
|
|
|
670
|
|
|
|
1
|
|
|
|
462
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
463
|
|
|
Issuance of stock options
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
126
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
126
|
|
|
Exercise of warrants and options
|
|
|
—
|
|
|
|
—
|
|
|
|
6,649
|
|
|
|
7
|
|
|
|
3,269
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
3,276
|
|
|
Stock-based compensation expense
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
824
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
824
|
|
|
Deemed dividends on replacement of warrants
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
537
|
|
|
|
—
|
|
|
|
(537
|
)
|
|
|
—
|
|
|
|
—
|
|
|
Acquisition of treasury stock
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(28
|
)
|
|
|
—
|
|
|
|
—
|
|
|
|
(28
|
)
|
|
Net Loss
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
(7,215
|
)
|
|
|
—
|
|
|
|
(7,215
|
)
|
|
BALANCE, December 31, 2013
|
|
|
2
|
|
|
$
|
1,139
|
|
|
|
70,479
|
|
|
$
|
71
|
|
|
$
|
101,840
|
|
|
$
|
(132
|
)
|
|
$
|
(103,025
|
)
|
|
$
|
—
|
|
|
$
|
(107
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The accompanying notes are
an integral part of these consolidated statements.
|
GUIDED THERAPEUTICS, INC. AND SUBSIDIARY
|
|
CONSOLIDATED STATEMENTS OF CASH FLOWS
|
|
FOR THE YEARS ENDED DECEMBER 31, 2013 AND 2012
|
|
(In Thousands)
|
|
|
|
2013
|
|
2012
|
|
CASH FLOWS FROM OPERATING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
$
|
(7,215
|
)
|
|
$
|
(4,353
|
)
|
|
Adjustments to reconcile net loss to net cash used in operating activities:
|
|
|
|
|
|
|
|
|
|
Bad debt (recovery) expense
|
|
|
7
|
|
|
|
(3
|
)
|
|
Depreciation
|
|
|
461
|
|
|
|
361
|
|
|
Stock-based compensation
|
|
|
824
|
|
|
|
645
|
|
|
Change in fair value of warrants
|
|
|
674
|
|
|
|
—
|
|
|
Changes in operating assets and liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(33
|
)
|
|
|
13
|
|
|
Inventory
|
|
|
(669
|
)
|
|
|
(4
|
)
|
|
Other current assets
|
|
|
97
|
|
|
|
(144
|
)
|
|
Other assets
|
|
|
(25
|
)
|
|
|
55
|
|
|
Accounts payable
|
|
|
126
|
|
|
|
(337
|
)
|
|
Deferred revenue
|
|
|
(26
|
)
|
|
|
(413
|
)
|
|
Accrued liabilities
|
|
|
223
|
|
|
|
513
|
|
|
Total adjustments
|
|
|
1,659
|
|
|
|
299
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in operating activities
|
|
|
(5,556
|
)
|
|
|
(3,666
|
)
|
|
|
|
|
|
|
|
|
|
|
|
CASH FLOWS FROM INVESTING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
Additions to fixed assets
|
|
|
(107
|
)
|
|
|
(552
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(107
|
)
|
|
|
(552
|
)
|
|
|
|
|
|
|
|
|
|
|
|
CASH FLOWS FROM FINANCING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
Net proceeds from issuance of preferred stock and warrants
|
|
|
2,214
|
|
|
|
—
|
|
|
Proceeds from debt financing
|
|
|
115
|
|
|
|
86
|
|
|
Payments made on notes payable
|
|
|
(374
|
)
|
|
|
(125
|
)
|
|
Proceeds from options and warrants exercised
|
|
|
3,276
|
|
|
|
3,102
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by financing activities
|
|
|
5,231
|
|
|
|
3,063
|
|
|
|
|
|
|
|
|
|
|
|
|
NET CHANGE IN CASH AND CASH EQUIVALENTS
|
|
|
(432
|
)
|
|
|
(1,155
|
)
|
|
|
|
|
|
|
|
|
|
|
|
CASH AND CASH EQUIVALENTS, beginning of year
|
|
|
1,045
|
|
|
|
2,200
|
|
|
|
|
|
|
|
|
|
|
|
|
CASH AND CASH EQUIVALENTS, end of year
|
|
$
|
613
|
|
|
$
|
1,045
|
|
|
|
|
|
|
|
|
|
|
|
|
SUPPLEMENTAL SCHEDULE OF:
|
|
|
|
|
|
|
|
|
|
Cash paid for:
|
|
|
|
|
|
|
|
|
|
Interest
|
|
$
|
31
|
|
|
$
|
48
|
|
|
NONCASH INVESTING AND FINANCING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
Acquisition of minority interest
|
|
$
|
—
|
|
|
$
|
104
|
|
|
Conversion of accrued expenses into common stock / options
|
|
$
|
126
|
|
|
$
|
162
|
|
|
Purchase of fixed assets by issuing notes payable
|
|
$
|
—
|
|
|
$
|
50
|
|
|
Issuance of common stock as board compensation
|
|
$
|
463
|
|
|
$
|
—
|
|
|
Deemed dividends in the form of warrants to purchase common stock.
|
|
$
|
537
|
|
|
$
|
2,656
|
|
|
Deemed dividends on preferred stock
|
|
$
|
3,148
|
|
|
$
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The accompanying notes are
an integral part of these consolidated statements.
GUIDED THERAPEUTICS, INC. AND SUBSIDIARY
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2013 AND 2012
1. Organization, Background, and Basis of Presentation
Guided Therapeutics, Inc. (formerly SpectRx,
Inc.), together with its wholly owned subsidiary, InterScan, Inc. (formerly Guided Therapeutics, Inc.), collectively referred to
herein as the “Company”, is a medical technology company focused on developing innovative medical devices that have
the potential to improve healthcare. The Company’s primary focus is the development of its LuViva™ non-invasive cervical
cancer detection device and extension of its cancer detection technology into other cancers, including esophageal. The Company’s
technology, including products in research and development, primarily relates to biophotonics technology for the non-invasive detection
of cancers.
Basis of Presentation
All information and footnote disclosures included
in the consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the
United States.
The Company’s prospects must be considered
in light of the substantial risks, expenses and difficulties encountered by entrants into the medical device industry. This industry
is characterized by an increasing number of participants, intense competition and a high failure rate. The Company has experienced
net losses since its inception and, as of December 31, 2013, it had an accumulated deficit of approximately $103.0 million. Through
December 31, 2013, the Company has devoted substantial resources to research and development efforts. The Company first generated
revenue from product sales in 1998, but does not have significant experience in manufacturing, marketing or selling its products.
The Company’s development efforts may not result in commercially viable products and it may not be successful in introducing
its products. Moreover, required regulatory clearances or approvals may not be obtained. The Company’s products may not ever
gain market acceptance and the Company may not ever achieve levels of revenue to sustain further development costs and support
ongoing operations or achieve profitability. The development and commercialization of the Company’s products will require
substantial development, regulatory, sales and marketing, manufacturing and other expenditures. The Company expects operating losses
to continue through the foreseeable future as it continues to expend substantial resources to complete development of its products,
obtain regulatory clearances or approvals and conduct further research and development.
Going Concern
The Company’s consolidated financial
statements have been prepared and presented on a basis assuming it will continue as a going concern. The factors below raise substantial
doubt about the Company’s ability to continue as a going concern. The financial statements do not include any adjustments
that might be necessary from the outcome of this uncertainty. Notwithstanding the foregoing, the Company believes it has made progress
in recent years in stabilizing its financial situation by execution of multiyear contracts from Konica Minolta Opto, Inc., a subsidiary
of Konica Minolta, Inc., a Japanese corporation based in Tokyo (“Konica Minolta”) and grants from the National Cancer
Institute (“NCI”), while at the same time simplifying its capital structure and significantly reducing debt. However,
the Company has replaced its prior agreements with Konica Minolta with a new licensing agreement, and therefore will no longer
receive direct payments from Konica Minolta, and will have to pay a royalty to Konica Minolta should the Company sell any products
licensed from Konica Minolta.
At December 31, 2013, the Company had working
capital of approximately $268,000, accumulated deficit of $103.0 million, and incurred a net loss of $7.2 million for the year
then ended. Stockholders’ deficit totaled approximately $107,000 at December 31, 2013, primarily due to recurring net losses
from operations, deemed dividends on warrants and preferred stock, offset by proceeds from the exercise of options and warrants
and proceeds from sales of stock.
The Company’s
capital-raising efforts are ongoing. If sufficient capital cannot be raised by the end of the second quarter of 2014, the Company
has plans to curtail operations by reducing discretionary spending and staffing levels, and attempting to operate by only pursuing
activities for which it has external financial support and additional NCI, NHI or other grant funding. However, there can be no
assurance that such external financial support will be sufficient to maintain even limited operations or that the Company will
be able to raise additional funds on acceptable terms, or at all. In such a case, the Company might be required to enter into unfavorable
agreements or, if that is not possible, be unable to continue operations, and to the extent practicable, liquidate and/or file
for bankruptcy protection.
The Company had
warrants exercisable for approximately 11.3 million shares of its common stock outstanding at December 31, 2013, with exercise
prices of $0.40, $0.80 and $1.08 per share. Exercises of these warrants would generate a total of approximately $7.6 million in
cash, assuming full exercise, although the Company cannot be assured that holders will exercise any warrants. Management may obtain
additional funds through the private sale of preferred stock or debt securities, public and private sales of common stock, and
grants, if available.
Assuming the Company
receives FDA approval for its LuViva cervical cancer detection device in 2014, the Company currently anticipates an early 2015
product launch in the United States. Product launch outside the United States began in the second half of 2013.
2. Summary of Significant Accounting Policies
Use of Estimates
The preparation of financial statements in
conformity with accounting principles generally accepted in the United States requires management to make estimates and assumptions
that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the
financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ
from those estimates. Significant areas where estimates are used include the allowance for doubtful accounts, inventory valuation
and input variables for Black-Scholes calculations.
Principles of Consolidation
The accompanying consolidated financial statements
include the accounts of Guided Therapeutics, Inc. and its wholly owned subsidiary. As disclosed in Note 4, the Company purchased
the remaining 49% interest in its subsidiary during December 2012.
Cash Equivalents
The Company considers all highly liquid investments
with an original maturity of three months or less when purchased to be a cash equivalent.
Concentrations of Credit Risk
The Company, from time to time during the years
covered by these consolidated financial statements, may have bank balances in excess of its insured limits. Management has deemed
this a normal business risk.
Inventory Valuation
All inventories are stated at lower of cost
or market, with cost determined substantially on a “first-in, first-out” basis. Selling, general, and administrative
expenses are not inventoried, but are charged to expense when purchased. At December 31, 2013 and December 31, 2012, our inventories
were as follows (in thousands):
|
|
|
December 31,
2013
|
|
December 31,
2012
|
|
Raw materials
|
|
$
|
1,013
|
|
|
$
|
518
|
|
|
Work in process
|
|
|
268
|
|
|
|
21
|
|
|
Finished goods
|
|
|
96
|
|
|
|
37
|
|
|
Inventory reserve
|
|
|
(184
|
)
|
|
|
(52
|
)
|
|
Total
|
|
$
|
1,193
|
|
|
$
|
524
|
|
Property and Equipment
Property and equipment are recorded at cost.
Depreciation is computed using the straight-line method over estimated useful lives of three to seven years. Leasehold improvements
are depreciated at the shorter of the useful life of the asset or the remaining lease term. Depreciation expense is included in
general and administrative expense on the statement of operations. Expenditures for repairs and maintenance are expensed as incurred.
Property and equipment are summarized as follows at December 31, 2013 and 2012 (in thousands):
|
|
|
Year Ended
December 31,
|
|
|
|
2013
|
|
2012
|
|
Equipment
|
|
$
|
1,277
|
|
|
$
|
1,196
|
|
|
Software
|
|
|
737
|
|
|
|
730
|
|
|
Furniture and fixtures
|
|
|
124
|
|
|
|
124
|
|
|
Leasehold Improvement
|
|
|
189
|
|
|
|
170
|
|
|
|
|
|
2,327
|
|
|
|
2,220
|
|
|
Less accumulated depreciation
|
|
|
(1,407
|
)
|
|
|
(946
|
)
|
|
Total
|
|
$
|
920
|
|
|
$
|
1,274
|
|
Patent Costs (Principally Legal Fees)
Costs incurred in filing, prosecuting, and
maintaining patents are recurring, and expensed as incurred. Maintaining patents are expensed as incurred as the Company has not
yet received FDA approval and recovery of these costs is uncertain. Such costs aggregated approximately $75,000 and $46,000 in
2013 and 2012, respectively.
Accounts Receivable
The Company performs periodic credit evaluations
of its customers’ financial conditions and generally does not require collateral. The Company reviews all outstanding accounts
receivable for collectability on a quarterly basis. An allowance for doubtful accounts is recorded for any amounts deemed uncollectable.
The Company does not accrue interest receivable on past due accounts receivable.
Capitalized Costs of Internally Developed
Software
Costs of producing product masters incurred
subsequent to establishing technological feasibility are capitalized. Those costs include coding and testing performed subsequent
to establishing technological feasibility.
Software production costs for computer
software that is to be used as an integral part of a product or process are not capitalized until technological feasibility has
been established for the software and all research and development activities for the other components of the product have been
completed.
Capitalization of computer software costs ceases
when the product is available for general release to customers. Costs of maintenance and customer support are charged to expense
when related revenue is recognized or when those costs are incurred, whichever occurs first.
Costs of internally developed software are
capitalized during the development stage of the software. The cost will be transferred to property and equipment and will be depreciated
over the expected life of the software, which is estimated to be three years once the software becomes functional.
Other Assets
Other assets primarily consist of long-term
deposits for various tooling projects that are being constructed for the Company. At December 31, 2013 and 2012, such balances
were approximately $326,000 and $283,000, respectively.
Accrued Liabilities
Accrued liabilities are summarized as follows
at December 31, 2013 and 2012 (in thousands):
|
|
|
As of
December 31,
|
|
|
|
2013
|
|
2012
|
|
Accrued compensation
|
|
$
|
426
|
|
|
$
|
706
|
|
|
Accrued professional fees
|
|
|
116
|
|
|
|
191
|
|
|
Deferred rent
|
|
|
68
|
|
|
|
77
|
|
|
Other accrued expenses
|
|
|
113
|
|
|
|
64
|
|
|
Total
|
|
$
|
723
|
|
|
$
|
1,038
|
|
Revenue Recognition
Revenue from the sale of the Company’s
products is recognized upon shipment of such products to its customers. The Company recognizes revenue from contracts on a straight
line basis, over the terms of the contracts. The Company recognizes revenue from grants based on the grant agreements, at the time
the expenses are incurred.
Significant
Customers
In 2013 and 2012, the majority of the Company’s
revenues were from three and two customers, respectively. Revenue from these customers totaled approximately $653,000 or 65% and
approximately $2.9 million or 85% of total revenue for the year ended December 31, 2013 and 2012, respectively. Accounts receivable
due from the customers represents 27% and 48% as of December 31, 2013 and 2012, respectively.
Deferred Revenue
The Company defers payments received
as revenue until earned based on the related contracts on a straight line basis, over the terms of the contract.
Research and Development
Research and development expenses consist of
expenditures for research conducted by the Company and payments made under contracts with consultants or other outside parties
and costs associated with internal and contracted clinical trials. All research and development costs are expensed as incurred.
Income Taxes
The Company uses the liability method of accounting
for income taxes. Under this method, deferred tax assets and liabilities are determined based on differences between the financial
reporting and tax bases of assets and liabilities and are measured using the enacted tax rates and laws that will be in effect
when the differences are expected to reverse. Management provides valuation allowances against the deferred tax assets for amounts
that are not considered more likely than not to be realized.
Uncertain Tax Positions
Effective January 1, 2007 the Company adopted
ASC guidance regarding accounting for uncertainty in income taxes. This guidance clarifies the accounting for income taxes by prescribing
the minimum recognition threshold an income tax position is required to meet before being recognized in the financial statements
and applies to all income tax positions. Each income tax position is assessed using a two-step process. A determination is first
made as to whether it is more likely than not that the income tax position will be sustained, based upon technical merits, upon
examination by the taxing authorities. If the income tax position is expected to meet the more likely than not criteria, the benefit
recorded in the financial statements equals the largest amount that is greater than 50% likely to be realized upon its ultimate
settlement. At December 31, 2013 and 2012, there were no uncertain tax positions.
The Company
is current with its federal and applicable state tax returns filings. Although we have been experiencing recurring losses, we are
obligated to file tax returns for compliance with Internal Revenue Service (“IRS”) regulations and that of applicable
state jurisdictions. As of December 31, 2013, the Company has approximately $59.8 million of net operating loss eligible to be
carried forward for tax purposes at federal and applicable states level.
None of the Company’s federal or state
income tax returns are currently under examination by the IRS or state authorities. However, fiscal years 2010 and later remain
subject to examination by the IRS and applicable states.
Warrants
The Company has issued warrants, which allow
the warrant holder to purchase one share of stock at a specified price for a specified period of time. The Company records equity
instruments including warrants issued to non-employees based on the fair value at the date of issue. The fair value of warrants
classified as equity instruments at the date of issuance is estimated using the Black-Scholes Model. The fair value of warrants
classified as liabilities at the date of issuance is estimated using the Monte Carlo Simulation model.
Stock Based Compensation
The Company records
compensation expense related to options granted to non-employees based on the fair value of the award.
Compensation cost is recorded as earned for
all unvested stock options outstanding at the beginning of the first year based upon the grant date fair value estimates, and for
compensation cost for all share-based payments granted or modified subsequently based on fair value estimates.
For the years ended December 31, 2013 and 2012,
share-based compensation for options attributable to employees and officers were approximately $824,000 and $645,000, respectively.
These amounts have been included in the Company’s statements of operations. Compensation costs for stock options which vest
over time are recognized over the vesting period. As of December 31, 2013, the Company had approximately $865,000 of unrecognized
compensation costs related to granted stock options to be recognized over the remaining vesting period of approximately three years.
3. FAIR VALUE OF FINANCIAL INSTRUMENTS
The guidance for fair value measurements, ASC820,
Fair Value Measurements and Disclosures
, establishes the authoritative definition of fair value, sets out a framework for
measuring fair value, and outlines the required disclosures regarding fair value measurements. Fair value is the price that would
be received to sell an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the
asset or liability in an orderly transaction between market participants at the measurement date. The Company uses a three-tier
fair value hierarchy based upon observable and non-observable inputs as follow:
|
|
·
|
Level 1 – Quoted market prices in active markets for identical assets and liabilities;
|
|
|
·
|
Level 2 – Inputs, other than level 1 inputs, either directly or indirectly observable;
and
|
|
|
·
|
Level 3 – Unobservable inputs developed using internal estimates and assumptions (there
is little or no market date) which reflect those that market participants would use.
|
The Company records its derivative activities
at fair value, which consisted of warrants as of December 31, 2013. The fair value of the warrants was estimated using the Monte
Carlo Simulation model. Gains and losses from derivative contracts are included in net gain (loss) from derivative contracts in
the statement of operations. The fair value of the Company’s derivative warrants is classified as a Level 3 measurement,
since unobservable inputs are used in the valuation.
The following table presents the fair value
for those liabilities measured on a recurring basis as of December 31, 2013:
FAIR VALUE MEASUREMENTS ( In Thousands)
|
|
Description
|
|
|
|
Level 1
|
|
|
|
Level 2
|
|
|
|
Level 3
|
|
|
|
Total
|
|
|
|
Asset/(Liability)
Total
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Warrants
|
|
|
$
|
—
|
|
|
$
|
—
|
|
|
$
|
(1,548
|
)
|
|
$
|
(1,548
|
)
|
|
$
|
(1,548
|
)
|
|
|
There were neither derivatives liabilities nor valuations of financial
liabilities at December 31, 2012.
4. Stockholders’ Equity
Common Stock
The Company has authorized 145 million shares
of common stock with $0.001 par value, of which 70.5 million were issued and outstanding as of December 31, 2013. For the year
ended December 31, 2012, there were 145 million authorized shares of common stock, of which 62.3 million were issued and outstanding.
In December 2012, the Company entered into
an agreement to purchase the remaining 49% interest in InterScan, Inc. In exchange, the Company agreed to issue to the seller warrants
equal to 49% of the fair value of InterScan, Inc., as determined by a third party. The agreement established a minimum value purchase
price of $147,000, or approximately 198,000 warrants, based upon the closing stock price at the date of the agreement, and a maximum
purchase price of 2,500,000 warrants. The agreement required the seller to exercise one quarter of his outstanding warrants, subject
to a minimum of $450,000 in warrant exercise payments, prior to March 1, 2013. The seller exercised all required warrants in accordance
with the agreement. The Company issued 439,883 warrants to purchase the Company’s common stock at $0.68 per share to the
seller, which will expire on March 31, 2016.
Preferred Stock; Series B Convertible Preferred Stock
The Company has authorized 5,000,000 shares
of preferred stock with a $.001 par value. The board of directors has the authority to issue these shares and to set dividends,
voting and conversion rights, redemption provisions, liquidation preferences, and other rights and restrictions. The board of directors
designated 525,000 shares of preferred stock as redeemable convertible preferred stock, none of which remain outstanding, and 3,000
shares of preferred stock as Series B Preferred Stock, of which 2,147 shares were issued and outstanding as of December 31, 2013.
Pursuant to the terms
of the Series B Preferred Stock set forth in the Certificate of Designations, Preferences and Rights designating the Preferred
Stock (the “Preferred Stock Designation”), shares of Series B Preferred Stock are convertible into common stock by
their holder at any time, and will be mandatorily convertible upon the achievement of certain conditions, including the receipt
of certain approvals from the U.S. Food and Drug Administration and the achievement by the Company of specified average trading
prices and volumes for the common stock. The original conversion price was $0.68 per share, such that each share of Preferred Stock
would convert into 1,471 shares of common stock, subject to customary adjustments, including any accrued but unpaid dividends and
pursuant to certain anti-dilution provisions, as set forth in the Preferred Stock Designation. As a result
of anti-dilution provisions,
the current conversion price is set at $0.40 per share, such that each share of Preferred Stock would convert into 2,500 shares
of common stock.
Holders of the Series
B Preferred Stock are entitled to quarterly dividends at an annual rate of 5.0%, for the quarter ended December 31, 2013, and at
an annual rate of 10% thereafter, in each case, payable in cash or, subject to certain conditions, common stock, at the Company’s
option. Accrued dividends totaled approximately $27,000 at December 31, 2013. Each share of Series B Preferred Stock is entitled
to a number of votes equal to the number of shares of common stock into which the Series B Preferred Stock is convertible. As long
as shares of the Series B Preferred Stock are outstanding, and until the receipt of certain approvals from the U.S. Food and Drug
Administration and the achievement by the Company of specified average trading prices and volumes for the common stock, the Company
may not incur indebtedness for borrowed money secured by the Company’s intellectual property or in excess of $2.0 million
without the prior consent of the holders of two-thirds of the outstanding shares of Series B Preferred Stock. The Company may redeem
the Series B Preferred Stock after the second anniversary of issuance, subject to certain conditions. Upon the Company’s
liquidation or sale to or merger with another corporation, each share of Series B Preferred Stock will be entitled to a liquidation
preference of $1,000 per share, plus any accrued but unpaid dividends.
The Series B Preferred
Stock was issued with Tranche A warrants to purchase 1,858,089 shares of common stock and Tranche B warrants purchasing 1,858,088
shares of common stock, both at an exercise price of $1.08 per share. Pursuant to the terms of the Tranche B warrants, their exercise
price will be reduced, and the number of shares of common stock into which those warrants are exercisable will be increased, if
the Company issues shares at a price below the then-current exercise price. The exercise price of Tranche B warrants is currently
$0.40 per share, convertible into 5,016,840 shares of common stock. As a result of these provisions, the Company is required to
account for the warrants as a liability recorded at fair value each period. The Company values the warrants using a Monte Carlo
Simulation model. Of the $2.6 million in proceeds from issuance of the Series B Preferred Stock, the Company originally allocated
$873,000 to the fair value of the warrants. At December 31, 2013, the fair value of these warrants was approximately $1.5 million.
Stock Options
Under the Company’s
1995 Stock Plan (the “Plan”), a total of 6,724,027 shares remained available at December 31, 2013 and 6,531,192 shares
were subject to stock options outstanding as of that date, bringing the total number of shares subject to stock options outstanding
and those remaining available for issue to 13,255,219 shares of common stock as of December 31, 2013. The Plan allows the issuance
of incentive stock options, nonqualified stock options, and stock purchase rights. The exercise price of options is determined
by the Company’s board of directors, but incentive stock options must be granted at an exercise price equal to the fair market
value of the Company’s common stock as of the grant date. Options historically granted have generally become exercisable
over four years and expire ten years from the date of grant.
The fair value of stock options granted in
2013 and 2012 were estimated using the Black-Scholes option pricing model. A summary of the assumptions used in determining the
fair value of options follows:
|
|
|
2013
|
|
2012
|
|
Expected volatility
|
|
|
174
|
%
|
|
|
141
|
%
|
|
Expected option life in years
|
|
|
10.0
|
|
|
|
10.0
|
|
|
Expected dividend yield
|
|
|
0.00
|
%
|
|
|
0.00
|
%
|
|
Risk-free interest rate
|
|
|
1.87
|
%
|
|
|
1.84
|
%
|
|
Weighted average fair value per option at grant date
|
|
$
|
0.69
|
|
|
$
|
0.76
|
|
Application of the Black-Scholes option
pricing model involves assumptions that are judgmental and affect compensation expense. Historical information is the primary basis
for the selection of expected volatility, expected option life and expected dividend yield. Expected volatility is based on the
most recent historical period equal to the
expected life of the option. The risk-free
interest rate is based on yields of U.S. Treasury zero-coupon issues with a term equal to the expected life of the option on the
date the stock options were granted.
Stock option activity for each of the two years ended December 31
is as follows:
|
|
2013
|
2012
|
|
|
|
Weighted
Average
Exercise
|
|
Weighted
Average
Exercise
|
|
|
Shares
|
Price
|
Shares
|
Price
|
|
Outstanding at beginning of year
|
6,463,206
|
$0.67
|
6,862,167
|
$0.70
|
|
Options granted
|
977,276
|
$0.50
|
96,500
|
$0.79
|
|
Options exercised
|
(580,540)
|
$0.31
|
(326,461)
|
$0.28
|
|
Options expired/forfeited
|
(328,750)
|
$1.15
|
(169,000)
|
$2.60
|
|
Outstanding at end of year
|
6,531,192
|
$0.66
|
6,463,206
|
$0.67
|
|
Options vested and exercisable at year-end
|
5,463,963
|
$0.58
|
4,373,807
|
$0.50
|
|
Options available for grant at year-end
|
6,724,027
|
|
6,792,013
|
|
|
Aggregate intrinsic value – options exercised
|
$ 236,059
|
|
$ 93,088
|
|
|
Aggregate intrinsic value – options outstanding
|
$ 625,412
|
|
$ 1,332,965
|
|
|
Aggregate intrinsic value – options vested and
exercisable
|
$ 612,946
|
|
$1,208,831
|
|
|
Options unvested, balance at beginning of year (1)
|
1,819,087
|
$1.18
|
-
|
-
|
|
Options granted (1)
|
977,276
|
$0.50
|
-
|
-
|
|
Vested (1)
|
(1,582,034)
|
$0.80
|
-
|
-
|
|
Cancelled/Forfeited
|
(147,100
|
$1.22
|
-
|
-
|
|
Balance, end of period (1)
|
1,067,229
|
$1.12
|
-
|
-
|
__________________
|
|
(1)
|
Includes awards not captured in valuation fragments
|
The Company estimates the fair value of stock
options using a Black-Scholes valuation model. Key input assumptions used to estimate the fair value of stock options include the
expected term, expected volatility of the Company’s common stock, the risk free interest rate, option forfeiture rates, and
dividends, if any. The expected term of the options is based upon the historical term until exercise or expiration of all granted
options. The expected volatility is derived from the historical volatility of the Company’s stock on the OTCQB marketplace
for a period that matches the expected term of the option. The risk-free interest rate is the constant maturity rate published
by the U.S. Federal Reserve Board that corresponds to the expected term of the option.
Warrants
In July 2012, the Company completed a warrant
exchange program, pursuant to which it exchanged warrants exercisable for a total of 15,941,640 shares of common stock, or 56.29%
of the warrants eligible to participate, for three classes of new warrants. These exchanges resulted in a deemed dividend of approximately
$2.66 million, reflected as a non-cash disclosure in this financial statement of cash flows. The first class of new warrants expired
on September 17, 2012 and carried an exercise price of $0.40, $0.45 or $0.50, depending on the date exercised. The second class
of new warrants carries a one-year extension from the original expiration date and is exercisable at $0.65. The third class of
new warrants carries a two-year extension from the original expiration date and is exercisable at $0.80.
In November 2013, the Company completed a warrant
exchange program, pursuant to which it exchanged warrants exercisable for a total of 3,560,869 shares of common stock, or 99% of
the warrants eligible to participate. These exchanges resulted in a deemed dividend of approximately $537,000, reflected as a non-cash
disclosure in this financial statement of cash flows.
The following table summarizes transactions
involving the Company’s outstanding warrants to purchase common stock for the year ended December 31, 2013:
|
|
|
Warrants
(Underlying Shares)
|
|
Outstanding, January 1, 2013
|
|
|
20,801,512
|
|
|
Issuances
|
|
|
6,874,929
|
|
|
Canceled / Expired
|
|
|
(10,349,659
|
)
|
|
Exercised
|
|
|
(6,067,843
|
)
|
|
Outstanding, December 31, 2013
|
|
|
11,258,939
|
|
The Company had the following shares reserved for the warrants as
of December 31, 2013:
Warrants
(Underlying Shares)
|
|
Exercise Price
|
|
Expiration Date
|
|
29,656
|
(1)
|
$0.65 per share
|
|
March 1, 2014
|
|
471,856
|
(1)
|
$0.80 per share
|
|
July 26, 2014
|
|
3,590,522
|
(1)
|
$0.80 per share
|
|
March 1, 2015
|
|
6,790
|
(2)
|
$1.01 per share
|
|
September 10, 2015
|
|
439,883
|
(3)
|
$0.68 per share
|
|
March 31, 2016
|
|
285,186
|
(4)
|
$1.05 per share
|
|
November 20, 2016
|
|
1,858,089
|
(5)
|
$1.08 per share
|
|
May 23, 2018
|
|
5,016,840
|
(6)
|
$0.40 per share
|
|
May 23, 2018
|
__________
|
|
(1)
|
Consists of outstanding warrants issued in connection with a warrant
exchange program in June 2012.
|
|
|
(2)
|
Consists of outstanding warrants issued in conjunction with a private
placement on September 10, 2010.
|
|
|
(3)
|
Consists of outstanding warrants issued in conjunction with a buy
back of our minority interest in December 2012, which were issued in February 2014.
|
|
|
(4)
|
Consists of outstanding warrants issued in conjunction with a private
placement on November 21, 2011.
|
|
|
(5)
|
Consists of outstanding warrants issued in conjunction with a private
placement on May 24, 2013.
|
|
|
(6)
|
Consists of outstanding warrants issued in conjunction with a private
placement on May 24, 2013. Underlying shares increased from 1,858,089 to 5,016,840, and exercise price decreased from $1.08 per
share to $0.40 per share, pursuant to the terms of the warrants, as a result of the 2013 warrant exchange program.
|
5. Income Taxes
The Company has incurred net operating losses
(“NOLs”) since inception. As of December 31, 2013, the Company had NOL carryforwards available through 2033 of
approximately $59.8 million to offset its future income tax liability. The NOL carryforwards began to expire in 2008. The Company
has recorded a valuation allowance for all deferred tax assets related to the NOLs. Utilization of existing NOL carry forwards
may be limited in future years based on significant ownership changes. The Company is in the process of analyzing its NOLs and
has not determined if it is subject to any restrictions in the Internal Revenue Code that could limit the future use of NOL.
Components of deferred taxes are as follows
at December 31 (in thousands):
|
|
|
2013
|
|
2012
|
|
Deferred tax assets:
|
|
$
|
287
|
|
|
$
|
277
|
|
|
Net operating loss carry forwards
|
|
|
22,737
|
|
|
|
23,474
|
|
|
Deferred tax liabilities:
|
|
|
|
|
|
|
|
|
|
Intangible assets and other
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
23,025
|
|
|
|
23,751
|
|
|
Valuation allowance
|
|
|
(23,025
|
)
|
|
|
(23,751
|
)
|
|
|
|
$
|
0
|
|
|
$
|
0
|
|
The following is a summary of the items that caused recorded income
taxes to differ from taxes computed using the statutory federal income tax rate for the years ended December 31:
|
|
|
2013
|
|
2012
|
|
Statutory federal tax rate
|
|
|
34
|
%
|
|
|
34
|
%
|
|
State taxes, net of federal benefit
|
|
|
4
|
|
|
|
4
|
|
|
Nondeductible expenses
|
|
|
—
|
|
|
|
—
|
|
|
Valuation allowance
|
|
|
(38
|
)
|
|
|
(38
|
)
|
|
|
|
|
0
|
%
|
|
|
0
|
%
|
6. Commitments and Contingencies
Operating Leases
In December 2009, the Company moved its offices,
which comprise its administrative, research and development, marketing and production facilities to 5835 Peachtree Corners East,
Suite D, Norcross, Georgia 30092. The Company leases approximately 23,000 square feet under a lease that expires in June 2017.
The fixed monthly lease expense is approximately $15,000 plus common charges. The Company also leases office and automotive equipment
under operating lease agreements with monthly payments ranging from $275 to $1,960. These leases expire at various dates through
April 2016. Future minimum rental payments at December 31, 2013 under non-cancellable operating leases for office space and equipment
are as follows (in thousands):
|
Year
|
|
Amount (,000)
|
|
2014
|
|
$ 207
|
|
2015
|
|
211
|
|
2016
|
|
201
|
|
2017
|
|
98
|
|
Total
|
|
$ 717
|
Rental expense was approximately $170,000 in 2013 and 2012.
Litigation and Claims
For the years ended December 31, 2013 and 2012, there was no accrual
needed for any potential losses related to pending litigation.
Contracts
Under the Company’s
prior collaboration agreements with Konica Minolta related to the development of lung and esophageal cancer detection products,
the Company received approximately $400,000 and $1.3 million, respectively, in 2012. In February 2013, the Company replaced its
existing agreements with Konica Minolta with a new agreement, pursuant to which, subject to the payment of a nominal license fee
due upon FDA approval, Konica Minolta has granted the Company a five-year, world-wide, non-transferable and non-exclusive right
and license to manufacture and to develop a non-invasive esophageal cancer detection product from Konica Minolta and based on the
Company’s biophotonic technology platform. The license permits the Company to use certain related intellectual property of
Konica Minolta. In return for the license, the Company has agreed to pay Konica Minolta a royalty for each licensed product the
Company sells.
7. License and Technology Agreements
As part of the Company’s efforts to conduct
research and development activities and to commercialize potential products, the Company, from time to time, enters into agreements
with certain organizations and individuals that further those efforts but also obligate the Company to make future minimum payments
or to remit royalties ranging from 1% to 3% of revenue from the sale of commercial products developed from the research. The Company
generally is required to make minimum royalty
payments for the exclusive license to develop certain technology.
8. Notes Payable
Short Term Notes Payable
At December 31, 2012, the Company maintained
a note payable to IQMS, an enterprise resources planning software provider, of approximately $34,000, as well as a note to Premium
Assignment Corporation, an insurance premium financing company, of approximately $33,000. These notes were 8 and 12 month, straight-line
amortizing loans dated June 29, 2012 and July 4, 2012, respectively, with monthly principal and interest payments of approximately
$4,300 and $11,000 per month, respectively. The notes carried annual interest rates ranging between 5-6%. The Premium Assignment
Corporate note was paid in full during the quarter ended March 31, 2013. The IQMS note was paid in full during the quarter ended
September 30, 2013.
At December 31, 2013, the Company maintained
an additional note payable to Premium Assignment Corporation of approximately $35,000. This note is an 8 month, straight-line amortizing
loan dated July 4, 2013 with monthly principal and interest payments of approximately $12,000 per month. The note carries an annual
interest rate of 5.34%.
Notes Payable
At December 31, 2012, the Company was past
due on two short-term notes totaling approximately $419,000 of principal and accrued interest. Interest charged on these notes
prior to amendment ranged between 15-18%. On February 27, 2013, the Company renegotiated one of the two past due notes. The new
note accrued interest at 6% and was paid in full during the quarter ended June 30, 2013. On April 16, 2013, the Company renegotiated
the other note. The renegotiated note accrues interest at 9.0%, requires monthly payments of $10,000, including interest, and matures
November 2015. The balance due on this note was approximately $208,000 at December 31, 2013, of which $103,000 is payable during
the year ending December 31, 2014 and $105,000 is payable during the year ending December 31, 2015.
9.
Related Party Transactions
None
10. Valuation and Qualifying Accounts
Allowance for Doubtful Accounts
The Company has the following allowances for doubtful accounts (in
thousands):
|
|
|
Year Ended
December 31,
|
|
|
|
2013
|
|
2012
|
|
Beginning balance
|
|
$
|
12
|
|
|
$
|
20
|
|
|
Additions / (Adjustments)
|
|
|
6
|
|
|
|
(8
|
)
|
|
Balance
|
|
$
|
18
|
|
|
$
|
12
|
|
|
|
|
|
|
|
|
|
|
|
Inventory Reserves
The Company has the following reserves for inventory balance (in
thousands):
|
|
|
Year Ended
December 31,
|
|
|
|
2013
|
|
2012
|
|
Beginning balance
|
|
$
|
52
|
|
|
$
|
64
|
|
|
Additions / (Adjustments)
|
|
|
132
|
|
|
|
(12
|
)
|
|
Balance
|
|
$
|
184
|
|
|
$
|
52
|
|
|
|
|
|
|
|
|
|
|
|
11. Loss Per Common Share
Basic net loss per share attributable to common
stockholders amounts are computed by dividing the net loss plus preferred stock dividends and deemed dividends on preferred stock
by the weighted average number of shares outstanding during the period.
On December 17, 2012, the Company entered into
a buy-back agreement with the holder of a 51 percent interest in the Company’s subsidiary, InterScan, Inc., pursuant to which
the original agreement, dated February 28, 2011, was canceled and ownership of InterScan reverted back to the Company. InterScan
is a non-active subsidiary of the Company.
12. Subsequent Events
On January 7, 2014 the Company announced the
appointment of Gene Cartwright, 59, as Chief Executive Officer, effective January 6, 2014. Dr. Cartwright replaced Mark L. Faupel,
who has transitioned to the role of Chief Scientific Officer. In accordance with Dr. Faupel’s employment agreement, all outstanding
unvested stock options became fully vested on January 6, 2014, resulting in compensation expense of approximately $111,000. The
Company also owes Dr. Faupel additional compensation payable of $40,000, as a result of the Company’s employment agreement
with Dr. Cartwright.
Effective January 31, 2014, Ronald W. Allen
resigned from the Board of Directors of the Company.
Between February 1 and March 25, 2014, the
Company received cash advances from certain affiliates totaling about $175,000.
Guided Therapeutics (QB) (USOTC:GTHP)
Historical Stock Chart
From Jun 2024 to Jul 2024
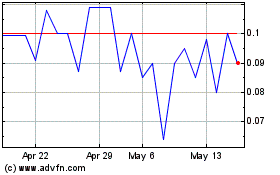
Guided Therapeutics (QB) (USOTC:GTHP)
Historical Stock Chart
From Jul 2023 to Jul 2024