UNITED STATES SECURITIES
AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
Date of report: March 8, 2024
Commission File Number: 001-38974
BIOPHYTIS S.A.
(Translation of registrant’s name into
English)
Stanislas Veillet
Biophytis S.A.
Sorbonne University—BC 9, Bâtiment
A 4ème étage
4 place Jussieu
75005 Paris, France
+33 1 44 27 23 00
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual
reports under cover of Form 20-F or Form 40-F:
x Form 20-F ¨ Form 40-F
Indicate
by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ¨
Indicate
by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ¨
On March 8, 2024, Biophytis S.A. issued a press release announcing
that the company presented the results of its recent clinical trials with RuvembriTM at the Muscular Dystrophy Association
(MDA) Clinical & Scientific Conference, held from 3 to 6 March 2024 in Orlando, USA. A copy of the press release
is attached as Exhibit 99.1 to this Form 6-K.
EXHIBIT LIST
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934,
the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| |
BIOPHYTIS S.A. |
| |
|
|
| Date: March 8, 2024 |
By: |
/s/ Stanislas Veillet |
| |
|
Name: Stanislas Veillet |
| |
|
Title: Chairman and Chief Executive Officer |
Exhibit 99.1

Press
release
Biophytis presented the potential
of RuvembriTM in the treatment of Duchenne Muscular Dystrophy
Paris (France) and Cambridge (Massachusetts,
USA), March 8, 2024 – 07:00am CET – Biophytis SA (Nasdaq CM : BPTS, Euronext Growth Paris : ALBPS), ("Biophytis"
or the "Company"), a clinical-stage biotechnology company specialized in the development of therapeutics that are aimed at slowing
the degenerative processes associated with aging and improving functional outcomes for patients suffering from age-related diseases, presented
the results of its recent clinical trials with RuvembriTM at the Muscular Dystrophy Association (MDA) Clinical &
Scientific Conference, held from 3 to 6 March 2024 in Orlando, USA.
Administered orally, RuvembriTM activates
the MAS receptor, stimulating respiratory and motor functions. The results of the SARA-INT (phase 2) and COVA (phase 3) clinical trials
demonstrated the efficacy and safety of RuvembriTM , respectively in sarcopenic patients and those suffering from severe forms
of COVID-19. The data collected on these two vulnerable populations confirm the potential of RuvembriTM in the treatment of
patients suffering from Duchenne Muscular Dystrophy (DMD).
RuvembriTM has already obtained the
orphan drug designation in Europe and the USA for this indication, which will accelerate clinical development and market authorization
procedures. Biophytis aims to start in 2024 a phase 1-2 clinical trial in non-ambulatory DMD patients suffering from respiratory failure.
Stanislas Veillet, CEO of Biophytis, stated: "Duchenne
Muscular Dystrophy is a severe orphan condition which generally appears between the ages of 2 and 5 years by progressive muscle weakness,
loss of walking capacity and later cardio-respiratory difficulties responsible for the early death of young adults. Despite some recent
advances and the great hope generated by gene therapy, there is no satisfactory treatment today and the medical need is still substantial,
particularly for non-ambulatory patients. The treatment of DMD, the world's most common neuromuscular disease, represents a market of
USD 2.5 billion, which is expected to grow rapidly to USD 4.3 billion by 2029."
The infographic presented at the conference can
be viewed by clicking on this link.
* * * *
About BIOPHYTIS
Biophytis SA is a clinical-stage biotechnology
company specializing in the development of drug candidates for age-related diseases. RuvembriTM, our lead drug candidate,
is a small molecule in development for age-related neuromuscular (sarcopenia and Duchenne muscular dystrophy) and cardiorespiratory (Covid-19)
diseases. Promising clinical results were obtained in the treatment of sarcopenia in an international phase 2 study, enabling the launch
of a phase 3 study in this indication (SARA project). The safety and efficacy of RuvembriTM in the treatment of severe COVID-19
were studied in a positive international phase 2-3 clinical trial (COVA project). A pediatric formulation of RuvembriTM is
currently being developed for the treatment of Duchenne Muscular Dystrophy (DMD, MYODA project). The company is based in Paris, France,
and Cambridge, Massachusetts. The Company's ordinary shares are listed on Euronext Growth (Ticker: ALBPS -ISIN: FR0012816825) and the
ADSs (American Depositary Shares) are listed on Nasdaq Capital Market (Ticker BPTS - ISIN: US09076G1040). For more information, visit
www.biophytis.com

Press
release
About MDA Clinical & Scientific Conference
The MDA Clinical & Scientific Conference
provides a comprehensive exploration of pre-clinical, translational, and clinical research and care for individuals with neuromuscular
disease, with particular attention to some of the changes the field is undergoing in response to the approval of new therapies. MDA is
the #1 voluntary health organization in the United States for people living with muscular dystrophy, ALS, and related neuromuscular diseases.
For over 70 years, MDA has led the way in accelerating research, advancing care, and advocating for the support of families. MDA's mission
is to empower the people they serve to live longer, more independent lives.
Disclaimer
This press release contains forward-looking statements.
Forward-looking statements include all statements that are not historical facts. In some cases, you can identify these forward-looking
statements by the use of words such as "outlook," "believes," "expects," "potential," "continues,"
"may," "will," "should," "could," "seeks," "predicts," "intends,"
"trends," "plans," "estimates," "anticipates" or the negative version of these words or other
comparable words. Such forward- looking statements are based on assumptions that Biophytis considers to be reasonable. However, there
can be no assurance that the statements contained in such forward-looking statements will be verified, which are subject to various risks
and uncertainties. The forward- looking statements contained in this press release are also subject to risks not yet known to Biophytis
or not currently considered material by Biophytis. Accordingly, there are or will be important factors that could cause actual outcomes
or results to differ materially from those indicated in these statements. Please also refer to the "Risk and uncertainties the Company
is to face» section from the Company’s 2022 Financial Report available on BIOPHYTIS website (www.biophytis.com) and as exposed
in the "Risk Factors" section of form 20-F as well as other forms filed with the SEC (Securities and Exchange Commission, USA).
We undertake no obligation to publicly update or review any forward-looking statement, whether as a result of new information, future
developments or otherwise, except as required by law.
Biophytis contacts
Investor relations
Nicolas Fellmann, CFO
Investors@biophytis.com
Media
Antoine Denry: antoine.denry@taddeo.fr –
+33 6 18 07 83 27
Nizar Berrada: nizar.berrada@taddeo.fr – +33 6 38 31
90 50
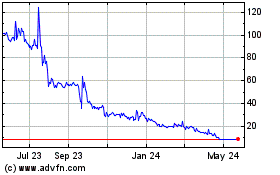
Biophytis (NASDAQ:BPTS)
Historical Stock Chart
From Dec 2024 to Jan 2025
Biophytis (NASDAQ:BPTS)
Historical Stock Chart
From Jan 2024 to Jan 2025