Denali Therapeutics Announces Broad Collaboration with Sanofi to Develop RIPK1 Inhibitors for the Treatment of Neurological a...
01 November 2018 - 5:00PM

Denali Therapeutics Inc. (NASDAQ: DNLI), a biopharmaceutical
company developing a broad portfolio of therapeutic candidates for
neurodegenerative diseases, today announced that it will
collaborate with Sanofi on the development of multiple RIPK1
inhibitor molecules with the potential to treat a range of
neurological and systemic inflammatory diseases.
The two lead molecules DNL747 and DNL758 target
a critical signaling protein known as the receptor-interacting
serine/threonine-protein kinase 1 (RIPK1) in the TNF receptor
pathway, which regulates inflammation and cell death in tissues
throughout the body. The companies plan to study DNL747 in
Alzheimer’s disease, amyotrophic lateral sclerosis and multiple
sclerosis, and DNL758 in systemic inflammatory diseases such as
rheumatoid arthritis and psoriasis.
Under the terms of the agreement, Sanofi will
make an upfront cash payment to Denali of $125 million, with future
development and commercial milestone payments that could exceed $1
billion. Sanofi and Denali will share commercial profits and losses
from DNL747 in the U.S. and China equally, while Denali will
receive a royalty from Sanofi for other territories for DNL747 and
worldwide for DNL758.
Phase 1b and 2 clinical development costs for
DNL747 will be fully funded by Sanofi for MS, ALS, and other
neurological indications, except in Alzheimer’s disease, which will
be funded by Denali. Phase 3 trials for all neurological
indications will be jointly funded by Sanofi (70%) and Denali
(30%). Sanofi will fully fund the clinical development costs for
DNL758 in systemic inflammatory diseases.
“This collaboration with Denali is yet another
example of Sanofi’s commitment to accelerate the development of
transformative and best-in-class treatments for patients living
with serious illnesses,” said Rita Balice-Gordon, Ph.D., Global
Head of Rare and Neurologic Diseases Research at Sanofi. “We look
forward to working with Denali on the RIPK1 program as we explore
the potential of this mechanism in neurologic and inflammatory
diseases.”
“RIPK1 is a promising target with the potential
to bring disease modifying medicines to patients suffering from
neurodegenerative diseases as well as systemic inflammatory
diseases. We are very excited to partner with Sanofi and expand our
RIPK1 program into new indications,” said Ryan Watts, Ph.D., CEO of
Denali. "With its considerable infrastructure and experience
in both clinical development and commercial functions, Sanofi is an
ideal partner for Denali to maximize the clinical and commercial
success of our RIPK1 program.”
RIPK1 Inhibitor Molecules
- DNL747 is a brain-penetrant small molecule inhibitor of RIPK1.
It is currently being evaluated in early clinical stage trials,
known as Phase 1. Phase 1b studies in Alzheimer’s disease and ALS
patients are expected to commence in the near-term. Denali will
lead Phase 2 clinical trials in Alzheimer’s disease while Sanofi
will lead Phase 2 clinical trials in MS and ALS, as well as future
Phase 3 trials in all neurological indications.
- DNL758 is a small molecule inhibitor of RIPK1 that does not
penetrate the brain. Sanofi will lead clinical development
activities for all systemic inflammatory diseases. The clinical
trials are expected to begin in 2019.
The collaboration also includes additional
pre-clinical RIPK1 inhibitor molecules.
The transaction is expected to close in the
coming months in accordance with customary regulatory
approvals.
About Denali Therapeutics
Denali is a biopharmaceutical company developing
a broad portfolio of product candidates for neurodegenerative
diseases. Denali pursues new treatments by rigorously assessing
genetically validated targets, engineering delivery across the
blood-brain barrier and guiding development with biomarker
monitoring to demonstrate target engagement and select patients.
Denali is based in South San Francisco. For additional information,
please visit www.denalitherapeutics.com.
Cautionary Note Regarding
Forward-Looking Statements
This press release contains forward-looking
statements within the meaning of the Private Securities Litigation
Reform Act of 1995. Forward-looking statements expressed or implied
in this press release include, but are not limited to, the
potential benefits of the collaboration; plans to commence Phase 1b
clinical studies of DNL747 in Alzheimer’s disease and ALS patients
in the near-term; the expectation as to when the transaction will
close; expectations for future clinical development activities and
the timing of future clinical trials; plans for Sanofi and Denali
to collaborate on development of RIPK1 inhibitor molecules; and
statements made by Denali’s Chief Executive Officer.
Actual results are subject to risks and
uncertainties and may differ materially from those indicated by
these forward-looking statements as a result of these risks and
uncertainties, including but not limited to, risks related to: the
risk that the transaction may not close in a timely manner or at
all; risks related to obtaining the requisite regulatory approvals;
the risk of the occurrence of any event, change or other
circumstance that could give rise to the termination of the
agreement (including without limitation the failure to timely
obtain requisite regulatory approvals); risks related to the effect
of the announcement of the transaction on Denali’s business
relationships, operating results and business generally; Denali’s
early stages of clinical drug development; Denali’s ability to
complete the development and, if approved, commercialization of its
product candidates; Denali’s ability to conduct or complete
clinical trials on expected timelines; implementation of Denali’s
strategic plans for its business, product candidates and BBB
platform technology; and other risks, including those described in
Denali’s Annual Report on Form 10-K filed with the SEC on March 19,
2018, Denali’s Quarterly Report on Form 10-Q filed with the SEC on
August 9, 2018 and Denali’s future reports to be filed with the
SEC. The forward-looking statements in this press release are based
on information available to Denali as of the date hereof. Denali
disclaims any obligation to update any forward-looking statements,
except as required by law.
Denali Media Relations
Contacts:
Morgan Warners (202)
295-0124mwarners@gpg.com
Lizzie Hyland(646) 495-2706lhyland@gpg.com
Denali Therapeutics (NASDAQ:DNLI)
Historical Stock Chart
From Jun 2024 to Jul 2024
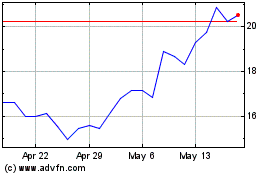
Denali Therapeutics (NASDAQ:DNLI)
Historical Stock Chart
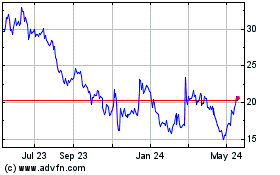
From Jul 2023 to Jul 2024