AKL-T01 is the first and only digital
therapeutic specifically designed to improve attention function
PureTech Health plc (Nasdaq: PRTC, LSE: PRTC) (“PureTech” or the
“Company”), a clinical-stage biotherapeutics company dedicated to
discovering, developing and commercializing highly differentiated
medicines for devastating diseases, is pleased to note that its
Founded Entity, Akili Interactive (Akili) today announced
collaborations with Weill Cornell Medicine, NewYork-Presbyterian
Hospital and Vanderbilt University Medical Center to evaluate Akili
digital therapeutic AKL-T01 as a treatment for patients with
cognitive dysfunction following COVID-19 (also known as “COVID
brain fog”). There are currently no approved treatments for
cognitive impairments in COVID-19 survivors.
Under each collaboration, Akili will work with research teams at
each institution to conduct two separate randomized, controlled
clinical studies evaluating AKL-T01’s ability to target and improve
cognitive functioning in COVID-19 survivors who have exhibited a
deficit in cognition. AKL-T01 is the first and only digital
therapeutic specifically designed to improve attention function.
The organizations aim to begin clinical recruitment for the studies
in the next month.
Since being named a global pandemic by the World Health
Organization in March 2020, clinicians continue to learn about the
vast ways in which COVID-19 manifests in patients. Evidence is
mounting on long-term neurological and cognitive symptoms that can
persist in some COVID-19 patients after initial diagnosis, even
after the virus is no longer detected in the body. A study
published in Neuropsychopharmacology led by Drs. Abhishek Jaywant
and Faith Gunning at Weill Cornell Medicine and
NewYork-Presbyterian found that difficulties in attention,
multitasking, and processing speed were common in hospitalized
patients recovering from COVID-191. Of the patients in their study,
81% exhibited some degree of cognitive impairment1. Recent research
also shows these cognitive impairments may persist
post-hospitalization and commonly occur in “post-COVID long
haulers” or “long COVID” patients. These impairments can have a
significant impact on survivors’ daily functioning and quality of
life, impacting the ability of most COVID-19 long haulers to work
for six months or more according to a recent study2. For more
information on COVID brain fog, click here.
The full text of the announcement from Akili is as follows:
Akili Collaborates with Weill Cornell
Medicine, NewYork-Presbyterian Hospital and Vanderbilt University
Medical Center to Study Digital Therapeutic AKL-T01 as Treatment
for Patients with COVID Brain Fog
AKL-T01 is the first and only digital
therapeutic specifically designed to improve attention function
BOSTON – April 7, 2021 – Akili Interactive (“Akili” or
“Company"), today announced collaborations with Weill Cornell
Medicine, NewYork-Presbyterian Hospital and Vanderbilt University
Medical Center to evaluate Akili digital therapeutic AKL-T01 as a
treatment for patients with cognitive dysfunction following
COVID-19 (also known as “COVID brain fog”). There are currently no
approved treatments for cognitive impairments in COVID-19
survivors.
Under each collaboration, Akili will work with research teams at
each institution to conduct two separate randomized, controlled
clinical studies evaluating AKL-T01’s ability to target and improve
cognitive functioning in COVID-19 survivors who have exhibited a
deficit in cognition. AKL-T01 is the first and only digital
therapeutic specifically designed to improve attention function.
The organizations aim to begin clinical recruitment for the studies
in the next month.
“As frontline healthcare workers continue to fight the immediate
acute symptoms of COVID-19, certain longer-term consequences of the
illness are beginning to emerge, including serious cognitive
impairments,” said Anil S. Jina, M.D., Chief Medical Officer at
Akili. “With more than 100 million infections globally and
counting, the potential impact of long-term cognitive impairments
in even a subset of these patients is devastating. We look forward
to working with leading researchers at Vanderbilt, Weill Cornell
and NewYork-Presbyterian to understand and improve COVID-19-related
cognitive deficits.”
Since being named a global pandemic by the World Health
Organization in March 2020, clinicians continue to learn about the
vast ways in which COVID-19 manifests in patients. Evidence is
mounting on long-term neurological and cognitive symptoms that can
persist in some COVID-19 patients after initial diagnosis, even
after the virus is no longer detected in the body. A study
published in Neuropsychopharmacology led by Drs. Abhishek Jaywant
and Faith Gunning at Weill Cornell Medicine and
NewYork-Presbyterian found that difficulties in attention,
multitasking, and processing speed were common in hospitalized
patients recovering from COVID-191. Of the patients in their study,
81% exhibited some degree of cognitive impairment1. Recent research
also shows these cognitive impairments may persist
post-hospitalization and commonly occur in “post-COVID long
haulers” or “long COVID” patients. These impairments can have a
significant impact on survivors’ daily functioning and quality of
life, impacting the ability of most COVID-19 long haulers to work
for six months or more according to a recent study2. For more
information on COVID brain fog, click here.
“Clinicians are seeing an increase in cognitive impairments
among COVID-19 patients and though we don’t yet know how long these
difficulties last, we are concerned about how these cognitive
difficulties may affect people in their daily lives,” said Gunning
Ph.D., Vice Chair of Research in the Department of Psychiatry,
associate professor of psychology in psychiatry at Weill Cornell
Medicine, and associate attending psychologist at
NewYork-Presbyterian/Weill Cornell Medical Center, who is the study
coordinator. “It’s critical that we identify therapeutics to help
the increasing number of people whose lives have been impacted by
cognitive impairments associated with COVID-19.”
“The chronic symptoms of COVID-19 long haulers represent a
serious and growing public health concern that will linger long
after the acute nature of COVID-19 has passed,” said James Jackson,
PsyD, Assistant Director of The ICU Recovery Center at Vanderbilt
and lead psychologist for the Critical Illness, Brain Dysfunction
and Survivorship (CIBS) Center at the Vanderbilt University Medical
Center. “We’re excited by the potential of new therapeutics that
target cognitive impairments to help COVID-19 survivors.”
AKL-T01 is built on the Akili Selective Stimulus Management
Engine (SSME™), a disease agnostic proprietary technology designed
to treat impaired cognitive function, specifically attention
control. Delivered through an action video game experience, the
first-in-class technology presents specific sensory stimuli and
simultaneous motor challenges designed to target and activate the
neural systems that play a key role in attention function while
using adaptive algorithms to personalize the treatment experience
for each individual patient. SSME has been evaluated as a potential
treatment for cognitive impairments associated with a number of
different disease areas and has been studied in more than 2600
patients across 30 clinical trials. AKL-T01, branded EndeavorRx™,
is cleared for use by the U.S. Food and Drug Administration (FDA)3
and has received Conformité Européenne (CE) Mark certification in
Europe for use in pediatric ADHD4. Product screenshots and b-roll
are available here.
Study Designs The Akili, Weill Cornell Medicine and
NewYork-Presbyterian Hospital randomized, controlled study will
evaluate AKL-T01 in approximately 100 COVID-19 survivors ages 18-89
who have exhibited a deficit in cognition. The study will take
place over 10 weeks, with 6 weeks of treatment and 4 weeks of
follow-up. Half of the study participants will receive the digital
treatment and half will serve as a control group. The primary
endpoint of the study is mean change in cognitive function, as
assessed by a measure of attention and processing speed. Secondary
endpoints include additional measures of cognitive functioning. The
study will be conducted remotely in patients’ homes, and patients
in the control arm will have the option to receive the AKL-T01
intervention after the conclusion of their participation in the
control condition.
The Akili and Vanderbilt randomized, controlled study will
evaluate AKL-T01 in approximately 100 COVID-19 survivors ages 18
and older who have exhibited a deficit in cognition. The study will
recruit from subjects who have completed the SARS-CoV-2 Household
Transmission Study. Half of the study participants will receive the
digital treatment for 4 weeks and half will serve as a control
group. The primary endpoint of the study is mean change in
cognitive function, as measured by CNS Vital Signs (composite score
of cognitive function, especially attention and processing speed).
Secondary endpoints include additional measures of cognitive
functioning. The study will be conducted remotely in patients’
homes.
About Akili Akili is combining scientific and clinical
rigor with the ingenuity of the tech and entertainment industries
to challenge the status quo of medicine. Akili has pioneered the
development of video game-based digital medicine to improve
cognitive function. Akili’s flagship product is a prescription
digital treatment to address inattention in children with attention
deficit hyperactivity disorder (ADHD). Akili’s patented technology
serves as the foundation of its products and is designed to
directly activate the networks in the brain responsible for
cognitive function. Driven by Akili’s belief that effective
medicine can also be fun and engaging, Akili’s treatments are
delivered through captivating action video game experiences that
drive engagement and compliance. For more information, please visit
AkiliInteractive.com.
About PureTech Health PureTech is a clinical-stage
biotherapeutics company dedicated to discovering, developing and
commercializing highly differentiated medicines for devastating
diseases, including intractable cancers, lymphatic and
gastrointestinal diseases, central nervous system disorders and
inflammatory and immunological diseases, among others. The Company
has created a broad and deep pipeline through the expertise of its
experienced research and development team and its extensive network
of scientists, clinicians and industry leaders. This pipeline,
which is being advanced both internally and through PureTech’s
Founded Entities, as of the date of PureTech’s most recently filed
Registration Statement on Form 20-F, was comprised of 24
therapeutics and therapeutic candidates, including two that have
received FDA clearance and European marketing authorization. All of
the underlying programs and platforms that resulted in this
pipeline of product candidates were initially identified or
discovered and then advanced by the PureTech team through key
validation points based on the Company’s unique insights into the
biology of the brain, immune and gut, or BIG, systems and the
interface between those systems, referred to as the BIG Axis.
For more information, visit www.puretechhealth.com or connect
with us on Twitter @puretechh.
Cautionary Note Regarding Forward-Looking Statements This
press release contains statements that are or may be
forward-looking statements, including statements that relate to the
company's future prospects, developments, and strategies. The
forward-looking statements are based on current expectations and
are subject to known and unknown risks and uncertainties that could
cause actual results, performance and achievements to differ
materially from current expectations, including, but not limited
to, our expectations regarding the potential therapeutic benefits
of Akili’s AKL-T01 as a treatment for patients with cognitive
dysfunction following COVID-19, and those risks and uncertainties
described in the risk factors included in the regulatory filings
for PureTech Health plc. These forward-looking statements are based
on assumptions regarding the present and future business strategies
of the company and the environment in which it will operate in the
future. Each forward-looking statement speaks only as at the date
of this press release. Except as required by law and regulatory
requirements, neither the company nor any other party intends to
update or revise these forward-looking statements, whether as a
result of new information, future events or otherwise.
1 Jaywant et al. Neuropsychopharmacol. (2021). 2 David et al.
Preprint. (2020). 3 EndeavorRx is an FDA-cleared medical device. It
is indicated to improve attention function as measured by
computer-based testing in children ages 8-12 years old with
primarily inattentive or combined-type ADHD, who have a
demonstrated attention issue. Patients who engage with EndeavorRx
demonstrate improvements in a digitally assessed measure Test of
Variables of Attention (TOVA) of sustained and selective attention
and may not display benefits in typical behavioral symptoms, such
as hyperactivity. EndeavorRx should be considered for use as part
of a therapeutic program that may include clinician-directed
therapy, medication, and/or educational programs, which further
address symptoms of the disorder. EndeavorRx is available by
prescription only. It is not intended to be used as a stand-alone
therapeutic and is not a substitution for a child’s medication. 4
In June, 2020, EndeavorRx received Conformité Européenne (CE) Mark
certification as a prescription-only digital therapeutic software
intended for the treatment of attention and inhibitory control
deficits in pediatric patients with ADHD.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210407005246/en/
Investors Allison Mead Talbot +1 617 651 3156
amt@puretechhealth.com
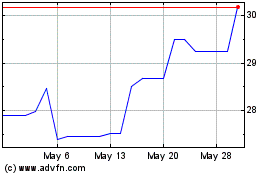
PureTech Health (NASDAQ:PRTC)
Historical Stock Chart
From Jun 2024 to Jul 2024
PureTech Health (NASDAQ:PRTC)
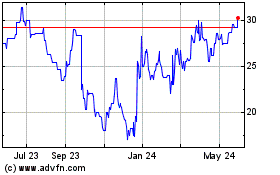
Historical Stock Chart
From Jul 2023 to Jul 2024