C2i Genomics Partners with Twist Bioscience to Launch Reference Materials for Whole-Genome Cancer Detection
09 February 2022 - 12:00AM
Business Wire
Twist will leverage C2i’s leadership in
whole-genome cancer signatures across solid cancers to generate
unique synthetic reference materials for cancer detection
validation in labs across the globe
C2i to integrate Twist’s library preparation
kits into minimal residual disease workflow
C2i Genomics, a cancer intelligence company, and Twist
Bioscience Corporation (NASDAQ: TWST), a company enabling customers
to succeed through its offering of high-quality synthetic DNA using
its silicon platform, today announced a partnership to develop
whole-genome cancer reference materials. This resource will provide
diagnostic labs around the globe with the ability to better
validate and monitor the quality of their whole-genome cancer
screening and minimal residual disease (MRD) products. In addition,
C2i will integrate Twist’s library preparation into their MRD
workflow.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20220208005517/en/
Today in cancer diagnostics, a major obstacle facing diagnostic
labs and regulators is the lack of standardized reference samples
for labs to use for the analytical validation of their whole-genome
assays. Especially in the complex field of oncology, there is a
heightened need for advanced technology to help execute better
treatment decisions, and ultimately, to save more lives.
Under the collaboration, C2i will use its artificial
intelligence (AI) to identify a very large set of variants across
the entire genome known to be associated with cancer signatures.
Twist will then synthesize each of these variants, tuning them up
or down to allow for similarity to natural samples, to create a
whole-genome sequencing-focused set of reference materials. The
reference standard kits will be available through Twist
Bioscience.
“There is a tremendous need to have a consistent reference
standard for the detection of cancer and minimal residual disease,”
said Emily M. Leproust, Ph.D., CEO and co-founder of Twist
Bioscience. “We expect that the development of this exceptionally
robust set of reference standards for whole-genome sequencing will
complement our cfDNA Pan-Cancer Reference Standards, which can be
used in liquid biopsy assays. We believe both of these products
will enable our customers to create innovative solutions that
accurately detect cancer initially and upon recurrence. C2i’s
approach through AI and WGS has the potential to truly change the
treatment paradigm.”
The reference standards will also be used as training samples,
tuning C2i's algorithm for patient sample analysis. Using
whole-genome sequencing and machine learning, C2i has developed a
way to use (AI) to enable highly precise cancer detection in even
the tiniest tumors. The development of this strategic collaboration
demonstrates C2i’s investment in furthering the utility and
accessibility to whole-genome cancer diagnostics in labs all over
the world.
“C2i is committed to increasing the accessibility of
whole-genome cancer detection and monitoring to support researchers
and clinicians in improving oncology care for patients. We’re proud
to work closely with Twist, as these new reference kits will solve
an urgent unmet need for lab validation and proficiency testing
materials in the cancer diagnosis space,” said Asaf Zviran, CEO and
Co-Founder at C2i Genomics.
C2i’s partnership with Twist comes after a year of significant
momentum. C2i announced several key partnerships within the last
year. The first being the initiation of its global commercial
launch through its partnership with NovogeneAIT Genomics. This was
followed by a network expansion with Premier Inc., a collaboration
with NuProbe Global, and most recently an expansion into Europe
with OncoDNA and the initiation of a first-of-its-kind
interventional clinical trial in France led by Gustave Roussy. For
companies interested in deploying the C2-Intelligence Platform,
please visit www.c2i-genomics.com.
About C2i Genomics
Founded in 2019, C2i Genomics has created the world’s leading
cancer treatment intelligence platform that uses low-input blood
(only 2mL blood) whole-genome sequencing to provide up to 100x more
sensitive detection than competing liquid biopsy companies. With a
headquarters in NYC, CLIA lab in Cambridge MA, and an R&D
center in Israel, C2i’s SaaS solution utilizes a cloud-based
platform to perform cancer tumor burden monitoring on a global
scale, leveraging the thousands of already installed genome
sequencers around the world. Using cutting-edge scientific
breakthroughs, growing genomic and clinical databases, and
sophisticated computation and AI, C2i enables high precision
personalized medicine, reduced cancer treatment costs, and
accelerated drug development. For more information, please visit
www.c2i-genomics.com.
About Twist Bioscience
Twist Bioscience is a leading and rapidly growing synthetic
biology and genomics company that has developed a disruptive DNA
synthesis platform to industrialize the engineering of biology. The
core of the platform is a proprietary technology that pioneers a
new method of manufacturing synthetic DNA by “writing” DNA on a
silicon chip. Twist is leveraging its unique technology to
manufacture a broad range of synthetic DNA-based products,
including synthetic genes, tools for next-generation sequencing
(NGS) preparation, and antibody libraries for drug discovery and
development. Twist is also pursuing longer-term opportunities in
digital data storage in DNA and biologics drug discovery. Twist
makes products for use across many industries including healthcare,
industrial chemicals, agriculture and academic research.
Legal Notice Regarding Forward-Looking Statements
This press release contains forward-looking statements. All
statements other than statements of historical facts contained
herein are forward-looking statements reflecting the current
beliefs and expectations of management made pursuant to the safe
harbor provisions of the Private Securities Litigation Reform Act
of 1995. Forward-looking statements include statements regarding
the development of a whole-genome sequencing-focused set of
reference materials, the potential for such products to change
treatment paradigms, and the ability of such reference materials to
aid in the development of solutions that accurately detect cancer.
Such forward-looking statements involve known and unknown risks,
uncertainties, and other important factors that may cause Twist
Bioscience’s actual results, performance, or achievements to be
materially different from any future results, performance, or
achievements expressed or implied by the forward-looking
statements. For a description of the risks and uncertainties that
could cause actual results to differ from those expressed in these
forward-looking statements, as well as risks relating to Twist
Bioscience’s business in general, see Twist Bioscience’s risk
factors set forth in Twist Bioscience’s [Annual Report Form 10-K
filed with the Securities and Exchange Commission on November 23,
2021] and subsequent filings with the SEC. Any forward-looking
statements contained in this press release speak only as of the
date hereof, and Twist Bioscience specifically disclaims any
obligation to update any forward-looking statement, whether as a
result of new information, future events or otherwise.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220208005517/en/
Jessica Flick PR for C2i Genomics VSC for C2i Genomics
c2i@vsc.co
Angela Bitting SVP, Corporate Affairs Twist Bioscience
abitting@twistbioscience.com
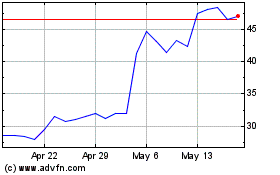
Twist Bioscience (NASDAQ:TWST)
Historical Stock Chart
From Jun 2024 to Jul 2024
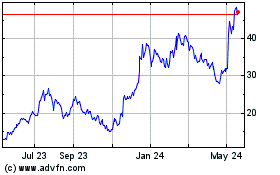
Twist Bioscience (NASDAQ:TWST)
Historical Stock Chart
From Jul 2023 to Jul 2024