Emergent BioSolutions Awarded U.S. Department of State Contract to Supply Medical Countermeasures For Chemical Warfare Agents...
01 March 2019 - 8:45AM

Emergent BioSolutions Inc. (NYSE: EBS) today announced that it has
signed an indefinite-delivery, indefinite-quantity (IDIQ) contract
with the U.S. Department of State to establish a long-term,
reliable, and stable supply chain for medical countermeasures that
address chemical warfare agents. The contract is comprised of a
five-year base period of performance along with five one-year
option periods with a total contract value of a minimum of
approximately $7 million to a maximum of $100 million over the
contract’s period of performance. Emergent will be supplying two of
its current medical countermeasures addressing chemical threats;
the Trobigard® atropine sulfate/obidoxime chloride auto-injector, a
drug-device combination product for emergency use in the event of
nerve agent or organophosphate poisoning, and RSDL® (Reactive Skin
Decontamination Lotion Kit), which is intended to remove or
neutralize chemical warfare agents and T-2 toxin from the skin.
“Emergent is pleased with this follow-on opportunity to meet the
U.S. government’s need for medical countermeasures that enhance the
security of U.S. diplomats and other Chief of Mission personnel
engaged in high-risk environments worldwide,” said Doug White,
senior vice president and head of the devices business unit at
Emergent BioSolutions. “Our mission – to protect and enhance life –
has been at the core of our 20-year history of partnering with
governments. We look forward to successfully completing deliveries
of our Trobigard auto-injector under our 2017 contract and to our
continued partnership with the State Department as we expand our
portfolio of solutions to address existing and emerging chemical
warfare agents for the long-term.”
“One of our most important responsibilities is to respond to
critical threats to Department diplomatic missions overseas and to
maintain the safety of deployed employees,” said William A.
Walters, M.D., managing director of operational medicine for the
U.S. Department of State. “This begins with establishing a stable
supply chain of medical countermeasures and ensuring adequate
stockpiles as we equip ourselves with the tools that best enable a
safe, accurate, and timely response to the highest risk chemical
agents.”
Under this IDIQ contract, Emergent will maintain the capability
to manufacture and deliver various medical countermeasures defined
by the Department of State, including the Trobigard auto-injector,
Reactive Skin Decontamination Lotion Kit, and auto-injector
training devices.
About Emergent’s Chemical Medical Countermeasure
ProgramsEmergent has proprietary medical countermeasure
products and product candidates that address accidental or
deliberate exposure to chemical agents. Emergent is currently
partnering with the U.S. government to develop new auto-injector
and intranasal products to defend against emerging chemical threats
such as nerve agents and cyanide. These drug-device combination
products are designed to support chemical defense programs by
governments around the world to protect military and civilian
populations.
About TrobigardTrobigard® atropine sulfate
2mg/obidoxime chloride 220mg auto-injector is Emergent’s first
nerve agent antidote product launched outside the United States. It
has been designed as a pre-hospital medical intervention during
nerve agent and organophosphate poisoning. Trobigard auto-injector
is manufactured in Germany and is currently stockpiled and fielded
by select European, Middle Eastern, and other U.S. allied countries
authorized to purchase emergency use products. The Trobigard
auto-injector has not been approved by the U.S. Food and Drug
Administration (FDA) and is not promoted or distributed in the
U.S.
About RSDLThe RSDL® (Reactive Skin
Decontamination Lotion Kit) is an FDA-cleared device consisting of
a lotion impregnated sponge in an easy-to-open packet. The RSDL kit
is intended to remove or neutralize chemical warfare agents and T-2
toxin from the skin. RSDL kit was initially developed by Defence
Research and Development Canada, an agency of the Canadian
Department of National Defence (DND), to prepare the Canadian
forces for chemical warfare attacks. The U.S. Department of Defense
became interested in RSDL kits and the military then filed with the
FDA. The FDA issued 510(k) clearance for RSDL kits in November
2002. The European CE Mark and Australian TGA clearances were later
issued. The RSDL kit has been adopted by several militaries around
the world with over 15 million packets of RSDL sold in over 35
countries.
About Emergent BioSolutionsEmergent
BioSolutions Inc. is a global life sciences company seeking to
protect and enhance life by focusing on providing specialty
products for civilian and military populations that address
accidental, deliberate, and naturally occurring public health
threats. We aspire to be a Fortune 500 company recognized for
protecting and enhancing life, driving innovation, and living our
values. Additional information about the company may be found at
www.emergentbiosolutions.com. Find us on LinkedIn and follow us on
Twitter @emergentbiosolu and Instagram @life_at_emergent.
Investor Contact:Robert G.
BurrowsVice President, Investor
Relations240-631-3280BurrowsR@ebsi.com
Media Contact:Lynn KiefferVice
President, Corporate Communications
240-631-3391KiefferL@ebsi.com
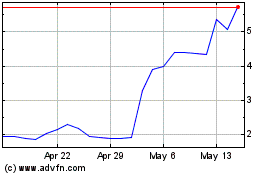
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From Jun 2024 to Jul 2024
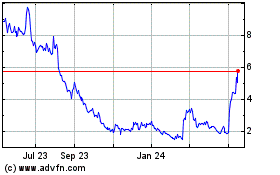
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From Jul 2023 to Jul 2024