Hemogenyx Pharmaceuticals Says FDA Accepted Plans to Address Leukemia Treatment Concerns
14 September 2023 - 6:11PM
Dow Jones News
By Anthony O. Goriainoff
Hemogenyx Pharmaceuticals said the U.S. Food and Drug
Administration has accepted its plans to address the agency's
concerns over its leukemia treatment product candidate.
The London-listed biopharmaceutical group--that develops new
therapies and treatments for blood diseases--said Thursday it had
submitted a detailed plan for its HEMO-CAR-T investigational new
drug, or IND, which was supported by laboratory tests to address
the FDA's concerns.
On July 10, the company said the FDA was concerned with the
splicing that occurs during the manufacturing of the lentivirus
used in its acute myeloid leukemia treatment product.
"We are now working hard to complete the schedule of work set
out in the plan and to resubmit the IND as expeditiously as
possible in order to move forward with clinical trials of
HEMO-CAR-T," Chief Executive Vladislav Sandler said.
Shares at 0734 GMT were up 0.02 pence, or 1.5%, at 1.68
pence.
Write to Anthony O. Goriainoff at
anthony.orunagoriainoff@dowjones.com
(END) Dow Jones Newswires
September 14, 2023 03:56 ET (07:56 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
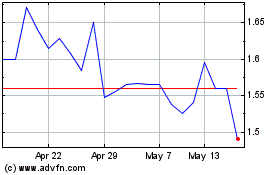
Hemogenyx Pharmaceuticals (LSE:HEMO)
Historical Stock Chart
From Apr 2024 to May 2024
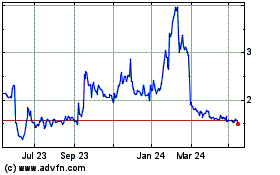
Hemogenyx Pharmaceuticals (LSE:HEMO)
Historical Stock Chart
From May 2023 to May 2024