UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the date of September 1, 2023
Commission File Number 001-39124
Centogene N.V.
(Translation of registrant's name into English)
Am Strande
7
18055 Rostock
Germany
(Address of principal executive offices)
Indicate by check mark whether the registrant files
or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-F..X.. Form 40-F.....
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ___
Indicate by check mark if the registrant is submitting
the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ____
Centogene N.V.
On September 1, 2023, Centogene N.V. issued a press release titled
“CENTOGENE Announces Publication Establishing Lyso-Gb1 as a Predictive Biomarker for Gaucher Disease Patients”.
A copy of the press release is attached hereto as Exhibit 99.1.
Signatures
Pursuant to the requirements of the Securities Exchange Act of 1934,
the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Date: September 1, 2023
| |
|
|
CENTOGENE N.V.
|
| |
|
|
|
|
|
| |
|
|
|
|
|
| |
|
|
By: |
/s/ Jose Miguel Coego Rios |
| |
|
|
|
Name: |
Jose Miguel Coego Rios |
| |
|
|
|
Title: |
Chief Financial Officer |
Exhibit Index
| Exhibit |
Description of Exhibit |
| |
|
| 99.1 |
Press Release dated September 1, 2023 |
Exhibit 99.1
PRESS
RELEASE
CENTOGENE
Announces Publication Establishing Lyso-Gb1 as a Predictive Biomarker for Gaucher Disease Patients
| • | Results
from pivotal study published in Diagnostics journal |
| • | Confirmation
of utility of lyso-Gb1 (glucosylsphingosine) as a sensitive biomarker for Gaucher disease |
| • | Could
predict clinical course of patients and improve personalized care |
CAMBRIDGE,
Mass. and ROSTOCK, Germany and BERLIN, September 1, 2023 (GLOBE NEWSWIRE) – CENTOGENE N.V. (Nasdaq: CNTG) (the “Company”),
the essential life science partner for data-driven answers in rare and neurodegenerative diseases, today announced data confirming the
utility of lyso-Gb1 (glucosylsphingosine) as a sensitive biomarker for Gaucher disease (GD). The findings from this landmark study which
were published in Diagnostics in a paper titled, “Insights into the value of lyso-Gb1 as a predictive biomarker in treatment-naïve
patients with Gaucher disease type 1 in the LYSO-PROOF study,” also indicate lyso-Gb1 could help to predict the clinical course
of patients and improve personalized care of GD patients in the future.
“Over
the past decade, we have leveraged all our multiomic technological know-how to establish and confirm lyso-Gb1 as the best biomarker for
Gaucher disease,” said Professor Peter Bauer, CENTOGENE’s Chief Medical and Genomic Officer. “This landmark study further
advances our understanding of Gaucher disease patients – demonstrating a significant correlation between lyso-Gb1 levels and disease
severity. The combination of state-of-the-art multiomic testing with access to data from our diverse, real-world CENTOGENE Biodatabank
provides the essential link between genetic variants and actionable clinical information.”
The
study, which included 160 treatment-naïve GD patients from Israel, Russia, Pakistan, Egypt, Iran, Morocco, Algeria, India, Spain,
Albania, Greece, Sweden, Columbia, and Tunisia, is one of the largest studies to examine Gaucher patients who have never received disease-specific
treatment.
CENTOGENE
utilized CentoCard®, the Company’s proprietary, CE-marked dried blood spot (DBS) collection kit in combination with state-of-the-art
biochemical and sequencing technologies to screen for mutations in the GBA1 gene and establish a GD diagnosis. The insights gained
were powered by the CENTOGENE Biodatabank, the world’s largest real-world integrated multiomic data repository in rare and neurodegenerative
diseases.
The
results of the study revealed a highly significant correlation between lyso-Gb1 and disease severity in all Gaucher patients, including
those with novel rare GBA1 variants.
“This
important international study has helped to set the standard for Gaucher disease care – from diagnosis and prognosis to treatment
and monitoring,” said Tobias Böttcher, M.D., Director of Clinical Neurogenetics at CENTOGENE. “The progressive increase
in lyso-Gb1 levels in untreated Gaucher patients suggests that these patients could benefit from treatment, such as enzyme replacement
therapy. The biomarker, combined with diagnostic and sequencing technologies, provides physicians with the tool to establish the best
therapeutic strategies for each individual patient, which could significantly improve their quality of life.”
To
read the full study, visit: https://link.centogene.com/lyso-gb1-publication
About
Gaucher Disease
Gaucher
disease (GD) is a rare inherited lysosomal storage disorder (LSD) that affects the body’s ability to break down a type of fat called
glucocerebroside. This results in the accumulation of this fat within cells, particularly in the liver, spleen, and bone marrow. GD can
cause a wide range of symptoms, including enlargement of the liver and spleen, anemia, bone pain, and increased susceptibility to fractures.
There are different types of GD, and the severity of symptoms can vary greatly. Treatment options may include enzyme replacement therapy
to help break down the accumulated fat and manage the associated symptoms. Early diagnosis and appropriate management are essential to
improve the quality of life for patients with GD.
About
Lyso-Gb1
Lyso-Gb1,
also known as glucosylsphingosine, is a biomarker associated with Gaucher disease (GD). Elevated levels of lyso-GB1 in the blood and
other tissues can indicate the presence of GD and help in its diagnosis and monitoring, as well as proactively predicting disease course.
This biomarker plays a crucial role in assessing the severity of the disease and guiding treatment decisions for affected individuals.
Due
to its utility, lyso-Gb1 could be used as a particularly valuable addition in Newborn Screening programs to shorten the diagnostic delay
in GD.
Lyso-Gb1
is one of CENTOGENE’s commercialized, proprietary biomarkers, which has patents globally, including in the U.S., Europe, and Asia.
About
CENTOGENE
CENTOGENE’s
mission is to provide data-driven, life-changing answers to patients, physicians, and pharma companies for rare and neurodegenerative
diseases. We integrate multiomic technologies with the CENTOGENE Biodatabank – providing dimensional analysis to guide the next
generation of precision medicine. Our unique approach enables rapid and reliable diagnosis for patients, supports a more precise physician
understanding of disease states, and accelerates and de-risks targeted pharma drug discovery, development, and commercialization.
Since
our founding in 2006, CENTOGENE has been offering rapid and reliable diagnosis – building a network of approximately 30,000 active
physicians. Our ISO, CAP, and CLIA certified multiomic reference laboratories in Germany utilize Phenomic, Genomic, Transcriptomic, Epigenomic,
Proteomic, and Metabolomic datasets. This data is captured in our CENTOGENE Biodatabank, with over 750,000 patients represented from
over 120 highly diverse countries, over 70% of whom are of non-European descent. To date, the CENTOGENE Biodatabank has contributed to
generating novel insights for more than 275 peer- reviewed publications.
By
translating our data and expertise into tangible insights, we have supported over 50 collaborations with pharma partners. Together, we
accelerate and de-risk drug discovery, development, and commercialization in target & drug screening, clinical development, market
access and expansion, as well as offering CENTOGENE Biodata Licenses and Insight Reports to enable a world healed of all rare and neurodegenerative
diseases.
To
discover more about our products, pipeline, and patient-driven purpose, visit www.centogene.com and follow us on LinkedIn.
Forward-Looking
Statements
This
press release contains “forward-looking statements” within the meaning of the U.S. federal securities laws. Statements contained
herein that are not clearly historical in nature are forward-looking, and the words “anticipate,” “believe,”
“continues,” “expect,” “estimate,” “intend,” “project,” “plan,”
“is designed to,” “potential,” “predict,” “objective” and similar expressions and future
or conditional verbs such as “will,” “would,” “should,” “could,” “might,”
“can,” and “may,” or the negative of these are generally intended to identify forward-looking statements. Such
forward-looking statements involve known and unknown risks, uncertainties, and other important factors that may cause CENTOGENE’s
actual results, performance, or achievements to be materially different from any future results, performance, or achievements expressed
or implied by the forward- looking statements. Such risks and uncertainties include, among others, negative economic and geopolitical
conditions and instability and volatility in the worldwide financial markets, possible changes in current and proposed legislation, regulations
and governmental policies, pressures from increasing competition and consolidation in our industry, the expense and uncertainty of regulatory
approval, including from the U.S. Food and Drug Administration, our reliance on third parties and collaboration partners, including our
ability to manage growth, execute our business strategy and enter into new client relationships, our dependency on the rare disease industry,
our ability to manage international expansion, our reliance on key personnel, our reliance on intellectual property protection, fluctuations
of our operating results due to the effect of exchange rates, our ability to streamline cash usage, our continued ongoing compliance
with covenants linked to financial instruments, our requirement for additional financing and our ability to continue as a going concern,
or other factors. For further information on the risks and uncertainties that could cause actual results to differ from those expressed
in these forward-looking statements, as well as risks relating to CENTOGENE’s business in general, see CENTOGENE’s risk factors
set forth in CENTOGENE’s Form 20-F filed on May 16, 2023, with the Securities and Exchange Commission (the “SEC”) and
subsequent filings with the SEC. Any forward- looking statements contained in this press release speak only as of the date hereof, and
CENTOGENE’s specifically disclaims any obligation to update any forward-looking statement, whether as a result of new information,
future events, or otherwise.
CONTACT
CENTOGENE
Melissa
Hall
Corporate
Communications
Press@centogene.com
Lennart
Streibel
Investor
Relations
IR@centogene.com
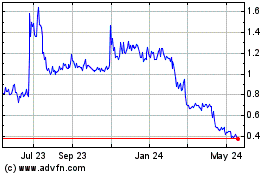
Centogene NV (NASDAQ:CNTG)
Historical Stock Chart
From Apr 2024 to May 2024
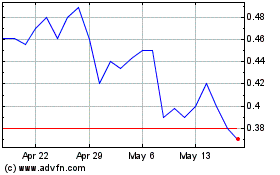
Centogene NV (NASDAQ:CNTG)
Historical Stock Chart
From May 2023 to May 2024