0001678660false00016786602024-08-122024-08-12
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): August 12, 2024
Prelude Therapeutics Incorporated
(Exact Name of Registrant as Specified in its Charter)
|
|
|
|
|
|
|
|
|
|
|
|
Delaware |
|
001-39527 |
|
81-1384762 |
(State or other jurisdiction of
incorporation or organization) |
|
(Commission
File Number) |
|
(I.R.S. Employer
Identification No.) |
|
|
|
175 Innovation Boulevard Wilmington, Delaware |
|
19805 |
(Address of principal executive offices) |
|
(Zip Code) |
Registrant’s telephone number, including area code: (302) 467-1280
Not Applicable
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
☐ Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
☐ Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
☐ Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
☐ Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
|
|
|
|
|
Title of each class |
|
Trading
Symbol(s) |
|
Name of each exchange on which registered |
Common Stock, $0.0001 par value per share |
|
PRLD |
|
Nasdaq Global Select Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☒
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 2.02 Results of Operations and Financial Condition
On August 12, 2024, Prelude Therapeutics Incorporated (the "Company") issued a press release announcing its financial results for the three months ended June 30, 2024. A copy of the press release is attached as Exhibit 99.1 to this report.
Item 7.01 Regulation FD Disclosure
The Company has prepared investor presentation materials with information about the Company, which it intends to use as part of investor presentations. A copy of the investor presentation materials to be used by management for presentations is attached as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated herein by reference.
The information in this Current Report on Form 8-K and in Exhibits 99.1 and 99.2 attached hereto is being furnished, but shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (“Exchange Act”), and is not incorporated by reference into any filing of the Company under the Securities Act of 1933, as amended, or the Exchange Act, whether made before or after the date hereof, regardless of any general incorporation language in such filing.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
|
|
|
|
|
|
PRELUDE THERAPEUTICS INCORPORATED |
|
|
|
Date: August 12, 2024 |
By: |
/s/ Bryant Lim |
|
|
Bryant Lim |
|
|
Chief Legal Officer, Corporate Secretary, and Interim Chief Financial Officer |

Prelude Therapeutics Reports Second Quarter 2024 Financial Results and Provides Corporate Update
Interim Phase 1 data for its first-in-class, highly selective IV SMARCA2 degrader, PRT3789, selected for an oral presentation at the European Society for Medical Oncology (ESMO) Congress 2024 in September
Received investigational new drug (IND) authorization for PRT7732, its first-in-class oral SMARCA2 degrader, from the U.S. Food and Drug Administration (FDA)
Announced clinical collaboration with Merck to evaluate PRT3789 in combination with Merck’s anti-PD-1 therapy KEYTRUDA® (pembrolizumab) in patients with SMARCA4-mutated cancers
Potentially best-in-class CDK9 inhibitor, PRT2527, remains on track to report interim Phase 1 data in Q4 2024
Current cash runway into 2026 with $179.8 million in cash, cash equivalents and marketable securities as of June 30, 2024
WILMINGTON, Del., Aug.12, 2024 – Prelude Therapeutics Incorporated (Nasdaq: PRLD), a clinical-stage precision oncology company, today reported its financial results for the second quarter ended June 30, 2024 and provided an update on its clinical development pipeline and other corporate developments.
“Our team continues to make solid progress towards the Company’s ambitious R&D objectives that we established for 2024 and beyond. We are focused on advancing our two lead clinical programs, including the first-in-class, highly selective SMARCA2 degrader, PRT3789 and a potent and selective CDK9 inhibitor, PRT2527, both on track to report initial clinical results this year,” stated Kris Vaddi, Ph.D., Chief Executive Officer of Prelude.
Dr. Vaddi continued, “We believe that targeting the SMARCA pathway has the potential to deliver a ‘pipeline in a program.’ We are building on our leadership position by advancing the industry’s first highly selective oral SMARCA2 degrader, PRT7732, into the clinic, and will initiate a study of PRT3789 in combination with KEYTRUDA in collaboration with Merck later this year. Additionally, in collaboration with AbCellera, we are developing precision ADCs with SMARCA payloads to extend the reach of our molecules to an even broader set of cancers without SMARCA4 mutations.”
“Regarding our clinical development programs, we are very pleased with the progress of both of our SMARCA2 degraders, PRT3789 and PRT7732,” added Jane Huang, M.D., President and Chief Medical Officer of Prelude. “We are looking forward to sharing the initial clinical data from the Phase 1 study of our highly selective SMARCA2 degrader which has been chosen as the subject of an oral presentation at the upcoming ESMO Congress in September.”
Dr. Huang continued, “Additionally, based on the continued progress of PRT2527, our selective CDK9 inhibitor, we intend to present interim phase 1 clinical data, including a potential best-in-class safety profile in the fourth quarter of this year.”
Clinical Program Updates and Upcoming Milestones
PRT3789 – A first-in-class, highly selective, intravenous SMARCA2 Degrader
PRT3789 is a first-in-class SMARCA2 degrader, highly selective for SMARCA2 and designed to treat patients with a SMARCA4 mutation. Cancer patients whose tumors have SMARCA4 mutations have a poor prognosis and as a result, this is an area of high unmet medical need.
PRT3789 is in Phase 1 clinical development in biomarker selected SMARCA4 mutant patients. Enrollment remains on track, and the Company expects to conclude monotherapy dose escalation in 2024 and identify a recommended Phase 2 dose. In addition, enrollment of patients into back-fill cohorts enriched for NSCLC and SMARCA4 loss-of-function mutations is ongoing, as is enrollment of the combination with docetaxel cohort.
Objectives for this first Phase 1 clinical trial are to establish the safety and tolerability profile of PRT3789 as both monotherapy and in combination with docetaxel, evaluate activity, pharmacokinetics and pharmacodynamics and determine a dose and potential indications for advancement into registrational clinical trial(s).
Prelude recently launched an educational video series focused on the science of SMARCA biology, the discovery of first-in-class, highly selective SMARCA2 degraders and the unmet medical need for patients with SMARCA4 mutated cancer. This series can be found on the Company’s website under Highly Selective SMARCA2 Degraders - Prelude Therapeutics (preludetx.com).
Interim Phase 1 data selected for an oral presentation at the ESMO Congress 2024
The abstract titled “First Clinical Results from a Phase 1 Trial of PRT3789, a First-in-Class Intravenous SMARCA2 Degrader, in Patients with Advanced Solid Tumors with a SMARCA4 Mutation,” will be presented by Robin Guo, M.D. from Memorial Sloan Kettering Cancer Center. The ESMO Congress 2024 Scientific Committee selected the abstract as an oral presentation.
The presentation is scheduled for September 13th, 2024, at 4:00 PM CEST (10:00 AM EST) in the Santander Auditorium (Hall 5) as part of the Developmental Therapeutics Session.
Abstracts are anticipated to be available on the ESMO website on September 9th, 2024 at 12:05 AM CEST (6:05 PM EST on September 8th).
Clinical collaboration announced with Merck to evaluate PRT3789 in combination with KEYTRUDA® in patients with SMARCA4-mutated cancers
In July 2024, Prelude announced a clinical collaboration with Merck to evaluate PRT3789 in combination with KEYTRUDA® in patients with SMARCA4-mutated cancers.
The mechanistic rationale and pre-clinical data to support the SMARCA2 and anti-PD-1 monoclonal antibody (mAb) combination was previously presented by the Company at the 2023 AACR International Conference on Molecular Targets and Cancer Therapeutics. In pre-clinical models, SMARCA2 degrader combined with an anti-PD-1 mAb in SMARCA4-mutated cancers enhanced anti-tumor immunity and demonstrated tumor regressions. Please see the Company’s website under Publications - Prelude Therapeutics (preludetx.com) for more information.
Under the terms of the Agreement, Merck will provide KEYTRUDA® to Prelude. Prelude will be the sponsor of the Phase 2 clinical combination trial, anticipated to initiate in the fourth quarter of 2024. Prelude and Merck each retain all commercial rights to their respective compounds, including as monotherapy or as combination therapies.
KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA.
PRT7732 – A potent, highly selective and orally bioavailable SMARCA2 Degrader
Prelude has identified a series of highly selective and orally bioavailable SMARCA2 degraders. The lead oral candidate, PRT7732, recently was granted IND authorization from the FDA and is expected to enter Phase 1 clinical development in the second half of 2024.
PRT2527 – A potent and highly selective CDK9 Inhibitor
PRT2527 is a potent and highly selective CDK9 inhibitor that has the potential to avoid off-target toxicities observed with other less selective CDK9 inhibitors. The Company is currently advancing PRT2527 as monotherapy in both lymphoid and myeloid hematological malignancies, and in combination with zanubrutinib in B-cell malignancies.
PRT2527 is expected to complete monotherapy dose escalation in B-cell malignancies this year. Initiation of dose escalation in myeloid malignancies occurred in the first half of 2024. Interim Phase 1 data is on track for presentation in the fourth quarter of 2024.
Second Quarter 2024 Financial Results
Cash, Cash Equivalents, and Marketable securities:
Cash, cash equivalents and marketable securities as of June 30, 2024 were $179.8 million. The Company anticipates that its existing cash, cash equivalents and marketable securities will fund Prelude’s operations into 2026.
Research and Development (R&D) Expenses:
For the second quarter of 2024, R&D expense increased to $29.5 million from $25.0 million for the prior year period. Research and development expenses increased primarily due to an increase in our chemistry, manufacturing, and controls (CMC) expense to support our pre-clinical and
clinical research programs. We expect our R&D expenses to vary from quarter to quarter, primarily due to the timing of our clinical development activities.
General and Administrative (G&A) Expenses:
For the second quarter of 2024, G&A expenses increased to $7.7 million from $7.4 million for the prior year period. The increase is primarily due to an increase in professional fees incurred as we expand our operations to support our research and development efforts.
Net Loss:
For the three months ended June 30, 2024, net loss was $34.7 million, or $0.46 per share compared to $30.4 million, or $0.54 per share, for the prior year period. Included in the net loss for the quarter ended June 30, 2024, was $6.1 million of non-cash expenses related to the impact of expensing share-based payments, including employee stock options, as compared to $6.7 million for the same period in 2023.
About Prelude Therapeutics
Prelude Therapeutics is a leading precision oncology company developing innovative medicines in areas of high unmet need for cancer patients. Our pipeline is comprised of several novel drug candidates including first-in-class, highly selective IV and oral SMARCA2 degraders, and a potentially best-in-class CDK9 inhibitor. We are also leveraging our expertise in targeted protein degradation to discover, develop and commercialize next generation degrader antibody conjugates (“Precision ADCs”) with partners. We are on a mission to extend the promise of precision medicine to every cancer patient in need. For more information, visit preludetx.com.
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the "safe harbor" provisions of the Private Securities Litigation Reform Act of 1995, including, but not limited to, anticipated discovery, preclinical and clinical development activities for Prelude’s product candidates, the potential safety, efficacy, benefits and addressable market for Prelude’s product candidates, the expected timeline for initial proof-of-concept data and clinical trial results for Prelude’s product candidates, and the sufficiency of Prelude’s cash runway into 2026. All statements other than statements of historical fact are statements that could be deemed forward-looking statements. The words “believes,” “anticipates,” “estimates,” “plans,” “expects,” “intends,” “may,” “could,” “should,” “potential,” “likely,” “projects,” “continue,” “will,” “schedule,” and “would” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. These forward-looking statements are predictions based on the Company’s current expectations and projections about future events and various assumptions. Although Prelude believes that the expectations reflected in such forward-looking statements are reasonable, Prelude cannot guarantee future events, results, actions, levels of activity, performance or achievements, and the timing and results of biotechnology development and potential regulatory approval is inherently uncertain. Forward-looking statements are subject to risks and uncertainties that may cause Prelude's actual activities or results to differ significantly from those expressed in any forward-looking statement, including risks and uncertainties related to Prelude's ability to advance its
product candidates, the receipt and timing of potential regulatory designations, approvals and commercialization of product candidates, clinical trial sites and our ability to enroll eligible patients, supply chain and manufacturing facilities, Prelude’s ability to maintain and recognize the benefits of certain designations received by product candidates, the timing and results of preclinical and clinical trials, Prelude's ability to fund development activities and achieve development goals, Prelude's ability to protect intellectual property, and other risks and uncertainties described under the heading "Risk Factors" in Prelude’s Annual Report on Form 10-K for the year ended December 31, 2023, its Quarterly Reports on Form 10-Q and other documents that Prelude files from time to time with the Securities and Exchange Commission. These forward-looking statements speak only as of the date of this press release, and Prelude undertakes no obligation to revise or update any forward-looking statements to reflect events or circumstances after the date hereof, except as may be required by law.
PRELUDE THERAPEUTICS INCORPORATED
STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(UNAUDITED)
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended June 30, |
|
(in thousands, except share and per share data) |
|
2024 |
|
|
2023 |
|
Operating expenses: |
|
|
|
|
|
|
Research and development |
|
$ |
29,509 |
|
|
$ |
24,966 |
|
General and administrative |
|
|
7,655 |
|
|
|
7,432 |
|
Total operating expenses |
|
|
37,164 |
|
|
|
32,398 |
|
Loss from operations |
|
|
(37,164 |
) |
|
|
(32,398 |
) |
Other income, net |
|
|
2,424 |
|
|
|
1,967 |
|
Net loss |
|
$ |
(34,740 |
) |
|
$ |
(30,431 |
) |
Per share information: |
|
|
|
|
|
|
Net loss per share of common stock, basic and diluted |
|
$ |
(0.46 |
) |
|
$ |
(0.54 |
) |
Weighted average common shares outstanding, basic
and diluted |
|
|
75,762,152 |
|
|
|
56,240,491 |
|
Comprehensive loss: |
|
|
|
|
|
|
Net loss |
|
$ |
(34,740 |
) |
|
$ |
(30,431 |
) |
Unrealized (loss) gain on marketable securities, net of tax |
|
|
(55 |
) |
|
|
(313 |
) |
Comprehensive loss |
|
$ |
(34,795 |
) |
|
$ |
(30,744 |
) |
PRELUDE THERAPEUTICS INCORPORATED
BALANCE SHEETS
|
|
|
|
|
|
|
|
|
(in thousands, except share data) |
|
June 30,
2024 |
|
|
December 31,
2023 |
|
Assets |
|
(unaudited) |
|
|
|
|
Current assets: |
|
|
|
|
|
|
Cash and cash equivalents |
|
$ |
27,828 |
|
|
$ |
25,291 |
|
Marketable securities |
|
|
152,016 |
|
|
|
207,644 |
|
Prepaid expenses and other current assets |
|
|
2,870 |
|
|
|
2,654 |
|
Total current assets |
|
|
182,714 |
|
|
|
235,589 |
|
Restricted cash |
|
|
4,044 |
|
|
|
4,044 |
|
Property and equipment, net |
|
|
7,554 |
|
|
|
7,325 |
|
Operating lease right-of-use asset |
|
|
29,574 |
|
|
|
30,412 |
|
Other assets |
|
|
405 |
|
|
|
295 |
|
Total assets |
|
$ |
224,291 |
|
|
$ |
277,665 |
|
Liabilities and stockholders’ equity |
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
Accounts payable |
|
$ |
6,170 |
|
|
$ |
4,580 |
|
Accrued expenses and other current liabilities |
|
|
11,426 |
|
|
|
15,768 |
|
Deferred revenue |
|
|
3,000 |
|
|
|
— |
|
Operating lease liability |
|
|
2,232 |
|
|
|
1,481 |
|
Finance lease liability |
|
|
507 |
|
|
|
— |
|
Total current liabilities |
|
|
23,335 |
|
|
|
21,829 |
|
Other liabilities |
|
|
3,215 |
|
|
|
3,339 |
|
Operating lease liability |
|
|
15,465 |
|
|
|
15,407 |
|
Total liabilities |
|
|
42,015 |
|
|
|
40,575 |
|
Commitments |
|
|
|
|
|
|
Stockholders’ equity: |
|
|
|
|
|
|
Voting common stock, $0.0001 par value: 487,149,741 shares authorized; 42,158,224 and 42,063,995 shares issued and outstanding at June 30, 2024 and December 31, 2023, respectively |
|
|
4 |
|
|
|
4 |
|
Non-voting common stock, $0.0001 par value: 12,850,259 shares authorized; 12,850,259 shares issued and outstanding at both June 30, 2024 and December 31, 2023 |
|
|
1 |
|
|
|
1 |
|
Additional paid-in capital |
|
|
705,122 |
|
|
|
693,252 |
|
Accumulated other comprehensive (loss) income |
|
|
(290 |
) |
|
|
223 |
|
Accumulated deficit |
|
|
(522,561 |
) |
|
|
(456,390 |
) |
Total stockholders’ equity |
|
|
182,276 |
|
|
|
237,090 |
|
Total liabilities and stockholders’ equity |
|
$ |
224,291 |
|
|
$ |
277,665 |
|
Investor Contact:
Robert A. Doody, Jr.
Senior Vice President, Investor Relations
Prelude Therapeutics Incorporated
484.639.7235
rdoody@preludetx.com

Corporate Presentation August 2024 Exhibit 99.2

Forward Looking Statements This presentation contains “forward-looking” statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including, but not limited to, anticipated discovery, preclinical and clinical development activities for Prelude’s product candidates, the potential safety, efficacy, benefits and addressable market for Prelude’s product candidates, the expected timeline for proof-of-concept data and clinical trial results for Prelude’s product candidates including its SMARCA2 degrader molecules. Any statements contained herein or provided orally that are not statements of historical fact may be deemed to be forward-looking statements. In some cases, you can identify forward-looking statements by such terminology as ‘‘believe,’’ ‘‘may,’’ ‘‘will,’’ ‘‘potentially,’’ ‘‘estimate,’’ ‘‘continue,’’ ‘‘anticipate,’’ ‘‘intend,’’ ‘‘could,’’ ‘‘would,’’ ‘‘project,’’ ‘‘plan,’’ ‘‘expect’’ and similar expressions that convey uncertainty of future events or outcomes, although not all forward-looking statements contain these words. Statements, including forward-looking statements, speak only to the date they are provided (unless an earlier date is indicated). Certain data in this presentation are based on cross-study comparisons and are not based on any head-to-head clinical trials. Cross-study comparisons are inherently limited and may suggest misleading similarities or differences. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy, nor shall there be any sale of these securities in any state or other jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or other jurisdiction. These forward-looking statements are based on the beliefs of our management as well as assumptions made by and information currently available to us. Although we believe the expectations reflected in such forward-looking statements are reasonable, we can give no assurance that such expectations will prove to be correct. If such assumptions do not fully materialize or prove incorrect, the events or circumstances referred to in the forward-looking statements may not occur. We undertake no obligation to update publicly any forward-looking statements for any reason after the date of this presentation to conform these statements to actual results or to changes in our expectations, except as required by law. Accordingly, readers are cautioned not to place undue reliance on these forward-looking statements. Additional risks and uncertainties that could affect our business are included under the caption “Risk Factors” in our filings with the Securities and Exchange Commission, including our Annual Report on Form 10-K for the year ended December 31, 2023.

We are on a mission to extend the promise of precision �medicine to every cancer patient in need Select the best modality to precisely target oncogenic mechanisms Strive for first- or best-in-class and anchor to patient unmet need Draw on decades of experience and proven leadership to drive innovation
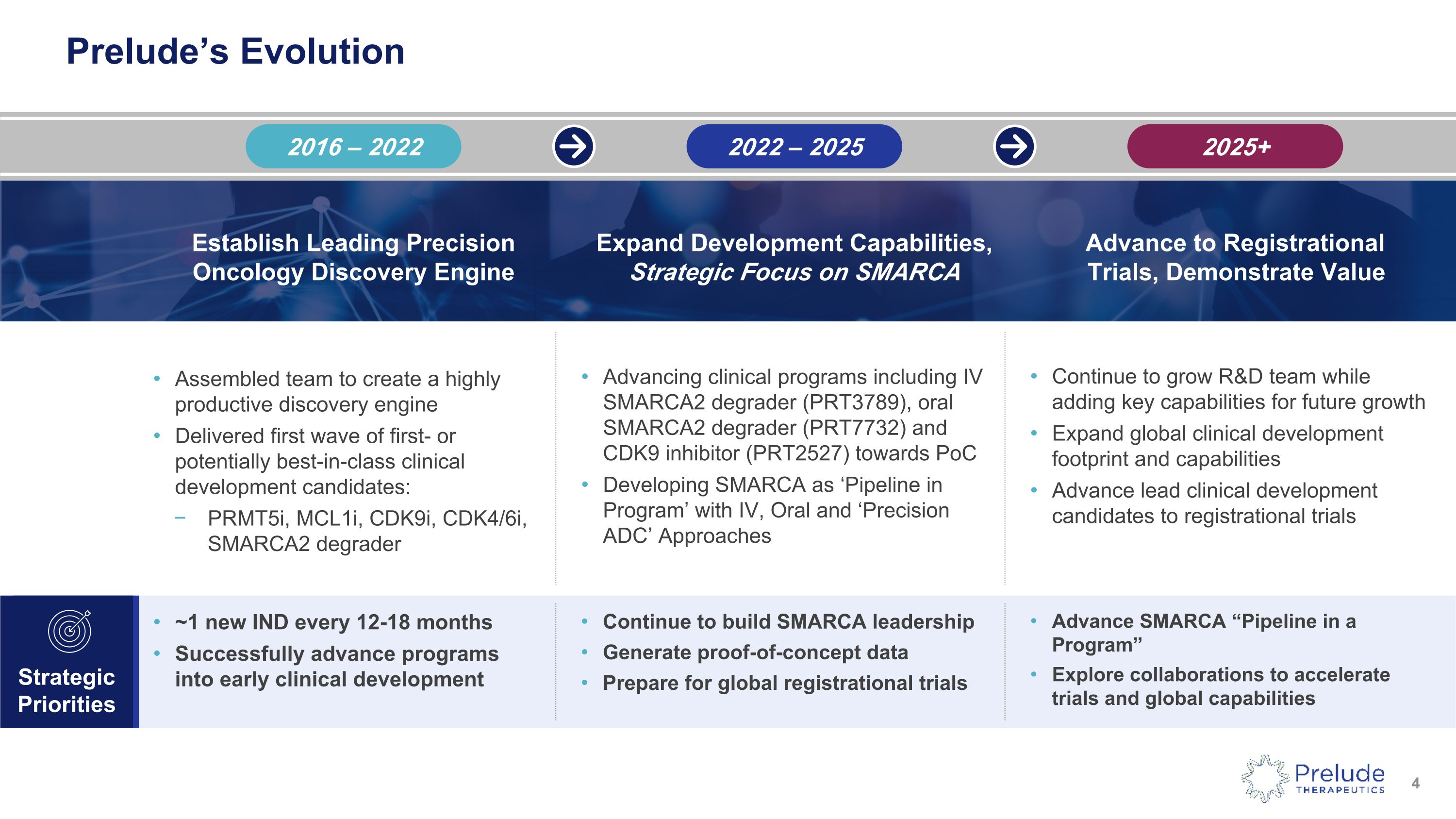
Prelude’s Evolution Strategic Priorities Continue to build SMARCA leadership Generate proof-of-concept data Prepare for global registrational trials Advance SMARCA “Pipeline in a Program” Explore collaborations to accelerate trials and global capabilities ~1 new IND every 12-18 months Successfully advance programs into early clinical development Assembled team to create a highly productive discovery engine Delivered first wave of first- or potentially best-in-class clinical development candidates: PRMT5i, MCL1i, CDK9i, CDK4/6i, SMARCA2 degrader Continue to grow R&D team while adding key capabilities for future growth Expand global clinical development footprint and capabilities Advance lead clinical development candidates to registrational trials Advancing clinical programs including IV SMARCA2 degrader (PRT3789), oral SMARCA2 degrader (PRT7732) and CDK9 inhibitor (PRT2527) towards PoC Developing SMARCA as ‘Pipeline in Program’ with IV, Oral and ‘Precision ADC’ Approaches 2016 – 2022 2022 – 2025 2025+ Establish Leading Precision �Oncology Discovery Engine Expand Development Capabilities, Strategic Focus on SMARCA Advance to Registrational �Trials, Demonstrate Value

Kris Vaddi, PhD Founder & Chief Executive Officer Jane Huang M.D. President and Chief Medical Officer Andrew Combs, PhD Chief Chemistry Officer Sean Brusky, MBA Chief Business Officer Peggy Scherle, PhD Chief Scientific Officer Bryant Lim, J.D. Chief Legal Officer, Corporate Secretary and Interim CFO Experienced Leadership Team With Proven Track Records in Precision Oncology

Prelude’s Precision Medicine Pipeline & Discovery Engine PROGRAM POTENTIAL INDICATIONS DISCOVERY PHASE 1 PHASE 2/3 Lead SMARCA2�Degrader (IV) SMARCA4-mutated NSCLC & other cancers Oral SMARCA2�Degrader SMARCA4-mutated NSCLC & other cancers SMARCA2/4�Precision ADCs* Interim Phase 1 Data at�ESMO Congress 2024 UPCOMING MILESTONES Expand SMARCA Portfolio to Address Cancers Without SMARCA4 Mutations Phase 1 Start Anticipated in Q4 2024 PRT3789 Next-Gen CDK9�Selective Inhibitor Myeloid and B-cell malignancies PRT2527 Interim Phase 1 Data in Q4 2024 Discovery Engine Deliver a First- or Best-in-Class �New Program Every 12-18 Months Hard-to-treat cancers, “undruggable” targets, high unmet need Precision ADCs* Broad range of cancers (heme & solid tumors) Co-Develop Up to 5 Novel Precision ADCs Broad range of cancers (heme & solid tumors) Highly selective, brain-penetrant CDK4-biased inhibitor (PRT3645) available for partnering * Precision ADCs are the focus of our strategic collaboration with AbCellera PRT7732

Prelude’s First-in-Class, Highly Selective SMARCA2 Degraders PRT3789 (IV) and PRT7732 (Oral) Click Here to Access Prelude’s Educational Video Series on SMARCA2 Degraders
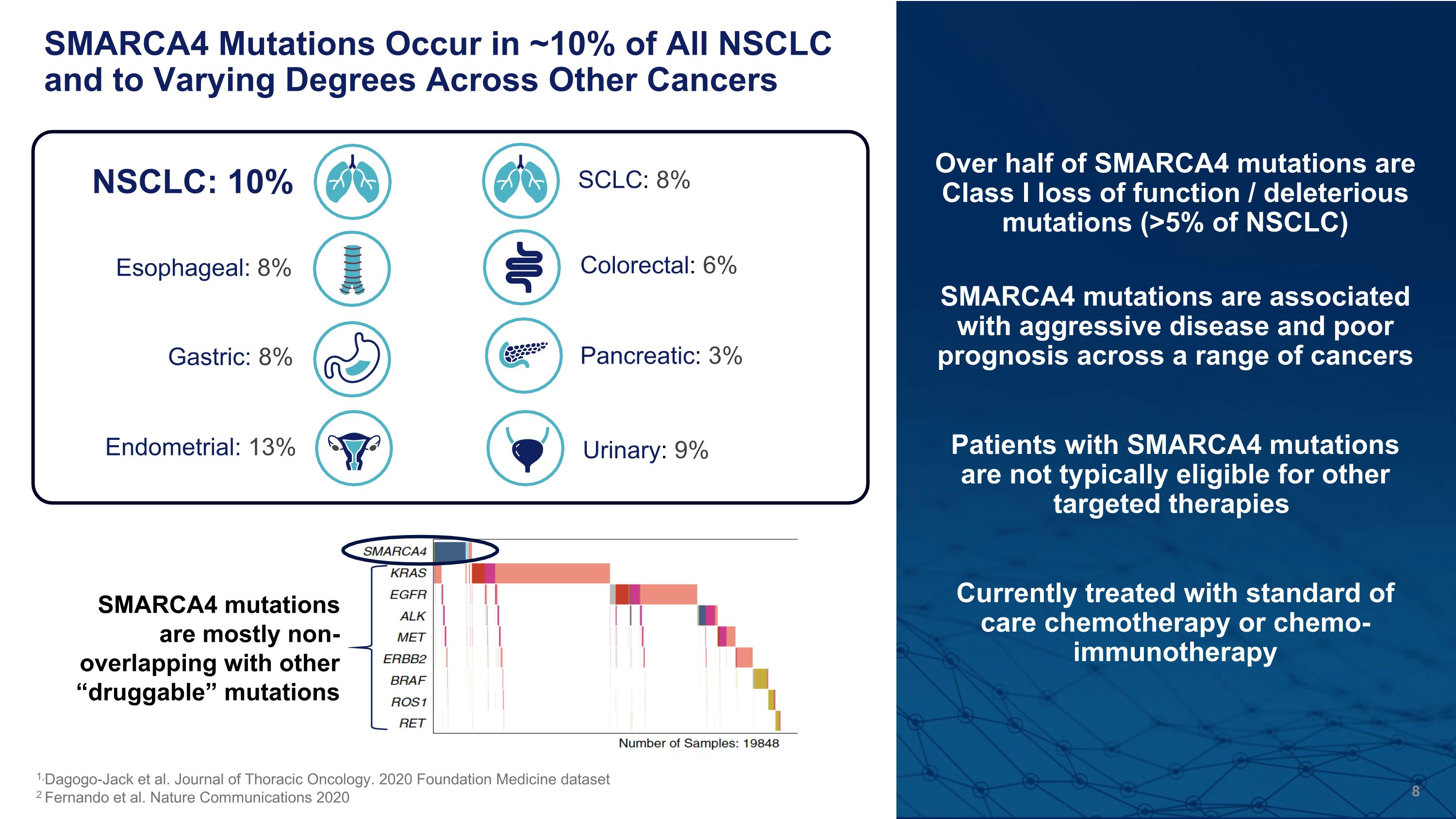
Over half of SMARCA4 mutations are Class I loss of function / deleterious mutations (>5% of NSCLC) SMARCA4 mutations are associated with aggressive disease and poor prognosis across a range of cancers Patients with SMARCA4 mutations are not typically eligible for other targeted therapies Currently treated with standard of care chemotherapy or chemo-immunotherapy SMARCA4 Mutations Occur in ~10% of All NSCLC and to Varying Degrees Across Other Cancers Pancreatic: 3% NSCLC: 10% Esophageal: 8% Gastric: 8% Endometrial: 13% SCLC: 8% Urinary: 9% Colorectal: 6% 1,Dagogo-Jack et al. Journal of Thoracic Oncology. 2020 Foundation Medicine dataset 2 Fernando et al. Nature Communications 2020 SMARCA4 mutations are mostly non-overlapping with other “druggable” mutations
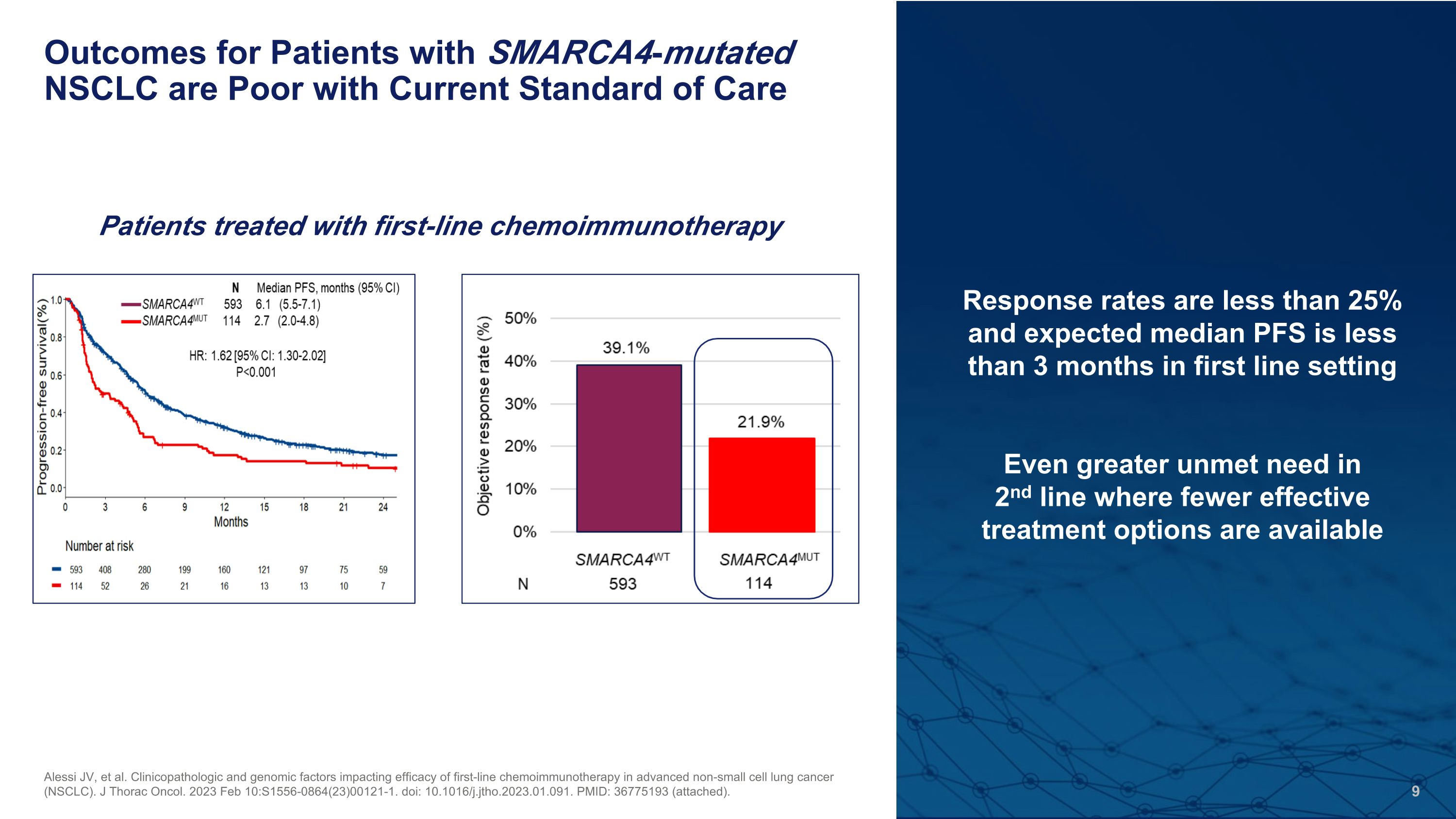
Outcomes for Patients with SMARCA4-mutated NSCLC are Poor with Current Standard of Care Alessi JV, et al. Clinicopathologic and genomic factors impacting efficacy of first-line chemoimmunotherapy in advanced non-small cell lung cancer (NSCLC). J Thorac Oncol. 2023 Feb 10:S1556-0864(23)00121-1. doi: 10.1016/j.jtho.2023.01.091. PMID: 36775193 (attached). Response rates are less than 25% and expected median PFS is less than 3 months in first line setting Even greater unmet need in �2nd line where fewer effective treatment options are available Patients treated with first-line chemoimmunotherapy

Selective Targeting of SMARCA2 is an Attractive Approach to Treat SMARCA4 Mutated Cancers Mutations in the chromatin �remodeling complex drive cancer growth and resistance Cancer cells with SMARCA4 mutations become highly dependent on SMARCA2 for survival Selectively degrading SMARCA2 induces "synthetic lethality" in SMARCA4-deficient cancers High selectivity for SMARCA2 has been challenging because of its high similarity to SMARCA4 SMARCA: SWI/SNF-related, Matrix-associated, Actin-dependent Regulator of Chromatin, subfamily A. SMARCA2 is also known as “BRM” // SMARCA4 is also known as “BRG1”
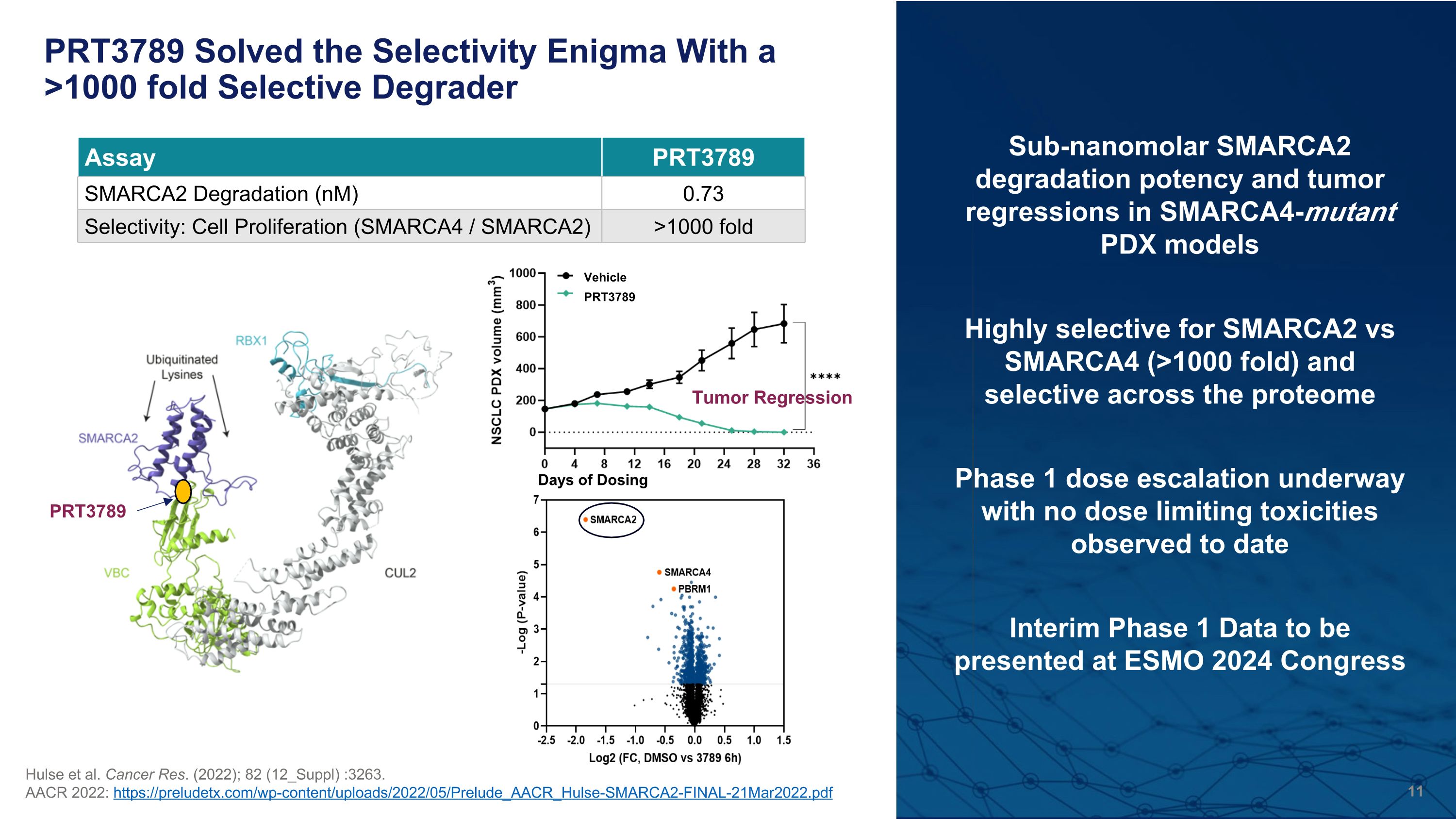
PRT3789 Solved the Selectivity Enigma With a >1000 fold Selective Degrader Hulse et al. Cancer Res. (2022); 82 (12_Suppl) :3263. AACR 2022: https://preludetx.com/wp-content/uploads/2022/05/Prelude_AACR_Hulse-SMARCA2-FINAL-21Mar2022.pdf Highly selective for SMARCA2 vs SMARCA4 (>1000 fold) and�selective across the proteome Assay PRT3789 SMARCA2 Degradation (nM) 0.73 Selectivity: Cell Proliferation (SMARCA4 / SMARCA2) >1000 fold Sub-nanomolar SMARCA2 degradation potency and tumor regressions in SMARCA4-mutant �PDX models PRT3789 Interim Phase 1 Data to be presented at ESMO 2024 Congress Phase 1 dose escalation underway with no dose limiting toxicities observed to date Tumor Regression PRT3789 Vehicle Days of Dosing
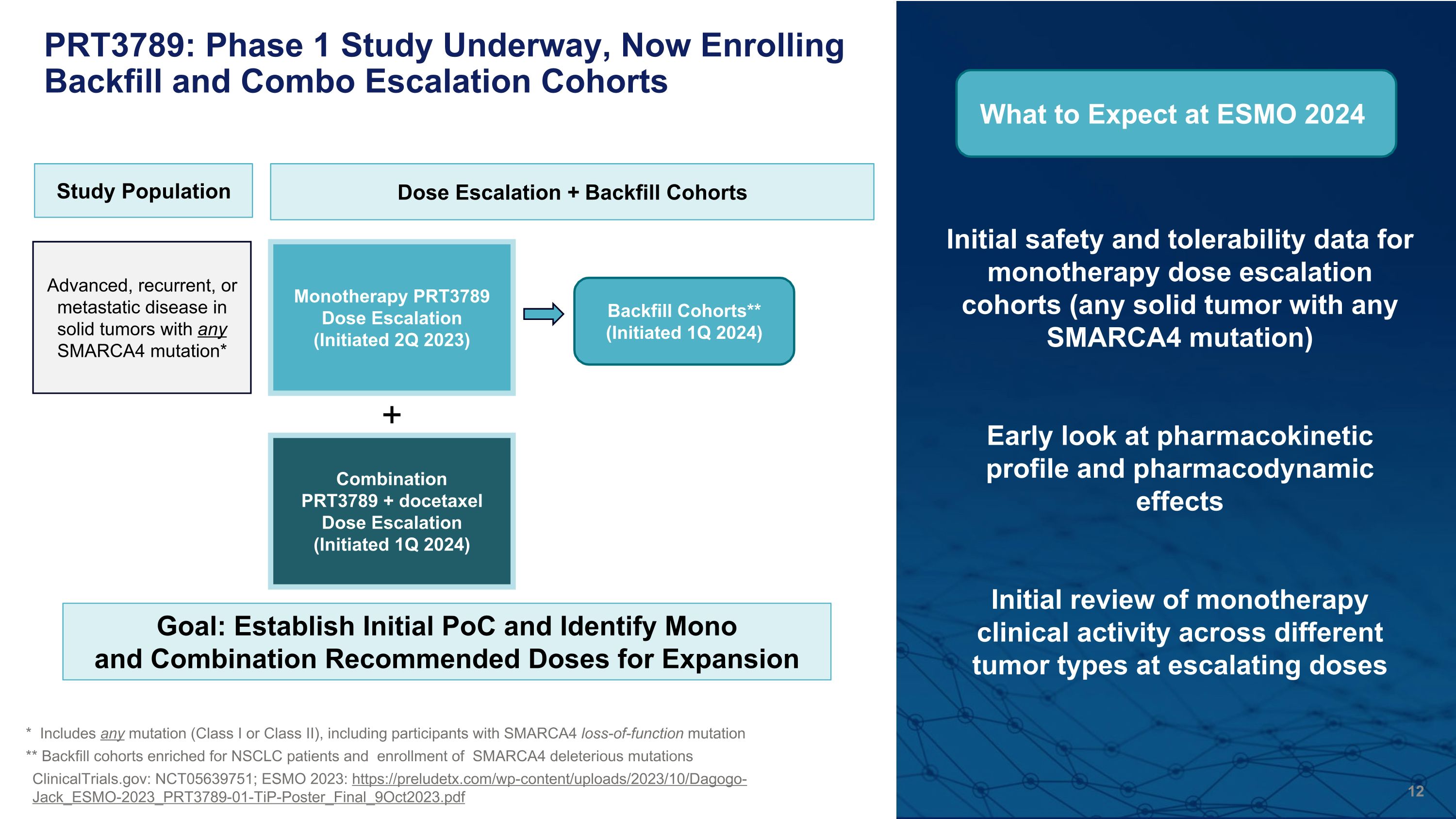
Initial safety and tolerability data for monotherapy dose escalation cohorts (any solid tumor with any SMARCA4 mutation) Early look at pharmacokinetic profile and pharmacodynamic effects Initial review of monotherapy clinical activity across different tumor types at escalating doses * Includes any mutation (Class I or Class II), including participants with SMARCA4 loss-of-function mutation ** Backfill cohorts enriched for NSCLC patients and enrollment of SMARCA4 deleterious mutations ClinicalTrials.gov: NCT05639751; ESMO 2023: https://preludetx.com/wp-content/uploads/2023/10/Dagogo-Jack_ESMO-2023_PRT3789-01-TiP-Poster_Final_9Oct2023.pdf PRT3789: Phase 1 Study Underway, Now Enrolling Backfill and Combo Escalation Cohorts Study Population Advanced, recurrent, or metastatic disease in solid tumors with any SMARCA4 mutation* Dose Escalation + Backfill Cohorts Monotherapy PRT3789 Dose Escalation (Initiated 2Q 2023) Combination�PRT3789 + docetaxel Dose Escalation (Initiated 1Q 2024) Backfill Cohorts** (Initiated 1Q 2024) + What to Expect at ESMO 2024 Goal: Establish Initial PoC and Identify Mono and Combination Recommended Doses for Expansion

Prelude to Initiate Phase 2 Combination Study of PRT3789 + Pembrolizumab in Q4 2024 PRT3789 upregulates genes encoding antigen processing and presentation machinery and may turn ‘cold’ tumors ‘hot’ Trial will explore safety and anti-tumor activity of the combination Preclinical rationale supportive of enhanced efficacy with PRT3789 and anti-PD1 therapy combination KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA.

Next Steps for Development of PRT3789 and Future Directions Full Phase I Trial Results: 2025 Full safety and tolerability data for monotherapy dose escalation, backfill, and chemotherapy combination cohorts Assessment of PK profile and PD effects to support recommended expansion/Phase 2 dose Assessment of clinical activity and ORR for a lead indication at the RDE/RP2D Engagement with regulators on potential registrational trial pathways Future Directions Further evaluation of potential of PRT3789 in combination with both chemotherapy and immunotherapy Potential for use in earlier lines of therapy and potentially early-stage disease as adjuvant or neo-adjuvant therapy Generate evidence across additional tumor types for patients with SMARCA4 mutations
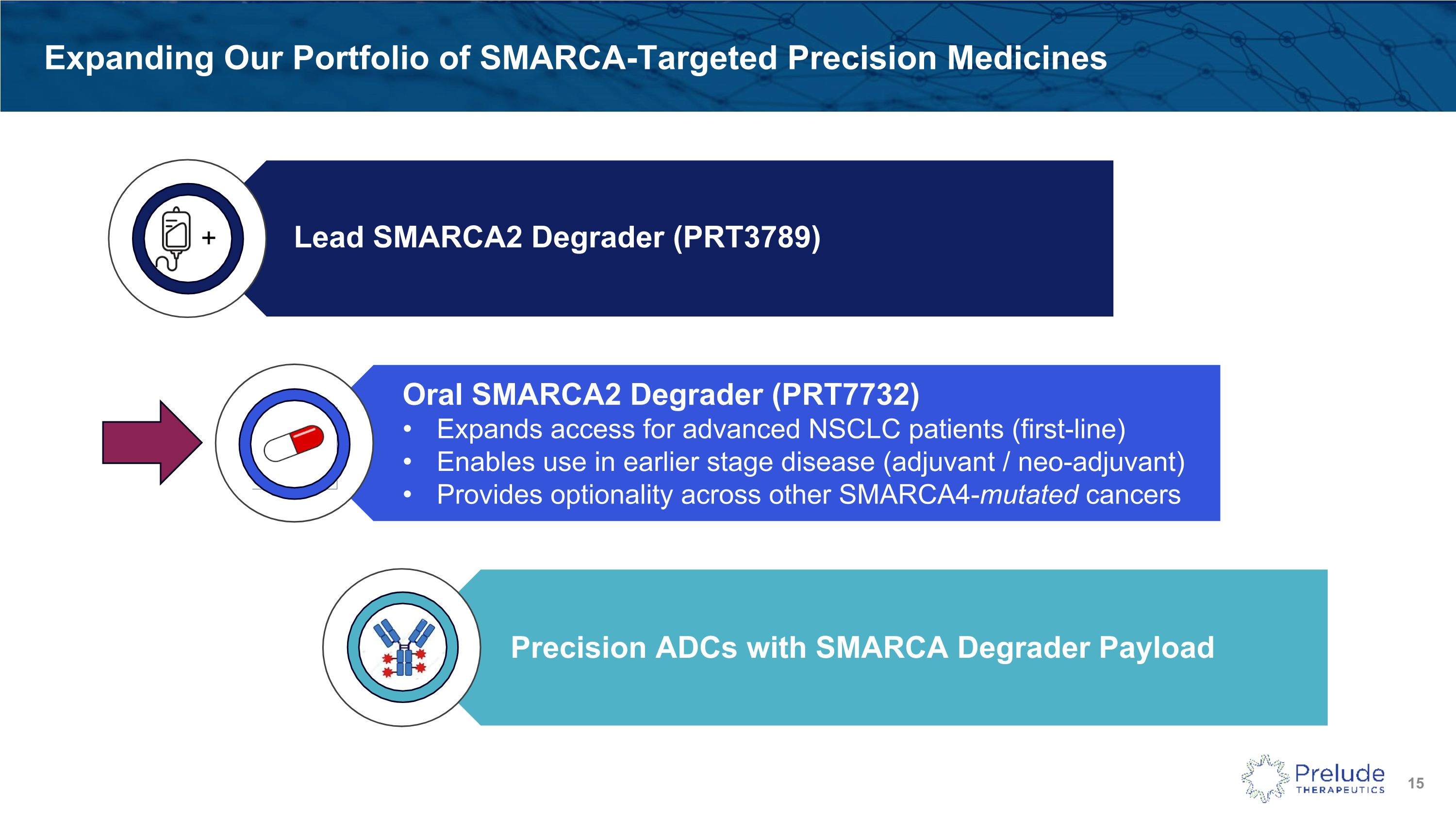
Expanding Our Portfolio of SMARCA-Targeted Precision Medicines SMARCA - DACs + Lead SMARCA2 Degrader (PRT3789) Oral SMARCA2 Degrader (PRT7732) Expands access for advanced NSCLC patients (first-line) Enables use in earlier stage disease (adjuvant / neo-adjuvant) Provides optionality across other SMARCA4-mutated cancers Precision ADCs with SMARCA Degrader Payload
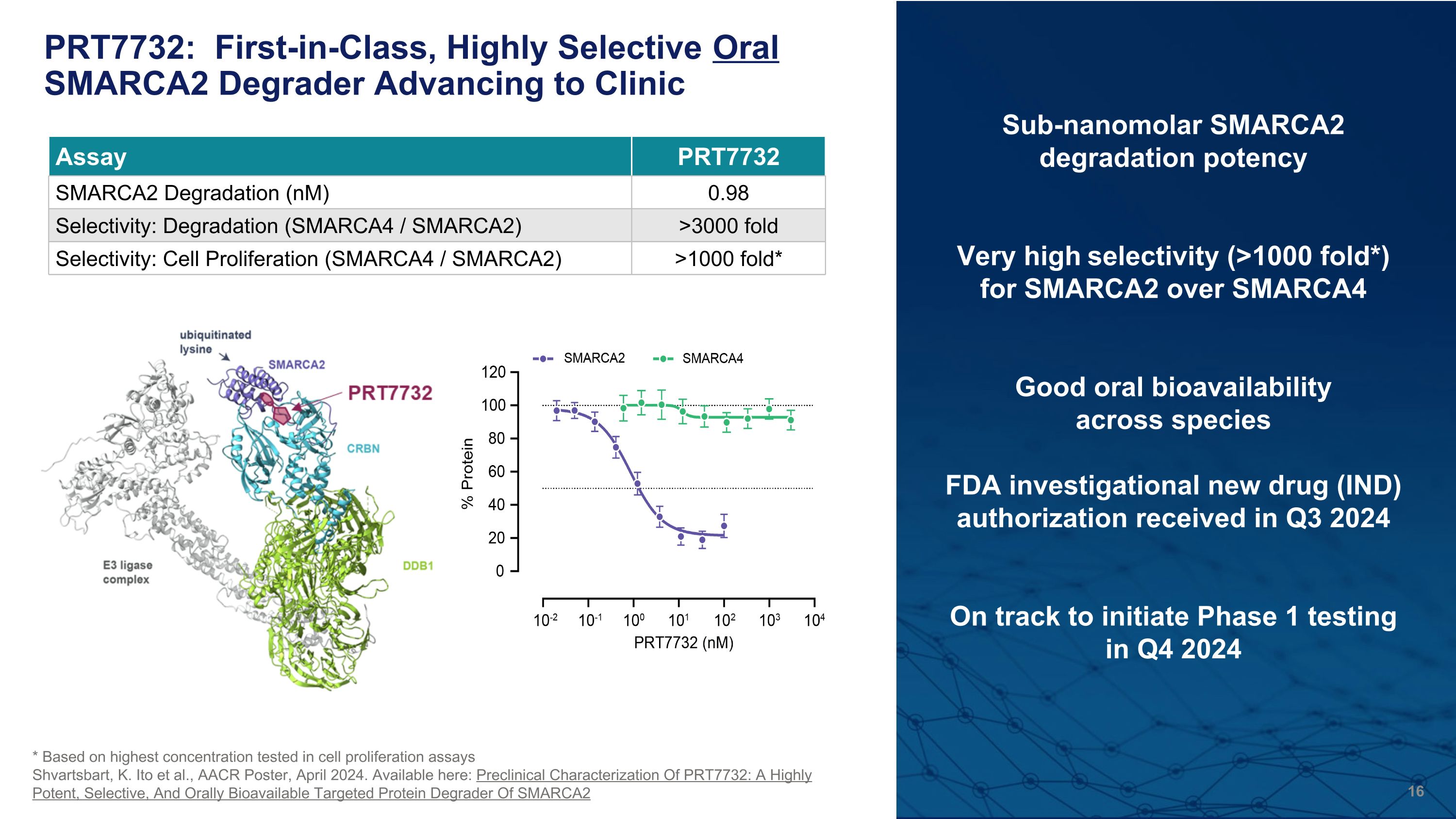
PRT7732: First-in-Class, Highly Selective Oral SMARCA2 Degrader Advancing to Clinic Sub-nanomolar SMARCA2 degradation potency Very high selectivity (>1000 fold*) for SMARCA2 over SMARCA4 Good oral bioavailability �across species FDA investigational new drug (IND) authorization received in Q3 2024 On track to initiate Phase 1 testing �in Q4 2024 * Based on highest concentration tested in cell proliferation assays Shvartsbart, K. Ito et al., AACR Poster, April 2024. Available here: Preclinical Characterization Of PRT7732: A Highly Potent, Selective, And Orally Bioavailable Targeted Protein Degrader Of SMARCA2 Assay PRT7732 SMARCA2 Degradation (nM) 0.98 Selectivity: Degradation (SMARCA4 / SMARCA2) >3000 fold Selectivity: Cell Proliferation (SMARCA4 / SMARCA2) >1000 fold*
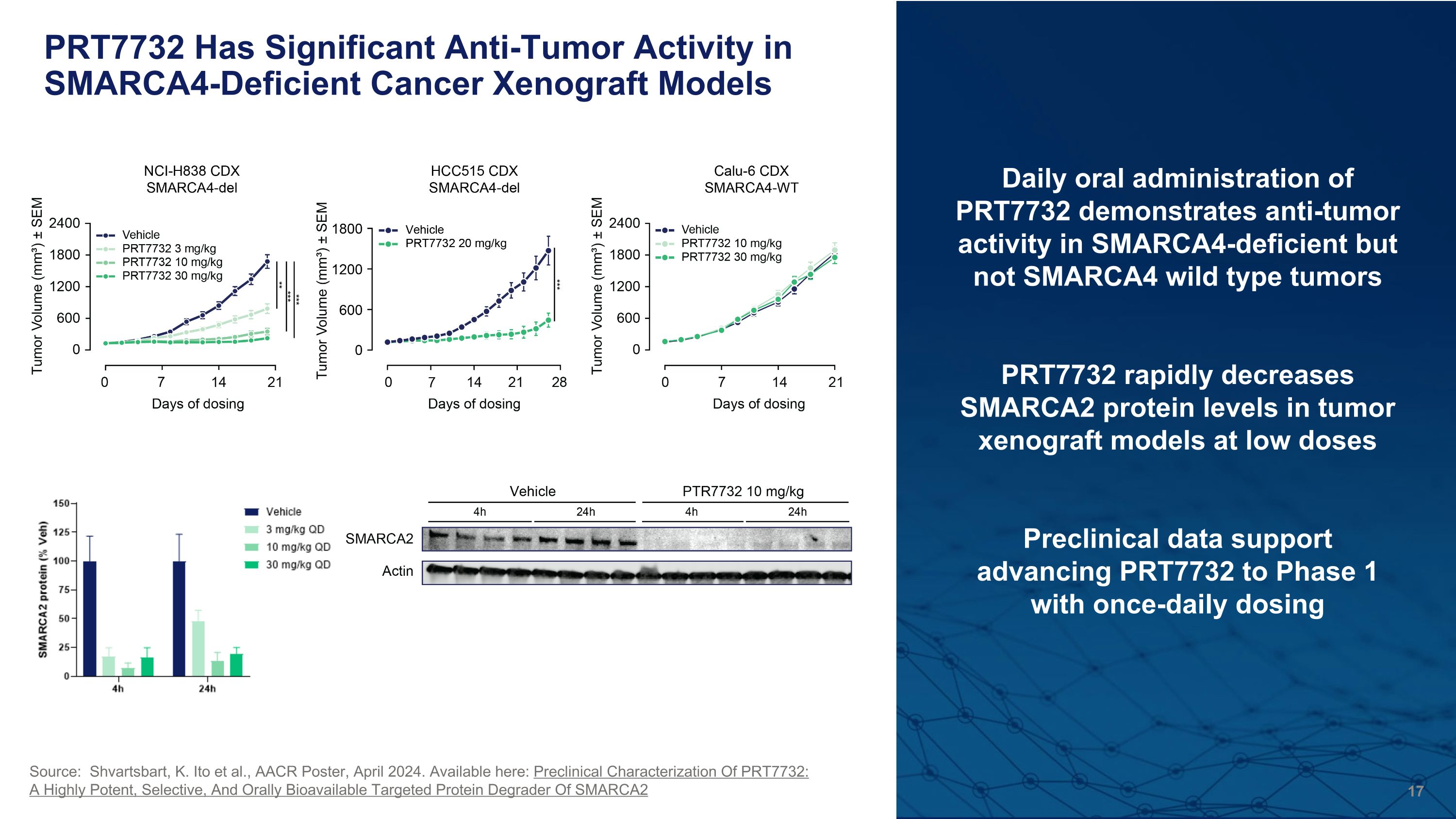
PRT7732 Has Significant Anti-Tumor Activity in SMARCA4-Deficient Cancer Xenograft Models Daily oral administration of PRT7732 demonstrates anti-tumor activity in SMARCA4-deficient but not SMARCA4 wild type tumors PRT7732 rapidly decreases SMARCA2 protein levels in tumor xenograft models at low doses Preclinical data support advancing PRT7732 to Phase 1 with once-daily dosing Source: Shvartsbart, K. Ito et al., AACR Poster, April 2024. Available here: Preclinical Characterization Of PRT7732: A Highly Potent, Selective, And Orally Bioavailable Targeted Protein Degrader Of SMARCA2
Expanding Our Portfolio of SMARCA-Targeted Precision Medicines Precision ADCs with SMARCA Degrader Payload Cancers with dysregulated SMARCA pathway Independent of SMARCA4-mutation status Initial focus of AbCellera collaboration SMARCA - DACs + Lead SMARCA2 Degrader (PRT3789) Oral SMARCA2 Degrader (PRT7732)
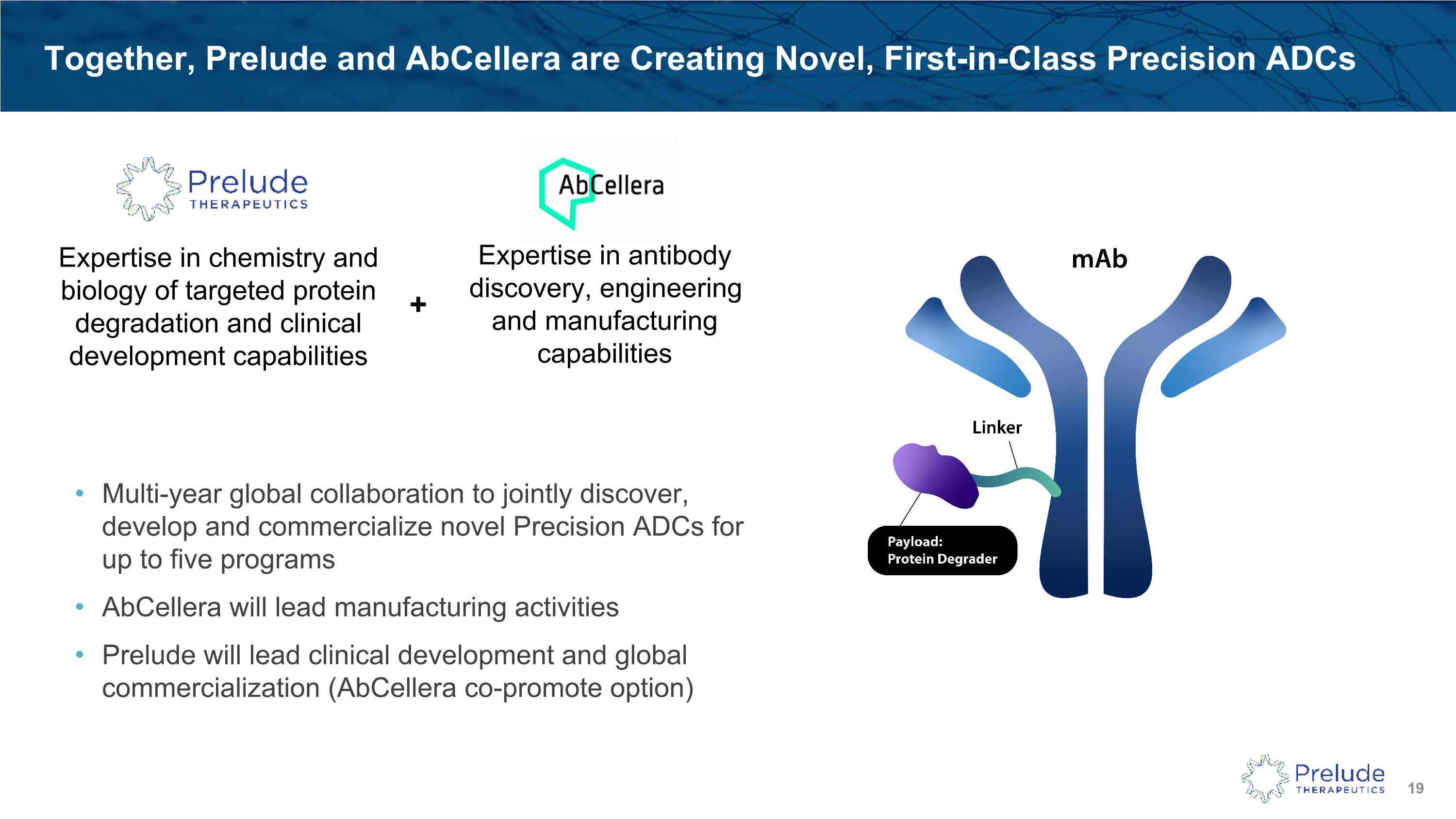
Together, Prelude and AbCellera are Creating Novel, First-in-Class Precision ADCs Multi-year global collaboration to jointly discover, develop and commercialize novel Precision ADCs for up to five programs AbCellera will lead manufacturing activities Prelude will lead clinical development and global commercialization (AbCellera co-promote option) Expertise in chemistry and biology of targeted protein degradation and clinical development capabilities Expertise in antibody discovery, engineering and manufacturing capabilities +
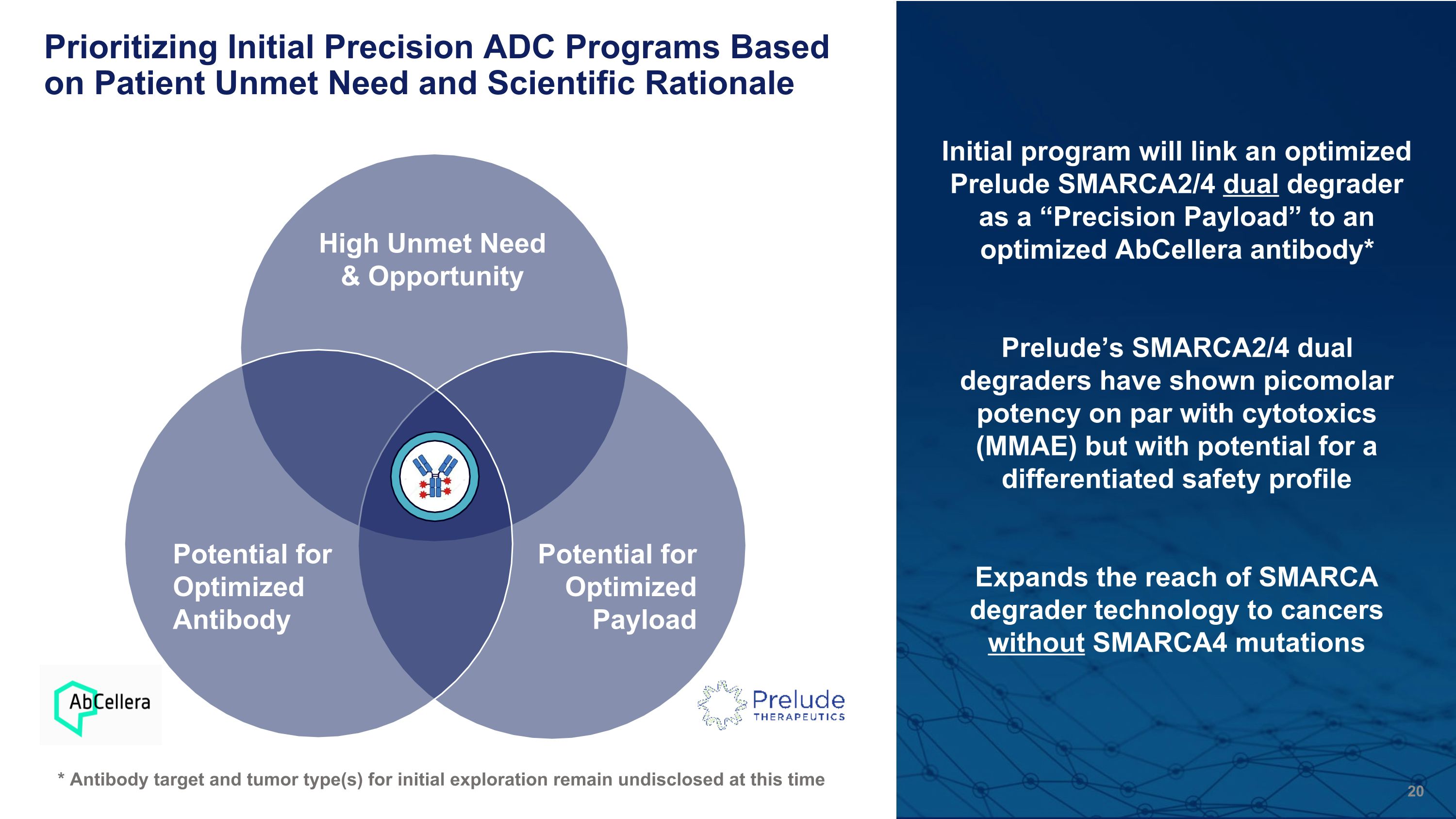
Prioritizing Initial Precision ADC Programs Based on Patient Unmet Need and Scientific Rationale High Unmet Need & Opportunity Potential for Optimized Payload Potential for Optimized Antibody Initial program will link an optimized Prelude SMARCA2/4 dual degrader as a “Precision Payload” to an optimized AbCellera antibody* Prelude’s SMARCA2/4 dual degraders have shown picomolar potency on par with cytotoxics (MMAE) but with potential for a differentiated safety profile Expands the reach of SMARCA degrader technology to cancers without SMARCA4 mutations * Antibody target and tumor type(s) for initial exploration remain undisclosed at this time
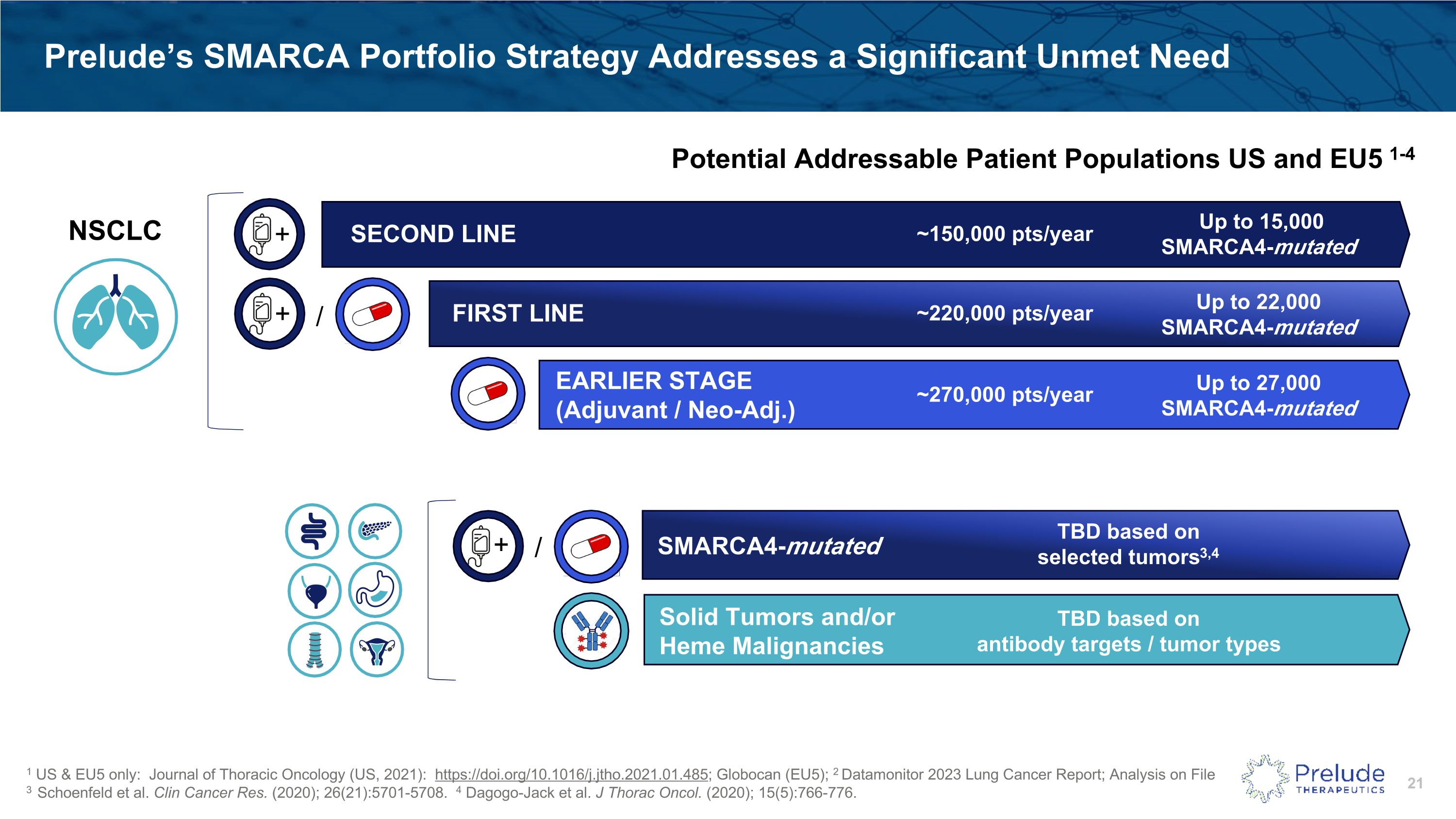
Prelude’s SMARCA Portfolio Strategy Addresses a Significant Unmet Need ~220,000 pts/year Up to 22,000 �SMARCA4-mutated FIRST LINE ~150,000 pts/year Up to 15,000 �SMARCA4-mutated SECOND LINE + 1 US & EU5 only: Journal of Thoracic Oncology (US, 2021): https://doi.org/10.1016/j.jtho.2021.01.485; Globocan (EU5); 2 Datamonitor 2023 Lung Cancer Report; Analysis on File 3 Schoenfeld et al. Clin Cancer Res. (2020); 26(21):5701-5708. 4 Dagogo-Jack et al. J Thorac Oncol. (2020); 15(5):766-776. ~270,000 pts/year Up to 27,000 �SMARCA4-mutated EARLIER STAGE�(Adjuvant / Neo-Adj.) + / Potential Addressable Patient Populations US and EU5 1-4 “Advanced NSCLC” includes patients with Stage IIIB & Stage IV disease1 Excludes patients eligible for other targeted therapy (EGFR, ALK, ROS1, etc.)2 Up to 10% of NSCLC SMARCA4-deficient (includes Class I & II mutations)3,4 Key Assumptions: + / SMARCA4-mutated Solid Tumors and/or Heme Malignancies TBD based on selected tumors3,4 TBD based on �antibody targets / tumor types NSCLC
Highly Selective CDK9 Inhibitor PRT2527
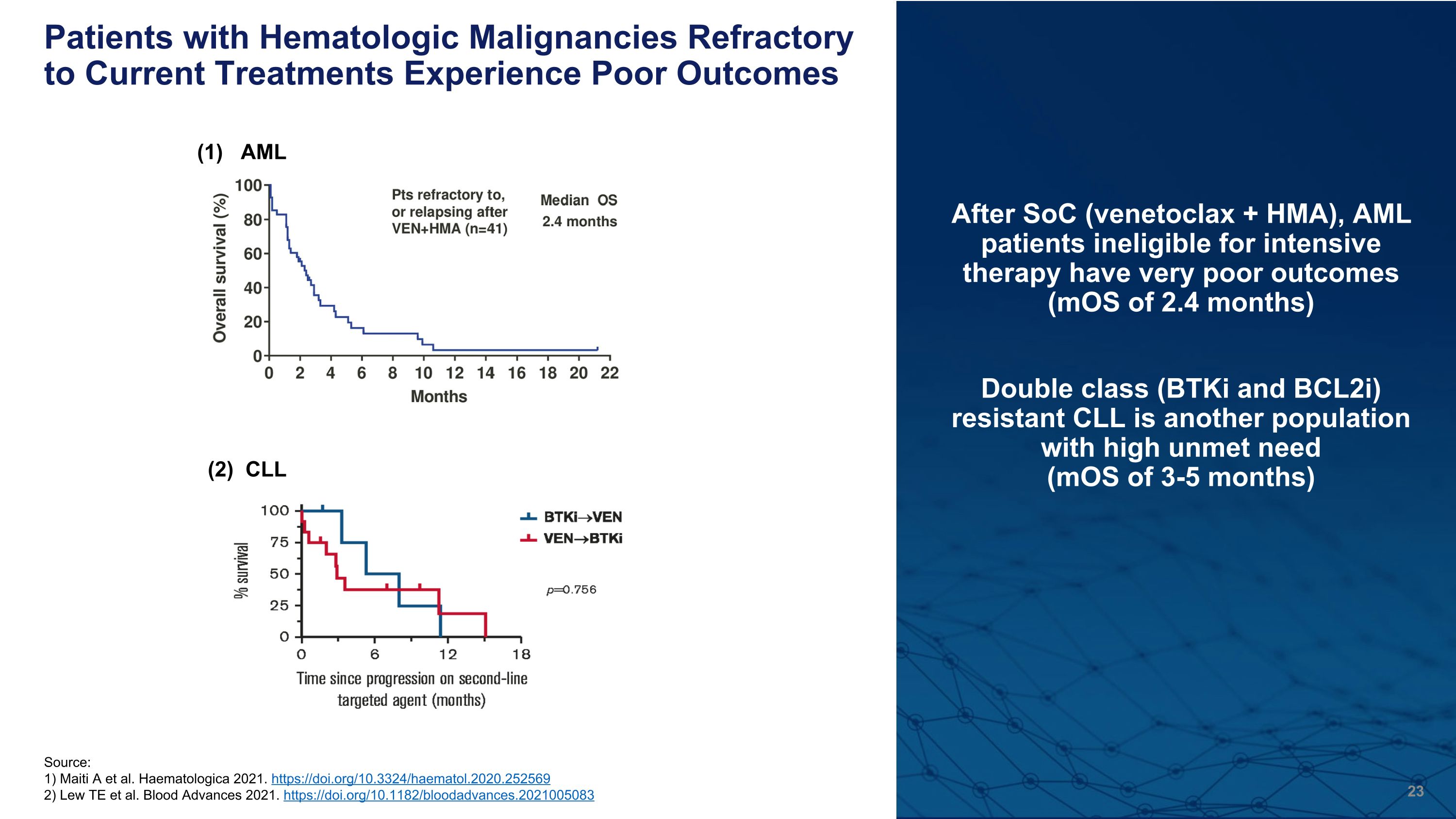
Source: 1) Maiti A et al. Haematologica 2021. https://doi.org/10.3324/haematol.2020.252569 2) Lew TE et al. Blood Advances 2021. https://doi.org/10.1182/bloodadvances.2021005083 Patients with Hematologic Malignancies Refractory to Current Treatments Experience Poor Outcomes After SoC (venetoclax + HMA), AML patients ineligible for intensive therapy have very poor outcomes (mOS of 2.4 months) Double class (BTKi and BCL2i) resistant CLL is another population with high unmet need �(mOS of 3-5 months) Median OS 3.6 months (1) AML (2) CLL
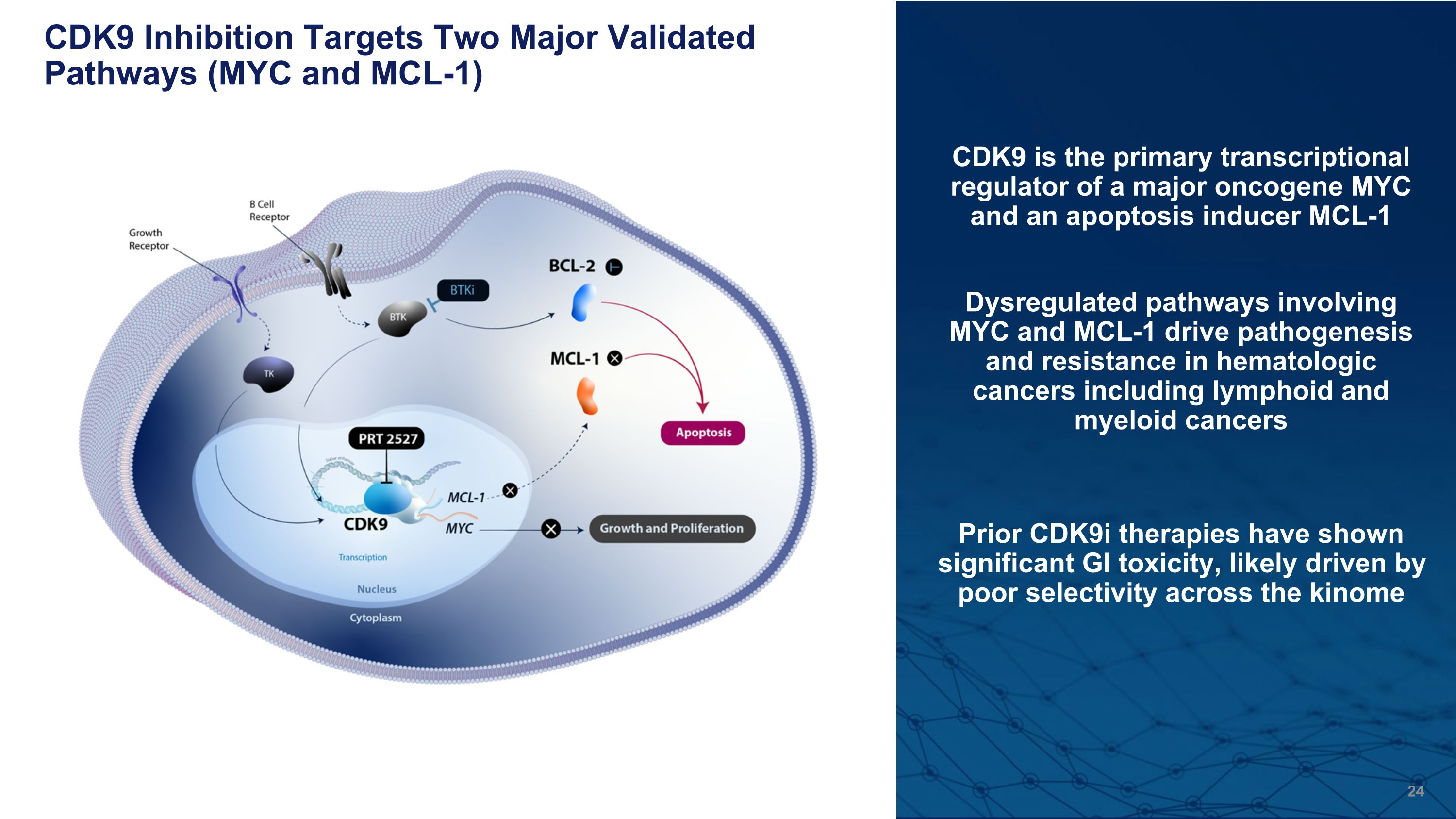
CDK9 Inhibition Targets Two Major Validated Pathways (MYC and MCL-1) CDK9 is the primary transcriptional regulator of a major oncogene MYC and an apoptosis inducer MCL-1 Dysregulated pathways involving MYC and MCL-1 drive pathogenesis and resistance in hematologic cancers including lymphoid and myeloid cancers Prior CDK9i therapies have shown significant GI toxicity, likely driven by poor selectivity across the kinome
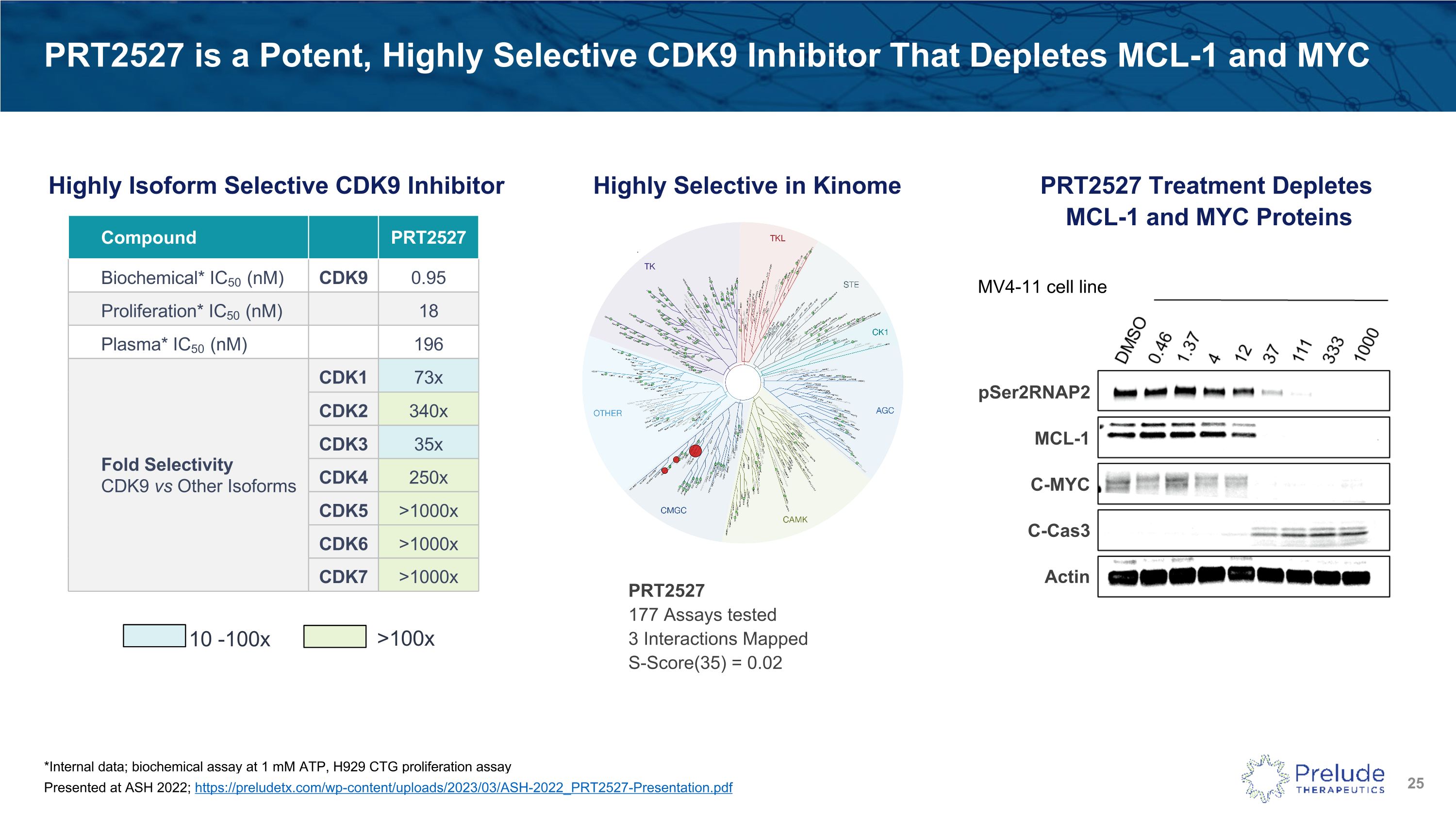
PRT2527 is a Potent, Highly Selective CDK9 Inhibitor That Depletes MCL-1 and MYC Highly Isoform Selective CDK9 Inhibitor Compound PRT2527 Biochemical* IC50 (nM) CDK9 0.95 Proliferation* IC50 (nM) 18 Plasma* IC50 (nM) 196 Fold Selectivity �CDK9 vs Other Isoforms CDK1 73x CDK2 340x CDK3 35x CDK4 250x CDK5 >1000x CDK6 >1000x CDK7 >1000x >100x 10 -100x PRT2527 �177 Assays tested�3 Interactions Mapped�S-Score(35) = 0.02 PRT2527 Treatment Depletes MCL-1 and MYC Proteins Highly Selective in Kinome pSer2RNAP2 MCL-1 C-MYC C-Cas3 Actin MV4-11 cell line *Internal data; biochemical assay at 1 mM ATP, H929 CTG proliferation assay Presented at ASH 2022; https://preludetx.com/wp-content/uploads/2023/03/ASH-2022_PRT2527-Presentation.pdf
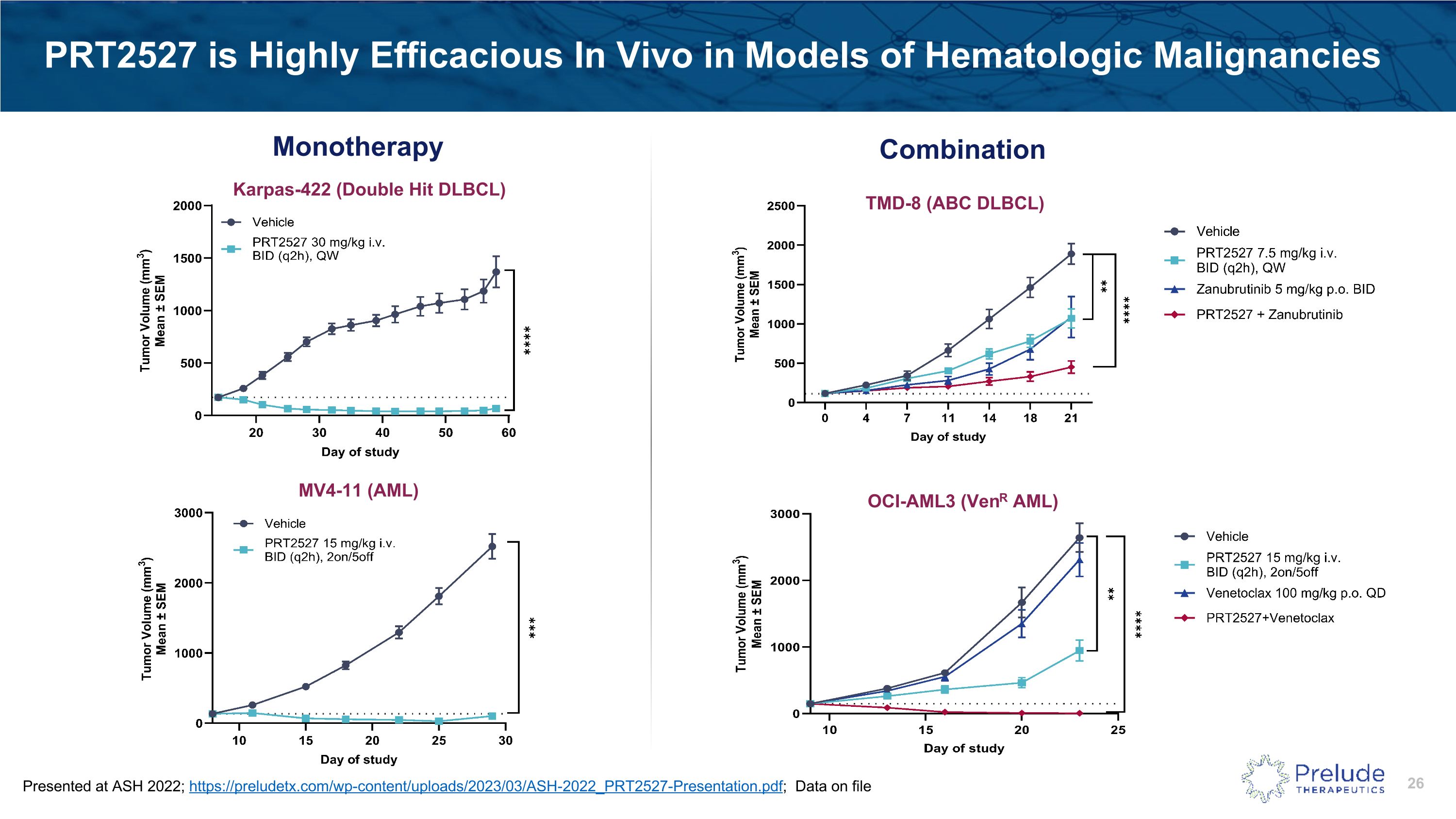
PRT2527 is Highly Efficacious In Vivo in Models of Hematologic Malignancies Monotherapy Karpas-422 (Double Hit DLBCL) MV4-11 (AML) Combination TMD-8 (ABC DLBCL) OCI-AML3 (VenR AML) Presented at ASH 2022; https://preludetx.com/wp-content/uploads/2023/03/ASH-2022_PRT2527-Presentation.pdf; Data on file
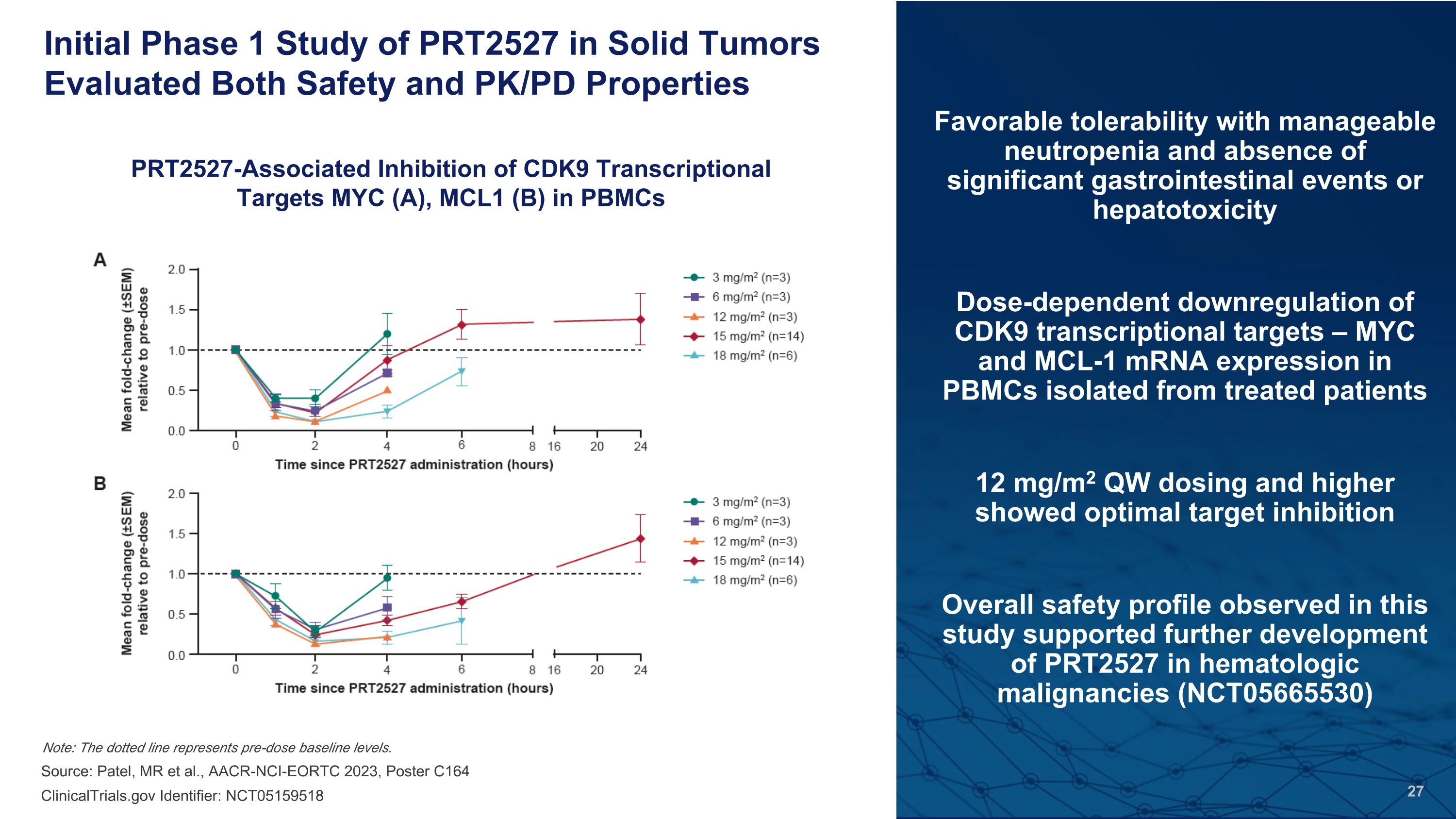
Favorable tolerability with manageable neutropenia and absence of significant gastrointestinal events or hepatotoxicity Dose-dependent downregulation of CDK9 transcriptional targets – MYC and MCL-1 mRNA expression in PBMCs isolated from treated patients 12 mg/m2 QW dosing and higher showed optimal target inhibition Overall safety profile observed in this study supported further development of PRT2527 in hematologic malignancies (NCT05665530) Initial Phase 1 Study of PRT2527 in Solid Tumors Evaluated Both Safety and PK/PD Properties Source: Patel, MR et al., AACR-NCI-EORTC 2023, Poster C164 PRT2527-Associated Inhibition of CDK9 Transcriptional Targets MYC (A), MCL1 (B) in PBMCs Note: The dotted line represents pre-dose baseline levels. ClinicalTrials.gov Identifier: NCT05159518
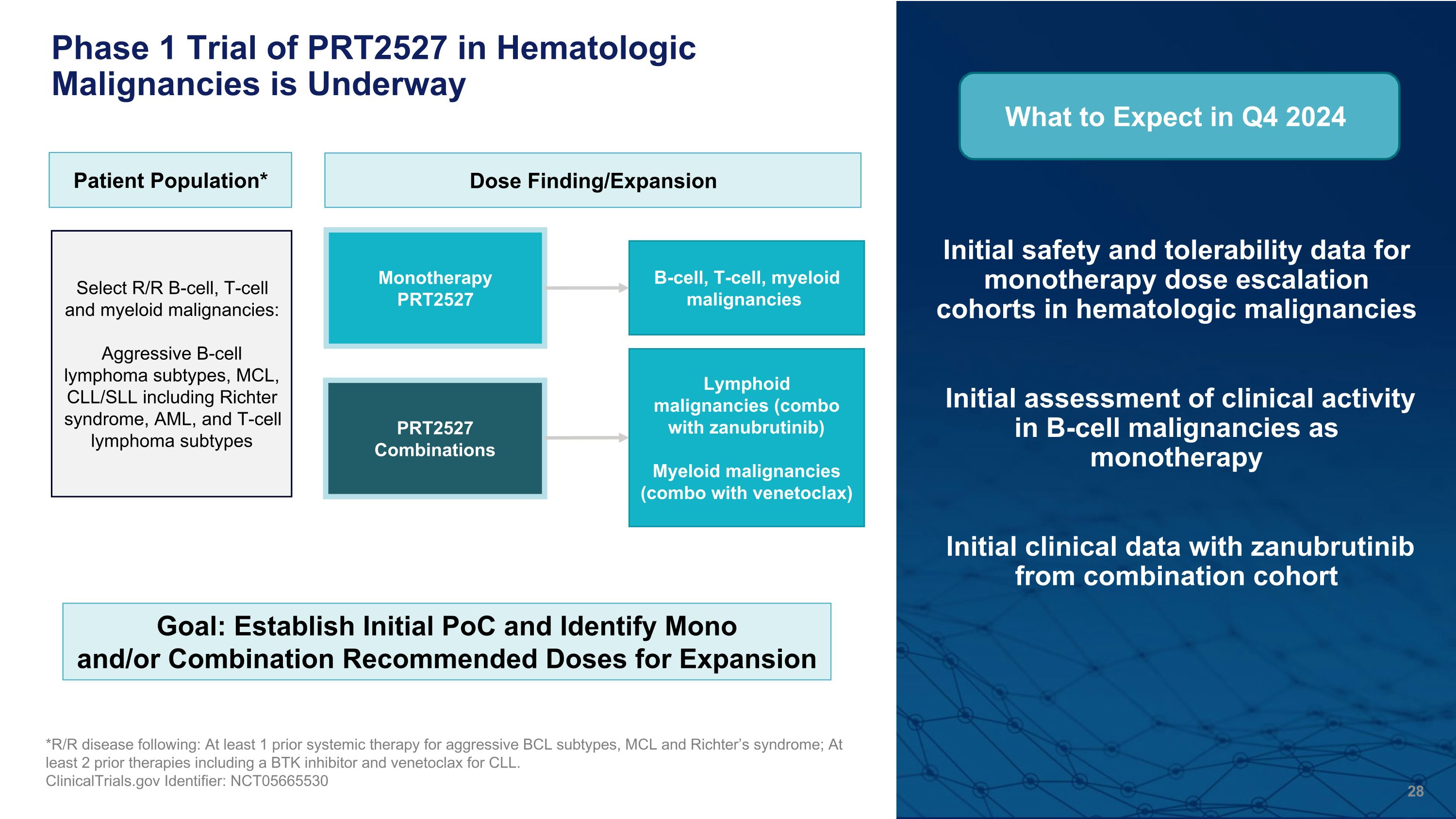
Initial safety and tolerability data for monotherapy dose escalation cohorts in hematologic malignancies Initial assessment of clinical activity in B-cell malignancies as monotherapy Initial clinical data with zanubrutinib from combination cohort *R/R disease following: At least 1 prior systemic therapy for aggressive BCL subtypes, MCL and Richter’s syndrome; At least 2 prior therapies including a BTK inhibitor and venetoclax for CLL. ClinicalTrials.gov Identifier: NCT05665530 Phase 1 Trial of PRT2527 in Hematologic Malignancies is Underway Patient Population* Dose Finding/Expansion Monotherapy PRT2527 Select R/R B-cell, T-cell and myeloid malignancies: Aggressive B-cell lymphoma subtypes, MCL, CLL/SLL including Richter syndrome, AML, and T-cell lymphoma subtypes PRT2527 Combinations What to Expect in Q4 2024 B-cell, T-cell, myeloid malignancies Lymphoid malignancies (combo with zanubrutinib) Myeloid malignancies (combo with venetoclax) Goal: Establish Initial PoC and Identify Mono and/or Combination Recommended Doses for Expansion

Continued Execution Across Strategic Priorities PRT2527 CDK9 Initiate zanubrutinib combination study Initiate myeloid cohort in the existing phase 1 study Complete monotherapy dose escalation in B-cell malignancies Report interim phase 1 clinical results in 2024 PROGRAM Initiate docetaxel combination study cohort Report interim Phase 1 clinical results in 2024 Complete monotherapy escalation and fully enroll backfill cohorts PRT3789 Lead IV SMARCA2 Degrader Selective CDK9 Inhibitor PRT2527 Investigational New Drug (IND) authorization from FDA Initiate Phase 1 in patients with SMARCA4 mutations PRT7732 Oral SMARCA2�Degrader Discovery�Engine Precision ADCs & Other Advance next first-in-class, novel small molecule discovery candidate Advance first SMARCA2/4 Precision ADC in partnership with AbCellera Advance second Precision ADC program in partnership with AbCellera DELIVERABLE MILESTONE Complete Sep 2024 YE 2024 Complete Q4 2024 Complete Complete 2H 2024 Q4 2024 2024 2025 2025 Cash, Cash Equivalents of $179.8 Million as of 6/30/2024

Thank You Contact Us: Robert Doody SVP, Investor Relations rdoody@preludetx.com
v3.24.2.u1
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14a
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Prelude Therapeutics (NASDAQ:PRLD)
Historical Stock Chart
From Feb 2025 to Mar 2025
Prelude Therapeutics (NASDAQ:PRLD)
Historical Stock Chart
From Mar 2024 to Mar 2025