Athira Pharma Announces Promotion of Kevin Church, Ph.D., to Chief Scientific Officer
31 January 2023 - 11:00PM

Athira Pharma, Inc. (NASDAQ: ATHA), a late
clinical-stage biopharmaceutical company focused on developing
small molecules to restore neuronal health and slow
neurodegeneration, today announced the promotion of Kevin Church,
Ph.D., to Chief Scientific Officer.
“Kevin is a driving force behind our research at Athira. He is
instrumental in leading and directing our efforts to elucidate and
validate the promise of the HGF/MET pathway and its potential
applications for the treatment of neurodegenerative disorders,”
said Mark Litton, Ph.D., President and Chief Executive Officer of
Athira. “Kevin’s leadership and dedication to exploring novel
approaches is guided by an unwavering commitment to innovation and
integrity that resonates not only within the research team but
throughout the company.”
Dr. Church joined Athira in 2016 and most recently served as
Executive Vice President, Research. In his current role, he leads
the discovery and preclinical development teams and is responsible
for leading the expansion and development of Athira’s novel
platform of small molecule therapeutic candidates, which offers the
potential to address a wide range of neurological diseases. Dr.
Church received his B.S. in Microbiology from the University of
Idaho and his Ph.D. in Molecular Biosciences from Washington State
University.
About Athira Pharma, Inc.Athira Pharma, Inc.,
headquartered in the Seattle, Washington area, is a late
clinical-stage biopharmaceutical company focused on developing
small molecules to restore neuronal health and slow
neurodegeneration. Athira aims to provide rapid cognitive
improvement and alter the course of neurological diseases with its
novel mechanism of action. Athira is currently advancing its
pipeline of therapeutic candidates targeting the HGF/MET
neurotrophic system for Alzheimer’s and Parkinson’s disease,
Dementia with Lewy bodies and amyotrophic lateral sclerosis (ALS).
For more information, visit www.athira.com. You can also
follow Athira on Facebook, LinkedIn and
@athirapharma on Instagram.
Forward-Looking StatementsThis communication
contains “forward-looking statements” within the meaning of Section
27A of the Securities Act of 1933, Section 21E of the Securities
Exchange Act of 1934 and the Private Securities Litigation Reform
Act of 1995. These forward-looking statements are not based on
historical fact and include statements regarding: product
candidates as a potential treatment for Alzheimer’s disease,
Parkinson’s disease dementia, Dementia with Lewy bodies,
amyotrophic lateral sclerosis, and other neurodegenerative
diseases; Athira’s platform technology and potential therapies;
future development plans; clinical and regulatory objectives and
the timing thereof; expectations regarding the potential efficacy
and commercial potential of Athira’s product candidates; and
Athira’s ability to advance its product candidates into later
stages of development. Forward-looking statements generally include
statements that are predictive in nature and depend upon or refer
to future events or conditions, and include words such as “may,”
“will,” “should,” “on track,” “would,” “expect,” “plan,” “believe,”
“intend,” “pursue,” “continue,” “suggest,” “potential,” and other
similar expressions, among others. Any forward-looking statements
are based on management’s current expectations of future events and
are subject to a number of risks and uncertainties that could cause
actual results to differ materially and adversely from those set
forth in or implied by such forward-looking statements. These risks
and uncertainties include, but are not limited to, the data for our
product candidates from our preclinical and clinical trials not
supporting the safety, efficacy and tolerability of our product
candidates; cessation or delay of Athira’s development of product
candidates may occur; future potential regulatory milestones for
product candidates, including those related to current and planned
clinical studies, may be insufficient to support regulatory
submissions or approval; the impact of the COVID-19 pandemic on
Athira’s business, research and clinical development plans and
timelines, and the regulatory process for Athira product
candidates; Athira may not be able to recruit sufficient patients
for its clinical trials; the outcome of legal proceedings that have
been or may in the future be instituted against us and certain of
our directors and officers; clinical trials may not demonstrate
safety and efficacy of any of Athira’s product candidates; possible
negative interactions of Athira's product candidates with other
treatments; Athira’s assumptions regarding the sufficiency of its
cash, cash equivalents and investments to fund its planned
operations may be incorrect; adverse conditions in the general
domestic and global economic markets; the impact of competition;
regulatory agencies may be delayed in reviewing, commenting on or
approving any of Athira’s clinical development plans as a result of
the COVID-19 pandemic, which could further delay development
timelines; the impact of expanded product development and clinical
activities on operating expenses; the impact of new or changing
laws and regulations; as well as the other risks detailed in
Athira’s filings with the Securities and Exchange Commission. These
forward-looking statements speak only as of the date hereof and
Athira undertakes no obligation to update forward-looking
statements. Athira may not actually achieve the plans, intentions,
or expectations disclosed in its forward-looking statements, and
you should not place undue reliance on the forward-looking
statements.
Investor & Media ContactJulie RathbunAthira
PharmaJulie.rathbun@athira.com206-769-9219
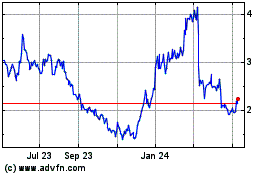
Athira Pharma (NASDAQ:ATHA)
Historical Stock Chart
From Apr 2024 to May 2024
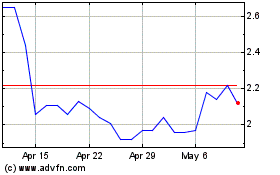
Athira Pharma (NASDAQ:ATHA)
Historical Stock Chart
From May 2023 to May 2024