Bruker Expects its FluoroType® SARS-CoV-2 varID Q PCR Assay to Reliably Detect and Tentatively Differentiate Omicron (B.1.1.529) Variant
03 December 2021 - 3:43AM
Business Wire
- Includes robust dual-target SARS-CoV-2 detection and
identification of 4 S-gene mutations
- Reliable detection and even tentative differentiation of
Omicron variant expected
- Validated with the GenoXtract® and
GenoXtract ® fleXT extraction system
- Validated with Bruker’s novel FluoroCycler® XT
real-time PCR thermocycler
Bruker Corporation (Nasdaq: BRKR) today announced that its
established CE-IVD marked FluoroType® SARS-CoV-2 varID Q
real-time multiplexed PCR assay reliably detects all SARS-CoV-2
variants, and in addition is expected to also provide a clear
indication of Omicron (B.1.1.529), recently declared a ‘variant of
concern’ by the WHO.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20211202005759/en/
FluoroType® SARS-CoV-2 varID Q PCR Kit
(Photo: Business Wire)
FluoroType® SARS-CoV-2 varID Q is a multiplexed CE-IVD
marked PCR assay for the detection and quantification of all major
SARS-CoV-2 variants, based on Bruker´s proprietary LiquidArray®
technology. In addition, the assay simultaneously detects the
S-gene mutations Del69-70 and N501Y, which are expected to be clear
indications that the variant is Omicron (B.1.1.529). The new
Omicron variant shares these mutations with the Alpha variant
(B.1.1.7), but the Alpha variant is meanwhile of low
epidemiological interest and only gets detected sporadically.
Accordingly, by identifying Del69-70 and N501Y, the laboratory has
an indication that the sample most probably is the SARS-CoV-2
Omicron variant. This can subsequently be confirmed by genomic
sequencing, as is standard practice.
The FluoroType® SARS-CoV-2 varID Q also provides a viral
load result according to WHO standard (IU/ml), as well as CP values
and easy interpretation of mutations. The test is validated for use
with nasopharyngeal and oropharyngeal swabs and contains reagents
for 96 reactions. Sample extraction, amplification and PCR results
are available in under 3-4 hours.
FluoroType® SARS-CoV-2 varID Q can be run on the
FluoroCycler® XT PCR instrument after sample preparation
with the GenoXtract® fleXT system, which provides fully
automated extraction and PCR set up. The results from the
FluoroCycler® XT are analysed by the FluoroSoftware®
delivering easy to read results and direct indication of
mutations.
Using the FluoroType® SARS-CoV-2 varID Q assay allows the
user to run a screening tool for detecting SARS-CoV-2, viral load
and an early indication of the Omicron variant.
Moreover, Bruker is pleased to confirm that their full range of
other FluoroType® SARS-CoV-2 assays are also expected to
reliably detect the Omicron variant (B.1.1.529). These assays
include:
CE-IVD assays: FluoroType® SARS-CoV-2 plus*
FluoroType® SARS-CoV-2 varID Q* FluoroType®
SARS-CoV-2/Flu/RSV*
RUO assay: FluoroType® SARS-CoV-2 evo (RUO)* **
* Not for Sale in the USA ** Not for use in clinical diagnostics
procedures
All our SARS-CoV-2 assays are CE-IVD-labeled according to
the European IVD Directive 98/79/EC and mainly sold into European
Markets. FluoroType® SARS-CoV-2 plus is also registered and
sold in some African markets including South Africa.
About Bruker Molecular Diagnostics
Bruker Molecular Diagnostics (MDx) is focused on MDx products
within Bruker´s Microbiology & Diagnostics Business Area. Hain
Lifescience GmbH is the legal manufacturer of the FluoroCycler®
XT, MTBDR 2.0 assay, GXT nucleic acid preparation kits and of
the FluoroType® SARS-CoV-2 assays. For more information:
www.hain-lifescience.de.
About Bruker Corporation (Nasdaq: BRKR)
Bruker is enabling scientists to make breakthrough discoveries
and develop new applications that improve the quality of human
life. Bruker’s high-performance scientific instruments and
high-value analytical and diagnostic solutions enable scientists to
explore life and materials at molecular, cellular and microscopic
levels. In close cooperation with our customers, Bruker is enabling
innovation, improved productivity and customer success in life
science molecular research, in applied and pharma applications, in
microscopy and nanoanalysis, and in industrial applications, as
well as in cell biology, preclinical imaging, clinical phenomics
and proteomics research and clinical microbiology. For more
information, please visit: www.bruker.com.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20211202005759/en/
Investor: T: +1 (978) 663–3660, ext. 1479 E:
investor.relations@bruker.com
Media and Customers: Philip Perry, Bruker Daltonics
Microbiology & Diagnostics T: +49-172-313-7216 E:
Philip.Perry@bruker.com
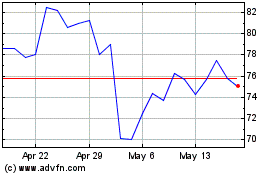
Bruker (NASDAQ:BRKR)
Historical Stock Chart
From Jun 2024 to Jul 2024
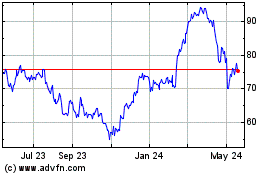
Bruker (NASDAQ:BRKR)
Historical Stock Chart
From Jul 2023 to Jul 2024