Bluebird Bio: FDA Extends Reviews of Beti-Cel, Eli-Cel
19 January 2022 - 12:07AM
Dow Jones News
By Colin Kellaher
Bluebird bio Inc. on Tuesday said the U.S. Food and Drug
Administration has extended by three months its review of the
company's applications seeking approval of a pair of gene
therapies.
The Cambridge, Mass., biotechnology company said the extensions
give the FDA more time to study additional clinical information
bluebird submitted in response to requests by the agency as part of
its reviews.
Bluebird said the revised target action date for betibeglogene
autotemcel, or beti-cel, which the FDA is reviewing for patients
with the inherited blood disorder beta thalassemia, is now Aug. 19,
while the new target date for and elivaldogene autotemcel, or
eli-cel, for cerebral adrenoleukodystrophy, is Sept 16.
Bluebird noted that the extended review isn't related to new
safety events for either drug, and that it doesn't expect the new
target action dates to affect the priority-review status of either
application or the potential for the company to receive
priority-review vouchers upon approval of either drug in 2022.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
January 18, 2022 07:52 ET (12:52 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
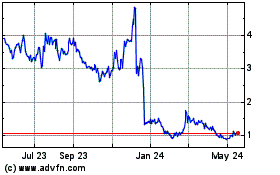
bluebird bio (NASDAQ:BLUE)
Historical Stock Chart
From Apr 2024 to May 2024
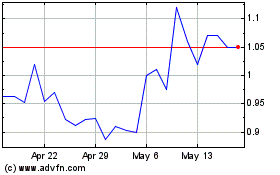
bluebird bio (NASDAQ:BLUE)
Historical Stock Chart
From May 2023 to May 2024