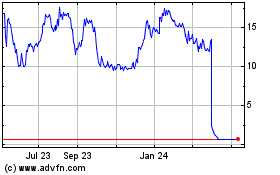
- Pulmonary function measures over 12
months comparable between CVT-301 and observational control
group
- Separate clinical studies assessed
safety profile of CVT-301 in people with asthma, smokers and early
morning OFF
- New Drug Application (NDA) submission
planned by end Q2 2017
Acorda Therapeutics, Inc. (Nasdaq: ACOR) today announced results
from two ongoing, long-term safety studies of CVT-301 in people
with Parkinson’s that showed no differences in pulmonary function
between the group receiving CVT-301 and an observational control
group. These results are consistent with previously reported data
from Phase 2b and Phase 3 clinical trials.
This Smart News Release features multimedia.
View the full release here:
http://www.businesswire.com/news/home/20170329005421/en/
Figure 1 (Graphic: Business Wire)
CVT-301 is an investigational, inhalable formulation of levodopa
(L-dopa). It is being studied as a treatment for symptoms of OFF
periods in people with Parkinson’s taking an oral carbidopa /
levodopa regimen. OFF periods are characterized by the re-emergence
of Parkinson’s symptoms.
“We are delighted with these results, and plan to move forward
with our NDA filing for CVT-301. These two studies, which include
approximately 700 participants, represent the largest safety
database evaluating long-term pulmonary function in people with
Parkinson’s,” said Burkhard Blank, M.D., Chief Medical Officer of
Acorda. “We thank the study volunteers and clinical investigators,
whose willingness to participate in these trials has been essential
to the progress of this program.”
The Company is conducting two separate long-term safety
studies:
- CVT-301-005: a 12-month, randomized,
open-label study in which 271 participants with Parkinson's who did
not have a history of asthma or other chronic lung disease receive
CVT-301 84 mg up to five times daily, along with usual Parkinson’s
standard of care. Safety findings for participants treated with
CVT-301 are compared to an observational control group of 127
participants managed with usual Parkinson’s standard of care. At
the time of this analysis, all ongoing participants completed their
36 week visit and 199 participants completed their 52 week
visit.
- CVT-301-004E: participants receive one
of two doses of CVT-301 (84 mg – 149 participants; 60 mg – 146
participants). There is no control arm in the study. At the time of
this analysis, 70 participants completed their 36 week visit and 49
participants completed their 52 week visit.
Data from both studies will be presented at a future medical
meeting.
The Company plans to file a New Drug Application (NDA) in the
United States by the end of the second quarter of 2017 and, pending
additional data analyses, plans to file a Marketing Authorization
Application (MAA) in Europe by the end of 2017.
CVT-301-005 Detailed Safety Findings
The primary objective of this study is to assess pulmonary
function. Measures include Forced Expiratory Volume in 1 second
(FEV1) and diffusing capacity of the lung for carbon monoxide
(DLCO). Further study details are available at
https://clinicaltrials.gov/ct2/show/NCT02352363.
The mean changes in FEV1 (see Figure 1) and DLCO (see Figure 2)
from baseline to Week 52 in the CVT-301 84 mg group were not
statistically different from the observational control group.
Participants reporting serious adverse events (SAEs) were as
follows: 13 (10.2%) in the observational control arm and 40 (14.9%)
in the CVT-301 84 mg arm. Urinary tract infection occurred in four
participants (1.4%) receiving CVT-301 84 mg. No other SAEs in the
CVT-301 treatment group were reported at greater than 1%. There was
one death in the study, a drowning in the CVT-301 84 mg group,
judged by the investigator to be not related to study drug.
The most common adverse events that were reported in any study
arm at >5% were:
Adverse Event
n (%)
Observational Control
(n=127)
CVT-301 84 mg
(n=271)
Cough 1 (0.8%) 35 (12.9%)
Nasopharyngitis 6 (4.7%) 17 (6.3%)
Dyskinesia 4 (3.1%) 15 (5.5%) Fall
3 (2.4%) 14 (5.2%) Bone Fracture
(various types) 3 (2.4%) 14 (5.2%)
All reported fractures were judged by the investigators to be
unlikely related or not related to study drug. Most reports of
cough were mild (91%), none were severe. Two of 271 participants
(0.7%) receiving CVT-301 discontinued the study due to cough.
Safety findings in the CVT-301-004E study, which did not have an
observational arm, were consistent with those observed among
participants receiving CVT-301 84 mg in the CVT-301-005 study.
Special Population Safety Studies
Asthmatics: The Company conducted a
study to evaluate the safety of CVT-301 in otherwise healthy people
with mild to moderate asthma. In this crossover study, 25
participants received three inhalations of CVT-301 separated by 4
hours, as well as three placebo inhalations separated by 4
hours.
The study found that 10 of the 25 subjects experienced acute
bronchoconstriction when receiving CVT-301, defined as a >15%
decrease in FEV1. This change was reversible, asymptomatic and did
not require rescue treatment. FEV1 in all 10 participants returned
to baseline within 24 hours. The most common adverse event reported
during CVT-301 administration was cough (n=15, 60%), and was
characterized as mild or moderate (20% and 80%, respectively).
Smokers: In a study to assess the
safety of CVT-301 in healthy volunteers who smoked, 25 smokers with
no history of chronic lung disease and 31 healthy non-smokers
received a single dose of CVT-301 84 mg. There were no significant
changes in pulmonary function between the two groups following
administration. The incidence of adverse events was similar across
groups. These events were classified as mild by study investigators
and resolved without treatment. Cough was the most frequently
reported adverse event (60% - smokers; 71% - non-smokers) and was
generally characterized as mild.
Early Morning OFF: A study of 36
participants assessed safety in people with Parkinson’s who
received CVT-301 concurrently with carbidopa / levodopa during
their first OFF period of the day (known as early morning OFF).
Using a cross-over design, participants received a single dose of
CVT-301 and a single dose of placebo. There were no serious adverse
events reported, and no participants discontinued because of
adverse events. There was no difference in the occurrence of
orthostatic hypotension between the groups. Cough was the most
frequently reported adverse event (11% - CVT-301; 2.8% - placebo)
and was generally characterized as mild.
About Parkinson’s Disease and OFF Periods
Approximately one million people in the U.S. and 1.2 million
Europeans are diagnosed with Parkinson’s disease (PD); OFF periods
are experienced by approximately 350,000 in the U.S. and 420,000 in
Europe.
Parkinson’s is a progressive neurodegenerative disorder
resulting from the gradual loss of certain neurons responsible for
producing dopamine. It causes a range of symptoms including
impaired movement, muscle stiffness and tremors. As PD progresses,
people with Parkinson’s experience OFF periods, which are
characterized by the re-emergence of PD symptoms. This re-emergence
can occur even when an individual’s treatment regimen has been
optimized.
OFF periods can be very disruptive to the lives of people with
Parkinson’s, their families and caregivers. OFF periods can
increase in frequency and severity during the course of the
disease.
About CVT-301 and ARCUS®
CVT-301 is being developed as a self-administered, inhaled
levodopa (L-dopa) therapy for the treatment of symptoms of OFF
periods in people with Parkinson’s disease taking an oral carbidopa
/ levodopa regimen.
CVT-301 utilizes Acorda’s investigational ARCUS® platform
for inhaled therapeutics. CVT-301 delivers a precise dose of a dry
powder formulation of L-dopa to the lung. Oral medication can be
associated with variable onset of action, as the medicine is
absorbed through the gastrointestinal (digestive) tract before
reaching the brain. Inhaled treatments enter the body through
the lungs and reach the brain shortly thereafter, bypassing the
digestive system.
About Acorda Therapeutics
Founded in 1995, Acorda Therapeutics is a biotechnology company
focused on developing therapies that restore function and improve
the lives of people with neurological disorders.
Acorda has an industry leading pipeline of novel neurological
therapies addressing a range of disorders, including Parkinson’s
disease, migraine and multiple sclerosis. Acorda markets three
FDA-approved therapies, including AMPYRA® (dalfampridine) Extended
Release Tablets, 10 mg.
For more information, please visit the Company’s website at:
www.acorda.com.
Forward-Looking Statement
This press release includes forward-looking statements. All
statements, other than statements of historical facts, regarding
management's expectations, beliefs, goals, plans or prospects
should be considered forward-looking. These statements are subject
to risks and uncertainties that could cause actual results to
differ materially, including: the ability to realize the benefits
anticipated from the Biotie and Civitas transactions, among other
reasons because acquired development programs are generally subject
to all the risks inherent in the drug development process and our
knowledge of the risks specifically relevant to acquired programs
generally improves over time; the ability to successfully integrate
Biotie’s operations and Civitas’ operations, respectively, into our
operations; we may need to raise additional funds to finance our
expanded operations and may not be able to do so on acceptable
terms; our ability to successfully market and sell Ampyra
(dalfampridine) Extended Release Tablets, 10 mg in the U.S.; third
party payers (including governmental agencies) may not reimburse
for the use of Ampyra or our other products at acceptable rates or
at all and may impose restrictive prior authorization requirements
that limit or block prescriptions; the risk of unfavorable results
from future studies of Ampyra or from our other research and
development programs, including CVT-301 or any other acquired or
in-licensed programs; we may not be able to complete development
of, obtain regulatory approval for, or successfully market CVT-301,
any other products under development, or the products that we will
acquire when we complete the Biotie transaction; the occurrence of
adverse safety events with our products; delays in obtaining or
failure to obtain and maintain regulatory approval of or to
successfully market Fampyra outside of the U.S. and our dependence
on our collaborator Biogen in connection therewith; competition;
failure to protect our intellectual property, to defend against the
intellectual property claims of others or to obtain third party
intellectual property licenses needed for the commercialization of
our products; and failure to comply with regulatory requirements
could result in adverse action by regulatory agencies.
These and other risks are described in greater detail in our
filings with the Securities and Exchange Commission. We may not
actually achieve the goals or plans described in our
forward-looking statements, and investors should not place undue
reliance on these statements. Forward-looking statements made in
this press release are made only as of the date hereof, and we
disclaim any intent or obligation to update any forward-looking
statements as a result of developments occurring after the date of
this press release.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20170329005421/en/
Acorda Therapeutics, Inc.Jeff Macdonald,
914-326-5232jmacdonald@acorda.com
Acorda Therapeutics (NASDAQ:ACOR)
Historical Stock Chart
From Apr 2024 to May 2024
Acorda Therapeutics (NASDAQ:ACOR)
Historical Stock Chart
From May 2023 to May 2024