Revance Shares Slide After FDA Rejects DAXI Application
18 October 2021 - 9:41PM
Dow Jones News
By Colin Kellaher
Shares of Revance Therapeutics Inc. tumbled in premarket trading
Monday after the biotechnology company said the U.S. Food and Drug
Administration had turned away its injection treatment for frown
lines due to issues related to the drug's manufacturing plant.
The Newark, Calif., company late Friday said the FDA had issued
a complete response letter, indicating that the agency wouldn't
approve the application for DaxibotulinumtoxinA for Injection, or
DAXI, in its current form.
Revance said the FDA indicated that it found deficiencies during
its delayed inspection of its manufacturing plant in California,
adding that the agency didn't identify any other issues.
Revance said it plans to request a meeting with the FDA as soon
as possible to address the deficiencies, adding that it is "very
disappointed by this unanticipated response."
Analysts at Mizuho said the latest development in the DAXI saga
likely means a delay of about 12 to 15 months for approval of the
drug.
The FDA in early 2020 had accepted Revance's application for
DAXI, with a decision expected before the end of the year, but the
agency later deferred its decision because it hadn't been able to
inspect the manufacturing plant due to travel restrictions related
to the Covid-19 pandemic. The FDA eventually conducted its
pre-approval inspection in late June and early July of this
year.
Revance shares fell sharply early last week after the FDA,
responding to a Freedom of Information Act request, released a
document outlining concerns raised by the inspection, but the stock
rebounded Wednesday after Revance said it had already responded,
and that it still expected FDA approval of DAXI by the end of this
year.
Mizuho said it now assumes a late 2022 approval of the drug,
with a launch in early 2023. The analysts cut their price target on
Revance shares to $26 from $36 to reflect the delay, but they
maintained their "buy" rating, saying they still expect DAXI will
ultimately win FDA approval and represents a long-term
opportunity.
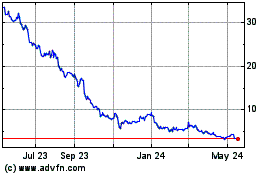
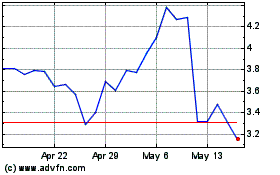
Revance shares, which closed Friday at $22.71, were recently
down nearly 32% to $15.47 in premarket trading.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
October 18, 2021 06:26 ET (10:26 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
Revance Therapeutics (NASDAQ:RVNC)
Historical Stock Chart
From Mar 2024 to Apr 2024
Revance Therapeutics (NASDAQ:RVNC)
Historical Stock Chart
From Apr 2023 to Apr 2024