Vanda Pharmaceuticals Gets Notice of Opportunity for FDA Meeting
08 September 2022 - 9:34AM
Dow Jones News
By Josh Beckerman
Vanda Pharmaceuticals Inc. received a notice of opportunity for
a Food and Drug Administration hearing on its Supplemental New Drug
Application for Hetlioz to treat jet lag disorder.
Hetlioz is currently available for treating Non-24-Hour
Sleep-Wake Disorder and nighttime sleep disturbances in
Smith-Magenis Syndrome. In 2019, the company received an FDA
complete response letter for its sNDA for treating jet lag
disorder.
Vanda said Wednesday that it "provided the FDA with four
clinical studies that Vanda believes are adequate and well
controlled and provide substantial evidence of efficacy." The
company said it "does not agree with the FDA's characterization of
these studies."
Shares rose 2% to $10.50 after hours.
Write to Josh Beckerman at josh.beckerman@wsj.com
(END) Dow Jones Newswires
September 07, 2022 19:19 ET (23:19 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
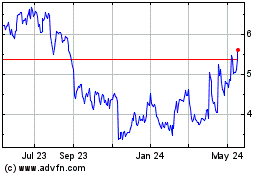
Vanda Pharmaceuticals (NASDAQ:VNDA)
Historical Stock Chart
From Apr 2024 to May 2024
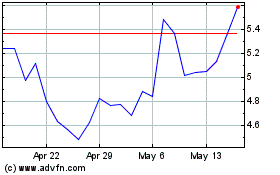
Vanda Pharmaceuticals (NASDAQ:VNDA)
Historical Stock Chart
From May 2023 to May 2024