Zenas BioPharma Launches as a Cross-Border Biopharmaceutical Company to Bring Innovation to the Development and Delivery of I...
23 March 2021 - 7:00PM

Zenas BioPharma (“Zenas”) launched today as a cross-border
biopharmaceutical company committed to becoming a global leader in
the development and delivery of immune-based therapies for patients
in China and throughout the world.
Zenas was founded and initially funded by Tellus BioVentures,
LLC (“Tellus”) and Fairmount Funds Management LLC (“Fairmount”) and
is additionally backed by a syndicate of leading global life
science investment funds including Quan Capital, WuXi Biologics
Healthcare Ventures and Wellington Management.
Lonnie Moulder, Managing Member of Tellus and Founder of Zenas,
serves as Executive Chairman and Interim Chief Executive Officer.
As a veteran life sciences founder and executive, he brings to
Zenas a track record of success and the necessary skills to
navigate the global biopharmaceutical landscape. Mr. Moulder has
assembled an experienced leadership team with strong credentials in
acquiring, developing and commercializing products across the
globe.
“We are excited about the launch of Zenas BioPharma and its
pipeline of differentiated product candidates. These potentially
best-in-class products are intended to bring innovation to patients
with underserved diagnoses while also maximizing value across the
healthcare ecosystem,” said Mr. Moulder. “The expanding Zenas
leadership team and our network of business partners will drive
operational excellence to deliver potentially transformative
therapies to improve the lives of people living with autoimmune and
rare diseases.”
“We are thrilled to invest in and support this new venture lead
by a proven and experienced team,” added Marietta Wu, MD, Ph.D.,
MBA, Managing Director at Quan Capital and Zenas Board Member.
“Immunology is an area of transformative innovation poised to solve
great unmet medical needs. We are confident that by leveraging
cross border research and development capabilities and resources,
Zenas will rapidly advance its current portfolio and establish its
global leadership with an expanded pipeline coupled with a strong
footprint in China.”
Rapidly Advancing Pipeline
Zenas debuts with a deep pipeline of seven innovative
immune-based therapeutics that will continue to expand through a
successful business development strategy. Zenas owns worldwide
rights to three of the seven pipeline programs, which uniquely
positions the Company to be global in scope.
Zenas innovation is defined by the selection of product
candidates that are designed with best-in-class potential and then
advanced utilizing differentiated development and commercial
strategies to address patient needs AND the value requirements of
the dynamic healthcare environment. Zenas innovation further
includes acquiring or discovering first-in-class product candidates
based upon new knowledge in the evolving field of immunology.
ZB001 and ZB011
Zenas is developing its lead product candidate ZB001, an
insulin-like growth factor-1 receptor (IGF1R) monoclonal antibody
(mAb), for the potential treatment of patients in China with
thyroid eye disease (TED). TED is a debilitating condition that
significantly impacts quality of life and can cause proptosis
(“bulging eyes”), double vision and vision loss. ZB001, a potential
first-in-class product in China, was exclusively in-licensed from
Viridian Therapeutics, Inc. (“Viridian”) (NASDAQ: VRDN).
More than 100 oncology patients have previously been treated
with the antibody in US and EU studies, under the name AVE-1642,
enabling understanding of its pharmacokinetic and pharmacodynamic
profile, as well as its safety and tolerability. Viridian maintains
all rights outside of China and is developing the IGF1R mAb under
the name VRDN-001. This program is advancing rapidly with an
expected Investigational New Drug (“IND”) application filing by
Viridian in late 2021, followed by a Zenas filing in China. Data
from the first clinical trial involving patients is expected to be
available in 2022.
In parallel with ZB001 development, Viridian and Zenas initiated
the ZB011 program, which seeks to improve IGF1R-targeted antibodies
to further enhance dosing and convenience.
ZB002, ZB003 and ZB004
The Zenas pipeline also includes three potentially best-in-class
mAbs (ZB002, ZB003, ZB004) exclusively in-licensed worldwide from
Xencor, Inc. (NASDAQ: XNCR). These three mAbs were engineered with
XmAb™ technology for improved potency, half-life, and stability and
are undergoing IND-enabling studies to support clinical development
for both new and established autoimmune disease indications.
ZB005 and ZB006
Zenas additionally entered a collaboration with Dianthus
Therapeutics, Inc. (“Dianthus”) that provides China rights for two
mAb programs, ZB005 and ZB006. Dianthus is a privately held
biotechnology company focused on the discovery and development of
monoclonal antibody therapeutics that modulate the immune
complement system for the treatment of autoimmune diseases.
IND-enabling activities are underway for ZB005, the first of the
two programs.
About Zenas BioPharma
Zenas BioPharma is a cross-border (China-USA based)
biopharmaceutical company committed to becoming a global leader in
the development and delivery of immune-based therapies for patients
in China and around the world. Zenas is rapidly advancing a deep
pipeline of innovative therapeutics that continues to grow through
our successful business development strategy. Our experienced
leadership team and network of business partners drive operational
excellence to deliver potentially transformative therapies to
improve the lives of those facing autoimmune and rare diseases. For
more information about Zenas BioPharma, please visit
www.zenasbio.com and follow us on Twitter at @ZenasBioPharma and
LinkedIn.
Contacts
Investor Contact:
Zenas BioPharmaIR@zenasbio.com
Media Contact:
Lauren BartlettZenas BioPharmalauren.bartlett@zenasbio.com
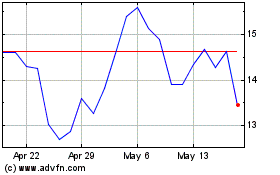
Viridian Therapeutics (NASDAQ:VRDN)
Historical Stock Chart
From Jun 2024 to Jul 2024
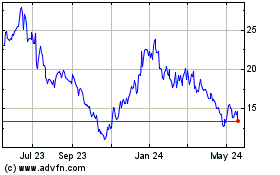
Viridian Therapeutics (NASDAQ:VRDN)
Historical Stock Chart
From Jul 2023 to Jul 2024