UNITED STATES
SECURITIES AND
EXCHANGE COMMISSION
Washington, D.C.
20549
FORM 10-K
(Mark One)
|
|
x
|
Annual
Report Pursuant to Section 13 or 15(d) of The Securities Exchange Act of 1934
|
For the Fiscal Year Ended March 31, 2019
Or
|
|
¨
|
Transition Report
Pursuant to Section 13 or 15(d) of The Securities Exchange Act of 1934
|
For the transition period from_____to _____
Commission File Number: 000-54717
Bionik
Laboratories Corp.
(Exact name of registrant as specified
in its charter)
|
Delaware
|
27-1340346
|
|
(State or other jurisdiction of
|
(I.R.S. Employer
|
|
incorporation or organization)
|
Identification No.)
|
483 Bay Street N105, Toronto, Ontario
Canada M5G 2C9
(Address of principal executive offices)
(Zip Code)
Registrant’s telephone number, including
area code:
(416) 640-7887 x 508
Securities registered pursuant to Section
12(b) of the Act:
|
Title of each class
|
|
Trading Symbol(s)
|
|
Name of Exchange on which
registered
|
|
N/A
|
|
N/A
|
|
N/A
|
Securities registered pursuant to Section
12(g) of the Act: Common Stock, $0.001 par value
Indicate by check mark if the registrant
is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes
¨
No
x
Indicate by check mark if the registrant
is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes
¨
No
x
Indicate by check mark whether the registrant:
(1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding
12 months (or for such shorter period that the registrant was required to file such reports), and has been subject to such filing
requirements for the past 90 days. Yes
x
No
¨
Indicate by check mark whether the registrant
has submitted electronically, every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§
232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit
such files). Yes
x
No
¨
Indicate by check mark whether the registrant
is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging growth
company. See definitions of “large accelerated filer”, “accelerated filer”, “smaller reporting company”
and “emerging growth company” in Rule 12b-2 of the Exchange Act.
|
Large accelerated filer
|
¨
|
Accelerated filer
|
¨
|
|
Non-accelerated filer
|
x
|
Smaller reporting company
|
x
|
|
|
Emerging Growth Company
|
¨
|
If an emerging growth company, indicate
by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial
accounting standards provided pursuant to 13(a) of the Exchange Act.
Indicate by check mark whether the registrant
is a shell company (as defined in Rule 12b-2 of the Act). Yes
¨
No
x
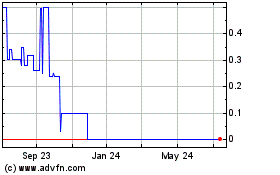
The aggregate market value of the voting
and non-voting common equity held by non-affiliates of the registrant based on the closing sales price, or the average bid and
asked price on such stock, at September 30, 2018 was $7,968,899.
The number of shares of the registrant’s
common stock outstanding as of June 27, 2019 was 3,702,398 shares of common stock, par value $0.001 per share.
BIONIK LABORATORIES CORP.
TABLE OF CONTENTS
BASIS OF PRESENTATION
Unless otherwise
noted, references in this Annual Report on Form 10-K to “Bionik,” the “Company,” “we,” “our,”
or “us” means Bionik Laboratories Corp., and, unless the context otherwise requires, together with its subsidiaries,
Bionik Laboratories, Inc., a Canadian corporation (“Bionik Canada”) and Bionik, Inc., a Massachusetts corporation
(formerly Interactive Motion Technologies, Inc., “IMT”). References to Bionik Canada refer to such company prior to
its acquisition by the Company on February 26, 2015 and references to IMT refer to such company prior to its acquisition by the
Company on April 21, 2016.
CAUTIONARY STATEMENT
REGARDING FORWARD-LOOKING STATEMENTS
The information
contained in this Annual Report on Form 10-K, including in documents that may be incorporated by reference into this Report, includes
some statements that are not purely historical and that are “forward-looking statements.” Such forward-looking statements
include, but are not limited to, statements regarding the Company and its management’s expectations, hopes, beliefs, intentions
or strategies regarding the future, including its financial condition and results of operations. In addition, any statements that
refer to projections, forecasts or other characterizations of future events or circumstances, including any underlying assumptions,
are forward-looking statements. The words “anticipates,” “believes,” “continue,” “could,”
“estimates,” “expects,” “intends,” “may,” “might,” “plans,”
“possible,” “potential,” “predicts,” “projects,” “seeks,” “should,”
“will,” “would” and similar expressions, or the negatives of such terms, may identify forward-looking
statements, but the absence of these words does not mean that a statement is not forward-looking.
The forward-looking
statements contained in this Annual Report on Form 10-K are based on current expectations and beliefs concerning future developments.
There can be no assurance that future developments actually affecting the Company will be those anticipated. These forward-looking
statements involve a number of risks, uncertainties (some of which are beyond the parties’ control) or other assumptions
that may cause actual results or performance to be materially different from those expressed or implied by these forward- looking
statements, some of which are described in the Section of this Form 10-K entitled “Risk Factors”.
Should one
or more of these risks or uncertainties materialize, or should any of the Company’s assumptions prove incorrect, actual
results may vary in material respects from those projected in these forward-looking statements. The Company undertakes no obligation
to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except
as may be required under applicable securities laws.
CAUTIONARY NOTE
REGARDING INDUSTRY DATA
Unless otherwise
indicated, information contained in this Annual Report on Form 10-K concerning our company, our business, the services we provide
and intend to provide, our industry and our general expectations concerning our industry are based on management estimates. Such
estimates are derived from publicly available information released by third party sources, as well as data from our internal research,
and reflect assumptions made by us based on such data and our knowledge of the industry, which we believe to be reasonable.
ITEM 1. BUSINESS
Company Overview
Bionik Laboratories
Corp. is a healthcare company focused on improving the quality of life of millions of people with neurological or mobility impairments
by combining artificial intelligence and innovative robotics technology to help individuals from hospital to home to regain mobility,
enhance autonomy, and regain self-esteem.
The Company uses artificial
intelligence and machine learning technologies to make rehabilitation methods and processes smarter and more intuitive to deliver
greater recovery for patients with neurological or mobility impairments. These technologies allow large amounts of data to be
collected and processed in real-time, enabling appropriately challenging and individualized therapy during every treatment session.
This is the foundation of the InMotion™ therapy. The Company’s rehabilitation therapy robots are built on an artificial
intelligence platform, measuring the position, the speed and the acceleration of the patient 200 times per second. The artificial
intelligence platform is designed to adapt in real time to the patient’s needs and progress while providing quantifiable
feedback of a patient’s progress and performance, in a way that the Company believes a trained clinician cannot.
Based on this foundational
work, the Company has a portfolio of products focused on upper and lower extremity rehabilitation for stroke and other mobility-impaired
individuals, including three InMotion™ robots currently in the market and two products in varying stages of development.
The InMotion™
therapy uses the Company’s robots to assist patients to rewire a segment of their brains after injury, also known as neuroplasticity.
The InMotion™ Robots - the InMotion
™
ARM, InMotion
™
WRIST and the InMotion
™
ARM/HAND
– are designed to provide intelligent, adaptive therapy in a manner that has been clinically shown to maximize neurorecovery.
The Company is also developing a home version of the InMotion™ upper-body rehabilitation technology, as well as a lower-body
wearable assistive product based on the Company’s existing ARKE lower body exoskeleton technology, which could allow certain
mobility impaired individuals to walk better. The Company intends to launch this mobility assistance solution into the consumer
market when the Company has sufficient funds to develop this product.
The InMotion
™
,
ARM InMotion
™
ARM/HAND, and InMotion
™
WRIST are robotic therapies for the upper limbs. InMotion™
robotic therapies have been characterized as Class II medical devices by the U.S. Food and Drug Administration, or FDA, and are
listed with the FDA to market and sell in the United States. More than 280 of our clinical robotic products for stroke rehabilitation
have been sold in over 15 countries, including the United States. In addition to these fully developed, clinical rehabilitation
solutions, we are also developing “InMotion™ Home”, which is an upper extremity product that allows the patient
to extend their therapy for as long as needed while rehabilitating at home. This rehabilitation solution is being developed on
the same design platform as the InMotion™ clinical products.
We believe recent
payment changes in the US marketplace proposed and finalized by the Centers for Medicare and Medicaid Services create a favorable
environment for greater clinical adoption of our robotic technology. For instance, the Improving Medicare Post-Acute Care Transformation
Act of 2014, or the Impact Act of 2014, began the shift toward standardizing patient assessment data for quality measures. The
updated Prospective Payment System (PPS), SNF QRP (Quality Reporting Program) and SNF VBP (Value Based Purchasing) programs have
further shifted reimbursement toward the needs of the patient and away from volume of services provided in the skilled nursing
setting. Other programs have caused a similar shift in the Inpatient Rehabilitation Facility setting, as well. We expect that
in the next 6-12 months, further incentives toward quality based care will be implemented, resulting in providers being publicly
ranked, as well as financially rewarded, for quality reporting and better outcomes.
We have a growing
body of clinical data for our products. More than 1,500 patients participated in trials using our InMotion™ robots, the
results of which have been published in peer-reviewed medical journals (including the New England Journal of Medicine, Nature
and Stroke).
An earlier model of
InMotion™ robots were used in a multicenter randomized controlled phase III interventional trial, funded by the National
Institute for Health Research Health Technology Assessment Program (RATULS) in the United Kingdom. The study was completed in
2018, included the enrollment of 770 stroke patients in a multi-center randomized controlled research trial to evaluate the clinical
and cost effectiveness of robot-assisted training in post-stoke care. The Company is pleased that the RATULS trial confirmed the
finding of previous research studies which demonstrated that robot assisted therapy improved upper limb impairment when compared
with conventional care of stroke victims. The primary outcome for upper limb success was determined by an Action Research Arm
Test (ARAT), with four distinct success criteria that varied according to baseline severity. This test with these success criteria
was developed by the RATULS trial team for this study and has not been used previously in clinical trials. The findings of this
major research trial demonstrated that robot assisted therapy improved upper limb impairment however using this ARAT measurement,
the trial was unable to conclude that robot assisted therapy or enhance upper limb therapy resulted in improved upper limb functionality
after stroke compared with usual care provided to patients with stroke related upper limb functional limitation. The study findings
also showed that the attrition rate was drastically reduced in the patient population following either robotic therapy or enhanced
upper limb therapy versus usual care only. Most of the withdrawals from the study were before 3 months of usual care due to the
disappointment with the treatment allocation.
In addition to our
proprietary in-house products, we had the exclusive right to market and sell the Morning Walk lower body rehabilitation technology
owned by Curexo Inc., a South Korean company, within the United States. The Company has decided not to distribute the Morning
Walk product due to market conditions in the U.S. and is renegotiating its contract with our distributor in Korea. We may in the
future further augment our product portfolio through technology acquisition opportunities should they come available and if we
are sufficiently capitalized to undertake these investments.
On December 14, 2018,
we entered into a Sale of Goods Agreement (the “Agreement”) with CHC Management Services, LLC, or Kindred, pursuant
to which, among other things, Kindred agreed to purchase from us in a first phase a minimum of 21 of the Company’s InMotion™
ARM Interactive Therapy Systems – a minimum of one for each of Kindred’s existing and soon-to-open affiliated inpatient
rehabilitation hospitals and similar facilities described in the Agreement, and in a second phase a minimum of one InMotion
™
ARM Interactive Therapy System for each future facilities of Kindred, during the four-year minimum term of the Agreement.
Kindred entered into an initial purchase order for nine InMotion™ ARM Interactive Therapy System that shipped before December
31, 2018, with a further 10 robots in the quarter ended March 31, 2019 for a total of 19 InMotion™ robots sold during the
year ended March 31, 2019.
On January 23, 2019,
we announced the commercial launch of our newest generation InMotion™ ARM/HAND robotic system for clinical rehabilitation
of stroke survivors and those with mobility impairments due to neurological conditions. The improved new generation InMotion
™
ARM/HAND was developed according to the same principals of motor learning and neuro plasticity that were incorporated into
the original InMotion
™
ARM robotic system and utilizes artificial intelligence and data analysis to provide individualized
therapy and reports that empower patients.
It includes
the following new features:
|
|
·
|
Enhanced
hand-rehabilitation technology: The updated hand robot provides therapy focused on hand
opening and grasping for patients ready to retrain reach and grasp functional tasks.
|
|
|
·
|
InMotion™ EVAL: The InMotion™ ARM/HAND offers the
ability to assess hand movements in a precise and objective manner, allowing the clinician to better measure and quantify
a patient’s progress and response to therapy.
|
|
|
·
|
Improved, comprehensive reporting: Optimized report formats
provide improved documentation of patient outcomes, improved ease of use and enhanced interpretation of evaluation results,
allowing clearer indications of progress over their complete rehabilitation journey, all on one screen.
|
We have worked with
industry leaders in manufacturing and design and have also expanded our development team through partnerships with researchers
and academia. On May 17, 2017, we entered into a Co-operative Joint Venture Contract with Ginger Capital Investment Holding Ltd.,
pursuant to which the Company has a 25% interest and Ginger Capital has a 75% interest. As of the date of this 10-K, Ginger Capital
is obligated to contribute $725,000 to the joint venture and is required to contribute an additional $725,000 by May 22, 2023.
To date the Chinese partners if the JV have contributed $1,100,000 to the JV. Three InMotion™ robots have been delivered
from us to the joint venture, which were used for product demonstration and for quality assessment by Chinese authorities during
the fiscal year March 31, 2019 and 8 additional robots were shipped by Bionik to the Chinese JV in the first quarter of fiscal
year 2020 according to contract terms.
On June 20, 2017,
we entered into a joint development and manufacturing agreement with Wistron Medical Tech Holding Company of Taiwan to jointly
develop a lower body assistive robotic product based on the ARKE technology for the consumer home market. As the lower body
assistive robotic device is on an engineering hold due to prioritizing the development of the InMotion
™
Home robotic
device, no work has been done with Wistron recently.
We have also entered
into an agreement with Cogmedix Inc., a wholly owned subsidiary of Coghlin Companies, a medical device development and manufacturing
company located in West Boylston, MA, for the production of InMotion
™
robots. The initial agreement is for turnkey,
compliant manufacturing with the capability of scaling faster production to meet increased volume as the Company grows. In addition,
our Massachusetts-based manufacturing facility is compliant with ISO- 13485 and FDA regulations.
We currently hold
an intellectual property portfolio that includes 4 U.S. patents and 1 U.S. pending patent, all 5 of which are pending internationally,
as well as other patents under development. We may file provisional patents from time to time, which may expire if we do not pursue
full patents within 12 months of the filing date. One of new provisional patent has recently been filed which the Company plans
to file as a full patent prior to the 12-month deadline. The provisional patents may not be filed as full patents and new provisional
patents may be filed as the technology evolves or changes. Additionally, we hold exclusive licenses to three additional patents
of which one is currently being used for the InMotion
™
Wrist and is licensed to us from the Massachusetts Institute
of Technology.
We currently sell
our products directly or can introduce customers to a third-party finance company to lease at a monthly fee over the term or other
fee structure for our products to hospitals, clinics, distribution companies and/or buying groups that supply those rehabilitation
facilities.
We introduced our
new enhanced commercial version of the InMotion
™
product line starting with the InMotion
™
Arm in December
2017 then the InMotion
™
Arm/Hand in January 2019. We sold 6 InMotion™ robots in the year ended March 31, 2017,
11 InMotion™ robots in the year ended March 31, 2018, and 33 InMotion™ robots in the for the year ended March 31,
2019.
We had $3,246,038
of revenue for the year ended March 31, 2019 (March 31, 2018 – $987,431).
History; Recent Developments
Bionik Laboratories
Corp. was incorporated on January 8, 2010 in the State of Colorado. At the time of our incorporation the name of our company was
Strategic Dental Management Corp. On July 16, 2013, we changed our name from Strategic Dental Management Corp. to Drywave Technologies,
Inc. and changed our state of incorporation from Colorado to Delaware. Effective February 13, 2015, we changed our name to Bionik
Laboratories Corp.
Bionik Laboratories
Inc., which we refer to in this 10-K as Bionik Canada, was incorporated on March 24, 2011 under the Canada Business Corporations
Act.
On February 26, 2015,
we entered into an Investment Agreement with Bionik Acquisition Inc., a company existing under the laws of Canada and our wholly
owned subsidiary, and Bionik Canada whereby we acquired 100 Class 1 common shares of Bionik Canada representing 100% of the outstanding
Class 1 common shares of Bionik Canada. After giving effect to this and related transactions, we commenced operations through
Bionik Canada. Subsequently, on April 21, 2016, we acquired Interactive Motion Technologies, Inc., or IMT, a Boston, Massachusetts-based
provider of effective robotic products for neurorehabilitation, including all of its owned and licensed products both commercialized
and in development.
We effected a one-for-one
hundred fifty reverse stock split on October 29, 2018. As a result of the reverse stock split, each one hundred fifty shares of
our common stock automatically combined into and became one share of our common stock. Accordingly, as of October 29, 2018, there
were 2,337,964 shares of our common stock issued and outstanding. Any fractional shares which would otherwise be due as a result
of the reverse stock split were rounded up to the nearest whole share. The reverse stock split automatically and proportionately
adjusted, based on the one-for-one hundred fifty reverse stock split ratio, all issued and outstanding shares of our common stock
and exchangeable shares, as well as common stock underlying stock options, warrants and other derivative securities outstanding
at the time of the effectiveness of the reverse stock split. The exercise price on outstanding equity based-grants was proportionately
increased, while the number of shares available under our equity-based plans was also proportionately reduced. Share and per share
data (except par value) for the periods presented reflect the effects of this reverse stock split. References to numbers of shares
of common stock and per share data in the accompanying financial statements and notes thereto relating to dates prior to the reverse
stock split have been adjusted to reflect the reverse stock split on a retroactive basis.
In June 2019, we commenced
an up to $9 million convertible note offering, of which $700,000 has been raised through June 27, 2019.
Corporate Information
Our principal executive
office is located at 483 Bay Street, N105, Toronto, ON, Canada M5G 2C9 and our main corporate telephone number is (416) 640-7887
x 508. Our principal US office is located at 80 Coolidge Hill Road, Watertown, MA, USA 02472, telephone number 617-926-4800. Our
website is www.bioniklabs.com. Information on our website does not constitute a part of this Annual Report on Form 10-K.
Products in Market
InMotion™ Robots
Our suite of robotic
rehabilitation products are the result of medical engineering research and original development at the Newman Laboratory for Biomechanics
and Human Rehabilitation at the Massachusetts Institute of Technology (MIT).
We believe that our
robotic products have exceptional capacity for measurement and immediate interactive response, which sets them apart from other
therapy systems:
|
|
·
|
Patient
can be set up to rehabilitate on the InMotion™ robots within 2 minutes:
|
|
|
·
|
InMotion™
robots senses the patient’s movement and responds to a patient’s continually
changing ability;
|
|
|
·
|
Using
artificial intelligence, the robots guide the exercise treatment accordingly:
|
|
|
·
|
If
the patient is unable to move, the robot assists the patient to initiate movement towards
the target;
|
|
|
·
|
If
coordination is a problem, using artificial intelligence, the robot “guides”
the movement, allowing the patient to move towards the target and confirming that the
patient is practicing the movement the correct way; and
|
|
|
·
|
As
the patient gains movement control, the robot provides less assistance and continually
challenges the patient; and Provides quantifiable feedback on progress and performance
that can be downloaded.
|
InMotion™ robots
have been tested by leading medical centers in controlled clinical trials, including large randomized controlled clinical studies.
Through research, we have determined that the best way to optimize robot therapy is by allowing the robots to focus on reducing
impairments and allowing the therapist to assist on translating the gains into function.
We believe that our
modular systems approach to neurorehabilitation is designed to optimize the use of robotics in a manner that is consistent with
the latest clinical research and neuroscience, taking into account the latest understanding on motor learning interference and
motor memory consolidation.
More than two hundred
eighty InMotion™ robots have been sold for research and rehabilitation in over 15 countries, including the United States.
Extensive research has shown the InMotion™ robots to be effective, especially for stroke, cerebral palsy and Parkinson’s
disease. Based on clinical trials using the InMotion
™
ARM, the American Heart Association (AHA) Stroke council and
the U.S. Department of Veterans Affairs recommended, in 2010, the use of robot-assisted therapy to improve upper extremity motor
coordination in individuals with some voluntary finger extension in outpatient and chronic care settings. In the trial conducted
by the Department of Veterans Affairs, results demonstrated efficacy and a reduction in healthcare expenses when using the InMotion
™
ARM when compared to non-robotic therapy.
InMotion™ ARM
The InMotion
™
ARM is an evidence-based intelligent interactive rehabilitation technology that senses patient movements and limitations,
providing assistance as needed in real time. It allows clinicians to effectively deliver optimum intensive sensor motor therapy
to the shoulder and elbow to achieve the development of new neural pathways and helps patients regain motor function following
a neurological condition or injury. We recently launched a new version of the InMotion™ ARM, which has a 40% smaller footprint
than the previous generation and has wireless report printing, among other improvements.
The
product is characterized as a Class II medical device by the U.S. Food and Drug Administration (FDA) and is listed with the FDA
as 510(k) exempt, allowing the product to be marketed in the United States.
InMotion™ ARM/HAND
The InMotion
™
ARM/HAND
provides support for therapy involving reaching with grasp and release movements,
and individual hand movements. It allows clinicians to efficiently deliver optimal intensive sensory motor therapy to the hand
to develop new neural pathways and helps patients regain motor function following a neurological condition or injury. The product
is characterized as a Class II medical device by the U.S. Food and Drug Administration (FDA) and is listed with the FDA as 510(k)
exempt allowing the product to be marketed in the United States. We recently launched our newest generation InMotion™ ARM/HAND,
which has enhanced hand-rehabilitation technology and improved, comprehensive reporting, among other improvements.
InMotion™ WRIST
The InMotion
™
WRIST is an evidence based interactive rehabilitation device that senses patient movements and limitations and provides assistance
as needed. It can accommodate the range of motion of a normal wrist in everyday tasks and can be used by clinicians as a stand-alone
treatment option or in addition to the InMotion™ ARM
™
. The InMotion™ WRIST enables clinicians to efficiently
deliver optimum intensive sensor motor wrist and forearm therapy to patients with neurological conditions. The product is characterized
as a Class II medical device by the U.S. and is listed with the FDA as 510(k) exempt allowing the product to be marketed in the
United States. The Company plans to launch a new version of the InMotion™ Wrist in early 2020.
Product Pipeline
InMotion
™
HOME
The InMotion™
Home is an upper extremity product that would allow patients to extend their therapy for as long as needed while rehabilitating
at home and is being developed on the same design platform as the InMotion™ clinical products described above. The InMotion™
Home is currently in development and we have not yet determined a release date for this product.
Lower Body Robotic Products
The ARKE is a robotic
lower body exoskeleton that was under development and designed for wheelchair bound individuals suffering from spinal cord injuries,
stroke and other mobility disabilities. As a result of a combination of our concentrating on the commercialization of the InMotion™
robots, our lack of additional funds, and changes in the marketplace, we determined to suspend the further development of the
ARKE as a rehabilitation device, and instead, building on our existing ARKE exoskeleton technology, a lower body robotic assistive
device as well as other technology targeting the consumer market, that could allow mobility impaired individuals to walk better.
This product development is on engineering hold at a technical development stage due to prioritizing the development of the InMotion™
Home robot.
Other Prospective Products
We have exclusively
licensed the rights to manufacture and sell products and methodologies covered by a patent for a lower limb robotic rehabilitation
apparatus and method for rehabilitating gait, owned in part by Dr. Hermano Igo Krebs, one of our former directors and executive
officers; however, this product has not yet been developed.
We may from time to
time expand our product offerings and enhance the strength of our Company through internal development, as well as through strategic
and accretive partnerships or acquisitions from time to time.
Competition and Competitive Advantage
The medical technology
equipment industry is characterized by strong competition and rapid technological change. There are a number of companies developing
technologies that are competitive to our existing and proposed products, many of them, when compared to our Company, having significantly
longer operational history and greater financial and other resources.
The primary competitor
for the InMotion
™
product line of upper-body rehabilitation robots is Hocoma, a Swiss-based company. Other competitors
include Motorika and Tyromotion as well as other known and unknown smaller potential competitors that may compete with us directly
or indirectly. We believe that the InMotion™ product line’s primary advantage over Hocoma is the evidence based, research
proven data that supports each of our products. Evidence based, research proven data is used to support reimbursement from health
systems, insurance companies and governments.
The prime competitors
for our lower body robotics assistive device in development are Honda, Cyberdyne and Ekso Bionics. We expect it, once developed,
to compete as a personal choice physical enhancement consumer product.
Our challenge will
be achieving rapid market awareness and adoption of our emerging technology in rehabilitation and mobility centers throughout
the U.S., Canada and any other market we may enter. Our existing InMotion™ robots and technologies are expected to significantly
help with third party clinical trials and our ability to launch our lower-extremity development products into the market, as we
intend to leverage third party clinical data on our rehabilitative products and international distributorships and relationships
with rehabilitation centers around the world.
Robotic technology
and its use in clinical settings is a new and emerging industry and is regulated by medical device regulatory agencies (such as
the US Food and Drug Administration). We believe that we will face challenges of increased regulatory scrutiny, possible changes
in regulator’s requirements, meeting quality control standards of various government regulators, increased competition in
the future based on other new technologies, additional features and customizability, reduced pricing, clinical outcomes and other
factors. Our strength in this market will depend on our ability to achieve market acceptance, develop new technologies, develop
new products, implement production plans, develop marketing strategies, secure regulatory approvals, secure necessary data for
reimbursement, protect our intellectual property and have sufficient funding to meet all these challenges.
The market for the
Company’s other prospective products also has competition and is subject to rapid technological change and regulatory requirements.
There can be no assurance that the Company will be in a strong position to respond quickly to potential acquisitions and other
market opportunities, new or emerging technologies and changes in customer requirements. Failure to maintain and enhance our competitive
position could materially affect the business and our prospects.
Market Strategy
The Company’s
current products are designed to be rehabilitation products and mobility solutions for patients in hospitals and clinics. We currently
have three robotic products that are listed with the FDA, which are the products sold through our own sales team in the United
States, as well as through third party distributors around the world. Our business plan in part relies on broad adoption of upper
and lower body robotic rehabilitation products to provide neuro rehabilitation to individuals who have suffered a neurological
injury or disorder.
The sales of our clinical
and proposed products could depend, in part, on the extent to which healthcare providers and facilities or individual users are
reimbursed by government authorities, private insurers and other third-party payers for the costs of our products or the services
performed with our products. The coverage policies and reimbursement levels of third-party payers, which can vary among public
and private sources and by country, may affect which products are purchased by customers and the prices they are willing to pay
for those products in a particular jurisdiction. Reimbursement rates can also affect the acceptance rate of new technologies.
Legislative or administrative reforms to reimbursement systems in the United States or abroad, or changes in reimbursement rates
by private payers, could significantly reduce reimbursement for procedures using the Company’s products or result in denial
of reimbursement for those products, which would adversely affect customer demand or the price customers may be willing to pay
for such products. The change expected in 2019 under certain US government plans to reimburse SNF’s (Skilled Nursing Facilities)
to be followed by IRF’s (Inpatient Rehabilitation Facilities) based on outcome data, is expected to be beneficial to the
Company in its sales efforts.
The Company has committed
to a commercial strategy to maximize its efforts to position its solutions with multi-location, high patient volume rehabilitation
organizations. The Company believes its robotic systems are a good match to the patient care and business objectives relevant
to these larger organizations operating on a regional or national basis.
Outside of the US,
we have used distributors to sell in local markets and we currently have a distributor in South Korea, as well as a joint venture
partner in China. We plan in the near term to hire a sales director in Europe to increase our market penetration in Europe and
surrounding areas. Our efforts to penetrate the European market are supported by having attained the CE marking which signifies
that products sold in the European Economic Area (EEA) have been assessed to meet high safety health and environmental protection
requirements.
We have not yet determined
a release date for the InMotion™ Home, our planned home version of our InMotion™ product line. Our market strategy
will be the development of hospital and clinic relationships that will allow us to gain acceptance of the technology among experts
and patients. We are also seeking a number of government grants in collaboration with various hospitals and clinics to allow us
to partially fund trials and research projects. We expect to gain traction among the doctors and experts involved in the distribution
and buying groups that are established within those selected partner hospitals.
We currently sell
our robots or can introduce customers to a third-party finance company to lease at a monthly fee over term or other fee structure
for our products to hospitals, clinics, distribution companies and/or buying groups that supply those rehabilitation facilities.
Our market strategy
also relies on identifying and entering into joint venture arrangements with third parties that can assist us with the development,
commercialization and distribution of our technologies and products. For instance, we have entered into a relationship with Curexo
Inc. of South Korea to distribute our InMotion™ robots to that market. Additionally, we established a cooperative joint
venture enterprise with Ginger Capital Investment Holding Ltd. for the purpose of selling and distributing our InMotion™
robots in the People’s Republic of China.
Intellectual Property
We use intellectual
property developed, acquired or licensed, including patents, trade secrets and technical innovations to provide our future growth
and to build our competitive position. We have 4 U.S. patents and 1 U.S. pending patent, all 5 of which are pending internationally,
and other patents under development. As we continue to expand our intellectual property portfolio, it is critical for us to continue
to invest in filing patent applications to protect our technology, inventions, and improvements. However, we can give no assurance
that we will have sufficient funds to do so or that competitors will not infringe on our patent rights or otherwise create similar
or non-infringing competing products that are technically patentable in their own right.
Our patents and pending
patents, all of which are expected to expire in 2033 or 2034, are as follows:
|
Patent
|
|
Status
|
|
Algorithms & Control Systems
|
|
Filed US & International
|
|
Sensory Technology
|
|
Issued in US & pending International
|
|
Robotics
|
|
Issued in US & pending International
|
|
Robotics
|
|
Issued in US & pending International
|
|
Robotics
|
|
Issued in US & pending International
|
We may file provisional
patents from time to time, which may expire if we do not pursue full patents within 12 months of the filing date. One new provisional
patent has recently been filed which the Company plans to file as a full patent prior to the 12-month deadline. The provisional
patents may not be filed as full patents and new provisional patents may be filed as the technology evolves or changes.
We have a trademark
filed for the InMotion
™
name in the US and European Union. This trademark is to be used for the robots and software
that Bionik develops and sells related to this product line.
In addition, the following
are the patents licensed to us that we acquired on April 21, 2016:
|
Patent
#
|
|
Description
|
|
Date
|
|
Expiration
|
|
|
|
|
|
|
|
|
|
7,618,381
|
|
Wrist and Upper Extremity
Motion (MIT License)
|
|
11/17/09
|
|
10/27/2024
|
|
|
|
|
|
|
|
|
|
7,556,606
|
|
Pelvis Interface: key components for
effective motor neuro- Rehabilitation of lower extremities (MIT License)
|
|
07/07/09
|
|
05/17/2027
|
|
|
|
|
|
|
|
|
|
8,613,691
|
|
Dynamic Lower Limb Rehabilitation Robotic
Apparatus and Method of Rehabilitating Human Gait (Krebs/Bosecker License)
|
|
12/24/13
|
|
4/16/2030
|
IMT entered into an
Agreement, executed on December 31, 1999, to license two of the above-referenced patents from MIT with a royalty of 3% on sales
within the United States and 1.5% for sales outside the United States, with a minimum annual royalty of $10,000. To date, we have
not determined whether we intend to commercialize the patent relating to the pelvis.
Dr. Krebs, a former
director and former executive officer and a founder of IMT, is a co-licensor pursuant to an Agreement dated June 8, 2009, of patent
#8,613,691, pursuant to which we are required to pay Dr. Krebs and Caitlyn Joyce Bosecker an aggregate royalty of 1% of sales
based on such patent. As this product connected to the patent is not yet commercialized, no sales have been made.
We have to date and
generally plan to continue to enter into non-disclosure, confidentially and intellectual property assignment agreements with all
new employees as a condition of employment. In addition, we also generally enter into confidentiality and non-disclosure agreements
with consultants, manufacturers’ representatives, distributors, suppliers, investors, financial partners and others to attempt
to limit access to, use and disclosure of our proprietary information.
Research and Development
Our research and development
programs are pursued by engineers and scientists employed by us in Toronto and Boston on a full- time basis or hired as per diem
consultants. InMotion™ robots are based on research and development originally done at MIT. Our InMotion™ Wrist product
is based on a patent that we license from MIT.
We also work with
advisors who are industry leaders in manufacturing and design and researchers and academia. Our leading robotic advisor is Dr.
Neville Hogan of MIT. We are also working with subcontractors in developing specific components of our technologies. The primary
objective of our research and development program is to advance the development of our existing and proposed products, to enhance
the commercial value of such products.
For the fiscal years
ended March 31, 2019 and March 31, 2018, the Company incurred $3,174,892 and $2,825,200 respectively, in research and development
costs. Research and development expenses have increased due to increased staff members however, project development project costs
are comparable to the prior year.
Government Regulations
General
Our medical technology
products and operations are subject to regulation by the U.S. Food and Drug Administration (“FDA”) and various other
federal and state agencies, as well as foreign governmental agencies in Canada, Europe, South America and Asia. These agencies
enforce laws and regulations that govern the development, testing, manufacturing, labeling, advertising, marketing and distribution,
and market surveillance of our medical device products.
In addition to the
below, other regulations we encounter are the regulations that are common to all businesses, such as employment legislation, implied
warranty laws, and environmental, health and safety standards, to the extent applicable. We will also encounter in the future
industry-specific government regulations that would govern our new products, if and when developed for commercial use. It may
become the case that other regulatory approvals will be required for the design and manufacture of our products and proposed products.
U.S. Regulation
Under
the U.S. Federal Food, Drug, and Cosmetic Act, medical devices are classified into one of three classes — Class I, Class
II or Class III — depending on the degree of risk associated with each medical device and the extent of control needed to
ensure safety and effectiveness. The InMotion™ robots are classified as Class II 510 (k) exempt products. Our manufacturing
facility in Boston is compliant with ISO 13485 and FDA regulations.
We also are required
to establish a suitable and effective quality management system, which establishes controlled processes for our product design,
manufacturing, and distribution. We are doing this in compliance with the internationally recognized standard ISO 13485 Quality
Management Systems. Following the introduction of a product, the FDA and foreign agencies may engage in periodic reviews of our
quality systems, as well as product performance and advertising and promotional materials. These regulatory controls, as well
as any changes in FDA or other foreign agencies’ policies, can affect the time and cost associated with the development,
introduction and continued availability of new products. Where possible, we anticipate these factors in our product development
processes. These agencies possess the authority to take various administrative and legal actions against us, such as product recalls,
product seizures and other civil and criminal sanctions.
Foreign Regulation
In
addition to regulations in the United States, we will be subject to a variety of foreign regulations governing clinical trials
and commercial sales and distribution of our products in foreign countries. InMotion™ robots have also been designated as
Class IIa devices in the EU. Whether or not we obtain FDA clearance for the marketing, sale and use of a product, we must obtain
approval of a product by the comparable regulatory authorities of foreign countries before we can commence clinical trials or
marketing of the product in those countries. The process varies from country to country, and the time may be longer or shorter
than that required by the FDA. The requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement
vary greatly from country to country.
The policies of the
FDA and foreign regulatory authorities may change and or additional government regulations may be enacted which could prevent
or delay regulatory approval of our products and could also increase the cost of regulatory compliance. We cannot predict the
likelihood, nature or extent of adverse governmental regulation that might arise from future legislative or administrative action,
either in the United States or abroad.
Employees
As of June 27, 2019,
we had 28 full-time employees, 2 part-time employees and 3 consultants who are based in our principal executive office located
in Toronto, Canada, and our Watertown, Massachusetts facility. These employees oversee day-to-day operations of the Company supporting
management, engineering, research and development, sales and marketing and administration functions of the Company. As required,
we also engage consultants to provide services to the Company, including quality assurance and corporate services. We have no
unionized employees.
Subject to available
funds, we plan to hire up to 5 additional full-time employees within the next 12 months whose principal responsibilities will
be the support of our research and development, clinical development, production, sales and marketing and commercialization/ business
development activities.
We consider relations
with our employees to be satisfactory.
Item 1A- RISK
FACTORS
The securities
of the Company involve a high degree of risk and should only be purchased by persons who can afford to lose all or part of their
investment. Prospective purchasers should carefully consider, among other things, the following risk factors and the other information
in this Annual Report on Form 10-K, including our financial statements and the notes to those statements, prior to making an investment
decision.
We have a limited operating history
upon which investors can evaluate our future prospects.
We have a limited
operating history based on our current business plan of commercializing and selling the InMotion™ robots, upon which an
evaluation of our business plan or performance and prospects can be made.
The business and prospects
of the Company must be considered in the light of the potential problems, delays, uncertainties and complications encountered
in connection with a relatively new business and creating a new industry. The risks include, but are not limited to, the possibility
that we will not be able to develop functional and scalable products and services, or that although functional and scalable, our
products and services will not be economical to market; that our competitors hold proprietary rights that preclude us from marketing
such products; that our competitors market a superior or equivalent product; that we are not able to upgrade and enhance our technologies
and products to accommodate new features and expanded service offerings; or the failure to receive necessary regulatory clearances
for our products. To successfully introduce and market our products at a profit, we must establish brand name recognition and
competitive advantages for our products. There are no assurances that we can successfully address these challenges. If it is unsuccessful,
we and our business, financial condition and operating results could be materially and adversely affected.
The current and future
expense levels are based largely on estimates of planned operations and future revenues. It is difficult to accurately forecast
future revenues because the robotics market has not been fully developed, and we can give no assurance that our products will
continue to fuel revenue growth. If our forecasts prove incorrect, the business, operating results and financial condition of
the Company will be materially and adversely affected. Moreover, we may be unable to adjust our spending in a timely manner to
compensate for any unanticipated reduction in revenue we expect to generate as a result of our products. As a result, the failure
to generate revenues would immediately and adversely affect the business, financial condition and operating results of the Company.
We cannot predict when we will achieve
profitability.
We have not been profitable
and cannot predict when we will achieve profitability. We have experienced net losses since our inception in 2010. We began generating
revenues after April 21, 2016 as a result of the acquisition of IMT and the sale of the InMotion™ robots, and we do not
anticipate generating significant revenues from other technologies in development until we successfully develop, commercialize
and sell products derived from those technologies, of which we can give no assurance. Although we sold 11 InMotion™ robots
during the year ended March 31, 2018 and 33 InMotion™ robots for the year ended March 31, 2019, we are unable to determine
when we will generate significant recurring revenues, if any, from the future sale of any of our products, or generate increased
recurring revenues from the sale of our commercialized InMotion™ robots.
We cannot predict
when we will achieve profitability, if ever. Our inability to become profitable has forced us to curtail or temporarily discontinue
certain of our research and development programs such as our lower body robotic assistive device and may force us to do so with
other commercialization programs and our day-to-day operations. Furthermore, there can be no assurance that profitability, if
achieved, can be sustained on an ongoing basis. As of March 31, 2019, we had an accumulated deficit of $46,357,373.
There is substantial doubt on our
ability to continue as a going concern.
Our independent registered
public accounting firm has issued a going concern qualification as part of its audit report that accompanies our 2019 audited
financial statements included herein. As stated in the notes to our audited financial statements for the fiscal year ended March
31, 2019, we have a negative working capital deficiency and have accumulated a significant deficit. Our continued existence is
dependent upon our ability to continue to execute our operating plan and to obtain additional debt or equity financing. There
can be no assurance that the additional necessary debt or equity financing will be available, or will be available on terms acceptable
to us, in which case we may be unable to meet our obligations or fully implement our business plan, if at all. Additionally, should
we be unable to realize our assets and discharge our liabilities in the normal course of business, the net realizable value of
our assets may be materially less than the amounts recorded in our financial statements.
We are subject to significant accounts
payable and other current liabilities.
We have accounts payable
and other liabilities of approximately $2.8 million as of March 31, 2019. We also incur indebtedness from time to time to fund
operations, which have historically been converted into equity but in the future may be required to be repaid at maturity. Our
operations are not currently able to generate sufficient cash flows to meet our payable and other liabilities, which could reduce
our financial flexibility, increase interest expenses and adversely impact our operations. We may not generate sufficient cash
flow from operations to enable us to repay this indebtedness and to fund other liquidity needs, including capital expenditure
requirements. Such indebtedness could affect our operations in several ways, including the following:
|
|
·
|
a
significant portion of our cash flows could be required to be used to service such indebtedness;
|
|
|
·
|
a
high level of indebtedness could increase our vulnerability to general adverse economic
and industry conditions;
|
|
|
·
|
any
covenants contained in the agreements governing such outstanding indebtedness could limit
our ability to borrow additional funds, dispose of assets, pay dividends and make certain
investments;
|
|
|
·
|
a
high level of indebtedness may place us at a competitive disadvantage compared to our
competitors that are less leveraged and, therefore, our competitors may be able to take
advantage of opportunities that our indebtedness may prevent us from pursuing;
|
|
|
·
|
debt
covenants may affect our flexibility in planning for, and reacting to, changes in the
economy and in our industry, if any; and
|
|
|
·
|
any
ability to convert or exchange such indebtedness for equity in the Company can cause
substantial dilution to existing stockholders of the Company.
|
We may need to refinance or restructure
all or a portion of our indebtedness and other liabilities on or before maturity. We may not be able to refinance any of our indebtedness
or other liabilities on commercially reasonable terms, or at all.
A high level of indebtedness
and other liabilities increases the risk that we may default on our debt obligations and other liabilities. We may not be able
to generate sufficient cash flows to pay the principal or interest on our debt. If we cannot service or refinance our indebtedness
and other liabilities or convert or exchange indebtedness for equity in the Company, we may have to take actions such as selling
significant assets, seeking additional equity financing (which will result in additional dilution to stockholders) or reducing
or delaying capital expenditures or our research and development programs, any of which could have a material adverse effect on
our operations and financial condition. The Company requires new funding which it does not yet have secured and if this new funding
is not received it will have a material adverse effect on our business, financial condition and results of operation.
Our acquisition of companies or
technologies could prove difficult to integrate and may disrupt our business and harm our operating results and prospects.
Potential acquisitions
will likely involve risks associated with our assumption of some or all of the liabilities of an acquired company, which may be
liabilities that we were or are unaware of at the time of the acquisition, potential write-offs of acquired assets and potential
loss of the acquired company’s key employees or customers.
We may encounter difficulties
in successfully integrating our operations, technologies, services and personnel with that of the acquired company, and our financial
and management resources may be diverted from our existing operations. For instance, we diverted some resources from our existing
technologies under development to focus on the InMotion™ robots acquired from IMT in April 2016. Offices outside of Canada
or in multiple states or provinces, including our offices in Massachusetts have created a strain on our ability to effectively
manage our operations and key personnel. We have consolidated accounting, finance and administration in Toronto. If we elect to
further consolidate our facilities, we may lose key personnel unwilling to relocate to the consolidated facility, may have difficulty
hiring appropriate personnel at the consolidated facility and may have difficulty providing continuity of service through the
consolidation.
End-user satisfaction
or performance problems with any acquired business, technology, service or device, including the InMotion™ robots, could
also have a material adverse effect on our reputation. Additionally, potential disputes with the seller of an acquired business
or its employees, suppliers or customers and amortization expenses related to intangible assets could adversely affect our business,
operating results and financial condition. If we fail to properly evaluate and execute acquisitions, our business may be disrupted,
and our operating results and prospects may be harmed.
We will require additional capital
to support our present business plan and our anticipated business growth, and such capital may not be available on acceptable
terms, or at all, which would adversely affect our ability to operate; and such capital may substantially dilute the interests
of existing stockholders.
We will require additional
funds to further develop our business plan and have been relying on convertible and term debt financing to fund the operation
of our business. Based on our current operating plans, our resources are currently not sufficient to fund our planned operations,
including those necessary to introduce development-stage products into the rehabilitation and mobility markets. Since it is unlikely
that we will generate sufficient revenues from our operating activities to fund all of our operating and development plans, we
will need to raise additional funds through debt, equity or equity-linked offerings or otherwise in order to meet our expected
future liquidity requirements, including development of existing products, introducing other products or pursuing new product
opportunities. Any such financing that we undertake will likely be dilutive to current stockholders or may require that we relinquish
rights to certain of our technologies or products. For instance, as of March 31, 2018 and June 2018, we converted approximately
$9.1 million of convertible promissory notes into approximately 1.25 million shares of common stock. As of July 20, 2018, we also
converted approximately $4.7 million of convertible promissory notes into 683,396 shares of common stock. On March 28, 2019, the
Company converted $4.65 million of convertible promissory notes and interest into 1,247,099 common shares. In June 2019, we commenced
a new up to $9 million convertible note offering, of which we have raised $700,000 through June 27, 2019. We are evaluating future
financing arrangements, as well.
We intend to continue
to make investments to support our business growth through introducing new products, including patent or other intellectual property
asset creation, the acquisition of other businesses or strategic assets and licensing of technology or other assets. To fully
execute on our business plan, we will need additional funds to respond to business opportunities and challenges, including ongoing
operating expenses, protecting our intellectual property, satisfying debt payment obligations, developing new lines of business
and enhancing our operating infrastructure. While we will need to seek additional funding for such purposes, we may not be able
to obtain financing on acceptable terms, or at all. In addition, the terms of our financings may be dilutive to, or otherwise
adversely affect, holders of our common stock or common stock equivalents. We have previously and may again seek additional funds
through arrangements with collaborators or other third parties. We may not be able to negotiate any such arrangements on acceptable
terms, if at all. If we are unable to obtain additional funding on a timely basis, we may be required to curtail or terminate
some or all of our business plans.
We may never complete the development
of any of our proposed products or product improvements into marketable products.
We do not know when
or whether we will successfully complete the development of the planned development-stage or next generation InMotion™ robots,
or any other proposed, developmental or contemplated product, for any of our target markets. We continue to seek to improve our
technologies before we are able to produce a commercially viable product. Failure to improve on any of our technologies could
delay or prevent their successful development for any of our target markets.
Developing any technology
into a marketable product is a risky, time consuming and expensive process. You should anticipate that we will encounter setbacks,
discrepancies requiring time consuming and costly redesigns and changes and that there is the possibility of outright failure.
We may not meet our product development, manufacturing, regulatory, commercialization and other milestones.
We have established
milestones, based upon our expectations regarding our technologies at that time, which we use to assess our progress toward developing
our products. These milestones relate to product roll-outs, technology and design improvements as well as to dates for achieving
development goals and regulatory approvals, among other things. If our products exhibit technical defects or are unable to meet
cost or performance goals or for any other reason, our commercialization schedule could be delayed and potential purchasers of
our initial commercial products, may decline to purchase such products or may opt to pursue alternative products. Due to our current
budgeting constraints and evolving timelines on our products in development, we are changing or delaying some of the timelines
and milestones for our other technologies being developed.
We can give no assurance
that our commercialization schedule will be met as we concentrate our efforts as we continue to develop our products.
Customers will be unlikely to buy
any of our proposed, developmental or contemplated products unless we can demonstrate that they can be produced for sale to consumers
at attractive prices.
During the past year,
we retained a third-party manufacturer to manufacture our products, in addition to our Boston-based manufacturing facility now
used primarily for research and development purposes but may continue to be used to manufacture and assemble some or all of our
products as needed. We can offer no assurance that either we or our manufacturing partners will continue to develop efficient,
automated, low-cost manufacturing capabilities and processes to meet the quality, price, engineering, design and production standards
or production volumes required to successfully mass market any of our existing or contemplated products. Even if we or our manufacturing
partners are successful in developing such manufacturing capability and processes, we do not know whether we or they will be timely
in meeting our product commercialization schedule or the production and delivery requirements of potential customers. A failure
to develop such manufacturing processes and capabilities could have a material adverse effect on our business and financial results.
The price of our existing
or contemplated products is in part dependent on material and other manufacturing costs. We are unable to offer any assurance
that either we or a manufacturing partner from time to time will be able to reduce costs to a level which will allow production
of a competitive product or that any product produced using lower cost materials and manufacturing processes will not suffer from
a reduction in performance, reliability and longevity. Furthermore, although we have implemented a pricing structure for our existing
products, we can give no assurance that this pricing structure will not require changes in the future that could affect the attractiveness
of our pricing.
Our products may not be accepted
in the market.
We cannot be certain
that our current products or any other products we may develop, or market will achieve or maintain market acceptance. Market acceptance
of our products depends on many factors, including our ability to convince key opinion leaders to provide recommendations regarding
our products, convince distributors and customers that our technology is an attractive alternative to other technologies, demonstrate
that our products are reliable and supported by us in the field, supply and service sufficient quantities of products directly
or through marketing alliances, and price products competitively in light of the current macroeconomic environment, which, particularly
in the case of the medical device industry, are becoming increasingly price sensitive.
We are subject to extensive governmental
regulations relating to the manufacturing, labeling and marketing of our products.
Our medical technology
products and operations are or are expected to be subject to regulation by the FDA, Health Canada and other governmental authorities
both inside and outside of the United States. These agencies enforce laws and regulations that govern the development, testing,
manufacturing, labeling, advertising, marketing and distribution, and market surveillance of our medical products.
Under the United States
Federal Food, Drug, and Cosmetic Act, medical devices are classified into one of three classes — Class I, Class II or Class
III — depending on the degree of risk associated with each medical device and the extent of control needed to ensure safety
and effectiveness. Class II devices require a 510(k) premarket submission to the US FDA. The Company’s InMotion™ robots
have been characterized as Class II devices by the FDA.
In addition to regulations
in the United States, we will be subject to a variety of foreign regulations governing clinical trials and commercial sales and
distribution of our products in foreign countries. Whether or not we obtain FDA approval for a product, we must obtain approval
of a product by the comparable regulatory authorities of foreign countries before we can market the product in those countries.
The approval process varies from country to country, and the time may be longer or shorter than that required for FDA approval.
The requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary greatly from country
to country.
The policies of the
FDA and foreign regulatory authorities may change and or additional government regulations may be enacted which could prevent
or delay regulatory approval of our products and could also increase the cost of regulatory compliance. We cannot predict the
likelihood, nature or extent of adverse governmental regulation that might arise from future legislative or administrative action,
either in the United States or abroad.
Following the introduction
of a product, these agencies will also periodically review our manufacturing processes and product performance. The process of
complying with the applicable good manufacturing practices, adverse event reporting, clinical trial and other requirements can
be costly and time consuming, and could delay or prevent the production, manufacturing or sale of our products. In addition, if
we fail to comply with applicable regulatory requirements, it could result in fines, delays or suspensions of regulatory clearances,
closure of manufacturing sites, seizures or recalls of products and damage to our reputation. Recent changes in enforcement practice
by the FDA and other agencies have resulted in increased enforcement activity, which increases the compliance risk for the Company
and other companies in our industry. In addition, governmental agencies may impose new requirements regarding registration, labeling
or prohibited materials that may require us to modify or re-register products already on the market or otherwise impact our ability
to market our products in those countries. Once clearance or approval has been obtained for a product, there is an obligation
to ensure that all applicable FDA, Health Canada and other regulatory requirements continue to be met.
We may be subject to penalties and
may be precluded from marketing our products if we fail to comply with extensive governmental regulations.
The InMotion™
robots and we believe certain other products under development will be categorized as a Class II device in the U.S. Class II devices
require a 510(k) premarket submission to the US FDA. However, the FDA has not made any determination about whether our proposed
medical products are Class II medical devices and, from time to time, the FDA may disagree with the classification of a new Class
II medical device and require the manufacturer of that device to apply for approval as a Class III medical device. In the event
that the FDA determines that our medical products should be reclassified as a Class III medical device, we could be precluded
from marketing the devices for clinical use within the United States for months, years or longer, depending on the specific changes
to the classification. Reclassification of our products as Class III medical devices could significantly increase our regulatory
costs, including the timing and expense associated with required clinical trials and other costs.
The FDA and non-U.S.
regulatory authorities require that our products be manufactured according to rigorous standards. These regulatory requirements
may significantly increase our production costs and may even prevent us from making our products in amounts sufficient to meet
market demand. If we change our manufacturing process, regulatory authorities may need to review the process before it may be
used. Failure to comply with applicable regulatory requirements discussed could subject us to enforcement actions, including warning
letters, fines, injunctions and civil penalties, recall or seizure of our products, operating restrictions, partial suspension
or total shutdown of our production and criminal prosecution.
Federal, state and
non-U.S. regulations regarding the manufacture and sale of medical devices are subject to future changes. The complexity, timeframes
and costs associated with obtaining marketing clearances are unknown. Although we cannot predict the impact, if any, these changes
might have on our business, the impact could be material.
If we are not able to both obtain
and maintain adequate levels of third-party reimbursement for our products, it would have a material adverse effect on our business.
Healthcare providers
and related facilities are generally reimbursed for their services through payment systems managed by various governmental agencies
worldwide, private insurance companies, and managed care organizations. The manner and level of reimbursement in any given case
may depend on the site of care, the procedure(s) performed, the final patient diagnosis, the device(s) utilized, available budget,
or a combination of these factors, and coverage and payment levels are determined at each payer’s discretion. The coverage
policies and reimbursement levels of these third-party payers may impact the decisions of healthcare providers and facilities
regarding which medical products they purchase and the prices they are willing to pay for those products. Thus, changes in reimbursement
levels or methods may either positively or negatively impact sales of our products.
We have no direct
control over payer decision-making with respect to coverage and payment levels for our medical device products. Additionally,
we expect many payers to continue to explore cost-containment strategies (e.g., comparative and cost-effectiveness analyses, so-called
“pay-for-performance” programs implemented by various public and private payers, and expansion of payment bundling
schemes such as Accountable Care Organizations, and other such methods that shift medical cost risk to providers) that may potentially
impact coverage and/or payment levels for our current products or products we develop.
As our product offerings
are expected to be diverse across healthcare settings, they will likely be affected to varying degrees by the many payment systems.
Therefore, individual countries, product lines or product classes may be impacted by changes to these systems.
Product defects could adversely
affect the results of our operations.
The design, manufacture
and marketing of our products involves certain inherent risks. Manufacturing or design defects, unanticipated use of our products,
or inadequate disclosure of risks relating to the use of our products can lead to injury or other adverse events. These events
could lead to recalls or safety alerts relating to our products (either voluntary or required by the FDA, Health Canada or similar
governmental authorities in other countries), and could result, in certain cases, in the removal of a product from the market.
A recall could result in significant costs, as well as negative publicity and damage to our reputation that could reduce demand
for our products. Personal injuries relating to the use of our products could also result in product liability claims being brought
against us. The Company has product liability insurance to mitigate this risk. In some circumstances, such adverse events could
also cause delays in new product approvals.
Changes in reimbursement practices
of third-party payers could affect the demand for our products and the prices at which they are sold.
The sales of our InMotion™
robot and proposed products could depend, in part, on the extent to which healthcare providers and facilities or individual users
are reimbursed by government authorities, private insurers and other third-party payers for the costs of our products or the services
performed with our products. The coverage policies and reimbursement levels of third-party payers, which can vary among public
and private sources and by country, may affect which products are purchased by customers and the prices they are willing to pay
for those products in a particular jurisdiction. Reimbursement rates can also affect the acceptance rate of new technologies.
Legislative or administrative reforms to reimbursement systems in the United States or abroad, or changes in reimbursement rates
by private payers, could significantly reduce reimbursement for procedures using the Company’s products or result in denial
of reimbursement for those products, which would adversely affect customer demand or the price customers may be willing to pay
for such products.
We could be exposed to significant
liability claims if we are unable to obtain insurance at acceptable costs and adequate levels or otherwise protect ourselves against
potential product liability claims.
The testing, manufacturing,
marketing and sale of medical devices entail the inherent risk of liability claims or product recalls. The Company currently maintains
product liability insurance; however, product liability insurance is expensive and may not be available on acceptable terms in
the future, if at all. A successful product liability claim, or product recall could inhibit or prevent the successful commercialization
of our products, cause a significant financial burden on the Company, or both, which in either case could have a material adverse
effect on our business and financial condition. Although we carry product liability insurance, there is no guarantee that our
insurance will adequately cover us against potential liability. If not, the results of our operations could be materially and
adversely affected. In addition, any product liability claims brought in connection with any alleged defect of our products, whether
with or without merit, could increase our product liability insurance rates or prevent us from securing continuing coverage at
rates we could afford.
The results of our research and
development efforts are uncertain and there can be no assurance of the commercial success of our products.
We believe that we
will need to incur additional research and development expenditures to continue development of our existing and proposed products
as well as research and development expenditures to develop new products and services. The products and services we are developing
and may develop in the future may not be technologically successful. In addition, the length of our product and service development
cycle may be greater than we originally expected, and we may experience delays in product development. If our resulting products
and services are not technologically successful, they may not achieve market acceptance or compete effectively with our competitors’
products and services.
If we fail to retain certain of
our key personnel and attract and retain additional qualified personnel, we might not be able to pursue our growth strategy.
Our future success
will depend upon the continued service of Eric Dusseux, our Chief Executive Officer, and his executive team or any qualified replacement
of those individuals. There can be no assurance that the services of any of these individuals will continue to be available to
us in the future. We do not carry any key man life insurance policies on any of our executive officers. The failure to retain,
or attract replacement, qualified personnel could have a material adverse effect on our business and our ability to pursue our
growth strategy.
Recent executive and legislative
actions to amend or impede the implementation of the Affordable Care Act and ongoing efforts to repeal, replace or further modify
the Affordable Care Act may adversely affect our business, financial condition and results of operations.
Recent executive and
legislative actions to amend or impede the implementation of the Affordable Care Act and ongoing efforts to repeal, replace or
further modify the Affordable Care Act may adversely affect our business, financial condition and results of operations.
Since its adoption
into law in 2010, the Affordable Care Act has been challenged before the U.S. Supreme Court, and several bills have been and continue
to be introduced in Congress to delay, defund, or repeal implementation of or amend significant provisions of the Affordable Care
Act. In addition, there continues to be ongoing litigation over the interpretation and implementation of certain provisions of
the law. The net effect of the Affordable Care Act, as currently in effect, on our business is subject to a number of variables,
including the law’s complexity, lack of complete implementing regulations and interpretive guidance, and the sporadic implementation
of the numerous programs designed to improve access to and the quality of healthcare services. Additional variables of the Affordable
Care Act impacting our business will be how states, providers, insurance companies, employers, and other market participants respond
during this period of uncertainty surrounding the future of the Affordable Care Act.
On January 20, 2017,
President Trump issued an executive order that, among other things, stated that it was the intent of his administration to repeal
the Affordable Care Act and, pending that repeal, instructed the executive branch of the federal government to defer or delay
the implementation of any provision or requirement of the Affordable Care Act that would impose a fiscal burden on any state or
a cost, fee, tax or penalty on any individual, family, health care provider, or health insurer. Additionally, on October 12, 2017,
President Trump issued another executive order requiring the Secretaries of the Departments of Health and Human Services, Labor
and the Treasury to consider proposing regulations or revising existing guidance to allow more employers to form association health
plans that would be allowed to provide coverage across state lines, increase the availability of short-term, limited duration
health insurance plans, which are generally not subject to the requirements of the Affordable Care Act, and increase the availability
and permitted use of health reimbursement arrangements. On October 13, 2017, the DOJ announced that HHS was immediately stopping
its cost sharing reduction payments to insurance companies based on the determination that those payments had not been appropriated
by Congress. Furthermore, on December 22, 2017, President Trump signed tax reform legislation into law that, in addition to overhauling
the federal tax system, also, effective as of January 1, 2019, repeals the penalties associated with the individual mandate.
We cannot predict
the impact that the President’s executive order will have on the implementation and enforcement of the provisions of the
Affordable Care Act or the current or pending regulations adopted to implement the law. In addition, we cannot predict the impact
that the repeal of the penalties associated with the individual mandate and the cessation of cost sharing reduction payments to
insurers will have on the availability and cost of health insurance and the overall number of uninsured. We also cannot predict
whether the Affordable Care Act will be repealed, replaced, or modified, and, if the Affordable Care Act is repealed, replaced
or modified, what the replacement plan or modifications would be, when the replacement plan or modifications would become effective,
or whether any of the existing provisions of the Affordable Care Act would remain in place.
Our operations in international
markets involve inherent risks that we may not be able to control.
Our business plan
includes the marketing and sale of our existing and proposed products in international markets. Accordingly, our results could
be materially and adversely affected by a variety of uncontrollable and changing factors relating to international business operations,
including:
|
|
·
|
macroeconomic
conditions adversely affecting geographies where we intend to do business;
|
|
|
·
|
foreign
currency exchange rates;
|
|
|
·
|
political
or social unrest or economic instability in a specific country or region;
|
|
|
·
|
higher
costs of doing business in foreign countries;
|
|
|
·
|
infringement
claims on foreign patents, copyrights or trademark rights;
|
|
|
·
|
difficulties
in staffing and managing operations across disparate geographic areas;
|
|
|
·
|
difficulties
associated with enforcing agreements and intellectual property rights through foreign
legal systems;
|
|
|
·
|
trade
protection measures and other regulatory requirements, which affect our ability to import
or export our products from or to various countries;
|
|
|
·
|
adverse
tax consequences;
|
|
|
·
|
unexpected
changes in legal and regulatory requirements;
|
|
|
·
|
military
conflict, terrorist activities, natural disasters and medical epidemics; and
|
|
|
·
|
our
ability to recruit and retain channel partners in foreign jurisdictions.
|
Our financial results may be affected
by fluctuations in exchange rates.
Our financial statements
are presented in U.S. dollars, while a portion of our business is conducted, and a portion of our operating expenses are payable,
in Canadian dollars. Due to possible substantial volatility of currency exchange rates, exchange rate fluctuations may have an
adverse impact on our future revenues or expenses presented in our financial statements. Our results of operations could be adversely
affected if we are unable to successfully manage currency fluctuations in the future.
Any weakness in internal control
over financial reporting or disclosure controls and procedures could result in a loss of investor confidence in our financial
reports and lead to a stock price decline.
We are required to
evaluate our internal control over financial reporting under Section 404 of the Sarbanes-Oxley Act of 2002 and report the results
in our Annual Report on Form 10-K. We are also required to maintain effective disclosure controls and procedures. If material
weaknesses arise and they are not remedied, we will be unable to assert that our internal controls are effective. Any failure
to have effective internal control over financial reporting or disclosure controls and procedures could cause investors to lose
confidence in the accuracy and completeness of our financial reports, limit our ability to raise financing or lead to regulatory
sanctions, any of which could result in a material adverse effect on our business or decline in the market price of our common
stock.
The industries in which we operate
are highly competitive and subject to rapid technological change. If our competitors are better able to develop and market products
that are safer, more effective, less costly, easier to use, or are otherwise more attractive, we may be unable to compete effectively
with other companies.
The medical technology
industry is characterized by intense competition and rapid technological change and we will face competition on the basis of product
features, clinical outcomes, price, services and other factors. Competitors may include large medical device and other companies,
some of which have significantly greater financial and marketing resources than we do, and firms that are more specialized than
we are with respect to particular markets. Our competition may respond more quickly to new or emerging technologies, undertake
more extensive marketing campaigns, have greater financial, marketing and other resources than ours or may be more successful
in attracting potential customers, employees and strategic partners.
Our competitive position
will depend on multiple, complex factors, including our ability to achieve market acceptance for our products, develop new products,
implement production and marketing plans, secure regulatory approvals for products under development and protect our intellectual
property. In some instances, competitors may also offer, or may attempt to develop, alternative therapies that may be delivered
without a medical device or a medical device superior to ours. The development of new or improved products, processes or technologies
by other companies may render our products or proposed products obsolete or less competitive. The entry into the market of manufacturers
located in low-cost manufacturing locations may also create pricing pressure, particularly in developing markets. Our future success
depends, among other things, upon our ability to compete effectively against current technology, as well as to respond effectively
to technological advances, and upon our ability to successfully implement our marketing strategies and execute our research and
development plan.
We face competition from other medical
device companies that focus on robotic rehabilitation solutions to individuals with neurological disorders.
We face competition
from other companies that also focus on robotic rehabilitation solutions to individuals with neurological disorders. Hocoma, Motorika
and Tyromotion are each currently selling products that may compete with our InMotion™ product and we believe that there
are other smaller potential competitors in various stages of development that may compete with us directly or indirectly. Cyberdyne
and Honda are the main competitors of one of our consumer development products. These companies have longer operating histories
and may have greater name recognition and substantially greater financial, technical and marketing resources than us. Many of
these companies also have FDA or other applicable governmental approval to market and sell their products, and more extensive
customer bases, broader customer relationships and broader industry alliances than us, including relationships with many of our
potential customers. Increased competition from any of these sources could result in our failure to achieve and maintain an adequate
level of customers and market share to support the cost of our operations. We expect similar strong competition with respect to
any other product or technology we develop or acquire.
Our industry is experiencing greater
scrutiny and regulation by governmental authorities, which may lead to greater governmental regulation in the future.
In recent years, the
medical device industry has been subject to increased regulatory scrutiny, including by the FDA, Health Canada and numerous other
federal, state, provincial and foreign governmental authorities. This has included increased regulation, enforcement, inspections,
and governmental investigations of the medical device industry and disclosure of financial relationships with health care professionals.
We anticipate that governments will continue to scrutinize our industry closely, and that additional regulation by governmental
authorities, both foreign and domestic, may increase compliance costs, exposure to litigation and other adverse effects to our
operations.
Unsuccessful clinical trials or
procedures relating to products under development could have a material adverse effect on our prospects.
The regulatory approval
process for new products and new indications for existing products requires extensive clinical trials and procedures, including
early clinical experiences and regulatory studies. Unfavorable or inconsistent clinical data from current or future clinical trials
or procedures conducted by us, our competitors, or third parties, or perceptions regarding this clinical data, could adversely
affect our ability to obtain necessary approvals and the market’s view of our future prospects. Such clinical trials and
procedures are inherently uncertain and there can be no assurance that these trials or procedures will be completed in a timely
or cost-effective manner or result in a commercially viable product. Failure to successfully complete these trials or procedures
in a timely and cost-effective manner could have a material adverse effect on our prospects. Clinical trials or procedures may
experience significant setbacks even after earlier trials have shown promising results. Further, preliminary results from clinical
trials or procedures may be contradicted by subsequent clinical analysis.
In addition, results
from our clinical trials or procedures may not be supported by actual long-term studies or clinical experience. If preliminary
clinical results are later contradicted, or if initial results cannot be supported by actual long-term studies or clinical experience,
our business could be adversely affected. Clinical trials or procedures may be suspended or terminated by us, the FDA or other
regulatory authorities at any time if it is believed that the trial participants face unacceptable health risks.
Intellectual property litigation
and infringement claims could cause us to incur significant expenses or prevent us from selling certain of our products.
The industry in which
we operate, including, in particular, the medical device industry, are characterized by extensive intellectual property litigation
and, from time to time, we might be the subject of claims by third parties of potential infringement or misappropriation. Regardless
of outcome, such claims are expensive to defend and divert the time and effort of our management and operating personnel from
other business issues. A successful claim or claims of patent or other intellectual property infringement against us could result
in our payment of significant monetary damages and/or royalty payments or negatively impact our ability to sell current or future
products in the affected category and could have a material adverse effect on its business, cash flows, financial condition or
results of operations.
If we are unable to protect our
patents or other proprietary rights, or if we infringe on the patents or other proprietary rights of others, our competitiveness
and business prospects may be materially damaged.
We own 4 U.S. patents
and 1 U.S. pending patent, all 5 of which are pending internationally, as well as other patents under development. We also have
exclusive licensing rights to three patents of which one relates to components of our InMotion™ robots. We intend to continue
to seek legal protection, primarily through patents, trade secrets and contractual provisions, for our proprietary technology,
as cash flow allows. Such methods may not be adequate to protect us or permit us to gain or maintain a competitive advantage.
Seeking patent protection is a lengthy and costly process, which we can give no assurance of success and there can be no assurance
that patents will be issued from any pending applications, or that any claims allowed from existing or pending patents will be
sufficiently broad or strong to protect our proprietary technology. There is also no guarantee that any patents we hold will not
be challenged, invalidated or circumvented, or that the patent rights granted will provide competitive advantages to us. Our competitors
have developed and may continue to develop and obtain patents for technologies that are similar or superior to our technologies.
In addition, the laws of foreign jurisdictions in which we develop, manufacture or sell our products may not protect our intellectual
property rights to the same extent, as do the laws of the United States and Canada.
Despite our efforts
to safeguard our unpatented and unregistered intellectual property rights, we may not be successful in doing so or the steps taken
by us in this regard may not be adequate to detect or deter misappropriation of our technologies or to prevent an unauthorized
third party from copying or otherwise obtaining and using our products, technologies or other information that we regard as proprietary.
Additionally, third parties may be able to design around our patents. Our inability to adequately protect our intellectual property
could allow our competitors and others to produce products based on our technologies, which could substantially impair our ability
to compete.
Adverse outcomes in
current or future legal disputes regarding patent and other intellectual property rights or our ability to bring or defend against
such actions due to lack of funds could result in the loss of our intellectual property rights, subject us to significant liabilities
to third parties, require us to seek licenses from third parties on terms that may not be reasonable or favorable to us, prevent
us from manufacturing, importing or selling our products, or compel us to redesign our products to avoid infringing third parties’
intellectual property. As a result, we may be required to incur substantial costs to prosecute, enforce or defend our intellectual
property rights if they are challenged. Any of these circumstances could have a material adverse effect on our business, financial
condition and resources or results of operations.
Our ability to develop
intellectual property depends in large part on hiring retaining and motivating highly qualified design and engineering staff with
the knowledge and technical competence to advance our technology and productivity goals. We have entered into confidentiality
and/or intellectual property assignment agreements with many of our employees and consultants as one of the ways we seek to protect
our intellectual property and other proprietary technologies. However, these agreements may not be enforceable or may not provide
meaningful protection for our trade secrets or other proprietary information in the event of unauthorized use or disclosure or
other breaches of the agreements.
Our employees and
consultants may unintentionally or willfully disclose our confidential information to competitors, and confidentiality agreements
may not provide an adequate remedy in the event of unauthorized disclosure of confidential information. Enforcing a claim that
a third party illegally obtained and is using our proprietary know-how is expensive and time-consuming, and the outcome is unpredictable.
In addition, courts outside the United States are sometimes less willing to protect know-how than courts in the United States.
Moreover, our competitors may independently develop equivalent knowledge, methods and know-how. Failure to obtain or maintain
intellectual property protection could adversely affect our competitive business position.
RISKS RELATED TO OUR SECURITIES
AND GOVERNANCE MATTERS
The concentration
of our capital stock ownership with insiders will likely limit your ability to influence corporate matters.
Our
executive officers, directors, and their affiliated entities together beneficially own approximately 40% of our outstanding common
stock. As a result, these stockholders, if they act together or in a block, could have significant influence over virtually all
matters that require approval by our stockholders, including the election of directors and approval of significant corporate transactions,
even if other stockholders oppose them. This concentration of ownership might also have the effect of delaying or preventing a
change of control of our company that other stockholders may view as beneficial.
We may
have undisclosed liabilities and any such liabilities could harm our revenues, business, prospects, financial condition and results
of operations.
Before
our going-public transaction in 2015 with Drywave, a public shell company that at the time was a start-up designer and manufacturer
of massage systems, Bionik Canada conducted due diligence on the Company it believed was customary and appropriate for similar
transactions. However, the due diligence process may not have revealed all material liabilities of the Company then existing or
which may be asserted in the future against us relating to the Company’s activities before the consummation of the going-public
transaction with Drywave. In addition, the agreement with the Company contains representations with respect to the absence of
any liabilities and indemnification for any breach thereof. However, there can be no assurance that the Company had no liabilities
upon the closing of the going-public transaction with Drywave or that we will be successful in enforcing the indemnification provisions
or that such indemnification provisions will be adequate to reimburse us. Any such liabilities of the Company that survive the
going-public transaction with Drywave could harm our revenues, business, prospects, financial condition and results of operations.
We do not expect to pay cash dividends
on our common stock.
We anticipate that
we will retain our earnings, if any, for future growth and therefore do not anticipate paying cash dividends on our common stock
in the future. Investors seeking cash dividends should not invest in our common stock for that purpose.
Anti-takeover provisions in the
Company’s charter and bylaws may prevent or frustrate attempts by stockholders to change the board of directors or current
management and could make a third-party acquisition of the Company difficult.
The Company’s
Amended and Restated Certificate of Incorporation and Amended and Restated Bylaws contain provisions that may discourage, delay
or prevent a merger, acquisition or other change in control that stockholders may consider favorable, including transactions in
which stockholders might otherwise receive a premium for their shares. These provisions could limit the price that investors might
be willing to pay in the future for shares of the Company’s common stock.
We cannot assure you that the Company’s
Common Stock will be listed on any national securities exchange or remain listed or quoted.
We cannot assure you
that the Company’s common stock or other securities will ever be listed on any national securities exchange. Our stock began
trading on the OTCQB market from the OTCQX market on August 14, 2017. If our Common Stock remains quoted on or reverts to an over-the-counter
system rather than being listed on a national securities exchange, an investor may find it more difficult to dispose of shares
or obtain accurate quotations as to the market value of the Company’s Common Stock.
We may not be able to establish
a liquid market for the Company’s Common Stock or attract the attention of research analysts at major brokerage firms
We have been unable
to establish a liquid market for the Company’s Common Stock. Moreover, we do not expect security analysts of brokerage firms
to provide coverage of the Company in the near future unless we successfully uplist to a national securities exchange. Investment
banks may be less likely to agree to underwrite secondary offerings on behalf of the Company or our stockholders due to our becoming
a public reporting company not by means of an initial public offering of Common Stock. If all or any of the foregoing risks occur,
it would have a material adverse effect on the Company.
We cannot predict
whether an active market for the Company’s Common Stock will ever develop in the future. In the absence of an active trading
market:
|
|
·
|
Investors may have difficulty buying and selling or obtaining
market quotations;
|
|
|
·
|
Market visibility for shares of the Company’s Common Stock
may be limited; and
|
|
|
·
|
A lack of visibility for shares of the Company’s Common
Stock may have a depressive effect on the market price for shares of the Company’s Common Stock.
|
The Company’s
Common Stock is quoted on the OTCQB marketplace operated by OTC Markets Group, Inc. since August 14, 2017 as a result of not meeting
the net tangible asset requirements of the OTCQX market. These markets are relatively unorganized, interdealer, over-the-counter
markets that provide significantly less liquidity than NASDAQ or the NYSE. No assurances can be given that our Common Stock will
ever actively trade on such markets, much less a senior market like the Nasdaq Capital Market. In any of these events, there could
remain a highly illiquid market for the Company’s Common Stock and you may be unable to dispose of your Common Stock at
desirable prices or at all.
An active and visible public trading
market for the Company’s Common Stock may not develop and the market for our Common Stock is limited.
Our Common Stock is
thinly traded, and any recently reported sales price may not be a true market-based valuation of our Common Stock. In addition,
the stock market in general has experienced extreme price and volume fluctuations that have often been unrelated or disproportionate
to operating performance. Consequently, holders of shares of our common stock may not be able to liquidate their investment in
the Company’s shares at prices that they may deem appropriate.
The market price for our Common
Stock may be volatile.
The market price for
our Common Stock may be volatile and subject to wide fluctuations in response to factors including the following:
|
|
·
|
Actual or anticipated fluctuations in our quarterly or annual
operating results;
|
|
|
·
|
Changes in financial or operational estimates or projections;
|
|
|
·
|
Conditions in markets generally;
|
|
|
·
|
Changes in the economic performance or market valuations of
companies similar to ours;
|
|
|
·
|
Announcements by us or our competitors of new products, acquisitions,
strategic partnerships, joint ventures or capital commitments;
|
|
|
·
|
Our intellectual property position; and
|
|
|
·
|
General economic or political conditions in the United States,
Canada or elsewhere.
|
In addition, the securities
market has from time to time experienced significant price and volume fluctuations that are not related to the operating performance
of particular companies. These market fluctuations may also materially and adversely affect the market price of shares of our
common stock.
As our Common Stock is subject to
the SEC’s penny stock rules, broker-dealers may experience difficulty in completing customer transactions and trading activity
in our securities may be adversely affected.
The SEC has adopted
regulations, which generally define “penny stock” to be an equity security that has a market price of less than $5.00
per share, subject to specific exemptions. Until recently when we effected our 1:150 reverse stock split, our common stock had
a market price consistently below $5.00 per share. The market price of our Common Stock is currently and may in the future continue
to be less than $5.00 per share and therefore would be a “penny stock” according to SEC rules, unless we are listed
on a national securities exchange. Under these rules, broker-dealers who recommend such securities to persons other than institutional
accredited investors must:
|
|
·
|
Make a special written suitability determination for the purchaser;
|
|
|
·
|
Receive the purchaser’s prior written agreement to the
transaction;
|
|
|
·
|
Provide the purchaser with risk disclosure documents which identify
certain risks associated with investing in “penny stocks” and which describe the market for these “penny
stocks” as well as a purchaser’s legal remedies; and
|
|
|
·
|
Obtain a signed and dated acknowledgment from the purchaser
demonstrating that the purchaser has actually received the required risk disclosure document before a transaction in a “penny
stock” can be completed.
|
When our common stock
is subject to these rules, broker-dealers may find it difficult to effectuate customer transactions and trading activity in our
securities may be adversely affected. As a result, the market price of our securities may be depressed, and you may find it more
difficult to sell your securities.
IN ADDITION TO
THE ABOVE RISKS, BUSINESSES ARE OFTEN SUBJECT TO RISKS NOT FORESEEN OR FULLY APPRECIATED BY MANAGEMENT. IN REVIEWING THIS ANNUAL
REPORT ON FORM 10-K, POTENTIAL INVESTORS SHOULD KEEP IN MIND THAT THERE MAY BE OTHER POSSIBLE RISKS THAT COULD BE IMPORTANT.
Item 1B – Unresolved Staff Comments
None
Item 2 – Properties
Our principal executive
office is located in premises of approximately 3,655 square feet at 483 Bay Street, N105, Toronto, Ontario Canada M5G 2C9. The
facilities have been leased on our behalf by Ryerson University and we receive a subsidy on lease payments to the University.
We are also renting additional storage space. Our U.S. base of operations is located in approximately 9,300 square feet of leased
space at 80 Coolidge Hill Road, Watertown, Mass. 02472. We plan to move our US operations to more suitable space at some point,
now that we have outsourced manufacturing; however, no such space has been identified. Otherwise, we believe these facilities
are adequate for our current needs.
We do not own any
real estate.
Item 3 –
Legal Proceedings
From time to time,
we may become involved in various lawsuits and legal proceedings, which arise in the ordinary course of business. However, litigation
is subject to inherent uncertainties, and an adverse result in these or other matters may arise from time to time that may harm
business.
We are not currently
a party in any legal proceeding or governmental regulatory proceeding nor are we currently aware of any pending or potential legal
proceeding or governmental regulatory proceeding proposed to be initiated against us that would have a material adverse effect
on us or our business.
Item 4 – Mine Safety Disclosures
N/A
PART II
ITEM 5 - MARKET FOR REGISTRANT’S
COMMON EQUITY RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Information
Our common stock is
traded on the OTCQB marketplace under the symbol “BNKL” since August 14, 2017. Prior to that, our common stock was
traded on the OTCQX marketplace under the symbol “BNKL” since August 19, 2015. Prior to that, our common stock was
traded on the OTC Pink marketplace and was traded on such market prior to March 13, 2015 under the symbol “DWTP”.
Our common stock did not trade between approximately July 15, 2013 and February 23, 2015. The closing price for our common stock
on June 27, 2019 was $3.50 per share.
The following table
sets forth for the periods indicated the high and low sale prices per share of our common stock as reported on OTCQB marketplace,
but as adjusted to reflect our October 29, 2018 1:150 reverse stock split:
|
Quarterly Period Ended
|
|
High
|
|
|
Low
|
|
|
March 31, 2019
|
|
$
|
100.00
|
|
|
$
|
3.70
|
|
|
June 30, 2019 (through June 27, 2019)
|
|
$
|
5.05
|
|
|
|
2.95
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2018
|
|
$
|
27.00
|
|
|
$
|
9.75
|
|
|
June 30, 2018
|
|
$
|
12.60
|
|
|
$
|
6.30
|
|
|
September 30, 2018
|
|
$
|
10.50
|
|
|
$
|
4.80
|
|
|
December 31, 2018
|
|
$
|
12.50
|
|
|
$
|
3.00
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2017
|
|
$
|
222.00
|
|
|
$
|
54.00
|
|
|
June 30, 2017
|
|
$
|
71.25
|
|
|
$
|
31.65
|
|
|
September 30, 2017
|
|
$
|
45.00
|
|
|
$
|
15.75
|
|
|
December 31, 2017
|
|
$
|
36.75
|
|
|
$
|
15.00
|
|
We consider our common
stock to be thinly traded and, accordingly, reported sales prices or quotations may not be a true market- based valuation of our
common stock.
Holders
As of June 27, 2019,
3,702,398 shares of Common Stock were issued and outstanding, which were held by approximately 308 holders of record and 156,239
Exchangeable Shares were issued and outstanding, which were held by approximately 32 holders of record. The number of record
holders was determined from the records of our transfer agent and does not include beneficial owners of common stock whose shares
are held in the names of various security brokers, dealers, and registered clearing agencies.
Dividends
We have not paid any
dividends and we do not anticipate paying any cash dividends in the foreseeable future and we intend to retain all of our earnings,
if any, to finance our growth and operations and to fund the expansion of our business. Payment of any dividends will be made
in the discretion of our Board of Directors, after our taking into account various factors, including our financial condition,
operating results, current and anticipated cash needs and plans for expansion.
Equity Compensation Plan Information
We adopted, and a
majority of our stockholders approved, the 2014 Equity Incentive Plan (the “2014 Plan”). Under such plan, we may grant
equity based incentive awards, including options, restricted stock, and other stock-based awards, to any directors, employees,
advisers, and consultants that provide services to us or any of our subsidiaries on terms and conditions that are from time to
time determined by us. An aggregate of up to 15% of our common stock and common stock reserved for issuance from the Exchangeable
Shares are reserved for issuance under the 2014 Plan, and options for the purchase of 182,996 shares of our common stock have
been granted and are outstanding as of March 31, 2019. The purpose of the 2014 Plan is to provide financial incentives for selected
directors, employees, advisers, and consultants of the Company and/or its subsidiaries, thereby promoting the long-term growth
and financial success of the Company.
The table below sets
forth information as of March 31, 2019 with respect to compensation plans under which our common stock or Exchangeable Shares
are authorized for issuance, as adjusted to reflect the one-for-one hundred fifty reverse stock split.
|
|
|
(a)
Number of securities
to be Issued upon
exercise of
outstanding options,
warrants and rights
|
|
|
(b)
Weighted-
average
exercise price of
outstanding
options,
warrants and
rights
|
|
|
(c)
Number of securities
remaining available for
future issuance under
equity compensation
plans (excluding
securities reflected in
column (a))
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Equity compensation plans approved by security
holders
|
|
|
101,560
|
|
|
$
|
48.62
|
|
|
|
208,764
|
|
|
Equity compensation plans not approved by security holders:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Executive Stock Options
|
|
|
81,436
|
|
|
$
|
24.15
|
|
|
|
-
|
|
|
Total
|
|
|
182,996
|
|
|
$
|
37.73
|
|
|
|
208,764
|
|
ITEM 6 – SELECTED FINANCIAL DATA
This item is not required for a smaller
reporting company.
ITEM 7. MANAGEMENT’S
DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS.
The following Management’s
Discussion and Analysis of Financial Condition and Results of Operations (“MD&A”) covers as of March 31, 2019
and 2018, Except as otherwise noted, the financial information contained in this MD&A and in the financial statements has
been prepared in accordance with accounting principles generally accepted in the United States of America. All amounts are expressed
in U.S. dollars unless otherwise noted.
The preparation
of consolidated financial statements in conformity with U.S. GAAP requires us to make estimates and assumptions that affect the
reported amounts of assets and liabilities, disclosure of any contingent liabilities at the financial statement date and reported
amounts of revenue and expenses during the reporting period. On an on-going basis we review our estimates and assumptions. The
estimates were based on historical experience and other assumptions that we believe to be reasonable under the circumstances.
Actual results are likely to differ from those estimates under different assumptions or conditions, but we do not believe such
differences will materially affect our financial position or results of operations.
Forward Looking Statements
Certain information
contained in this MD&A includes “forward-looking statements.” Statements which are not historical reflect our
current expectations and projections about our future results, performance, liquidity, financial condition and results of operations,
prospects and opportunities and are based upon information currently available to us and our management and their interpretation
of what is believed to be significant factors affecting our existing and proposed business, including many assumptions regarding
future events. In some cases, you can identify forward-looking statements by terminology such as “may,” “will”
“should,” “expect,” “intend,” “plan,” anticipate,” “believe,”
“estimate,” “predict,” “potential,” “continue,” or similar terms, variations of
such terms or the negative of such terms. These statements are only predictions and involve known and unknown risks, uncertainties
and other factors. Although forward- looking statements, and any assumptions upon which they are based, are made in good faith
and reflect our current judgment, actual results could differ materially from those anticipated in such statements. Actual results,
performance, liquidity, financial condition and results of operations, prospects and opportunities could differ materially and
perhaps substantially from those expressed in, or implied by, these forward-looking statements as a result of various risks, uncertainties
and other factors, including those risks described in detail in the section of this Annual Report on Form 10-K entitled “Risk
Factors” as well as elsewhere in this Annual Report on Form 10-K.
In light of these
risks and uncertainties, and especially given the nature of our existing and proposed business, there can be no assurance that
the forward-looking statements contained in this section and elsewhere in this Annual Report on Form 10-K will in fact occur.
Potential investors should not place undue reliance on any forward-looking statements. Except as expressly required by the federal
securities laws, there is no undertaking to publicly update or revise any forward-looking statements, whether as a result of new
information, future events, changed circumstances or any other reason.
Plan of Operation and Corporate Developments
We are a global pioneering
robotics company focused on providing rehabilitation solutions to individuals with neurological disorders, specializing in the
designing, developing and commercializing of cost-effective physical rehabilitation technologies, prosthetics, and assisted robotic
products. We strive to innovate and build devices that improve an individual’s health, comfort, accessibility and quality
of life through the use of advanced algorithms and sensing technologies that anticipate a user’s ever move. Our product
line includes three FDA-listed upper extremity clinical rehabilitation products currently on the market for clinical use, a gait
rehabilitation product, a lower-body product being developed for the consumer market, as well as a potential pipeline to other
new product candidates.
Bionik Laboratories
Corp. was incorporated on January 8, 2010 in the State of Colorado. At the time of our incorporation the name of our company was
Strategic Dental Management Corp. On July 16, 2013, the Company changed its name from Strategic Dental Management Corp. to Drywave
Technologies, Inc. and changed its state of incorporation from Colorado to Delaware. Effective February 13, 2015, we changed our
name to Bionik Laboratories Corp.
Bionik Canada was incorporated on March
24, 2011 under the Canada Business Corporations Act. On February 26, 2015, we:
|
|
·
|
Acquired
100 Class 1 common shares of Bionik Canada representing 100% of the outstanding Class
1 common shares of Bionik Canada. After giving effect to this transaction, we commenced
operations through Bionik Canada; and
|
|
|
·
|
Immediately
prior thereto, we transferred all of the legacy business, properties, assets, operations
and goodwill of the Company (other than cash and cash equivalents), and liabilities,
so that as of the Company’s acquisition of Bionik Canada, the Company had no material
assets or liabilities.
|
As a result of the shareholders of Bionik
Canada having a controlling interest in the Company subsequent to the February 2015 transaction, for accounting purposes the transaction
did not constitute a business combination, and instead has been accounted for as a recapitalization of the Company with Bionik
Canada being the accounting acquirer even though the legal acquirer is the Company.
On April 21, 2016,
we acquired all of the outstanding shares and, accordingly, all assets and liabilities of IMT, a Boston, Massachusetts-based global
pioneer and leader in providing effective robotic tools for neurorehabilitation, pursuant to an Agreement and Plan of Merger,
dated March 1, 2016, with IMT, Hermano Igo Krebs, and Bionik Mergerco Inc., a Massachusetts corporation and our wholly owned subsidiary,
which provided for the merger of Bionik Mergerco with and into IMT, with IMT surviving the merger as our wholly-owned subsidiary.
In return for acquiring IMT, IMT shareholders received 23,650,000 shares of our common stock.
Significant Accounting Policies and
Estimates
The discussion and
analysis of the financial condition and results of operations are based upon the financial statements, which have been prepared
in accordance with accounting principles generally accepted in the United States. The preparation of consolidated financial statements
in conformity with accounting principles generally accepted in the United States requires us to make estimates and assumptions
that affect the reported amounts of assets and liabilities, disclosure of any contingent liabilities at the financial statement
date and reported amounts of revenue and expenses during the reporting period. On an on-going basis we review our estimates and
assumptions. The estimates were based on historical experience and other assumptions that we believe to be reasonable under the
circumstances. Actual results are likely to differ from those estimates under different assumptions or conditions, but we do not
believe such differences will materially affect our financial position or results of operations.
The adoption of the
FASB issued, ASU No. 2017-11,
Earnings Per Share (Topic 260) Distinguishing Liabilities From Equity (Topic 480) Derivatives
and Hedging (Topic 815): I. Accounting for Certain Financial Instruments With Down Round Features II. Replacement of the Indefinite
Deferral for Mandatorily Redeemable Financial Instruments of Certain Nonpublic Entities and Certain Mandatorily Redeemable Noncontrolling
Interests With a Scope Exception
, allows a financial instrument with a down-round feature to no longer automatically be classified
as a liability solely based on the existence of the down-round provision. The update means the instrument does not have to be
accounted for as a derivative and be subject to an updated fair value measurement each reporting period. The Company adopted ASU
No. 2017-11 in the quarter ended September 30, 2017. Accordingly, we have reissued our audited financial statements for the fiscal
years ended March 31, 2017 and 2016 in accordance with SEC rules to reflect this adoption.
Results of Operations
From the inception
of Bionik Canada on March 24, 2011 through to March 31, 2019, we have generated a deficit of $46,357,373.
We expect to incur
additional operating losses through the fiscal year ending March 31, 2019 and beyond, principally as a result of our continuing
research and development, building the sales and marketing team, long sales cycles and general and administrative costs predominantly
associated with being a public company.
For the Fiscal Year Ended
March 31, 2019 Compared to the Fiscal Year Ended March 31, 2018
Sales
Sales were $3,246,038
for the fiscal year ended March 31, 2019 (March 31, 2018 - $987,431). The revenues for the fiscal year ended March 31, 2019 are
comprised of sales of 33 (March 31, 2018 – 11) InMotion™ robots, service and warranty income.
Cost of Sales and Gross Margin
Cost of sales was
$1,630,166 for the fiscal year ended March 31, 2019 (March 31, 2018- $402,665), In fiscal year 2019, cost of sales included inventory
write downs totaling $62,589 (2018 - $38,860) and product cost of sales of $1,567,577 (March 31, 2018 - $363,805).
If the inventory write
downs were excluded from the gross margin of $1,615,872 (March 31, 2018- $584,766), it would result in a gross margin before inventory
write-downs of $1,678,461 (March 31, 2018- $623,626).
Operating Expenses
Total operating expenses
for the fiscal year ended March 31, 2019 were $11,103,252 and for the year ended March 31, 2018 was $10,354,032, as further described
below.
For the fiscal year
ended March 31, 2019, the Company incurred $2,339,359 in sales and marketing expenses and for the year ended March 31, 2018 –
$1,989,837. The increase in these expenses by $349,522 is due to the cost of hiring additional marketing and sales support employees,
increased sales related expenses including commissions, as well as increased presence at conferences in fiscal 2019 over fiscal
2018.
For the fiscal year
ended March 31, 2019, the Company incurred research and development expenses of $3,174,892 (March 31, 2018– $2,825,200).
The increase in research and development expenses relates primarily to the additional hires to strengthen the development team
to support our new development projects.
The Company incurred
general and administrative expenses of $3,893,393 for the fiscal year ended March 31, 2019 and $3,585,484 for the fiscal year
ended March 31, 2018. The increase in general and administrative expenses in 2019 over 2018 resulted from higher audit and legal
related costs related to company financing activities and the addition of four new members to the Board of Directors and the write-off
of $293,188 associated with an unsuccessful public capital raise.
Stock compensation
expense was $1,347,399 for the fiscal year ended March 31, 2019, compared to $1,540,580 for the fiscal year ended March 31, 2018,
due to fewer option grants in the year ended March 31, 2019 compared to the fiscal year ended March 31, 2018.
Amortization of technology
and other assets allocated from the purchase of IMT was $278,997 for the fiscal year ended March 31, 2019 (March 31, 2018 –
$323,905). The amortization has decreased as certain assets acquired have been fully amortized. Assets acquired were workforce
and non-compete agreements which is now fully amortized. Customer relationships is amortized over 10 years, patents and our exclusive
license agreements over their lifetime and trademarks are indefinite and therefore are not amortized.
Depreciation amounted
to $69,212 for the fiscal year ended March 31, 2018 (March 31, 2017 – $89,026).
Other Expenses
For the fiscal year
ended March 31, 2019, the Company recorded $3,266,918 as accretion expense compared to $1,937,308 for the fiscal year ended March
31, 2018 due to the amortization of the fair value of the Company’s March 28, 2019 convertible notes offering as well as
the conversion feature recorded in connection with the conversion of the July 20,2019 convertible debt financing.
For the fiscal year
ended March 31, 2019, we expensed share premium expense of $Nil (March 31, 2018 – $1,249,994). For the fiscal year ended
March 31, 2018, the share premium expense was related to the Company’s convertible promissory notes and represents 25% of
the principle investment amount of the original convertible promissory loans.
For the fiscal year
ended March 31, 2019, we had a gain in a fair value adjustment of $337,923 (March 31, 2018 - $Nil) related to not having enough
shares at March 31, 2018 to fully convert certain loans and issuance of shares in June 2018, resulted in this gain.
For the fiscal year
ended March 31, 2019, we had a gain of $2,048,697 (March 31, 2018 – ($376,674)) on the mark to market reevaluation of the
shares to be issued as of March 31, 2018 due to not having enough authorized shares to issue the shares of common stock upon conversion
of our convertible promissory notes on March 31, 2018 as referred to above.
For the fiscal year
ended March 31, 2019, we incurred other expense of $262,596 (March 31, 2018 – $1,297,205). The decrease in other expenses
relates to lower interest expense in connection with indebtedness in fiscal year ended March 31, 2019 compared to the fiscal year
ended March 31, 2018 due to the lowering of the interest rate from 3% to 1% per month.
For the fiscal year
ended March 31, 2019, we incurred a foreign exchange gain of $507 (March 31, 2018 – ($102,999)). On April 1, 2015, our subsidiaries
changed their functional currency from the Canadian Dollar to the U.S. Dollar. This reflects the fact that the majority of the
Company’s business is influenced by an economic environment denominated in U.S. currency as well as that the Company anticipates
revenues to be earned in U.S. dollars.
Other Income
For the fiscal year
ended March 31, 2019, other income was $73,166 and for the fiscal year ended March 31, 2018, other income was $107,656, in each
case related to interest and other income.
Comprehensive Loss
Comprehensive loss
for the fiscal year ended March 31, 2019 was ($10,556,601) resulting in loss per share of $(4.47) and for the fiscal year ended
March 31, 2018, after retroactive adoption of ASU 2017-11 noted above, comprehensive loss was $(14,625,790), resulting in loss
per share of ($21.73). The decrease in the comprehensive loss is primarily due to higher revenue and gains from mark to market
reevaluation related to shares issued at March 31, 2018 in the current year and the decrease in the earnings per shares is due
to the increase in common shares issued due to the convertible loan conversions.
Liquidity and Capital Resources
We have funded operations
through the issuance of capital stock, loans, grants and investment tax credits received from the Government of Canada. The Company
raised in its 2015 private offering net proceeds of $11,341,397. Since 2015, the Company also obtained funds through additional
government tax credits, incurring new convertible indebtedness totaling $18,469,681 that has since been converted into equity,
a short-term loan of $400,000, that was repaid and raising $1,125,038 in June 2017 from its warrant solicitation. At March 31,
2019, the Company had cash and cash equivalents of $446,779 (March 31, 2018- $507,311). Subsequent to March 31, 2019, the Company
has obtained a $500,000 term loan from its Chairman and commenced an up to $9 million convertible loan raise, of which $700,000
has been raised through June 27, 2019.
Based on our current
burn rate, we need to raise additional capital in the short term to fund operations and meet expected future liquidity requirements,
as well as to repay our remaining existing indebtedness, or we will be required to curtail or terminate some or all of our product
lines or our operations. We are continuously in discussions to raise additional capital, which may include or be a combination
of convertible loans and equity which, if successful, will enable us to continue operations based on our current burn rate, for
the next 12 months; however, we cannot give any assurance at this time that we will successfully raise all or some of such capital
or any other capital. While a director and major stockholder has committed to invest additional funds to allow us to continue
to operate through June 15, 2019, we do not have any definitive agreement with such person. We recently were not successful in
raising funds through the sale of equity in a public offering and have since commenced a new up to $9 million convertible notes
financing round of which $700,000 has been raised through June 27, 2019. Furthermore, we do not have an established source of
funds sufficient to cover operating costs after July 30 , 2019 at this time and accordingly, there can be no assurance that the
$9 million convertible note financing round will be successful or other necessary debt or equity financing will be available,
or will be available on terms acceptable to us, in which case we may be unable to meet our obligations or fully implement our
business plan, if at all. These conditions however raise substantial doubt about the Company’s ability to continue as a
going concern. The accompanying condensed consolidated interim financial statements do not include any adjustments to reflect
the possible future effects on recoverability and reclassification of assets or the amounts and classification of liabilities
that may result from the outcome of this uncertainty.
Additionally, we will
need additional funds to respond to business opportunities including potential acquisitions of complementary technologies, protect
our intellectual property, develop new lines of business and enhance our operating infrastructure. While we may need to seek additional
funding for any such purposes, we may not be able to obtain financing on acceptable terms, or at all. In addition, the terms of
our financings may be dilutive to, or otherwise adversely affect, holders of our common stock. We will also seek additional funds
through arrangements with collaborators or other third parties. We may not be able to negotiate any such arrangements on acceptable
terms, if at all. If we are unable to obtain additional funding on a timely basis, we may be required to curtail or terminate
some or all of our product lines or our operations.
Net Cash Used in Operating Activities
During the fiscal
year ended March 31, 2019, we used cash in operating activities of $(9,232,411). The increased use of cash in the fiscal year
ended March 31, 2019, compared to a use of $(7,710,862) for the year ended March 31, 2018 is mainly attributable to the build-up
of inventory and accounts receivable due to the increase in sales in the current fiscal year.
Net Cash Used in Investing Activities
During the fiscal
year ended March 31, 2019, net cash used in investing activities was $(101,779), compared to $(21,567) for the fiscal year ended
March 31, 2018. In the fiscal years ended March 31, 2019 and 2018, there was no investment activity.
Net cash used in investing
activities in 2019 and 2018 was used for the acquisition of equipment related to the Company’s purchase of additional computer
equipment due to the increase in engineers, equipment to help with the development of our technology and demo units to assist
in the sales process.
Net Cash Provided by Financing Activities
Net cash provided
by financing activities was $9,273,658 for the fiscal year ended March 31, 2019 compared to $7,696,090 for the fiscal year ended
March 31, 2018. The increase from the 2019 fiscal period to the 2018 fiscal period is due to successfully raising more capital
in the 2019 fiscal period than the 2018 fiscal period.
Newly Adopted and Recently Issued Accounting
Pronouncements
Newly Adopted
In May 2014, the Financial
Accounting Standards Board (“FASB”) issued Accounting Standards Update (“ASU”) No. 2014-09, Revenue from
Contracts with Customers (Topic 606). The updated standard will replace most existing revenue recognition guidance in U.S. GAAP.
The new standard introduces a five-step process to be followed in determining the amount and timing of revenue recognition. It
also provides guidance on accounting for costs incurred to obtain or fulfill contracts with customers and establishes disclosure
requirements which are more extensive than those required under existing U.S. GAAP. The FASB has issued numerous amendments to
ASU 2014-09 from August 2015 through January 2018, which provide supplemental and clarifying guidance, as well as amend the effective
date of the new standard. ASU 2014-09, as amended, is effective for the Company in the interim period ended June 30, 2018. The
standard permits the use of either the retrospective or modified retrospective (cumulative effect) transition method. The Company
adopted the new standard using the modified retrospective transition method. The Company has adopted ASU-2014-1 for the fiscal
year ended March 31, 2019 and it did not have a material effect on the consolidated financial position and the consolidated results
of operations.
In November 2015,
the FASB issued ASU No. 2015-17, “Balance Sheet Classification of Deferred Taxes,” which require that deferred tax
liabilities and assets be classified on our Consolidated Balance Sheets as noncurrent based on an analysis of each taxpaying component
within a jurisdiction. ASU No. 2015-17 is effective for the fiscal year commencing after December 15, 2017. The Company has adopted
ASU-2015-17 for the fiscal year ended March 31, 2019 and it did not have a material effect on the consolidated financial position
or the consolidated results of operations.
In January 2016, the
FASB issued ASU No. 2016-01 Financial Instruments - Overall (Subtopic 825-10): Recognition and Measurement of Financial Assets
and Financial Liabilities. The updates make several modifications to Subtopic 825-10, including the elimination of the available-for-sale
classification of equity investments, and it requires equity investments with readily determinable fair values to be measured
at fair value with changes in fair value recognized in operations.
The update is effective
for fiscal years beginning after December 2017. The Company has adopted ASU 2016-01 for the year ended March 31, 2019 and it did
not have a material effect on the consolidated financial position and the consolidated results of operations.
In February 2016,
the FASB issued ASU 2016-02, Leases. This update requires organizations that lease assets to recognize on the balance sheet the
assets and liabilities for the rights and obligations created by those leases. The new guidance will also require additional disclosure
about the amount, timing and uncertainty of cash flows arising from leases. The provisions of this update are effective for annual
and interim periods beginning after December 15, 2018. The Company adopted ASU 2016-02 and it did not have a material effect on
the consolidated financial position and the consolidated results of operations.
In August 2016, the
FASB issued ASU 2016-15, “Statement of Cash Flows: Classification of Certain Cash Receipts and Cash Payments”. This
ASU provides eight targeted changes to how cash receipts and cash payments are presented and classified in the statement of cash
flows. ASU 2016-15 is effective for the fiscal year commencing after December 15, 2017. The Company has adopted ASU 2016-15 for
the fiscal year ended March 31, 2019 and it did not have material effect on the consolidated financial position or on the consolidated
statement of cash flows.
In May 2017, the FASB
issued ASU No. 2017-09, Compensation - Stock Compensation (Topic 718): Scope of Modification Accounting (ASU 2017-09). The FASB
issued the update to provide clarity and reduce the cost and complexity when applying the guidance in Topic 718. The amendments
in this update provide guidance about which changes to the terms or conditions of a share-based payment award require an entity
to apply modification accounting in Topic 718. The Company adopted ASU 2017-09 during the year ended March 31, 2019 and it did
not have a material effect on the consolidated financial statements and the consolidated results of operations.
Recently Issued
In January 2017, the
FASB issued ASU 2017-01, “Business Combinations: Clarifying the definition of a Business” which amends the current
definition of a business. Under ASU 2017-01, to be considered a business, an acquisition would have to include an input and a
substantive process that together significantly contributes to the ability to create outputs. ASU 2017-01 further states that
when substantially all of the fair value of gross assets acquitted is concentrated in a single asset (or a group of similar assets),
the assets acquired would not represent a business. The new guidance also narrows the definition of the term “outputs”
to be consistent with how it is described in Topic 606, Revenue from Contracts with Customers. The changes to the definition of
a business will likely result in more acquisitions being accounted for as asset acquisitions. ASU 2017-01 is effective for acquisitions
commencing on or after June 30, 2019, with early adoption permitted. Adoption of this guidance will be applied prospectively on
or after the effective date and the Company does not expect this policy will have a material effect on the consolidated financial
position or consolidated statement of cash flows.
In January 2017, the
FASB issued ASU 2017-04, “Intangibles – Goodwill and Other” ASU 2017-04 simplifies the accounting for goodwill
impairment by eliminating Step 2 of the current goodwill impairment test, which required a hypothetical purchase price allocation.
Goodwill impairment will now be the amount by which the reporting unit’s carrying value exceeds its fair value, limited
to the carrying value of the goodwill. ASU 2017-04 is effective for financial statements issued for fiscal years, and interim
periods beginning after December 15, 2019. The Company is still assessing the impact that the adoption of ASU 2017-04 will have
on the consolidated statement of financial position and consolidated statement of operations.
In June 2016, the
FASB issued ASU 2016-13 Financial Instruments – Credit Losses (Topic 326): Measurement of Credit Losses of Financial Instruments,
which introduces an expected credit loss methodology for the impairment of financial assets measured at amortized cost basis.
The methodology replaces the probable, incurred loss model for those assets. The update if effective for fiscal years beginning
after December 15, 2019. The Company is still assessing the impact that the adoption of ASU 2016-13 will have on the consolidated
statement of financial position and consolidated statement of operations.
In February 2016,
the FASB issued ASU 2016-02, Leases. This update requires organizations that lease assets to recognize on the balance sheet the
assets and liabilities for the rights and obligations created by those leases. The new guidance will also require additional disclosure
about the amount, timing and uncertainty of cash flows arising from leases. The provisions of this update are effective for annual
and interim periods beginning after December 15, 2018. The Company is still assessing the impact that the adoption of ASU 2016-02
will have on the consolidated statement of financial position and consolidated statement of operations.
Management does not
believe that any other recently issued, but not yet effective accounting pronouncements, if adopted, would have a material effect
on the accompanying condensed consolidated interim financial statements
Off-Balance Sheet Arrangements
We had no off-balance
sheet arrangements that have or are reasonably likely to have a current or future effect on our financial condition, changes in
financial condition, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources.
ITEM 7A. QUANTITATIVE
AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK.
This item is not required
for a smaller reporting company.
ITEM 8. FINANCIAL
STATEMENTS AND SUPPLEMENTARY DATA.
Our consolidated financial
statements and corresponding notes thereto called for by this item appear at the end of this document commencing on page F-1.
ITEM 9. CHANGES
IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE.
None.
ITEM 9A. CONTROLS
AND PROCEDURES.
Evaluation of Disclosure Controls and
Procedures
Under the supervision
and with the participation of our management, including our principal executive officer and the principal financial officer, we
have conducted an evaluation of the effectiveness of the design and operation of our disclosure controls and procedures, as defined
in Rules 13a-15(e) and 15d-15(e) under the Securities and Exchange Act of 1934, as of the end of the period covered by this report.
Based on this evaluation, our principal executive officer and principal financial officer concluded as of the evaluation date
that our disclosure controls and procedures were not effective to ensure that the material information required to be included
in our Securities and Exchange Commission reports is accumulated and communicated to our management, including our principal executive
and financial officer, recorded, processed, summarized and reported within the time periods specified in Securities and Exchange
Commission rules and forms relating to our company.
Management’s Annual Report on
Internal Control Over Financial Reporting
Management is responsible
for establishing and maintaining adequate internal control over financial reporting for our company and its subsidiaries. Our
management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is
defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act, for the Company.
Internal control over
financial reporting includes those policies and procedures that: (1) pertain to the maintenance of records that, in reasonable
detail, accurately and fairly reflect the transactions and dispositions of our assets; (2) provide reasonable assurance that transactions
are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles,
and that our receipts and expenditures are being made only in accordance with authorizations of its management and directors;
and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition
of our assets that could have a material effect on the financial statements.
Management recognizes
that there are inherent limitations in the effectiveness of any system of internal control, and accordingly, even effective internal
control can provide only reasonable assurance with respect to financial statement preparation and may not prevent or detect material
misstatements. In addition, effective internal control at a point in time may become ineffective in future periods because of
changes in conditions or due to deterioration in the degree of compliance with our established policies and procedures.
A material weakness
is a significant deficiency, or combination of significant deficiencies, that results in there being a more than remote likelihood
that a material misstatement of the annual or interim financial statements will not be prevented or detected.
Under the supervision
and with the participation of our Chief Executive Officer, management conducted an evaluation of the effectiveness of our internal
control over financial reporting, as of March 31, 2019 based on the framework set forth in Internal Control-Integrated Framework
issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). Based on our evaluation under this framework,
management concluded that our internal control over financial reporting was not effective as of the evaluation date due to the
factors stated below.
Management assessed
the effectiveness of the Company’s internal control over financial reporting as of the evaluation date and identified the
following material weaknesses:
|
|
·
|
Inadequate
Segregation of Duties: we have a lack of segregation of duties with internal accounting
control functions which is limited to a relatively few individuals in the accounting
department.
|
|
|
·
|
During
the fiscal year ended March 31, 2019, the Company appointed an independent Audit Committee
on May 30, 2018 and achieved a majority of independent outside Directors on the Company’s
Board of Directors by September 7, 2018. As we have only recently appointed an independent
audit committee and added board members to have a majority of Board members to be independent,
this results in ineffective oversight in the establishment and monitoring of required
internal controls and procedures for the entire fiscal year.
|
Management is committed to improving its
internal controls and will:
|
|
·
|
Continue
to use third party specialists to address shortfalls in staffing and to assist the Company
with accounting and finance responsibilities; and
|
|
|
·
|
Increase
the frequency of independent reconciliations of significant accounts, which will mitigate
the lack of segregation of duties until there are enough personnel.
|
Management, including
our Chief Executive Officer and Chief Financial Officer, has discussed the material weaknesses noted above with our independent
registered public accounting firm. Due to the nature of these material weaknesses, there is a more than remote likelihood that
misstatements which could be material to the annual or interim financial statements could occur that would not be prevented or
detected.
This annual report
does not include an attestation report of our registered public accounting firm regarding internal control over financial reporting.
Management’s report was not subject to attestation by the Company’s registered public accounting firm pursuant to
rules of the SEC that permit us to provide only management’s report in this annual report.
Changes in Internal Controls
There was no change
in our internal controls over financial reporting that occurred during the period covered by this report, which has materially
affected, or is reasonably likely to materially affect, our internal controls over financial reporting.
ITEM 9B. OTHER
INFORMATION.
Not applicable
PART
III
ITEM 10. -DIRECTORS, EXECUTIVE OFFICERS
AND CORPORATE GOVERNANCE
Directors and Executive Officers
Our executive officers and directors are as follows:
|
Name
|
|
Age
|
|
Position
|
|
|
|
|
|
|
|
Andre Auberton-Herve
|
|
57
|
|
Chairman of the Board
|
|
Eric Dusseux
|
|
51
|
|
Chief Executive Officer and Director
|
|
Michal Prywata
|
|
27
|
|
Chief Technology Officer and Director
|
|
Remi Gaston Dreyfus
|
|
64
|
|
Director
|
|
P. Gerald Malone
|
|
68
|
|
Director
|
|
Joseph Martin
|
|
71
|
|
Director
|
|
Charles Matine
|
|
61
|
|
Director
|
|
Audrey Thevenon
|
|
41
|
|
Director
|
|
Leslie Markow
|
|
58
|
|
Chief Financial Officer
|
Andre Auberton-Herve:
Chairman of the Board
. Mr. Auberton-Herve has been the Chairman of the Company’s Board of Directors since January
24, 2018. Mr. Auberton-Herve brings substantial leadership experience within strategic, operational, and financial activities
from past roles.
Mr. Auberton-Herve is the founder of 4A Consulting & Engineering, which
provides strategic advice and consulting services with respect to renewable energy and digital innovation and has served as its
President and CEO since its founding in July 2015. 4A Consulting provided consulting services to the Company from February 2017
until Mr. Auberton-Herve’s appointment as Chairman. Mr. Auberton-Herve co-founded Soitec SA, a publicly traded company on
the Euronext Paris stock exchange which designs and manufactures innovative semiconductor materials which are used in many smartphone
platforms and computing activities, where he was President and CEO from July 1992 until January 2015, then Chairman and Chairman
Emeritus since September 2015.
While at Soitec SA, Mr. Auberton-Herve was responsible for overseeing the strategic, operational
and financial activities of the company. He built an international high-tech group in ten countries and five manufacturing facilities
in Europe, Asia and the U.S. Mr. Auberton-Herve also led the company through its listing on Euronext in 1999, raising significant
amounts of capital since then with some of the world’s largest investment banks. He has been nominated Knight of the Legion
of Honor and Knight of the Order of Merit in France. Mr. Auberton-Herve holds a Doctorate degree in Semiconductor Physics and
a Master’s degree in Materials Science from Ecole Centrale de Lyon in France.
The
Company believes that Mr. Auberton-Herve is qualified as a board member of the Company because of his substantial strategic, operational
and leadership experience.
Dr. Eric Dusseux:
Chief Executive Officer and Director
. Dr. Dusseux has served as the Company’s Chief Executive Officer since September
1, 2017 and has served as a director since July 22, 2017. He is also a director of Mc10 Inc., a private company, which is developing
a hardware and software platform for biometric healthcare analytics. He was previously the President Europe at Auregen BioTherapeutics
SA and was a director at Auregen BioTherapeutics Inc., which is translating 3D bioprinting technology for innovative treatments
for patients with rare disorders, since February 2017. Prior to that, from November 2016 through January 2017, Dr. Dusseux was
President Europe at Bemido SA, a family office. From September 2012 to October 2016, Dr. Dusseux was an Executive Committee Member
in the Corporate Strategy Department of Sanofi Pasteur SA, the vaccines division of Sanofi, a global healthcare leader, where
he led corporate strategy, business intelligence, and international business development. He has also served in key roles at GlaxoSmithKline
Biologicals from January 2008 to June 2012, leading product development and business growth strategy. Dr. Dusseux also gained
significant experience providing strategic advice for numerous pharmaceutical, medical device, payer and biotechnology clients,
while working for the Boston Consulting Group from 2002 to 2007. Dr. Dusseux is a Medical Doctor, specializing in Public Health.
Dr. Dusseux also holds a Master of Science in Physical Chemistry and is a graduate of the French Business School H.E.C. in Paris
(MBA, Isa). We believe that Dr. Dusseux is qualified as a board member of the Company because of his substantial strategic and
leadership experience within the healthcare industry.
Michal Prywata:
Chief Technology Officer and Director
. Mr. Prywata is the co-founder of Bionik Canada and has served as our Chief Technology
Officer since June 2017, Chief Operating Officer from April 2013 to June 2017, as a director from March 2011 to September 2018,
and again since March 2019. Mr. Prywata previously served as our Chief Executive Officer from March 2011 to April 2013. Mr. Prywata
studied biomedical engineering at Ryerson University until the end of his second year, with a focus on electronics and software
development for medical products. He has a track record of winning technology showcases and inventing technologies that address
significant unmet needs and untapped markets. He has spent the past 5 years with Bionik Canada, managing technological advancements,
managing day-to-day operations, and developing concepts into products. In addition, Mr. Prywata, together with the Company’s
other co-founder and its former CEO, was responsible for raising and securing initial seed capital and subsequent capital raises.
Mr. Prywata is the co-inventor of the Company’s ARKE technology platform. Mr. Prywata serves as a member of the Board of
Directors due to his being a founder of the Company and his current executive position with the Company. We also believe that
Mr. Prywata is qualified due to his experience in the medical device industry.
Remi Gaston-Dreyfus:
Director
. Mr. Gaston-Dreyfus has been a director of the Company since September 1, 2017. Since 2007, Mr. Gaston-Dreyfus
has been the CEO and Founder of RGD Investissements S.A.S. in Paris, a developer of and investor in real estate assets in Paris.
Prior to 2007, Mr. Gaston-Dreyfus was a shareholder, Chairman and CEO of the Photo-Journalism group A.G.I. (including Gamma Press
Agency). Mr. Gaston-Dreyfus was a co-founder of a Parisian law firm in 1984 and was a French lawyer until 1992. We believe that
Mr. Gaston-Dreyfus is qualified to serve as a member of the Board of Directors due to his experience as an entrepreneur and his
legal training
P. Gerald Malone:
Director
. Mr. Malone has been a director of the Company since March 19, 2018. Since 1997, Mr. Malone has held a number
of directorships and chairmanships in private and AIM listed companies in the healthcare, IT and energy sectors in the UK and
the USA. He has extensive experience within the financial services sector, serving since 2001 as a board member and ultimately
Chairman of Aberdeen Asia-Pacific Income Fund (FAX), a U.S. closed-end mutual fund. He also serves as a director of a number of
other U.S. and Canadian closed- and open-end mutual funds, and of the Washington, D.C.-based Mutual Fund Directors Forum, a body
representing independent fund directors. A Scottish lawyer by profession, Mr. Malone was previously a Member of Parliament in
the U.K. from 1983 to 1997 and served as Minister of State for Health in John Major’s government from 1994 to 1997. Mr.
Malone is qualified as a board member of the Company because of his substantial commercial strategic, government and leadership
experience.
Joseph Martin:
Director
. Mr. Martin currently serves as Chairman of Brooks Automation, a global provider of automation, vacuum and instrumentation
solutions. He also serves as a director of Collectors Universe, Inc., a third-party grading and authentication service for high-value
collectibles, of Allegro Microsystems, a manufacturer of high-performance semiconductors for the automotive market, Fairchild
Semiconductor, ChipPAC Inc. and Soitec Inc. In 2000
CFO Magazine
awarded Mr. Martin the CFO of the Year award
for turnaround operations. Mr. Martin holds an Executive Masters certification from The American College of Corporate Directors.
We believe Mr. Martin is qualified to serve as a member of the Board of Directors due to his extensive board and financial expertise.
Charles Matine:
Director.
Mr. Matine serves as an Advisory Board Member of Enlaps, a start-up company providing a time-lapse solution
to photographers, since February 2018. Since July 2015, Mr. Matine has served as a strategic advisor to C4 Ventures, a London-based
venture fund supporting media, e-commerce and hardware startups. In April 2014, Mr. Matine founded B & Associates, a marketing
and digital transformation consultancy firm, and has served as its CEO since April 2014. Prior to that, Mr. Matine served as a
Business Unit Director of Apple France from July 2010 to April 2014, where he led the Education and Research business unit, and
as a Senior Marketing Manager of Apple Europe from April 2006 to June 2010, where he was responsible for promoting Apple products
and defining marketing, PR and branding strategies within central Europe, the Middle East and Africa. Prior to Apple, Mr. Matine
worked extensively in marketing and advertising, promoting technology products and brands throughout Europe. Mr. Matine studied
at Sciences Po (the Paris Institute for Political Studies, Section Public Service) and holds the IFA-Sciences Po non-executive
director certificate.
We believe that Mr. Matine is qualified as to serve as a member of
the Board of Directors because of his experience with product marketing and go-to-market strategies.
Audrey Thevenon,
Ph.D.: Director.
Dr. Thevenon serves as a Program Officer on the Board of Life Sciences at the National Academies of Sciences,
Engineering and Medicine (“NASEM”), a private, nonprofit institution that provides high-quality, objective advice
on science, engineering, and health matters, since October 2016, and previously served as the Associate Program Officer of NASEM
from August 2014 to October 2016. Dr. Thevenon also serves as the Managing Editor of the journal Institute for Laboratory Animal
Research at NASEM. From February 2012 to July 2014, Dr. Thevenon was a Postdoctoral Fellow at the Uniformed Services University
of the Health Sciences in Bethesda, MA. Dr. Thevenon has also completed a Postdoctoral Fellowship at the University of Hawaii
in placental pharmacology. Dr. Thevenon has a Ph.D. and an MS both in Biology from Georgetown University, as well as an MS in
Cell Biology & Physiology and a BS in Life Sciences and Environment from the University of Rennes 1 in France. We believe
that Dr. Thevenon is qualified as to serve as a member of the Board of Directors because of her experience in medicine and scientific
innovation.
Leslie Markow:
Chief Financial Officer
. Ms. Markow has served as the Company’s Chief Financial Officer since September 2014. She
is a CPA CA in Canada, a US CPA (Illinois) and Chartered Director. From 2002 to 2004 and since 2010, Ms. Markow has provided outsourced
CFO, controller and financial services on a part-time basis to numerous public and private companies. In addition, in 2012-2013,
Ms. Markow was the Chief Financial Officer of Stewardship Ontario, a supply chain operator of Blue Box and Orange Drop Programs
for industry in the Province of Ontario. In 2010-2012, Ms. Markow was the Chief Financial Officer of Blue Ocean NutraSciences
Inc. (formerly Solutions4CO2 Inc.), a public CO2 solution industrial company. From 2004 to 2010, Ms. Markow was the Director of
Client Service for Resources Global Professionals, a NASDAQ-listed global consulting firm. From 1991-2002, she held various positions
at SunOpta Inc. a TSX-NASDAQ listed company, which at that time was named Stake Technology Ltd. and was an industrial technology
manufacturer, including as Chief Administrative Officer, Vice-President Regulatory Reporting & Compliance, Chief Financial
Officer and Vice-President–Finance and Controller. Ms. Markow started her career in 1983 with predecessors of PricewaterhouseCoopers,
ultimately holding a position as Senior Audit Manager and in 1991, she moved to SunOpta Inc. Ms. Markow is a member of the Board
of Directors and Chairperson of the Audit Committee of Jemtec Inc., a Canadian public company that sells monitoring hardware and
software. She also is a member of Financial Executives Canada, where she is a past National Board Director, Toronto Board Director,
Toronto Chapter President and the winner of the Toronto Leadership Award, and is a faculty member of The Directors College, which
is a joint venture of McMaster University and The Conference Board of Canada.
There are no family
relationships among any of our current or proposed officers and directors.
Involvement in Certain Legal Proceedings
To the best of our
knowledge, none of our directors or executive officers has been convicted in a criminal proceeding, excluding traffic violations
or similar misdemeanors, or has been a party to any judicial or administrative proceeding during the past ten years that resulted
in a judgment, decree, or final order enjoining the person from future violations of, or prohibiting activities subject to, federal
or state securities laws, or a finding of any violation of federal or state securities laws, except for matters that were dismissed
without sanction or settlement. Each of our executive officers and directors has informed us that he or she, as the case may be,
has not been involved in any of the events specified in clauses (1) through (8) of Regulation S-K, Item 401(f). Except as set
forth in our discussion below in “Certain Relationships and Related Transactions, and Director Independence – Transactions
with Related Persons,” none of our directors, director nominees, or executive officers has been involved in any transactions
with us or any of our directors, executive officers, affiliates, or associates that are required to be disclosed pursuant to the
rules and regulations of the Commission.
Term of Office
Directors are appointed
to hold office until the next annual general meeting of stockholders or until removed from office in accordance with our bylaws.
Our officers are appointed by our Board and hold office until removed by our Board.
All officers and directors
listed above will remain in office until the next annual meeting of our stockholders, and until their successors have been duly
elected and qualified. Our bylaws provide that officers are appointed annually by our Board and each executive officer serves
at the discretion of our Board.
Section 16(a) Beneficial Ownership
Reporting Compliance
Section 16(a) of the
Securities Exchange Act requires the Company’s officers and directors, and persons who beneficially own more than ten (10%)
percent of a class of equity securities registered pursuant to Section 12 of the Exchange Act, to file reports of ownership and
changes in ownership with the Securities and Exchange Commission and the principal exchange upon which such securities are traded
or quoted. Reporting Persons are also required to furnish copies of such reports filed pursuant to Section 16(a) of the Exchange
Act with the Company.
Based on our review
of the copies of such forms received by us, and to the best of our knowledge, all executive officers, directors and greater than
10% stockholders filed the required reports in a timely manner in the fiscal year ended March 31, 2019, except for Mr. Maloberti
who failed to timely file his Form 3, Mr. Gaston-Dryfus, who failed to timely file 2 Form 4’s showing 2 transactions and
Mr. Auberton-Herve, who failed to timely file a Form 4 showing 1 transaction.
Code of Business Conduct and Ethics
Policy
We adopted a Code
of Business Conduct and Ethics that applies to, among other persons, our principal executive officers, principal financial officer,
principal accounting officer or controller, and persons performing similar functions. Our Code of Business Conduct and Ethics
is available on our website
www.bioniklabs.com
.
Corporate Governance
The business and affairs
of the Company are managed under the direction of our Board of Directors which as of June 27, 2019 is comprised of Messrs.
Auberton-Herve
,
Dusseux, Gaston-Dreyfus, Martin, Malone, Matine, Prywata and Dr. Thevenon.
There have been no
changes in any state law or other procedures by which security holders may recommend nominees to our board of directors.
Committees of the Board of Directors
Presently,
the Board has two standing committees — the Audit Committee and the Compensation Committee. All members of the Audit Committee
and the Compensation Committee are required by the charters of the respective committees to be, independent.
Audit Committee
On
May 30, 2018, our Board formed an Audit Committee, of whom Messrs. Martin (Chairman) and Malone were initial members, with Charles
Matine joining the Audit Committee on September 7, 2018. Each member of the Audit Committee is independent, and the Board has
determined that Messrs. Martin, Malone and Matine are all independent and Mr. Martin is an “audit committee financial expert,”
as defined in SEC rules. The Audit Committee acts pursuant to a written charter which is available through our website at www.bioniklabs.com.
The
primary functions of the Audit Committee are to assist the Board in overseeing (i) the effectiveness of the Company’s accounting
and financial reporting processes and internal controls and the audits of the Company’s financial statements, (ii) the qualifications,
independence, appointment, retention, compensation and performance of the Company’s registered public accounting firm and
(iii) the performance of the Company’s internal audit department or department or person(s) having the equivalent responsibility
and functions.
Compensation Committee
On
May 30, 2018, our Board formed a Compensation Committee, of whom Messrs. Malone (Chairman) and Martin were initial members, with
Dr. Thevenon joining on September 7, 2018. Each of the members of the Compensation Committee is independent. The Compensation
Committee acts pursuant to a written charter which is available through our website at www.bioniklabs.com.
The primary functions
of the Compensation Committee are to (i) review and approve corporate goals and objectives relevant to executive compensation,
(ii) determine and review the CEO’s and other executive officers’ compensation, and (iii) make recommendations to
the Board concerning (a) compensation and (b) adoption of equity incentive plans.
Director Independence
We use the definition
of “independence” of The NASDAQ Stock Market to make this determination. NASDAQ Listing Rule 5605(a)(2) provides that
an “independent director” is a person other than an officer or employee of the company or any other individual having
a relationship, which, in the opinion of the Company’s Board, would interfere with the exercise of independent judgment
in carrying out the responsibilities of a director. The NASDAQ listing rules provide that a director cannot be considered independent
if:
|
|
·
|
The
director is, or at any time during the past three years was, an employee of the company;
|
|
|
·
|
The
director or a family member of the director accepted any compensation from the company
in excess of $120,000 during any period of 12 consecutive months within the three years
preceding the independence determination (subject to certain exclusions, including, among
other things, compensation for board or board committee service);
|
|
|
·
|
The
director or a family member of the director is, or at any time during the past three
years was, an executive officer of the company;
|
|
|
·
|
The
director or a family member of the director is a partner in, controlling stockholder
of, or an executive officer of an entity to which the company made, or from which the
company received, payments in the current or any of the past three fiscal years that
exceed 5% of the recipient’s consolidated gross revenue for that year or $200,000,
whichever is greater (subject to certain exclusions);
|
|
|
·
|
The
director or a family member of the director is employed as an executive officer of an
entity where, at any time during the past three years, any of the executive officers
of the company served on the compensation committee of such other entity; or
|
|
|
·
|
The
director or a family member of the director is a current partner of the company’s
outside auditor, or at any time during the past three years was a partner or employee
of the company’s outside auditor, and who worked on the company’s audit.
|
Under such definitions, Messrs. Martin,
Malone, Matine, Gaston-Dreyfus and Dr. Thevenon are considered independent directors.
ITEM 11 - EXECUTIVE COMPENSATION
Compensation of Executive
Officers
The following table
sets forth information regarding each element of compensation that was paid or awarded to our named executive officers for the
periods indicated.
Name and
Principal
Position
|
|
Year (1)
|
|
Salary
($)
|
|
|
Bonus
($)
|
|
|
Stock
Awards
($)
|
|
|
Option
Awards (2)
($)
|
|
|
Non-Equity
Incentive Plan
Compensation
($)
|
|
|
All Other
Compensation
($)
|
|
|
Total
($)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Eric Dusseux (3)
|
|
2019
|
|
|
381,158
|
|
|
|
225,564
|
|
|
|
–
|
|
|
|
363,714
|
|
|
|
–
|
|
|
|
61,333
|
|
|
|
1,031,769
|
|
|
Chief Executive Officer (CEO)
|
|
2018
|
|
|
229,987
|
|
|
|
136,719
|
|
|
|
–
|
|
|
|
983,602
|
|
|
|
–
|
|
|
|
12,547
|
|
|
|
1,362,855
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Michal Prywata
|
|
2019
|
|
|
210,000
|
|
|
|
12,600
|
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
10,836
|
|
|
|
233,436
|
|
|
Chief Technology Officer
|
|
2018
|
|
|
210,000
|
|
|
|
103,590
|
|
|
|
–
|
|
|
|
67,450
|
|
|
|
–
|
|
|
|
11,247
|
|
|
|
392,647
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Leslie Markow
|
|
2019
|
|
|
210,000
|
|
|
|
31,500
|
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
10,968
|
|
|
|
252,468
|
|
Chief Financial Officer
|
|
2018
|
|
|
210,000
|
|
|
|
116,550
|
|
|
|
–
|
|
|
|
40,470
|
|
|
|
–
|
|
|
|
11,068
|
|
|
|
378,088
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Renaud Maloberti (5)
Former Chief Commercial Officer
|
|
2019
|
|
|
239,215
|
|
|
|
28,025
|
|
|
|
–
|
|
|
|
30,341
|
|
|
|
–
|
|
|
|
559
|
|
|
|
298,140
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Timothy McCarthy (4)
|
|
2019
|
|
|
31,372
|
|
|
|
69,000
|
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
100,372
|
|
|
Former Chief Commercialization Officer
|
|
2018
|
|
|
260,000
|
|
|
|
97,500
|
|
|
|
–
|
|
|
|
691,106
|
|
|
|
–
|
|
|
|
-
|
|
|
|
1,048,606
|
|
|
|
(1)
|
“2019” represents the
fiscal year ended March 31, 2019 and “2018” represents the fiscal year ended
March 31, 2018.
|
|
|
(2)
|
For assumptions made in such valuation,
see Note 10 to the Company’s audited consolidated financial statements included
in this Annual Report on Form 10-K, commencing on page F-39.
|
|
|
(3)
|
On September 1, 2017, Mr. Dusseux
was hired as our Chief Executive Officer at an annual base salary of CDN $500,000.
|
|
|
(4)
|
On August 8, 2016, Mr. McCarthy was
hired as our Chief Commercialization Officer with a base salary of $260,000. Mr. McCarthy
left the Company on April 27, 2018.
|
|
|
(5)
|
On June 10, 2018, Mr. Renaud Maloberti
was hired as our Chief Commercialization Officer with a base salary of $295,000 and resigned
from all positions which the Company on May 13, 2019
|
Outstanding Equity Awards at Fiscal Year-End
The following table
presents the outstanding equity awards held by each of the named executive officers as of the end of the fiscal year ended March
31, 2019.
|
Option Awards
|
|
Name
|
|
Number of Securities
Underlying
Unexcercised Options
Excercisable
|
|
|
Number of Securities
Underlying
Unexcercised Options
Unexcercisable
|
|
|
Option Exercise Price
|
|
|
Option Expiration
Date
|
|
Eric Michel Dusseux
|
|
|
13,573
|
(1)
|
|
|
27,145
|
(1)
|
|
$
|
24.15
|
|
|
September 1, 2027
|
|
|
|
|
1,111
|
(2)
|
|
|
2,223
|
(2)
|
|
$
|
23.25
|
|
|
January 24, 2025
|
|
|
|
|
40,000
|
(3)
|
|
|
|
|
|
$
|
9.735
|
|
|
April 19, 2028
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Michal Prywata
|
|
|
6,606
|
(4)
|
|
|
-
|
|
|
$
|
34.50
|
|
|
July 1, 2021
|
|
|
|
|
2,667
|
(5)
|
|
|
-
|
|
|
$
|
150.00
|
|
|
December 14, 2022
|
|
|
|
|
1,111
|
(2)
|
|
|
2,223
|
(2)
|
|
$
|
23.25
|
|
|
January 24, 2025
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Leslie N. Markow
|
|
|
944
|
(6)
|
|
|
-
|
|
|
$
|
34.50
|
|
|
February 17, 2022
|
|
|
|
|
2,667
|
(7)
|
|
|
-
|
|
|
$
|
183.00
|
|
|
November 24, 2022
|
|
|
|
|
666
|
(2)
|
|
|
1,334
|
(2)
|
|
$
|
23.250
|
|
|
January 24, 2025
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Renaud Maloberti
|
|
|
|
|
|
|
5,000
|
(8)
|
|
$
|
6.93
|
|
|
August 8, 2023
|
|
|
(1)
|
On September 1, 2017, we issued 40,718
options to Mr. Dusseux at an exercise price of $24.15. 13,573 options have vested and
50% of the remaining options vest on performance being met and 50% vest annually over
5 years.
|
|
|
(2)
|
On January 24, 2018, the Company granted
3,334 options to Mr. Dusseux, 3,334 options to Mr. Prywata, 2,000 options to Ms. Markow
at $23.25 that vest equally on January 24, 2019, 2020 and 2021.
|
|
|
(3)
|
On April 19, 2018 we issued 40,000
options to Mr. Dusseux at an exercise price of $9.735. The options vested on the grant
date and expire in 10 years.
|
(4) On
July 1, 2014, Bionik Canada issued 6,606 options (adjusted for post-going public transaction) to Mr. Prywata at an exercise price
of $34.50 with a term of 7 years, which vested May 27, 2015. All of such options were issued subject to and contingent on the
successful consummation of the Offering and the going public transaction, which took place on February 26, 2015. Accordingly,
such options are deemed issued as of February 26, 2015.
|
|
(5)
|
On December 14, 2015, we issued 2,667
options to Mr. Prywata at an exercise price of $150.00 that vest equally over three years
on the anniversary date starting December 14, 2016. As of March 31, 2019, all options
are fully vested and expire December 14, 2022.
|
|
|
(6)
|
On February 17, 2015, we issued 944
options to Ms. Markow at an exercise price of $34.50, that vested one-third immediately
and two-thirds over the next two anniversary dates with an expiry date of seven years.
|
|
|
(7)
|
On November 24, 2015, we issued 2,667
options to Ms. Markow at an exercise price of $183.00, that vest equally over three years
on the anniversary date starting November 24, 2016. As of March 31, 2019, all options
are fully vested.
|
|
|
(8)
|
On June 10, 2018,
we issued 5,000 options to Mr. Maloberti at an exercise price of $6.93 that will vest
equally over three years on the anniversary date starting June 10, 2019. Mr. Maloberti
resigned from all positions with the Company on May 13, 2019 and all of these options
expired the same day.
|
On February 25, 2015,
1,752 common shares were issued to two former lenders connected with a $241,185 loan received and repaid in fiscal 2013. As part
of the consideration for the initial loan, Mr. Prywata and Mr. Caires, a former executive of the Company, collectively transferred
2,098 common shares to the lenders. For contributing the common shares to the lenders, the Company intends to reimburse them 2,134
common shares; however, these shares have not yet been issued.
Long-Term Incentive Plans and Awards
Since our incorporation
on January 8, 2010 through March 31, 2019 we did not have any long-term incentive plans that provided compensation intended to
serve as incentive for performance. No individual grants or agreements regarding future payouts under non-stock price-based plans
have been made to any executive officer or any director or any employee or consultant since our inception through March 31, 2019.
Director Compensation
The following table sets forth a summary
of the compensation we paid or accrued to our non-employee directors during the fiscal year ended March 31, 2019.
|
Name
|
|
Fees Earned
or Paid in
Cash
|
|
|
Stock
Awards
|
|
|
Option
Awards
|
|
|
Non-Equity
Incentive Plan
Compensation
|
|
|
Nonqualified
Deferred
Compensation
Earnings
|
|
|
All Other
Compensation
|
|
|
Total
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Andre Auberton-Herve
|
|
$
|
180,000
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
180,000
|
|
|
Marc Mathieu (1)
|
|
$
|
16,667
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
16,667
|
|
|
Remi Gaston Dreyfus
|
|
$
|
50,000
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
50,000
|
|
|
P. Gerald Malone
|
|
$
|
50,000
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
50,000
|
|
|
Joseph Martin
|
|
$
|
50,000
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
50,000
|
|
|
Charles Matine
|
|
$
|
32,527
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
32.527
|
|
|
Audrey Thevenon
|
|
$
|
32,527
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
32.527
|
|
|
|
(1)
|
Mr. Mathieu
resigned as a director of the Company on August 1, 2018.
|
Other than Mr. Auberton-Herve’s
annual fee as Chairman of $180,000, our non-employee directors are entitled to receive an annual cash payment of up to $50,000,
as well as reimbursement for expenses incurred by them in connection with attending board meetings. Our directors also are eligible
for stock option grants.
Employment Agreements
Eric Michel Dusseux
The Company entered
into an employment agreement with Dr. Dusseux on September 1, 2017, pursuant to which he serves as our Chief Executive Officer
(the “Dusseux Employment Agreement”). Under the Dusseux Employment Agreement, Dr. Dusseux will receive an initial
annual base salary of CDN$500,000. In addition, Dr. Dusseux may receive up to 50% of his base salary as a target bonus based on
measurable performance goals to be mutually agreed upon once employment starts on a pro-rata basis in the first fiscal year.
The Company also entered
into an Equity Compensation Agreement, dated September 1, 2017 (the “Dusseux Equity Compensation Agreement”), pursuant
to which the Company is required to grant Dr. Dusseux a stock option representing a right to acquire 6% of the aggregate amount
of the Company’s outstanding common stock and exchangeable shares as of the date of grant, which grant is required to be
made as soon as practicable following September 1, 2017. The exercise price of the option is $0.161, and the expiration date will
be the tenth anniversary of the date of grant. One-sixth of the option will be vested and exercisable as of its date of grant,
and the unvested portion of the option will become vested and exercisable as follows:
|
|
·
|
50%
in 5 equal annual installments on each of the five anniversaries of the date of the issuance
of the option; and
|
|
|
·
|
50%
in 5 equal separate tranches annually based on Dr. Dusseux’s achievement of annual
performance goals to be established by the Board in consultation with Dr. Dusseux. The
extent to which each separate tranche becomes vested shall be determined by reference
to Dr. Dusseux’s annual performance as measured by reference to the performance
targets set for that performance period. In the event a specific tranche is not fully
vested, that tranche shall not be forfeited, but shall remain outstanding, and may become
vested as a result of Dr. Dusseux’s future performance at an above target level
or as a result of accelerated vesting on the occurrence of any other event that triggers
accelerated vesting.
|
The option, including
any portion that is subject to vesting based on the period of Dr. Dusseux’s service and any portion that is subject to vesting
on the basis of performance, shall be fully vested on the occurrence of any of the following conditions: (a) A Change of Control
(as defined in the Company’s 2014 Equity Incentive Plan) or (b) Termination of Dr. Dusseux’s employment that constitutes
a “separation from service” (as the phrase is used for purpose of Section 409A of the Internal Revenue Code of 1986,
as amended), other than where such termination is for Cause (as defined in the Company’s 2014 Equity Incentive Plan) or
if Dr. Dusseux resigns other than for Good Reason (as defined in the Company’s 2014 Equity Incentive Plan).
Dr. Dusseux
is also entitled to receive a target annual cash bonus of up to 50% of base salary.
Dr. Dusseux
is entitled to reimbursement of housing costs of up to $4,000 per month for 24 months and the costs of immigration and annual
tax compliance and an annual executive medical provided by Medcan or similar supplier over the time he is employed.
In the event
that Dr. Dusseux employment is terminated as a result of death, Dr. Dusseux’s estate would be entitled to receive the annual
salary and a portion of the annual bonus earned up to the date of death. In addition, all vested options as of the date of death
would continue in full force and effect, subject to their terms and conditions of the Equity Incentive Plan.
In the event
that Dr. Dusseux’s employment is terminated as a result of disability, Dr. Dusseux would be entitled to receive the annual
salary, benefits, a portion of the annual bonus earned up to the date of disability and expenses incurred up to the date of termination.
In addition, all vested options as of the date of death would continue in full force and effect, subject to their terms and conditions
of the Equity Incentive Plan
In the event
that Dr. Dusseux’s employment is terminated by the Company for cause Dr. Dusseux would be entitled to receive his annual
salary, benefits and expenses incurred up to the date of termination.
In the event
that Dr. Dusseux’s employment is terminated by the Company without cause, he would be entitled to receive 12 months’
pay and benefit coverage plus one month for each year of service. Payment of pro-rata bonus for the fiscal year up to the date
of termination will also be paid.
The agreement
contains customary non-competition and non-solicitation provisions pursuant to which Dr. Dusseux agrees not to compete and solicit
with the Company. Dr. Dusseux also agreed to customary terms regarding confidentiality and ownership of intellectual property.
Michal Prywata
Bionik Canada
entered into an employment agreement with Michal Prywata on July 7, 2014, pursuant to which he serves as our Chief Operating Officer
on an indefinite basis, subject to the termination provisions described in the agreement. Pursuant to the terms of the agreement,
Mr. Prywata has received an annual base salary of $210,000 since February 26, 2015. The salary is reviewed on an annual basis
to determine potential increases based on Mr. Prywata’s performance and that of the Company. On June 29, 2017, the Company
changed his title to Chief Technology Officer.
Mr. Prywata
is also entitled to receive a target annual cash bonus of up to 30% of base salary. Mr. Prywata is further entitled to a cash
and option bonus based on a per patent creation basis, as determined by the Board of Directors.
In the event
Mr. Prywata’s employment is terminated as a result of death, Mr. Prywata’s estate would be entitled to receive the
annual salary and a portion of the annual bonus earned up to the date of death. In addition, all vested options and warrants as
of the date of death would continue in full force and effect, subject to their terms and conditions.
In the event
Mr. Prywata’s employment is terminated as a result of disability, Mr. Prywata would be entitled to receive the annual salary,
benefits, a portion of the annual bonus earned up to the date of disability and expenses incurred up to the date of termination.
In the event
Mr. Prywata’s employment is terminated by the Company for cause, Mr. Prywata would be entitled to receive his annual salary,
benefits and expenses incurred up to the date of termination.
In the event
Mr. Prywata’s employment is terminated by the Company without cause, he would be entitled to receive 12 months’ pay
and full benefits, plus one month for each year of service. Furthermore, Mr. Prywata will have six months after termination to
exercise all vested options in accordance with the terms of the 2014 Incentive Plan. All unvested options would immediately forfeit
upon such notice of termination.
The agreement
contains customary non-competition and non-solicitation provisions pursuant to which Mr. Prywata agrees not to compete and solicit
with the Company. Mr. Prywata also agreed to customary terms regarding confidentiality and ownership of intellectual property.
Leslie N. Markow
Bionik Canada
entered into an employment agreement with Leslie Markow on September 3, 2014, pursuant to which she serves as our Chief Financial
Officer on a part-time, indefinite basis, subject to the termination provisions described in the agreement. On September 16, 2015,
Ms. Markow was promoted to full time. Pursuant to the terms of the agreement, as amended, Ms. Markow receives an annual base salary
of $210,000 payable semi-monthly in arrears. The salary is reviewed on an annual basis to determine potential increases based
on Ms. Markow’s performance and that of the Company. Ms. Markow is also entitled to receive a target annual cash bonus of
up to 30% of base salary, and a grant of options in an amount to be determined at the price of the Company’s going public
transaction, upon the closing of the Company’s going public transaction, to vest over three years in equal annual installments.
In the event
Ms. Markow’s employment is terminated as a result of death, Ms. Markow’s estate would be entitled to receive the annual
salary and a portion of the annual bonus earned up to the date of death. In addition, all vested options and warrants as of the
date of death would continue in full force and effect, subject to the terms and conditions of the plan.
In the event
Ms. Markow’s employment is terminated as a result of disability, Ms. Markow would be entitled to receive the annual salary,
benefits, a portion of the annual bonus earned up to the date of disability and expenses incurred up to the date of termination.
In the event
Ms. Markow’s employment is terminated by the Company for cause, Ms. Markow would be entitled to receive her annual salary,
benefits and expenses incurred up to the date of termination.
In the event
Ms. Markow’s employment is terminated by us without cause, or she decides to leave the Company, she would be entitled to
receive six months, but no more than nine months’ pay and full benefits. Furthermore Ms. Markow will have six months after
termination to exercise all vested options in accordance with the terms of the plan. All unvested options would immediately forfeit
upon such notice of termination.
The agreement
contains customary non-competition and non-solicitation provisions pursuant to which Ms. Markow agrees not to compete and solicit
with the Company. Ms. Markow also agreed to customary terms regarding confidentiality and ownership of intellectual property.
Limits on Liability and Indemnification
We provide
directors and officers insurance for our current directors and officers.
Our certificate
of incorporation eliminates the personal liability of our directors to the fullest extent permitted by law. The certificate of
incorporation further provides that the Company will indemnify its officers and directors to the fullest extent permitted by law.
We believe that this indemnification covers at least negligence on the part of the indemnified parties. Insofar as indemnification
for liabilities under the Securities Act may be permitted to our directors, officers, and controlling persons under the foregoing
provisions or otherwise, we have been advised that in the opinion of the Securities and Exchange Commission such indemnification
is against public policy as expressed in the Securities Act of 1933 and is therefore unenforceable.
ITEM 12 - SECURITY
OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following
table shows the beneficial ownership of our Common Stock as of June 27, 2019 held by (i) each person known to us to be the beneficial
owner of more than five percent (5%) of our Common Stock; (ii) each director; (iii) each executive officer; and (iv) all directors
and executive officers as a group, as adjusted to reflect the one-for-one hundred fifty reverse stock split.
Beneficial
ownership is determined in accordance with the rules of the SEC, and generally includes voting power and/or investment power with
respect to the securities held. Shares of Common Stock subject to options and warrants currently exercisable or which may become
exercisable within 60 days of June 27, 2019 are deemed outstanding and beneficially owned by the person holding such options or
warrants for purposes of computing the number of shares and percentage beneficially owned by such person, but are not deemed outstanding
for purposes of computing the percentage beneficially owned by any other person. Except as indicated in the footnotes to this
table, the persons or entities named have sole voting and investment power with respect to all shares of our Common Stock shown
as beneficially owned by them.
The following
table provides for percentage ownership assuming 3,858,115 shares are issued outstanding as of June 27, 2019, consisting of 3,702,398
shares of Common Stock and 155,717 Common Stock equivalents through the Exchangeable Shares. The percentages below also assume
the exchange by all of the holders of Exchangeable Shares for an equal number of shares of our Common Stock in accordance with
the terms of the Exchangeable Shares. Unless otherwise indicated, the address of each beneficial holder of our Common Stock is
our corporate address.
|
Name of Beneficial Owner
|
|
Shares of Common Stock
Beneficially Owned
|
|
|
% of Shares of Common Stock
Beneficially Owned
|
|
|
Remi Gaston-Dreyfus (1)(2)
|
|
|
1,182,324
|
|
|
|
30.13
|
%
|
|
E.C.I SA (1)(3)
|
|
|
188,617
|
|
|
|
4.87
|
%
|
|
Solomar SA (1)(4)
|
|
|
153,211
|
|
|
|
3.96
|
%
|
|
Andre Auberton–Herve (5)
|
|
|
252,887
|
|
|
|
6.51
|
%
|
|
Olivier Dassault
|
|
|
543,277
|
|
|
|
14.08
|
%
|
|
SFP Capital
|
|
|
366,584
|
|
|
|
9.50
|
%
|
|
Eric Michel Dusseux (6)
|
|
|
91,635
|
|
|
|
2.32
|
%
|
|
Michal Prywata(1)(7)
|
|
|
60,360
|
|
|
|
1.56
|
%
|
|
Leslie N. Markow (6)
|
|
|
6,743
|
|
|
|
*
|
|
|
P. Gerald Malone (6)
|
|
|
1,666
|
|
|
|
*
|
|
|
Audrey Thevenon (6)
|
|
|
1,666
|
|
|
|
*
|
|
|
Charles Matine (6)
|
|
|
1,666
|
|
|
|
*
|
|
|
Joseph Martin (6)
|
|
|
1,666
|
|
|
|
*
|
|
|
|
|
|
|
|
|
|
|
|
|
All directors and executive officers as a group (9 persons)
|
|
|
1,600,613
|
|
|
|
39.35
|
%
|
|
|
(1)
|
Such shares include
Exchangeable Shares for tax purposes. The Exchangeable Shares have the following attributes,
among others:
|
|
|
·
|
Be, as
nearly as practicable, the economic equivalent of the Common Stock as of the consummation
of the Company’s going public transaction;
|
|
|
·
|
Have dividend
entitlements and other attributes corresponding to the Common Stock;
|
|
|
·
|
Be exchangeable,
at each holder’s option, for Common Stock; and
|
|
|
·
|
Upon the
direction of our Board of Directors, be exchanged for Common Stock on the 10-year anniversary
of the first closing of the Company’s 2015 offering, subject to applicable law,
unless exchanged earlier upon the occurrence of certain events.
|
The holders of the Exchangeable
Shares, through The Special Voting Preferred Stock, will have voting rights and other attributes corresponding to the Common Stock.
|
|
(2)
|
Includes options
to acquire 3,333 shares of Common Stock, (ii) an aggregate of 22,473 Exchangeable Shares
held through Lombard International Assurance SA and RGD Investissements and (iii) warrants
to purchase an aggregate of 61,465 shares of Common Stock held through Lombard International
Assurance SA and RGD Investissements.
|
|
|
(3)
|
Includes 9,321
Exchangeable Shares. Also includes warrants to purchase an aggregate of 11,524 shares
of Common Stock.
|
|
|
(4)
|
Includes 16,312
Exchangeable Shares. Also includes warrants to purchase an aggregate of 10,671 shares
of Common Stock
|
|
|
(5)
|
Includes (i) warrants
to purchase 10,671 shares of Common Stock held through Star SCI, (ii) an aggregate of
15,241 options to acquire Common Stock held through 4A Consulting and Engineering and
(iii) 2,500 options to acquire Common Stock held in his own name.
|
|
|
(6)
|
Represents options
to acquire shares of our Common Stock.
|
|
|
(7)
|
Includes 10,384
options to acquire shares of our Common Stock and 49,976 Exchangeable Shares.
|
ITEM 13
- CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS
Procedures and Policies
We consider
“related party transactions” to be transactions between our Company and (i) a director, officer, director nominee
or beneficial owner of greater than five percent of our stock; (ii) the spouse, parents, children, siblings or in-laws of any
person named in (i); or (iii) an entity in which one of our directors or officers is also a director or officer or has a material
financial interest.
Our Board
of Directors is vested with the responsibility of evaluating and approving any potential related party transaction, unless a special
committee consisting solely of independent directors is appointed by the Board of Directors. We do not have any formal policies
or procedures for related party transactions.
Transactions with Related
Parties
Since April 2017 through
June 27, 2019, entities controlled by Mr. Gaston-Dreyfus have made the following loans to the Company:
|
|
·
|
Between
August and December 2017, entities controlled by Mr. Gaston-Dreyfus loaned the Company
an aggregate of $2,580,000 evidenced by convertible promissory notes. Mr. Gaston-Dreyfus
received warrants as part of this financing.
|
|
|
·
|
On
December 19, 2017, an entity controlled by Mr. Gaston-Dreyfus loaned the Company $400,000
evidenced by a promissory note which was paid back January 4, 2018.
|
|
|
·
|
From
January 2018 through March 31, 2018, the Company borrowed an aggregate of $1,250,000
from an entity controlled by Mr. Gaston-Dreyfus, evidenced by convertible promissory
notes.
|
All convertible loans were exchanged
for common shares on March 31, 2018 and Mr. Gaston-Dreyfus and his affiliates received an aggregate of 608,028 shares of common
stock. As part of such transaction, 61,645 warrants were issued to affiliates of Mr. Gaston-Dreyfus.
|
|
·
|
From
April 2018 through June 25, 2018, the Company borrowed an aggregate of $1,991,673 from
an entity controlled by Mr. Gaston-Dreyfus, evidenced by convertible promissory notes.
Effective as of July 20, 2018, such convertible notes converted in accordance with their
terms into 289,791 shares of common stock.
|
|
|
·
|
On
January 22, 2019, the Company borrowed an aggregate of $750,000 from an affiliate of
Mr. Gaston-Dreyfus evidenced by a convertible promissory note, and such note and interest
was converted into common shares of the Company pursuant to the terms of such notes and
197,234 common shares were issued on March 28, 2019.
|
|
|
·
|
On
June 11, 2019, the Company borrowed $500,000 from an affiliate of Mr. Gaston-Dreyfus
evidenced by a convertible promissory note pursuant to an up to $9 million convertible
note offering.
|
In June 2018, the
Company borrowed an aggregate of $306,255 from an entity controlled by Mr. Andre Auberton–Herve, evidenced by a convertible
promissory note. Effective as of July 20, 2018, such convertible note converted in accordance with its terms into 44,590 shares
of common stock.
On October 10, 2018,
the Company borrowed an aggregate of $300,000 from an affiliate of Mr. Andre Auberton-Herve evidenced by a convertible promissory
note, and such note and interest was converted into common shares of the Company pursuant to the terms of such notes and 81,492
common shares were issued on March 28, 2019.
As of March 31, 2019,
we had aggregate advances repayable by Mr. Prywata of $18,585. The loan to Mr. Prywata bears interest at a prescribed rate of
1% until March 31, 2018 and 2% thereafter and is repayable on demand in Canadian dollars.
At March 31, 2019,
there was $229,473 owing to Eric Dusseux, $14,851 owing to Michal Prywata and $33,387 owing to Leslie Markow and $28,025 to Renaud
Malobert for sums paid by them on behalf of Bionik for business expense and bonus amounts. In addition, at March 31, 2019, the
Company owed nil (March 31, 2018- $587,019) as severance to its former CEO Peter Bloch, which is being paid over-time and was
fully repaid in February 2019.
In connection with
a CDN$250,000 loan obtained by Bionik Canada (which loan has been repaid), Bionik agreed to transfer pre-transaction 83,574 common
shares to the lenders. In addition, Messrs. Caires and Prywata also transferred 100,000 pre- transaction common shares to the
loan holder and this will be reimbursed by the issuance of 2,134 exchangeable shares (exchangeable to common shares) to Messrs.
Caires and Prywata. These shares have not yet been issued.
On May 8, 2019, the
Company borrowed $500,000 from an entity controlled by Mr. Auberton-Herve evidenced by a promissory note.
Other than the above
transactions, there have been no related party transactions, or any other transactions or relationships required to be disclosed
pursuant to Item 404 Regulation S-K. The Company is currently not a subsidiary of any company.
ITEM 14. PRINCIPAL
ACCOUNTING FEES AND SERVICES.
The Board of Directors
has reviewed and discussed the audited consolidated financial statements of Bionik Laboratories Corp. for the fiscal year ended
March 31, 2019, with management and have reviewed related written disclosures of MNP LLP, our independent accountants of the matters
required to be discussed by SAS 114 (Codification of Statements on Auditing Standards, AU Section 380), as amended, with respect
to those statements. We have reviewed the written disclosures and the letter from MNP LLP required by regulatory and professional
standards and have discussed with MNP LLP its independence in connection with its audit of our most recent financial statements.
Based on this review and these discussions, the Board of Directors recommends that the financial statements be included in this
Form 10-K for the year ended March 31, 2019.
We have also reviewed
the various fees that we paid or accrued to MNP LLP during the year ended March 31, 2019 and , 2018, for services they rendered
in connection with our annual audits and quarterly reviews, as well as for any other non-audit services they rendered.
The following table
shows the fees for professional services rendered by MNP LLP for the audit of our financial statements for the years ended March
31, 2019 and fees billed for other services rendered by MNP LLP during those periods:
|
Fee Category
|
|
2019
|
|
|
2018
|
|
|
Audit Fees
|
|
$
|
73,542
|
|
|
$
|
122,162
|
|
|
Audited related fees
|
|
|
-
|
|
|
|
-
|
|
|
Tax Fees
|
|
|
21,580
|
|
|
|
33,804
|
|
|
All Other Fees
|
|
|
211,900
|
|
|
|
26,606
|
|
|
Total Fees
|
|
$
|
307,022
|
|
|
$
|
182,572
|
|
Audit fees consist
of fees billed for professional services rendered for the audit of our financial statements and review of the interim financial
statements included in quarterly reports and services that are normally provided by the above auditors in connection with statutory
and regulatory fillings or engagements. Audit-related fees consist of fees billed for professional services rendered for the review
of SEC filings or other reports containing the audited financial statements. Tax fees consist of fees to prepare the Company’s
federal and state income tax returns. Other fees relate to advisory services related research on accounting or other regulatory
matters.
Pre-Approval Policies and Procedures
Our board of directors
is in the process of adopting a policy on pre-approval of audit and permissible non-audit services.
PART
IV
ITEM 15. EXHIBITS, FINANCIAL
STATEMENT SCHEDULES
Our financial statements as set
forth in the Index to Consolidated Financial Statements attached hereto commencing on page F-1 are hereby incorporated by reference.
The following
exhibits, which are numbered in accordance with Item 601 of Regulation S-K, are filed herewith or, as noted, incorporated by reference
herein.
|
2.3
|
|
Waiver
and Amendment Agreement, dated as of March 14, 2016, by and among Bionik Laboratories Corp., Hermano Igo Krebs, Bionik Mergerco
Inc. and Interactive Motion Technologies, Inc. (incorporated by reference to the Company’s Annual Report on Form 10-K
for the fiscal year ended December 31, 2015, filed on March 18, 2016)
|
|
3.1
|
|
Articles
of Conversion, dated June 25, 2013 (incorporated by reference to the Company’s 10-K filing on April 15, 2014)
|
|
3.2
|
|
Certificate
of Conversion, dated June 25, 2013 (incorporated by reference to the Company’s 10-K filing on April 15, 2014)
|
|
3.3
|
|
Certificate
of Incorporation, dated June 25, 2013 (incorporated by reference to the Company’s 10-K filing on April 15, 2014)
|
|
3.4
|
|
Delaware
By-laws, dated June 25, 2013 (incorporated by reference to the Company’s 10-K filing on April 15, 2014)
|
|
3.5
|
|
Amended
and Restated Certificate of Incorporation dated February 10, 2015 (incorporated by reference to the Company’s 8-K filing
on March 4, 2015)
|
|
3.6
|
|
Amended
and Restated By-Laws (incorporated by reference to the Company’s 8-K filing on March 4, 2015)
|
|
3.7
|
|
Certificate
of Amendment of the Certificate of Incorporation, dated November 8, 2017 (incorporated by reference to the Company’s
Current Report on Form 8-K filed on November 8, 2017).
|
|
3.8
|
|
Certificate
of Amendment of the Certificate of Incorporation, dated June 11, 2018 (incorporated by reference to the Company’s Current
Report on Form 8-K filed on June 13, 2018).
|
|
3.9
|
|
Certificate
of Amendment of the Certificate of Incorporation, dated October 26, 2018 (incorporated by reference to the Company’s
Current Report on Form 8-K filed on October 29, 2018).
|
|
4.1
|
|
Certificate
of Designation of Preferences, Rights and Limitations of Special Voting Preferred Stock of Bionik Laboratories Corp. (incorporated
by reference to the Company’s 8-K filing on March 4, 2015)
|
|
4.2
|
|
Schedule
A to Articles of Amendment of Bionik Laboratories Inc., relating to the Exchangeable Shares of Bionik Laboratories Inc. (incorporated
by reference to the Company’s 8-K filing on March 4, 2015)
|
|
4.3
|
|
Form
of Warrant (incorporated by reference to the Company’s 8-K filing on March 4, 2015)
|
|
4.4
|
|
Form
of Common Stock Purchase Warrant (incorporated by reference to the Company’s 8-K filing on March 4, 2015)
|
|
4.5
|
|
Form
of Warrant (incorporated by reference to the Company’s Annual Report on Form 10-K for the Fiscal Year ended March 31,
2017, filed with the Commission on June 29, 2017)
|
|
10.1
|
|
Investment
Agreement, dated February 26, 2015, among Bionik Laboratories Inc., Bionik Acquisition Inc. and Bionik Laboratories Corp.
(incorporated by reference to the Company’s 8-K filing on March 4, 2015)
|
|
10.2
|
|
Voting
and Exchange Trust Agreement, made as of February 26, 2015, among Bionik Laboratories Corp., Bionik Laboratories, Inc. and
Computershare Trust Company of Canada dated February 26, 2015 (incorporated by reference to the Company’s 8-K filing
on March 4, 2015)
|
|
10.3
|
|
Support
Agreement, made as of February 26, 2015, among Bionik Laboratories Inc., Bionik Acquisition Inc. and Bionik Laboratories Corp.
(incorporated by reference to the Company’s 8-K filing on March 4, 2015)
|
|
10.4
|
|
Registration
Rights Agreement, made as of February 26, 2015, by and between Bionik Laboratories Inc. and each of the several shareholders
signatory thereto (incorporated by reference to the Company’s 8-K filing on March 4, 2015)
|
|
10.5
|
|
Novation
Agreement, dated as of February 26, 2015, between Bionik Laboratories Corp. and Bionik Laboratories Inc. (incorporated by
reference to the Company’s 8-K filing on March 4, 2015)
|
|
10.6
|
|
Spin-Off
Agreement, dated as of February 26, 2015, by and among Bionik Laboratories Corp., and Brian E. Ray and Jon Lundgreen (incorporated
by reference to the Company’s 8-K filing on March 4, 2015)
|
|
10.7
|
|
Assignment
and Assumption Agreement, dated as of February 26, 2015, by and between Bionik Laboratories Corp. and Tungsten 74 LLC (incorporated
by reference to the Company’s 8-K filing on March 4,2015)
|
|
10.8
|
|
Form
of Subscription Agreement (incorporated by reference to the Company’s 8-K filing on March 4, 2015)
|
|
10.9
*
|
|
Peter
Bloch Employment Agreement (incorporated by reference to the Company’s 8-K filing on March 4, 2015)
|
|
10.10
*
|
|
Michal
Prywata Employment Agreement (incorporated by reference to the Company’s 8-K filing on March 4, 2015)
|
|
10.11
*
|
|
Leslie
Markow’s Employment Agreement (incorporated by reference to the Company’s 8-K filing on March 4, 2015)
|
|
10.12
*
|
|
Bionik
Laboratories Corp. f/k/a Drywave Technologies, Inc. 2014 Equity Incentive Plan (incorporated by reference to the Company’s
Definitive Information Statement on Schedule 14C filing on October 6, 2014)
|
|
10.13
|
|
Minutes
of Settlement (incorporated by reference to the Company’s Registration Statement on Form S-1 (Registration No.: 333-207581)
|
|
10.14
|
|
License
Agreement with The Massachusetts Institute of Technology, as amended (incorporated by reference to the Company’s Registration
Statement on Form S-1 (Registration No.: 333-207581)
|
|
10.15
|
|
Exclusive
Patent Application and Patent License Agreement between Interactive Motion Technologies, Inc., and Hermano Igo Krebs and Caitlyn
Joyce Bosecker (incorporated by reference to the Company’s Registration Statement on Form S-1 (Registration No.: 333-207581)
|
|
10.16
*
|
|
Employment
Agreement with Timothy McCarthy (incorporated by reference to the Registrant’s Current Report on Form 8- K filed on
August 8, 2016)
|
|
10.17
|
|
Registration
Rights Agreement dated April 21, 2016 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed
on April 26, 2016)
|
|
10.18
|
|
Allonge
#3 to Secured Promissory Note (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on March
2, 2017)
|
|
10.19
|
|
Engagement
Agreement dated May 3, 2017, by and between the Company and Garden State Securities Inc. (Incorporated by reference to Exhibit
(d)(1) to the Company’s Schedule TO filed on May 25, 2017)
|
|
10.20
|
|
Convertible
Promissory Note dated March 28, 2017 (incorporated by reference to the Company’s Annual Report on Form 10-K for the
Fiscal Year ended March 31, 2017, filed with the Commission on June 29, 2017)
|
|
10.21
|
|
Form
of Allonge to Promissory Notes dated as of March 28, 2017 (incorporated by reference to the Company’s Annual Report
on Form 10-K for the Fiscal Year ended March 31, 2017, filed with the Commission on June 29, 2017)
|
|
10.22
|
|
Cooperative
Joint Venture Contract dated May 23, 2017, by and between Ginger Capital Investment Holding Ltd. and Bionik Laboratories Corp.
(incorporated by reference to the Company’s Annual Report on Form 10-K for the Fiscal Year ended March 31, 2017, filed
with the Commission on June 29, 2017)
|
|
10.23
|
|
Convertible
Promissory Notes in the principal amount of $200,000 to Leizhang, as holder (incorporated by reference to the Company’s
Annual Report on Form 10-K for the Fiscal Year ended March 31, 2017, filed with the Commission on June 29, 2017)
|
|
10.24
|
|
Convertible
Promissory Notes in the principal amount of $150,000 to Bluestone International Capital LLC, as holder (incorporated by reference
to the Company’s Annual Report on Form 10-K for the Fiscal Year ended March 31, 2017, filed with the Commission on June
29, 2017)
|
|
10.25
|
|
Convertible
Promissory Notes in the principal amount of $150,000 to Ginger Capital, LLC, as holder (incorporated by reference to the Company’s
Annual Report on Form 10-K for the Fiscal Year ended March 31, 2017, filed with the Commission on June 29, 2017)
|
|
10.26
|
|
Demand
Notes in favor of Neville Hogan, in the aggregate principal amount of $50,000 (incorporated by reference to the Company’s
Annual Report on Form 10-K for the Fiscal Year ended March 31, 2017, filed with the Commission on June 29, 2017)
|
|
10.27
|
|
Amendments
to Demand Notes with Neville Hogan (incorporated by reference to the Company’s Annual Report on Form 10-K for the Fiscal
Year ended March 31, 2017, filed with the Commission on June 29, 2017)
|
|
10.28
|
|
Demand
Notes in favor of Hermano Igo Krebs, in the aggregate principal amount of $120,000 (incorporated by reference to the Company’s
Annual Report on Form 10-K for the Fiscal Year ended March 31, 2017, filed with the Commission on June 29, 2017)
|
|
10.29
|
|
Amendments
to Demand Notes with Hermano Igo Krebs (incorporated by reference to the Company’s Annual Report on Form 10-K for the
Fiscal Year ended March 31, 2017, filed with the Commission on June 29, 2017)
|
|
10.30
|
|
Demand
Notes in favor of Rodolfo Rohr, in the aggregate principal amount of $130,000 (incorporated by reference to the Company’s
Annual Report on Form 10-K for the Fiscal Year ended March 31, 2017, filed with the Commission on June 29, 2017)
|
|
10.31
|
|
Amendments
to Demand Notes with Rodolfo Rohr (incorporated by reference to the Company’s Annual Report on Form 10-K for the Fiscal
Year ended March 31, 2017, filed with the Commission on June 29, 2017)
|
|
10.32
|
|
License
Agreement by and between Bionik Laboratories Corp. and China Bionik Medical Rehabilitation Technology Ltd. dated May 17, 2017
(incorporated by reference to the Company’s Annual Report on Form 10-K for the Fiscal Year ended March 31, 2017, filed
with the Commission on June 29, 2017)
|
|
10.33
|
|
Distribution
Agreement by and between Bionik Laboratories Corp. and China Bionik Medical Rehabilitation Technology Ltd. dated May 17, 2017
(incorporated by reference to the Company’s Annual Report on Form 10-K for the Fiscal Year ended March 31, 2017, filed
with the Commission on June 29, 2017)
|
|
10.34
|
|
Joint
Development and Manufacturing Agreement by and between Bionik Laboratories Corp. and Wistron Medical Tech Holding Company
(incorporated by reference to the Company’s Annual Report on Form 10-K for the Fiscal Year ended March 31, 2017, filed
with the Commission on June 29, 2017)
|
|
10.35
*
|
|
First
Amendment to Tim McCarthy Employment Agreement (incorporated by reference to the Company’s Current Report on Form 8-K
filed on August 9, 2017)
|
|
10.36
*
|
|
Equity
Compensation Agreement between the Company and 4A Consulting and Engineering (incorporated by reference to the Company’s
Current Report on Form 8-K filed on September 11, 2017)
|
|
10.37
|
|
Form
of Convertible Promissory Note in the principal amount of up to $2,000,000 (incorporated by reference to the Company’s
Current Report on Form 8-K filed on September 11, 2017)
|
|
10.38
*
|
|
Peter
Bloch Separation Agreement (incorporated by reference to the Company’s Current Report on Form 8-K filed on September
11, 2017)
|
|
10.39
*
|
|
Eric
Dusseux Employment Agreement (incorporated by reference to the Company’s Current Report on Form 8-K filed on September
11, 2017)
|
|
10.40
*
|
|
Equity
Compensation Agreement between the Company and Eric Dusseux (incorporated by reference to the Company’s Current Report
on Form 8-K filed on September 11, 2017)
|
|
10.41
|
|
Form
of Subscription Agreement for the sale of up to $2,000,000 in Convertible Promissory Notes (incorporated by reference to the
Company’s Current Report on Form 8-K filed on September 20, 2017)
|
|
10.42
|
|
Form
of Convertible Promissory Note (incorporated by reference to the Company’s Current Report on Form 8-K filed on September
20, 2017)
|
|
10.43
|
|
Form
of Common Stock Purchase Warrant (incorporated by reference to the Company’s Current Report on Form 8-K filed on September
20, 2017)
|
|
10.44
|
|
Allonge
#1 to Convertible Promissory Note (incorporated by reference to the Company’s Current Report on Form 8-K filed on September
20, 2017)
|
|
10.45
|
|
Form
of Allonge #2 to Convertible Promissory Notes (incorporated by reference to the Company’s Current Report on Form 8-K
filed on September 20, 2017)
|
|
10.46
|
|
Form
of Allonge to Common Stock Purchase Warrant (incorporated by reference to the Company’s Current Report on Form 8-K filed
on September 20, 2017)
|
|
10.47
|
|
Allonge
to Demand Note (incorporated by reference to the Company’s Current Report on Form 8-K filed on December 14, 2017)
|
|
10.48
|
|
Allonge
to Demand Note (incorporated by reference to the Company’s Current Report on Form 8-K filed on December 14, 2017)
|
|
10.49
|
|
Amendment
No. 1 to Convertible Promissory Notes (Incorporated by reference to the Company’s Current Report on Form 8-K filed on
February 5, 2018)
|
|
10.50
|
|
Promissory
Note, dated February 2, 2018 (Incorporated by reference to the Company’s Current Report on Form 8-K filed on February
5, 2018)
|
|
10.51
|
|
Form
of Subscription (Incorporated by reference to the Company’s Quarterly Report for the fiscal quarter ended December 31,
2017, filed on February 13, 2018)
|
|
10.52
|
|
Form
of Convertible Promissory Note (Incorporated by reference to the Company’s Quarterly Report for the fiscal quarter ended
December 31, 2017, filed on February 13, 2018)
|
|
10.53
**
|
|
Distribution
Agreement (Incorporated by reference to the Company’s Current Report on Form 8-K, filed on March 7, 2018)
|
|
10.54
*
|
|
Amended
Separation Agreement, effective as of March 13, 2018, by and between the Company and Peter Bloch (Incorporated by reference
to the Company’s Current Report on Form 8-K filed on March 14, 2018)
|
|
10.55
|
|
Exchange
Agreement, dated as of March 12, 2018 (Incorporated by reference to the Company’s Current Report on Form 8-K filed on
March 14, 2018)
|
|
10.56
|
|
Promissory
Note, dated March 14, 2018 (Incorporated by reference to the Company’s Current Report on Form 8-K filed on March 14,
2018)
|
|
10.57
|
|
Allonge
to Convertible Promissory Notes (Incorporated by reference to the Company’s Current Report on Form 8-K filed on April
3, 2018)
|
|
10.58
|
|
Allonge
to Common Stock Purchase Warrants (Incorporated by reference to the Company’s Current Report on Form 8-K filed on April
3, 2018)
|
|
10.59
|
|
Exchange
Agreement, dated March 30, 2018 (Incorporated by reference to the Company’s Current Report on Form 8-K filed on April
3, 2018)
|
|
10.60
|
|
Promissory
Note, dated as of April 12, 2018 (Incorporated by reference to the Company’s Current Report on Form 8-K filed on April
18, 2018)
|
|
10.61
|
|
Promissory
Note, dated as of May 24, 2018 (Incorporated by reference to the Company’s Current Report on Form 8-K filed on May 31,
2018)
|
|
10.62
|
|
Promissory
Note, dated as of April 26, 2018 (Incorporated by reference to the Company’s Annual Report on Form 10-K, filed on June
27, 2018)
|
|
10.63
|
|
Promissory
Note, dated as of May 10, 2018 (Incorporated by reference to the Company’s Annual Report on Form 10-K, filed on June
27, 2018)
|
|
10.64
*
|
|
Employment
Agreement with Renaud Maloberti (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on June
11, 2018)
|
|
10.65
|
|
Promissory
Note, dated as of June 12, 2018 (Incorporated by reference to the Company’s Annual Report on Form 10-K, filed on June
27, 2018)
|
|
10.66
|
|
Promissory
Note, dated as of June 22, 2018 (Incorporated by reference to the Company’s Annual Report on Form 10-K, filed on June
27, 2018)
|
|
10.67
|
|
Form
of Stock Option Agreement (Incorporated by reference to the Company’s Annual Report on Form 10-K, filed on June 27,
2018)
|
|
10.68
|
|
Form
of Subscription Agreement (Incorporated by reference to the Company’s Current Report on Form 8-K, filed on July 5, 2018)
|
|
10.69
|
|
Form
of Convertible Promissory Note (Incorporated by reference to the Company’s Current Report on Form 8-K, filed on July
5, 2018)
|
|
10.70
|
|
Exchange
Agreement, dated as of June 28, 2018 (Incorporated by reference to the Company’s Current Report on Form 8-K, filed on
July 5, 2018)
|
|
10.71
|
|
Form
of Subscription Agreement (Incorporated by reference to the Company’s Current Report on Form 8-K, filed on October 12,
2018)
|
|
10.72
|
|
Form
of Convertible Promissory Note (Incorporated by reference to the Company’s Current Report on Form 8-K, filed on October
12, 2018)
|
|
10.73
|
|
Sale
of Goods Agreement, dated as of December 13, 2018, by and between Bionik Inc. and CHC Management Services, LLC (Incorporated
by reference to the Company’s Current Report on Form 8-K, filed on December 17, 2018)
|
|
10.74
|
|
Form
of Allonge to Short-Term Convertible Promissory Notes (Incorporated by reference to the Company’s Current Report
on Form 8-K, filed on February 14, 2019)
|
|
10.75
|
|
Form
of Allonge to Bridge Notes (Incorporated by reference to the Company’s Current Report on Form 8-K, filed on February
14, 2019)
|
|
10.76
|
|
Promissory
Note, dated as of May 8, 2019 (Incorporated by reference to the Company’s Current Report on Form 8-K, filed on May 10,
2019)
|
|
10.77
|
|
Form
of Subscription Agreement (Incorporated by reference to the Company’s Current Report on Form 8-K, filed on June 17,
2019)
|
|
10.78
|
|
Form
of Promissory Note (Incorporated by reference to the Company’s Current Report on Form 8-K, filed on June 17, 2019)
|
|
14.1
|
|
Code
of Business Conduct and Ethics (Incorporated by reference to the Company’s Annual Report on Form 10-K for the fiscal
year ended December 31, 2014)
|
* Management contract or compensatory
plan or arrangement.
** Portions of this document have been
omitted and submitted separately with the Securities and Exchange Commission pursuant to a request for “Confidential Treatment”.
SIGNATURES
Pursuant to
the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to
be signed on its behalf by the undersigned, thereunto duly authorized.
|
|
Bionik Laboratories Corp.
|
|
|
|
|
|
|
By:
|
/s/ Eric Dusseux
|
|
|
|
Eric Dusseux
|
|
|
|
Chief Executive Officer
|
Dated: July 1, 2019
Pursuant to the requirements
of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant
and in the capacities and on the dates indicated.
|
Signature
|
|
Title
|
|
Date
|
|
|
|
|
|
|
|
/s/ Eric Dusseux
|
|
Chief Executive Officer and Director
|
|
July 1, 2019
|
|
Eric Dusseux
|
|
(Principal Executive Officer)
|
|
|
|
|
|
|
|
|
|
/s/ Leslie Markow
|
|
Chief Financial Officer
|
|
July 1, 2019
|
|
Leslie Markow
|
|
(Principal Financial and Accounting Officer)
|
|
|
|
|
|
|
|
|
|
/s/
Michal Prywata
|
|
Chief Technology Officer and Director
|
|
July 1, 2019
|
|
Michal Prywata
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Chairman and Director
|
|
|
|
Andre Auberton
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Remi Gaston Dreyfus
|
|
Director
|
|
July 1, 2019
|
|
Remi Gaston Dreyfus
|
|
|
|
|
|
|
|
|
|
|
|
/s/
P. Gerald Malone
|
|
Director
|
|
July 1, 2019
|
|
P. Gerald Malone
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Joseph Martin
|
|
Director
|
|
July 1, 2019
|
|
Joseph Martin
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Charles Matine
|
|
Director
|
|
July 1, 2019
|
|
Charles Matine
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Audrey Thevenon
|
|
Director
|
|
July 1, 2019
|
|
Audrey Thevenon
|
|
|
|
|
|
|
|
|
|
|
BIONIK LABORATORIES
CORP.
CONSOLIDATED FINANCIAL
STATEMENTS
March 31, 2019
and 2018
(Amounts expressed
in US Dollars)
Index
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Shareholders of Bionik Laboratories
Corp.
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated
balance sheets of Bionik Laboratories Corp. (the Company) as of March 31, 2019 and 2018, and the related consolidated statements
of operations and comprehensive loss, shareholders’ equity, and cash flows for each of the years in the two-year period
ended March 31, 2019, and the related notes comprising a summary of significant accounting policies and other explanatory information
(collectively referred to as the consolidated financial statements).
In our opinion, the consolidated financial
statements present fairly, in all material respects, the consolidated financial position of the Company as of March 31, 2019 and
2018, and the results of its consolidated operations and its consolidated cash flows for each of the years in the two-year period
ended March 31, 2019, in conformity with accounting principles generally accepted in the United States of America.
Material Uncertainty Related to Going Concern
The accompanying consolidated financial
statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the consolidated
financial statements, the Company’s accumulated deficit, recurring losses and negative cash flows from operations raise
substantial doubt about its ability to continue as a going concern. Management's plans in regard to these matters are also described
in Note 1. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements
are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated
financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight
Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal
securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance
with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about
whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is
not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our
audits, we are required to obtain an understanding of internal control over financial reporting, but not for the purpose of expressing
an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such
opinion.
Our audits included performing procedures
to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing
procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and
disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant
estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe
that our audits provide a reasonable basis for our opinion.

|
|
Chartered
Professional Accountants
|
|
|
Licensed
Public Accountants
|
We have served as the Company’s auditor since 2015
Toronto, Ontario
June 25, 2019
Bionik Laboratories Corp.
Consolidated Balance Sheets
(Amounts expressed in US Dollars)
|
|
|
As at
|
|
|
As at
|
|
|
|
|
March 31,
|
|
|
March 31,
|
|
|
|
|
2019
|
|
|
2018
|
|
|
|
|
$
|
|
|
$
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
Current
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
|
446,779
|
|
|
|
507,311
|
|
|
Accounts receivable, net of allowance for doubtful accounts of $Nil (March 31, 2018 - $19,694)
|
|
|
1,523,193
|
|
|
|
212,730
|
|
|
Prepaid expenses and other receivables (Note 5)
|
|
|
1,355,032
|
|
|
|
433,655
|
|
|
Inventories (Note 6)
|
|
|
405,682
|
|
|
|
237,443
|
|
|
Due from related parties (Note 9(a))
|
|
|
18,585
|
|
|
|
18,897
|
|
|
Total Current Assets
|
|
|
3,749,271
|
|
|
|
1,410,036
|
|
|
Equipment (Note 7)
|
|
|
192,528
|
|
|
|
159,961
|
|
|
Technology and other assets (Note 4)
|
|
|
4,427,722
|
|
|
|
4,706,719
|
|
|
Goodwill (Note 3)
|
|
|
22,308,275
|
|
|
|
22,308,275
|
|
|
Total Assets
|
|
|
30,677,796
|
|
|
|
28,584,991
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and Shareholders' Equity
|
|
|
|
|
|
|
|
|
|
Current
|
|
|
|
|
|
|
|
|
|
Accounts Payable (Notes 9(b))
|
|
|
1,148,852
|
|
|
|
724,673
|
|
|
Accrued liabilities (Notes 8 and 9(b))
|
|
|
1,653,233
|
|
|
|
1,530,305
|
|
|
Demand Loans (Note 8)
|
|
|
-
|
|
|
|
51,479
|
|
|
Deferred revenue - Contract Liabilities
|
|
|
467,778
|
|
|
|
122,667
|
|
|
Shares to be issued, stock options and warrants (Note 10)
|
|
|
-
|
|
|
|
5,692,853
|
|
|
Total Current Liabilities
|
|
|
3,269,863
|
|
|
|
8,121,977
|
|
|
Shareholders' Equity
|
|
|
|
|
|
|
|
|
|
Preferred Stock, par value $0.001; Authorized 10,000,000 Special Voting Preferred Stock, par
value
|
|
|
|
|
|
|
|
|
|
$0.001; Authorized; Issued and outstanding - 1 (March 31, 2018 – 1)
|
|
|
-
|
|
|
|
-
|
|
|
Common Shares, par value $0.001; Authorized - 500,000,000 (March 31, 2018 – 250,000,000);
Issued and outstanding 3,661,838 and 196,799 Exchangeable Shares (March 31, 2018 – 1,368,856 and 295,146 Exchangeable
Shares)
|
|
|
3,858
|
|
|
|
1,664
|
|
|
Additional paid in capital
|
|
|
73,719,299
|
|
|
|
56,195,541
|
|
|
Deficit
|
|
|
(46,357,373
|
)
|
|
|
(35,776,340
|
)
|
|
Accumulated other comprehensive income
|
|
|
42,149
|
|
|
|
42,149
|
|
|
Total Shareholders' Equity
|
|
|
27,407,933
|
|
|
|
20,463,014
|
|
|
Total Liabilities and Shareholders' Equity
|
|
|
30,677,796
|
|
|
|
28,584,991
|
|
|
Commitments and Contingencies (Note 15)
|
|
Subsequent Events (Note 16)
|
|
The accompanying notes are an integral part of these consolidated financial statements.
|
|
The Financial Statements have been updated to reflect the 150 to 1 reverse stock
split on October 29, 2018, Note 2(a)
|
Bionik Laboratories Corp.
Consolidated Statements of Operations and Comprehensive
Loss for the years ended March 31, 2019 and 2018
(Amounts expressed in U.S. Dollars)
|
|
|
March 31, 2019
|
|
|
March 31, 2018
|
|
|
|
|
$
|
|
|
$
|
|
|
Sales
|
|
|
3,246,038
|
|
|
|
987,431
|
|
|
Cost of Sales
|
|
|
1,630,166
|
|
|
|
402,665
|
|
|
Gross Margin
|
|
|
1,615,872
|
|
|
|
584,766
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating expenses
|
|
|
|
|
|
|
|
|
|
Sales and marketing
|
|
|
2,339,359
|
|
|
|
1,989,837
|
|
|
Research and development
|
|
|
3,174,892
|
|
|
|
2,825,200
|
|
|
General and administrative
|
|
|
3,893,393
|
|
|
|
3,585,484
|
|
|
Share-based compensation expense (Notes 11)
|
|
|
1,347,399
|
|
|
|
1,540,580
|
|
|
Amortization (Note 4)
|
|
|
278,997
|
|
|
|
323,905
|
|
|
Depreciation (Note 7)
|
|
|
69,212
|
|
|
|
89,026
|
|
|
Total operating expenses
|
|
|
11,103,252
|
|
|
|
10,354,032
|
|
|
|
|
|
|
|
|
|
|
|
|
Other (income) expenses
|
|
|
|
|
|
|
|
|
|
Accretion expense (Note 8)
|
|
|
3,266,918
|
|
|
|
1,937,308
|
|
|
Share premium
|
|
|
-
|
|
|
|
1,249,994
|
|
|
Fair Value Adjustment (Note 8)
|
|
|
(337,923
|
)
|
|
|
-
|
|
|
Gain/Loss on mark to market re-evaluation (Note 10(c))
|
|
|
(2,048,697
|
)
|
|
|
376,674
|
|
|
Other expense
|
|
|
262,596
|
|
|
|
1,297,205
|
|
|
Other income
|
|
|
(73,166
|
)
|
|
|
(107,656
|
)
|
|
Foreign exchange
|
|
|
(507
|
)
|
|
|
102,999
|
|
|
Total other expenses
|
|
|
1,069,221
|
|
|
|
4,856,524
|
|
|
Net loss and comprehensive loss for the period
|
|
|
(10,556,601
|
)
|
|
|
(14,625,790
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Loss per share - basic and diluted
|
|
|
(4.47
|
)
|
|
|
(21.73
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of shares outstanding – basic and diluted
|
|
|
2,363,107
|
|
|
|
673,203
|
|
The accompanying notes are an integral part of these consolidated
financial statements
The Financial Statements have been updated to reflect the 150
to 1 reverse stock split on October 29, 2018, Note 2(a)
Bionik Laboratories Corp.
Consolidated Statements of Changes in Shareholders' Equity
for the years ended March 31, 2019 and March 31, 2018
(Amounts expressed in U.S. Dollars)
|
|
|
Special Voting
|
|
|
Common Shares
|
|
|
Additional
Paid
|
|
|
|
|
|
Comprehensive
|
|
|
|
|
|
|
|
Shares
|
|
|
Amount
|
|
|
Shares
|
|
|
Amount
|
|
|
in Capital
|
|
|
Deficit
|
|
|
Income
|
|
|
Total
|
|
|
|
|
|
|
|
$
|
|
|
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
Balance, March 31, 2017
|
|
|
1
|
|
|
|
-
|
|
|
|
645,297
|
|
|
|
645
|
|
|
|
45,184,320
|
|
|
|
(21,076,464
|
)
|
|
|
42,149
|
|
|
|
24,150,650
|
|
|
Warrant exercised
|
|
|
-
|
|
|
|
-
|
|
|
|
33,335
|
|
|
|
33
|
|
|
|
1,125,005
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,125,038
|
|
|
Share compensation expense
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,540,580
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,540,580
|
|
|
Shares to be issued for services
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Fair value of warrants on convertible loans
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
548,179
|
|
|
|
-
|
|
|
|
-
|
|
|
|
548,179
|
|
|
Loss on warrant down round feature (Note 12)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
74,086
|
|
|
|
(74,086
|
)
|
|
|
-
|
|
|
|
-
|
|
|
Conversion of European Promissory notes - 1st tranche
|
|
|
-
|
|
|
|
-
|
|
|
|
326,563
|
|
|
|
327
|
|
|
|
3,059,977
|
|
|
|
-
|
|
|
|
-
|
|
|
|
3,060,304
|
|
|
Conversion of European Promissory notes - 2nd tranche
|
|
|
-
|
|
|
|
-
|
|
|
|
452,898
|
|
|
|
453
|
|
|
|
4,243,767
|
|
|
|
-
|
|
|
|
-
|
|
|
|
4,244,220
|
|
|
Conversion of European Promissory notes - 3rd tranche
|
|
|
-
|
|
|
|
-
|
|
|
|
143,280
|
|
|
|
143
|
|
|
|
1,342,562
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,342,705
|
|
|
Conversion of Promissory notes
|
|
|
-
|
|
|
|
-
|
|
|
|
62,629
|
|
|
|
63
|
|
|
|
533,493
|
|
|
|
-
|
|
|
|
-
|
|
|
|
533,556
|
|
|
Stock option and warrant reclassification (Notes 11 & 12)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(2,845,557
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
(2,845,557
|
)
|
|
Beneficial Conversion Feature on convertible debt
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,389,129
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,389,129
|
|
|
Net loss for the year
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(14,625,790
|
)
|
|
|
-
|
|
|
|
(14,625,790
|
)
|
|
Balance, March 31, 2018
|
|
|
1
|
|
|
|
-
|
|
|
|
1,664,002
|
|
|
|
1,664
|
|
|
|
56,195,541
|
|
|
|
(35,776,340
|
)
|
|
|
42,149
|
|
|
|
20,463,014
|
|
|
Conversion of Promissory notes (Note 10(b))
|
|
|
-
|
|
|
|
-
|
|
|
|
263,639
|
|
|
|
264
|
|
|
|
2,470,358
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,470,622
|
|
|
Conversion of Promissory notes - July 20, 2018 (Note 8(d5))
|
|
|
-
|
|
|
|
-
|
|
|
|
683,395
|
|
|
|
683
|
|
|
|
4,732,170
|
|
|
|
-
|
|
|
|
-
|
|
|
|
4,732,853
|
|
|
Conversion of Promissory notes - March 28, 2019 (Note 8 (d6))
|
|
|
-
|
|
|
|
-
|
|
|
|
1,247,099
|
|
|
|
1,247
|
|
|
|
6,009,370
|
|
|
|
-
|
|
|
|
-
|
|
|
|
6,010,617
|
|
|
Stock option and warrant reclassification (Notes 11 and 12)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,173,534
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,173,534
|
|
|
Share compensation expense (Note 11)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,347,399
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,347,399
|
|
|
Fair value of Anti-dilution feature
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,766,495
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,766,495
|
|
|
Loss on warrant down round feature (Note 12)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
24,432
|
|
|
|
(24,432
|
)
|
|
|
-
|
|
|
|
-
|
|
|
Net loss for the year
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(10,556,601
|
)
|
|
|
-
|
|
|
|
(10,556,601
|
)
|
|
Adjustment due to 1:150 share consolidation round-up
|
|
|
-
|
|
|
|
-
|
|
|
|
502
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Balance, March 31, 2019
|
|
|
1
|
|
|
|
-
|
|
|
|
3,858,637
|
|
|
|
3,858
|
|
|
|
73,719,299
|
|
|
|
(46,357,373
|
)
|
|
|
42,149
|
|
|
|
27,407,933
|
|
The accompanying notes are an integral part of these consolidated
financial statements
The Financial Statements have been updated to reflect the 150
to 1 reverse stock split on October 29, 2018, Note 2(a)
Bionik Laboratories Corp.
Consolidated Statements of Cash Flows
For the years ended March 31, 2019 and 2018
(Amounts expressed in U.S. Dollars)
|
|
|
March 31, 2019
|
|
|
March 31, 2018
|
|
|
|
|
$
|
|
|
$
|
|
|
Operating activities
|
|
|
|
|
|
|
|
|
|
Net loss for the year
|
|
|
(10,556,601
|
)
|
|
|
(14,625,790
|
)
|
|
Adjustment for items not affecting cash
|
|
|
|
|
|
|
|
|
|
Depreciation
|
|
|
69,212
|
|
|
|
89,026
|
|
|
Amortization
|
|
|
278,997
|
|
|
|
323,905
|
|
|
Interest expense
|
|
|
255,833
|
|
|
|
1,294,005
|
|
|
Share based compensation expense
|
|
|
1,347,399
|
|
|
|
1,540,580
|
|
|
Shares premium
|
|
|
-
|
|
|
|
1,249,994
|
|
|
Accretion expense
|
|
|
3,266,918
|
|
|
|
1,937,308
|
|
|
Fair Value Adjustment
|
|
|
(337,923
|
)
|
|
|
-
|
|
|
Gain/Loss on mark to market re-evaluation
|
|
|
(2,048,697
|
)
|
|
|
376,674
|
|
|
Allowance for doubtful accounts
|
|
|
(19,694
|
)
|
|
|
(19,694
|
)
|
|
|
|
|
(7,744,556
|
)
|
|
|
(7,833,992
|
)
|
|
Changes in non-cash working capital items
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(1,290,769
|
)
|
|
|
190,867
|
|
|
Prepaid expenses and other receivables
|
|
|
(921,377
|
)
|
|
|
(205,608
|
)
|
|
Due from related parties
|
|
|
312
|
|
|
|
(166
|
)
|
|
Inventories
|
|
|
(168,239
|
)
|
|
|
(9,194
|
)
|
|
Accounts payable
|
|
|
424,179
|
|
|
|
(60,098
|
)
|
|
Accrued liabilities
|
|
|
122,928
|
|
|
|
304,048
|
|
|
Customer advances
|
|
|
-
|
|
|
|
(120,762
|
)
|
|
Deferred revenue
|
|
|
345,111
|
|
|
|
24,043
|
|
|
Net cash (used in) operating activities
|
|
|
(9,232,411
|
)
|
|
|
(7,710,862
|
)
|
|
Investing activities
|
|
|
|
|
|
|
|
|
|
Acquisition of equipment
|
|
|
(101,779
|
)
|
|
|
(21,567
|
)
|
|
Net cash (used in) investing activities
|
|
|
(101,779
|
)
|
|
|
(21,567
|
)
|
|
Financing activities
|
|
|
|
|
|
|
|
|
|
Proceeds on exercise of warrants
|
|
|
-
|
|
|
|
1,125,038
|
|
|
Proceeds from convertible loans
|
|
|
9,326,633
|
|
|
|
7,111,375
|
|
|
Repayment of Promissory notes principal
|
|
|
-
|
|
|
|
(200,000
|
)
|
|
Repayment of Promissory notes interest
|
|
|
-
|
|
|
|
(49,505
|
)
|
|
Repayment of Demand notes principal
|
|
|
(50,000
|
)
|
|
|
(208,359
|
)
|
|
Repayment of Demand notes interest
|
|
|
(2,975
|
)
|
|
|
(79,259
|
)
|
|
Proceeds from short-term loan
|
|
|
-
|
|
|
|
400,000
|
|
|
Repayment of short-term loan
|
|
|
-
|
|
|
|
(400,000
|
)
|
|
Repayment of short-term loan interest
|
|
|
-
|
|
|
|
(3,200
|
)
|
|
Net cash provided by financing activities
|
|
|
9,273,658
|
|
|
|
7,696,090
|
|
|
Net (decrease) in cash and cash equivalents for the year
|
|
|
(60,532
|
)
|
|
|
(36,339
|
)
|
|
Cash and cash equivalents, beginning of the year
|
|
|
507,311
|
|
|
|
543,650
|
|
|
Cash and cash equivalents, end of the year
|
|
|
446,779
|
|
|
|
507,311
|
|
The accompanying notes are an integral part of these consolidated
financial statements.
The Financial Statements have been updated to reflect the 150
to 1 reverse stock split on October 29, 2018, Note 2(a)
BIONIK LABORATORIES
CORP.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
For the years ended March 31, 2019 and
2018
(Amounts expressed in U.S. Dollars)
|
|
1.
|
NATURE
OF OPERATIONS AND GOING CONCERN
|
The Company and its Operations
Bionik Laboratories Corp., (the “Company”
or “Bionik”) was incorporated on January 8, 2010 in the State of Colorado as Strategic Dental Management Corp. On
July 16, 2013, the Company changed its name to Drywave Technologies Inc. (“Drywave”) and its state of incorporation
from Colorado to Delaware. Effective February 13, 2015, the Company changed its name to Bionik Laboratories Corp. and reduced
the authorized number of shares of common stock from 200,000,000 to 150,000,000. Concurrently, the Company implemented a 1-for-0.831105
reverse stock split of the common stock, which had previously been approved on September 24, 2014. On October 29, 2018, the Company
implemented at 1 for 150 reverse stock split of the common and exchangeable shares.
On February 26, 2015, the Company entered
into a Share Exchange Agreement and related transactions whereby it acquired Bionik Laboratories Inc., a Canadian Corporation
(“Bionik Canada”) and Bionik Canada issued 333,334 Exchangeable Shares, representing a 3.14 exchange ratio, for 100%
of the then outstanding common shares of Bionik Canada (the “Merger”). The Exchangeable Shares are exchangeable at
the option of the holder, each into one share of the common stock of the Company. In addition, the Company issued one Special
Preferred Voting Share (the “Special Preferred Share”) (Note 10).
On April 21, 2016, the Company acquired
all of the outstanding shares and, accordingly, all assets and liabilities of Interactive Motion Technologies, Inc. (IMT), a Boston,
Massachusetts-based global pioneer and leader in providing effective robotic products for neurorehabilitation, pursuant to an
Agreement and Plan of Merger (the “Merger Agreement”) dated March 1, 2016, with IMT, Hermano Igo Krebs, and Bionik
Mergerco Inc., a Massachusetts corporation and the Company’s wholly owned subsidiary (Bionik Mergerco). The merger agreement
provided for the merger of Bionik Mergerco with and into IMT, with IMT surviving the merger as the Company’s wholly owned
subsidiary. In return for acquiring IMT, IMT shareholders received an aggregate of 157,667 shares of the Company’s common
stock (Note 4).
On November 6, 2017, the Company approved
the authorization of a common share capital share increase to 250,000,000 from 150,000,000 and on June 12, 2018, the Company approved
the authorization of a common share increase to 500,000,000 from 250,000,000.
References to the Company refer to the
Company and its wholly owned subsidiaries, Bionik Inc., Bionik Acquisition Inc. and Bionik Canada.
The Company is a global pioneering robotics
company focused on providing rehabilitation solutions to individuals with neurological disorders, specializing in designing, developing
and commercializing cost-effective physical rehabilitation technologies, prosthetics, and assisted robotic products. The Company
strives to innovate and build devices that can rehabilitate and improve an individual’s health, comfort, accessibility and
quality of life through the use of advanced algorithms and sensing technologies that anticipate a user’s every move.
These consolidated financial statements
have been prepared in accordance with accounting principles generally accepted in the United States of America (“US GAAP”),
which contemplates continuation of the Company as a going concern, which assumes the realization of assets and the satisfaction
of liabilities and commitments in the normal course of business.
The Company’s principal offices
are located at 483 Bay Street, N105, Toronto, Ontario, Canada M5G 2C9 and its U.S. address is 80 Coolidge Hill Road, Watertown,
MA 02472.
Going Concern
As at March 31, 2019, the Company had
a working capital surplus of $479,408 (working capital deficit as at March 31, 2018, of $(6,711,941)) and an accumulated deficit
of $46,357,373 (March 31, 2018 - $35,776,340) and the Company incurred a net loss and comprehensive loss of $10,556,601 for the
year ended March 31, 2019 (March 31, 2018 - $(14,625,790)).
There is no certainty that the Company
will be successful in generating sufficient cash flow from operations or achieving and maintaining profitable operations in the
future to enable it to meet its obligations as they come due and consequently continue as a
BIONIK LABORATORIES CORP.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
For the years ended March 31, 2019 and
2018
(Amounts expressed in U.S. Dollars)
|
|
1.
|
NATURE
OF OPERATIONS AND GOING CONCERN – Continued
|
going concern. The Company will require
additional financing this year to fund its operations and it is currently working on securing this funding through corporate collaborations,
public or private equity offerings or debt financings. Sales of additional equity securities by the Company would result in the
dilution of the interests of existing stockholders. There can be no assurance that financing will be available when required.
In the event that the necessary additional financing is not obtained, the Company would reduce its discretionary overhead costs
substantially or otherwise curtail operations. The Company expects the forgoing, or a combination thereof, to meet the Company’s
anticipated cash requirements for the next 12 months; however, these conditions raise substantial doubt about the Company’s
ability to continue as a going concern. The accompanying consolidated financial statements do not include any adjustments to reflect
the possible future effects on recoverability and reclassification of assets or the amounts and classification of liabilities
that may result from the outcome of this uncertainty.
All adjustments, consisting only of normal
recurring items, considered necessary for fair presentation have been included in these consolidated financial statements.
During the fiscal year, holders of the
common stock and exchangeable shares of the Company approved, through a majority shareholder vote, an amendment to the Company’s
Amended and Restated Certificate of Incorporation authorizing the Board of Directors to effect a reverse stock split of Bionik’s
common stock and exchangeable shares at a ratio up to one-to-one hundred and fifty.
On October 29, 2018, the Company effected
a reverse stock split and thereafter Bionik’s common stock began trading on the OTCQB market on a one-for-one hundred and
fifty (1:150) split-adjusted basis. All owners of record on October 29, 2018 received one issued and outstanding share of Bionik
common stock or exchangeable share in exchange for one hundred and fifty issued and outstanding shares of Bionik common stock
or Bionik exchangeable stock. No fractional shares were issued in connection with the reverse stock split. All fractional shares
created by the one-for-one hundred and fifty reverse split were rounded up to the next whole share. The reverse stock split had
no impact on the par value per share of Bionik common stock, which remains at $0.001. All current and prior period amounts related
to shares, share prices and earnings per share, presented in the Company’s consolidated financial statements and the accompanying
Notes have been restated to give retrospective presentation for the reverse stock split.
|
|
3.
|
SIGNIFICANT
ACCOUNTING POLICIES
|
Newly Adopted and Recently Issued Accounting
Pronouncements
Newly Adopted
In May 2014, the Financial Accounting
Standards Board (“FASB”) issued Accounting Standards Update (“ASU”) No. 2014-09, Revenue from Contracts
with Customers (Topic 606). The updated standard will replace most existing revenue recognition guidance in U.S. GAAP. The new
standard introduces a five-step process to be followed in determining the amount and timing of revenue recognition. It also provides
guidance on accounting for costs incurred to obtain or fulfill contracts with customers and establishes disclosure requirements
which are more extensive than those required under existing U.S. GAAP. The FASB has issued numerous amendments to ASU 2014-09
from August 2015 through January 2018, which provide supplemental and clarifying guidance, as well as amend the effective date
of the new standard. ASU 2014-09, as amended, is effective for the Company in the interim period ended June 30, 2018. The standard
permits the use of either the retrospective or modified retrospective (cumulative effect) transition method. The Company adopted
the new standard using the modified retrospective transition method. The Company has adopted ASU-2014-1 for the fiscal year ended
March 31, 2019 and it did not have a material effect on the consolidated financial position and the consolidated results of operations.
BIONIK LABORATORIES
CORP.
NOTES TO CONSOLIDATED
FINANCIAL STATEMENTS
For the years
ended March 31, 2019 and 2018
(Amounts expressed
in U.S. Dollars)
|
|
3.
|
SIGNIFICANT
ACCOUNTING POLICIES – Continued
|
In November 2015, the FASB issued ASU
No. 2015-17, “Balance Sheet Classification of Deferred Taxes,” which require that deferred tax liabilities and assets
be classified on our Consolidated Balance Sheets as noncurrent based on an analysis of each taxpaying component within a jurisdiction.
ASU No. 2015-17 is effective for the fiscal year commencing after December 15, 2017. The Company has adopted ASU-2015-17 for the
fiscal year ended March 31, 2019 and it did not have a material effect on the consolidated financial position or the consolidated
results of operations.
In January 2016, the FASB issued ASU No.
2016-01 Financial Instruments - Overall (Subtopic 825-10): Recognition and Measurement of Financial Assets and Financial Liabilities.
The updates make several modifications to Subtopic 825-10, including the elimination of the available-for-sale classification
of equity investments, and it requires equity investments with readily determinable fair values to be measured at fair value with
changes in fair value recognized in operations. The update is effective for fiscal years beginning after December 2017. The Company
has adopted ASU 2016-01 for the year ended March 31, 2019 and it did not have a material effect on the consolidated financial
position and the consolidated results of operations.
In August 2016, the FASB issued ASU 2016-15,
“Statement of Cash Flows: Classification of Certain Cash Receipts and Cash Payments”. This ASU provides eight targeted
changes to how cash receipts and cash payments are presented and classified in the statement of cash flows. ASU 2016-15 is effective
for the fiscal year commencing after December 15, 2017. The Company has adopted ASU 2016-15 for the fiscal year ended March 31,
2019 and it did not have material effect on the consolidated financial position or on the consolidated statement of cash flows.
In May 2017, the FASB issued ASU No. 2017-09,
Compensation - Stock Compensation (Topic 718): Scope of Modification Accounting (ASU 2017-09). The FASB issued the update to provide
clarity and reduce the cost and complexity when applying the guidance in Topic 718. The amendments in this update provide guidance
about which changes to the terms or conditions of a share-based payment award require an entity to apply modification accounting
in Topic 718. The Company adopted ASU 2017-09 during the year ended March 31, 2019 and it did not have a material effect on the
consolidated financial statements and the consolidated results of operations.
Recently Issued
In January 2017, the FASB issued ASU 2017-01,
“Business Combinations: Clarifying the definition of a Business” which amends the current definition of a business.
Under ASU 2017-01, to be considered a business, an acquisition would have to include an input and a substantive process that together
significantly contributes to the ability to create outputs. ASU 2017-01 further states that when substantially all of the fair
value of gross assets acquitted is concentrated in a single asset (or a group of similar assets), the assets acquired would not
represent a business. The new guidance also narrows the definition of the term “outputs” to be consistent with how
it is described in Topic 606, Revenue from Contracts with Customers. The changes to the definition of a business will likely result
in more acquisitions being accounted for as asset acquisitions. ASU 2017-01 is effective for acquisitions commencing on or after
June 30, 2019, with early adoption permitted. Adoption of this guidance will be applied prospectively on or after the effective
date and the Company does not expect this policy will have a material effect on the consolidated financial position or consolidated
statement of cash flows.
In January 2017, the FASB issued ASU 2017-04,
“Intangibles – Goodwill and Other” ASU 2017-04 simplifies the accounting for goodwill impairment by eliminating
Step 2 of the current goodwill impairment test, which required a hypothetical purchase price allocation. Goodwill impairment will
now be the amount by which the reporting unit’s carrying value exceeds its fair value, limited to the carrying value of
the goodwill. ASU 2017-04 is effective for financial statements issued for fiscal years, and interim periods beginning after December
15, 2019. The Company is still assessing the impact that the adoption of ASU 2017-04 will have on the consolidated statement of
financial position and consolidated statement of operations.
BIONIK LABORATORIES
CORP.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
For the years ended March 31, 2019 and
2018
(Amounts expressed in U.S. Dollars)
|
|
3.
|
SIGNIFICANT
ACCOUNTING POLICIES – Continued
|
In June 2016, the FASB issued ASU 2016-13
Financial Instruments – Credit Losses (Topic 326): Measurement of Credit Losses of Financial Instruments, which introduces
an expected credit loss methodology for the impairment of financial assets measured at amortized cost basis. The methodology replaces
the probable, incurred loss model for those assets. The update if effective for fiscal years beginning after December 15, 2019.
The Company is still assessing the impact that the adoption of ASU 2016-13 will have on the consolidated statement of financial
position and consolidated statement of operations.
In February 2016, the FASB issued ASU
2016-02, Leases. This update requires organizations that lease assets to recognize on the balance sheet the assets and liabilities
for the rights and obligations created by those leases. The new guidance will also require additional disclosure about the amount,
timing and uncertainty of cash flows arising from leases. The provisions of this update are effective for annual and interim periods
beginning after December 15, 2018. The Company is still assessing the impact that the adoption of ASU 2016-02 will have on the
consolidated statement of financial position and consolidated statement of operations.
Inventory
Inventory is stated at the lower of cost
or net realizable value. Cost is recorded at actual cost, on the first-in first-out basis. The Company only has finished goods
inventory recoded based on actual cost from outsourced manufacturing partner.
Revenue Recognition
The Company has adopted ASU-2014-9 with initial application
date of April 1, 2018. The Company adopted the new standard using the modified retrospective transition method. The updated accounting
policies and the impact on the consolidated audited financial statements and additional disclosures are as follows:
The Company determines revenue through
the following steps: a) identification of the contract with the customer; b) identification of the performance obligations in
the contract; c) determination of the transaction price; d) allocation of the transaction price for the performance obligations
in the contract; and e) recognition of revenue when or as the Company satisfies a performance obligation. Revenue is recognized
when control of a product is transferred to a customer. Revenue is measured based on the consideration specified in the contract
with the customer, net of returns and discounts. Accruals for sales returns are calculated based on the best estimate of the amount
of product that will ultimately be returned by customers, reflecting historical experience and the magnitude of non-conforming
inventory claims made by customer that have either been approved or are pending review. Contract liabilities are recorded when
cash payments are received or due in advance of the Company’s performance. The Company defers revenue from extended warranty
sales and recognizes them over the period of extended warranty and from training services when the training is provided.
In the comparative period, the revenue
was measured at the fair value of the consideration received or receivable, net of returns and discounts and was recognized when
the risks and rewards of ownership has transferred to the customer. No revenue was recognized if there were significant uncertainties
regarding recovery of the consideration due, the costs incurred or to be incurred could not be measured reliably, or there was
continuing management involvement with the goods.
Allowance for doubtful accounts
The Company extends
unsecured credit to its customers in the ordinary course of business but mitigates the associated credit risk by supplying products
to customers with pre-approved capital expenditure budgets or rental credit, and by actively pursuing past due accounts. An allowance
for doubtful accounts is estimated and recorded based on management’s assessment of the credit history with the customer
and the current relationships with them. On this basis management has determined that an allowance for doubtful accounts of $Nil
and $19,694 was appropriate as of March 31, 2019 and 2018, respectively.
BIONIK LABORATORIES CORP.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
For the years ended March 31, 2019 and
2018
(Amounts expressed in U.S. Dollars)
|
|
3.
|
SIGNIFICANT
ACCOUNTING POLICIES – Continued
|
Warranty Reserve and Deferred Warranty
Revenue
The Company provides a one-year warranty
as part of its normal sales offering. When products are sold, the Company provides warranty reserves, which, based on the historical
experience of the Company are sufficient to cover warranty claims. Accrued warranty reserves are included in accrued liabilities
on the consolidated balance sheets and amounted to $143,500 at March 31, 2019 (March 31, 2018 - $64,957). The Company also sells
extended warranties for additional periods beyond the standard warranty. Extended warranty revenue is deferred and recognized
as revenue over the extended warranty period. The Company recognized $84,038 of expenses related to warranty expenses and recorded
this expense in cost of goods sold for the year ended March 31, 2019 (March 31, 2018 - $Nil).
Foreign Currency Translation
The functional and presentation currency
of the Company and its wholly owned subsidiaries is the U.S. dollar. Transactions denominated in a currency other than the functional
currency are recorded on the initial recognition at the exchange rate at the date of the transaction. After initial recognition
monetary assets and liabilities denominated in foreign currency are translated at the end of each reporting period into the functional
currency at the exchange rate at that date. Exchange differences are recognized in profit and loss. Non- monetary assets and liabilities
measured at cost are translated at the exchange rate at the date of the transaction.
Equipment
Equipment is recorded at cost. Depreciation
is computed using the declining balance method, over the estimated useful lives of these assets. The costs of improvements that
extend the life of equipment are capitalized. All ordinary repair and maintenance costs are expensed as incurred. Equipment is
depreciated as follows:
|
Computer and Electronics
|
50% per annum
|
|
Furniture and Fixtures
|
20% per annum
|
|
Demonstration Equipment
|
50% per annum
|
|
Manufacturing Equipment
|
20% per annum
|
|
Tools and Parts
|
20% per annum
|
|
Assets under capital lease
|
Life of lease (60 months)
|
Use of Estimates
The preparation of the consolidated financial
statements in conformity with US GAAP requires management to make estimates and assumptions that affect the reported amounts of
assets and liabilities and the disclosure of contingent assets and liabilities at the date of the consolidated financial statements
and the reported amounts of revenues and expenses during the reporting periods. The estimates based on management’s best
knowledge of current events and actions of the Company may undertake in the future. Significant areas requiring the use of estimates
relate to the valuation of inventory, the useful life of equipment and intangible assets, impairment of goodwill and intangible
assets, share based compensation, warranty accruals, accretion, fair value adjustment and fair value determination of warrants.
Actual results could differ from these estimates.
Fair Value of Financial Instruments
ASC Topic 820 defines fair value, establishes
a framework for measuring fair value, and expands disclosures about fair value measurements. Included in the ASC Topic 820 framework
is a three level valuation inputs hierarchy with Level 1 being inputs and transactions that can be effectively fully observed
by market participants spanning to Level 3 where estimates are unobservable by market participants outside of the Company and
must be estimated using assumptions developed by the Company. The Company discloses the lowest level input significant to each
category of asset or liability valued within the scope of ASC Topic 820 and the valuation method as exchange, income or use. The
Company uses inputs, which are as observable as possible, and the methods most applicable to the specific situation of each company
or valued item.
BIONIK LABORATORIES
CORP.
NOTES TO CONSOLIDATED
FINANCIAL STATEMENTS
For the years
ended March 31, 2019 and 2018
(Amounts expressed
in U.S. Dollars)
|
|
3.
|
SIGNIFICANT
ACCOUNTING POLICIES – Continued
|
The carrying amounts reported in the balance
sheets for cash and cash equivalents, accounts receivable, other receivables, accounts payable, accrued liabilities, due from
related parties, demand loans, convertible loans and promissory note payable approximate fair value because of the short period
of time between the origination of such instruments, their expected realization and their current market rates of interest. Per
ASC Topic 820 framework these are considered Level 2 inputs where inputs other than Level 1 that are observable, either directly
or indirectly, such as quoted prices in active markets for similar assets or liabilities, quoted prices for identical or similar
assets or liabilities in markets that are not active, or other inputs that are observable or can be corroborated by observable
market data for substantially the full term of the assets or liabilities.
The Company’s policy is to recognize
transfers into and out of Level 3 as of the date of the event or change in the circumstances that caused the transfer. There were
no such transfers during the year.
Segment Reporting
ASC 280-10, “Disclosures about Segments
of an Enterprise and Related Information”, establishes standards for the way that public business enterprises report information
about operating segments in the Company’s consolidated financial statements. Operating segment are components of an enterprise
about which separate financial information is available that is evaluated regularly by the chief operating decision maker in deciding
how to allocate resources and in assessing performance.
Approximately 99% of the Company’s
assets are US-based and all sales for the years ended March 31, 2019 and 2018 were made by the Company’s US subsidiary,
Bionik, Inc. In addition, all of the Company’s technology and other assets and goodwill are connected to the acquisition
by the Company in April 2016 of Bionik, Inc. Equipment connected to Bionik Inc. amounts to $148,618 (March 31, 2018 -$120,910)
and $43,910 (March 31, 2018 - $39,051) is connected to equipment at the Company’s Canadian subsidiary Bionik Laboratories
Inc.
Cash and Cash Equivalents
Cash and cash equivalents include highly
liquid investments with original terms to maturity of 90 days or less at the date of purchase. For all periods presented cash
and cash equivalents consisted entirely of cash on deposit with Canadian and US banks.
Research and Development
The Company is engaged in research and
development work. Research and development costs are charged to operating expenses of the Company as incurred.
Share based compensation
At grant date share-based compensation
is valued using the Black-Scholes option pricing model based on key assumptions determined by the Company. The value is recognized
based on the straight- line method during the vesting period or based on the fulfillment of predetermined milestones in case of
performance-based vesting.
Income Taxes
Income taxes are computed in accordance
with the provisions of ASC Topic 740, which requires, among other things, a liability approach to calculating deferred income
taxes. The Company recognizes deferred tax liabilities and assets for the expected future tax consequences of events that have
been recognized in its consolidated financial statements or tax returns. Under this method, deferred tax liabilities and assets
are determined based on the difference between the financial statement carrying amounts and tax bases of assets and liabilities
using enacted tax rates in effect in the years in which the differences are expected to reverse. The Company is required to make
certain estimates and judgments about the application of tax law, the expected resolution of uncertain tax positions and other
matters. In the event that uncertain tax positions are resolved for amounts different than the Company’s estimates, or the
related statutes of limitations expire without the assessment of additional income taxes, the Company will be required to adjust
the amounts of related assets and liabilities in the period in which such events occur. Such adjustment may have a material impact
on the Company’s income tax provision and results of operations.
BIONIK LABORATORIES
CORP
.
NOTES TO CONSOLIDATED
FINANCIAL STATEMENTS
For the years
ended March 31, 2019 and 2018
(Amounts expressed
in U.S. Dollars)
|
|
3.
|
SIGNIFICANT
ACCOUNTING POLICIES – Continued
|
Basic and Diluted Loss Per Share
Basic and diluted loss per share has been
determined by dividing the net loss available to shareholders for the applicable period by the basic and diluted weighted average
number of shares outstanding, respectively. The diluted weighted average number of shares outstanding is calculated as if all
dilutive options had been exercised or vested at the later of the beginning of the reporting period or date of grant, using the
treasury stock method.
Loss per common share is computed by dividing
the net loss by the weighted average number of shares of common shares outstanding during the period. Common share equivalents,
options and warrants were excluded from the computation of diluted loss per share because their effect was anti-dilutive.
Impairment of Long-Lived Assets
The Company follows the ASC Topic 360,
which requires that long-lived assets be reviewed for impairment whenever events or changes in circumstances indicate that the
assets’ carrying amounts may not be recoverable. In performing the review for recoverability, if future undiscounted cash
flows (excluding interest charges) from the use and ultimate disposition of the assets are less than their carrying values, an
impairment loss represented by the difference between its fair value and carrying value, is recognized. When properties are classified
as held for sale, they are recorded at the lower of the carrying amount or the expected sales price less costs to sell.
Goodwill and Indefinite Lived Intangible Assets
The Company records goodwill when the
purchase price of an acquisition exceeds the fair value of the net tangible and identified intangible assets acquired. Goodwill
and indefinite lived intangible assets, consisting of the trademarks acquired (Note 4), are assessed for impairment annually,
or more frequently if indicators of potential impairment exist, which includes evaluating qualitative and quantitative factors
to assess the likelihood of an impairment of goodwill or indefinite lived intangible assets. The qualitative factors used in the
analysis include microeconomic conditions, industry and market conditions, cost factors, overall financial performance and other
relevant entity specific events. The Company performs impairment tests using a fair value approach when necessary. None of the
Company’s goodwill or indefinite lived intangibles was impaired as of March 31, 2019. Accordingly, no impairment loss has
been recognized in the year ended March 31, 2019.
|
|
4.
|
TECHNOLOGY
AND OTHER ASSETS
|
The schedule below reflects
the intangible assets acquired in the IMT acquisition and the assets amortization period and expense for the year ended March
31, 2019:
|
|
|
Amortization
period (years)
|
|
Value
acquired
|
|
|
Expense
March 31,
2018
|
|
|
Value at
March 31,
2018
|
|
|
Expenses
March 31, 2019
|
|
|
Value at
March 31, 2019
|
|
|
Intangible assets acquired
|
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
Patents and exclusive Licence Agreement
|
|
9.74
|
|
|
1,306,031
|
|
|
|
134,126
|
|
|
|
1,045,530
|
|
|
|
134,090
|
|
|
|
911,440
|
|
|
Trademark
|
|
Indefinite
|
|
|
2,505,907
|
|
|
|
-
|
|
|
|
2,505,907
|
|
|
|
-
|
|
|
|
2,505,907
|
|
|
Customer relationships
|
|
10
|
|
|
1,431,680
|
|
|
|
143,206
|
|
|
|
1,153,543
|
|
|
|
143,168
|
|
|
|
1,010,375
|
|
|
Non compete agreement
|
|
2
|
|
|
61,366
|
|
|
|
30,709
|
|
|
|
1,739
|
|
|
|
1,739
|
|
|
|
-
|
|
|
Assembled workforce
|
|
1
|
|
|
275,720
|
|
|
|
15,864
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
|
|
5,580,704
|
|
|
|
323,905
|
|
|
|
4,706,719
|
|
|
|
278,997
|
|
|
|
4,427,722
|
|
The aggregate amortization expense for
the technology and other assets was $1,152,982 and $873,985 at March 31, 2019 and 2018 respectively.
BIONIK LABORATORIES CORP.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
For the years ended March 31, 2019 and
2018
(Amounts expressed in U.S. Dollars)
|
|
5.
|
PREPAID
EXPENSES AND OTHER RECEIVABLES
|
|
|
|
March 31,
2019
|
|
|
March 31,
2018
|
|
|
|
|
$
|
|
|
$
|
|
|
Prepaid expenses and other receivables
|
|
|
92,170
|
|
|
|
86,957
|
|
|
Prepaid inventory
|
|
|
1,144,392
|
|
|
|
301,104
|
|
|
Prepaid insurance
|
|
|
66,320
|
|
|
|
36,497
|
|
|
Sales taxes receivable (i)
|
|
|
52,150
|
|
|
|
9,097
|
|
|
|
|
|
1,355,032
|
|
|
|
433,655
|
|
i) Sales tax receivable represents net
harmonized sales taxes (HST) input tax credits receivable from the Government of Canada.
|
|
|
March
31,
2019
|
|
|
March
31,
2018
|
|
|
|
|
$
|
|
|
$
|
|
|
Raw Materials
|
|
|
-
|
|
|
|
237,443
|
|
|
Finished Goods
|
|
|
405,682
|
|
|
|
-
|
|
|
|
|
|
405,682
|
|
|
|
237,443
|
|
For the year ended March 31, 2019, $62,589
(March 31, 2018 - $38,860) of inventory has been written off to Cost of Sales as it is not expected to be used as a result of
an introduction of new versions of existing InMotion™ products.
Equipment consisted of the following as at March 31, 2019 and
March 31, 2018:
|
|
|
March 31, 2019
|
|
|
March 31, 2018
|
|
|
|
|
Cost
|
|
|
Accumulated
Depreciation
|
|
|
Net
|
|
|
Cost
|
|
|
Accumulated
Depreciation
|
|
|
Net
|
|
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
$
|
|
|
Computers and electronics
|
|
|
286,855
|
|
|
|
243,346
|
|
|
|
43,509
|
|
|
|
256,505
|
|
|
|
223,750
|
|
|
|
32,755
|
|
|
Furniture and fixtures
|
|
|
36,795
|
|
|
|
29,648
|
|
|
|
7,147
|
|
|
|
36,795
|
|
|
|
28,051
|
|
|
|
8,744
|
|
|
Demonstration equipment
|
|
|
271,615
|
|
|
|
147,257
|
|
|
|
124,358
|
|
|
|
200,186
|
|
|
|
105,441
|
|
|
|
94,745
|
|
|
Manufacturing equipment
|
|
|
88,742
|
|
|
|
86,230
|
|
|
|
2,512
|
|
|
|
88,742
|
|
|
|
85,668
|
|
|
|
3,074
|
|
|
Tools and parts
|
|
|
11,422
|
|
|
|
6,779
|
|
|
|
4,643
|
|
|
|
11,422
|
|
|
|
5,741
|
|
|
|
5,681
|
|
|
Assets under capital lease
|
|
|
23,019
|
|
|
|
12,660
|
|
|
|
10,359
|
|
|
|
23,019
|
|
|
|
8,057
|
|
|
|
14,962
|
|
|
|
|
|
718,448
|
|
|
|
525,920
|
|
|
|
192,528
|
|
|
|
616,669
|
|
|
|
456,708
|
|
|
|
159,961
|
|
Equipment is recorded at cost less accumulated
depreciation. Depreciation expense during the year ended March 31, 2019 was $69,212 (March 31, 2018 - $89,026).
BIONIK LABORATORIES
CORP.
NOTES TO CONSOLIDATED
FINANCIAL STATEMENTS
For the years
ended March 31, 2019 and 2018
(Amounts expressed
in U.S. Dollars)
The Company had an outstanding note payable
(“Notes”) of $Nil at March 31, 2019 ($51,479 – March 31, 2018), which was acquired from IMT on April 21, 2016.
The Note and interest was repaid during the year ended March 31, 2019.
|
Balance March 31, 2017
|
|
|
330,600
|
|
|
Accrued interest
|
|
|
8,497
|
|
|
Repayment
|
|
|
(287,618
|
)
|
|
Balance, March 31, 2018
|
|
$
|
51,479
|
|
|
Accrued interest
|
|
|
1,496
|
|
|
Repayment
|
|
|
(52,975
|
)
|
|
Balance, March 31, 2019
|
|
$
|
-
|
|
Interest expense incurred on the Note
totaled $1,496 for the year ended March 31, 2019 (March 31, 2018 - $8,497), which was included in accrued liabilities until it
was paid off.
|
|
(b)
|
Promissory
Notes Payable
|
In February
2014, the Company borrowed $200,000 for an existing investor under the terms of a secured promissory note (“Promissory Note”).
The Promissory Note bears interest at a simple interest rate equal to 10% per annum and interest is payable quarterly. Interest
expenses incurred on the Promissory Note totaled $Nil for year ended March 31, 2019 (March 31, 2018 - $12,957). The Promissory
Note was paid in full during the year ended March 31, 2018.
In December
2017, a company controlled by a Board member made a short-term loan to the Company of $400,000 with interest at 1.5% per month.
Interest expenses totaled $Nil for the year ended March 31, 2019 (March 31, 2018 -$3,200). The Company repaid this note with interest
of $3,200 during the year ended March 31, 2018.
|
|
(d)
|
Convertible Loans Payable
|
(1) In December 2016,
several shareholders of the Company agreed to advance the Company $1,500,000 of convertible notes in three tranches: $500,000
upon origination of the convertible loans and $500,000 on each of January 15, 2017 and February 15, 2017. A further $500,000 was
advanced in March 2017 to bring the total of these convertible loans to approximately $2,000,000. The convertible loans bore interest
at 6% until the original due date of March 31, 2017 and $17,488 was accrued and expensed as interest on these loans for the year
ended March 31, 2017.
The convertible loans contain the following
terms: convertible at the option of the holder at the price of the equity financing or payable on demand upon the completion of
an equity financing greater than $5,000,000; automatically convertible at the price of the equity financing upon completion of
an equity financing between $3,500,000 and $5,000,000; if no such equity financing is completed by November 15, 2017, then the
loans shall become secured by a general security agreement over all assets of the Company; and, upon a change in control would
either be payable on demand or convertible at the lesser of a price per share equal to that received by the parties in the change
in control transaction or the market price of the shares. These conversion features were analyzed and determined to be contingent
conversion features, accordingly, until the triggering event no beneficial conversion feature is recognized.
On August 14, 2017, the Company entered
into an amendment to these convertible loans, whereby the interest was changed to a fixed rate of 12% per year from April 1, 2017
to August 14, 2017, and 3% per month from August 14, 2017 to maturity, which was extended to the earlier of March 31, 2018 or
consummation of a qualified financing. The conversion feature was modified to contain the following terms: upon the consummation
of an equity or equity-linked round of with an aggregate gross proceeds of $7,000,000, without any action on part of the Holder,
the outstanding principal, accrued and unpaid interest and premium amount equal to 25% of the principal amount less the accrued
and unpaid interest, will be converted into shares of new round stock based upon the lesser of (a) the lowest issuance (or conversion)
price of new round stock in case there is more than one tranche of new round stock or (b) $0.25.
BIONIK LABORATORIES CORP.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
For the years ended March 31, 2019 and
2018
(Amounts expressed in U.S. Dollars)
|
|
8.
|
NOTES
PAYABLE - Continued
|
Further, the Company issued warrants to
these debt holders amounting to 20% of the aggregate principal of the convertible loans divided by the exercise price, which would
be determined as the lowest of a new round stock in a qualified financing, the average volume weighted average price for the sixty
trading days prior to January 31, 2018 or $0.25. The warrants have a term of five years. These amendments were treated as an extinguishment
of the original debt; however, there was no gain or loss recognized and the new and amended debts were recognized as shown below.
An additional $2,999,975 was received
from these shareholders during the year ended March 31, 2018 for a total of $4,999,975. For the year ended March 31, 2018, an
additional $1,037,067 of interest was accrued and expensed on these convertible loans.
The Company has recognized a discount
against the convertible loans for the relative fair value of the warrants and is accreting the discount using the effective interest
rate method. The assumptions used in valuing the warrants using the binomial valuation model were as follows: exercise price of
$0.25, volatility of 114%, risk-free interest rate of 1.91% and a term of five years. The Company evaluated the fair value of
the warrants attached to the convertible notes as $548,178 and recorded $548,178 of accretion expense in the year ended March
31, 2018.
|
Balance, March 31, 2016
|
|
$
|
-
|
|
|
Additional principal investment
|
|
|
2,000,000
|
|
|
Accrued Interest
|
|
|
17,488
|
|
|
Balance, March 31, 2017
|
|
|
2,017,488
|
|
|
Additional principal investment
|
|
|
2,999,975
|
|
|
Fair value of warrants
|
|
|
(548,178
|
)
|
|
Accretion expense
|
|
|
548,178
|
|
|
Accrued Interest
|
|
|
1,037,067
|
|
|
Conversion of principal and interest
|
|
|
(6,054,530
|
)
|
|
Balance, March 31, 2018 and March 31, 2019
|
|
$
|
-
|
|
(2) In May
2017, the Company’s Chinese joint venture partners loaned the Company $500,000 at an interest rate of 8% convertible into
the Company’s common shares upon a capital raise (“Qualified Financing”) where gross proceeds exceed $3,000,000
at the lesser of $0.50 and the quotient of the outstanding balance on the conversion date by the price of the Qualified Financing.
Additionally, the holders are entitled to warrants equaling 25% of the number of conversion shares to be issued at conversion.
During the year ended March 31, 2018 $33,556 of interest was accrued and expensed on these convertible loans.
|
Balance, March 31, 2017
|
|
$
|
-
|
|
|
Additional principal investment
|
|
|
500,000
|
|
|
Accrued Interest
|
|
|
33,556
|
|
|
Conversion of principal and interest
|
|
|
(533,556
|
)
|
|
Balance, March 31, 2018 and March 31, 2019
|
|
$
|
-
|
|
(3) In December
2017, investors of the Company advanced funds under a new convertible loan offering. These convertible loans bear interest at
a fixed rate of 3% per month until the earlier of (a) January 31, 2018 and (b) the consummation of a qualified financing defined
as gross proceeds of no less than $7,000,000 and up to $14,000,000 raised in one or more tranches. On the maturity date, without
any action on the part of the Holder, the outstanding principal and accrued and unpaid interest under the notes will be converted
into shares of new round stock based upon a (15%) discount to the lesser of (i) (A) the VWAP average of the last 30 days ending
on the closing of the qualified financing (or, in the event of multiple closings, the lowest VWAP average of the last 30 days
ending on each closing of a qualified financing) in the event of a maturity date referred to in clause (b) of the definition thereof,
or (B) the VWAP average of the last 30 days before the maturity date in the event of a maturity date referred to in clause (a)
of the definition thereof, and (ii) ($0.18). In January 2018, the terms of the new convertible loan offering were amended to extend
the maturity date until March 31, 2018 and in March 2018 the terms of the loans were amended to change the definition of qualified
financing as gross proceeds of no less than $2,000,000 and up to $14,000,000 raised in one or more tranches.
BIONIK LABORATORIES
CORP.
NOTES TO CONSOLIDATED
FINANCIAL STATEMENTS
For the years
ended March 31, 2019 and 2018
(Amounts expressed
in U.S. Dollars)
|
|
8.
|
NOTES
PAYABLE - Continued
|
$3,611,400 was received from these investors
during the twelve months ended March 31, 2018 and $201,928 of interest was accrued and expensed on these convertible loans for
the twelve months ended March 31, 2018.
|
Balance, March 31, 2017
|
|
|
-
|
|
|
Additional principal investment
|
|
|
3,611,400
|
|
|
Accrued Interest
|
|
|
201,928
|
|
|
Conversion of principal and interest
|
|
|
(3,813,328
|
)
|
|
Balance, March 31, 2018 and March 31, 2019
|
|
$
|
-
|
|
|
|
(4)
|
Conversion of
Notes Payable
|
|
|
|
March 31, 2018
|
|
|
|
|
Principal
|
|
|
Interest
|
|
|
Premium
|
|
|
Total
Conversion
Amount
|
|
|
Beneficial
Conversion
Feature
|
|
|
Number of
Shares
Converted
|
|
|
Convertible Notes Payable (December 2016 to December 2017)
|
|
$
|
4,999,975
|
|
|
$
|
1,054,555
|
|
|
$
|
1,249,994
|
|
|
$
|
7,304,523
|
|
|
$
|
762,301
|
|
|
|
779,461
|
|
|
Chinese Convertible Loan
|
|
$
|
500,000
|
|
|
$
|
33,556
|
|
|
|
-
|
|
|
$
|
533,556
|
|
|
$
|
76,230
|
|
|
|
62,629
|
|
|
Convertible Notes Payable (December 2017 to March 2018)
|
|
$
|
3,611,400
|
|
|
$
|
201,928
|
|
|
|
-
|
|
|
$
|
3,813,328
|
|
|
$
|
550,598
|
|
|
|
406,918
|
|
|
Total
|
|
$
|
9,111,375
|
|
|
$
|
1,290,039
|
|
|
$
|
1,249,994
|
|
|
$
|
11,651,407
|
|
|
$
|
1,389,129
|
|
|
|
1,249,008
|
|
|
|
(5)
|
Between April 1, 2018 and July 20,
2018, the Company received loans totaling $4,708,306 (which is inclusive of $31,673 that
was capitalized interest which carried an interest rate of 1% per month) and of which
$2,297,928 came from related parties. $4,732,853 of the loans and accrued and unpaid
interest thereon were converted into 683,396 common shares as of July 20, 2018 at a 10%
discount to the 30-day volume weighted average price (“VWAP”) of the Company’s
stock price.
|
The tables below reflect the fair value and anti-dilution features
of the convertible loans, which resulted in accretion expense related to the loans:
|
|
|
At issuance
|
|
|
At July 20, 2018
|
|
|
|
|
Principal
|
|
|
Conversion feature fair
value
|
|
|
|
|
|
|
|
|
|
|
|
Ending
|
|
|
|
|
|
|
|
Conversion
|
|
|
Anti-dilution
|
|
|
Fair value of
debt
|
|
|
Accretion
expense
|
|
|
Interest
|
|
|
balance
before
conversion
|
|
|
Convertible promissory note
|
|
$
|
4,708,306
|
|
|
$
|
406,744
|
|
|
$
|
1,697,674
|
|
|
$
|
2,603,888
|
|
|
$
|
2,104,418
|
|
|
$
|
24,547
|
|
|
$
|
4,732,853
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Conversion
|
|
|
Anti-dilution
|
|
|
Total
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Conversion feature fair value
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
At Issuance
|
|
|
|
|
|
$
|
406,744
|
|
|
$
|
1,697,674
|
|
|
$
|
2,104,418
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fair value adjustment
|
|
|
|
|
|
$
|
(406,744
|
)
|
|
$
|
68,821
|
|
|
$
|
(337,923
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance allocated to equity on conversion
|
|
|
|
|
|
$
|
-
|
|
|
$
|
(1,766,495
|
)
|
|
$
|
(1,766,495
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Ending balance at March 31, 2019
|
|
|
|
|
|
$
|
-
|
|
|
$
|
-
|
|
|
$
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
|
BIONIK LABORATORIES
CORP.
NOTES TO CONSOLIDATED
FINANCIAL STATEMENTS
For the years
ended March 31, 2019 and 2018
(Amounts expressed
in U.S. Dollars)
|
|
8.
|
NOTES
PAYABLE - Continued
|
(6) Between
October 1, 2018 and March 31, 2019, the Company received $4,650,000 in new convertible loans (“New Loans”), which
carry an interest rate of 1% per month and of which $1,050,000 came from related parties.
The schedules below reflect the accretion
expense of $1,162,500 and interest expense of $198,117 being expensed in relation to the New Loans for the year ended March 31,
2019. The loan and accrued interest of $6,010,617 was converted into 1,247,099 common shares on March 28, 2019 based on a 20%
discount to the 30-day VWAP of the Company’s stock price.
|
|
|
At issuance
|
|
|
At March 28, 2019
|
|
|
|
|
Principal
|
|
|
Accretion expense
|
|
|
Interest
|
|
|
Ending Balance
|
|
|
Convertible promissory note
|
|
$
|
4,650,000
|
|
|
$
|
1,162,500
|
|
|
$
|
198,117
|
|
|
$
|
6,010,617
|
|
(7) During
the year ended March 31, 2019, the Company received loans totaling $9,326,633 (March 31, 2018 - $7,111,375) noted in (5) and (6),
which carry an interest rate of 1% per month and of which $3,347,928 came from related parties. An accretion expense of $3,266,918
and a fair value adjustment of $337,923 was expensed for the year ended March 31, 2019 (March 31, 2018 - $1,937,308 accretion
and $Nil and $Nil fair value adjustment).
|
|
9.
|
RELATED
PARTY TRANSACTIONS AND BALANCES
|
|
|
(a)
|
Due
from related parties
|
An outstanding loan to the
Chief Technology Officer (“CTO”) of the Company is for $18,585 (March 31, 2018 - $18,897). The loan had an interest
rate of 1% until March 31, 2018 and 2% after based on the Canada Revenue Agency’s prescribed rate for such advances and
is denominated in Canadian dollars. During the year ended March 31, 2019, the Company accrued interest receivable in the amount
of $353 (March 31, 2018 – $590); the remaining fluctuation in the balance from the prior year is due to changes in foreign
exchange.
|
|
(b)
|
Accounts payable and accrued liabilities
|
As at March 31, 2019, $229,473
(March 31, 2018 - $208,567) was owing to the CEO of the Company; $14,851 (March 31, 2018 – $135,039) was owing to the Chief
Technology Officer; $33,387 (March 31, 2018 - $116,624) was owing to the Chief Financial Officer (“CFO”), $28,025
was owing to the Chief Commercial Officer (“CCO”), and $Nil (March 31, 2018 – $587,019) was owing to the former
CEO, all related to severance, bonuses and business expenses.
BIONIK LABORATORIES CORP
.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
For the years ended March 31, 2019 and
2018
(Amounts expressed in U.S. Dollars)
|
|
|
March 31, 2019
|
|
|
March 31, 2018
|
|
|
|
|
Number of shares
|
|
|
$
|
|
|
Number of shares
|
|
|
$
|
|
|
Exchangeable Shares
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance beginning of year
|
|
|
295,146
|
|
|
|
295
|
|
|
|
319,396
|
|
|
|
319
|
|
|
Converted into common shares (d)
|
|
|
(98,347
|
)
|
|
|
(98
|
)
|
|
|
(24,250
|
)
|
|
|
(24
|
)
|
|
Balance at end of year
|
|
|
196,799
|
|
|
|
197
|
|
|
|
295,146
|
|
|
|
295
|
|
|
Common Shares
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at beginning of the year
|
|
|
1,368,856
|
|
|
|
1,369
|
|
|
|
325,901
|
|
|
|
326
|
|
|
Shares issued to exchangeable shareholders (d)
|
|
|
98,347
|
|
|
|
98
|
|
|
|
24,250
|
|
|
|
24
|
|
|
Shares issued on conversion of loans (b) (c)
|
|
|
2,194,133
|
|
|
|
2,194
|
|
|
|
985,370
|
|
|
|
986
|
|
|
Warrants exercised (a)
|
|
|
-
|
|
|
|
-
|
|
|
|
33,335
|
|
|
|
33
|
|
|
Share consolidation rounding adjustment
|
|
|
502
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Balance at end of the year
|
|
|
3,661,838
|
|
|
|
3,661
|
|
|
|
1,368,856
|
|
|
|
1,369
|
|
|
TOTAL SHARES
|
|
|
3,858,637
|
|
|
|
3,858
|
|
|
|
1,664,002
|
|
|
|
1,664
|
|
|
|
(a)
|
During the year ended March 31,
2018, the Company consummated an offer to amend and exercise to its warrant holders,
enabling them to exercise their outstanding warrants for $37.50 per share, and as a result,
33,335 common shares were issued for net proceeds of $1,125,038 (Note 12).
|
|
|
(b)
|
During the year ended March 31,
2018, the Company converted $9,058,708 of notes payable and interest into 985,370 common
shares. Under the terms of this conversion the remaining $1,342,705 of principal and
interest was required to be converted into 263,639 common shares, but they were unable
to be issued as a result of the Company not having enough authorized shares. The $2,470,622
value of these shares at March 31, 2018 has been classified as a liability until the
common shares can be issued. In addition, there was a $376,674 loss recorded in the year
connected to the difference of the $2,847,296 market value of the shares at March 31,
2018 and the value of these shares which resulted on the conversion of notes payable,
the exercise price of which was based on a 30-day VWAP. The fair value of these shares
($2,847,296), outstanding warrants ($1,394,164) and options ($1,451,393) at March 31,
2018 represented the $5,692,853 liability on the Company’s balance sheet.
|
|
|
(c)
|
During the year ended March 31,
2019, after the increase of the number of authorized shares to 500,000,000, the company
issued the outstanding 263,639 common shares related to the March 31, 2018 promissory
note conversion. In addition, there was a $2,048,697 gain recorded in the year connected
to the difference of the market value of the shares, outstanding options and warrants
at March 31, 2018 and their value at June 12, 2018, the time of the authorized share
increase and share issuance. On July 20, 2018 the Company converted $4,708,306 of notes
payable and interest into 683,395 common shares and on March 28, 2019 the Company converted
$4,848,117 of notes payable and interest into 1,247,099 common shares. (Note 8)
|
|
|
(d)
|
During the
year ended March 31, 2019, 98,347 exchangeable shares were exchanged for common shares
on a 1 for 1 basis in accordance with their terms. (March 31, 2018 – 24,250 shares)
|
|
|
(e)
|
On October
29, 2018, the Company completed a one-for-one hundred and fifty to one (1:150) reverse
stock consolidation that has been reflected in all shares and per share amounts, warrants
and options.
|
Special Voting Preferred Share
In connection with the Merger (Note 1),
on February 26, 2015, the Company entered into a voting and exchange trust agreement (the “Trust Agreement”). Pursuant
to the Trust Agreement, the Company issued one Special Voting Preferred Share to the Trustee, and the parties created a trust
for the Trustee to hold the Special Voting Preferred Share for the benefit of the holders of the Exchangeable Shares (the “Beneficiaries”).
Pursuant to the Trust Agreement, the Beneficiaries will have voting rights in the Company equivalent to what they would have had,
had they received shares of common stock in the same amount as the Exchangeable Shares held by the Beneficiaries. In connection
with the Merger and the Trust Agreement, effective February 20, 2015, the Company filed a certificate of designation of the Special
Voting Preferred Share (the “Special Voting Certificate of Designation”) with the Delaware Secretary of State. Pursuant
to the Special Voting Certificate of Designation, one share of the Company’s blank check preferred stock was designated
as Special Voting Preferred Share. The Special Voting Preferred Share entitles the Trustee to exercise the number of votes equal
to the number of Exchangeable Shares outstanding on a one-for-one basis during the term of the Trust Agreement. The Special Voting
Preferred Share is not entitled to receive any dividends or to receive any assets of the Company upon liquidation and is not convertible
into shares of common stock of the Company. The voting rights of the Special Voting Preferred Share will terminate pursuant to
and in accordance with the Trust Agreement and the Special Voting Preferred Share will be automatically cancelled.
BIONIK LABORATORIES CORP.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
For the years ended March 31, 2019 and
2018
(Amounts expressed in U.S. Dollars)
The purpose of the Company’s equity
incentive plan, is to attract, retain and motivate persons of training, experience and leadership to the Company, including their
directors, officers and employees, and to advance the interests of the Company by providing such persons with the opportunity,
through share options, to acquire an increased proprietary interest in the Company.
Options or other securities may be granted
in respect of authorized and unissued shares, provided that the aggregate number of shares reserved for issuance upon the exercise
of all options or other securities granted under the Plan shall not exceed 15% of the shares of common stock and Exchangeable
Shares issued and outstanding (determined as of January 1 of each year). Optioned shares in respect of which options are not exercised
shall be available for subsequent options.
On November 24, 2015, the Company issued
4,334 options granted to employees that vest at an exercise price of $183.00 over three years at the anniversary date. The grant
date fair value of the options was $694,384. During the year ended March 31, 2016, 1,667 options were cancelled. During the year
ended March 31, 2019, $92,585, (March 31, 2018 -$142,438) in stock compensation expense was recognized and these options are now
fully expensed.
On December 14, 2015, the Company issued
16,634 options granted to employees, directors and consultants at an exercise price of $150 that vest over three years at the
anniversary date. The grant date fair value of the options was $1,260,437. In years previous to March 31, 2018, 434 options were
cancelled. For the year ended March 31, 2019 1,889 options were cancelled (March 31, 2018 - 2,912), $105,121, (March 31, 2018
- $479,315) of stock compensation expense was recognized and these options are now fully expensed.
On April 21, 2016, the Company issued
20,000 stock options to employees of Bionik Inc., the Company’s wholly-owned subsidiary (formerly IMT) in exchange for 3,895,000
options that existed before the Company purchased IMT, of which 6,667 have an exercise price of $37.50 per share, 6,667 have an
exercise price of $142.50 per share and 6,666 have an exercise price of $157.50 per share. The grant date fair value of vested
options was $2,582,890 and has been recorded in fiscal 2016, $Nil (March 31, 2018 -$29,524) has been recognized as stock compensation
expense for the year ended March 31, 2019. These options are now fully expensed.
On April 26, 2016, the Company issued
1,667 options to an employee with an exercise price of $150.00 per share that will vest over three years at the anniversary date.
The grant fair value was $213,750. During the year ended March 31, 2019 557 options were cancelled (March 31, 2018 – Nil)
and $63,333 (March 31, 2018 - $71,250) was recognized as stock compensation expense.
On August 8, 2016, the Company issued
5,000 options to an employee with an exercise price of $150.00 per share that will vest over three years at the anniversary date.
The grant fair value was $652,068. During the year ended March 31, 2019, $48,301, (March 31, 2018 -$217,356) of stock compensation
expense was recognized, 3,335 of the options were cancelled during the year ended March 31, 2019 as a result of this employee
leaving.
On February 6, 2017, the Company issued
2,667 options to an employee with an exercise price of $105.00 per share that will vest over three years at the anniversary date.
The grant fair value was $245,200. During the year ended March 31, 2019, $81,733 (March 31, 2018 - $81,733) of stock compensation
expense was recognized.
On February 13, 2017, the Company issued
1,667 options to a consultant with an exercise price of $102.00 per share that will vest over one and one- half years, every six
months. The grant fair value was $148,750. During the year ended March 31, 2019, $92,821, (March 31, 2018 -$49,583) of stock compensation
expense was recognized and these options are now fully expensed.
BIONIK LABORATORIES
CORP
.
NOTES TO CONSOLIDATED
FINANCIAL STATEMENTS
For the years
ended March 31, 2019 and 2018
(Amounts expressed
in U.S. Dollars)
|
|
11.
|
STOCK
OPTIONS – Continued
|
On August 3, 2017, 13,334 options at $31.50
per share to an executive officer, which vest equally over three years. In addition, this executive officer was also granted up
to 10,000 additional performance options based on meeting sales targets for the years ended March 31, 2018 and 2019. The performance
options will vest at market price if the performance objectives are met. This grant had a grant date fair value of $387,209 and
$7,546 (March 31,2018 - $60,371) was recognized as share compensation expense for the year ended March 31, 2019. This officer
left in April 2018 and all options were cancelled.
On September 1, 2017, the Company granted
81,436 options at $24.15 per share equally to an executive officer and a consultant, who is now the Chairman of the Company. 27,148
options have vested and 50% of the remaining options vest on performance being met and 50% vest annually over 5 years for the
CEO, for our Chairman the options vest over 5 years. The grant date fair value was $1,832,304 and $343,557 is the current expense
for the year ended March 31, 2019. (March 31, 2018 - $381,730)
On January 24, 2018, the Company granted
24,267 options at $23.25 per share to employees that vest equally on January 24, 2019, 2020 and 2021. 7,334 options were cancelled
for the year ended March 31, 2019 (March 31, 2018 - $Nil). The grant fair value was $491,036 and $140,540 is the current stock
compensation expense for the year ended March 31, 2019. (March 31, 2018 - $27,280)
On April 30, 2018, the Company granted
to an executive officer, 40,000 options with an exercise price of $9.74 that vest immediately with a 10-year expiry. These options
were valued using the Black Scholes model and the following inputs were used: expected life 10 years, expected volatility 114%
and a risk-free rate of 1.59%. As these options vested immediately as of the grant date and $363,714 of stock compensation expense
was recorded for the year ended March 31, 2019.
On June 11, 2018, the Company granted
to a sales executive officer, 5,000 options with an exercise price of $6.93 per share that vest over three years from the anniversary
of the grant and expire in 7 years. The options were valued using the Black Scholes model and the following inputs were used:
expected life of 7 years, expected volatility of 114% and a risk-free rate of 1.59%. The grant fair value was $30,341 and $8,147
of stock compensation was recognized for year ended March 31, 2019.
During the year ended March 31, 2019,
the Company recorded $1,347,399 in share-based compensation related to the vesting of stock options (March 31, 2018 - $1,540,580).
The following is a summary of stock options outstanding and
exercisable as of March 31, 2019.
These options at their respective grant dates were valued using
the Black-Scholes option pricing model with the following key assumptions:
|
|
|
Expected life
|
|
|
Risk free
|
|
|
Dividend
|
|
|
Forfeiture
|
|
|
Expected
|
|
|
Grant date
|
|
|
Grant date
|
|
in years
|
|
|
Rate
|
|
|
Rate
|
|
|
Rate
|
|
|
volatility
|
|
|
fair value
|
|
|
February 17, 2015
|
|
|
2.89
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
136,613
|
|
|
July 1, 2014
|
|
|
2.25
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
1,259,487
|
|
|
June 20, 2014
|
|
|
2.22
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
118,957
|
|
|
November 24, 2015
|
|
|
3.65
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
694,384
|
|
|
December 14, 2015
|
|
|
3.71
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
1,260,437
|
|
|
April 21, 2016
|
|
|
5.11
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
2,582,890
|
|
|
April 26, 2016
|
|
|
4.07
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
213,750
|
|
|
February 6, 2017
|
|
|
4.86
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
245,200
|
|
|
February 13, 2017
|
|
|
4.88
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
148,750
|
|
|
September 1, 2017
|
|
|
8.43
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
1,832,304
|
|
|
January 24, 2018
|
|
|
5.82
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
491,036
|
|
|
April 30, 2018
|
|
|
9.06
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
363,714
|
|
|
June 11, 2018
|
|
|
6.20
|
|
|
|
1.59
|
%
|
|
|
0
|
%
|
|
|
0
|
%
|
|
|
114
|
%
|
|
$
|
30,341
|
|
BIONIK LABORATORIES
CORP
.
NOTES TO CONSOLIDATED
FINANCIAL STATEMENTS
For the years
ended March 31, 2019 and 2018
(Amounts expressed
in U.S. Dollars)
|
|
11.
|
STOCK
OPTIONS – Continued
|
|
|
|
Number of
Options
|
|
|
Weighted-Average
Exercise Price
|
|
|
Outstanding, March 31, 2018
|
|
|
170,675
|
|
|
|
75.00
|
|
|
Issued
|
|
|
45,000
|
|
|
|
9.42
|
|
|
Cancelled
|
|
|
(32,679
|
)
|
|
|
65.93
|
|
|
Outstanding, March 31, 2019
|
|
|
182,996
|
|
|
|
37.73
|
|
The following is a summary of stock options outstanding and
exercisable as of March 31, 2019:
|
Exercise Price ($)
|
|
|
Number of Options
|
|
|
Expiry Date
|
|
Exercisable Options
|
|
|
|
34.500
|
|
|
|
630
|
|
|
20-Jun-21
|
|
|
630
|
|
|
|
34.500
|
|
|
|
13,212
|
|
|
01-Jul-21
|
|
|
13,212
|
|
|
|
34.500
|
|
|
|
944
|
|
|
17-Feb-22
|
|
|
944
|
|
|
|
183.000
|
|
|
|
2,667
|
|
|
24-Nov-22
|
|
|
2,667
|
|
|
|
150.000
|
|
|
|
11,400
|
|
|
14-Dec-22
|
|
|
11,400
|
|
|
|
142.500
|
|
|
|
359
|
|
|
28-Mar-23
|
|
|
359
|
|
|
|
157.500
|
|
|
|
1,387
|
|
|
28-Mar-23
|
|
|
1,387
|
|
|
|
150.000
|
|
|
|
1,112
|
|
|
26-Apr-23
|
|
|
1,112
|
|
|
|
105.000
|
|
|
|
2,667
|
|
|
06-Feb-24
|
|
|
1,778
|
|
|
|
102.000
|
|
|
|
1,667
|
|
|
13-Feb-24
|
|
|
1,667
|
|
|
|
142.500
|
|
|
|
106
|
|
|
03-Mar-24
|
|
|
106
|
|
|
|
157.500
|
|
|
|
408
|
|
|
03-Mar-24
|
|
|
408
|
|
|
|
142.500
|
|
|
|
43
|
|
|
14-Mar-24
|
|
|
43
|
|
|
|
157.500
|
|
|
|
164
|
|
|
14-Mar-24
|
|
|
164
|
|
|
|
142.500
|
|
|
|
485
|
|
|
30-Sep-24
|
|
|
485
|
|
|
|
157.500
|
|
|
|
1,876
|
|
|
30-Sep-24
|
|
|
1,876
|
|
|
|
142.500
|
|
|
|
24
|
|
|
02-Jun-25
|
|
|
24
|
|
|
|
157.500
|
|
|
|
90
|
|
|
02-Jun-25
|
|
|
90
|
|
|
|
37.500
|
|
|
|
221
|
|
|
30-Dec-25
|
|
|
221
|
|
|
|
142.500
|
|
|
|
164
|
|
|
30-Dec-25
|
|
|
164
|
|
|
|
24.150
|
|
|
|
81,436
|
|
|
01-Sep-27
|
|
|
27,148
|
|
|
|
23.250
|
|
|
|
16,934
|
|
|
24-Jan-25
|
|
|
5,867
|
|
|
|
9.735
|
|
|
|
40,000
|
|
|
19-Apr-28
|
|
|
40,000
|
|
|
|
6.930
|
|
|
|
5,000
|
|
|
10-Jun-25
|
|
|
-
|
|
|
|
|
|
|
|
182,996
|
|
|
|
|
|
111,752
|
|
The weighted-average remaining contractual term of the outstanding
options is 7.20 years (March 31, 2018 – 7.46 years) and for the options that are exercisable the weighted average is 6.80
years (March 31, 2018 – 5.74 years).
BIONIK LABORATORIES CORP
.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
For the years ended March 31, 2019 and
2018
(Amounts expressed in U.S. Dollars)
The following is a continuity schedule of the Company’s
common share purchase warrants:
|
|
|
Number of
Warrants
|
|
|
Weighted
-Average
Exercise Price
($)
|
|
|
Outstanding and exercisable, March 31, 2017
|
|
|
117,589
|
|
|
|
202.50
|
|
|
Exercised
|
|
|
(33,335
|
)
|
|
|
(37.50
|
)
|
|
Issued in connection with anti-dilution provision connected warrant transaction
|
|
|
559
|
|
|
|
112.35
|
|
|
Issued in connection with anti-dilution provision connected warrant transaction
|
|
|
6,275
|
|
|
|
194.00
|
|
|
Issued in connection to the warrant transaction to the broker
|
|
|
2,667
|
|
|
|
37.50
|
|
|
Issued in connection with conversion of loans and interest into common shares
|
|
|
106,709
|
|
|
|
9.375
|
|
|
Issued in connection with conversion of loans and interest into common shares
|
|
|
15,658
|
|
|
|
90.00
|
|
|
Issued in connection with anti-dilution provision connected with issuance of common shares
|
|
|
136,388
|
|
|
|
73.02
|
|
|
Issued in connection with anti-dilution provision connected with issuance
of common shares
|
|
|
13,464
|
|
|
|
44.28
|
|
|
Outstanding and exercisable, March 31, 2018
|
|
|
365,974
|
|
|
|
53.19
|
|
|
Issued in connection with anti-dilution provision connected warrant transaction
|
|
|
67,952
|
|
|
|
55.71
|
|
|
Issued in connection with anti-dilution provision connected warrant transaction
|
|
|
6,305
|
|
|
|
34.50
|
|
|
Issued in connection with anti-dilution provision connected warrant transaction
|
|
|
52,590
|
|
|
|
38.91
|
|
|
Expired
|
|
|
(204,304
|
)
|
|
|
(51.85
|
)
|
|
Outstanding and exercisable, March 31, 2019
|
|
|
288,517
|
|
|
|
40.27
|
|
During the year ended March 31, 2019,
204,304 warrants expired in accordance with their terms (March 31, 2018 - Nil)
During the year ended March 31, 2018,
the Company consummated an offer to amend and exercise its then outstanding warrants, enabling the holders of the warrants to
exercise such warrants for $37.50 per share. The Company received net proceeds of $1,125,038. The Company also converted loans
and interest due.
Due to an anti-dilution clause in the
warrant agreements for such outstanding warrants during the year ended March 31, 2019, an additional 67,952 warrants were issued
to the $73.02 per share warrant holders and 6,305 warrants to the $44.28 per share warrant holders. As a result of the anti-dilution
clause, the exercise price of the warrants changed from $73.02 per share to $55.71 per share and from $44.28 per share to $34.50
per share as a result of this warrant transaction. The $34.49 per share warrants expired during the fiscal year ended March 31,
2019.
Furthermore, due to an anti-dilution clause
in the warrant agreements for such outstanding warrants during the year ended March 31, 2019, an additional 52,590 warrants were
issued to the $55.71 per share warrant holders. As a result of the anti-dilution clause, the exercise price of the warrants changed
from $55.71 per share to $38.91 per share as a result of this warrant transaction.
During the fiscal year ended March 31,
2018, an additional 559 warrants were issued to the $120.00 per share warrant holders and 6,275 warrants were issued to the $210.00
per share warrant holders. Furthermore, as a result of the anti-dilution clause, the exercise price of the warrants changed from
$120.00 per share to $112.35 per share and from $210.00 per share to $194.00 per share as a result of this warrant transaction.
BIONIK LABORATORIES CORP
.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
For the years ended March 31, 2019 and
2018
(Amounts expressed in U.S. Dollars)
During the fiscal year ended March 31,
2018, due to an anti-dilution clause in the warrant agreements for such outstanding warrants an additional 13,464 warrants were
issued to the $112.35 per share warrant holders and 136,388 warrants were issued to the $194.00 per share warrant holders. Furthermore,
as a result of the anti-dilution clause, the exercise price of the warrants changed from $112.35 per share to $44.28 per share
and from $194.00 per share to $73.02 per share as a result of loan and interest conversion transaction for shares that have been
issued and shares that will be issued.
The Company measured the effects of the
above transactions, which triggered anti-dilution clause using the binomial option pricing model and recorded a loss for the year
ended March 31, 2019 of $24,432 (March 31, 2018 -$74,086) against deficit.
The Company issued 2,667 warrants exercisable
at $37.50 per share for four years expiring June 27, 2020 to the firm who facilitated the warrant offer.
The Company issued 15,658 warrants at
$90.00 per share which expire in 5 years on March 31, 2023 and 106,709 warrants at $9.375 which also expire on March 31, 2023
in connection with a loan and interest conversion transaction.
Common share purchase warrants
The following is a summary of common share
purchase warrants outstanding after the warrant offer to amend the additional warrant issue and the re-pricing of the warrants
as of March 31, 2019.
|
Exercise
Price
($)
|
|
|
Number
of
Warrants
|
|
|
Expiry
Date
|
|
|
90.00
|
|
|
|
15,658
|
|
|
March 31, 2023
|
|
|
38.91
|
|
|
|
84,562
|
|
|
April 21, 2019
|
|
|
38.91
|
|
|
|
39,922
|
|
|
May 27, 2019
|
|
|
38.91
|
|
|
|
38,998
|
|
|
June 30, 2019
|
|
|
37.50
|
|
|
|
2,667
|
|
|
June 27, 2020
|
|
|
9.375
|
|
|
|
64,025
|
|
|
August 14, 2022
|
|
|
9.375
|
|
|
|
42,684
|
|
|
March 31, 2022
|
|
|
|
|
|
|
288,517
|
|
|
|
The weighted-average remaining contractual term of the outstanding
warrants was 1.51 years (March 31, 2018 – 2.27 years).
The exercise price and number of underlying
shares with respect to the remaining $38.91 per share warrants are expected to be further adjusted pursuant to the anti-dilution
provisions therein, as a result of any further issuance of common shares.
BIONIK LABORATORIES
CORP
.
NOTES TO CONSOLIDATED
FINANCIAL STATEMENTS
For the years
ended March 31, 2019 and 2018
(Amounts expressed in U.S. Dollars)
Components of net loss before income taxes consists of the
following:
|
|
|
March 31
|
|
|
March 31
|
|
|
|
|
2019
|
|
|
2018
|
|
|
|
|
$
|
|
|
$
|
|
|
U.S.
|
|
|
(8,849,294
|
)
|
|
|
(12,281,398
|
)
|
|
Canada
|
|
|
(1,707,307
|
)
|
|
|
(2,344,392
|
)
|
|
|
|
|
(10,556,601
|
)
|
|
|
(14,625,790
|
)
|
|
Net loss for the year before recovery of income taxes
|
|
|
(10,556,601
|
)
|
|
|
(14,625,790
|
)
|
|
Statutory rate
|
|
|
25.30
|
%
|
|
|
34.04
|
%
|
|
Expected income tax (recovery)
|
|
|
(2,670,579
|
)
|
|
|
(4,978,619
|
)
|
|
Tax rate changes and other basis adjustments
|
|
|
(525,472
|
)
|
|
|
1,748,278
|
|
|
Stock-based compensation
|
|
|
340,861
|
|
|
|
524,412
|
|
|
Difference in Foreign Tax Rates
|
|
|
-
|
|
|
|
-
|
|
|
Accretion
|
|
|
890,797
|
|
|
|
184,414
|
|
|
Change in fair value
|
|
|
(85,487
|
)
|
|
|
659,458
|
|
|
(Gain) loss on mark to market re-evaluation
|
|
|
(518,274
|
)
|
|
|
-
|
|
|
Share premium
|
|
|
-
|
|
|
|
425,497
|
|
|
Non-deductible expense
|
|
|
83,578
|
|
|
|
339,296
|
|
|
Net DTA acquired
|
|
|
-
|
|
|
|
-
|
|
|
Change in valuation allowance
|
|
|
2,484,576
|
|
|
|
1,097,264
|
|
|
Recovery of income taxes
|
|
|
-
|
|
|
|
-
|
|
The following deferred tax assets have
not been recognized. Deferred tax reflects the tax effects of temporary differences that gave rise to significant portions of
deferred tax assets and liabilities and consisted of the following:
|
|
|
March 31,
2019
|
|
|
March
31,
2018
|
|
|
|
|
$
|
|
|
$
|
|
|
Equipment
|
|
|
70,650
|
|
|
|
70,350
|
|
|
Share issue costs
|
|
|
-
|
|
|
|
510
|
|
|
SR&ED pool
|
|
|
844,001
|
|
|
|
690,320
|
|
|
Other
|
|
|
1,022,309
|
|
|
|
535,510
|
|
|
Non-capital losses – Canada
|
|
|
2,796,469
|
|
|
|
2,515,170
|
|
|
Net operating losses – U.S.
|
|
|
5,911,320
|
|
|
|
4,331,850
|
|
|
Valuation allowance
|
|
|
(9,502,006
|
)
|
|
|
(7,017,430
|
)
|
|
|
|
|
1,142,743
|
|
|
|
1,126,280
|
|
|
Intangibles and other
|
|
|
(1,142,743
|
)
|
|
|
(1,126,280
|
)
|
|
|
|
|
-
|
|
|
|
-
|
|
The Company has non-capital losses in
its Canadian subsidiary of approximately $10,552,713, which will expire between 2032 and 2039. The Company has net operating losses
in the U.S. parent Company of $14,190,773, and net operating losses in the U.S. subsidiary of approximately $14,190,773, which
will expire in 2038. Certain tax attributes are subject to an annual limitation as a result of the acquisition of the US subsidiary,
which constitutes a change of ownership as defined under IRC Section 382.
Income taxes are provided based on the
liability method, which results in deferred tax assets and liabilities arising from temporary differences. Temporary differences
are differences between the tax basis of assets and liabilities and their reported amounts in the consolidated financial statements
that will result in taxable or deductible amounts in future years. The liability method requires the effect of tax rate changes
on current and accumulated deferred taxes to be reflected in the period in which the rate change was enacted. The liability method
also requires that deferred tax assets be reduced by a valuation allowance unless it is more likely than not that the assets will
be realized.
BIONIK LABORATORIES
CORP.
NOTES TO CONSOLIDATED
FINANCIAL STATEMENTS
For the years
ended March 31, 2019 and 2018
(Amounts expressed
in U.S. Dollars)
|
|
13.
|
INCOME
TAXES – Continued
|
The Company recognizes the financial statement
benefit of a tax position only after determining that the relevant tax authority would more likely than not sustain the position
following an audit. For tax positions meeting the more-likely-than-not threshold, the amount recognized in the consolidated financial
statements is the largest benefit that has a greater than 50% likelihood of being realized upon ultimate settlement with the relevant
tax authority. The Company recognizes interest accrued on uncertain tax positions as well as interest received from favorable
tax settlements within interest expense. The Company recognizes penalties accrued on unrecognized tax benefits within general
and administrative expenses. As of March 31, 2019, the Company had no uncertain tax positions.
In many cases the Company’s uncertain
tax positions are related to tax years that remain subject to examination by tax authorities. The following describes the open
tax years, by major tax jurisdiction, as of March 31, 2019:
|
United States – Federal
|
2015 – present
|
|
United States – State
|
2015 – present
|
|
Canada – Federal
|
2014 – present
|
|
Canada – Provincial
|
2014 – present
|
Concentrations
of Credit Risk and Economic Dependence
Cash and cash equivalents include highly
liquid investments with original terms to maturity of 90 days or less at the date of purchase. For all periods presented cash
and cash equivalents consisted entirely of cash on deposit with Canadian and US banks.
The Company’s
cash balances are maintained in various banks in Canada and the United States. Deposits held in banks in the United States are
insured up to $250,000 per depositor for each bank by the Federal Deposit Insurance Corporation. Deposits held in banks in Canada
are insured up to $100,000 Canadian per depositor for each bank by The Canada Deposit Insurance Corporation, a federal crown corporation.
Actual balances at times may exceed these limits.
Three of the
Company’s customers accounted for 83.3%, 8.9% and 4.6% and 88.1%, 1.9% and 1.5%, of the Company’s gross accounts receivables
as at March 31, 2019 and 2018, respectively,.
|
|
15.
|
COMMITMENTS
AND CONTINGENCIES
|
Contingencies
From time to time, the Company may be
involved in a variety of claims, suits, investigations and proceedings arising in the ordinary course of our business, collections
claims, breach of contract claims, labor and employment claims, tax and other matters. Although claims, suits, investigations
and proceedings are inherently uncertain and their results cannot be predicted with certainty, the Company believes that the resolution
of current pending matters will not have a material adverse effect on its business, financial position, results of operations
or cash flow. Regardless of the outcome, litigation can have an adverse impact on the Company because of legal costs, diversion
of management resources and other factors.
Commitments
|
|
(a)
|
On February
25, 2015, 1,753 common shares were issued to two former lenders connected with a $241,185
loan received and repaid during fiscal 2013. The common shares were valued at $210,323
based on the value of the concurrent private placement and recorded in stock-based compensation
on the consolidated statement of operations and comprehensive loss. As part of the consideration
for the initial loan, the Company’s then-CTO and COO had transferred 2,098 common
shares to the lenders. For contributing the common shares to the lenders, the Company
intends to reimburse the former CTO and COO 2,134 common shares. As at March 31, 2019
these shares have not yet been issued.
|
BIONIK LABORATORIES
CORP.
NOTES TO CONSOLIDATED
FINANCIAL STATEMENTS
For the years
ended March 31, 2019 and 2018
(Amounts expressed
in U.S. Dollars)
|
|
15.
|
COMMITMENTS
AND CONTINGENCIES – Continued
|
|
|
(b)
|
On May 17,
2017, the Company entered into a Co-operative Joint Venture Contract (the “JV Contract”)
with Ginger Capital Investment Holding, Ltd. (the “JV Partner”) to form China
Bionik Medical Rehabilitation Technology Ltd. (“China JV”), in which the
Company will have a 25% interest and the JV Partner 75%. The China JV was formally established
on receiving a business license on May 22, 2018. Under the terms of the JV Contract,
the JV Partner is required to contribute $290,000 within 30 days of the date of establishment,
$435,000 12 months later and $725,000, 60 months after the date of establishment. The
Company is required to license certain intellectual property to the China JV. The Company
is applying the equity method of accounting to the joint venture. As of March 31, 2019,
the Company has provided certain technical information to the Chinese JV in order to
obtain Chinese regulatory approvals.
|
|
|
(c)
|
In connection with the acquisition
of IMT, the Company acquired a license agreement dated June 8, 2009, with a former director
as a co- licenser, pursuant to which the Company pays the director and the co-licenser
an aggregate royalty of 1% of sales based on patent #8,613,691. No sales have been made,
as the technology under this patent has not been commercialized.
|
|
|
(d)
|
The Company has committed to
upgrading two robots previously sold to a customer to the newest version when released.
As part of this transaction, the customer will enter into an extended warranty agreement
that will be approximately equal to the manufacturing value of robots.
|
|
|
(a)
|
Subsequent
to March 31, 2019, an exchangeable shareholder exchanged 6,083,900 pre-split exchangeable
shares into 40,560 post -split common shares.
|
|
|
(b)
|
Subsequent
to March 31, 2019, the Company’s Board granted 169,882 options at $3.16 to the
Board members and employees of the Company that will vest over one year, every six months,
starting immediately at the grant date of May 31, 2019 and expire in 7 years.
|
|
|
(c)
|
Subsequent
to March 31, 2019, an affiliate of the Company’s Chairman of the Board provided
a $500,000 term loan to the Company that bears interest at a fixed rate of 1% per month
and matures May 8, 2021.
|
|
|
(d)
|
Subsequent to
March 31, 2019, an affiliate of one of the Company’s major shareholders who is
also a director provided an aggregate amount of $500,000 and from an existing shareholder $200,000 in convertible loans to
the Company under the newly launched up to $9,000,000 convertible note offering
(“Offering”) that bears interest at a fixed rate of 1% per month and
matures on the earlier of March 30, 2020 and the consummation of the Offering,
provided that the Company raises in one or more tranches aggregate gross proceeds of
no less than US$9,000,000. The convertible loans contain an anti-dilution provision
effective when the Company converts the above Notes into Common Stock and raises
additional capital through the sale of Common Stock for cash during the period
ending on the three (3) year anniversary of the earliest Issue Date of these Notes,
and the price per share is less than the Conversion Price of these Notes, then the
Company shall issue to the Holders additional shares of Common Stock equal to the
number of Conversion Shares the Holders would have received upon conversion if the
Conversion Price equaled to the new Offering Price, less the number of shares of
Conversion Shares actually issued on or as of the Conversion Date.
|
Bionik Laboratories (CE) (USOTC:BNKL)
Historical Stock Chart
From Jun 2024 to Jul 2024
Bionik Laboratories (CE) (USOTC:BNKL)
Historical Stock Chart
From Jul 2023 to Jul 2024