Agile Therapeutics Acknowledges New Government Guidance Reinforcing Requirement to Cover Contraceptives that a Woman and Her Heathcare Provider Determine are Medically Appropriate at No Cost
13 January 2022 - 9:29AM

On January 10, 2022, the tri-agencies, Department of Labor (DOL),
Health and Human Services (HHS) and Treasury issued new guidance
that supports women’s access to U.S. Food and Drug Administration
(FDA) approved contraceptive products. Additionally, on January 11,
2022, the Health Resources and Services Administration (HRSA)
updated the Women’s Preventative Services Guidelines including
contraception services.
In their guidance, the tri-agencies reminded plans and issuers
of their responsibility to fully comply with the requirements of
the Affordable Care Act and the HRSA Guidelines, including the
requirement that, if an individual and their attending provider
determine that a particular service or FDA-approved, cleared, or
granted contraceptive product is medically appropriate for the
individual (whether or not the item or service is identified in the
current FDA Birth Control Guide), the plan or issuer must cover
that service or product without cost sharing.
They also provided a path for consumers to report concerns if
they experience issues obtaining the contraceptive deemed most
medically appropriate for them.
The updated HRSA Women's Preventive Services Guidelines, which
take effect for plan years beginning 2023, restates the requirement
that women have access to the full range of FDA-approved, granted
or cleared contraceptives as part of preventative services
available without cost sharing.
"Birth control is a very individual decision, and we agree that
women should have access to the contraceptive product determined to
be most appropriate for them based on discussions with their
healthcare provider. The recent guidelines and recommendations will
help reduce barriers by providing women with access to the birth
control most appropriate for them at no cost.” said Kimberly
Whelan, Agile Therapeutics Vice President, Policy, Advocacy and
Market Access.
Agile Therapeutics Chairman and CEO Al Altomari added, “We agree
with the recent recommendations that will help eliminate barriers
to contraceptive access for women and we will work with the plans
and issuers to achieve this goal.”
About Agile Therapeutics, Inc.Agile
Therapeutics is a women's healthcare company dedicated to
fulfilling the unmet health needs of today’s women. Our
product and product candidates are designed to provide women with
contraceptive options that offer freedom from taking a daily pill,
without committing to a longer-acting method. Our initial
product, Twirla®, (levonorgestrel and ethinyl estradiol)
transdermal system, is a non-daily prescription contraceptive.
Twirla is based on our proprietary transdermal patch technology,
called Skinfusion®, which is designed to allow drug delivery
through the skin. For more information, please visit the company
website at www.agiletherapeutics.com. The
Company may occasionally disseminate material, nonpublic
information on the Company’s website.
Certain information contained in this press release includes
“forward-looking statements” within the meaning of Section 27A of
the Securities Act of 1933, as amended, and Section 21E of the
Securities Exchange Act of 1934, as amended. We may in some cases
use terms such as “predicts,” “believes,” “potential,” “continue,”
“anticipates,” “estimates,” “expects,” “plans,” “intends,” “may,”
“could,” “might,” “likely,” “will,” “should” or other words that
convey uncertainty of the future events or outcomes to identify
these forward-looking statements. Our forward-looking statements
are based on current beliefs and expectations of our management
team that involve risks, potential changes in circumstances,
assumptions, and uncertainties, including statements regarding
third party coverage of contraceptives and women’s affordable
access to the contraceptives of their choice. Any or all of the
forward-looking statements may turn out to be wrong or be affected
by inaccurate assumptions we might make or by known or unknown
risks and uncertainties. These forward-looking statements are
subject to risks and uncertainties including risks related to
regulatory and legislative developments in the United States and
foreign countries, legal and regulatory compliance of third parties
outside our control, and the other risks set forth in our filings
with the U.S. Securities and Exchange Commission, including our
Annual Report on Form 10-K and our Quarterly Reports on Form 10-Q.
For all these reasons, actual results and developments could be
materially different from those expressed in or implied by our
forward-looking statements. You are cautioned not to place undue
reliance on these forward-looking statements, which are made only
as of the date of this press release. We undertake no obligation to
publicly update such forward-looking statements to reflect
subsequent events or circumstances.
Contact: Matt RileyHead of Investor
Relationsmriley@agiletherapeutics.com
Agile Therapeutics (NASDAQ:AGRX)
Historical Stock Chart
From Apr 2024 to May 2024
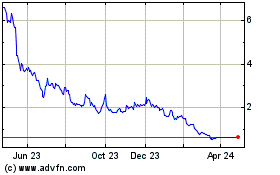
Agile Therapeutics (NASDAQ:AGRX)
Historical Stock Chart
From May 2023 to May 2024