Anika Announces Regulatory Approval for MONOVISC® in Australia for the Treatment of Pain Associated with Osteoarthritis of a...
17 October 2017 - 7:05AM
Business Wire
Anika Therapeutics, Inc. (NASDAQ: ANIK), a global, integrated
orthopedics medicines company specializing in therapeutics based on
its proprietary hyaluronic acid ("HA") technology, today announced
that regulatory authorities in Australia granted approval to
MONOVISC®, Anika’s single injection viscosupplement for the
treatment of pain associated with osteoarthritis of all synovial
joints, including the hip and knee. MONOVISC is commercially
available in over twenty countries, including the United States,
Canada, and various European countries. MONOVISC also recently
received approval in the Asia-Pacific countries of India and
Taiwan. Anika intends to further expand MONOVISC into additional
international markets in 2018.
“The Asia-Pacific region presents an important growth
opportunity for our global orthobiologics franchise, and we’re
excited to bring MONOVISC to patients in Australia and New
Zealand,” said Charles H. Sherwood, Ph.D., Chief Executive Officer
of Anika Therapeutics. “With this approval, MONOVISC is poised to
become the most widely available single-injection viscosupplement
in the world and will be a key driver in growing global market
share.”
The global expansion of MONOVISC is an important international
orthobiologics revenue driver for Anika, and Australia and New
Zealand represent a large and growing market opportunity. Anika has
a multi-year, exclusive distribution agreement with Surgical
Specialties Pty. Ltd. to market MONOVISC in Australia and New
Zealand. Established in 2006, Surgical Specialties is an
independent distributor of innovative medical devices, with a team
of over 70 people in sales offices throughout Australia and New
Zealand. Surgical Specialties is an ideal partner for Anika with
its established relationships with leading orthopaedic surgeons and
sports physicians, a successful track record, and a highly skilled
and dedicated national sales force.
About MONOVISC
MONOVISC is Anika’s next-generation HA-based therapy for
treating osteoarthritis that features enhanced durability in a
safe, easy-to-use, single injection regimen. MONOVISC is made from
highly purified, non-animal, natural hyaluronan. Hyaluronan occurs
naturally throughout the body, especially in articular cartilage,
synovial fluid in joints and in the skin. For more information
about MONOVISC, please visit www.monovisc.com.
About Anika Therapeutics, Inc.
Anika Therapeutics, Inc. (NASDAQ: ANIK) is a global,
integrated orthopedic medicines company based in Bedford,
Massachusetts. Anika is committed to improving the lives of
patients with degenerative orthopedic diseases and traumatic
conditions with clinically meaningful therapies along the continuum
of care, from palliative pain management to regenerative cartilage
repair. The Company has over two decades of global expertise
developing, manufacturing, and commercializing more than 20
products based on its proprietary hyaluronic acid (HA)
technology. Anika's orthopedic medicine portfolio
includes ORTHOVISC®, MONOVISC, and CINGAL®, which
alleviate pain and restore joint function by replenishing depleted
HA, and HYALOFAST®, a solid HA-based scaffold to aid cartilage
repair and regeneration. For more information about Anika, please
visit www.anikatherapeutics.com.
Forward-Looking Statements
The statements made in the last sentences of the first and
second paragraphs and first sentence of the third paragraph of this
press release, which are not statements of historical fact, are
forward-looking statements within the meaning of Section 27A of the
Securities Act of 1933, as amended, and Section 21E of the
Securities Exchange Act of 1934, as amended. These statements
include, but are not limited to, those relating to the Company’s
international expansion plans for MONOVISC, the market for the
Company’s products in foreign countries, including Australia and
New Zealand, and the status of MONOVISC as a global revenue driver
for the Company. These statements are based upon the current
beliefs and expectations of the Company's management and are
subject to significant risks, uncertainties, and other factors. The
Company's actual results could differ materially from any
anticipated future results, performance, or achievements described
in the forward-looking statements as a result of a number of
factors including, but not limited to, (i) the Company's ability to
successfully commence and/or complete clinical trials of its
products on a timely basis or at all; (ii) the Company's ability to
obtain pre-clinical or clinical data to support domestic and
international pre-market approval applications, 510(k)
applications, or new drug applications, or to timely file and
receive FDA or other regulatory approvals or clearances of its
products; (iii) that such approvals will not be obtained in a
timely manner or without the need for additional clinical trials,
other testing or regulatory submissions, as applicable; (iv) the
Company's research and product development efforts and their
relative success, including whether we have any meaningful sales of
any new products resulting from such efforts; (v) the cost
effectiveness and efficiency of the Company's clinical studies,
manufacturing operations, and production planning; (vi) the
strength of the economies in which the Company operates or will be
operating, as well as the political stability of any of those
geographic areas; (vii) future determinations by the Company to
allocate resources to products and in directions not presently
contemplated; (viii) the Company's ability to successfully
commercialize its products, in the U.S. and abroad; (ix) the
Company's ability to provide an adequate and timely supply of its
products to its customers; and (x) the Company's ability to achieve
its growth targets. Additional factors and risks are described in
the Company's periodic reports filed with the Securities and
Exchange Commission, and they are available on the SEC's website at
www.sec.gov. Forward-looking statements are made based on
information available to the Company on the date of this press
release, and the Company assumes no obligation to update the
information contained in this press release.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20171016006027/en/
For Investor Inquiries:Anika Therapeutics, Inc.Sylvia Cheung,
781-457-9000Chief Financial OfficerorFor Media Inquiries:Pure
CommunicationsSonal Vasudev,
917-523-1418sonal@purecommunicationsinc.com
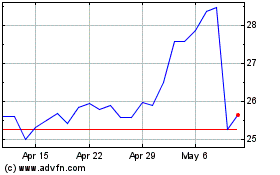
Anika Therapeutics (NASDAQ:ANIK)
Historical Stock Chart
From Apr 2024 to May 2024
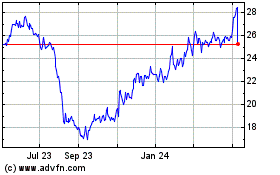
Anika Therapeutics (NASDAQ:ANIK)
Historical Stock Chart
From May 2023 to May 2024