Dynavax Announces Publication of Preclinical Study of TLR9 Agonist in Lung Cancer
04 September 2018 - 8:30PM

Dynavax Technologies Corporation (NASDAQ: DVAX) today announced
publication of a preclinical study demonstrating that inhalation of
a TLR9 agonist can stimulate effective immunity against lung tumors
and complement the actions of PD-1 blockade to generate durable,
systemic anti-tumor immunity. The paper titled Inhaled TLR9 Agonist
Renders Lung Tumors Permissive to PD-1 Blockade by Promoting
Optimal CD4+ and CD8+ T cell Interplay, by Dynavax scientists
M.Gallotta, H. Assi, E. Degagné, S. Kannan, R.Coffman and C.
Guiducci was published in the journal Cancer Research. The study
demonstrated that combining an inhaled TLR9 agonist with systemic
anti-PD-1 led to long-term survival in two different mouse lung
tumor models, mediated by systemic immunity that eradicated tumors
both in the lung and in distal organs. The study further delineated
the distinctive mechanisms of action of these agents in the lung
environment.
Administration of the TLR9 agonist SD-101 into
the lungs of mice with metastatic tumors generated anti-tumor
responses that controlled or eliminated tumor growth in the lungs
as well as in non-treated organs, including liver. Treatment with
SD-101 resulted in ~90% decrease in tumor burden in both the lung
and liver. This led to a significant increase in survival time,
with a majority of mice surviving beyond 90-100 days. Treatment
with SD-101 and anti-PD-1 resulted in a large increase of
tumor-reactive T cells, which were required for anti-tumor
activity. The durable control of liver metastases shows that local
administration of SD-101 to the lung generates an anti-tumor T cell
response capable of controlling tumor growth beyond the lung
itself.
The TLR9 agonist used in these studies was SD-101, Dynavax's
lead clinical candidate currently being developed as an
intratumoral agent in combination with anti-PD-1 therapy in
patients with advanced melanoma and head and neck squamous cell
carcinoma. Unpublished data demonstrates that another TLR9 agonist,
DV281 - optimized for delivery to primary lung tumors and lung
metastases - has equivalent activity in these models. These studies
provide the preclinical rationale for the Phase 1b dose escalation
study of inhaled DV281 currently being conducted by Dynavax in
advanced non-small lung cancer patients (NCT03326752). DV281
and SD-101 stimulate potent Type 1 interferon induction along with
maturation of dendritic cells into effective antigen-presenting
cells. These combined actions lead to the increased numbers of
cytotoxic T cells that are critical for the induction of effective
systemic anti-tumor immunity.
About DynavaxDynavax is a fully-integrated
biopharmaceutical company focused on leveraging the power of the
body's innate and adaptive immune responses through toll-like
receptor (TLR) stimulation. Dynavax discovers and develops novel
vaccines and immuno-oncology therapeutics. The Company’s first
commercial product, HEPLISAV-B® [Hepatitis B Vaccine (Recombinant),
Adjuvanted], was approved by the United States Food and Drug
Administration in November 2017 for prevention of infection caused
by all known subtypes of hepatitis B virus in adults age 18 years
and older. Dynavax's lead immunotherapy product, SD-101, is an
investigational cancer immunotherapeutic currently being evaluated
in Phase 1/2 studies and its second cancer immunotherapeutic,
DV281, is in Phase 1 development. For more information, visit
www.dynavax.com.
Forward Looking StatementsThis press release
contains "forward-looking" statements, including statements
regarding preclinical studies of Dynavax’s TLR9 agonist and the
conduct of clinical trials of SD-101 and DV281. Actual results may
differ materially from those set forth in this press release due to
the risks and uncertainties inherent in our business, including
whether we can timely provide adequate clinical supplies;
initiation, enrollment and completion of clinical trials of the
TLR9 agonist, SD-101 and DV281; the results of clinical trials and
the impact of those results on the initiation or continuation of
subsequent trials and issues arising in the regulatory process; the
ability to successfully develop and commercialize these
investigational compounds; and whether or not Dynavax and parties
with whom we are collaborating may reach any future agreement on
further studies or a more extensive collaboration beyond the
clinical trials contemplated under the existing agreements, as well
as other risks detailed in the "Risk Factors" section of our Annual
Report on Form 10-K for the fiscal year ended December 31, 2017 and
in our Quarterly Report on Form 10-Q for the quarter ended June 30,
2018, as well as discussions of potential risks, uncertainties and
other important factors in our other filings with the U.S.
Securities and Exchange Commission. We undertake no obligation to
revise or update information herein to reflect events or
circumstances in the future, even if new information becomes
available. Information on Dynavax's website at www.dynavax.com is
not incorporated by reference in our current periodic reports with
the SEC.
Contact: Ryan SpencerVP, Corporate Strategy
& Communications510.665.4618
Dynavax Technologies (NASDAQ:DVAX)
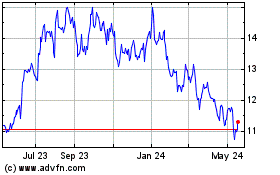
Historical Stock Chart
From Apr 2024 to May 2024
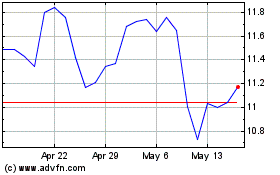
Dynavax Technologies (NASDAQ:DVAX)
Historical Stock Chart
From May 2023 to May 2024