-Includes highlights of safety and efficacy
data for ZURAMPIC® (lesinurad) from extension studies-
-Additional presentations elucidate the
potential long-term health consequences of gout-
Ironwood Pharmaceuticals, Inc. (NASDAQ: IRWD) today
announced a series of oral and poster presentations to be presented
at the upcoming American College of Rheumatology (ACR) Annual
Meeting in Washington, D.C. from November 11 to 16, 2016.
The poster presentations include data from two lesinurad
extension studies that enrolled patients from the pivotal Phase III
CLEAR1, CLEAR2 and CRYSTAL trials, as well as a pooled analysis of
renal safety from pivotal Phase III and extension studies of
lesinurad, and an integrated safety study of lesinurad’s three
pivotal trials and extension studies. Results from the CLEAR1
pivotal trial were published in the August 2016 issue of Arthritis
& Rheumatology.
Other presentations will address the potential long-term health
consequences of gout with respect to such topics as heart failure,
cardiometabolic risk, and progression of chronic kidney
disease.
The titles and scheduled times of the presentations are as
follows:
Safety and Efficacy of Lesinurad:Examination of Serum
Uric Acid (sUA) Lowering and Safety With Extended Lesinurad +
Allopurinol Treatment in Subjects With Gout (CLEAR Extension)
(abstract #208, poster), to be presented during the Metabolic and
Crystal Arthropathies Poster Session I: Clinical Practice on
Sunday, Nov. 13, 2016, 9:00 a.m. – 11:00 a.m. Eastern Time, by
Kenneth G. Saag, M.D., M.Sc., University of Alabama at
Birmingham.
Clinical Response of Tophus and Flares to Extended Use of
Lesinurad in Combination With a Xanthine Oxidase Inhibitor in
Patients With Gout (CLEAR/CRYSTAL Extension) (abstract #209,
poster), to be presented during the Metabolic and Crystal
Arthropathies Poster Session I: Clinical Practice on Sunday, Nov.
13, 2016, 9:00 a.m. – 11:00 a.m. Eastern Time, by Thomas Bardin,
M.D., Lariboisière Hospital, Paris, France.
Renal Safety of Lesinurad: A Pooled Analysis of Phase III and
Extension Studies (abstract #206, poster), to be presented during
the Metabolic and Crystal Arthropathies Poster Session I: Clinical
Practice on Sunday, Nov. 13, 2016, 9:00 a.m. – 11:00 a.m. Eastern
Time, by Robert Terkeltaub, M.D., University of California, San
Diego.
Integrated Safety of Lesinurad, A Novel Uric Acid Reabsorption
Inhibitor for the Treatment of Gout (abstract #207, poster), to be
presented during the Metabolic and Crystal Arthropathies Poster
Session I: Clinical Practice on Sunday, Nov. 13, 2016, 9:00 a.m. –
11:00 a.m. Eastern Time, by Michael A. Becker, M.D., University of
Chicago.
Potential Long-Term Health Consequences of Gout:Rate of
Hospitalization for Heart Failure Is Lower in Patients with
Controlled Gout Versus Uncontrolled Gout (abstract #1242, poster),
to be presented during the Health Services Research Poster Session
II on Monday, Nov. 14, 2016, 9:00 a.m. – 11:00 a.m. Eastern Time,
by Robert Morlock, Ph.D., Your Care Choice, Ann Arbor, MI.
Cardiometabolic Risk and Subclinical Urate Deposits in Patients
with Symptomatic Hyperuricemia and Metabolic Syndrome (abstract
#2293, poster), to be presented during the Innate Immunity and
Rheumatic Disease Poster Session II: Epidemiology and Mechanisms of
Disease on Tuesday, Nov. 15, 2016, 9:00 a.m.-11:00 a.m. Eastern
Time, by Seoyoung C. Kim, M.D., Sc.D., MSCE, Brigham and Women's
Hospital and Harvard Medical School.
Association of Gout with Risk of Advanced Chronic Kidney Disease
(abstract #3188, oral), to be presented during the Epidemiology and
Public Health Oral Session III: Psoriatic Arthritis and More on
Wednesday, Nov. 16, 2016, 11:00 a.m. – 12:30 p.m. Eastern Time, by
Austin Stack, M.D., M.Sc., FRCPI, University Hospital Limerick
& Health Research Institute, University of Limerick, Limerick,
Ireland.
Disease State and Mechanism of Action Studies:Presence of
Monosodium Urate Crystals by Dual-Energy Computed Tomography in
Gout Patients Treated with Allopurinol (abstract #219, poster), to
be presented during the Metabolic and Crystal Arthropathies Poster
Session I: Clinical Practice on Sunday, Nov. 13, 2016, 9:00 a.m. –
11:00 a.m. Eastern Time, by Nicola Dalbeth, MBChB, M.D., FRACP,
University of Auckland, Auckland, New Zealand.
Evidence of Phospho-Degron Regulating Expression of Urate
Secretory Transporter ABCG2 (abstract #2274, poster), to be
presented during the Metabolic and Crystal Arthropathies Poster
Session II: Epidemiology and Mechanisms of Disease on Tuesday, Nov.
15, 2016, 9:00 a.m. – 11:00 a.m. Eastern Time, by Alexis Hofherr,
M.D., Ph.D., University of Freiburg Medical Center, Freiburg im
Breisgau, Germany.
Fructose Amplifies Inflammatory Potential in Human Monocytic
Cells Via Reduction of AMP-Activated Protein Kinase Activity
(abstract #2268, poster), to be presented during the Innate
Immunity and Rheumatic Disease Poster Session II on Tuesday, Nov.
15, 2016, 9:00 a.m. – 11:00 a.m. Eastern Time, by Xihua Cao, Ph.D.,
Veterans Medical Research Foundation (VMRF), San Diego,
California.
Health Economics Studies:Accuracy of HumaSens-plus
Point-of-Care Uric Acid Meter Using Capillary Blood Obtained by
Fingertip Puncture (abstract #218, poster), to be presented during
the Metabolic and Crystal Arthropathies Poster Session I: Clinical
Practice on Sunday, Nov. 13, 2016, 9:00 a.m. - 11:00 a.m. Eastern
Time, by Stephanie Fabre, M.D., M.Sc., Lariboisière Hospital,
Paris, France.
Comparing the Burden of Illness of Patients with Tophaceous and
Non-Tophaceous Gout in France, Germany, Italy, Spain, UK, and USA
(abstract #226, poster), to be presented during the Metabolic and
Crystal Arthropathies Poster Session I: Clinical Practice on
Sunday, Nov. 13, 2016, 9:00 a.m. – 11:00 a.m. Eastern Time, by Puja
Khanna, M.D., M.P.H., University of Michigan.
Relationship Between Patient and Disease Factors and Severity of
Gout in a Real-World Population (abstract #229, poster), to be
presented during the Metabolic and Crystal Arthropathies Poster
Session I: Clinical Practice on Sunday, Nov. 13, 2016, 9:00 a.m. –
11:00 a.m. Eastern Time, by Michael Pillinger, M.D., New York
University.
Health Economics of Uncontrolled Gout in the United States: A
Systematic Literature Review (abstract #2241, poster), to be
presented during the Health Services Research - Poster Session III
on Tuesday, Nov. 15, 2016, 9:00 a.m. – 11:00 a.m. Eastern Time, by
Shaum Kabadi, Ph.D., M.P.H., AstraZeneca.
Development and Pilot Testing of an Online Educational Tool for
Gout Patients — Mygoutcare® (abstract #3130, oral), to be presented
during the Metabolic and Crystal Arthropathies Oral Session II:
Clinical Practice on Wednesday, Nov. 16, 2016, 9:00 a.m. - 10:30
a.m. Eastern Time, by Puja Khanna, M.D., M.P.H., University of
Michigan.
Serum Uric Acid Testing Practices Over Five Years Among Incident
Gout Cases (abstract #1225, poster), to be presented during the
Health Services Research – Poster Session II on Monday, Nov. 14,
2016, 9:00 a.m. - 11:00 a.m. Eastern Time, by Dena Jaffe, Ph.D.,
Kantar Health, Tel Aviv, Israel.
About Hyperuricemia and GoutGout is a highly symptomatic
and painful form of inflammatory arthritis affecting an estimated
eight million people in the U.S. It is caused by an underlying
metabolic disorder, hyperuricemia - high levels of uric acid in the
blood - and can lead to painful flares, characterized by
excruciating pain, inflammation, swelling and tenderness in one or
more joints. Gout is commonly hereditary and not only a lifestyle
disease. While diet and lifestyle changes are important in managing
gout and its comorbidities, they are often not enough to get
patient serum uric acid (sUA) levels to target.
Approximately four million patients are treated with a xanthine
oxidase inhibitor (XOI), either allopurinol or febuxostat, for gout
in the U.S. Of these, an estimated two million patients are
uncontrolled and are not achieving target serum uric acid (sUA)
levels <6 mg/dL as recommended by the American College of
Rheumatology, despite treatment with an XOI alone. These patients
continue to suffer from flares despite treatment with an XOI alone,
and may face serious long-term consequences that can result from
having uncontrolled sUA levels.
About ZURAMPIC® (lesinurad) 200 mg
tabletsZURAMPIC® (lesinurad) is a URAT1 inhibitor approved by
the FDA for use in combination with a xanthine oxidase inhibitor
(XOI) for the treatment of hyperuricemia associated with gout in
patients who have not achieved target serum uric acid levels (sUA)
with an XOI alone. ZURAMPIC is not recommended for the treatment of
asymptomatic hyperuricemia and should not be used as a monotherapy.
XOIs reduce the production of uric acid; ZURAMPIC increases renal
excretion of uric acid by selectively inhibiting the action of
URAT1, the UA transporter responsible for the majority of renal UA
reabsorption. The dual-mechanism combination of ZURAMPIC plus an
XOI (allopurinol or febuxostat) can address both inefficient
excretion and overproduction of UA, thereby lowering sUA levels.
The safety and efficacy of ZURAMPIC were established in three Phase
III clinical trials that evaluated a once-daily dose of ZURAMPIC in
combination with the XOI allopurinol or febuxostat compared to XOI
alone. Visit www.zurampic.com for more information about
ZURAMPIC.
Important Safety Information
WARNING: RISK OF ACUTE RENAL FAILURE
MORE COMMON WHEN USED
WITHOUT A XANTHINE OXIDASE INHIBITOR
(XOI)
-- Acute renal failure has occurred
with ZURAMPIC and was more common when ZURAMPIC was given
alone
-- ZURAMPIC should be used in
combination with an XOI
Contraindications:
- Severe renal impairment (eCLcr less
than 30 mL/min), end-stage renal disease, kidney transplant
recipients, or patients on dialysis
- Tumor lysis syndrome or Lesch-Nyhan
syndrome
Warnings and Precautions:
- Renal events: Adverse reactions
related to renal function have occurred after initiating ZURAMPIC.
A higher incidence was observed at the 400-mg dose, with the
highest incidence occurring with monotherapy use. Monitor renal
function at initiation and during therapy with ZURAMPIC,
particularly in patients with eCLcr below 60 mL/min or with serum
creatinine elevations 1.5 to 2 times the pre-treatment value, and
evaluate for signs and symptoms of acute uric acid nephropathy.
Interrupt treatment with ZURAMPIC if serum creatinine is elevated
to greater than 2 times the pre-treatment value or if there are
symptoms that may indicate acute uric acid nephropathy. ZURAMPIC
should not be restarted without another explanation for the serum
creatinine abnormalities. ZURAMPIC should not be initiated in
patients with an eCLcr less than 45 mL/min.
- Cardiovascular events: In
clinical trials, major adverse cardiovascular events (defined as
cardiovascular deaths, non-fatal myocardial infarctions, or
non-fatal strokes) were observed with ZURAMPIC. A causal
relationship has not been established.
Adverse Reactions:
- Most common adverse reactions with
ZURAMPIC (in combination with an XOI and more frequently than on an
XOI alone) were headache, influenza, blood creatinine increased,
and gastroesophageal reflux disease
Indication and Limitations of Use for ZURAMPIC:ZURAMPIC
is a URAT1 inhibitor indicated in combination with an XOI for the
treatment of hyperuricemia associated with gout in patients who
have not achieved target serum uric acid levels with an XOI
alone.
- ZURAMPIC is not recommended for the
treatment of asymptomatic hyperuricemia
- ZURAMPIC should not be used as
monotherapy
Please see full Prescribing Information, including Boxed
WARNING, http://www.azpicentral.com/zurampic/zurampic.pdf.
About Ironwood PharmaceuticalsIronwood
Pharmaceuticals (NASDAQ: IRWD) is a commercial biotechnology
company focused on creating medicines that make a difference for
patients, building value for our fellow shareholders, and
empowering our passionate team. We are advancing a pipeline of
innovative medicines in areas of significant unmet need, including
irritable bowel syndrome with constipation (IBS-C)/chronic
idiopathic constipation (CIC), uncontrolled gout, refractory
gastroesophageal reflux disease, and vascular and fibrotic
diseases. We discovered, developed and are commercializing
linaclotide, the U.S. branded prescription market leader in the
IBS-C/CIC category, and we are applying our proven R&D and
commercial capabilities to advance multiple internally-developed
and externally-accessed product opportunities. Ironwood was founded
in 1998 and is headquartered in Cambridge, Mass. For more
information, please visit
www.ironwoodpharma.com or www.twitter.com/ironwoodpharma;
information that may be important to investors will be routinely
posted in both these locations.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20161025005376/en/
Ironwood Pharmaceuticals, Inc.Media RelationsTrista Morrison,
617-374-5095Director, Corporate
Communicationstmorrison@ironwoodpharma.comorInvestor
RelationsMeredith Kaya, 617-374-5082Director, Investor
Relationsmkaya@ironwoodpharma.com
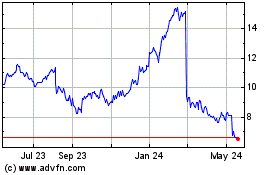
Ironwood Pharmaceuticals (NASDAQ:IRWD)
Historical Stock Chart
From Mar 2024 to May 2024
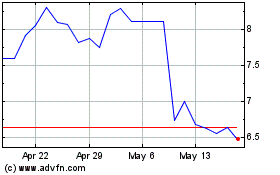
Ironwood Pharmaceuticals (NASDAQ:IRWD)
Historical Stock Chart
From May 2023 to May 2024