ADDING MULTIMEDIA Lucid Diagnostics Appoints Suman Verma MD, PhD as Chief Scientific Officer
22 October 2021 - 3:59AM
Business Wire
Lucid Diagnostics Inc. (Nasdaq: LUCD) (“Lucid”) a
commercial-stage, cancer prevention medical diagnostics company,
and subsidiary of PAVmed Inc. (Nasdaq: PAVM, PAVMZ) (“PAVmed”),
today announced the appointment of highly accomplished molecular
biologist Suman M. Verma, M.D., Ph.D. as Lucid’s Chief Scientific
Officer and PAVmed’s VP, Molecular Diagnostics.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20211021005428/en/
Dr. Suman Verma appointed Chief
Scientific Officer of Lucid Diagnostics (Photo: Business Wire)
“I am very pleased to welcome Dr. Verma to the Lucid Diagnostics
team,” said Lishan Aklog M.D., Lucid’s Chairman and Chief Executive
Officer. “In her role as VP of Genomic Services at our CLIA
laboratory partner, ResearchDx, Dr. Verma played a central role in
transferring the EsoGuard® Esophageal DNA Test from the Case
Western Reserve University research laboratory to a commercial
laboratory setting, completing the CLIA validation process, and
launching EsoGuard as a commercially available Laboratory Developed
Test. We are so fortunate to have someone with such deep experience
and expertise in cutting-edge molecular diagnostics techniques and
technology, as well as intimate knowledge of all aspects of the
EsoGuard assay, to serve as our Chief Scientific Officer.”
“I am excited to join this remarkable company and team, and I
look forward to contributing to Lucid’s important mission of
preventing deadly esophageal cancer,” said Dr. Verma. “I look
forward to working with my Lucid colleagues, as well as our
industry and academic partners to advance the underlying science of
our assay, continually improve its performance, and expand our
portfolio of molecular diagnostic tools in esophageal disease and
beyond.”
Dr. Verma most recently served as VP, Research & Development
at Irvine, CA-based Bridge Diagnostics, a National Molecular
Laboratory focused on infectious disease and women’s health. Prior
to that, over the previous decade, she served in various research
scientist and senior executive roles at Irvine, CA-based
ResearchDx, Inc. a leading contract diagnostics organization (CDO)
for the biopharmaceutical and diagnostic industries and long-time
CLIA laboratory partner of Lucid Diagnostics. As VP, Genomic
Services she led the development and implementation of numerous
molecular diagnostic assays using the full spectrum of cutting edge
molecular biologic techniques and technologies. Dr. Verma earned
her Ph.D. in Molecular Biology and completed a post-doctoral
fellowship at the University of California Irvine, during which she
performed and published on important research in breast cancer
biology and targets for therapeutic intervention in breast cancer.
Prior to immigrating to the U.S., Dr. Verma earned her medical
degree at Maharishi Dayanand University in Rohtak, India, completed
her internal medicine training at Civil hospital, Ballabgarh,
India, and served as a staff physician active in clinical research
in Faridabad, India.
About Lucid Diagnostics
Lucid Diagnostics Inc. (Nasdaq: LUCD) is a commercial-stage,
cancer prevention medical diagnostics company, and subsidiary of
PAVmed Inc. (Nasdaq: PAVM). Lucid is focused on the millions of
patients with gastroesophageal disease (GERD), also known as
chronic heartburn, who are at risk of developing esophageal
precancer and cancer. Lucid’s EsoGuard® Esophageal DNA Test,
performed on samples collected in a brief noninvasive office
procedure with its EsoCheck® Esophageal Cell Collection Device, is
the first and only commercially available diagnostic test capable
of serving as a widespread screening tool to prevent cancer and
cancer deaths through early detection of esophageal precancer in
at-risk GERD patients. EsoGuard is commercialized in the U.S. as a
Laboratory Developed Test (LDT). EsoCheck is commercialized in the
U.S. as a 510(k)-cleared esophageal cell collection device.
EsoGuard, used with EsoCheck, was granted FDA Breakthrough Device
designation and is the subject of two large, actively enrolling,
international multicenter clinical trials to support FDA PMA
approval. Lucid is building a network of Lucid Test Centers where
at-risk GERD patients can undergo the EsoCheck procedure for
EsoGuard testing.
Forward-Looking Statements
This press release includes forward-looking statements.
Forward-looking statements are any statements that are not
historical facts. Such forward-looking statements, which are based
upon the current beliefs and expectations of Lucid’s management,
are subject to risks and uncertainties, which could cause actual
results to differ from the forward-looking statements. Risks and
uncertainties that may cause such differences include, among other
things, volatility in the price of Lucid’s common stock; general
economic and market conditions; the uncertainties inherent in
research and development, including the cost and time required
advance Lucid’s products to regulatory submission; whether
regulatory authorities will be satisfied with the design of and
results from Lucid’s clinical and preclinical studies; whether and
when Lucid’s products are cleared by regulatory authorities; market
acceptance of Lucid’s products once cleared and commercialized;
Lucid’s ability to raise additional funding as needed; and other
competitive developments. In addition, Lucid has been monitoring
the COVID-19 pandemic and the pandemic’s impact on Lucid’s
businesses. Lucid expects the significance of the COVID-19
pandemic, including the extent of its effect on its financial and
operational results, to be dictated by, among other things, the
success of efforts to contain the pandemic and the impact of such
efforts on Lucid’s businesses. All of these factors are difficult
or impossible to predict accurately and many of them are beyond
Lucid’s control. In addition, new risks and uncertainties may arise
from time to time and are difficult to predict. For a further list
and description of these and other important risks and
uncertainties that may affect Lucid’s future operations, see
Lucid’s registration statement on Form S-1 filed with the
Securities and Exchange Commission. Lucid disclaims any intention
or obligation to publicly update or revise any forward-looking
statement to reflect any change in its expectations or in events,
conditions, or circumstances on which those expectations may be
based, or that may affect the likelihood that actual results will
differ from those contained in the forward-looking statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20211021005428/en/
Investors Lisa DeScenza
LaVoieHealthScience (617) 351-0243
ldescenza@lavoiehealthscience.com
Media Kristi Bruno
LaVoieHealthScience (617) 865-3940
PAVmed@lavoiehealthscience.com
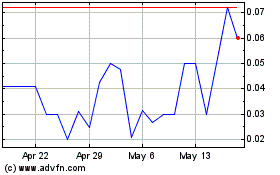
PAVmed (NASDAQ:PAVMZ)
Historical Stock Chart
From Apr 2024 to May 2024
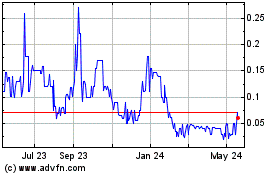
PAVmed (NASDAQ:PAVMZ)
Historical Stock Chart
From May 2023 to May 2024