Simulations Plus Forms Scientific Advisory Board for RENAsym Consortium
16 March 2021 - 11:30PM
Business Wire
RENAsym SAB to Provide Insights and Guidance
for Development of Kidney Injury Software
Simulations Plus, Inc. (Nasdaq: SLP), a leading provider of
modeling and simulation software and services for pharmaceutical
safety and efficacy, today announced that its DILIsym Services
(DSS) division has formed a Scientific Advisory Board (SAB) to
provide guidance and direction for development of its RENAsym
quantitative systems toxicology (QST) kidney injury software
platform.
The SAB has been formed within the RENAsym Consortium, and
consists of the following experts:
- Dr. Lauren Aleksunes, Professor of Pharmacology and Toxicology
at the Rutgers University Ernest Mario School of Pharmacy;
- Dr. Zheng Dong, Regents’ Professor and Leon H. Charbonnier
Endowed Chair at the Medical College of Georgia of Augusta
University and the Director of Research & Development at the
Charlie Norwood VA Medical Center;
- Dr. K. Melissa Hallow, Associate Professor at the University of
Georgia School of Chemical, Materials, and Biomedical
Engineering;
- Dr. Ravindra Mehta, Professor at the University of California
at San Diego School of Medicine;
- Dr. Frank D. Sistare, Former Scientific Associate Vice
President at Merck Research Laboratories and Captain in the U.S.
Public Health Service at the Food and Drug Administration’s Center
for Drug Evaluation and Research;
- Dr. Paul B. Watkins, RENAsym and DILI-sim SAB Chair, Howard Q.
Ferguson Distinguished Professor and Director of the Institute for
Drug Safety Sciences at the University of North Carolina.
For the past 10 years, DSS has coordinated the DILI-sim
Initiative, which is a public-private partnership that has guided
development of the DILIsym® software package. The RENAsym
Consortium was recently launched in a similar fashion for the
development of the RENAsym® software program, which uses properties
of drugs to predict their risk of causing kidney injury in
patients. RENAsym version 1A will be released in the latter half of
2021 for drug developers to use in-house or engage via consulting.
The combined liver and kidney safety capabilities of DILIsym and
RENAsym will allow DSS to address a larger portion of the safety
issues facing drug developers.
Dr. Paul B. Watkins, chair of the Scientific Advisory Board of
the RENAsym Consortium, remarked: “Given the tremendous success of
the DILIsym® software in predicting liver injury due to drugs, many
stakeholders urged the team to take on kidney toxicity and we are
delighted to see this happening. The members of the Scientific
Advisory Board are internationally recognized scientists with
expertise spanning basic kidney biology, novel new tests to detect
kidney injury, and the various ways drugs can harm kidneys in
patients. I am very excited to be working with this team.”
Newly appointed SAB member, Dr. Frank Sistare, commented: “I am
honored to be part of this effort. Drug safety toolboxes are
expanding in areas of region-specific translational kidney safety
biomarkers, advanced in vitro kidney models, and mechanistic
molecular tissue biomarkers. It makes perfect sense to work
collaboratively to expand mathematical modeling capabilities that
can pull these data streams together to provide more accurate
perspectives on kidney effects at target clinical exposures and
improve drug development decision making.”
Dr. Brett A. Howell, president of DILIsym Services, added, “All
of our new RENAsym SAB members are extremely knowledgeable and
distinguished scientific leaders. Our establishment of this new SAB
under the RENAsym Consortium umbrella is just another example of
our commitment to top level science within our products and
services.”
About Simulations Plus, Inc.
Simulations Plus, Inc., is a leading provider of modeling and
simulation software and consulting services supporting drug
discovery, development research, and regulatory submissions. With
our subsidiaries, Cognigen, DILIsym Services, and Lixoft, we offer
solutions which bridge machine learning, physiologically based
pharmacokinetics, quantitative systems pharmacology/toxicology, and
population PK/PD modeling approaches. Our technology is licensed
and applied by major pharmaceutical, biotechnology, chemical,
consumer goods companies and regulatory agencies worldwide. For
more information, visit our website at www.simulations-plus.com.
Follow us on Twitter | Read our Environmental, Social, and
Governance (ESG) Report.
The DILIsym Services division of Simulations Plus has developed
DILIsym® and NAFLDsym® QSP software, and is developing IPFsym™ and
RENAsym™ QSP software, to provide the pharmaceutical industry with
the tools and resources to efficiently develop safe and effective
drug therapies. DILIsym and RENAsym are designed to address
drug-induced liver injury (DILI) and drug-induced acute kidney
injury, respectively. NAFLDsym and IPFsym are designed for target
or compound evaluation of therapeutic efficacy in nonalcoholic
fatty liver disease (NAFLD or NASH) and idiopathic pulmonary
fibrosis (IPF), respectively. More information is available on the
company’s web page.
Safe Harbor Statement Under the Private Securities Litigation
Reform Act of 1995 – With the exception of historical
information, the matters discussed in this press release are
forward-looking statements that involve a number of risks and
uncertainties. Words like “believe,” “expect,” and “anticipate”
mean that these are our best estimates as of this writing, but that
there can be no assurances that expected or anticipated results or
events will actually take place, so our actual future results could
differ significantly from those statements. Factors that could
cause or contribute to such differences include, but are not
limited to: our ability to maintain our competitive advantages,
acceptance of new software and improved versions of our existing
software by our customers, the general economics of the
pharmaceutical industry, our ability to finance growth, our ability
to continue to attract and retain highly qualified technical staff,
our ability to identify and close acquisitions on terms favorable
to the Company, and a sustainable market. Further information on
our risk factors is contained in our quarterly and annual reports
and filed with the U.S. Securities and Exchange Commission.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210316005399/en/
Simulations Plus Investor Relations
Ms. Renee Bouche 661-723-7723 renee@simulations-plus.com
Hayden IR Mr. Cameron Donahue
651-653-1854 slp@haydenir.com
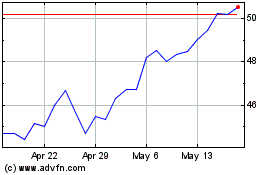
Simulations Plus (NASDAQ:SLP)
Historical Stock Chart
From Apr 2024 to May 2024
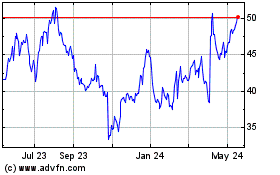
Simulations Plus (NASDAQ:SLP)
Historical Stock Chart
From May 2023 to May 2024