- Results from Study 0170 show a benefit in study patients
with multiple system atrophy (MSA)
- Company beginning discussions with potential
strategic partners and planning health
authority interactions to expedite ampreloxetine as a
possible treatment option for patients with symptomatic
nOH
- Company remains focused on respiratory therapeutics, value
creation for shareholders and reiterates plan to be sustainably
cash-flow positive by the second half of the year
DUBLIN,
April 4, 2022
/PRNewswire/ -- Theravance Biopharma, Inc. ("Theravance
Biopharma" or the "Company") (NASDAQ: TBPH) today announced
results from the second Phase 3 study, Study 0170 (n=128 out of
n=154 planned), assessing the durability of clinical effect of
ampreloxetine compared to placebo for the treatment of symptomatic
nOH. Study 0170 was a 22-week Phase 3 study comprised of a
16-week open-label period followed by a 6-week double-blind,
placebo-controlled, randomized withdrawal period. The primary
endpoint of treatment failure at week 6 of the randomized
withdrawal period was defined as a worsening of both Orthostatic
Hypotension Symptom Assessment Scale (OHSA) question #1 and Patient
Global Impression of Severity (PGI-S) scores by 1.0 point.
The primary endpoint was not statistically significant for the
overall population of patients which included patients with
Parkinson's disease (PD), pure autonomic failure (PAF) and MSA
(odds ratio=0.6; p-value=0.196). The odds ratio suggests that
patients receiving ampreloxetine had a 40% reduction in the
odds of treatment failure compared to placebo.
The pre-specified subgroup analysis by disease type suggests the
benefit seen in patients receiving ampreloxetine was largely driven
by MSA patients (n=40). An odds ratio of 0.28 (95% CI: 0.05, 1.22)
was observed in MSA patients indicating a 72% reduction in the odds
of treatment failure with ampreloxetine compared to placebo. The
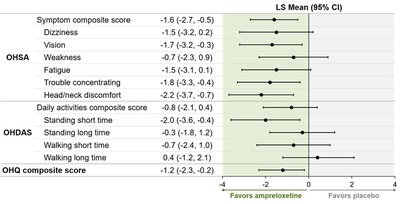
benefit to MSA patients was observed in multiple endpoints
including OHSA composite, Orthostatic Hypotension Daily Activities
Scale (OHDAS) composite, Orthostatic Hypotension Questionnaire
(OHQ) composite and OHSA #1 (see figure below). Notably, patients
withdrawn to placebo had a clinically relevant decrease in standing
blood pressure; there was no decrease for patients remaining on
ampreloxetine. While the same benefit was not apparent in patients
with PD or PAF, the Company continues to analyze the data to better
understand this observation. Throughout the study, there was no
indication of worsening of supine hypertension based on 24-hour
monitoring. Data suggest that ampreloxetine was well-tolerated and
no new safety signals were identified.
"MSA is a devastating and debilitating disease with no
effective disease modifying treatment. There is an urgency to treat
MSA patients suffering with nOH due to the impact on quality of
life and the extreme caregiver burden. Ampreloxetine appears to
improve a narrow, but critically important group of symptoms
related to blood pressure control, and along with the safety
profile, may represent a potential therapy for MSA patients,"
said Roy Freeman, MBChB, Professor
of Neurology, Director, Center for Autonomic and Peripheral Nerve
Disorders, Beth Israel Deaconess Medical Center, who assisted in
the design and interpretation of the study.
"At Theravance Biopharma, we are guided by patient outcomes.
Given the clear unmet need for MSA patients suffering from
symptomatic nOH, we are engaging potential partners and planning
health authority interactions to determine a path forward in hopes
of expediting ampreloxetine as a possible treatment
option for people with MSA," said Rick E
Winningham, Chief Executive Officer, Theravance Biopharma.
"Concurrently, we are executing against our business plan and
remain disciplined in capital allocation, maximizing shareholder
value and re-iterate our goal to be sustainably cash-flow positive
by the second half of this year."
Disclosure: Dr. Freeman is a consultant serving as an
advisor for drug development and clinical trial design for
Theravance Biopharma.
Study 0170 (REDWOOD) Ampreloxetine Phase 3
Results
About the Phase 3 Study
Study 0170 (NCT03829657) was a 22-week Phase 3 study comprised of a
16-week open-label period and a 6-week double-blind,
placebo-controlled, randomized withdrawal period. The primary
endpoint of treatment failure at week 6 was defined as a worsening
of both Orthostatic Hypotension Symptom Assessment Scale (OHSA)
question #1 and Patient Global Impression of Severity (PGI-S)
scores by 1.0 point. After Study 0169 did not meet its primary
endpoint, the Company took actions to close out the ongoing
clinical program including Study 0170. The study was more than 80%
enrolled (n=128/154 planned) despite stopping early.
About Symptomatic nOH
Neurogenic orthostatic
hypotension (nOH) is a rare disorder defined as a sustained
orthostatic fall in systolic blood pressure (SBP) of ≥ 20 mm Hg or
diastolic blood pressure (DBP) of ≥ 10 mm Hg within three minutes
of standing. Severely affected patients are unable to stand for
more than a few seconds because of their decrease in blood
pressure, leading to cerebral hypoperfusion and syncope. A
debilitating condition, nOH results in a range of symptoms
including dizziness, lightheadedness, fainting, fatigue, blurry
vision, weakness, trouble concentrating, and head and neck pain.
nOH is caused by autonomic nervous system malfunction and is
associated with several underlying medical conditions including
multiple system atrophy (MSA), pure autonomic failure (PAF), and
Parkinson's disease (PD).
About Multiple System Atrophy (MSA)
MSA (formerly Shy-Drager syndrome) is a rare, rapidly
progressive, severely debilitating, and fatal neurodegenerative
disease that leads to death within a median of seven to 10 years
after the onset of symptoms. MSA shares many Parkinson's
disease-like symptoms, such as slow movement, rigid muscles and
poor balance. The primary sign of multiple system atrophy is
postural (orthostatic) hypotension. A person with MSA can also
develop dangerously high blood pressure levels while lying down
(supine hypertension). Other manifestations include: urinary and
bowel dysfunction, constipation, incontinence, sweating
abnormalities, sleep disorders, sexual dysfunction, cardiovascular
problems and psychiatric problems. The most common causes of death
in MSA are infection and cardiopulmonary complications. Currently,
MSA patients receive only symptomatic and palliative therapies as
there are no disease-modifying treatments and no cure.
About Ampreloxetine
Ampreloxetine (TD-9855) is an investigational, Theravance
Biopharma-discovered, potent, long-acting, oral, once-daily
norepinephrine reuptake inhibitor in development for the treatment
of symptomatic neurogenic orthostatic hypotension (nOH) with patent
protection until 2037 in the US. In addition to the Phase 3
clinical program, Theravance Biopharma has conducted a
comprehensive non-clinical and clinical pharmacology program.
Ampreloxetine has been evaluated in over 800 patients in clinical
studies of fibromyalgia, attention deficit hyperactivity disorder
(ADHD) and symptomatic nOH.
About Theravance Biopharma
Theravance Biopharma, Inc. is a biopharmaceutical company
primarily focused on the discovery, development and
commercialization of respiratory medicines. Its core purpose is to
create medicines that help improve the lives of patients suffering
from respiratory illness.
In pursuit of its purpose, Theravance Biopharma leverages
decades of respiratory expertise to discover and develop
transformational medicines that make a difference. These efforts
have led to the development of FDA-approved
YUPELRI® (revefenacin) inhalation solution
indicated for the maintenance treatment of patients with chronic
obstructive pulmonary disease (COPD). Its respiratory pipeline of
internally discovered programs is targeted to address significant
patient respiratory needs.
Theravance Biopharma has an economic interest in potential
future payments from Glaxo Group Limited or one of its
affiliates (GSK) pursuant to its agreements with Innoviva, Inc.
relating to certain programs, including TRELEGY.
For more information, please visit www.theravance.com.
THERAVANCE BIOPHARMA®, THERAVANCE®, and
the Cross/Star logo are registered trademarks of
the Theravance Biopharma group of companies (in
the US and certain other countries).
YUPELRI® is a registered trademark of Mylan
Specialty L.P., a Viatris Company. Trademarks, trade names or
service marks of other companies appearing on this press release
are the property of their respective owners.
Forward-Looking Statements
This press release contains certain "forward-looking" statements as
that term is defined in the Private Securities Litigation Reform
Act of 1995 regarding, among other things, statements relating to
goals, plans, objectives, expectations and future events.
Theravance Biopharma intends such forward-looking statements
to be covered by the safe harbor provisions for forward-looking
statements contained in Section 21E of the Securities Exchange Act
of 1934 and the Private Securities Litigation Reform Act of 1995.
Examples of such statements include statements relating to: the
Company's goals, designs, strategies, plans and objectives, ability
to provide value to shareholders, the Company's regulatory
strategies, the potential characteristics, benefits and mechanisms
of action of the Company's product and product candidates, the
Company's expectations for product candidates through development
and the market for products being commercialized, the Company's
expectations regarding its allocation of resources, potential
regulatory actions and commercialization, product sales or profit
share revenue and the Company's expectations for its expenses,
excluding share-based compensation and other financial results.
These statements are based on the current estimates and assumptions
of the management of Theravance Biopharma as of the date
of the press release and are subject to risks, uncertainties,
changes in circumstances, assumptions and other factors that may
cause the actual results of Theravance Biopharma to be
materially different from those reflected in the forward-looking
statements. Important factors that could cause actual results to
differ materially from those indicated by such forward-looking
statements include, among others, risks related to:
additional future analysis of the data resulting from our clinical
trial(s), the potential that results from clinical or non-clinical
studies indicate the Company's compounds, products or product
candidates are unsafe, ineffective or not differentiated, risks of
decisions from regulatory authorities that are unfavorable to the
Company, delays or failure to achieve and maintain regulatory
approvals for product candidates, risks of collaborating with or
relying on third parties to discover, develop, manufacture and
commercialize products, and ability to retain key personnel,
the impact of the Company's restructuring actions on its employees,
partners and others. In addition, while we expect the effects of
COVID-19 to continue to adversely impact our business operations
and financial results, the extent of the impact on our ability to
generate revenue from YUPELRI® (revefenacin), our
clinical development programs, and the value of and market for our
ordinary shares, will depend on future developments that are highly
uncertain and cannot be predicted with confidence at this time.
Other risks affecting Theravance Biopharma are in the
Company's Form 10-K filed with the SEC on February
28, 2022, and other periodic reports filed with the SEC.
In addition to the risks described above and in Theravance
Biopharma's filings with the SEC, other unknown or
unpredictable factors also could affect Theravance
Biopharma's results. No forward-looking statements can be
guaranteed, and actual results may differ materially from such
statements. Given these uncertainties, you should not place undue
reliance on these forward-looking statements. Theravance
Biopharma assumes no obligation to update its forward-looking
statements on account of new information, future events or
otherwise, except as required by law.
Contact: Gail B. Cohen
Corporate Communications / 917-214-6603
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/theravance-biopharma-inc-announces-results-from-study-0170-a-second-phase-3-study-of-ampreloxetine-in-patients-with-symptomatic-neurogenic-orthostatic-hypotension-noh-301517024.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/theravance-biopharma-inc-announces-results-from-study-0170-a-second-phase-3-study-of-ampreloxetine-in-patients-with-symptomatic-neurogenic-orthostatic-hypotension-noh-301517024.html
SOURCE Theravance Biopharma, Inc.