Regulatory Expert Henrietta Ukwu, M.D. Appointed Novavax Chief Regulatory and Quality Officer
16 January 2021 - 12:00AM

Novavax, Inc. (Nasdaq: NVAX), a late-stage biotechnology company
developing next-generation vaccines for serious infectious
diseases, today announced the appointment of Henrietta Ukwu, M.D.,
FACP, FRAPS, to the position of Senior Vice President, Chief
Regulatory and Quality Officer. In this newly created role, Dr.
Ukwu will lead global regulatory strategy and execution as well as
the quality assurance function for the company’s development-stage
vaccine candidates, including its recombinant protein-based
COVID-19 candidate vaccine, NVX-CoV2373.
“Dr. Ukwu brings extensive regulatory affairs
and quality expertise and leadership experience at precisely the
right time for Novavax, as we advance toward data readouts and
regulatory submissions in 2021,” said Stanley C. Erck, President
and Chief Executive Officer, Novavax. “We welcome her medical
perspective and intricate knowledge of the regulatory process at
this extraordinary time for the company and the race against the
global pandemic.”
Dr. Ukwu most recently served as Chief
Regulatory Officer and Global Head of Quality and Regulatory at
Otsuka Pharmaceuticals. While there, she globalized and led the
regulatory and quality organizations with a focus on compliance
excellence, inspection readiness, engagement with health
authorities and regulatory agencies and successful outcomes. Prior
to Otsuka, she held senior leadership roles at PPD as Senior Vice
President and Head of Global Regulatory Affairs, where she had
responsibilities for regulatory services across all PPD lines of
business; Wyeth Pharmaceuticals, Inc. (Pfizer), where she was Vice
President, Regulatory Affairs, with responsibility that spanned
vaccines, biologics and pharmaceuticals across all regions; and at
Merck & Co., Inc., as Head of Vaccine Regulatory Development,
Vice President of Global Regulatory Affairs and Global Regulatory
Policy. Dr. Ukwu has led approval of many products, including for
Varivax®, a chickenpox varicella vaccine, and Crixivan®,
(indinavir), an HIV protease inhibitor that significantly impacted
the treatment paradigm of HIV/AIDS.
Dr. Ukwu’s significant contributions to
medicine, science and the pharmaceutical industry include
developing and enriching the regulatory and quality profession, as
well as serving as a mentor to others who have progressed into
senior leadership positions. These contributions have been
recognized with numerous prestigious awards.
Dr. Ukwu earned her medical and surgical degrees
from the University of Jos School of Medicine in Nigeria. She
completed her residency in internal medicine at Baptist Hospital,
and a fellowship in infectious diseases at Vanderbilt University,
both in Nashville, Tennessee. She is a Fellow of both the American
College of Physicians (FACP) and the Regulatory Affairs
Professional Society (FRAPS) and is a pioneer professor at the
Graduate School of Pharmacy at Temple University in Ambler,
Pennsylvania.
About NovavaxNovavax,
Inc. (Nasdaq: NVAX) is a late-stage biotechnology company that
promotes improved health globally through the discovery,
development and commercialization of innovative vaccines to prevent
serious infectious diseases. The company’s proprietary recombinant
technology platform combines the power and speed of genetic
engineering to efficiently produce highly immunogenic nanoparticles
designed to address urgent global health needs. Novavax is
conducting late-stage clinical trials for NVX-CoV2373, its vaccine
candidate against SARS-CoV-2, the virus that causes COVID-19.
NanoFlu™, its quadrivalent influenza nanoparticle vaccine, met all
primary objectives in its pivotal Phase 3 clinical trial in older
adults and will be advanced for regulatory submission. Both vaccine
candidates incorporate Novavax’ proprietary saponin-based Matrix-M™
adjuvant to enhance the immune response and stimulate high levels
of neutralizing antibodies.
For more information, visit www.novavax.com and
connect with us on Twitter and LinkedIn.
Novavax Forward Looking
StatementsStatements herein relating to the future of
Novavax and the ongoing development of its vaccine and adjuvant
products are forward-looking statements. Novavax cautions that
these forward-looking statements are subject to numerous risks and
uncertainties, which could cause actual results to differ
materially from those expressed or implied by such statements.
These risks and uncertainties include those identified under the
heading “Risk Factors” in the Novavax Annual Report on Form 10-K
for the year ended December 31, 2019, and Quarterly Report on Form
10-Q for the period ended September 30, 2020, as filed with the
Securities and Exchange Commission (SEC). We caution investors not
to place considerable reliance on forward-looking statements
contained in this press release. You are encouraged to read our
filings with the SEC, available at sec.gov, for a discussion of
these and other risks and uncertainties. The forward-looking
statements in this press release speak only as of the date of this
document, and we undertake no obligation to update or revise any of
the statements. Our business is subject to substantial risks and
uncertainties, including those referenced above. Investors,
potential investors, and others should consider these risks and
uncertainties.
Contacts:
Investors
Erika Trahan
ir@novavax.com
240-268-2022
Media
Edna Kaplan
media@novavax.com
617-974-8659
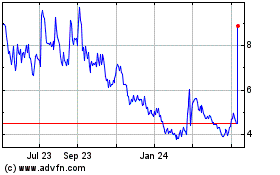
Novavax (NASDAQ:NVAX)
Historical Stock Chart
From Mar 2024 to Apr 2024
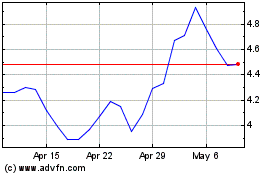
Novavax (NASDAQ:NVAX)
Historical Stock Chart
From Apr 2023 to Apr 2024