Registration No. 333-_______
UNITED
STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C.
20549
FORM S-8
REGISTRATION STATEMENT Under
THE SECURITIES ACT OF 1933
Protalix
BioTherapeutics, Inc.
(Exact name of registrant as specified
in its Charter)
| Florida |
|
65-0643773 |
| (State or other jurisdiction |
|
(I.R.S. Employer |
| of incorporation or organization) |
|
Identification Number) |
2 Snunit Street
Science Park
P.O. Box 455
Carmiel, Israel 20100
972-4-988-9488
(Address of principal executive offices)
Protalix BioTherapeutics, Inc. 2006
Stock Incentive Plan, as amended November 10, 2014
(Full title of plan)
Moshe Manor
President and Chief Executive Officer
2 Snunit Street, Science Park
P.O. Box 455
Carmiel 20100, Israel
972-4-988-9488
CT Corporation System
111 Eighth Avenue
New York, NY 10011
Tel: (212) 894-8400
(Name, address,
including zip code, and telephone number, including area code, of agent for service)
With a Copy to:
James R. Tanenbaum, Esq.
Morrison & Foerster LLP
1290 Avenue of the Americas
New York, New York 10104
(212) 468-8000
CALCULATION OF REGISTRATION FEE
Title
of each class of securities
to be registered | |
Amount
to be registered
(1) | | |
Proposed
maximum offering
price | | |
Proposed
maximum aggregate
offering price | | |
Amount
of registration
fee | |
| Common stock, par value $0.001 per share | |
| 2,500,000 | | |
$ | 2.13 | (2) | |
$ | 5,325,000 | (2) | |
$ | 618.77 | |
(1) This
Registration Statement also registers additional securities to be offered or issued upon adjustments or changes made to registered
securities by reason of any stock splits, stock dividends or similar transactions as permitted by Rule 416(a) and Rule 416(b)
under the Securities Act of 1933, as amended, or the Securities Act.
(2) Estimated
pursuant to Rule 457(h) and Rule 457(c) solely for purposes of calculating the aggregate offering price and the amount of the
registration fee based upon the average of the high and low prices reported for the shares on the NYSE MKT on May 5, 2015.
EXPLANATORY NOTE
Protalix BioTherapeutics, Inc., previously
filed a registration statement on Form S-8 (SEC File No. 333-148983) with the Securities and Exchange Commission, or the Commission,
in connection with the registration of an aggregate of 9,741,655 shares of our common stock to be issued under the Protalix BioTherapeutics,
Inc. 2006 Stock Incentive Plan, or the Plan, and subsequently filed a second registration statement on Form S-8 (SEC File No.
333-182677) with the Commission in connection with the registration of an aggregate of 2,500,000 additional shares of our common
stock to be issued under the Plan.
Pursuant to General Instruction E of Form S-8,
we are filing this registration statement on Form S-8 solely to register an additional 2,500,000 shares of our common stock,
par value $.001 per share, available for issuance under the Plan. This increase was approved by our Board of Directors and our
shareholders. Pursuant to Instruction E, the contents of the Registration Statement on Form S-8 (SEC File No. 333-148983
and 333-182677), including without limitation periodic reports that we filed, or will file, after this Registration Statement
to maintain current information about our company, are hereby incorporated by reference into this Registration Statement pursuant
to General Instruction E of Form S-8, with the exception of Items 3 and 8 of Part II of such prior Registration Statement,
each of which is amended and restated in its entirety herein.
This Registration Statement contains two
parts:
The first part contains a Reoffer Prospectus
prepared in accordance with the requirements of Part I of Form S-3. The Reoffer Prospectus covers reoffers and resales of
shares of our common stock, par value $.001 per share, by certain of our executive officers (as such term is defined under Rule 405
of the Securities Act of 1933, as amended (the ‘‘Securities Act’’)) with respect to options granted prior
to the date hereof (3,847,000 shares) pursuant the Protalix BioTherapeutics, Inc. 2006 Stock Incentive Plan, as amended. All such
individuals are listed on the selling securityholder table set forth herein.
The second part contains ‘‘Information
Required in the Registration Statement’’ prepared in accordance with the requirements of Part II of Form S-8 with
respect to the authorized issuance, as of the date hereof and subsequent to the date hereof, equity awards granted under the Protalix
BioTherapeutics, Inc. 2006 Stock Incentive Plan that relate to, in the aggregate, 13,841,655 shares of our common stock, including
the 3,847,000 shares of our common stock being offered under the Reoffer Prospectus. The Reoffer Prospectus does not contain all
of the information included in the Registration Statement, certain items of which are contained in exhibits to the Registration
Statement as permitted by the rules and regulations of the Securities and Exchange Commission (the ‘‘Commission’’).
Statements contained in the Reoffer Prospectus as to the contents of any agreement, instrument or other document referred to therein
are not necessarily complete. With respect to each such agreement, instrument or other document filed as an exhibit to the Registration
Statement, we refer you to the exhibit for a more complete description of the matter involved, and each such statement shall be
deemed qualified in its entirety by this reference.
PART I
INFORMATION REQUIRED IN THE SECTION 10(a)
PROSPECTUS
Item 1. Plan Information.
The documents containing the information
specified in Part I (plan and registrant information) will be delivered in accordance with Rule 428(b)(1) under the Securities
Act. Such documents are not required to be, and are not, filed with the Commission, either as part of this Registration Statement
or as prospectuses or prospectus supplements pursuant to Rule 424 under the Securities Act. These documents, and the documents
incorporated by reference in this Registration Statement pursuant to Item 3 of Part II of this Form S-8, taken together, constitute
a prospectus that meets the requirements of Section 10(a) of the Securities Act.
Item 2. Registrant Information
and Employee Plan Annual Information.
Upon written or oral request, any of the
documents incorporated by reference in Item 3 of Part II of this Registration Statement, which are also incorporated by reference
in the Section 10(a) prospectus, other documents required to be delivered to eligible participants pursuant to Rule 428(b), or
additional information about the Plan, will be available without charge by contacting the Corporate Secretary, Protalix BioTherapeutics,
Inc., 2 Snunit Street, Science Park, P.O. Box 455, Carmiel 20100, Israel, Telephone: +972-4-988-9488.
REOFFER PROSPECTUS
3,847,000
Shares

Common Stock
Issued or issuable under certain awards
granted under the
Protalix BioTherapeutics, Inc. 2006 Stock Incentive Plan, as amended
This Reoffer Prospectus relates to the
public resale, from time to time, of an aggregate of 3,847,000 shares of our common stock by certain securityholders identified
herein in the section entitled ‘‘Selling Securityholders’’. Such shares have been or may be acquired in
connection with awards granted under the Protalix BioTherapeutics, Inc. 2006 Stock Incentive Plan. You should read this prospectus
carefully before you invest in our common stock.
We will not receive any proceeds from
the sale by the selling securityholders of the shares covered by this Reoffer Prospectus.
We have not entered into any underwriting
arrangements in connection with the sale of the shares covered by this Reoffer Prospectus. The selling securityholders identified
in this Reoffer Prospectus, or their pledgees, donees, transferees or other successors-in-interest, may offer the shares covered
by this Reoffer Prospectus from time to time through public or private transactions at prevailing market prices, at prices related
to prevailing market prices or at privately negotiated prices.
Our common stock is listed on the NYSE
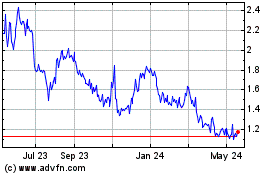
MKT under the symbol “PLX” and on the Tel Aviv Stock Exchange under the symbol “PLX.” On May 6, 2015,
the last reported sale price of our common stock was $2.14 per share on the NYSE MKT and NIS 8.08 per share on the Tel Aviv Stock
Exchange.
Investing in our common stock involves
a high degree of risk. See “Risk Factors” beginning on page 6 of this prospectus and the documents incorporated
by reference into this prospectus.
Neither the Securities and Exchange
Commission, the Israeli Securities Authority nor any state securities commission has approved or disapproved of these securities
or determined if the prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this Prospectus is May 7, 2015
TABLE OF CONTENTS
Except where the context otherwise requires, the terms,
“we,” “us,” “our” or “the Company,” refer to the business of Protalix BioTherapeutics,
Inc. and its consolidated subsidiaries, and “Protalix” or “Protalix Ltd.” refers to the business of Protalix
Ltd., our wholly-owned subsidiary and sole operating unit.
CAUTIONARY STATEMENT
REGARDING FORWARD-LOOKING STATEMENTS
The statements set forth and incorporated by reference in this
prospectus, which are not historical, constitute “forward-looking statements” within the meanings of Section 27A of
the Securities Act of 1933, as amended, or the Securities Act, and Section 21E of the Securities Exchange Act of 1934, as amended,
or the Exchange Act, including statements regarding expectations, beliefs, intentions or strategies for the future. When used
in this report, the terms “anticipate,” “believe,” “estimate,” “expect,” “can,”
“continue,” “could,” “intend,” “may,” “plan,” “potential,”
“predict,” “project,” “should,” “will,” “would” and words or phrases
of similar import, as they relate to our company or our subsidiaries or our management, are intended to identify forward-looking
statements. We intend that all forward-looking statements be subject to the safe-harbor provisions of the Private Securities Litigation
Reform Act of 1995. These forward-looking statements are only predictions and reflect our views as of the date they are made with
respect to future events and financial performance, and we undertake no obligation to update or revise, nor do we have a policy
of updating or revising, any forward-looking statement to reflect events or circumstances after the date on which the statement
is made or to reflect the occurrence of unanticipated events, except as may be required under applicable law. Forward-looking
statements are subject to many risks and uncertainties that could cause our actual results to differ materially from any future
results expressed or implied by the forward-looking statements.
Examples of the risks and uncertainties include, but are not
limited to, the following:
| · | risks
relating to the compliance by Fundação Oswaldo Cruz, or Fiocruz, an arm
of the Brazilian Ministry of Health, with its purchase obligations under our supply and
technology transfer agreement which may result in the termination of such agreement which
may have a material adverse effect on our company; |
| · | risks
related to the commercialization efforts for taliglucerase alfa in the United States,
Israel, Brazil, Canada, Australia and other countries; |
| · | risks
related to the supply of drug product pursuant to our supply arrangement with Fiocruz; |
| · | the
risk of significant delays in the commercial introduction of taliglucerase alfa in the
United States, Brazil, Israel, Canada, Australia and other markets as planned; |
| · | risks
related to the acceptance and use of taliglucerase alfa or any of our product candidates,
if approved, by physicians, patients and third-party payors; |
| · | the
risk that we will not be able to develop a successful sales and marketing organization
for taliglucerase alfa in Israel, or for any other product candidate, in a timely manner,
if at all; |
| · | failure
or delay in the commencement or completion of our preclinical studies and clinical trials
which may be caused by several factors, including: unforeseen safety issues; determination
of dosing issues; lack of effectiveness during clinical trials; slower than expected
rates of patient recruitment; inability to monitor patients adequately during or after
treatment; inability or unwillingness of medical investigators and institutional review
boards to follow our clinical protocols; or lack of sufficient funding to finance our
clinical trials; |
| · | the
risk that the results of our clinical trials will not support the applicable claims of
safety or efficacy, that our product candidates will not have the desired effects or
include undesirable side effects or other unexpected characteristics; |
| · | our
dependence on performance by third party providers of services and supplies, including
without limitation, clinical trial services; |
| · | delays
in the approval or the potential rejection of any application filed with or submitted
to the regulatory authorities reviewing taliglucerase alfa outside of the United States,
Israel, Brazil, Canada, Australia and other countries in which taliglucerase alfa is
already approved; |
| · | our
ability to establish and maintain strategic license, collaboration and distribution arrangements,
and to manage our relationships with Pfizer Inc., Fiocruz and any other collaborator,
distributor or partner; |
| · | risks
relating to our ability to make scheduled payments of the principal of, to pay interest
on or to refinance our 2018 convertible notes, or any other indebtedness; |
| · | risks
relating to our ability to finance our research programs, the expansion of our manufacturing
capabilities and the ongoing costs in the case of delays in regulatory approvals for
taliglucerase alfa outside of the United States, Israel, Brazil, Canada, Australia and
other countries in which taliglucerase alfa is already approved; |
| · | delays
in our preparation and filing of applications for regulatory approval of our other product
candidates in the United States, the European Union and elsewhere; |
| · | our
expectations with respect to the potential commercial value of our product and product
candidates; |
| · | the
risk that products that are competitive to our product candidates may be granted orphan
drug status in certain territories and, therefore, will be subject to potential marketing
and commercialization restrictions; |
| · | the
impact of development of competing therapies and/or technologies by other companies; |
| · | any
lack of progress of our research and development activities and our clinical activities
with respect to any product candidate; |
| · | the
inherent risks and uncertainties in developing the types of drug platforms and products
we are developing; |
| · | potential
product liability risks, and risks of securing adequate levels of product liability and
clinical trial insurance coverage; |
| · | the
possibility of infringing a third party’s patents or other intellectual property
rights; |
| · | the
uncertainty of obtaining patents covering our products and processes and in successfully
enforcing our intellectual property rights against third parties; |
| · | risks
relating to changes in healthcare laws, rule s and regulations in the United States or
elsewhere; and |
| · | the
possible disruption of our operations due to terrorist activities and armed conflict,
including as a result of the disruption of the operations of regulatory authorities,
our subsidiaries, our manufacturing facilities and our customers, suppliers, distributors,
collaborative partners, licensees and clinical trial sites. |
Companies in the pharmaceutical and biotechnology industries
have suffered significant setbacks in advanced or late-stage clinical trials, even after obtaining promising earlier trial results
or preliminary findings for such clinical trials. Even if favorable testing data is generated from clinical trials of a drug product,
the U.S. Food and Drug Administration or foreign regulatory authorities may not accept or approve a marketing application filed
by a pharmaceutical or biotechnology company for the drug product.
These forward-looking statements reflect our current views
with respect to future events and are based on assumptions and subject to risks and uncertainties. Given these uncertainties,
you should not place undue reliance on these forward-looking statements. These and other risks and uncertainties are detailed
under the heading “Risk Factors” herein and in our filings with the
Securities and Exchange Commission, or the Commission, incorporated by reference in this prospectus.
OUR BUSINESS
The Commission allows us to ‘‘incorporate
by reference’’ certain information that we file with the Commission, which means that we can disclose important information
to you by referring you to those documents. The information incorporated by reference is considered to be part of this prospectus,
and information that we file later with the Commission will update automatically, supplement and/or supersede the information
disclosed in this prospectus. Any statement contained in a document incorporated or deemed to be incorporated by reference in
this prospectus shall be deemed to be modified or superseded for purposes of this prospectus to the extent that a statement contained
in this prospectus or in any other document that also is or is deemed to be incorporated by reference in this prospectus modifies
or supersedes such statement. Any such statement so modified or superseded shall not be deemed, except as so modified or superseded,
to constitute a part of this prospectus. You should read the following summary together with the more detailed information regarding
our company, our common stock and our financial statements and notes to those statements appearing elsewhere in this prospectus
or incorporated herein by reference.
We are a biopharmaceutical company focused
on the development and commercialization of recombinant therapeutic proteins based on our proprietary ProCellEx®
protein expression system, or ProCellEx. Using our ProCellEx system, we are developing a pipeline of proprietary, clinically superior
versions of recombinant therapeutic proteins that primarily target large, established pharmaceutical markets and that in most
cases rely upon known biological mechanisms of action. Our initial commercial focus has been on complex therapeutic proteins,
including proteins for the treatment of genetic disorders, such as Gaucher disease and Fabry disease. With our experience, and
having successfully developed ElelysoTM, our first drug product, we believe ProCellEx will enable us to develop additional
proprietary recombinant proteins that are therapeutically superior to existing recombinant proteins currently marketed for the
same indications. We are now also applying the unique properties of our ProCellEx system for the oral delivery of therapeutic
proteins.
On May 1, 2012, the U.S. Food and Drug Administration,
or the FDA, approved for sale our first commercial product, taliglucerase alfa for injection, which is being marketed in the United
States and Israel under the brand name Elelyso, as an enzyme replacement therapy, or ERT, for the long-term treatment of adult
patients with a confirmed diagnosis of type 1 Gaucher disease. Subsequently, taliglucerase alfa was approved by the Brazilian
National Health Surveillance Agency (Agencia Nacional de Vigilancia Sanitaria, or ANVISA) in March 2013, and by the Israeli Ministry
of Health, or the Israeli MOH, in September 2012. It has also been approved by other regulatory agencies for other countries.
Taliglucerase alfa is being marketed under the name UplysoTM in Brazil and certain other Latin American countries.
In August 2014, the FDA approved Elelyso
for injection for pediatric patients and the Israeli MOH approved the pediatric indication in January 2015. Prior to the U.S.
pediatric approval, Elelyso was approved for pediatric indications in Australia and Canada but in no other jurisdiction. In September
2014, CONITEC, the National Commission for Incorporation of Technologies in Brazil’s Unified Healthcare System, announced
that it had decided to give a positive funding recommendation for Uplyso in the treatment of adult patients with types 1 and 3
Gaucher disease, and established that Uplyso will be the first choice for treatment for new adult Gaucher patients in Brazil.
Since May 2012, taliglucerase alfa has been
marketed in the United States by Pfizer Inc., or Pfizer, our commercialization partner, as provided in the exclusive license and
supply agreement by and between Protalix Ltd., our wholly-owned subsidiary, and Pfizer, which we refer to as the Pfizer Agreement.
We granted Pfizer an exclusive, worldwide license to develop and commercialize taliglucerase alfa under the Pfizer Agreement,
but we retained those rights in Israel, and later in Brazil. We have agreed to a specific allocation between Protalix Ltd. and
Pfizer of the responsibilities for the continued development efforts for taliglucerase alfa outside of Israel and Brazil. Protalix
Ltd. has been marketing taliglucerase alfa in Israel since 2013 and in Brazil since January 2014.
On
June 18, 2013, we entered into a Supply and Technology Transfer Agreement, or the Brazil Agreement, with Fiocruz, for taliglucerase
alfa. The agreement became effective in January 2014. The technology transfer is designed to be completed in four stages and is
intended to transfer to Fiocruz the capacity and skills required for the Brazilian government to construct its own manufacturing
facility, at its sole expense, and to produce a sustainable, high-quality, and cost-effective supply of taliglucerase alfa. The
initial term of the technology transfer is seven years. Under the agreement, Fiocruz committed to purchase at least approximately
$40 million worth of taliglucerase alfa during the first two years of the term. Since the agreement went into effect, we have
recorded revenues of approximately $3.5 million for sales of taliglucerase alfa to Fiocruz in 2014, and revenues of approximately
$1.7 million for sales of taliglucerase alfa to Fiocruz during the three months ended March 31, 2015.
In subsequent years, Fiocruz is required to purchase at least approximately $40 million worth of taliglucerase alfa per year.
We are not required to complete the final stage of the technology transfer until Fiocruz purchases at least approximately $280
million worth of taliglucerase alfa.
Under the agreement, if Fiocruz does not
purchase an additional approximately $30 million of Uplyso by July 31, 2015, we will have the right to terminate the agreement,
in which case all rights to the technology that were transferred to Fiocruz will be returned to our company.
The Brazil Agreement may be extended for
an additional five-year term, as needed, to complete the technology transfer. All of the terms of the arrangement, including the
minimum annual purchases, will apply during the additional term. Upon completion of the technology transfer, and subject to Fiocruz
receiving approval from ANVISA to manufacture taliglucerase alfa in its facility in Brazil, the agreement will enter into the
final term and will remain in effect until our last patent in Brazil expires. During such period, Fiocruz will be the sole provider
of this important treatment option for Gaucher patients in Brazil and shall pay us a single-digit royalty on net sales.
To facilitate the arrangement with Fiocruz,
we and Pfizer agreed to an amendment of our exclusive license and supply agreement, which amendment provides for the transfer
of the commercialization and other rights to taliglucerase alfa in Brazil back to us. As consideration for the transfer of the
commercialization and supply rights, we agreed to pay Pfizer a maximum amount of approximately $12.5 million from its net profits
(as defined in the license and supply agreement) per year. Pfizer has also agreed to perform certain transitional services in
Brazil on our behalf in connection with the supply of taliglucerase alfa to Fiocruz.
We will pay a fee equal to 5% of the net
proceeds generated in Brazil to our agent for services provided in assisting us complete the Brazil Agreement pursuant to an agency
agreement between us and the agent. The agency agreement will remain in effect with respect to the Brazil Agreement until the
termination thereof.
We are cooperating with Pfizer to obtain
marketing approval for taliglucerase alfa in additional countries and jurisdictions. In addition to those countries in which taliglucerase
alfa has been approved, marketing authorization applications have been filed in other countries.
Currently, patients are being treated with
taliglucerase alfa on a commercial basis mainly in the United States, Brazil, Israel and Chile.
In addition to taliglucerase alfa, we are
developing an innovative product pipeline using our ProCellEx protein expression system. Our product pipeline currently includes,
among other candidates:
(1)
PRX-102, or alpha-GAL-A, a therapeutic protein candidate for the
treatment of Fabry disease, a rare, genetic lysosomal disorder in humans, currently in an ongoing phase I/II clinical trial. We
expect to report the second interim efficacy and safety results for the second dose group of 1 mg/kg of the trial during the third
quarter of 2015 and to report final efficacy and safety results for the 0.2mg, 1 mg and 2mg/kg dose groups of the trial during
the fourth quarter of 2015.
(2)
PRX-106, our oral antiTNF product candidate which is being developed
as an orally-delivered anti inflammatory treatment using plant cells as a natural capsule for the expressed protein, currently
in an ongoing phase I clinical trial. We expect to initiate a proof
of concept efficacy study around year end.
(3) PRX-110, a proprietary plant cell recombinant
human Deoxyribonuclease 1, or DNase, under development for the treatment of cystic fibrosis, to be administered by inhalation.
We expect to initiate a proof of concept efficacy study around year end.
(4) PRX-112, an orally administered glucocerebrosidase
enzyme for the treatment of Gaucher patients utilizing oral delivery of the recombinant GCD enzyme produced and encapsulated within
carrot cells. PRX-102 has been the subject of successful proof of concept clinical trials, as described below, and we intend to
focus our efforts on a new formulation of the treatment during 2015 before proceeding to more advanced clinical trials.
Except
for the rights to commercialize taliglucerase alfa worldwide (other than Brazil and Israel), which we licensed to Pfizer, we hold
the worldwide commercialization rights to all of our proprietary development candidates.
We have built an internal marketing team designed to serve the Israeli and Brazilian market for taliglucerase alfa and we intend
to establish internal commercialization and marketing teams for our other product candidates in North America, the European Union
and in other significant markets, including Israel, subject to required marketing approvals, as the need arises. In addition,
we continuously evaluate potential strategic marketing partnerships as well as collaboration programs with biotechnology and pharmaceutical
companies and academic research institutes.
RISK FACTORS
Investment in our securities involves
a high degree of risk. Our business, financial condition or results of operations could be adversely affected by any of these
risks. If any of these risks occur, the value our common stock and our other securities may decline. You should carefully consider
the risk factors discussed in this section with the other information included in this prospectus, as well as the discussion set
forth under the caption ‘‘Risk Factors’’ in our Annual Report on Form 10-K, as amended, for the year ended
December 31, 2014, before making your investment decision, as well as those contained in any filing with the Commission subsequent
to the date of the Annual Report. Our business, financial condition or results of operations could be adversely affected by any
of these risks. If any of these risks occur, the value of our common stock could decline.
Risks Related to Our Financial Condition
and Capital Requirements
We currently have no significant product
revenues and may need to raise additional capital to operate our business, which may not be available on favorable terms, or at
all, and which will have a dilutive effect on our shareholders.
To date, we have not generated significant
revenues from product sales and only minimal revenues from research and development services and other fees, other than the milestone
payments we have received in connection with our license and supply agreement with Pfizer. For the years ended December 31, 2014,
2013 and 2012, we had net losses of $29.9 million, $27.8 million and $11.6 million, respectively, primarily as a result of expenses
incurred through a combination of research and development activities and expenses supporting those activities, which includes
share-based compensation expense. Drug development and commercialization is very capital intensive. Our lead product was first
approved for marketing by the FDA in May 2012, by the Israeli MOH in September 2012 and, subsequently, in certain other countries.
We fund all of our operations and capital expenditures from the revenues we generate from sales of taliglucerase alfa supplemented
with our cash on hand, other licensing fees and grants and the net proceeds of any equity or debt offerings. Based on our current
plans and capital resources, we believe that our cash and cash equivalents will be sufficient to enable us to meet our planned
operating needs for at least 12 months. However, changes may occur that could consume our existing capital at a faster rate than
projected, including, among others, the cost and timing of regulatory approvals, changes in the progress of our research and development
efforts and the costs of protecting our intellectual property rights.
We may seek additional financing to implement and fund product
development, preclinical studies and clinical trials for the drugs in our pipeline, as well as additional drug candidates and
other research and development projects. If we are unable to secure additional financing in the future on acceptable terms, or
at all, we may be unable to commence or complete planned preclinical and clinical trials or obtain approval of our drug candidates
from the FDA and other regulatory authorities. In addition, we may be forced to reduce or discontinue product development or product
licensing, reduce or forego sales and marketing efforts and other commercialization activities or forego attractive business opportunities
in order to improve our liquidity and to enable us to continue operations which would have a material adverse effect on our business
and results of operations. Any additional source of financing will likely involve the issuance of our equity securities, which
will have a dilutive effect on our shareholders.
Risks Related to the Commercialization of taliglucerase
alfa
We cannot predict the share in net income we will receive
from Pfizer’s sales of taliglucerase alfa.
Taliglucerase alfa has been approved for marketing in the United
States, Israel, Brazil and other territories. Otherwise, we have no other products approved for marketing. As we have invested
a significant portion of our efforts and financial resources in the development of taliglucerase alfa, our ability to generate
product revenue depends heavily on the successful commercialization of taliglucerase alfa. Under the Pfizer Agreement, Pfizer
holds an exclusive worldwide license to develop and commercialize taliglucerase alfa, except in Israel and Brazil. Sales of taliglucerase
alfa worldwide (except Israel and Brazil) are dependent upon Pfizer’s sales and marketing efforts, which we do not control
and may not be able to effectively influence, and on the actions and decisions of foreign regulatory authorities. Upon the approval
of taliglucerase alfa in additional markets, if at all, Pfizer may experience delays in, or be unable to achieve, the commercial
introduction of taliglucerase alfa in those markets, which would have a material adverse effect on our business, results of operations
and financial condition.
Our future revenues under our collaboration
with Pfizer from Pfizer’s sales of taliglucerase alfa will depend on a number of factors, including:
| · | the
number of Gaucher patients who will be treated with taliglucerase alfa; |
| · | the
willingness of Gaucher patients to switch from other ERTs to taliglucerase alfa; |
| · | competition
from Cerezyme, VPRIV and Cerdelga, and other current or future approved treatments of
Gaucher disease; |
| · | Pfizer’s
efforts under the Pfizer Agreement and the effectiveness of Pfizer’s commercial
strategy and its execution of that strategy, including its pricing strategy and the effectiveness
of its efforts to obtain adequate third-party reimbursements; |
| · | obtaining
marketing approvals and reimbursement from additional regulatory authorities; |
| · | a
continued acceptable safety and efficacy profile of our product candidates following
approval; |
| · | the
successful audit of our facilities by additional regulatory authorities; |
| · | maintaining the
cGMP compliance of our manufacturing facility or establishing manufacturing arrangements
with third parties; and |
| · | the capacity
of physicians and health care providers to provide treatment to Gaucher patients. |
We
cannot accurately predict the amount of revenues we will generate under our collaboration with Pfizer in future periods, if any.
Any failure to commercialize taliglucerase alfa or the experience of significant delays in doing so will have a material
adverse effect on our business, results of operations and financial condition.
The
market share and/or other indicators of market acceptance of taliglucerase alfa may not meet the expectations of investors or
public market analysts, which would have a material adverse effect on our business, results of operations and financial
condition and the market price of our common stock would likely decline.
Fiocruz may not comply with the terms
and conditions of the Supply and Technology Transfer Agreement.
Uplyso was first approved for marketing in Brazil in March
2013. Under our Supply and Technology Transfer Agreement with Fiocruz,
we are not required to complete the final stage of the technology transfer for the production of Uplyso until Fiocruz purchases
at least approximately $280 million worth of Uplyso. However, we do not control and may not be able to effectively influence
Fiocruz’s ability to distribute Uplyso in Brazil. With respect to the first required purchase amount, we have recorded revenues
of approximately $3.5 million for sales of Uplyso to Fiocruz in 2014 and, during the first quarter of 2015, we received a purchase
order for approximately $5.7 million of Uplyso out of which we have delivered approximately $1.7 million of the product.
If Fiocruz fails to comply with the purchase requirements of the Supply and Technology Transfer Agreement, we may terminate
the agreement and market Uplyso in Brazil on our own or through Pfizer. Any failure by Fiocruz to comply with the purchase requirements
of the Supply and Technology Transfer Agreement, or any other material breach by Fiocruz of the agreement, may have a material
adverse effect on our business, results of operations and financial condition.
We
cannot accurately predict the amount of revenues we will generate under our Supply and Technology Transfer with Fiocruz in future
periods, if any. Any failure by Fiocruz to distribute Uplyso in Brazil, or the experience of significant delays in doing
so, may have a material adverse effect on our business, results of operations and financial condition.
If safety issues regarding taliglucerase
alfa that were not known at the time of approval are discovered, or if we or Pfizer fail to comply with continuing U.S. and applicable
foreign regulations, commercialization efforts for taliglucerase alfa could be adversely affected and taliglucerase alfa could
lose its approval or its sales could be suspended.
Drug products remain subject to continuing
regulatory oversight after they are approved for marketing, including the review of additional safety information. Drugs are more
widely used by patients once approved for sale and, therefore, side-effects and other problems may be observed after approval
that were not seen or anticipated, or were not as prevalent or severe, during clinical trials or nonclinical studies. The subsequent
discovery of previously unknown problems with a product could negatively affect commercial sales of the product, result in restrictions
on the product or lead to the withdrawal of the product from the market. The reporting of adverse safety events involving taliglucerase
alfa or public speculation about such events could cause our stock price to decline or experience periods of volatility and may
have a material adverse effect on our business, results of operations and financial condition.
If we or Pfizer fail to comply with applicable
continuing regulatory requirements, we or Pfizer may be subject to fines and/or criminal prosecutions, and taliglucerase may become
subject to suspension or withdrawal of regulatory approval, product recalls and seizures and operating restrictions. In addition,
the manufacturers we and Pfizer engage to produce taliglucerase alfa and the manufacturing facilities in which taliglucerase alfa
is made are subject to periodic review and inspection by the FDA and foreign regulatory authorities. If problems are identified
during the review or inspection of these manufacturers or manufacturing facilities, it could result in our inability to use the
facility to make our product or a determination that inventories are not safe for commercial sale, which may have a material adverse
effect on our business, results of operations and financial condition.
If physicians, patients, third party payors
and others in the medical community do not accept and use taliglucerase alfa, or any of our other product candidates, if approved,
our ability to generate revenue from product sales will be materially impaired.
Physicians and
patients, and other healthcare providers, may not accept and use taliglucerase alfa or any of our other product candidates,
if approved for marketing. Future acceptance and use of taliglucerase alfa or any of our other
product candidates, if approved, will depend upon a number of factors including:
| · | perceptions
by physicians, patients, third party payors and others in the medical community about
the safety and effectiveness of taliglucerase alfa or our other drug candidates; |
| · | the
willingness of the target patient population to try new therapies and of physicians to
prescribe these therapies; |
| · | the
prevalence and severity of any side effects, including any limitations or warnings contained
in our products’ approved labeling; |
| · | pharmacological
benefits of taliglucerase alfa or our other drug candidates relative to competing products
and products under development; |
| · | the
efficacy and potential advantages relative to competing products and products under development; |
| · | relative
convenience and ease of administration; |
| · | effectiveness
of education, marketing and distribution efforts by us and our licensees and distributors,
if any; |
| · | publicity
concerning taliglucerase alfa or our other drug candidates or competing products and
treatments; |
| · | coverage
and reimbursement of our products by third party payors; and |
| · | the
price for our products and competing products. |
Because we expect
sales of taliglucerase alfa to generate substantially all of our product revenues for the foreseeable
future, any lack of market acceptance of taliglucerase alfa would have a material adverse effect on our business, results of operations
and financial condition.
If the market opportunities for taliglucerase
alfa or our other product candidates are smaller than we believe they are, our revenues may be adversely affected and our business
may suffer.
To date, our development efforts have focused
mainly on relatively rare disorders with small patient populations, in particular Gaucher disease and Fabry disease. Currently,
most reported estimates of the prevalence of these diseases are based on studies of small subsets of the population of specific
geographic areas, which are then extrapolated to estimate the prevalence of the diseases in the broader world population. As new
studies are performed, the estimated prevalence of these diseases may change. There can be no assurance that the prevalence of
Gaucher disease or Fabry disease in the study populations, particularly in these newer studies, accurately reflect the prevalence
of these diseases in the broader world population. If the market opportunities for our current product candidates are smaller
than we believe they are, our revenues may be adversely affected and our business may suffer.
Coverage
and reimbursement may not be available for taliglucerase alfa or any of our other product candidates, if approved,
in all territories which could diminish our sales or affect our ability to sell taliglucerase alfa or any other products profitably.
Market acceptance and sales of taliglucerase
alfa or any of our other product candidates, if approved, will depend on worldwide coverage and reimbursement policies. Government
authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which drugs they
will pay for and establish reimbursement levels. Although, to date taliglucerase alfa is covered and reimbursed in all the approved
territories, coverage might not be available for taliglucerase alfa or any of our other product candidates, if approved, in all
territories. Obtaining reimbursement approval for an approved product from governments and other third
party payors is a time consuming and costly process that requires our collaborators or us, as the case may be, to provide supporting
scientific, clinical and cost-effectiveness data for the use of our products, if and when approved, to every payor. We may not
be able to provide data sufficient to gain acceptance with respect to coverage and reimbursement or we might need to conduct post-marketing
studies in order to demonstrate the cost-effectiveness of approved products, if any, to such payors’ satisfaction. Such
studies might require our collaborators or us to commit a significant amount of management time and financial and other resources.
Even if a payor determines that taliglucerase alfa, or any other approved product, if any, is eligible for reimbursement, the
payor may impose coverage limitations that preclude payment for some uses that are approved by the FDA or other regulatory authorities.
In addition, full reimbursement may not be available for high priced products. Moreover, eligibility for coverage does not imply
that any approved product will be reimbursed in all cases or at a rate that allows us to make a profit or even cover our costs.
Also, limited reimbursement amounts may reduce the demand for, or the price of, our product candidates. Except with respect
to taliglucerase alfa, we have not commenced efforts to have our product candidates covered and reimbursed by government or third-party
payors. If coverage and reimbursement are not available or are available only to limited levels, the sales of our products, if
approved may be diminished or we may not be able to sell such products profitably.
We and our collaborating partners may
be subject, directly or indirectly, to federal and state healthcare fraud and abuse and false claims laws and regulations. If
we or our collaborating partners are unable to comply, or have not fully complied, with such laws, we could face substantial penalties.
All marketing activities associated with
taliglucerase alfa in the United States, as well as marketing activities in the United States related to any other products for
which we obtain regulatory approval, if any, will be, directly or indirectly
through our customers, subject to numerous federal and state laws governing the marketing and promotion of pharmaceutical
products in the United States, including, without limitation, the federal
Anti-Kickback Statute, the federal False Claims Act and HIPAA. These laws may adversely impact, among other things, our proposed
sales, marketing and education programs.
The
federal Anti-Kickback Statute prohibits persons from knowingly and willfully soliciting, receiving, offering or paying remuneration,
directly or indirectly, to induce either the referral of an individual, or the furnishing, recommending, or arranging for a good
or service, for which payment may be made under a federal healthcare program, such as the Medicare and Medicaid programs. The
term “remuneration” has been broadly interpreted to include anything of value, including for example, gifts, discounts,
the furnishing of supplies or equipment, credit arrangements, payments of cash, waivers of co-payments and deductibles, ownership
interests and providing anything at less than its fair market value. The reach of the Anti-Kickback Statute was also broadened
by the Patient Protection and Affordable Care Act, as amended by the Health Care and Education
Affordability Reconciliation Act, or the PPACA, which, among other
things, amends the intent requirement of the federal Anti-Kickback Statute and the applicable criminal healthcare fraud statutes
contained within 42 U.S.C. § 1320a-7b, effective March 23, 2010. Pursuant to the statutory amendment, a person or entity
no longer needs to have actual knowledge of this statute or specific intent to violate it in order to have committed a violation.
In addition, PPACA provides that the government may assert that a claim including items or services resulting from a violation
of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act (discussed
below) or the civil monetary penalties statute, which imposes penalties against any person who is determined to have presented
or caused to be presented a claim to a federal health program that the person knows or should know is for an item or service that
was not provided as claimed or is false or fraudulent. The federal Anti-Kickback Statute is broad, and despite a series of narrow
safe harbors, prohibits many arrangements and practices that are lawful in businesses outside of the healthcare industry. Penalties
for violations of the federal Anti-Kickback Statute include criminal penalties and civil sanctions such as fines, imprisonment
and possible exclusion from Medicare, Medicaid and other state or federal healthcare programs. Many states have also adopted laws
similar to the federal Anti-Kickback Statute, some of which apply to the referral of patients for healthcare items or services
reimbursed by any source, not only the Medicare and Medicaid programs, and do not contain identical safe harbors.
The federal False Claims Act imposes liability
on any person who, among other things, knowingly presents, or causes to be presented, a false or fraudulent claim for payment
by a federal healthcare program. The “qui tam” provisions of the False Claims Act allow a private individual to bring
civil actions on behalf of the federal government alleging that the defendant has submitted a false claim to the federal government,
and to share in any monetary recovery. In addition, various states have enacted false claims laws analogous to the False Claims
Act. Many of these state laws apply where a claim is submitted to any third-party payer and not merely a federal healthcare program.
When an entity is determined to have violated the False Claims Act, it may be required to pay up to three times the actual damages
sustained by the government, plus civil penalties of $5,500 to $11,000 for each separate false claim.
The Health Insurance Portability and Accountability
Act of 1996, or HIPAA, created several new federal crimes, including health care fraud, and false statements relating to health
care matters. The health care fraud statute prohibits knowingly and willfully executing a scheme to defraud any health care benefit
program, including private third-party payers. The false statements statute prohibits knowingly and willfully falsifying, concealing
or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery
of or payment for health care benefits, items or services.
We are unable to predict whether we could
be subject to actions under any of these or other fraud and abuse laws, or the impact of such actions. Moreover, to the extent
that taliglucerase alfa, or any of our products, if approved for marketing, will be sold in a foreign country, we and our collaborators,
including Pfizer, may be subject to similar foreign laws and regulations. If we or any of our collaborators are found to be in
violation of any of the laws described above and other applicable state and federal fraud and abuse laws, we may be subject to
penalties, including civil and criminal penalties, damages, fines, exclusion from government healthcare reimbursement programs
and the curtailment or restructuring or our operations, any of which could have a material adverse effect on our business, results
of operations and financial condition.
Risks Related to Our Business
We have a limited operating history which
may limit the ability of investors to make an informed investment decision.
Taliglucerase alfa is our only product with
commercial approvals. The successful commercialization of our drug candidates will require us to perform a variety of functions,
including:
| · | continuing
to perform preclinical development and clinical trials; |
| · | participating
in regulatory approval processes; |
| · | formulating
and manufacturing products; and |
| · | conducting
sales and marketing activities. |
Our operations have been limited to organizing
and staffing our company, acquiring, developing and securing our proprietary technology and undertaking, through third parties,
preclinical trials and clinical trials of our principal drug candidates. To date, we have commenced a phase III clinical trial
in connection with only one drug candidate, taliglucerase alfa, which trial was completed in August 2009. These operations provide
a limited basis for investors to assess our ability to commercialize our drug candidates and whether to invest in our company.
Our strategy, in certain cases, is to
enter into collaboration agreements with third parties to leverage our ProCellEx system to develop product candidates. If we fail
to enter into these agreements or if we or the third parties do not perform under such agreements or terminate or elect to discontinue
the collaboration, it could have a material adverse effect on our revenues.
Our strategy,
in certain cases, is to enter into arrangements with pharmaceutical companies to leverage our ProCellEx system to develop additional
product candidates. Under these arrangements, we may grant to our partners rights to license
and commercialize pharmaceutical products developed under the applicable agreements. Our partners may control key decisions relating
to the development of the products and we may depend on our partners’ expertise and dedication of sufficient resources to
develop and commercialize our product candidates. The rights of our partners limit our flexibility in considering alternatives
for the commercialization of our product candidates. If we or any of our current or future partners breach or terminate the agreements
that make up such arrangements, our partners otherwise fail to conduct their obligations under such arrangements in a timely manner,
there is a dispute about their obligations or if either party terminates the applicable agreement or elects not to continue the
arrangement, we may not enjoy the benefits of the agreements or receive a sufficient amount of royalty or milestone payments from
them, if any, which may have a material adverse effect on our business, results of operations and financial condition.
We have limited experience in selling,
marketing or distributing products and limited internal capability to do so.
We currently have very limited sales, marketing
or distribution capabilities and no experience in building a sales force and distribution capabilities. Pfizer holds an exclusive,
worldwide right to develop and commercialize taliglucerase alfa worldwide, except in Israel and Brazil. If we decide to market
any of our other products directly, if any, we must commit significant financial and managerial resources to develop a marketing
and sales force with technical expertise and with supporting distribution capabilities. Factors that may inhibit our efforts to
commercialize our products directly and without strategic partners include:
| · | the
inability to recruit and retain adequate numbers of effective sales and marketing personnel; |
| · | the
inability of sales personnel to obtain access to an adequate numbers of physicians or
to pursuance them to prescribe our products; |
| · | the
lack of complementary products to be offered by sales personnel, which may put us at
a competitive disadvantage relative to companies with more extensive product lines; and |
| · | unforeseen
costs and expenses associated with creating and sustaining an independent sales and marketing
organization. |
We may not be successful in recruiting or
retaining the sales and marketing personnel necessary to sell any of our products upon approval, if at all, which would have a
material adverse effect on our business, results of operations and financial condition.
We may enter into distribution arrangements
and marketing alliances for certain products and any failure to successfully identify and implement these arrangements on favorable
terms, if at all, may impair our ability to commercialize our product candidates.
While we do sell directly taliglucerase alfa
in Israel and Brazil and we intend to build a sales force to market other product candidates at least in certain regions if approved,
we do not anticipate having the resources in the foreseeable future to develop global sales and marketing capabilities for all
of the products we develop. We may pursue arrangements regarding the sales and marketing and distribution of one or more of our
product candidates, such as our current license and supply agreement with Pfizer for taliglucerase alfa, and our future revenues
may depend, in part, on our ability to enter into and maintain arrangements with other companies having sales, marketing and distribution
capabilities and the ability of such companies to successfully market and sell any such products. Any
failure to enter into such arrangements and marketing alliances on favorable terms, if at all, could delay or impair our ability
to commercialize our product candidates and could increase our costs of commercialization. Any use of distribution arrangements
and marketing alliances to commercialize our product candidates will subject us to a number of risks, including the following:
| · | we
may be required to relinquish important rights to our products or product candidates; |
| · | we
may not be able to control the amount and timing of resources that our distributors or
collaborators may devote to the commercialization of our product candidates; |
| · | our
distributors or collaborators may experience financial difficulties; |
| · | our
distributors or collaborators may not devote sufficient time to the marketing and sales
of our products; and |
| · | business
combinations or significant changes in a collaborator’s business strategy may adversely
affect a collaborator’s willingness or ability to complete its obligations under
any arrangement. |
We may need to enter into additional co-promotion
arrangements with third parties where our own sales force is neither well situated nor large enough to achieve maximum penetration
in the market. We may not be successful in entering into any co-promotion arrangements, and the terms of any co-promotion arrangements
we enter into may not be favorable to us.
Our ProCellEx protein expression system
is based on our proprietary plant cell-based expression technology which has a limited history and any material problems with
the system, which may be unforeseen, may have a material adverse effect on our business, results of operations and financial condition.
Our ProCellEx protein expression system is
based on our proprietary plant cell-based expression technology. The success of our business is dependent upon the successful
development and approval of our product and product candidates produced through this technology. Although taliglucerase alfa is
produced through ProCellEx, the technology remains novel. Accordingly, the technology remains subject
to certain risks. Mammalian cell-based protein expression systems have been used in connection with recombinant therapeutic protein
expression for more than 20 years and are the subject of a wealth of data; in contrast, there is not a significant amount of data
generated regarding plant cell-based protein expression and, accordingly, plant cell-based protein expression systems may be subject
to unknown risks. In addition, the protein glycosilation pattern created by our protein expression system is not identical to
the natural human glycosilation pattern and, although to date clinical data for up to five years of follow-up on taliglucerase
alfa has not demonstrated any sign of any effect, the longer term effect of the protein glycosilation pattern created by our protein
expression system on human patients, if any, is still unknown. Lastly, as our protein expression system is a new technology, we
cannot always rely on existing equipment; rather, there is a need to design custom-made equipment and to generate specific growth
media for the plant cells which may not be available at favorable prices, if at all. Any material problems with the technology
underlying our plant cell-based protein expression system may have a material adverse effect on our business, results of operations
and financial condition.
We currently depend heavily on the success
of taliglucerase alfa, our first commercial product. Any failure to successfully commercialize taliglucerase alfa, or the experience
of significant delays in doing so, will have a material adverse effect on our business, results of operations and financial condition.
We have invested a significant portion of
our efforts and financial resources in the development of taliglucerase alfa. Our ability to generate product revenue, depends
heavily on the successful development and commercialization of taliglucerase alfa. Pfizer holds an exclusive worldwide license
to develop and commercialize taliglucerase alfa, except in Israel and Brazil. The successful commercialization of taliglucerase
alfa will depend on several factors, including the following:
| · | obtaining
marketing approvals and reimbursement from additional regulatory authorities; |
| · | the
successful audit of our facilities by additional regulatory authorities; |
| · | maintaining
the cGMP compliance of our manufacturing facility or establishing manufacturing arrangements
with third parties; |
| · | Pfizer’s
efforts under the Pfizer Agreement; |
| · | Fiocruz’s
activities in Brazil; |
| · | Fiocruz’s
purchasing the minimum quantities under the Technology Transfer Agreement; |
| · | our
marketing efforts in Israel; |
| · | a
continued acceptable safety and efficacy profile of taliglucerase alfa; |
| · | the
availability of reimbursement to patients from healthcare payors for taliglucerase alfa;
and |
| · | other
risks described in these Risk Factors. |
Any failure to successfully commercialize
taliglucerase alfa or the experience of significant delays in doing so will have a material adverse effect on our business, results
of operations and financial condition.
If we are unable to develop and commercialize
our other product candidates, our business will be adversely affected.
A key element of our strategy is to develop
and commercialize a portfolio of new products in addition to taliglucerase alfa. We seek to do so through our internal research
programs and strategic collaborations for the development of new products. Research programs to identify new product candidates
require substantial technical, financial and human resources, whether or not any product candidates are ultimately identified.
Our research programs may initially show promise in identifying potential product candidates, yet fail to yield product candidates
for clinical development for many reasons, including the following:
| · | a
product candidate is not capable of being produced in commercial quantities at an acceptable
cost, or at all; |
| · | a
product candidate may not be accepted by patients, the medical community or third-party
payors; |
| · | competitors
may develop alternatives that render our product candidates obsolete; |
| · | the
research methodology used may not be successful in identifying potential product candidates;
or |
| · | a
product candidate may on further study be shown to have harmful side effects or other
characteristics that indicate it is unlikely to be effective or otherwise does not meet
applicable regulatory approval. |
Any failure to develop or commercialize any
of our other product candidates may have a material adverse effect on our business, results of operations and financial condition.
Clinical trials are very expensive, time-consuming
and difficult to design and implement and may result in unforeseen costs which may have a material adverse effect on our business,
results of operations and financial condition.
Human clinical trials are very expensive
and difficult to design and implement, in part because they are subject to rigorous regulatory requirements. The clinical trial
process is also time-consuming. Other than taliglucerase alfa, all of our other drug candidates are in the clinical, preclinical
or research stages and will take at least several years to complete. Preliminary and initial results from a clinical trial do
not necessarily predict final results, and failure can occur at any stage of the trials. We may encounter problems that cause
us to abandon or repeat preclinical studies or clinical trials. Companies in the pharmaceutical and biotechnology industries have
suffered significant setbacks in advanced clinical trials, even after obtaining promising results in earlier trials. Data obtained
from tests are susceptible to varying interpretations which may delay, limit or prevent regulatory approval. Failure or delay
in the commencement or completion of our clinical trials may be caused by several factors, including:
| · | unforeseen
safety issues; |
| · | determination
of dosing issues; |
| · | lack
of effectiveness during clinical trials; |
| · | slower
than expected rates of patient recruitment; |
| · | inability
to monitor patients adequately during or after treatment; |
| · | inability
or unwillingness of medical investigators and institutional review boards to follow our
clinical protocols; and |
| · | lack
of sufficient funding to finance the clinical trials. |
Any failure or delay in commencement or completion
of any clinical trials may have a material adverse effect on our business, results of operations and financial condition. In addition,
we or the FDA or other regulatory authorities may suspend any clinical trial at any time if it appears that we are exposing participants
in the trial to unacceptable safety or health risks or if the FDA or such other regulatory authorities, as applicable, find deficiencies
in our IND submissions or the conduct of the trial. Any suspension of a clinical trial may have a material adverse effect on our
business, results of operations and financial condition.
If the results of our clinical trials
do not support our claims relating to any drug candidate or if serious side effects are identified, the completion of development
of such drug candidate may be significantly delayed or we may be forced to abandon development altogether, which will significantly
impair our ability to generate product revenues.
The results of our clinical trials with respect
to any drug candidate might not support our claims of safety or efficacy, the effects of our drug candidates may not be the desired
effects or may include undesirable side effects or the drug candidates may have other unexpected characteristics. Further,
success in preclinical testing and early clinical trials does not ensure that later clinical trials will be successful, and the
results of later clinical trials may not replicate the results of prior clinical trials and preclinical testing. The
clinical trial process may fail to demonstrate that our drug candidates are safe for humans and effective for indicated uses. In
addition, our clinical trials may involve a specific and small patient population. Results of early clinical trials
conducted on a small patient population may not be indicative of future results. Adverse or inconclusive results may
cause us to abandon a drug candidate and may delay development of other drug candidates. Any delay in, or termination
of, our clinical trials will delay the filing of NDAs with the FDA, or other filings with other foreign regulatory authorities,
and, ultimately, significantly impair our ability to commercialize our drug candidates and generate product revenues which would
have a material adverse effect on our business, results of operations and financial condition.
We may find it difficult to enroll patients
in our clinical trials, which could cause significant delays in the completion of such trials or may cause us to abandon one or
more clinical trials.
Some of the diseases or disorders that our
drug candidates are intended to treat are relatively rare and we expect only a subset of the patients with these diseases to be
eligible for our clinical trials. Our clinical trials generally mandate that a patient cannot be involved in another clinical
trial for the same indication. Therefore, subjects that participate in ongoing clinical trials for products that are competitive
with our drug candidates are not available for our clinical trials. An inability to enroll a sufficient number of patients for
any of our current or future clinical trials would result in significant delays or may require us to abandon one or more clinical
trials altogether, which will have a material adverse effect on our business, results of operations and financial condition.
Patients may discontinue their participation
in our clinical trials, which may negatively impact the results of these studies and extend the timeline for completion of our
development programs.
Patients enrolled in our clinical trials
may discontinue their participation at any time during the study as a result of a number of factors, including withdrawing their
consent or experiencing adverse clinical events, which may or may not be judged related to our drug candidates under evaluation.
The discontinuation of patients in any one of our studies may cause the results from that study not to be positive or to not support
a filing for regulatory approval of the applicable drug candidate, which would have a material adverse effect on our business,
results of operations and financial condition.
Because our clinical trials depend upon
third-party researchers, the results of our clinical trials and such research activities are subject to delays and other risks
which are, to a certain extent, beyond our control, which could impair our clinical development programs and our competitive position.
We depend upon independent investigators
and collaborators, such as universities and medical institutions, to conduct our preclinical and clinical trials. These collaborators
are not our employees, and we cannot control the amount or timing of resources that they devote to our clinical development programs.
The investigators may not assign as great a priority to our clinical development programs or pursue them as diligently as we would
if we were undertaking such programs directly. If outside collaborators fail to devote sufficient time and resources to our clinical
development programs, or if their performance is substandard, the approval of our NDA and other marketing applications, and our
introduction of new drugs, if any, may be delayed which could impair our clinical development programs and would have a material
adverse effect on our business and results of operations. The collaborators may also have relationships with other commercial
entities, some of whom may compete with us. If our collaborators also assist our competitors, our competitive position could be
harmed.
The manufacture of our products is an
exacting and complex process, and if we or one of our materials suppliers encounter problems manufacturing our products, it will
have a material adverse effect on our business and results of operations.
The FDA and foreign
regulators require manufacturers to register manufacturing facilities. The FDA and foreign regulators also inspect these facilities
to confirm compliance with cGMP or similar requirements that the FDA or foreign regulators establish. We or our materials suppliers
may face manufacturing or quality control problems causing product production and shipment delays or a situation where we or the
supplier may not be able to maintain compliance with the FDA’s cGMP requirements, or those of foreign regulators, necessary
to continue manufacturing our drug candidates. Any failure to comply with cGMP requirements or other FDA or foreign regulatory
requirements could adversely affect our clinical research activities and our ability to market and develop our products.
To date, our current facility has passed audits by the FDA, the Israeli MOH, ANVISA and the IMB on behalf of the EMA but remains
subject to audit by other foreign regulatory authorities. There can be no assurance that we will be able to comply with FDA or
foreign regulatory manufacturing requirements for our current facility or any facility we may establish in the future, which would
have a material adverse effect on our business, results of operations and financial condition.
We rely on third parties for final processing
of taliglucerase alfa and our other product candidates, which exposes us to a number of risks that may delay development, regulatory
approval and commercialization of taliglucerase alfa and our other product candidates or result in higher product costs.
We have no experience in the final filling
and freeze drying steps of the drug manufacturing process. We have engaged a European contract manufacturer to act as an additional
source of fill and finish activities for taliglucerase alfa and have engaged other parties for our other product candidates. We
currently rely primarily on other third-party contractors to perform the final manufacturing steps for taliglucerase alfa on a
commercial scale. We may be unable to identify manufacturers and/or replacement manufacturers on acceptable terms or at all because
the number of potential manufacturers is limited and the FDA and other regulatory authorities, as applicable, must approve any
manufacturer and/or replacement manufacturer, including us, and we or any such third party manufacturer might be unable to formulate
and manufacture our drug products in the volume and of the quality required to meet our clinical and commercial needs. If we engage
any contract manufacturers, such manufacturers may not perform as agreed or may not remain in the contract manufacturing business
for the time required to supply our clinical or commercial needs. In addition, contract manufacturers are subject to the rules
and regulations of the FDA and comparable foreign regulatory authorities and face the risk that any of those authorities may find
that they are not in compliance with applicable regulations. Each of these risks could delay our clinical trials, the approval,
if any, of taliglucerase alfa and our other potential drug candidates by the FDA and other regulatory authorities, or the commercialization
of taliglucerase alfa and our other drug candidates or could result in higher product costs or otherwise deprive us of potential
product revenues.
Developments by competitors may render
our products or technologies obsolete or non-competitive which would have a material adverse effect on our business, results of
operations and financial condition.
We compete against
fully integrated pharmaceutical companies and smaller companies that are collaborating with larger pharmaceutical companies, academic
institutions, government agencies and other public and private research organizations. Our drug candidates will have to
compete with existing therapies and therapies under development by our competitors. In addition, our commercial opportunities
may be reduced or eliminated if our competitors develop and market products that are less expensive, more effective or safer than
our drug products. Other companies have drug candidates in various stages of preclinical or clinical development to treat diseases
for which we are also seeking to develop drug products. Some of these potential competing drugs are further advanced in development
than our drug candidates and may be commercialized earlier. Even if we are successful in developing effective drugs, our products
may not compete successfully with products produced by our competitors.
We specifically face competition from companies
with approved treatments of Gaucher disease. In addition to Elelyso, there are two other ERTs for the treatment of Gaucher disease;
Cerezyme and VPRIV. To a much lesser extent, we also compete with Actelion. In addition, Cerdelga, Genzyme’s orally-delivered
small molecule drug for the treatment of Gaucher disease, was approved in 2014.
There are two approved ERTs for the treatment
of Fabry disease; Fabrazyme and Replagal. Fabrazyme is available in the United States and the European Union. Replagal is available
in the European Union. In 2012, Shire elected to withdraw its BLA for Replagal in the United States. In addition, we are aware
of other clinical stage, early clinical stage and experimental drugs which are being developed for the treatment of Fabry disease.
We also face competition from companies that
are developing other platforms for the expression of recombinant therapeutic pharmaceuticals. We are aware of companies that are
developing alternative technologies to develop and produce therapeutic proteins in anticipation of the expiration of certain patent
claims covering marketed proteins. Competitors developing alternative expression technologies include Crucell N.V. (which was
acquired by Johnson & Johnson during 2010), Shire and GlycoFi, Inc. (which was acquired by Merck & Co. Inc.). Other companies
are developing alternate plant-based technologies, include iBio, Medicago and Greenovation Biotech GmbH, none of which are cell-based.
Rather, such companies base their product development on transgenic plants or whole plants.
Most of our competitors, either alone or
together with their collaborative partners, operate larger research and development programs, staff and facilities and have substantially
greater financial resources than we do, as well as significantly greater experience in:
| · | undertaking
preclinical testing and human clinical trials; |
| · | obtaining
marketing approvals from the FDA and other regulatory authorities; |
| · | formulating
and manufacturing drugs; and |
| · | launching,
marketing and selling drugs. |
These organizations also compete with us
to attract qualified personnel, acquisitions and joint ventures candidates and for other collaborations. Activities of our competitors
may impose unanticipated costs on our business which would have a material adverse effect on our business, results of operations
and financial condition.
If we in-license drug candidates, we may
delay or otherwise adversely affect the development of our existing drug candidates, which may negatively impact our business,
results of operations and financial condition.
In addition to our own internally developed
drug candidates, we proactively seek opportunities to in-license and advance other drug candidates that are strategic and have
value-creating potential to take advantage of our development know-how and technology. If we in-license any additional drug candidate,
our capital requirements may increase significantly. In addition, in-licensing additional drug candidates may place a strain on
the time of our existing personnel, which may delay or otherwise adversely affect the development of our existing drug candidates
or cause us to re-prioritize our drug pipeline if we do not have the necessary capital resources to develop all of our drug candidates,
which may delay the development of our drug candidates and materially and adversely impact our business, results of operations
and financial condition.
If we are unable to successfully manage
our growth, there could be a material adverse impact on our business, results of operations and financial condition.
We have grown rapidly and expect to continue
to grow. To manage our anticipated future growth, we must continue to implement and improve our managerial, operational and financial
systems, expand our facilities and continue to recruit and train additional qualified personnel. Due to our limited resources,
we may not be able to effectively manage the expansion of our operations or recruit and train additional qualified personnel.
The expansion of our operations may lead to significant costs and may divert our management and business development resources.
Any inability on the part of our management to manage growth could delay the execution of our business plans or disrupt our operations.
If we are unable to manage our growth effectively, we may not use our resources in an efficient manner, which may delay the development
of our drug candidates and materially and adversely impact our business, results of operations and financial condition.
If we acquire companies, products or technologies,
we may face integration risks and costs associated with those acquisitions that could negatively impact our business, results
of operations and financial condition.
If we are presented with appropriate opportunities,
we may acquire or make investments in complementary companies, products or technologies. We may not realize the anticipated benefit
of any acquisition or investment. If we acquire companies or technologies, we will face risks, uncertainties and disruptions associated
with the integration process, including difficulties in the integration of the operations of an acquired company, integration
of acquired technology with our products, diversion of our management’s attention from other business concerns, the potential
loss of key employees or customers of the acquired business and impairment charges if future acquisitions are not as successful
as we originally anticipate. In addition, our operating results may suffer because of acquisition-related costs or amortization
expenses or charges relating to acquired intangible assets. Any failure to successfully
integrate other companies, products or technologies that we may acquire may have a material adverse
effect on our business and results of operations. Furthermore, we may have to incur debt or issue equity securities to
pay for any additional future acquisitions or investments, the issuance of which could be dilutive to our existing shareholders.
We depend upon key employees and consultants
in a competitive market for skilled personnel. If we are unable to attract and retain key personnel, it could adversely affect
our ability to develop and market our products.
We are highly
dependent upon the principal members of our management team, especially our President and Chief Executive Officer, Moshe
Manor, as well as the Chairman of our Board of Directors, Shlomo Yanai, our other directors, our scientific advisory board members,
consultants and collaborating scientists. Many of these people have been involved with us for many
years and have played integral roles in our progress, and we believe that they will continue to provide value to us. A loss of
any of these personnel may have a material adverse effect on aspects of our business, clinical development and regulatory
programs. We have employment agreements with Moshe Manor and our other executive officers that may be terminated by us or the
applicable officer at any time with varying notice periods of 60 to 90 days. Although these employment
agreements generally include non-competition covenants, the applicable noncompetition provisions can be difficult and costly to
monitor and enforce. The loss of any of these persons’ services may adversely affect our ability to develop and market our
products and obtain necessary regulatory approvals. Further, we do not maintain key-man life insurance.
We also depend in part on the continued service
of our key scientific personnel and our ability to identify, hire and retain additional personnel, including marketing and sales
staff. We experience intense competition for qualified personnel, and the existence of non-competition agreements between prospective
employees and their former employers may prevent us from hiring those individuals or subject us to suit from their former employers.
While we attempt to provide competitive compensation packages to attract and retain key personnel, many of our competitors are
likely to have greater resources and more experience than we have, making it difficult for us to compete successfully for key
personnel.
Our collaborations with outside scientists
and consultants may be subject to restriction and change.
We work with medical
experts, chemists, biologists and other scientists at academic and other institutions, and consultants who assist us in our research,
development, regulatory and commercial efforts, including the members of our scientific advisory board. These scientists and consultants
have provided, and we expect that they will continue to provide, valuable advice regarding our programs. These scientists and
consultants are not our employees, may have other commitments that would limit their future availability to us and typically will
not enter into non-compete agreements with us. If a conflict of interest arises between their work for us and their work for another
entity, we may lose their services. In addition, we will be unable to prevent them from establishing competing businesses or developing
competing products. For example, if a key scientist acting as a principal investigator in any of our clinical trials identifies
a potential product or compound that is more scientifically interesting to his or her professional interests, his or her availability
to remain involved in our clinical trials could be restricted or eliminated, which may have a material adverse effect on our
business, financial condition and results of operations.
Under current U.S. and Israeli law, we
may not be able to enforce employees’ covenants not to compete and therefore may be unable to prevent our competitors from
benefiting from the expertise of some of our former employees.
We have entered
into non-competition agreements with substantially all of our employees. These agreements prohibit our employees, if they cease
working for us, from competing directly with us or working for our competitors for a limited period. Under current U.S. and Israeli
law, we may be unable to enforce these agreements against most of our employees and it may be difficult for us to restrict our
competitors from gaining the expertise our former employees gained while working for us. If we cannot enforce our employees’
non-compete agreements, we may be unable to prevent our competitors from benefiting from the expertise of our former employees,
which may have a material adverse effect on our business, financial condition and results of operations.
If product liability claims are brought
against us, it may result in reduced demand for our products and product candidates or damages that exceed our insurance coverage.
The clinical testing, marketing and use of
our products and product candidates exposes us to product liability claims if the use or misuse of those products or product candidates
cause injury or disease, or results in adverse effects. Use of our products or product candidates, whether in clinical trials
or post approval, could result in product liability claims. We presently carry clinical trial liability insurance with coverages
of up to $10.0 million per occurrence and $10.0 million in the aggregate, an amount we consider reasonable and customary. However,
this insurance coverage includes various deductibles, limitations and exclusions from coverage, and in any event might not fully
cover any potential claims. We may need to obtain additional clinical trial liability coverage prior to initiating additional
clinical trials. We expect to obtain product liability insurance coverage before commercialization of our product candidates;
however, such insurance is expensive and insurance companies may not issue this type of insurance when we need it. We may not
be able to obtain adequate insurance in the future at an acceptable cost. Any product liability claim, even one that was not in
excess of our insurance coverage or one that is meritless and/or unsuccessful, may adversely affect our cash available for other
purposes, such as research and development, which may have a material adverse effect on our business, results of operations and
financial condition. Product liability claims, even if without merit, may result in reduced demand for our products, if approved,
which would have a material adverse effect on our business, financial condition and results of operations. In addition, the existence
of a product liability claim could affect the market price of our common stock.
Reforms in the healthcare industry and
the uncertainty associated with pharmaceutical pricing, reimbursement and related matters could adversely affect the marketing,
pricing and demand for our products, if approved.
Increasing healthcare expenditures have been
the subject of considerable public attention in the United States. Both private and government entities are seeking ways to reduce
or contain healthcare costs. Numerous proposals that would result in changes in the U.S. healthcare system have been introduced
or proposed in the U.S. Congress and in some state legislatures within the United States, including reductions in the pricing
of prescription products and changes in the levels at which consumers and healthcare providers are reimbursed for purchases of
pharmaceutical products. Legislation passed in recent years has imposed certain changes to the way in which drugs, including our
product candidates, are covered and reimbursed in the United States. For example, federal legislation and regulations have implemented
new reimbursement methodologies for certain drugs, created a voluntary prescription drug benefit, Medicare Part D, and have imposed
significant revisions to the Medicaid Drug Rebate Program. The PPACA imposes yet additional changes to these programs. We believe
that legislation that reduces reimbursement for our product candidates could adversely impact how much or under what circumstances
healthcare providers will prescribe or administer our products, if approved. This could materially and adversely impact our business
by reducing our ability to generate revenue, raise capital, obtain additional collaborators and market our products, if approved.
In addition, we believe the increasing emphasis on managed care in the United States has and will continue to put pressure on
the price and usage of pharmaceutical products, which may adversely impact product sales, upon approval, if at all.
Governments outside the United States
tend to impose strict price controls and reimbursement approval policies, which may adversely affect our prospects for generating
revenue.
In some countries,
particularly European Union member states, the pricing of prescription pharmaceuticals is subject to governmental control. In
these countries, pricing negotiations with governmental authorities can take considerable time (six to 12 months or longer)
after the receipt of marketing approval for a product. To obtain reimbursement or pricing approval in some countries with respect
to any product candidate that achieves regulatory approval, we may be required to conduct a clinical trial that compares the cost-effectiveness
of our product candidate to other available therapies. If reimbursement of our products upon approval, if at all, is unavailable
or limited in scope or amount, or if pricing is set at unsatisfactory levels, our prospects for generating revenue, if any, could
be adversely affected which would have a material adverse effect on our business, results of operations and financial condition.
Further, if we achieve regulatory approval of any product, we must successfully negotiate product pricing for such product in
individual countries. As a result, the pricing of our products, if approved, in different countries may vary widely, thus creating
the potential for third-party trade in our products in an attempt to exploit price differences between countries. This third-party
trade of our products could undermine our sales in markets with higher prices which
could have a material adverse effect on our business, results of operations and financial condition.
Our ability to utilize net operating loss
carryforwards may be limited.
Our net operating
loss carryforwards, or “NOLs”, as of December 31, 2014, are equal to approximately $138.8 million,
of which approximately $17 million may be restricted under Section 382 of the Internal Revenue Code of 1986, as amended, or the
“Code.” Section 382 of the Code imposes limitations on a corporation’s ability to utilize NOLs to offset taxable
income if the corporation experiences an “ownership change.” In general terms, an “ownership change” may
result from transactions increasing the ownership of certain stockholders in the stock of a corporation by more than 50% over
a three-year period. In the event that an ownership change has occurred, or were to occur, utilization of our NOLs would be subject
to an annual limitation under Section 382, which is generally the fair market value of the pre-change entity multiplied by the
long-term tax exempt rate, which is published monthly by the Internal Revenue Service.
We are a holding company with no operations
of our own.
We are a holding company with no operations
of our own. Accordingly, our ability to conduct our operations, service any debt that we may incur in the future and pay dividends,
if any, is dependent upon the earnings from the business conducted by Protalix Ltd. The distribution of those earnings or advances
or other distributions of funds by our subsidiary to us, as well as our receipt of such funds, are contingent upon the earnings
of our subsidiary and are subject to various business considerations and U.S. and Israeli law. If Protalix Ltd. is unable to make
sufficient distributions or advances to us, or if there are limitations on our ability to receive such distributions or advances,
we may not have the cash resources necessary to conduct our corporate operations which would have a material adverse effect on
our business, results of operations and financial condition.
Risks Related to Regulatory Matters
We are subject to extensive governmental
regulation including the requirement of FDA or comparable approval before our drug candidates may be marketed.
Both before and after approval of our drug
candidates, we, our drug candidates, our suppliers, our contract manufacturers and our contract testing laboratories are subject
to extensive regulation by the FDA and comparable foreign regulatory authorities. Failure to comply with applicable requirements
of the FDA or comparable foreign regulatory authorities could result in, among other things, any of the following actions:
| · | fines
and other monetary penalties; |
| · | unanticipated
expenditures; |
| · | delays
in the FDA’s or other foreign regulatory authorities’ approving, or the refusal
of any regulatory authority to approve, any drug candidate; |
| · | product
recall or seizure; |
| · | interruption
of manufacturing or clinical trials; |
In addition to the approval requirements,
other numerous and pervasive regulatory requirements apply, both before and after approval, to us, our drug candidates, and our
suppliers, contract manufacturers, and contract laboratories. These include requirements related to:
| · | reporting
to the FDA certain adverse experiences associated with use of the drug candidate; and |
| · | obtaining
additional approvals for certain modifications to the drug candidate or its labeling
or claims. |
We also are subject to inspection by the
FDA and comparable foreign regulatory authorities, to determine our compliance with regulatory requirements, as are our suppliers,
contract manufacturers, and contract testing laboratories, and there can be no assurance that the FDA or any other comparable
foreign regulatory authority, will not identify compliance issues that may disrupt production or distribution, or require substantial
resources to correct. We may be required to make modifications to our manufacturing operations in response to these inspections
which may require significant resources and may have a material adverse effect upon our business, results of operations and financial
condition.
The approval process for any drug candidate
may also be delayed by changes in government regulation, future legislation or administrative action or changes in policy of the
FDA and comparable foreign authorities that occur prior to or during their respective regulatory reviews of such drug candidate.
Delays in obtaining regulatory approvals with respect to any drug candidate may:
| · | delay
commercialization of, and our ability to derive product revenues from, such drug candidate; |
| · | delay
any regulatory-related milestone payments payable under outstanding collaboration agreements; |
| · | require
us to perform costly procedures with respect to such drug candidate; or |
| · | otherwise
diminish any competitive advantages that we may have with respect to such drug candidate. |
Delays in the approval process for any drug
candidate may have a material adverse effect upon our business, results of operations and financial condition.
We may not obtain the necessary U.S.,
EMA or other worldwide regulatory approvals to commercialize our drug candidates in a timely manner, if at all, which would have
a material adverse effect on our business, financial condition and results of operations.
We need FDA approval to commercialize our
drug candidates in the United States, EMA approval to commercialize our drug candidates in the European Union and approvals from
foreign regulators to commercialize our drug candidates elsewhere. In order to obtain FDA approval of any of our drug candidates,
we must submit to the FDA an NDA or a Biologic License Application (BLA) demonstrating that the drug candidate is safe for humans
and effective for its intended use. This demonstration requires significant research and animal tests, which are referred to as
preclinical studies, as well as human tests, which are referred to as clinical trials. In the European Union, we must submit an
MAA to the EMA. Satisfaction of the FDA’s, the EMA’s and foreign regulatory authorities’ regulatory requirements
typically takes many years, depends upon the type, complexity and novelty of the drug candidate and requires substantial resources
for research, development and testing. Taliglucerase alfa has been approved for marketing in the United States, Israel, Brazil,
Mexico, Chile and Uruguay, and marketing applications are outstanding in other countries. Even if we comply with all the requests
of regulatory authorities, the authorities may ultimately reject the marketing applications filed for taliglucerase alfa or that
we file for our other product candidates in the future, if any, or we might not obtain regulatory clearance in a timely manner
for taliglucerase alfa in certain countries or for any of our other drug candidates. Companies in the pharmaceutical and biotechnology
industries have suffered significant setbacks in advanced or late-stage clinical trials, even after obtaining promising earlier
trial results or in preliminary findings or other comparable authorities for such clinical trials. Further, even if
favorable testing data is generated by the clinical trials of a drug candidate, the applicable regulatory authority may not accept
or approve the marketing application filed by a pharmaceutical or biotechnology company for the drug candidate. Failure to obtain
approval of the FDA, EMA or comparable foreign authorities of any of our drug candidates in a timely manner, if at all, will severely
undermine our business, financial condition and results of operation by reducing our potential marketable products and our ability
to generate corresponding product revenues.
Our research and clinical efforts may not
result in drugs that the FDA, EMA or foreign regulatory authorities consider safe for humans and effective for indicated uses,
which would have a material adverse effect on our business, financial condition and results of operations. After clinical
trials are completed for any drug candidate, if at all, the FDA, EMA and foreign regulatory authorities have substantial discretion
in the drug approval process of the drug candidate in their respective jurisdictions and may require us to conduct additional
clinical testing or perform post-marketing studies which would cause us to incur additional costs. Incurring such costs may have
a material adverse effect on our business, financial condition and results of operations.
We have only limited experience in regulatory
affairs, and some of our drug candidates may be based on new technologies. These factors may affect our ability or the time we
require to obtain necessary regulatory approvals.
We have only limited experience in filing
and prosecuting the applications necessary to gain regulatory approvals for medical devices and drug candidates. Moreover, some
of the drug candidates that are likely to result from our development programs may be based on new technologies that have not
been extensively tested in humans. The regulatory requirements governing these types of drug candidates may be less well defined
or more rigorous than for conventional products. As a result, we may experience a longer regulatory process in connection with
obtaining regulatory approvals of any products that we develop.
If any of our other competitors are able
to obtain orphan drug exclusivity for any products that are competitive with our products, we may be precluded from selling or
obtaining approval of our competing products by the applicable regulatory authorities for a significant period of time.
In the United States, the European Union
and other countries, a drug may be designated as having orphan drug status, subject to certain conditions. There can be no assurance
that a drug candidate that receives orphan drug designation will receive orphan drug marketing exclusivity and more than one drug
can have orphan designation for the same indication.
Foreign regulations
regarding orphan drugs are similar to those in the United States but there are several conceptual differences. For example, the
exclusivity period in the European Union is generally 10 years.
The EMA/European Commission has granted orphan drug designation and exclusivity to VPRIV in the European Union. For this reason,
CHMP has recommended that the EC not issue a Marketing Authorization for taliglucerase alfa
in the European Union. Therefore, VPRIV
has orphan market exclusivity in the European Union for a 10-year period commencing on its authorization in August 2010 which
may have a material adverse
effect on our business, results of operations and financial condition.
From time to time, we may apply to the FDA
or any comparable foreign regulatory authority for orphan drug designation for any one or more of our drug candidates. None of
our currently developed drug candidates have been designated as an orphan drug and there is no guarantee that the FDA or any other
regulatory authority will grant such designation in the future. In addition, neither orphan drug designation nor orphan drug exclusivity
prevents competitors from developing or marketing different drugs for that indication. Even if we obtain orphan drug exclusivity
for one or more indications for one of our drug candidates, we may not be able to maintain the exclusivity. For example, if a
competitive product that is the same drug or biologic as one of our drug candidates is shown to be clinically superior to the
drug candidate, any orphan drug exclusivity granted to the drug candidate will not block the approval of the competitive product.
Risks Related to Intellectual Property
Matters
If we fail to adequately protect or enforce
our intellectual property rights or secure rights to third party patents, the value of our intellectual property rights would
diminish and our business, competitive position and results of operations would suffer.
As of December 31, 2014, we had 94 pending
patent applications and one joint pending patent application with a third party. However, the filing of a patent application does
not mean that we will be issued a patent, or that any patent eventually issued will be as broad as requested in the patent application
or sufficient to protect our technology. Any modification required to a current patent application may delay the approval of such
patent application which would have a material adverse effect on our business, results of operations and financial condition.
In addition, there are a number of factors that could cause our patents, if granted, to become invalid or unenforceable or that
could cause our patent applications to not be granted, including known or unknown prior art, deficiencies in the patent application
or the lack of originality of the technology. Our competitive position and future revenues will depend in part on our ability
and the ability of our licensors and collaborators to obtain and maintain patent protection for our products, methods, processes
and other technologies, to preserve our trade secrets, to prevent third parties from infringing on our proprietary rights and
to operate without infringing the proprietary rights of third parties. We have filed U.S. and international patent applications
for process patents, as well as composition of matter patents,
for taliglucerase alfa and other product candidates. However, we cannot predict:
| · | the
degree and range of protection any patents will afford us against competitors and those
who infringe upon our patents, including whether third parties will find ways to invalidate
or otherwise circumvent our licensed patents; |
| · | if
and when patents will issue; |
| · | whether
or not others will obtain patents claiming aspects similar to those covered by our licensed
patents and patent applications; or |
| · | whether
we will need to initiate litigation or administrative proceedings, which may be costly,
and whether we win or lose. |
As of December
31, 2014, we hold, or have license rights to, 64 patents. If patent rights covering our products or technologies are not sufficiently
broad, they may not provide us with sufficient proprietary protection or competitive advantages against competitors with similar
products and technologies. Furthermore, if the U.S. Patent and Trademark Office or foreign patent
offices issue patents to us or our licensors, others may challenge the patents or circumvent the patents, or the patent office
or the courts may invalidate the patents. Thus, any patents we own or license from or to third parties may not provide any protection
against our competitors and those who infringe upon our patents.
Furthermore, the life of our patents is limited.
The patents we hold, and the patents that may be issued in the future based on patent applications from the patent families, relating
to our ProCellEx protein expression system are expected to expire between 2017 and 2025.
We rely on confidentiality agreements
that could be breached and may be difficult to enforce which could have a material adverse effect on our business and competitive
position.
Our policy is to enter agreements relating
to the non-disclosure of confidential information with third parties, including our contractors, consultants, advisors and research
collaborators, as well as agreements that purport to require the disclosure and assignment to us of the rights to the ideas, developments,
discoveries and inventions of our employees and consultants while we employ them. However, these agreements can be difficult and
costly to enforce. Moreover, to the extent that our contractors, consultants, advisors and research collaborators apply or independently
develop intellectual property in connection with any of our projects, disputes may arise as to the proprietary rights to the intellectual
property. If a dispute arises, a court may determine that the right belongs to a third party, and enforcement of our rights can
be costly and unpredictable. In addition, we rely on trade secrets and proprietary know-how that we seek to protect in part by
confidentiality agreements with our employees, contractors, consultants, advisors and others. Despite the protective measures
we employ, we still face the risk that:
| · | these
agreements may be breached; |
| · | these
agreements may not provide adequate remedies for the applicable type of breach; or |
| · | our
trade secrets or proprietary know-how will otherwise become known. |
Any breach of our confidentiality agreements
or our failure to effectively enforce such agreements may have a material adverse effect on our business and competitive position.
If we infringe the rights of third parties
we could be prevented from selling products, forced to pay damages and required to defend against litigation which could result
in substantial costs and may have a material adverse effect on our business, results of operations and financial condition.
We have not received to date any claims of
infringement by any third parties. However, as our drug candidates progress into clinical trials and commercialization, if at
all, our public profile and that of our drug candidates may be raised and generate such claims. Defending against such claims,
and occurrence of a judgment adverse to us, could result in unanticipated costs and may have a material adverse effect on our
business and competitive position. If our products, methods, processes and other technologies infringe the proprietary rights
of other parties, we may incur substantial costs and we may have to:
| · | obtain
licenses, which may not be available on commercially reasonable terms, if at all; |
| · | redesign
our products or processes to avoid infringement; |
| · | stop
using the subject matter claimed in the patents held by others, which could cause us
to lose the use of one or more of our drug candidates; |
| · | defend
litigation or administrative proceedings that may be costly whether we win or lose, and
which could result in a substantial diversion of management resources; or |
Any costs incurred in connection with such
events or the inability to sell our products may have a material adverse effect on our business, results of operations and financial
condition.
If we cannot meet requirements under our
license agreements, we could lose the rights to our products, which could have a material adverse effect on our business.
We depend on licensing
agreements with third parties to maintain the intellectual property rights to certain of our product candidates. Our license
agreements require us to make payments and satisfy performance obligations in order to maintain our
rights under these agreements. All of these agreements last either throughout the life of the patents that are the subject of
the agreements, or with respect to other licensed technology, for a number of years after the first commercial sale of the relevant
product.
In addition, we are responsible for the cost
of filing and prosecuting certain patent applications and maintaining certain issued patents licensed to us. If we do not meet
our obligations under our license agreements in a timely manner, we could lose the rights to our proprietary technology which
could have a material adverse effect on our business, results of operations and financial condition.
Risks Relating to Our Operations in Israel
Potential political, economic and military
instability in the State of Israel, where the majority of our senior management and our research and development facilities are
located, may adversely affect our results of operations.
Our executive
office and operations are located in the State of Israel. Accordingly, political, economic and military conditions in Israel directly
affect our business. Since the State of Israel was established in 1948, a number of armed conflicts have occurred between Israel
and its Arab neighbors. Any hostilities involving Israel or the interruption or curtailment of trade between Israel and its present
trading partners, or a significant downturn in the economic or financial condition of Israel, could affect adversely our operations
and product development. Although Israel has entered into various agreements with Egypt, Jordan and the Palestinian Authority,
there have been times since October 2000 when Israel has experienced an increase in unrest and terrorist activity. The establishment
in 2006 of a government in the Palestinian Authority by representatives of the Hamas militant group has created additional unrest
and uncertainty in the region. Starting in December 2008, for approximately three weeks, Israel engaged in an armed conflict with
Hamas in the Gaza Strip. Armed conflicts have taken place between Israel and Hamas in the Gaza Strip in 2008, 2012 and 2014. Our
facilities in northern Israel are in range of rockets that were fired from Lebanon into Israel during a 2006 war with the Hizbollah
in Lebanon, and suffered minimal damages during one of the rocket attacks. Our insurance policies do not cover us for the
damages incurred in connection with these conflicts or for any resulting disruption in our operations. The Israeli government,
as a matter of law, provides coverage for the reinstatement value of direct damages that are caused by terrorist attacks or acts
of war; however, the government may cease providing such coverage or the coverage might not be enough to cover potential damages.
If our facilities are damaged as a result of hostile action, our operations may be materially adversely affected.
In addition to the foregoing, since the end
of 2010, numerous acts of protest and civil unrest have taken place in several countries in the Middle East and North Africa,
many of which involved significant violence. Civil unrest in Egypt, which borders Israel, has resulted in significant changes
to the country’s government. There is currently a civil war in Syria, also bordering Israel, and Israel has been hit by
rockets and mortars originating from Syria. The ultimate effect of these developments on the political and security situation
in the Middle East and on Israel’s position within the region is not clear at this time.
Our operations may be disrupted by the
obligations of our personnel to perform military service which could have a material adverse effect on our business.
Many of our male employees in Israel, including
members of senior management, are obligated to perform up to one month (in some cases more) of annual military reserve duty until
they reach the age of 45 and, in the event of a military conflict, could be called to active duty. Our operations could be disrupted
by the absence of a significant number of our employees related to military service or the absence for extended periods of military
service of one or more of our key employees. A disruption could have a material adverse effect on our business.
Because a certain portion of our expenses
is incurred in New Israeli Shekels, or NIS, our results of operations may be seriously harmed by currency fluctuations and inflation.
We report our financial statements in U.S.
dollars, our functional currency. Although most of our expenses are incurred in U.S. dollars, we pay a portion of our expenses
in New Israeli Shekels, or NIS, and as a result, we are exposed to risk to the extent that the inflation rate in Israel exceeds
the rate of devaluation of the NIS in relation to the U.S. dollar or if the timing of these devaluations lags behind inflation
in Israel. In that event, the U.S. dollar cost of our operations in Israel will increase and our U.S. dollar-measured results
of operations will be adversely affected. To the extent that the value of the NIS increases against the dollar, our expenses on
a dollar cost basis increase. Our operations also could be adversely affected if we are unable to guard against currency fluctuations
in the future. To date, we have not engaged in hedging transactions. In the future, we may enter into currency hedging transactions
to decrease the risk of financial exposure from fluctuations in the exchange rate of the U.S. dollar against the NIS. These measures,
however, may not adequately protect us from material adverse effects.
The tax benefits available to us require
that we meet several conditions and may be terminated or reduced in the future, which would increase our taxes.
We are
able to take advantage of tax exemptions and reductions resulting from the “Approved Enterprise”
status of our facilities in Israel. To remain eligible for these tax benefits, we must continue to meet certain conditions,
including making specified investments in property and equipment, and financing at least 30% of such investments with share capital.
If we fail to meet these conditions in the future, the tax benefits would be canceled and we may be required to refund any tax
benefits we already have enjoyed. These tax benefits are subject to investment policy by the Investment Center and may not be
continued in the future at their current levels or at any level. In recent years the Israeli government has reduced the benefits
available and has indicated that it may further reduce or eliminate some of these benefits in the future. The termination or reduction
of these tax benefits or our inability to qualify for additional “Approved Enterprise” approvals may increase our
tax expenses in the future, which would reduce our expected profits and adversely affect our business and results of operations.
Additionally, if we increase our activities outside of Israel, for example, by future acquisitions, such increased activities
generally may not be eligible for inclusion in Israeli tax benefit programs.
The Israeli government grants we have
received for certain research and development expenditures restrict our ability to manufacture products and transfer technologies
outside of Israel and require us to satisfy specified conditions. If we fail to satisfy these conditions, we may be required to
refund grants previously received together with interest and penalties which could have a material adverse effect on our business
and results of operations.
Our research and development efforts have
been financed, in part, through grants that we have received from the OCS. We, therefore, must comply with the requirements of
the Research Law.
Under the Research Law we are prohibited
from manufacturing products developed using these grants outside of the State of Israel without special approvals, although the
Research Law does enable companies to seek prior approval for conducting manufacturing activities outside of Israel without being
subject to increased royalties. We may not receive the required approvals for any proposed transfer
of manufacturing activities. Even if we do receive approval to manufacture products developed with government grants outside
of Israel, we may be required to pay an increased total amount of royalties (possibly up to 300% of the grant amounts plus interest),
depending on the manufacturing volume that is performed outside of Israel, as well as at a possibly increased royalty rate. This
restriction may impair our ability to outsource manufacturing or engage in similar arrangements for those products or technologies.
Additionally, under the Research Law, we
are prohibited from transferring the OCS-financed technologies and related intellectual property rights outside of the State of
Israel, except under limited circumstances and only with the approval of the OCS’ Research Committee. We may not receive
the required approvals for any proposed transfer and, if received, we may be required to pay the OCS a portion of the consideration
that we receive upon any sale of such technology by a non-Israeli entity. The scope of the support received, the royalties that
we have already paid to the OCS, the amount of time that has elapsed between the date on which the know-how was transferred and
the date on which the OCS grants were received and the sale price and the form of transaction will be taken into account in order
to calculate the amount of the payment to the OCS. Approval of the transfer of technology to residents of the State of Israel
is required, and may be granted in specific circumstances only if the recipient abides by the provisions of applicable laws, including
the restrictions on the transfer of know-how and the obligation to pay royalties. No assurance can be made that approval to any
such transfer, if requested, will be granted.
These restrictions
may impair our ability to sell our technology assets or to outsource manufacturing outside of Israel. The restrictions will continue
to apply for a certain period of time even after we have repaid the full amount of royalties payable for the grants. For
the years ended December 31, 2013 and 2014, we recorded grants totaling $3.4 million and $5.1 million from the OCS, respectively.
The grants represent 10.4% and 17.2%, respectively, of our gross research and development expenditures for the years ended December
31, 2013 and 2014. If we fail to satisfy the conditions of the Research Law, we may be required to
refund certain grants previously received together with interest and penalties, and may become subject to criminal charges, any
of which could have a material adverse effect on our business, results of operations and financial condition.
Investors may have difficulties enforcing
a U.S. judgment, including judgments based upon the civil liability provisions of the U.S. federal securities laws against us,
our executive officers and most of our directors or asserting U.S. securities laws claims in Israel.
Most of our directors and none of our officers
are residents of the United States, and most of their assets and our assets are located outside the United States. Service of
process upon our non-U.S. resident directors and officers and enforcement of judgments obtained in the United States against us,
some of our directors and executive officers may be difficult to obtain within the United States. We have been informed by our
legal counsel in Israel that investors may find it difficult to assert claims under U.S. securities laws in original actions instituted
in Israel or obtain a judgment based on the civil liability provisions of U.S. federal securities laws against us, our officers
and our directors. Israeli courts may refuse to hear a claim based on a violation of U.S. securities laws against us or our officers
and directors because Israel is not the most appropriate forum to bring such a claim. In addition, even if an Israeli court agrees
to hear a claim, it may determine that Israeli law and not U.S. law is applicable to the claim. If U.S. law is found to be applicable,
the content of applicable U.S. law must be proved as a fact which can be a time-consuming and costly process. Certain matters
of procedure will also be governed by Israeli law. There is little binding case law in Israel addressing the matters described
above.
Israeli courts might not enforce judgments
rendered outside Israel which may make it difficult to collect on judgments rendered against us. Subject to certain time limitations,
an Israeli court may declare a foreign civil judgment enforceable only if it finds that:
| · | the
judgment was rendered by a court which was, according to the laws of the state of the
court, competent to render the judgment; |
| · | the
judgment may no longer be appealed; |
| · | the
obligation imposed by the judgment is enforceable according to the rules relating to
the enforceability of judgments in Israel and the substance of the judgment is not contrary
to public policy; and |
| · | the
judgment is executory in the state in which it was given. |
Even if these conditions are satisfied, an
Israeli court will not enforce a foreign judgment if it was given in a state whose laws do not provide for the enforcement of
judgments of Israeli courts (subject to exceptional cases) or if its enforcement is likely to prejudice the sovereignty or security
of the State of Israel. An Israeli court also will not declare a foreign judgment enforceable if:
| · | the
judgment was obtained by fraud; |
| · | there
is a finding of lack of due process; |
| · | the
judgment was rendered by a court not competent to render it according to the laws of
private international law in Israel; |
| · | the
judgment is at variance with another judgment that was given in the same matter between
the same parties and that is still valid; or |
| · | at
the time the action was brought in the foreign court, a suit in the same matter and between
the same parties was pending before a court or tribunal in Israel. |
Risks Related to Investing in our Common
Stock
The market price of our common stock may
fluctuate significantly.
The market price of our common stock may
fluctuate significantly in response to numerous factors, some of which are beyond our control, such as:
| · | Purchases
of Uplyso by Fiocruz; |
| · | Pfizer’s
and our commercialization efforts for Elelyso; |
| · | the
results of our ongoing studies regarding our product candidates; |
| · | announcements
regarding partnerships or collaborations by us or our competitors; |
| · | developments
concerning intellectual property rights and regulatory approvals; |
| · | the
announcement of new products or product enhancements by us or our competitors; |
| · | variations
in our and our competitors’ results of operations; |
| · | changes
in earnings estimates or recommendations by securities analysts; |
| · | developments
in the biotechnology industry; and |
| · | general
market conditions and other factors, including factors unrelated to our operating performance. |
Further, stock markets in general, and the
market for biotechnology companies in particular, have recently experienced price and volume fluctuations. Continued market fluctuations
could result in extreme volatility in the price of our common stock, which could cause a decline in the value of our common stock.
Price volatility of our common stock may be worse if the trading volume of our common stock is low. We have not paid, and do not
expect to pay, any cash dividends on our common stock as any earnings generated from future operations will be used to finance
our operations. As a result, investors will not realize any income from an investment in our common stock until and unless their
shares are sold at a profit.
Future sales of our common stock could
reduce our stock price.
The market price of our common stock could
drop significantly if our existing shareholders sell a large number of shares of our common stock or are perceived by the market
as intending to sell them. All of our outstanding shares of our common stock are freely tradable without restriction or further
registration under the federal securities laws, unless owned by our affiliates. At December 31, 2014, there were options
to purchase common stock issued and outstanding and unvested restricted shares covering 7,465,025 shares of our common stock with
a weighted average exercise price of $3.96 per share. Also at December 31, 2014, there were 1,923,444 shares of common stock
remaining available for future for issuance in connection with future grants of incentives under our amended 2006 stock incentive
plan.
Servicing our debt requires a significant
amount of cash, and we may not have sufficient cash flow from our business to pay our debt.
Our ability to pay interest on, or to make
any scheduled payment of the principal of, the Notes, depends on our future performance, which is subject to economic, financial,
competitive and other factors beyond our control. Our business may not generate cash flow from operations in the future sufficient
to service our debt and make necessary expenditures. If we are unable to generate such cash flow, we may be required to adopt
one or more alternatives, such as selling assets, restructuring debt or obtaining additional equity capital on terms that may
be onerous or highly dilutive. Our ability to refinance our indebtedness will depend on the capital markets and our financial
condition at such time. If we raise additional debt, it would increase our interest expense, leverage and operating and financial
costs. In addition, the terms of the indenture governing the Notes and the agreements governing future indebtedness may restrict
us from adopting any of these alternatives. We may not be able to engage in any of these activities or engage in these activities
on desirable terms, which could result in a default on our debt obligations. The failure to generate sufficient cash flow or to
effect any of these alternatives could significantly adversely affect the value of the Notes and our ability to pay amounts due
under the Notes.
Our significant level of indebtedness
could adversely affect our business, financial condition and results of operations and prevent us from fulfilling our obligations
under the Notes and our other indebtedness.
The outstanding Notes represent a significant
amount of indebtedness and substantial debt service requirements. We may also incur additional indebtedness to meet future financing
needs. Our substantial indebtedness could have material adverse effects on our business, financial condition and results of operations.
For example, it could:
| · | make
it more difficult for us to satisfy our financial obligations, including with respect
to the Notes; |
| · | result
in an event of default if we fail to comply with the financial and other restrictive
covenants contained in agreements governing any future indebtedness, which event of default
could result in all of our debt becoming immediately due and payable; |
| · | increase
our vulnerability to general adverse economic, industry and competitive conditions; |
| · | reduce
the availability of our cash flow to fund working capital, capital expenditures, acquisitions
and other general corporate purposes because we will be required to dedicate a substantial
portion of our cash flow from operations to the payment of principal and interest on
our indebtedness; |
| · | limit
our flexibility in planning for, or reacting to, and increasing our vulnerability to
changes in our business, the industry in which we operate and the general economy; |
| · | prevent
us from raising funds necessary to purchase Notes surrendered to us by holders upon a
fundamental change (as described in the indenture governing the Notes), which failure
would result in an event of default with respect to the Notes; |
| · | place
us at a competitive disadvantage compared to our competitors that have less indebtedness
or are less highly leveraged and that, therefore, may be able to take advantage of opportunities
that our debt levels or leverage prevent us from exploiting; and |
| · | limit
our ability to obtain additional financing. |
Each of these factors may have a material
and adverse effect on our business, financial condition and results of operations and our ability to meet our payment obligations
under the Notes and our other indebtedness. Our ability to make payments with respect to the Notes and to satisfy any other debt
obligations will depend on our future operating performance and our ability to generate significant cash flow in the future, which
will be affected by prevailing economic conditions and financial, business, competitive, legislative and regulatory factors as
well as other factors affecting our company and industry, many of which are beyond our control.
Any conversion of the Notes will dilute
the ownership interest of our existing stockholders, including holders who had previously converted their notes.
The conversion of some or all of the Notes
will dilute the ownership interests of our existing stockholders. Any sales in the public market of our common stock issuable
upon such conversion could adversely affect prevailing market prices of our common stock. In addition, the existence of the Notes
may encourage short selling by market participants because the conversion of the Notes could depress the price of our common stock.
Our common stock is listed for trade on
more than one stock exchange, and this may result in price variations.
Our common stock is listed for trade on both
the NYSE MKT and the TASE. Dual-listing may result in price variations between the exchanges due to a number of factors. First,
our common stock is traded in U.S. dollars on the NYSE MKT and in NIS on the TASE. In addition, the exchanges are open for trade
at different times of the day and on different days. For example, the TASE opens generally during Israeli business hours, Sunday
through Thursday while the NYSE MKT opens generally during U.S. business hours, Monday through Friday. The two exchanges also
have differing vacation schedules. Differences in the trading schedules, as well as volatility in the exchange rate of the two
currencies, among other factors, may result different trading prices for our common stock on the two exchanges. Other external
influences may have different effects on the trading price of our common stock on the two exchanges.
Directors, executive officers, principal
shareholders and affiliated entities own a significant percentage of our capital stock, and they may make decisions that an investor
may not consider to be in the best interests of our shareholders.
Our directors,
executive officers, principal shareholders and affiliated entities beneficially own, in the aggregate, approximately 33%
of our outstanding common stock. As a result, if some or all of them acted together, they would have the ability to exert substantial
influence over the election of our Board of Directors and the outcome of issues requiring approval by our shareholders. This concentration
of ownership may have the effect of delaying or preventing a change in control of our company that may be favored by other shareholders.
This could prevent the consummation of transactions favorable to other shareholders, such as a transaction in which shareholders
might otherwise receive a premium for their shares over current market prices.
Failure to maintain effective internal
controls in accordance with Section 404 of the Sarbanes-Oxley Act could have a material adverse effect on our business and operating
results. In addition, current and potential shareholders could lose confidence in our financial reporting, which could have a
material adverse effect on the price of our common stock.
Effective internal controls are necessary
for us to provide reliable financial reports and effectively prevent fraud. If we cannot provide reliable financial reports or
prevent fraud, our results of operation could be harmed.
Section 404 of the Sarbanes-Oxley Act of
2002 requires annual management assessments of the effectiveness of our internal controls over financial reporting and a report
by our independent registered public accounting firm addressing these assessments. We continuously monitor our existing internal
controls over financial reporting systems to confirm that they are compliant with Section 404, and we may identify deficiencies
that we may not be able to remediate in time to meet the deadlines imposed by the Sarbanes-Oxley Act. This process may divert
internal resources and will take a significant amount of time and effort to complete.
If, at any time, it is determined that we
are not in compliance with Section 404, we may be required to implement new internal control procedures and reevaluate our financial
reporting. We may experience higher than anticipated operating expenses as well as increased independent auditor fees during the
implementation of these changes and thereafter. Further, we may need to hire additional qualified personnel. If we fail to maintain
the adequacy of our internal controls, as such standards are modified, supplemented or amended from time to time, we may not be
able to conclude on an ongoing basis that we have effective internal controls over financial reporting in accordance with Section
404 of the Sarbanes-Oxley Act, which could result in our being unable to obtain an unqualified report on internal controls from
our independent auditors. Failure to maintain an effective internal control environment could also cause investors to lose confidence
in our reported financial information, which could have a material adverse effect on the price of our common stock.
Compliance with changing regulation of
corporate governance and public disclosure may result in additional expenses, divert management’s attention from operating
our business which could have a material adverse effect on our business.
There have been
other changing laws, regulations and standards relating to corporate governance and public disclosure in addition to the Sarbanes-Oxley
Act, as well as new regulations promulgated by the Commission and rules promulgated by the national securities exchanges, including
the NYSE MKT and the NASDAQ. These new or changed laws, regulations and standards are subject
to varying interpretations in many cases due to their lack of specificity, and as a result, their application in practice may
evolve over time as new guidance is provided by regulatory and governing bodies, which could result in continuing uncertainty
regarding compliance matters and higher costs necessitated by ongoing revisions to disclosure and governance practices. As a result,
our efforts to comply with evolving laws, regulations and standards are likely to continue to result in increased general and
administrative expenses and a diversion of management time and attention from revenue-generating activities to compliance activities.
Our board members, Chief Executive Officer and Chief Financial Officer could face an increased risk of personal liability in connection
with the performance of their duties. As a result, we may have difficulty attracting and retaining qualified board members and
executive officers, which could have a material adverse effect on our business. If our efforts to comply with new or changed laws,
regulations and standards differ from the activities intended by regulatory or governing bodies, we may incur additional expenses
to comply with standards set by regulatory authorities or governing bodies which would have a material adverse effect on our business,
results of operations and financial condition.
The issuance of preferred stock or additional
shares of common stock could adversely affect the rights of the holders of shares of our common stock.
Our Board of Directors is authorized to issue
up to 100,000,000 shares of preferred stock without any further action on the part of our shareholders. Our Board of Directors
has the authority to fix and determine the voting rights, rights of redemption and other rights and preferences of preferred stock.
Currently, we have no shares of preferred stock outstanding.
Our Board of Directors may, at any time,
authorize the issuance of a series of preferred stock that would grant to holders the preferred right to our assets upon liquidation,
the right to receive dividend payments before dividends are distributed to the holders of common stock and the right to the redemption
of the shares, together with a premium, before the redemption of our common stock, which may have a material adverse effect on
the rights of the holders of our common stock. In addition, our Board of Directors, without further shareholder approval, may,
at any time, issue large blocks of preferred stock. In addition, the ability of our Board of Directors to issue shares of preferred
stock without any further action on the part of our shareholders may impede a takeover of our company and may prevent a transaction
that is favorable to our shareholders.
Under the rules of the TASE, other than incentives
under our amended 2006 stock incentive plan, we were prohibited from issuing any securities of any class or series different than
the common stock that is listed on the TASE for the 12-month period immediately succeeding our initial listing, which occurred
on September 6, 2010. As of the date hereof, the rules of the TASE allow us to issue securities with preferential rights
with respect to dividends but such other securities may not include voting rights. The foregoing does not limit our liability
to issue and grant options and warrants for the purchase of shares of our common stock.
USE OF PROCEEDS
We will not receive any proceeds from the
sale of shares covered by this Prospectus. While we may receive sums upon the exercise of certain awards by the selling securityholders,
we currently have no plans for their application, other than for general corporate purposes. We cannot assure you that any of
such options will be exercised.
SELLING SECURITYHOLDERS
We are registering for resale the shares
covered by this prospectus to permit the selling securityholders identified below and their pledgees, donees, transferees and
other successors-in-interest that receive their securities from a securityholder as a gift, partnership distribution or other
non-sale related transfer after the date of this prospectus to resell the shares when and as they deem appropriate. The selling
securityholders acquired, or may acquire, these shares from us pursuant to the Protalix BioTherapeutics, Inc. 2006 Stock Incentive
Plan, as amended. The shares may not be sold or otherwise transferred by the selling securityholders unless and until the applicable
awards vest and are exercised, as applicable, in accordance with the terms and conditions of such plan.
The following table sets forth:
| · | the
name of each selling securityholder; |
| · | the
position(s), office or other material relationship with our company and its predecessors
or affiliates, over the last three years of each selling securityholder; |
| · | the
number and percentage of shares of our common stock that each selling securityholder
beneficially owned as of May 1, 2015 prior to the offering for resale of the shares under
this prospectus; |
| · | the
number of shares of our common stock that may be offered for resale for the account of
each selling securityholder under this prospectus; and |
| · | the
number and percentage of shares of our common stock to be beneficially owned by each
selling securityholder after the offering of the resale shares (assuming all of the offered
resale shares are sold by such selling securityholder). |
The number of shares in the column ‘‘Number
of Shares Being Offered’’ represents all of the shares of our common stock that each selling securityholder may offer
under this prospectus. We do not know how long the selling securityholders will hold the shares before selling them or how many
shares they will sell, and we currently have no agreements, arrangements or understandings with any of the securityholders regarding
the sale of any of the resale shares. The shares of our common stock offered by this prospectus may be offered from time to time
by the securityholders listed below. We cannot assure you that any of the selling securityholders will offer for sale or sell
any or all of the shares of common stock offered by them by this prospectus.
| | |
Number
of Shares Beneficially Owned
Prior to Offering(1) | | |
Number
of Shares
Being | | |
Number
of Shares Beneficially Owned
After Offering(2) | |
| Securityholders | |
Number | | |
Percent
(%) | | |
Offered | | |
Number | | |
Percent
(%) | |
| Moshe Manor (3) | |
| 900,000 | | |
| * | | |
| 900,000 | | |
| — | | |
| — | |
| Yoseph Shaaltiel, Ph.D. (4) | |
| 1,529,644 | | |
| 1.6 | | |
| 943,728 | | |
| 585,916 | | |
| * | |
| Einat Brill Almon, Ph.D. (5) | |
| 876,272 | | |
| * | | |
| 876,272 | | |
| — | | |
| — | |
| Yossi Maimon (6) | |
| 740,000 | | |
| * | | |
| 740,000 | | |
| — | | |
| — | |
| Tzvi Palash (7) | |
| 387,000 | | |
| * | | |
| 387,000 | | |
| — | | |
| — | |
*less than 1%
(1) Applicable
percentage ownership is based on 93,602,152 shares of common stock outstanding as of May 1, 2015, plus any common stock equivalents
or convertible securities held and shares beneficially owned by each such holder as set forth herein.
(2) Assumes
that all shares of common stock to be offered, as set forth above, are sold pursuant to this offering and that no other shares
of common stock are acquired or disposed of by the selling securityholders prior to the termination of this offering. Because
the selling securityholders may sell all, some or none of their shares of common stock or may acquire or dispose of other shares
of common stock, no reliable estimate can be made of the aggregate number of shares of common stock that will be sold pursuant
to this offering or the number or percentage of shares of common stock that each selling securityholder will own upon completion
of this offering.
(3) Mr.
Manor is a director of our company and serves as our President and Chief Executive Officer. Consists of 900,000 shares of our
common stock issuable upon the exercise of vested and unvested options.
(4) Dr.
Shaaltiel serves as our Executive Vice President, Research and Development. Consists of 463,754 shares of our common stock, 733,728
shares of our common stock issuable upon the exercise of vested and unvested options and 210,000 vested and unvested restricted
shares of our common stock.
(5) Dr. Brill Almon
serves as our Senior Vice President, Product Development. Consists of 691,272 shares of our common stock issuable upon the exercise
of vested and unvested options and 185,000 vested and unvested restricted shares of our common stock.
(6) Mr.
Maimon serves as our Vice President, Chief Financial Officer. Consists of 555,000 shares of our common stock issuable upon the
exercise of vested and unvested options and 185,000 vested and unvested restricted shares of our common stock.
(7) Mr.
Palash serves as our Chief Operating Officer. Consists of 285,000 shares of our common stock issuable upon the exercise of vested
and unvested options and 285,000 vested and unvested restricted shares of our common stock.
PLAN OF DISTRIBUTION
The selling securityholders and any of their
respective pledgees, donees, assignees and other successors-in-interest may, from time to time, sell any or all of their shares
of our common stock on any stock exchange, market or trading facility on which the shares are traded or in private transactions.
These sales may be at fixed or negotiated prices. The selling securityholders may use any one or more of the following methods
when selling shares:
| · | ordinary
brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| · | block
trades in which the broker-dealer will attempt to sell the shares as agent, but may position
and resell a portion of the block as principal to facilitate the transaction; |
| · | purchases
by a broker-dealer as principal and resale by the broker-dealer for its account; |
| · | an
exchange distribution in accordance with the rules of the applicable exchange; |
| · | privately
negotiated transactions; |
| · | short
sales after this registration statement becomes effective; |
| · | broker-dealers
may agree with the selling securityholders to sell a specified number of such shares
at a stipulated price per share; |
| · | through
the writing of options on the shares; |
| · | a
combination of any such methods of sale; and |
| · | any
other method permitted pursuant to applicable law. |
The selling securityholders may also sell
shares under Rule 144 under the Securities Act of 1933, as amended, if available, rather than under this prospectus. The selling
securityholders will have the sole and absolute discretion not to accept any purchase offer or make any sale of shares if they
deem the purchase price to be unsatisfactory at any particular time.
The selling securityholders may also engage
in short sales against the box after this registration statement becomes effective, puts and calls and other transactions in our
securities or derivatives of our securities and may sell or deliver shares in connection with these trades.
The selling securityholders or their respective
pledges, donees, transferees or other successors in interest, may also sell the shares directly to market makers acting as principals
and/or broker-dealers acting as agents for themselves or their customers. Such broker-dealers may receive compensation in the
form of discounts, concessions or commissions from the selling securityholders and/or the purchasers of shares for whom such broker-dealers
may act as agents or to whom they sell as principal or both, which compensation as to a particular broker-dealer might be in excess
of customary commissions. Market makers and block purchasers purchasing the shares of common stock in block transactions to market
makers or other purchasers at a price per share which may be below the then market price. The selling securityholders cannot assure
that all or any of the shares offered in this prospectus will be issued to, or sold by, the selling securityholders. The selling
securityholders and any brokers, dealers or agents, upon effecting the sale of any of the shares offered in this prospectus, may
be deemed to be ‘‘underwriters’’ as that term is defined under the Securities Act or the Securities Exchange
Act of 1934, as amended, or the rules and regulations under such acts. In such event, any commissions received by such broker-dealers
or agents and any profit on the resale of the shares purchased by them may be deemed to be underwriting commissions or discounts
under the Securities Act.
Discounts, concessions, commissions and similar
selling expenses, if any, attributable to the sale of shares will be borne by the selling securityholders. The selling securityholders
may agree to indemnify any agent, dealer or broker-dealer that participates in transactions involving sales of the shares if liabilities
are imposed on that person under the Securities Act.
The selling securityholders may from time
to time pledge or grant a security interest in some or all of the shares of common stock owned by them and, if they default in
the performance of their secured obligations, the pledge or secured parties may offer and sell the shares of common stock from
time to time under this prospectus after we have filed an amendment to this prospectus under Rule 424(b)(3) or any other
applicable provision of the Securities Act amending the list of selling securityholders to include the pledge, transferee or other
successors in interest as selling securityholders under this prospectus.
The selling securityholders also may transfer
the shares of common stock in other circumstances, in which case the transferees, pledges or other successors in interest will
be the selling beneficial owners for purposes of this prospectus and may sell the shares of common stock from time to time under
this prospectus after we have filed an amendment to this prospectus under Rule 424(b)(3) or other applicable provision of the
Securities Act amending the list of selling securityholders to include the pledge, transferee or other successors in interest
as selling securityholders under this prospectus.
Each of the selling securityholders acquired
the securities offered hereby in the ordinary course of business and have advised us that they have not entered into any agreements,
understandings or arrangements with any underwriters or broker-dealers regarding the sale of their shares of common stock, nor
is there an underwriter or coordinating broker acting in connection with a proposed sale of shares of common stock by any selling
securityholder. If we are notified by any selling securityholder that any material arrangement has been entered into with a broker-dealer
for the sale of shares of common stock, if required, we will file a supplement to this prospectus. If the selling securityholders
use this prospectus for any sale of the shares of common stock, they will be subject to the prospectus delivery requirements of
the Securities Act.
The anti-manipulation rules of Regulation
M under the Exchange Act may apply to sales of our common stock and activities of the selling securityholders.
WHERE YOU CAN FIND MORE INFORMATION
This prospectus is part of a registration
statement on Form S-8 that we filed with the Commission under the Securities Act. You should rely only on the information contained
in this prospectus or incorporated by reference in this prospectus. We have not authorized anyone else to provide you with different
information. You should not assume that the information in this prospectus is accurate as of any date other than the date on the
front cover of this prospectus, regardless of the time of delivery of this prospectus or any sale of common stock.
We file annual, quarterly and current reports,
proxy statements and other information with the Commission. Our Commission filings, including the registration statement and exhibits,
are available to the public at the Commission’s website at http://www.sec.gov. You may also read, without charge, and copy
the documents we file, at the Commission’s public reference rooms at 100 F Street, N.E., Room 1580, Washington, D.C. 20549.
You can request copies of these documents by writing to the Commission and paying a fee for the copying cost. Please call the
Commission at 1-800-SEC-0330 for further information on the public reference rooms. In addition, since we are also listed on the
Tel Aviv Stock Exchange, we submit copies of all our filings with the Commission to the Israeli Securities Authority and the Tel
Aviv Stock Exchange. Such copies can be retrieved electronically through the Tel Aviv Stock Exchange’s internet messaging
system (www.maya.tase.co.il) and through the MAGNA distribution site of the Israeli Securities Authority (www.magna.isa.gov.il).
We maintain an Internet site at www.protalix.com.
Webcasts of presentations we make at certain conferences may also be available on our website from time to time. We have not incorporated
by reference into this prospectus the information on our website, and you should not consider it to be a part of this prospectus.
This prospectus does not
contain all of the information included in the registration statement. We have omitted certain parts of the registration statement
in accordance with the rules and regulations of the Commission. For further information, we refer you to the registration statement,
including its exhibits and schedules, which may be found at the Commission’s website at http://www.sec.gov. Statements
contained in this prospectus about the provisions or contents of any contract, agreement or any other document referred to are
not necessarily complete. Please refer to the actual exhibit for a more complete description of the matters involved.
INCORPORATION
OF CERTAIN INFORMATION BY REFERENCE
The Commission allows
us to “incorporate by reference” the information we file with the Commission, which means we can disclose important
information to you by referring you to those documents. The information we incorporate by reference is an important part of this
prospectus, and certain information that we will later file with the Commission will automatically update and supersede this information.
We incorporate by reference the documents listed below as well as any future filings made with the Commission under Sections 13(a),
13(c), 14 or 15(d) of the Exchange Act from the date of the initial registration statement and prior to the effectiveness of this
registration statement, and any filings made after the date of this prospectus until we sell all of the securities under this
prospectus, except that we do not incorporate any document or portion of a document that is “furnished” to the Commission,
but not deemed “filed.” The following documents filed with the Commission are incorporated by reference in this prospectus:
| · | our
Annual Report on Form 10-K, for the year ended December 31, 2014; |
| · | our
Quarterly Report on Form 10-Q for the quarter ended March 31, 2015; |
| · | our
Current Reports on Form 8-K filed with the Commission on January 5, 2105 (but not including
Item 2.02 furnished therewith); January 8, 2105; February 2, 2015; February 12, 2015;
and May 7, 2015 (but not including Item 2.02 furnished therewith); and |
| · | the
description of our common stock included in our registration statement on Form 8-A12B
(File No. 001-33357) filed with the Commission on March 9, 2007, including any amendment
or reports filed for the purpose of updating such description. |
Copies of these filings are available at
no cost on our website, www.protalix.com. In addition, you may request a copy of these filings and any amendments thereto at no
cost, by writing or telephoning us. Those copies will not include exhibits to those documents unless the exhibits are specifically
incorporated by reference in the documents or unless you specifically request them. You may also request copies of any exhibits
to the registration statement at no cost. Please direct your request to:
Yossi Maimon
2 Snunit Street, Science Park
P.O. Box 455
Carmiel, Israel 20100
+972-4-988-9488
You should rely only on the information in
this prospectus and the documents that are incorporated by reference. We have not authorized anyone else to provide you with different
information. We are not offering these securities in any state where the offering is prohibited by law. You should not assume
that the information in this prospectus or any incorporated document is accurate as of any date other than the date of the document.
LEGAL MATTERS
The validity of the issuance of the securities
offered by this prospectus will be passed upon for us by Morrison & Foerster LLP, New York, New York.
EXPERTS
The financial statements and management’s
assessment of the effectiveness of internal control over financial reporting (which is included in Management’s Report on
Internal Control over Financial Reporting) incorporated in this Prospectus by reference to the Annual Report on Form 10-K for
the year ended December 31, 2014 have been so incorporated in reliance on the report of Kesselman & Kesselman, certified public
accountants in Israel, a member firm of PricewaterhouseCoopers International Limited, an independent registered public accounting
firm, given on the authority of said firm as experts in auditing and accounting.
DISCLOSURE OF
COMMISSION POSITION ON INDEMNIFICATION
FOR SECURITIES ACT LIABILITIES
Insofar as indemnification
for liabilities arising under the Securities Act may be permitted to directors, officers or persons controlling the registrant,
the registrant has been informed that in the opinion of the Commission such indemnification is against public policy as expressed
in the Securities Act and is therefore unenforceable.
PART II
INFORMATION REQUIRED IN THE REGISTRATION
STATEMENT
Item 3. Incorporation
of Documents by Reference.
The Commission allows us to “incorporate
by reference” the information we file with the Commission, which means we can disclose important information to you by referring
you to those documents. The information we incorporate by reference is an important part of this prospectus, and certain information
that we will later file with the Commission will automatically update and supersede this information. We incorporate by reference
the documents listed below as well as any future filings made with the Commission under Sections 13(a), 13(c), 14 or 15(d) of
the Exchange Act after the date of this prospectus until we sell all of the securities under this prospectus, except that we do
not incorporate any document or portion of a document that is “furnished” to the Commission, but not deemed “filed.”
The following documents filed with the Commission are incorporated by reference in this prospectus:
| · | our
Annual Report on Form 10-K, for the year ended December 31, 2014; |
| · | our
Quarterly Report on Form 10-Q for the quarter ended March 31, 2015; |
| · | our
Current Reports on Form 8-K filed with the Commission on January 5, 2105 (but not including
Item 2.02 furnished therewith); January 8, 2105; February 2, 2015; February 12, 2015;
and May 7, 2015 (but not including Item 2.02 furnished therewith); and |
| · | the
description of our common stock included in our registration statement on Form 8-A12B
(File No. 001-33357) filed with the Commission on March 9, 2007, including any amendment
or reports filed for the purpose of updating such description. |
Copies of these filings are available at
no cost on our website, www.protalix.com. In addition, you may request a copy of these filings and any amendments thereto at no
cost, by writing or telephoning us. Those copies will not include exhibits to those documents unless the exhibits are specifically
incorporated by reference in the documents or unless you specifically request them. You may also request copies of any exhibits
to the registration statement at no cost. Please direct your request to:
Yossi Maimon
2 Snunit Street, Science Park
P.O. Box 455
Carmiel, Israel 20100
+972-4-988-9488
You should rely only on the information in
this prospectus and the documents that are incorporated by reference. We have not authorized anyone else to provide you with different
information. We are not offering these securities in any state where the offering is prohibited by law. You should not assume
that the information in this prospectus or any incorporated document is accurate as of any date other than the date of the document.
Item 4. Description
of Securities.
Not
applicable.
Item 5. Interests
of Named Experts and Counsel.
Not
applicable.
Item 6. Indemnification
of Directors and Officers.
We indemnify our directors and officers to
the maximum extent permitted by Florida law for the costs and liabilities of acting or failing to act in an official capacity.
In addition, we have entered into indemnification agreements with each of our executive officers and directors to provide them
with the maximum indemnification allowed under our amended and restated bylaws and applicable Florida law, including indemnification
for all judgments and expenses incurred as the result of any lawsuit in which such person is named as a defendant by reason of
being our director, officer or employee, to the extent indemnification is permitted by the laws of Florida. We believe that the
indemnification agreements will enhance our ability to continue to attract and retain qualified individuals to serve as directors
and officers.
The articles of association of Protalix Ltd.,
our wholly-owned subsidiary, allow it to exculpate, indemnify, and insure its office holders to the fullest extent permitted by
Israeli law, as described below. Accordingly, Protalix Ltd. has entered into indemnification agreements with each of its officers
and directors undertaking to indemnify them to the fullest extent permitted by Israeli law. As required by law, the indemnification
is limited to events determined as foreseeable by the board of directors of Protalix Ltd. based on the activities of Protalix
Ltd. upon adoption of the form of indemnification in 2006, and to an amount determined by the board of directors as reasonable
under the circumstances.
We purchase and maintain a directors and
officers liability insurance policy which covers the liability of our directors and officers on a "claims made" basis
for their ongoing activity since December 28, 2006, with a limit of $20,000,000 for any one occurrence and in the aggregate for
the annual period with the addition of 20% of the above limit for legal defense costs (in Israel).
Furthermore, we purchase and maintain a Side
A Excess and Difference In Conditions insurance which covers the non indemnifiable liability of our directors and officers on
a "claims made" basis for their ongoing activity since December 28, 2006, with a limit of $5,000,000 for any one occurrence
and in the aggregate for the annual period with the addition of 20% of the above limit for legal defense costs (in Israel).
As of the date of hereof, no claims for directors’
and officers’ liability insurance have been filed under those policies and neither us nor Protalix Ltd. is aware of any
pending or threatened litigation or proceeding involving any of our directors or officers, or those of Protalix Ltd., in which
indemnification is sought.
We have undertaken to fulfill and honor in
all respects the obligations of Protalix Ltd. pursuant to any indemnification agreements between Protalix Ltd. and its directors
in effect prior to December 31, 2006 and to provide any substitute director and the officers of Protalix Ltd. with similar
indemnification agreements. We further agreed that any provision of Protalix Ltd.’s charter documents that relate to exculpation
and indemnification of officers and directors of Protalix Ltd. will not be amended, repealed, or otherwise modified in any manner
that would adversely affect the rights of the directors and officers, unless such modification is required by any applicable law.
Under Israeli law, an Israeli company may
not exculpate an office holder from liability for a breach of the duty of loyalty of the office holder. An Israeli company may
exculpate an office holder in advance from liability, in whole or in part, for damages caused due to a breach of duty of care
owed to the company (other than in the event that such liability arises out of a breach of duty of care to the company upon distribution)
but only if a provision authorizing such exculpation is inserted in its articles of association. Protalix Ltd.’s articles
of association include such a provision.
An Israeli company may indemnify an office
holder in respect of certain liabilities or expenses either in advance of an event or following an event provided a provision
authorizing such indemnification is inserted in its articles of association. Protalix Ltd.’s articles of association contain
such an authorization. An Israeli company may indemnify an office holder against the following liabilities or expenses incurred
for acts performed as an office holder:
(A) financial
liability imposed on or incurred by him or her in favor of another person pursuant to a judgment, settlement or arbitrator’s
award approved by a court;
(B) reasonable
litigation expenses, including attorneys’ fees, incurred by the office holder as a result of an investigation or proceeding
instituted against him or her by an authority authorized to conduct such investigation or proceeding, provided that (i) no indictment
was filed against such office holder as a result of such investigation or proceeding; and (ii) no financial liability, such as
a criminal penalty, was imposed upon him or her as a substitute for the criminal proceeding as a result of such investigation
or proceeding or, if such financial liability was imposed, it was imposed with respect to an offense that does not require proof
of criminal intent or with regard to a monetary sanction;
(C) reasonable
litigation expenses, including attorneys’ fees, incurred by the office holder or imposed by a court in proceedings instituted
against him or her by the company, on its behalf or by a third party or in connection with criminal proceedings in which the office
holder was acquitted or as a result of a conviction for a crime that does not require proof of criminal intent; and
(D) A
payment which the office holder is obligated to make to an injured party as set forth in Section 52(54)(a)(1)(a) of the Israeli
Securities Law, 5278-1968, as amended (“Israeli Securities Law”), and reasonable litigation expenses, including attorneys’
fees, that the office holder incurred in connection with a proceeding under Chapters H’3, H’4 or I’1 of the
Israeli Securities Law.
An undertaking provided in advance by an
Israeli company to indemnify an office holder with respect to (A), as abovementioned, must be limited to events which, in the
opinion of the board of directors, can be foreseen based on the company’s activities when the undertaking to indemnify is
given, and to an amount or according to criteria determined by the board of directors as reasonable under the circumstances, and
such undertaking shall detail the abovementioned events and amount or criteria. We are permitted to provide an office holder advanced
payments to cover expenses covered under (D) above.
An Israeli company may insure an office holder
against the following liabilities incurred for acts performed as an office holder:
| · | a
breach of duty of care to the company or to a third party; |
| · | a
breach of duty of loyalty to the company, to the extent that the office holder acted
in good faith and had a reasonable basis to believe that the act would not be detrimental
to the interests of the company; |
| · | a
financial liability imposed on the office holder in favor of a third party in respect
of an act performed in his or her capacity as an office holder; and |
| · | A
payment which the office holder is obligated to make to an injured party as set forth
in Section 52(54)(a)(1)(a) of the Israeli Securities Law and reasonable litigation expenses,
including attorneys’ fees, that the office holder incurred in connection with a
proceeding under Chapters H’3, H’4 or I’1 of the Israeli Securities
Law. |
An Israeli company may not insure, indemnify
or exculpate an office holder against any of the following:
| · | a
breach of duty of loyalty, except to the extent that the office holder acted in good
faith and had a reasonable basis to believe that the act would not be detrimental to
the interests of the company; |
| · | a
grossly negligent or intentional violation of an office holder’s duty of care; |
| · | an
act or omission committed with intent to derive illegal personal benefit; or |
| · | a
fine, civil fine, monetary sanction or ransom levied against the office holder. |
Under the Israeli law, exculpation, indemnification,
and insurance of office holders must be approved by the board of directors of Protalix Ltd. and, in respect of directors of Protalix
Ltd., also by the shareholders of Protalix Ltd. Such approvals were properly obtained.
Insofar as indemnification for liabilities
arising under the Securities Act may be permitted to our directors and officers or persons controlling us pursuant to the foregoing
provisions, or otherwise, we have been advised that, in the opinion of the Commission, such indemnification is against public
policy as expressed in the Securities Act, and is, therefore, unenforceable. With respect to insurance and indemnification for
liabilities and expenses arising under the Israeli Securities Law, such insurance and indemnification is permissible only to the
extent described in this Item 6.
We also have purchased insurance in the aggregate
amount of $1,000,000 for our directors and officers against all of the costs of such indemnification or against liabilities arising
from acts or omissions of the insured person in cases where we may not have power to indemnify the person against such liabilities.
Such policy will be in a run-off “tail” coverage phase as of December 31, 2006 and will cover those individuals who
were our officers and directors prior to the merger we effected on such date for a period of six-years after such individual resigned
his or her position with our company.
Item 7. Exemption from
Registration Claims.
Not
applicable.
Item 8. Exhibits.
Exhibit
Number |
|
Exhibit Description |
|
Method of Filing |
| |
|
|
|
|
| 4.1 |
|
Protalix BioTherapeutics, Inc. 2006 Stock Incentive Plan, as amended November 10, 2014 |
|
(Incorporated herein by reference to Appendix A to our Definitive Proxy Statement on Schedule 14A (File No. 001-33357),
filed with the Commission on October 9, 2014) |
| |
|
|
|
|
| 5.1 |
|
Opinion of Morrison & Foerster LLP, New York, New York as to the legality of the securities
being registered |
|
Filed herewith |
| |
|
|
|
|
| 23.1 |
|
Consent of Morrison & Foerster LLP, New York, New York (included in Exhibit 5.1) |
|
Filed herewith |
| |
|
|
|
|
| 23.2 |
|
Consent of Kesselman & Kesselman, Certified Public Accountant (Isr.), a member of PricewaterhouseCoopers
International Limited, independent registered public accounting firm for the Registrant |
|
Filed herewith |
| |
|
|
|
|
| 24.1 |
|
Power of Attorney (included on signature page) |
|
Filed herewith |
Item 9. Undertakings.
The undersigned registrant
hereby undertakes:
(1) to
file, during any period in which offers or sales are being made, a post-effective amendment to this Registration Statement;
(i) to
include any prospectus required by Section 10(a)(3) of the Securities Act;
(ii) to
reflect in the prospectus any facts or events arising after the effective date of the Registration statement (or most recent post-effective
amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the
Registration Statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total
dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the
estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to Rule 424(b)
if, in the aggregate, the change in volume and price represent no more than a 20 percent change in the maximum aggregate offering
price set forth in the “Calculation of Registration Fee” table in the effective Registration Statement; and
(iii) to
include any material information with respect to the plan of distribution not previously disclosed in this Registration Statement
or any material change to such information in this Registration Statement.
provided, however, that paragraphs
(1)(i) and (1)(ii) of this section do not apply if the information required to be included in a post-effective amendment by those
paragraphs is contained in reports filed with or furnished to the Commission by the registrant pursuant to Section 13 or Section
15(d) of the Exchange Act that are incorporated by reference in the Registration Statement;
(2) that,
for the purpose of determining any liability under the Securities Act, each such post-effective amendment shall be deemed to be
a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall
be deemed to be the initial bona fide offering thereof; and
(3) to
remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at
the termination of the offering.
The undersigned registrant
hereby undertakes that, for purposes of determining any liability under the Securities Act, each filing of the registrant’s
annual report pursuant to Section 13(a) or Section 15(d) of the Exchange Act (and, where applicable, each filing of an employee
benefit plan’s annual report pursuant to Section 15(d) of the Exchange Act) that is incorporated by reference in the Registration
Statement shall be deemed to be a new registration statement relating to the securities offered herein, and the offering of such
securities at that time shall be deemed to be the initial bona fide offering thereof.
Insofar as indemnification
for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant
pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Commission such
indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that
a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by
a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted
by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless
in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction
the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed
by the final adjudication of such issue.
SIGNATURES
Pursuant to the requirements of the Securities
Act of 1933, as amended, the Registrant certifies that it has reasonable grounds to believe that it meets all of the requirements
for filing on Form S-8 and has duly caused this Registration Statement to be signed on its behalf by the undersigned, thereunto
duly authorized, in the City of Tel Aviv, State of Israel, on May 7, 2015.
| |
PROTALIX BIOTHERAPEUTICS, INC. |
| |
By: |
/s/ Moshe Manor |
| |
|
Moshe Manor |
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that
each person whose signature appears below constitutes and appoints Moshe Manor and Yossi Maimon, and each of them, as his true
and lawful attorneys-in-fact and agents, with full power of substitution and resubstitution, for the undersigned and in his or
her name, place and stead, in any and all capacities, to sign any or all amendments (including post-effective amendments) to the
Registration Statement and to file the same, with all exhibits thereto, and all documents in connection therewith, including any
subsequent registration statement for the same offering that may be filed under Rule 462(b), with the Securities and Exchange
Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each
and every act and thing requisite and necessary to be done in connection therewith, as fully to all intents and purposes as he
or she might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents, or any of them
or their or his substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities
Act of 1933, the following persons in the capacities and on the dates indicated have signed this Registration Statement below
on behalf of the Registrant.
| /s/ Moshe Manor |
President, Chief Executive Officer |
May 7, 2015 |
| Moshe Manor. |
(Principal Executive Officer) and Director |
|
| |
|
|
| /s/ Yossi Maimon |
Chief Financial Officer, |
May 7, 2015 |
| Yossi Maimon |
Treasurer and Secretary
(Principal Financial and Accounting
Officer) |
|
| |
|
|
| /s/ Shlomo Yanai |
Chairman of the Board |
May 7, 2015 |
| Shlomo Yanai |
|
|
| |
|
|
| /s/ Zeev Bronfeld |
Director |
May 7, 2015 |
| Zeev Bronfeld |
|
|
| |
|
|
| /s/ Amos Bar Shalev |
Director |
May 7, 2015 |
| Amos Bar Shalev |
|
|
| |
|
|
| /s/ Yodfat Harel Buchris |
Director |
May 7, 2015 |
| Yodfat Harel Buchris |
|
|
| |
|
|
INDEX TO EXHIBITS
Exhibit
Number |
|
Exhibit Description |
|
Method of Filing |
| |
|
|
|
|
| 4.1 |
|
Protalix BioTherapeutics, Inc. 2006 Stock Incentive Plan, as amended November 10, 2014 |
|
(Incorporated herein by reference to Appendix A to our Definitive Proxy Statement on Schedule 14A (File No. 001-33357),
filed with the Commission on October 9, 2014) |
| |
|
|
|
|
| 5.1 |
|
Opinion of Morrison & Foerster LLP, New York, New York as to the legality of the securities
being registered |
|
Filed herewith |
| |
|
|
|
|
| 23.1 |
|
Consent of Morrison & Foerster LLP, New York, New York (included in Exhibit 5.1) |
|
Filed herewith |
| |
|
|
|
|
| 23.2 |
|
Consent of Kesselman & Kesselman, Certified Public Accountant (Isr.), a member of PricewaterhouseCoopers
International Limited, independent registered public accounting firm for the Registrant |
|
Filed herewith |
| |
|
|
|
|
| 24.1 |
|
Power of Attorney (included on signature page) |
|
Filed herewith |
Exhibit 5.1
[LETTERHEAD OF MORRISON
& FOERSTER LLP]
May 7, 2015
Protalix BioTherapeutics, Inc.
2 Snunit Street
Science Park
POB 455
Carmiel 20100, Israel
| Re: | Protalix BioTherapeutics,
Inc. – Registration Statement on Form S-8 |
Ladies and Gentlemen:
We have acted as counsel
to Protalix BioTherapeutics, Inc., a Florida corporation (the “Company”), in connection with the preparation and filing
with the Securities and Exchange Commission (the “Commission”) of a Registration Statement on Form S-8 (the “Registration
Statement”), relating to the registration by the Company under the Securities Act of 1933, as amended (the “Securities
Act”), of up to 13,841,655 shares of its common stock, $.001 par value per share (the “Common Stock”), which
will be issuable under the Protalix BioTherapeutics, Inc. 2006 Stock Incentive Plan, as amended on November 10, 2014 (the “Plan”).
This opinion is being rendered solely in connection with the registration of the offering and sale of the shares of common stock
under the Plan (the “Plan Shares”) under the registration requirements of the Securities Act.
As your counsel in
connection with the Registration Statement, we have examined the proceedings taken by you in connection with the adoption of the
Plan and the authorization of the issuance of the Plan Shares, and such documents as we have deemed necessary to render this opinion.
For the purpose of the opinion rendered below, we have assumed that in connection with the issuance of the Plan Shares, the Company
will receive consideration in an amount not less than the aggregate par value of the Plan Shares covered by each such issuance.
In our examination, we have assumed the genuineness of all signatures, the legal capacity of natural persons, the authenticity
of all documents submitted to us as originals, the conformity to original documents of all documents submitted to us as certified
or photostatic copies and the authenticity of the originals of such copies.
Based upon and subject
to the foregoing, it is our opinion that the Plan Shares, when issued and outstanding pursuant to the terms of the Plan, will
be validly issued, fully paid and nonassessable.
Please note that we
are opining only as to the matters expressly set forth herein and that no opinion should be inferred as to any other matter. In
rendering the foregoing opinion, we have relied, for matters involving Florida law, solely on the opinion of Shutts & Bowen
LLP, Miami, Florida. This opinion is based upon currently existing statutes, rules, regulations and judicial decisions, and we
disclaim any obligation to advise you of any change in any of these sources of law or subsequent legal or factual developments
which might affect any matters or opinions set forth herein.
We hereby consent
to the use of our name under the heading “Legal Matters” in the Registration Statement to be filed by the Company
with the Commission. We further consent to your filing a copy of this opinion as Exhibit 5.1 to the
Registration Statement. In giving such permission, we do not admit hereby that we come within the category of persons whose consent
is required under Section 7 of the Securities Act, or the rules and regulations of the Commission thereunder. We disclaim any
undertaking to advise you of any subsequent changes of the facts stated or assumed herein or any subsequent changes in applicable
law.
Very truly yours,
/s/ Morrison & Foerster LLP
Exhibit 23.2

CONSENT OF INDEPENDENT
REGISTERED PUBLIC ACCOUNTING FIRM
We hereby consent to
the incorporation by reference in this Registration Statement on Form S-8 of our report dated March 12, 2015 relating to the financial
statements and the effectiveness of internal control over financial reporting, which appears in Protalix BioTherapeutics, Inc.’s
Annual Report on Form 10-K for the year ended December 31, 2014. We also consent to the reference to us under the heading “Experts”
in such Registration Statement.
| |
/s/ Kesselman & Kesselman |
| Tel-Aviv, Israel |
Kesselman & Kesselman |
| May 7, 2015 |
Certified Public Accountants (Isr.) |
| |
A member firm of PricewaterhouseCoopers
International Limited |
Kesselman & Kesselman, Trade Tower, 25 Hamered Street,
Tel-Aviv 68125, Israel,
P.O Box 50005 Tel-Aviv 61500 Telephone: +972 -3- 7954555,
Fax:+972 -3- 7954556, www.pwc.com/il
Protalix BioTherapeutics (AMEX:PLX)
Historical Stock Chart
From Apr 2024 to May 2024
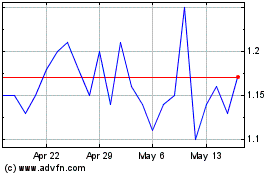
Protalix BioTherapeutics (AMEX:PLX)
Historical Stock Chart
From May 2023 to May 2024