As
filed with the Securities and Exchange Commission on October 22, 2020
Registration
No. 333-249401
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
AMENDMENT NO.
1
TO
FORM S-4
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
Zomedica Corp.
(Exact Name of Registrant as Specified in Its Charter)
Alberta, Canada*
(State or Other Jurisdiction
of Incorporation or Organization)
|
|
8071
(Primary Standard Industrial
Classification Code Number)
|
|
Not Applicable**
(I.R.S. Employer
Identification Number)
|
100 Phoenix
Drive, Suite 180
Ann Arbor, Michigan,
48108
(734) 369-2555
(Address, including
zip code, and telephone number, including area code,
of Registrant's principal executive offices)
Ann Marie Cotter
Chief Financial Officer
Zomedica Corp.
100 Phoenix Drive, Suite 180
Ann Arbor, Michigan 48108
(734) 369-2555
(Name, address, including zip code, and
telephone number, including area code, of agent for service)
Copies to:
|
John D. Hogoboom
Lowenstein Sandler LLP
1251 Avenue of the Americas New York, NY
10020
(646) 414-6846
|
K. Vanessa A. Grant
Andrea Brewer
Norton Rose Fulbright Canada LLP / S.E.N.C.R.L.,
s.r.l.
222 Bay Street, Suite 3000, P.O. Box 53, Toronto ON M5K 1E7 Canada
(416) 216-4056
|
Approximate date of commencement of
proposed sale of the securities to the public: As soon as practicable after this Registration Statement becomes effective and
the consummation of the domestication transaction covered hereby.
If the securities being
registered on this Form are being offered in connection with the formation of a holding company and there is compliance with General
Instruction G, check the following box. ☐
If this Form is filed
to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following box
and list the Securities Act registration statement number of the earliest effective registration statement for the same offering. ☐
If this Form is a post-effective
amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration
statement number of the earlier effective registration statement for the same offering. ☐
Indicate by check mark
whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company,
or emerging growth company. See the definitions of "large accelerated filer," "accelerated filer," "smaller
reporting company" and "emerging growth company" in Rule 12b-2 of the Exchange Act:
|
Large accelerated filer ☐
|
Accelerated filer ☐
|
|
Non-accelerated filer ☒
|
Smaller reporting company ☒
|
|
|
Emerging growth company ☒
|
If an emerging growth
company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any
new or revised financial accounting standards provided pursuant to Section 7(a)(2)(B) of the Securities Act. ☒
_______________________________
The
registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until
the registrant files a further amendment that specifically states that this registration statement shall thereafter become
effective in accordance with Section 8(a) of the Securities Act of 1933, as amended, or until the registration statement shall
become effective on such date as the Securities and Exchange Commission, acting pursuant to said Section 8(a), may determine.
The information contained herein
is subject to completion or amendment. A registration statement relating to these securities has been filed with the Securities
and Exchange Commission. These securities may not be sold nor may offers to buy be accepted prior to the time the registration
statement becomes effective. This document shall not constitute an offer to sell or the solicitation of any offer to buy nor shall
there be any sale of these securities in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to
registration or qualification under the securities laws of any such jurisdiction.
PRELIMINARY—SUBJECT TO
COMPLETION—DATED AS OF OCTOBER 22, 2020
ZOMEDICA CORP.
PROPOSED DOMESTICATION—YOUR VOTE
IS VERY IMPORTANT
Dear Shareholders:
We
are furnishing this proxy circular/prospectus to shareholders of Zomedica Corp. in connection with the solicitation of proxies
by our management for use at a special meeting of our shareholders, or the Special Meeting, to be held on December 15, 2020 at 8:30 a.m. (Toronto
time). Due to concerns regarding the coronavirus outbreak and to assist in protecting the health and well-being of our shareholders
and employees, we are holding the Special Meeting as a virtual-only meeting, which will be conducted via live audio online webcast,
where registered shareholders and duly appointed proxyholders, regardless of geographic location, will have an opportunity to participate
at the Special Meeting. Shareholders will not be able to attend the Special Meeting in person. Registered shareholders and duly
appointed proxyholders will be able to attend and participate at the Special Meeting online at https://web.lumiagm.com/157200202,
password “zomedica2020”. Online access begins at 7:30 a.m. (Toronto time).
The primary purpose of the Special Meeting is
to obtain shareholder approval to change our jurisdiction of incorporation from the Province of Alberta in Canada to the State
of Delaware in the United States of America. We refer to this transaction as the ‘‘domestication’’ throughout
this letter and the proxy circular/prospectus that accompanies it. We are also seeking common shareholder approval of the adoption
of the Zomedica Corp. 2020 Stock Incentive Plan, or the 2020 Plan.
We are pursuing the
domestication for a number of reasons. Our domestication is intended to reduce the regulatory burden and cost of being subject
to the laws and regulations of both the United States and Canada and to enhance shareholder value over the long term by, among
other things, reducing our operating costs and enhancing the marketability of our capital stock by becoming a Delaware corporation.
In addition, our corporate offices and operations are located in the United States and a large percentage of our shareholders are
located there. We believe that the domestication will save approximately $500,000 per year in duplicative, unnecessary compliance
costs, which will directly benefit shareholders.
We chose the State
of Delaware to be our domicile principally because the Delaware General Corporation Law, or DGCL, expressly accommodates a continuance
authorized by Section 189 of the Business Corporations Act (Alberta), or the ABCA. We also chose the State of Delaware because
of the substantial body of case law that has evolved over the years interpreting various provisions of the DGCL.
If we complete the domestication, we will continue
our legal existence in Delaware as if we had originally been incorporated under Delaware law. In addition, each outstanding common
share and each outstanding Series 1 preferred share of Zomedica Corp. as an Alberta corporation will then represent one share of
common stock or one share of Series 1 preferred stock, respectively, of Zomedica Corp. as a Delaware corporation. Our common shares
are currently traded and will continue to be traded on the NYSE American exchange under the symbol “ZOM.”
After the domestication, our management will comprise the same directors and executive officers who served in such capacities immediately
prior to the domestication.
The record date for
the determination of shareholders entitled to receive notice of, and to vote at, the Special Meeting is October 22, 2020.
At such date, 564,051,438 common shares and 12 Series 1 preferred shares were outstanding. The holders of at least two thirds
of our common shares and Series 1 preferred shares present at the Special Meeting virtually or by proxy (and assuming a quorum
of our outstanding shares are represented at the Special Meeting virtually or by proxy) must vote together as a single class to
approve the domestication proposal.
Dissenting shareholders
have the right to dissent from the proposed domestication and, if they follow the procedures specified in the ABCA, to be paid
the fair value of their shares in accordance with Section 191 of the ABCA. If approved by our shareholders, the domestication is
expected to become effective as soon as practicable after the Special Meeting. Our Board of Directors has reserved the right to
terminate or abandon our domestication at any time prior to its effectiveness, notwithstanding shareholder approval, if it determines
for any reason that the consummation of our domestication would be inadvisable or not in our best interests. In addition, the holders
of a majority of our common shares present at the Special Meeting virtually or by proxy (and assuming a quorum of our outstanding
common shares are represented at the Special Meeting virtually or by proxy) must vote to approve the adoption of the 2020 Plan.
Your existing certificates representing your
Zomedica Corp. common shares and Series 1 preferred shares will represent the same number of shares of Zomedica Corp. common stock
and Series 1 preferred stock, respectively, after the domestication without any action on your part. You will not have to exchange
any share certificates. We will issue new certificates to you representing shares of common stock or preferred stock of Zomedica
Corp. as a Delaware corporation upon a transfer of the shares by you or at your request.
This proxy circular/prospectus
provides a detailed description of our proposed domestication and other information to assist you in considering the proposals
on which you are being asked to vote. We urge you to review this information carefully and, if you require assistance, to consult
with your financial, tax or other professional advisers.
For the reasons set
forth in this proxy circular/prospectus, our Board of Directors unanimously believes that the proposed domestication is in our
best interests and in the best interests of our shareholders. Our Board of Directors unanimously recommends that you vote FOR
approval of our domestication and FOR approval of the adoption of the 2020 Plan.
Your vote is very important.
Only registered shareholders or their duly appointed proxyholders, may participate at the Special Meeting. Please review this proxy
circular/prospectus, which contains important information about the Special Meeting and the voting process.
We appreciate your
continued interest in our company.
Very truly yours,
/s/ Robert Cohen
Robert Cohen, Interim Chief Executive Officer
We
are an “emerging growth company” as defined under the federal securities laws and, as such, may elect to comply with
certain reduced public company reporting requirements for future filings. Investing in our common stock involves a high degree
of risk. See “Risk Factors” beginning on page 14 of this proxy circular/prospectus for a discussion of specified matters
that should be considered.
Neither the Securities
and Exchange Commission nor any state securities commission or similar authority in Canada has approved or disapproved of these
securities or determined if this proxy circular/prospectus is truthful or complete. Any representation to the contrary is a criminal
offense.
This proxy circular/prospectus is dated October ,
2020, and is first being mailed to shareholders on or about October , 2020.
ZOMEDICA
CORP.
100 Phoenix Drive, Suite 180
Ann Arbor, Michigan 48108
(734) 369-2555
NOTICE OF VIRTUAL-ONLY SPECIAL MEETING
OF SHAREHOLDERS
TO BE HELD ON DECEMBER 15, 2020
TAKE NOTICE that
the virtual-only special meeting (the “Special Meeting”) of holders of Common Shares (the “Common Shares”)
and Series 1 Preferred Shares (the “Series 1 Preferred Shares”) of ZOMEDICA CORP. (“Zomedica” or the “Corporation”)
will be held on December 15, 2020 at 8:30 a.m. (Toronto time). Due to social distancing measures related to the coronavirus outbreak
and to assist in protecting the health and well-being of our shareholders and employees, the Corporation is holding the Special
Meeting as a virtual-only meeting, which will be conducted via live audio online webcast, where registered shareholders and duly
appointed proxyholders, regardless of geographic location, will have an opportunity to participate at the Special Meeting. Shareholders
will not be able to attend the Special Meeting in person. Registered shareholders and duly appointed proxyholders will be able
to attend and participate at the Special Meeting online at https://web.lumiagm.com/157200202, password “zomedica2020”.
Online access begins at 7:30 a.m. (Toronto time).
The Special Meeting
will cover the following items of business:
|
|
(a)
|
to consider, and if deemed advisable, approve a special resolution authorizing the Corporation
to make an application under Section 189 of the Business Corporations Act (Alberta) to change its jurisdiction of incorporation
from the Province of Alberta, Canada to the State of Delaware, United States of America, and to approve the certificate of incorporation
authorized in the special resolution to be effective as of the date of the Corporation’s domestication (‘‘Proposal
1’’ or the ‘‘domestication’’);
|
|
|
(b)
|
to consider and vote upon a proposal to approve, assuming the domestication proposal is approved,
the Zomedica Corp. 2020 Stock Incentive Plan, or the 2020 Plan, a copy of which is attached to the proxy circular/prospectus as
Exhibit G (‘‘Proposal 2’’ or the ‘‘incentive plan proposal’’);
and
|
|
|
(c)
|
to transact such further or other business as may properly come before the Special Meeting or any
adjournment(s) or postponement(s) thereof.
|
The board of directors
of the Corporation has fixed the close of business on October 22, 2020 as the record date for determination of shareholders entitled
to notice of the Special Meeting, or any adjournment or postponement thereof, and the right to vote thereat. At such date, 564,051,438
Common Shares and 12 Series 1 Preferred Shares were outstanding. In accordance with Section 189(3) of the ABCA, each share of the
Corporation carries the right to vote in respect the domestication whether or not it otherwise carries the right to vote. As such,
the holders of at least two thirds of our Common Shares and Series 1 Preferred Shares present at the Special Meeting virtually
or by proxy (and assuming a quorum of our outstanding shares are represented at the Special Meeting virtually or by proxy) must
vote together as a single class to approve the domestication proposal. The holders of a majority of our Common Shares present at
the Special Meeting virtually or by proxy (and assuming a quorum of our outstanding common shares are represented at the Special
Meeting virtually or by proxy) must vote to approve the incentive plan proposal. No cumulative voting rights are authorized.
Accompanying this Notice is the proxy circular/prospectus dated October ,
2020 of Zomedica (the “proxy circular/prospectus”) in respect of the Special Meeting, which includes information relating
to matters to be addressed at the Special Meeting, together with an Instrument of Proxy (the “Instrument of Proxy”).
Registered shareholders who are unable to participate in the Special Meeting are requested to complete, sign, date and return the
enclosed Instrument of Proxy. An Instrument of Proxy will not be valid unless it is deposited at the office of AST Trust Company
(Canada), not less than forty-eight (48) hours (excluding Saturdays, Sundays and statutory holidays) before the time for holding
the Special Meeting or any adjournment(s) or postponement(s) thereof.
The enclosed Instrument
of Proxy appoints nominees of management as proxyholder and you may amend the Instrument of Proxy, if you wish, by inserting in
the space provided the name of the person you wish to represent you as proxyholder at the Special Meeting.
If you are a non-registered
holder of Common Shares and received these materials through your broker or another intermediary (each, an “Intermediary”),
please complete and return the voting instruction form provided by your Intermediary in accordance with instructions provided to
you by your Intermediary.
Only registered shareholders
or their duly appointed proxyholders, may participate at the Special Meeting. Please review the proxy circular/prospectus, which
contains important information about the Special Meeting and the voting process.
|
|
DATED:OCTOBER , 2020
|
|
|
|
|
|
By Order of the Board of Directors
|
|
|
|
|
|
(signed)”Robert Cohen”
|
|
|
|
|
|
Robert Cohen
|
|
|
Interim Chief Executive Officer
|
Proxy Circular/ Prospectus
TABLE OF CONTENTS
PAGES
|
Summary
|
|
|
QUESTIONS AND ANSWERS ABOUT THE PROPOSALS
|
3
|
|
PROPOSAL 1 – THE DOMESTICATION PROPOSAL
|
5
|
|
Accounting Treatment of the Domestication
|
8
|
|
DISSENTING RIGHTS OF SHAREHOLDERS
|
8
|
|
COMPARISON OF SHAREHOLDER RIGHTS
|
8
|
|
PROPOSAL 2 – THE STOCK INCENTIVE PLAN PROPOSAL
|
8
|
|
Risk Factors
|
14
|
|
CAUTIONARY NOTE REGARDING Forward-Looking Statements
|
40
|
|
Business
|
41
|
|
Market PRICE OF OUR Common SHARES and Related SHAREholder Matters
|
50
|
|
Management’s Discussion and Analysis of Financial Condition and Results of Operation
|
51
|
|
MANAGEMENT
|
63
|
|
EXECUTIVE AND DIRECTOR COMPENSATION
|
67
|
|
Securities Ownership of Certain beneficIal owners and management
|
73
|
|
Certain Relationships and Related PARTY Transactions
|
75
|
|
THE SPECIAL MEETING
|
77
|
|
VOTING SECURITIES AND PRINCIPAL HOLDERS OF VOTING SECURITIES
|
82
|
|
The Domestication
|
84
|
|
Description of Capital Stock
|
111
|
|
INTEREST OF CERTAIN PERSONS OR COMPANIES IN MATTERS TO BE ACTED ON
|
113
|
|
Interest of INFORMED PERSONS IN MATERIAL TRANSACTIONS
|
114
|
|
Legal Matters
|
114
|
|
Experts
|
114
|
|
other business
|
114
|
|
WHERE YOU CAN FIND MORE INFORMATION
|
114
|
|
INDEX TO FINANCIAL STATEMENTS
|
F-1
|
|
EXHIBIT A – SPECIAL RESOLUTION
|
A-1
|
|
EXHIBIT B – FORM OF CERTIFICATE of CORPORATE DOMESTICATION
|
B-1
|
|
EXHIBIT C – FORM OF CERTIFICATE OF INCORPORATION
|
C-1
|
|
EXHIBIT D – FORM OF BYLAWS
|
D-1
|
|
EXHIBIT E – SECTION 191 of the BUSINESS CORPORATIONS ACT (ALBERTA)
|
E-1
|
|
EXHIBIT F – FORM OF PROXY CARD
|
F-1
|
|
EXHIBIT G – 2020 STOCK INCENTIVE PLAN
|
G-1
|
ZOMEDICA CORP.
PROXY CIRCULAR/PROSPECTUS
SUMMARY
This summary highlights
selected information appearing elsewhere in this proxy circular/prospectus and does not contain all the information that you should
consider in making a decision with respect to the proposals described herein. You should read this summary, together with the more
detailed information, including our financial statements and the related notes appearing elsewhere in this proxy circular/prospectus,
and the exhibits attached hereto. You should carefully consider, among other things, the matters discussed in ‘‘Risk
Factors’’ and ‘‘Management’s Discussion and Analysis of Financial Condition and Results of Operations,’’
which are included in this proxy circular/prospectus. You should read this proxy circular/prospectus in its entirety.
Zomedica
Corp.
We are a veterinary
health company creating products for companion animals by focusing on the unmet needs of clinical veterinarians. We expect that
our product portfolio will include innovative diagnostics and medical devices that emphasize patient health and practice health.
With a team that includes clinical veterinary professionals, our goal is to provide veterinarians the opportunity to increase productivity
and grow revenue while better serving the animals in their care.
Our strategic focus
is on the final development and commercialization of our TRUFORMA™ diagnostic biosensor platform and the first five assays
for the detection of adrenal and thyroid disorders in cats and dogs. The TRUFORMA™ platform uses Bulk Acoustic Wave (BAW)
technology to provide a non-optical and fluorescence free detection system for use at the point-of-care. We believe that BAW technology
will enable precise and repeatable test results at the point-of-care during a typical veterinary appointment. We believe that the
TRUFORMA™ diagnostic platform does not require pre-market regulatory approval for use with companion animals in the United
States.
Following the commercial
launch of TRUFORMA™, we expect to continue the development of another point-of-care diagnostic platform, which is based on
miniaturized laser-based Raman spectroscopy technology and is designed to detect pathogens in companion animals. We believe this
platform will enable the identification of biological and biochemical signatures in complex biological samples and has the potential
to achieve reference lab sensitivity/specificity to screen for a wide range of pathogens in companion animal feces, urine, respiratory,
and dermatological samples in minutes without the need for extensive sample prep or the use of reagents. The diagnostic platform
requires a small fecal sample preparation. Additionally, the platform has automated analysis and does not require specialized staff
training. Assuming development work is successfully completed we expect the commercial launch of our fecal test to occur by 2022
and urine tests by 2023. We believe that this diagnostic platform does not require pre-market regulatory approval for use with
companion animals in the United States.
We have performed initial
development work on a circulating tumor cell (CTC) “liquid biopsy” platform for use in a reference lab setting as a
canine cancer diagnostic. This platform is intended for use to detect canine cancers faster, more affordably and less invasively
compared to existing methods, which can be expensive and cost-prohibitive for pet owners. We have worked on the development of
an assay for use with this platform that targets hard-to-diagnose canine cancers, such as hemangiosarcoma and osteosarcoma.
Consistent with our
focus on the development of point-of-care diagnostic platforms, we intend to seek one or more partners for the further development
and commercialization of the liquid biopsy platform.
Corporate
Information
Zomedica Corp. (formerly,
Wise Oakwood Ventures Inc.) was originally incorporated as Wise Oakwood Ventures Inc. on January 7, 2013 under the Business Corporations
Act (Alberta). On October 28, 2013, we completed our initial public offering in Canada and became classified as a Capital Pool
Company, as defined under the rules of the TSX Venture Exchange (“TSX-V”). On April 21, 2016, we changed our name to
Zomedica Pharmaceuticals Corp. and consolidated our common shares on a one-for-two and one-half basis. ZoMedica Pharmaceuticals
Inc. (“ZoMedica Inc.”) was incorporated on May 14, 2015 under the Canada Business Corporations Act. On April 21, 2016,
we completed a qualifying transaction (the “Qualifying Transaction”) under TSX-V Policy 2.4 – Capital Pool Companies,
consisting of a three- cornered amalgamation among our Company, ZoMedica Inc. and our wholly-owned subsidiary. Under the Qualifying
Transaction, ZoMedica Inc. and our subsidiary were amalgamated to form Zomedica Pharmaceuticals Ltd. (“Zomedica Ltd.”).
As consideration for the amalgamation, shareholders of ZoMedica Inc. became the owners of 97.6% (non-diluted) of our common shares,
and ZoMedica Ltd. became our wholly-owned subsidiary. Subsequent to the Qualifying Transaction, Zomedica Ltd. was vertically amalgamated
into our Company. On October 2, 2020, we changed our name to Zomedica Corp. Our principal executive offices are located at 100
Phoenix Drive, Suite 180, Ann Arbor, MI 48108, and our telephone number is (734) 369-2555. Our website address is www.zomedica.com.
We have not incorporated by reference into this proxy circular/prospectus the information on our website, and you should not consider
it to be a part of this document.
Implications
of Being an Emerging Growth Company
As a company with less
than $1.07 billion in revenue during our last fiscal year, we qualify as an “emerging growth company” as defined in
the JOBS Act. An “emerging growth company” may take advantage of reduced reporting requirements that are otherwise
applicable to public companies. These provisions include, but are not limited to:
|
|
·
|
not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley
Act;
|
|
|
·
|
reduced disclosure obligations regarding executive compensation in our periodic reports, proxy
statements and registration statements; and
|
|
|
·
|
exemptions from the requirements of holding a non-binding advisory vote on executive compensation
and shareholder approval of any golden parachute payments not previously approved.
|
We may take advantage
of these provisions until December 31, 2022. However, if certain events occur prior to December 31, 2022, including if we become
a “large accelerated filer,” our annual gross revenues exceed $1.07 billion or we issue more than $1.0 billion of non-convertible
debt in any three-year period, we will cease to be an emerging growth company before such date.
In addition, the JOBS
Act provides that an emerging growth company may delay adopting new or revised accounting standards until such time as those standards
apply to private companies. We have irrevocably elected not to avail ourselves of this exemption and, therefore, we will be subject
to the same new or revised accounting standards as other public companies that are not emerging growth companies.
Significant additional information about Zomedica
is set forth in the section of this proxy circular/prospectus entitled “Business,” beginning at page 41.
Set forth below in
a question and answer format is general information regarding the Special Meeting to which this proxy circular/prospectus relates.
This general information regarding the Special Meeting is followed by a more detailed summary of the process relating to, reasons
for and effects of our proposed change in jurisdiction of incorporation from the Province of Alberta in Canada to the State of
Delaware in the United States (Proposal 1 in the Notice of Special Meetings), which we refer to in this proxy circular/prospectus
as the ‘‘domestication’’ or the ‘‘continuance’’ and
the adoption of the 2020 Plan (Proposal 2 in the Notice of Special Meeting).
QUESTIONS AND ANSWERS ABOUT THE PROPOSALS
Q. What
is the purpose of the Special Meeting?
A. The primary purpose of the Special Meeting is to vote on the proposal to approve a special
resolution authorizing us to make an application to change our jurisdiction of incorporation to Delaware and adopt a certificate
of incorporation of Zomedica Corp. to be effective as of the date of our domestication. Assuming the approval of the domestication,
our shareholders also are being asked to consider and vote upon a proposal to adopt the 2020 Zomedica Corp. Stock Incentive Plan,
or the 2020 Plan, and to transact such other business as is proper at the Special Meeting.
Q. Where and when will
the Special Meeting be held?
A. The
Special Meeting will be held via live audio online webcast at https://web.lumiagm.com/157200202, password “zomedica2020”,
on December 15, 2020, at 8:30 a.m. (Toronto time).
Q. Who is soliciting
my vote?
A. Our
management is soliciting your proxy to vote at the Special Meeting.
This proxy circular/prospectus and form of proxy were first mailed to our shareholders on or about October , 2020.
Only the form of proxy accompanying this proxy circular/prospectus will be counted. Your vote is important. We encourage you to
vote as soon as possible after carefully reviewing this proxy circular/prospectus.
Q. Who is entitled to
vote?
A. The
record date for the determination of shareholders entitled to receive notice of the Special Meeting is October 22, 2020, or the
Record Date. In accordance with the provisions of the ABCA, we will prepare a list of our registered shareholders as of the Record
Date. If you were a shareholder as of the Record Date, you will be entitled to vote at the Special Meeting. The holders of at least
two thirds of our common shares and Series 1 preferred shares present at the Special Meeting virtually or by proxy (and assuming
a quorum of our outstanding shares are represented at the Special Meeting virtually or by proxy) must vote together as a single
class to approve the domestication proposal. The holders of a majority of our common shares present at the Special Meeting virtually
or by proxy (and assuming a quorum of our outstanding common shares are represented at the Special Meeting virtually or by proxy)
must vote to approve the incentive plan proposal. No cumulative voting rights are authorized.
Q. What am I voting
on?
A. The shareholders
are entitled to vote on a special resolution authorizing us to make an application under Section 189 of the ABCA to change our
jurisdiction of incorporation from the Province of Alberta, Canada to the State of Delaware, United States of America, by way of
a continuance under Section 189 of the ABCA and a domestication under Section 388 of the Delaware General Corporation Law, or the
DGCL, and to approve the certificate of incorporation of Zomedica Corp. authorized in the special resolution to be effective as
of the date of our domestication, and our common shareholders are entitled to vote on the approval of the 2020 Plan.
Q. What is the voting
recommendation of the Board of Directors?
A. The board of
directors of the Corporation, or the Board, recommends a vote FOR the special resolution authorizing us to make an application
under Section 189 of the ABCA to change our jurisdiction of incorporation from the Province of Alberta, Canada to the State of
Delaware, United States of America, by way of a continuance, and to approve the certificate of incorporation of Zomedica Corp.
authorized in the special resolution to be effective as of the date of our domestication. The Board also recommends a vote FOR
the approval of the 2020 Plan.
Q. Will any other matters
be voted on?
A. The Board does
not intend to present any other matters at the Special Meeting. The Board does not know of any other matters that will be brought
before our shareholders for a vote at the Special Meeting. If any other matter is properly brought before the Special Meeting,
your signed proxy card gives authority to Robert Cohen and, failing him, Ann Marie Cotter, as proxies, with full power of substitution,
to vote on such matters at their discretion.
Q. How
many votes do I have?
A. Shareholders
are entitled to one vote for each common share or Series 1 preferred share held as of the close of business on the Record Date.
Voting Securities
We are authorized to
issue an unlimited number of common shares and an unlimited number of preferred shares. As of the Record Date, 564,051,438 common
shares were issued and outstanding as fully paid and non-assessable and 12 Series 1 preferred shares were issued and outstanding
as fully paid and non-assessable. No other shares of any other class are issued or outstanding. In accordance with Section 189(3)
of the ABCA, each share of the Corporation carries the right to vote in respect the domestication whether or not it otherwise carries
the right to vote. As such, the holders of at least two thirds of our common shares and Series 1 preferred shares present at the
Special Meeting virtually or by proxy (and assuming a quorum of our outstanding shares are represented at the Special Meeting virtually
or by proxy) must vote together as a single class to approve the domestication proposal. The holders of a majority of our common
shares present at the Special Meeting virtually or by proxy (and assuming a quorum of our outstanding common shares are represented
at the Special Meeting virtually or by proxy) must vote to approve the incentive plan proposal. No cumulative voting rights are
authorized.
Shareholders of record
at the close of business on the Record Date are entitled to vote their shares at the Meeting on the basis of one vote for each
common share or Series 1 preferred share held except to the extent that, (a) the holder has transferred the ownership of any of
his shares after the Record Date, and (b) the transferee of those shares produces properly endorsed share certificates, or otherwise
establishes that he owns the shares, and demands not later than ten (10) days before the day of the Meeting that his name be included
in the list of persons entitled to vote at the Special Meeting, in which case the transferee will be entitled to vote his shares
at the Meeting as described above.
To our knowledge, no person owned more than 10% of our outstanding
common shares as of the Record Date. Wickfield Capital LLC and its affiliate, Wickfield Bridge Fund LLC, were the record owners
of all of our outstanding Series 1 preferred shares as of the Record Date. For additional beneficial ownership information, see
“Securities Ownership of Certain Beneficial Owners and Management.”
PROPOSAL NO. 1
THE DOMESTICATION PROPOSAL
Our Board is proposing
to change our jurisdiction of incorporation from the Province of Alberta in Canada to the State of Delaware in the United States
through a transaction called a ‘‘continuance’’ under Section 189 of the ABCA and a ‘‘domestication’’
under Section 388 of the DGCL. Under the DGCL, a corporation becomes domesticated in the State of Delaware by filing a certificate
of corporate domestication and a certificate of incorporation with the Secretary of State of the State of Delaware. The domesticated
corporation, which will be called Zomedica Corp., will become subject to the DGCL on the date of its domestication, but will be
deemed for the purposes of the DGCL to have commenced its existence in Delaware on the date it originally commenced existence in
Canada.
The Board has unanimously
approved the domestication, believes it to be in our best interests, and unanimously recommends its approval.
Our Board has determined to pursue the domestication for a number
of reasons. Our domestication is intended to reduce the regulatory burden and cost of being subject to the laws and regulations
of both the United States and Canada and to enhance shareholder value over the long term by, among other things, reducing our operating
costs and enhancing the marketability of our capital stock by becoming a Delaware corporation. In addition, our corporate offices
and operations are located in the United States and a large percentage of our shareholders are located there. We believe that the
domestication will save approximately $500,000 per year in duplicative, unnecessary compliance costs, which will directly benefit
shareholders.
We chose the State
of Delaware to be our domicile principally because the DGCL expressly accommodates continuances under the ABCA, and also because
of the comprehensive body of case law interpreting the DGCL that has evolved over the years, including case law interpreting the
duties and obligations of directors and officers.
The domestication will change the corporate
laws that apply to our shareholders from the laws of the Province of Alberta, and the laws of Canada applicable therein, to the
laws of the State of Delaware. There are material differences between the ABCA and the DGCL. Our shareholders may have more or
fewer rights under Delaware law depending on the specific set of circumstances.
We plan to complete the proposed domestication
as soon as possible following approval by our shareholders. The domestication will be effective on the date set forth in the certificate
of corporate domestication and certificate of incorporation, as filed with the Secretary of State of the State of Delaware. Thereafter,
Zomedica Corp. will be subject to the certificate of incorporation filed in Delaware. Simultaneously, we will continue out of Alberta
upon the issuance from the Alberta Registrar of Corporations of confirmation of the continuance, which is expected to be the same
date as the date of the filing of the certificate of corporate domestication and certificate of incorporation in Delaware. However,
the Board may decide to delay the domestication or not to proceed with the domestication after receiving approval from our shareholders
if it determines that the domestication is no longer advisable. The Board has not considered any alternative action if the domestication
is not approved or if it decides to abandon the domestication.
The domestication will not interrupt our corporate
existence, our operations, our outstanding agreements and obligations, or the trading market of our common shares. Upon consummation
of the domestication, (i) the property of Zomedica Corp. (the Alberta corporation) will continue to be the property of Zomedica
Corp. (the Delaware corporation); (ii) Zomedica Corp. (the Delaware corporation) will continue to be liable for the obligations
of Zomedica Corp. (the Alberta corporation); (iii) any existing cause of action, claim or liability to prosecution in respect of
Zomedica Corp. (the Alberta corporation) shall be unaffected; (iv) any civil, criminal or administrative action or proceeding pending
by or against Zomedica Corp. (the Alberta corporation) may be continued to be prosecuted by or against Zomedica Corp. (the Delaware
corporation); and (v) any conviction against, or ruling, order or judgment in favor of or against Zomedica Corp. (the Alberta corporation)
may be enforced by or against Zomedica Corp (the Delaware corporation). Each outstanding common share and Series 1 preferred share
at the time of the domestication will remain issued and outstanding as a share of common stock or Series 1 preferred stock, as
applicable, of Zomedica Corp. (the Delaware corporation) after our corporate existence is continued from the Province of Alberta
in Canada under the ABCA and domesticated in the State of Delaware in the United States under the DGCL. Following the completion
of the domestication, Zomedica Corp.’s common stock will continue to be listed on the NYSE American exchange under the symbol
“ZOM.”
Holders of our outstanding
warrants and options will continue to hold the same securities, which will remain exercisable for an equivalent number of shares
of the same class of common stock of Zomedica Corp. (the Delaware corporation), for the equivalent exercise price per share, without
any action by the holder.
Regulatory and Other Approvals
The continuance is subject to the authorization
of the Registrar of Corporations appointed under the ABCA. The Registrar is empowered to authorize the continuance if, among other
things, Registrar is satisfied that the continuance will not adversely affect our creditors or shareholders.
Tax Consequences of the Domestication
U.S. Federal Income
Tax Consequences. We believe that the change in our jurisdiction of incorporation should constitute a tax-free reorganization
within the meaning of Section 368(a) of the United States Internal Revenue Code of 1986, as amended (the “Code”) and,
generally, neither Zomedica Corp., the Alberta corporation (hereinafter referred to as “we” or the “Corporation”),
nor Zomedica Corp., the Delaware corporation, should recognize any gain or loss for U.S. federal income tax purposes as a result
of the domestication, other than as described later herein in “United States Federal Income Tax Consequences.”
For
U.S. Holders (as defined below in “United States Federal Income Tax Consequences”), the domestication also would generally
be tax free. However, Code Section 367(b) has the effect of potentially imposing income tax on such U.S. Holders in connection
with the domestication. Pursuant to the Treasury Regulations under Code Section 367(b), any 10% Shareholder (as defined below
in “United States Federal Income Tax Consequences”) will have to recognize a deemed dividend on the domestication
equal to the “all earnings and profits amount,” within the meaning of Treasury Regulations Section 1.367(b)-2, attributable
to such U.S. Holder’s shares in the Corporation. Any U.S. Holder that is not a 10% Shareholder and whose shares have a fair
market value of less than $50,000 on the date of the domestication will recognize no gain or loss pursuant to Code Section 367(b)
as a result of the domestication. A U.S. Holder that is not a 10% Shareholder but whose shares have a fair market value of at
least $50,000 on the date of the domestication must generally recognize gain (but not loss) on the domestication equal to the
difference between the fair market value of the Zomedica Corp. stock received at the time of the domestication over the U.S. Holder’s
tax basis in the Corporation’s shares. Such a holder, however, instead of recognizing gain, may elect to include in income
as a deemed U.S. dividend the “all earnings and profits amount” attributable to such holder’s shares in the
Corporation, which we refer to as a “Deemed Dividend Election.”
Based on the Corporation’s
limited activity at the holding company level and the size of the Corporation’s existing earnings and profits deficit, we
believe that no U.S. Holder should have a positive ‘‘all earnings and profits amount’’ attributable to
such holder’s shares in the Corporation, and accordingly no 10% Shareholder or U.S. Holder who makes a Deemed Dividend Election
should be required to include any such amount in income on the domestication. Our belief with respect to the “all earnings
and profits amount” results from calculations performed by our accounting firm based on information provided to them by
us. However, no assurance can be given that the IRS will agree with us. If it does not agree, then a U.S. Holder may be subject
to adverse U.S. federal income tax consequences.
In
addition, the domestication might be a taxable event to U.S. Holders under the passive foreign investment company (“PFIC”)
rules of the Code. The Corporation believes that it is likely a PFIC for U.S. federal income tax purposes. In the event that the
Corporation is considered a PFIC, then proposed Treasury Regulations under Code Section 1291(f) (which were promulgated in 1992
with a retroactive effective date), if finalized in their current form, generally would require a U.S. Holder to recognize gain
on the exchange of equity securities of the Corporation for equity securities of Zomedica Corp. pursuant to the domestication.
The tax on any such gain so recognized would be imposed at the rate applicable to ordinary income and an interest charge would
apply based on a complex set of computational rules designed to offset the tax deferral to such U.S. Holders on the Corporation’s
undistributed earnings. Any “all earnings and profits amount” included in income by a U.S. Holder as a result of the
domestication generally would be treated as gain subject to these rules. However, it is difficult to predict whether, in what
form and with what effective date final Treasury Regulations under Code Section 1291(f) will be adopted. U.S. Holders that make
or have made certain elections discussed further under ‘‘United States Federal Income Tax Consequences’’
with respect to their common shares of the Corporation are generally not subject to the same gain recognition rules under the
current proposed Treasury Regulations.
Following the domestication, a U.S. Holder’s tax
basis in the equity securities of Zomedica Corp. received in the exchange will be equal to such holder’s tax basis in the
equity securities of the Corporation surrendered in the exchange, increased by the amount of gain (if any) recognized in connection
with the domestication or the amount of the “all earnings and profits amount” included in such holder’s income.
A U.S. Holder’s holding period in the equity securities of Zomedica Corp. received in the exchange should include the period
of time during which such holder held its equity securities in the Corporation.
The tax consequences
of the domestication are complex and will depend on a holder’s particular circumstances. All holders are strongly urged to
consult their tax advisors for a full description and understanding of the tax consequences of the domestication, including the
applicability and effect of U.S. federal, state, local and foreign income and other tax laws. For a more complete discussion of
the U.S. federal income tax considerations of the domestication, see ‘‘United States Federal Income Tax Consequences.’’
Canadian Federal
Income Tax Consequences. Under the Income Tax Act (Canada), or the Tax Act, the change in our jurisdiction from Canada to the
United States will cause our tax year to end immediately before the domestication. Furthermore, we will be deemed to have disposed
of all of our property immediately before the continuance for proceeds of disposition equal to the fair market value of the property
at that time. We will be subject to a separate corporate emigration tax imposed on the amount, if any, by which the fair market
value of all of our property (principally consisting of all of the outstanding shares of capital stock of our wholly owned U.S.
subsidiary, Zomedica Pharmaceuticals, Inc.) immediately before the continuance exceeds the aggregate of our liabilities at that
time (other than dividends payable and taxes payable in connection with the emigration tax) and the amount of paid-up capital on
all of our outstanding common shares.
We have reviewed our
assets, liabilities, paid-up capital and other tax balances with the assistance of our professional advisors. Based on our calculations,
under current law and based on current applicable tax rates, we estimate that we will incur no Canadian income tax liability as
a result of the domestication. This conclusion is based in part on determinations of factual matters, including determinations
regarding the fair market value of our assets and tax attributes, any or all of which could change prior to the effective time
of the domestication.
Our shareholders
who hold our common shares or Series 1 preferred shares at the time of the domestication will not be considered to have disposed
of their shares for Canadian income tax purposes by reason only of the domestication. Accordingly, the domestication will not
cause Canadian resident shareholders to realize a capital gain or loss on their shares and there will be no effect on the adjusted
cost base of their shares.
The foregoing is a
brief summary of the principal income tax considerations only and is qualified in its entirety by the more detailed description
of income tax considerations in ‘‘United States and Canadian Tax Considerations’’ in this proxy circular/prospectus,
which shareholders are urged to read. This summary does not discuss all aspects of United States and Canadian tax consequences
that may apply in connection with the domestication. Shareholders should consult their own tax advisors as to the tax consequences
of the domestication applicable to them. In addition, please note that other tax consequences may arise under applicable law in
other countries.
Accounting
Treatment of the Domestication
Our domestication as
a Delaware corporation represents a transaction between entities under common control. Assets and liabilities transferred between
entities under common control are accounted for at carrying value. Accordingly, the assets and liabilities of Zomedica Corp. will
be reflected at their carrying value to us. Any of our shares that we acquire from dissenting shareholders will be treated as an
acquisition of treasury stock at the amount paid for the shares.
DISSENTING
RIGHTS OF SHAREHOLDERS
If you wish to dissent and do so in compliance
with Section 191 of the ABCA, and we proceed with the domestication, you will be entitled to be paid the fair value of the common
shares you hold. Fair value is determined as of the close of business on the day before the domestication is approved by our shareholders.
If you wish to dissent, you must send written objection to the domestication to us at or before the Special Meeting. If you vote
in favor of the domestication, you in effect lose your rights to dissent. If you do not vote or vote against the domestication,
you preserve your dissent rights to the extent you comply with Section 191 of the ABCA.
However, it is not
sufficient to vote against the domestication or to refrain from voting. You must also provide a separate dissent notice at or before
the Special Meeting. If you grant a proxy and intend to dissent, the proxy must instruct the proxy holder to vote against the domestication
in order to prevent the proxy holder from voting such shares in favor of the domestication and thereby voiding your right to dissent.
Under the ABCA, you have no right of partial dissent. Accordingly, you may dissent only as to all your common shares. Section 191
of the ABCA is reprinted in its entirety as Exhibit E to this proxy circular/prospectus.
COMPARISON
OF SHAREHOLDER RIGHTS
Upon completion
of the domestication, shareholders will be holders of capital stock of Zomedica Corp., a Delaware corporation, and their rights
will be governed by the DGCL as well as Zomedica Corp.’s certificate of incorporation and bylaws. Shareholders should be
aware that the rights they currently have under the ABCA may, with respect to certain matters, be different than the rights they
will have as stockholders under the DGCL. For example, under the ABCA, a company has the authority to issue an unlimited number
of shares whereas, under the DGCL, a Delaware corporation may only issue the number of shares that is authorized by its certificate
of incorporation and board and stockholder approval must be obtained to amend the certificate of incorporation to increase the
number of authorized shares before the issuance of additional shares. We refer you to the section titled ‘‘The Domestication—Comparison
of Shareholder Rights’’ for a more detailed description of the material differences between the rights of Canadian
shareholders and Delaware stockholders.
PROPOSAL NO. 2
THE STOCK INCENTIVE PLAN PROPOSAL
At
the Special Meeting, holders of our common shares will be asked to approve the Zomedica Corp. 2020 Equity Incentive Plan, or the
2020 Plan. The Board approved the 2020 Plan on October 7, 2020, subject to its approval by the common shareholders. If the common
shareholders approve the 2020 Plan, it will become effective upon completion of the domestication.
General
Our Board believes
that it is in the best interests of our company and its shareholders to have a new equity compensation plan adopted by the Board
and approved by the shareholders so that our company can continue to provide a means whereby eligible employees, officers, non-employee
directors and other individual service providers develop a sense of proprietorship and personal involvement in the development
and financial success of our company and to encourage them to devote their best efforts to the business of our company, thereby
advancing the interests of our company and its stockholders.
Approval of the 2020
Plan by our company’s shareholders is required, among other things, in order to: (i) comply with NYSE American rules
requiring stockholder approval of equity compensation plans and (ii) allow the grant to eligible employees of options that
qualify as “incentive stock options” (or ISOs) under Section 422 of the Code.
In the event that the
common shareholders do not approve this proposal, the 2020 Plan will not become effective. Approval of the 2020 Plan by the common
shareholders will allow us to grant stock options, restricted stock unit awards and other awards at levels determined appropriate
by our Board and/or compensation committee. The 2020 Plan will also allow us to utilize a broad array of equity incentives and
performance cash incentives in order to secure and retain the services of our employees, directors and consultants, and to provide
long-term incentives that align the interests of our employees, directors and consultants with the interests of our stockholders.
The following is a
summary of the principal features of the 2020 Plan. This summary does not purport to be a complete description of all of the provisions
of the 2020 Plan and it is qualified in its entirety by reference to the full text of the 2020 Plan, a copy of which is attached
to this proxy statement/prospectus as Exhibit G hereto. As used below, “Zomedica Corp.” refers to Zomedica Corp.,
a Delaware corporation.
Summary of the 2020 Plan
Available
Shares. An aggregate of 200,000,000 shares of Zomedica Corp.’s common stock (representing approximately 10% of the
outstanding common stock determined on a fully-diluted basis) may be issued under the 2020 Plan, subject to equitable adjustment
in the event of stock splits and other capital changes, all which may be issued in respect of Incentive Stock Options (or ISOs)
that meet the requirements of Section 422 of the Code.
In applying the aggregate
share limitation under the 2020 Plan, shares of common stock (i) subject to awards that are forfeited, cancelled, returned to
our company for failure to satisfy vesting requirements or otherwise forfeited, or terminated without payment being made thereunder
and (ii) that are surrendered in payment or partial payment of the exercise price of an option or taxes required to be withheld
with respect to the exercise of stock options or in payment with respect to any other form of award are not counted and, therefore,
may be made subject to new awards under the 2020 Plan.
Non-Employee
Director Compensation Limit. Under the 2020 Plan, no more than 10,000,000 shares of Zomedica Corp.’s common stock
may be subject to stock awards granted under the 2020 Plan during any one calendar year to any non-employee director.
Administration.
The 2020 Plan will be administered by the Compensation Committee of Zomedica Corp.’s Board, or the Compensation Committee.
The Compensation Committee has discretion to determine the individuals to whom awards may be granted under the 2020 Plan, the number
of shares of common stock, units or other rights subject to each award, the type of award, the manner in which such awards will
vest, and the other conditions applicable to awards. The Compensation Committee is authorized to interpret the 2020 Plan, to prescribe,
amend and rescind any rules and regulations relating to the 2020 Plan and to make any other determinations necessary or desirable
for the administration of the Plan. All interpretations, determinations and actions by the Compensation Committee are final, conclusive
and binding on all parties.
Eligibility.
Any employee, officer, director, consultant, advisor or other individual service provider of Zomedica Corp. or any of its
subsidiaries, or any person who is determined by our Compensation Committee to be a prospective employee, officer, director, consultant,
advisor or other individual service provider of Zomedica Corp. or any of its subsidiaries is eligible to participate in the 2020
Plan. As of the Record Date, we had approximately 17 full-time employees, including 4 executive officers, 5 non- employee directors,
and no consultants, advisors and/or other individual service providers. As of the Record Date, no person is eligible to participate
as a result of a determination by the Compensation Committee that that person is a prospective employee, director or consultant
of our Company or any of our subsidiaries. As awards under the 2020 Plan are within the discretion of the Compensation Committee,
we cannot determine how many individuals in each of the categories described above will receive awards.
Types of Awards.
Under the 2020 Plan, the Compensation Committee may grant nonqualified stock options (or NSOs), ISOs, stock appreciation
rights (or SARs), restricted stock, stock units, performance shares, performance units, other cash-based awards and other stock-based
awards. The terms of each award will be set forth in a written agreement with the recipient.
Stock Options.
The Compensation Committee will determine the exercise price and other terms for each option and whether the options will be NSOs
or ISOs. The exercise price per share of each option will not be less than 100% of the fair market value of Zomedica Corp.’s
common stock on the date of grant or, if there are no trades on such date, then the closing price of a share of Zomedica Corp.’s
common stock on the most recent date preceding the date of grant on which shares of common stock were publicly traded (or 110%
of the fair market value per share in the case of ISOs granted to a ten-percent or more shareholder). However, if permissible
under law and the rules of the exchange on which our company is listed, options to participants who are not residents of the U.S.
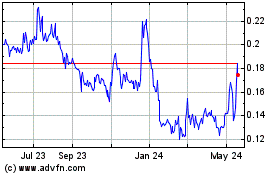
may be granted at a price below fair market value on the date of grant. On October 7, 2020, the closing sale price of our common
shares on the NYSE American exchange was $0.0927 per share.
ISOs may be granted
only to employees and are subject to certain other restrictions. To the extent an option intended to be an ISO does not qualify
as an ISO, it will be treated as a nonqualified option.
A participant may exercise
an option by written notice and payment of the exercise price in cash, or, as determined by the Compensation Committee, through
delivery of previously owned shares, the withholding of shares deliverable upon exercise, a cashless exercise program implemented
by the Compensation Committee in connection with the 2020 Plan, and/or such other method as approved by the Compensation Committee
and set forth in an award agreement. The maximum term of any option granted under the 2020 Plan is ten years from the date of grant
(five years in the case of an ISO granted to a ten-percent or more shareholder). The Compensation Committee may, in its discretion,
permit a holder of an NSO to exercise the option before it has otherwise become exercisable, in which case the shares of our company’s
common stock issued to the recipient will be restricted stock having analogous vesting restrictions to the unvested NSO before
exercise.
No option may be exercisable
for more than ten years (five years in the case of an ISO granted to a ten-percent or more shareholder) from the date of grant.
Options granted under the 2020 Plan will be exercisable at such time or times as the Compensation Committee prescribes at the time
of grant. No employee may receive ISOs that first become exercisable in any calendar year in an amount exceeding $100,000.
Unless an award agreement
provides otherwise, if a participant’s Service (as defined in the 2020 Plan) terminates (i) by reason of his or her death
or Disability (as defined in the 2020 Plan), any option held by such participant may be exercised, to the extent otherwise exercisable,
by the participant or his or her estate or personal representative, as applicable, at any time in accordance with its terms for
up to one year after the date of such participant’s death or termination of Service, as applicable, (ii) for Cause (as defined
in the 2020 Plan), any option held by such participant will be forfeited and cancelled as of the date of termination of Service
and (iii) for any reason other than death, Disability or Cause, any option held by such participant may be exercised, to the extent
otherwise exercisable, up until ninety (90) days following termination of Service.
Stock Appreciation
Rights. The Compensation Committee may grant SARs independent of or in connection with an option. The Compensation Committee
will determine the other terms applicable to SARs. The base price per share of each SAR will not be less than 100% of the closing
price of a share of Zomedica Corp.’s common stock on the date of grant or, if there are no trades on such date, then the
closing price of a share of Zomedica Corp.’s common stock on the most recent date preceding the date of grant on which shares
of common stock were publicly traded. The maximum term of any SAR granted under the 2020 Plan will be ten years from the date of
grant. Generally, each SAR will entitle a participant upon exercise to an amount equal to the excess of the fair market value on
the exercise date of one share of our common stock over the base price, multiplied by the number of shares of common stock as to
which the SAR is exercise. Payment may be made in shares of Zomedica Corp.’s common stock, in cash, or partly in shares of
Company common stock and partly in cash, all as determined by the Compensation Committee.
Restricted Stock
and Stock Units. The Compensation Committee may award restricted common stock and/or stock units under the 2020 Plan. Restricted
stock awards consist of shares of stock that are transferred to a participant subject to restrictions that may result in forfeiture
if specified conditions are not satisfied. Stock units confer the right to receive shares of Zomedica Corp.’s common stock,
cash, or a combination of shares and cash, at a future date upon or following the attainment of certain conditions specified by
the Compensation Committee, subject to applicable tax withholding requirements. The Compensation Committee will determine the restrictions
and conditions applicable to each award of restricted stock or stock units, which may include performance-based conditions. Unless
the Compensation Committee determines otherwise at the time of grant, holders of restricted stock will have the right to vote the
shares and receive all dividends and other distributions.
Performance Shares
and Performance Units. The Compensation Committee may award performance shares and/or performance units under the 2020 Plan.
Performance shares and performance units are awards, payable in shares of Zomedica Corp.’s common stock, cash or a combination
thereof, which are earned during a specified time period subject to the attainment of performance goals, as established by the
Compensation Committee. The Compensation Committee will determine the restrictions and conditions applicable to each award of performance
shares and performance units.
Incentive Bonus
Awards. The Compensation Committee may award incentive bonus awards payable in cash or shares of common stock, as set
forth in an award agreement. Incentive bonus awards may be based upon the attainment of specified levels of Zomedica Corp. or subsidiary
performance. The amount of an incentive bonus award to be paid upon the attainment of each targeted level of performance will equal
a percentage of a participant’s base salary for the fiscal year, a fixed dollar amount or such other formula, as determined
by the Compensation Committee. The Compensation Committee will determine the terms and conditions applicable to each incentive
bonus award.
Other Stock-Based
and Cash-Based Awards. The Compensation Committee may award other types of stock-based or cash-based awards under the 2020
Plan, including the grant or offer for sale of unrestricted shares of Zomedica Corp.’s common stock, in such amounts and
subject to such terms and conditions as the Compensation Committee determines.
Transferability.
Awards granted under the 2020 Plan will not be transferable other than by will or by the laws of descent and distribution,
except that the Compensation Committee may permit NSOs, share-settled SARs, restricted stock, performance share or share-settled
other stock-based awards to be transferred to family members and/or for estate planning or charitable purposes.
Change in Control.
The Compensation Committee may, at the time of the grant of an award, provide for the effect of a change in control (as
defined in the 2020 Plan) on any award, including (i) accelerating or extending the time periods for exercising, vesting in,
or realizing gain from any award, (ii) eliminating or modifying the performance or other conditions of an award, (iii) providing
for the cash settlement of an award for an equivalent cash value, as determined by the Compensation Committee, or (iv) such
other modification or adjustment to an award as the Compensation Committee deems appropriate to maintain and protect the rights
and interests of participants upon or following a change in control. Unless otherwise provided by an award agreement, the Compensation
Committee may, in its discretion and without the need for the consent of any recipient of an award, also take one or more of the
following actions contingent upon the occurrence of a change in control: (a) cause any or all outstanding options and SARs
to become immediately exercisable, in whole or in part; (b) cause any other awards to become non-forfeitable, in whole or
in part; (c) cancel any option or SAR in exchange for a substitute option and/or SAR; (d) cancel any award of restricted
stock, stock units, performance shares or performance units in exchange for a similar award of the capital stock of any successor
corporation; (e) redeem any restricted stock for cash and/or other substitute consideration with a value equal to the fair
market value of an unrestricted share of Zomedica Corp.’s common stock on the date of the change in control; or (f) terminate
any award in exchange for an amount of cash and/or property equal to the amount, if any, that would have been attained upon the
exercise of such award or realization of the participant’s rights as of the date of the occurrence of the Change in Control
(the “Change in Control Consideration”); provided, however that if the Change in Control Consideration with respect
to any option or SAR does not exceed the exercise price of such option or SAR, the Compensation Committee may cancel the option
or SAR without payment of any consideration therefor. Any such Change in Control Consideration may be subject to any escrow, indemnification
and similar obligations, contingencies and encumbrances applicable in connection with the change in control to holders of Zomedica
Corp.’s common stock. Without limitation of the foregoing, if as of the date of the occurrence of the change in control the
Compensation Committee determines that no amount would have been attained upon the realization of the participant’s rights,
then such award may be terminated by Zomedica Corp. without payment. The Compensation Committee may cause the Change in Control
Consideration to be subject to vesting conditions (whether or not the same as the vesting conditions applicable to the award prior
to the change in control) and/or make such other modifications, adjustments or amendments to outstanding Awards or the 2020 Plan
as the Compensation Committee deems necessary or appropriate.
Term;
Amendment and Termination. No award may be granted under the 2020 Plan on or after October 7, 2030. The board of directors
may suspend, terminate, or amend the 2020 Plan in any respect at any time, provided, however, that (i) no amendment, suspension
or termination may materially impair the rights of a participant under any awards previously granted, without his or her consent,
(ii) Zomedica Corp. shall obtain stockholder approval of any 2020 Plan amendment as required to comply with any applicable law,
regulation or stock exchange rule and (iii) stockholder approval is required for any amendment to the 2020 Plan that (x) increases
the number of shares of common stock available for issuance thereunder or (y) changes the persons or class of persons eligible
to receive awards.
New Plan Benefits
No awards will be granted
under the 2020 Plan prior to its approval by our shareholders. The grant of options and other awards under the 2020 Plan is discretionary,
and we cannot determine now the number or type of options or other awards to be granted in the future to any particular person
or group.
Existing Options under current Equity Incentive Plan
Upon completion of the domestication, holders
of our outstanding options will continue to hold the same options, which will remain exercisable for an equivalent number of shares
of the same class of common stock of Zomedica Corp. (the Delaware corporation), for the equivalent exercise price per share, without
any action by the holder, and in accordance with their terms.
Material United States Federal Income
Tax Consequences of the 2020 Plan
Following is a summary
of the principal U.S. federal income tax consequences of option grants and other awards under the 2020 Plan. Optionees and recipients
of other rights and awards granted under the 2020 Plan are advised to consult their personal tax advisors before exercising an
option or stock appreciation right or disposing of any stock received pursuant to the exercise of an option or stock appreciation
right or following vesting of a restricted stock award or restricted stock unit or upon grant of an unrestricted stock award. In
addition, the following summary is based upon an analysis of the Code as currently in effect, existing laws, judicial decisions,
administrative rulings, Treasury Regulations and proposed Treasury Regulations, all of which are subject to change, and does not
address state, local or other tax laws.
Nonqualified Stock
Options. There will be no U.S. federal income tax consequences to a participant or to Zomedica Corp. upon the grant of a nonqualified
stock option. When the participant exercises a nonqualified option, he or she will recognize ordinary income in an amount equal
to the excess of the fair market value of the option shares on the date of exercise over the exercise price, and Zomedica Corp.
will be allowed a corresponding tax deduction, subject to any applicable limitations under Code Section 162(m). Any gain that a
participant realizes when the participant later sells or disposes of the option shares will be short-term or long-term capital
gain, depending on how long the participant held the shares.
Incentive Stock
Options. There will be no U.S. federal income tax consequences to a participant or to Zomedica Corp. upon the grant of an ISO.
If the participant holds the option shares for the required holding period of at least two years after the date the option was
granted and one year after exercise of the option, the difference between the exercise price and the amount realized upon sale
or disposition of the option shares will be long-term capital gain or loss, and Zomedica Corp. will not be entitled to a U.S. federal
income tax deduction. If the participant disposes of the option shares in a sale, exchange, or other disqualifying disposition
before the required holding period ends, the participant will recognize taxable ordinary income in an amount equal to the difference
between the exercise price and the lesser of the fair market value of the shares on the date of exercise or the disposition price,
and Zomedica Corp. will be allowed a U.S. federal income tax deduction equal to such amount, subject to any applicable limitations
under Code Section 162(m). Any amount received by the participant in excess of the fair market value on the exercise date
will be taxed to the participant as capital gain, and Zomedica Corp. will receive no corresponding deduction. While the exercise
of an ISO does not result in current taxable income, the excess of the fair market value of the option shares at the time of exercise
over the exercise price will be a tax preference item that could subject a participant to alternative minimum tax in the year of
exercise.
Stock Appreciation
Rights. A participant will not recognize income, and Zomedica Corp. will not be allowed a tax deduction, at the time a Stock
Appreciation Right (“SAR”) is granted. When a participant exercises a SAR, the cash or fair market value of any common
stock received will be taxable to the participant as ordinary income, and Zomedica Corp. will be allowed a U.S. federal income
tax deduction equal to such amount, subject to any applicable limitations under Code Section 162(m).
Restricted Stock.
Unless a participant makes an election to accelerate recognition of income to the grant date as described below, the participant
will not recognize income, and Zomedica Corp. will not be allowed a compensation tax deduction, at the time restricted stock is
granted. When the restrictions lapse, the participant will recognize ordinary income equal to the fair market value of the common
stock as of that date, less any amount paid for the stock, and Zomedica Corp. will be allowed a corresponding tax deduction, subject
to any applicable limitations under Code Section 162(m). If the participant files an election under Code Section 83(b)
within 30 days after the grant date, the participant will recognize ordinary income as of the grant date equal to the fair market
value of the stock as of that date, less any amount paid for the stock, and Zomedica Corp. will be allowed a corresponding compensation
tax deduction at that time, subject to any applicable limitations under Code Section 162(m). Any future appreciation in the
stock will be taxable to the participant at capital gains rates. However, if the stock is later forfeited, such participant will
not be able to recover the tax previously paid pursuant to the Code Section 83(b) election.
Stock Units, Performance
Awards, and Incentive Bonus Awards. A participant will not recognize income, and Zomedica Corp. will not be allowed a compensation
tax deduction, at the time a stock unit, performance award or incentive bonus award is granted. When a participant receives payment
under a stock unit, performance award or incentive bonus award, the amount of cash received and the fair market value of any shares
of stock received will be ordinary income to the participant, and Zomedica Corp. will be allowed a corresponding compensation tax
deduction at that time, subject to any applicable limitations under Code Section 162(m).
Section 409A. If
an award is subject to Section 409A of the Code, but does not comply with the requirements of Section 409A of the Code,
the taxable events as described above could apply earlier than described, and could result in the imposition of additional taxes
and penalties. Participants are urged to consult with their tax advisors regarding the applicability of Section 409A of the
Code to their awards.
Potential Limitation
on Zomedica Corp. Deductions. Section 162(m) of the Code generally disallows a tax deduction for compensation in excess of
$1 million paid in a taxable year by a publicly held corporation to its chief executive officer and certain other “covered
employees”. Zomedica Corp.’s board of directors and the Compensation Committee intend to consider the potential impact
of Section 162(m) on grants made under the 2020 Plan, but reserve the right to approve grants of options and other awards for an
executive officer that exceeds the deduction limit of Section 162(m).
Tax Withholding.
As and when appropriate, each optionee purchasing shares of Zomedica Corp.’s common stock and each grantee receiving
an award of shares of Zomedica Corp.’s common stock under the 2020 Plan will be required to pay any U.S. federal, state or
local taxes required by law to be withheld.
Vote Required
The holders of a majority of our common shares
present at the Special Meeting virtually or by proxy (and assuming a quorum of our outstanding common shares are represented at
the Special Meeting virtually or by proxy) must vote to approve the incentive plan proposal.
The Board
of Directors recommends a vote FOR Proposal 2.
Risk
Factors
You should carefully
consider the risks and uncertainties described below, together with all of the other information and risks included in this proxy
circular/prospectus, including our consolidated financial statements and the related notes thereto, before making a decision whether
to vote for the matters described in this proxy circular/prospectus. As used below, “Corporation” refers to Zomedica
Corp., an Alberta corporation.
Risks Relating to the Domestication
While we
believe the domestication will be tax-free to U.S. Holders for U.S. federal income tax purposes, if the IRS does not agree with
our calculation of the “all earnings and profits amount” attributable to a holder’s shares in the Corporation,
Zomedica Corp.’s U.S. Holders may owe U.S. federal income taxes as a result of the domestication under Code Section 367(b).
Code Section
367(b) has the effect of potentially imposing income tax on U.S. Holders (as defined below in “United States Federal Income
Tax Consequences”) in connection with the domestication. Pursuant to the Treasury Regulations under Code Section 367(b),
any 10% Shareholder (as defined below in “United States Federal Income Tax Consequences”) will have to recognize a
deemed dividend on the domestication equal to the “all earnings and profits amount,” within the meaning of Treasury
Regulations Section 1.367(b)-2, attributable to such U.S. Holder’s shares in the Corporation. Any U.S. Holder that is not
a 10% Shareholder and whose shares have a fair market value of less than $50,000 on the date of the domestication will recognize
no gain or loss pursuant to Code Section 367(b) as a result of the domestication. A U.S. Holder that is not a 10% Shareholder
but whose shares have a fair market value of at least $50,000 on the date of the domestication must generally recognize gain (but
not loss) on the domestication equal to the difference between the fair market value of the Zomedica Corp. stock received at the
time of the domestication over the U.S. Holder’s tax basis in the Corporation’s shares. Such a holder, however, instead
of recognizing gain, may elect to include in income as a deemed U.S. dividend the “all earnings and profits amount”
attributable to such holder’s shares in the Corporation, which we refer to as a “Deemed Dividend Election.”
Based on the Corporation’s
limited activity at the holding company level and the size of the Corporation’s existing earnings and profits deficit, we
believe that no U.S. Holder should have a positive ‘‘all earnings and profits amount’’ attributable to
such holder’s shares in the Corporation, and accordingly no 10% Shareholder or U.S. Holder who makes a Deemed Dividend Election
should be required to include any such amount in income on the domestication. Our belief with respect to the “all earnings
and profits amount” results from calculations performed by our accounting firm based on information provided to them by us.
However, no assurance can be given that the IRS will agree with us. If it does not agree, then a U.S. Holder may be subject to
adverse U.S. federal income tax consequences.
While we
believe the domestication will be tax-free to U.S. Holders for U.S. federal income tax purposes, if proposed Treasury Regulations
under Code Section 1291(f) are finalized in their current form, Zomedica Corp.’s U.S. Holders may owe U.S. federal income
taxes as a result of the domestication under the PFIC rules.
The Corporation believes
that it is likely a PFIC (as defined below under ‘‘United States Federal Income Tax Consequences”) for U.S.
federal income tax purposes. In the event that the Corporation is considered a PFIC, then proposed Treasury Regulations under
Code Section 1291(f) (which were promulgated in 1992 with a retroactive effective date), if finalized in their current form,
generally would require a U.S. Holder to recognize gain on the exchange of equity securities of the Corporation for equity
securities of Zomedica Corp. pursuant to the domestication. The tax on any such gain so recognized would be imposed at the
rate applicable to ordinary income and an interest charge would apply based on a complex set of computational rules designed
to offset the tax deferral to such U.S. Holders on the Corporation’s undistributed earnings. Any “all earnings
and profits amount” included in income by a U.S. Holder as a result of the domestication generally would be treated as
gain subject to these rules. However, it is difficult to predict whether, in what form and with what effective date final
Treasury Regulations under Code Section 1291(f) will be adopted. U.S. Holders that make or have made certain elections
discussed further under ‘‘United States Federal Income Tax Consequences’’ with respect to their
common shares of the Corporation are generally not subject to the same gain recognition rules under the current proposed
Treasury Regulations. For additional information on the U.S. federal income tax consequences of the domestication, see
‘‘United States Federal Income Tax Consequences.’’
The rights of our shareholders under Canadian law will differ
from their rights under Delaware law, which will, in some cases, provide less protection to shareholders following the domestication.
Upon consummation of the domestication, our
shareholders will become stockholders of a Delaware corporation. There are material differences between the ABCA and the DGCL and
our current and proposed charter and bylaws. For example, under Canadian law, many significant corporate actions such as amending
a corporation’s articles of incorporation, effecting a share consolidation or consummating a merger require the approval
of two-thirds of the votes cast by shareholders, whereas under Delaware law, a majority of the total voting power of all of those
entitled to vote may approve the matter. Furthermore, Shareholders under Canadian law are entitled to dissent and appraisal rights
under a number of extraordinary corporate actions, including an amalgamation with another unrelated corporation, certain amendments
to a corporation’s articles of incorporation or the sale of all or substantially all of a corporation’s assets; under
Delaware law, stockholders are entitled to dissent and appraisal rights for certain specified corporate transactions such as mergers
or consolidations. If the domestication is approved, shareholders may be afforded less protection under the DGCL than they had
under the ABCA in certain circumstances. See ‘‘The Domestication—Comparison of Shareholder Rights.’’
The proposed
domestication will result in additional direct and indirect costs whether or not it is completed.
The domestication will
result in additional direct costs. We will incur attorneys’ fees, accountants’ fees, filing fees, mailing expenses
and financial printing expenses in connection with the domestication. The domestication will also temporarily divert the attention
of our management and employees from the day-to-day management of the business to a limited extent.
The amount
of corporate tax payable by us will be affected by the value of our property on the date of the domestication.
For Canadian tax purposes,
on the date of the domestication we will be deemed to have a year end and to have disposed of all of our property for proceeds
equal to the fair market value of those properties. We will also be subject to an additional corporate emigration tax imposed on
the amount, if any, by which the fair market value of our property, net of certain liabilities, exceeds the paid-up capital of
our issued and outstanding shares. We have completed certain calculations of our tax accounts with the assistance of tax advisors,
and we have estimated that the domestication will not result in any Canadian tax liability.
Provisions
in Zomedica Corp.’s certificate of incorporation and bylaws and Delaware law might discourage, delay or prevent a change
of control of our company or changes in our management and, therefore, depress the trading price of our common stock.
Zomedica Corp.’s
certificate of incorporation and bylaws will contain provisions that could depress the trading price of our common stock by acting
to discourage, delay or prevent a change in control of our company or changes in our management that the stockholders of our company
may deem advantageous. These provisions:
|
|
·
|
establish a classified board of directors so that not all members of our board are elected at one
time;
|
|
|
·
|
provide that directors may only be removed ‘‘for cause;’’
|
|
|
·
|
authorize the issuance of ‘‘blank check’’ preferred stock that our board
of directors could issue from time to time to increase the number of outstanding shares and discourage a takeover attempt;
|
|
|
·
|
eliminate the ability of our stockholders to call special meetings of stockholders;
|
|
|
·
|
prohibit stockholder action by written consent, which has the effect of requiring all stockholder
actions to be taken at a meeting of stockholders;
|
|
|
·
|
provide that the board of directors is expressly authorized to make, alter or repeal our bylaws;
|
|
|
·
|
establish advance notice requirements for nominations for election to our board of directors or
for proposing matters that can be acted upon by stockholders at stockholder meetings; and
|
|
|
·
|
require supermajority approvals to remove the protective provisions in our certificate of incorporation
and bylaws listed above or to amend our bylaws.
|
Such provisions could
impede any merger, consolidation, takeover or other business combination involving the company or discourage a potential acquirer
from making a tender offer or otherwise attempting to obtain control of the company. See the section entitled ‘‘The
Domestication -- Comparison of Shareholder Rights.”
The certificate
of incorporation of Zomedica Corp. will designate the Court of Chancery of the State of Delaware as the sole and exclusive forum
for certain types of actions and proceedings that may be initiated by Zomedica Corp.’s stockholders, which could limit the
ability of Zomedica Corp.’s stockholders to obtain a favorable judicial forum for disputes with us or our directors, officers
or other employees.
The certificate of
incorporation of Zomedica Corp. will require that, unless we consent in writing to the selection of an alternative forum:
|
|
·
|
any derivative action or proceeding brought on our behalf;
|
|
|
·
|
any action asserting a claim of breach of any fiduciary duty owed by any current or former director,
officer, other employee or stockholder of ours to our company or our stockholders;
|
|
|
·
|
any action asserting a claim arising pursuant to any provision of the DGCL, our certificate of
incorporation or bylaws or as to which the DGCL confers jurisdiction on the Court of Chancery of the State of Delaware; or
|
|
|
·
|
any action asserting a claim governed by the internal affairs doctrine;
|
the Court of Chancery of the State of Delaware
will, to the fullest extent permitted by law, be the exclusive forum or if the Court of Chancery of the State of Delaware does
not have subject matter jurisdiction thereof, the federal district court of the State of Delaware.
Furthermore, Section
22 of the Securities Act creates concurrent jurisdiction for federal and state courts over all such Securities Act actions. Accordingly,
both state and federal courts have jurisdiction to entertain such claims. To prevent having to litigate claims in multiple jurisdictions
and the threat of inconsistent or contrary rulings by different courts, among other considerations, Zomedica Corp.’s certificate
of incorporation will provide that the federal district courts of the United States of America will be the exclusive forum for
resolving any complaint asserting a cause of action arising under the Securities Act.
The exclusive forum
provisions described above will not apply to claims arising under the Securities Exchange Act of 1934, as amended, or the Exchange
Act.
While the Delaware
courts have determined that such choice of forum provisions are facially valid, a stockholder may nevertheless seek to bring such
a claim arising under the Securities Act against Zomedica Corp., its directors, officers, or other employees in a venue other than
in the federal district courts of the United States of America. In such instance, we would expect to vigorously assert the validity
and enforceability of the exclusive forum provisions of Zomedica Corp.’s certificate of incorporation.
Although we believe
this provision benefits Zomedica Corp. by providing increased consistency in the application of Delaware law in the types of lawsuits
to which it applies, this provision may limit or discourage a stockholder’s ability to bring a claim in a judicial forum
that it finds favorable for disputes with Zomedica Corp. or its directors, officers or other employees, which may discourage such
lawsuits against Zomedica Corp. and its directors, officers and other employees. Alternatively, if a court were to find the choice
of forum provision contained in the certificate of incorporation to be inapplicable or unenforceable in an action, Zomedica Corp.
may incur additional costs associated with resolving such action in other jurisdictions, which could adversely affect its business
and financial condition.
We note that there
is uncertainty as to whether a court would enforce the provision and that investors cannot waive compliance with the federal securities
laws and the rules and regulations thereunder. Although we believe this provision will benefit Zomedica Corp. by providing increased
consistency in the application of Delaware law in the types of lawsuits to which it applies, the provision may have the effect
of discouraging lawsuits against Zomedica Corp.’s directors and officers.
The anticipated
benefits of the domestication may not be realized.
We may not realize
the benefits we expect from the domestication. If we do not, we will have expended considerable resources and management efforts
in completing the domestication without benefiting the company or our shareholders. Such expenditure of time and resources would
adversely affect our business, operating results, and financial condition if the anticipated benefits are not achieved.
Risks Related to our Business
We have a
limited operating history, are not profitable and may never become profitable.
We have not generated
any revenue to date, and we expect to continue to incur significant research and development costs and other expenses. Our net
loss and comprehensive loss for (i) the three months ended June 30, 2020 and June 30, 2019 was $5,307,990 and $2,404,427, respectively,
(ii) for the six months ended June 30, 2020 and June 30, 2019 was $7,758,607 and $14,081,337, respectively, and (iii) for the years
ended December 31, 2019 and December 31, 2018 was $19,784,054 and $16,647,687, respectively. Our accumulated deficit as of June
30, 2020 was $59,816,448. As of June 30, 2020, we had total shareholders' equity of $29,578,961. We expect to continue to incur
losses for the foreseeable future, as we continue our product development and commercialization activities. Even if we succeed
in developing and broadly commercializing one or more of our product candidates, we expect to continue to incur losses for the
foreseeable future, and we may never become profitable. If we fail to achieve or maintain profitability, then we may be unable
to continue our operations at planned levels and be forced to reduce or cease operations.
We may need
to raise additional capital to achieve our goals.
We do not have any
products available for sale at this time. Although we believe that we do not require pre-market approval from the U.S. Food and
Drug Administration’s Center for Veterinary Medicine, or the FDA-CVM, or the United States Department of Agriculture Animal
and Health Inspection Service’s Center for Veterinary Biologics, or USDA-CVB, to market and sell TRUMFORMATM ,
our Raman spectroscopy-based point-of-care diagnostic platform, nor our circulating tumor cell, or CTC, diagnostic assay that we
are developing, the COVID-19 pandemic has impacted our expected timing for the development and commercialization of our TRUFORMA™
platform and the five initial assays.
We are also seeking
to identify potential complementary opportunities in the animal health sectors. We will continue to expend substantial resources
for the foreseeable future to develop our existing products and any other product that we may develop or acquire. These expenditures
will include: costs of developing and validating our diagnostic products and related assays and consumables; costs associated with
conducting any required clinical trials; costs associated with completing other research and development activities; costs of identifying
additional potential products; costs associated with payments to technology licensors and maintaining other intellectual property;
costs of obtaining regulatory approvals; costs associated with securing contract manufacturers to meet our commercial manufacturing
and supply capabilities; and costs associated with marketing and selling our products. In addition, our existing and future development
agreements may require us to make significant cash milestone payments to our development partners and to pay certain development
costs. We will not control the timing of these payments. We also may incur unanticipated costs. Because the outcome of our development
activities and commercialization efforts is inherently uncertain, the actual amounts necessary to successfully complete the development
and commercialization of our existing or future products may be greater or less than we anticipate.
As a result, we may
need to obtain additional capital to fund the development of our business. Except for our unsecured working capital line we have
no existing agreements or arrangements with respect to any financings, and any such financings may result in dilution to our shareholders,
the imposition of debt covenants and repayment obligations or other restrictions that may adversely affect our business or the
value of our common shares.
Our future capital
requirements depend on many factors, including, but not limited to:
|
|
·
|
the scope, progress, results and costs of researching and developing our existing or future diagnostics
and medical device products;
|
|
|
·
|
the extent to which any of our future diagnostic assays or medical devices may be subject to USDA-CVB
pre-market regulation;
|
|
|
·
|
the timing of, and the costs involved in, obtaining regulatory approvals for any of our existing
or future diagnostics or medical device products;
|
|
|
·
|
the number and characteristics of the diagnostics and/or medical device products we pursue;
|
|
|
·
|
the cost of contract manufacturers to manufacture our existing and future diagnostic and medical
device products and any additional products we seek to commercialize;
|
|
|
·
|
the cost of commercialization activities, including marketing, sales and distribution costs;
|
|
|
·
|
the expenses needed to attract and retain skilled personnel;
|
|
|
·
|
the costs associated with being a public company;
|
|
|
·
|
our ability to establish and maintain strategic partnerships, licensing or other arrangements and
the financial terms of such agreements; and
|
|
|
·
|
the costs involved in preparing and filing patent applications, maintaining any successfully obtained
patents and protecting and enforcing any such patents.
|
Additional funds may
not be available when we need them on terms that are acceptable to us, or at all. If adequate funds are not available to us on
a timely basis, we may be required to delay, limit, reduce or terminate one or more of our product development programs or any
future commercialization efforts.
The “Novel
Coronavirus Disease 2019” (“COVID-19”) pandemic has materially and adversely affected the development and commercialization
of our TRUFORMA™ platform.
The COVID-19 pandemic
materially and adversely affected the development and commercialization of our TRUFORMA™ platform and the initial five assays.
In response to the pandemic, our development partner had reduced the number of employees working in its facilities for a period
of time which has delayed the completion of the verification of the five initial TRUFORMA™ assays and the manufacturing of
commercial quantities of the TRUFORMA™ platform and the related assays. Veterinary hospitals and clinics that had agreed
to participate in the validation of our initial TRUFORMA™ assays either shut down for a period of time or limited their operations
to those involving only life-threatening conditions, which we have mitigated to a certain extent with our recent ability to successfully
complete remote installations. Potential customers have at times restricted access to their facilities which has affected and may
continue to affect our ability to perform on-site demonstrations and other marketing activities. The extent to which the COVID-19
pandemic may impact our business will depend on future developments, which are highly uncertain and cannot be predicted with confidence,
such as the duration of the outbreak, the spread and severity of COVID-19, and the effectiveness of governmental actions in response
to the pandemic.
The COVID-19
outbreak has disrupted our development partners and the COVID-19 pandemic and any future outbreak of a health epidemic or other
adverse public health developments could materially and adversely affect our business and operating results.
The COVID-19 outbreak
disrupted our development partners and the COVID-19 pandemic and any future outbreak of a health epidemic or other adverse public
health developments could materially and adversely affect our business and operating results. For example, our development partner
for our TRUFORMA™ platform and the related assays had reduced the number of employees working in its facility which significantly
impacted our expected timing for the completion of the development and the commencement of the commercialization of our TRUFORMA™
platform and the related assays. If our suppliers are unable or fail to fulfill their obligations to us for any reason, we may
not be able to manufacture our products and satisfy customer demand or our obligations under sales agreements in a timely manner,
and our business could be harmed as a result. As noted above, there is continuing uncertainty relating to the potential effect
of COVID-19 on our business. Infections may become more widespread and should that cause supply disruptions it would have a negative
impact on our business, financial condition and operating results. In addition, a significant health epidemic could adversely affect
the economies and financial markets of many countries, resulting in an economic downturn that could affect the market for our products,
which could have a material adverse effect on our business, operating results and financial condition.
The COVID-19
pandemic and any future outbreak of a health epidemic or other adverse public health developments could materially and adversely
affect the sales of our products.
The COVID-19 pandemic
resulted in a significant spike in unemployment and a concomitant decline in economic activity in the U.S. and many other countries.
A worsening of the COVID-19 pandemic, any future outbreak of a health epidemic or other adverse public health developments may
have similar effects. Pet owners may be unwilling or unable to seek treatment for their pets in such circumstances, thereby decreasing
demand for our products. In addition, as noted above, potential customers for our products have either shut down or limited their
operations which has affected and may continue to affect our ability to perform on-site demonstrations and other marketing activities.
Potential customers also may be unwilling or unable to invest in new equipment or to introduce new treatments for their patients.
As a result, the COVID-19 pandemic and any future outbreak of a health epidemic or other adverse public health developments could
materially and adversely affect the sales of our products.
The audit
opinion on our financial statements contains a going concern modification.
As a result of our
recurring losses from operations and our accumulated deficit, the opinion of our independent registered public accountants on our
financial statements as of and for the year ended December 31, 2019 contains a going concern modification. If we are unable to
continue as a going concern, we might have to liquidate our assets and the values we receive for our assets in liquidation or dissolution
could be significantly lower than the values reflected in our financial statements. In addition, the inclusion of a going concern
modification by our independent registered public accountants, our recurring losses, our accumulated deficit and our potential
inability to continue as a going concern may materially adversely affect our share price and our ability to raise new capital or
to enter into contractual relationships with third parties.
We have generated net operating loss
carryforwards for U.S. income tax purposes, but our ability to use these net operating losses may be limited by our inability to
generate future taxable income.
Our U.S. businesses
have generated consolidated net operating loss carryforwards (“U.S. NOLs”) for U.S. federal and state income tax purposes
of approximately $16,140,344 as of December 31, 2019. These U.S. NOLs can be available to reduce income taxes that might otherwise
be incurred on future U.S. taxable income. The utilization of these U.S. NOLs would have a positive effect on our cash flow. However
there can be no assurance that we will generate the taxable income in the future necessary to utilize these U.S. NOLs and realize
the positive cash flow benefit. A portion of our U.S. NOLs have expiration dates. There can be no assurance that, if and when
we generate taxable income in the future from operations or the sale of assets or businesses, we will generate such taxable income
before such portion of our U.S. NOLs expire. Under the Tax Cuts and Jobs Act (the “TCJA”), federal NOLs generated
in tax years ending after December 31, 2017 may be carried forward indefinitely. Under the Coronavirus Aid, Relief, and Economic
Security Act (the “CARES Act”), federal NOL carryforwards arising in tax years beginning after December 31, 2017 and
before January 1, 2021 may be carried back to each of the five tax years preceding the tax year of such loss. Due to our cumulative
losses through June 30, 2020 we do not anticipate that such provision of the CARES Act will be relevant to us. The deductibility
of federal NOLs, particularly for tax years beginning after December 31, 2020, may be limited. It is uncertain if and to what
extent various states will conform to TCJA or the CARES Act.
We have generated U.S. NOLs, but
our ability to reserve and use these U.S. NOLs may be limited or impaired by future ownership changes.
Our ability to utilize
the U.S. NOLs after an “ownership change” is subject to the rules of Code Section 382. An ownership change occurs
if, among other things, the shareholders (or specified groups of shareholders) who own or have owned, directly or indirectly,
five (5%) percent or more of the value of our shares or are otherwise treated as five (5%) percent shareholders under Code Section
382 and the Treasury Regulations promulgated thereunder increase their aggregate percentage ownership of the value of our shares
by more than 50 percentage points over the lowest percentage of the value of the shares owned by these shareholders over a three-year
rolling period. An ownership change could also be triggered by other activities, including the sale of our shares that are owned
by our five (5%) shareholders. In the event of an ownership change, Section 382 would impose an annual limitation on the amount
of taxable income we may offset with U.S. NOLs. This annual limitation is generally equal to the product of the value of our shares
on the date of the ownership change multiplied by the long-term tax-exempt rate in effect on the date of the ownership change.
The long-term tax-exempt rate is published monthly by the IRS. Any unused Section 382 annual limitation may be carried over to
later years until the applicable expiration date for the respective U.S. NOLs (if any). In the event an ownership change as defined
under Section 382 were to occur, our ability to utilize our U.S. NOLs would become substantially limited. The consequence of this
limitation would be the potential loss of a significant future cash flow benefit because we would no longer be able to substantially
offset future taxable income with U.S. NOLs. There can be no assurance that such ownership change will not occur in the future.
We are substantially
dependent on the success of our TRUFORMATM platform
and cannot be certain that it will be successfully commercialized.
We are focused primarily
on the development of our TRUFORMATM diagnostic platform and the related assays. Accordingly, our near-term prospects,
including our ability to generate material product revenue, or enter into potential strategic transactions, will depend heavily
on the successful development and commercialization of this product and the related assays, which in turn will depend on a number
of factors, including the following:
|
|
·
|
the successful completion of clinical validation and verification of our TRUFORMATM
diagnostic platform and the related assays, which may take significantly longer than we anticipate and will depend, in part, upon
the satisfactory performance of our strategic partner and third-party contractors;
|
|
|
·
|
the ability of our strategic partner to manufacture supplies of our TRUFORMATM diagnostic
instrument and the related assays and to develop, validate and maintain viable commercial manufacturing processes that are compliant
with Good Manufacturing Practices, or GMP, to the extent applicable;
|
|
|
·
|
our ability to successfully market our TRUFORMATM diagnostic platform and the related
assays, whether alone or in partnership with others;
|
|
|
·
|
the availability, perceived advantages, relative cost, relative safety and relative efficacy of
TRUFORMATM diagnostic platform and the related assays compared to alternative and competing products;
|
|
|
·
|
the acceptance of our TRUFORMATM diagnostic platform and the related assays by veterinarians,
pet owners and the animal health community;
|
|
|
·
|
our ability to achieve and maintain compliance with all regulatory requirements applicable to our
business; and
|
|
|
·
|
our ability to obtain and enforce intellectual property rights, and avoid or prevail in any third-party
patent interference, patent infringement claims or administrative patent proceedings initiated by third parties or the United States
Patent and Trademark Office (“USPTO”).
|
Many of these factors
are beyond our control. Accordingly, we cannot assure you that we will be successful in developing or commercializing our TRUFORMATM
diagnostic platform and the related assays or any of our future products. If we are unsuccessful or are significantly delayed in
developing and commercializing our products, our business and prospects will be materially adversely affected, and you may lose
all or a portion of your investment.
We face unproven
markets for our products.
The companion animal
diagnostic and medical device markets are less developed than the related human markets and as a result no assurance can be given
that our products will be successful. Veterinarians, pet owners or other veterinary health providers in general may not accept
or utilize any products that we may develop.
The companion
animal care industry is subject to rapidly changing technology, which could make our products obsolete.
The companion animal
care industry is characterized by rapid technological changes, frequent new product introductions and enhancements, and evolving
industry standards, all of which could make our products obsolete. Our future success will depend on our ability to keep pace with
the evolving needs of our customers on a timely and cost-effective basis and to pursue new market opportunities that develop as
a result of technological and scientific advances. We must continuously enhance our product offerings to keep pace with evolving
standards of care. If we do not update our product offerings to reflect new scientific knowledge or new standards of care, our
products could become obsolete, which would have a material adverse effect on our business, financial condition, and results of
operations.
Our ability to successfully
develop and commercialize our existing and any future products will depend on several factors, including:
|
|
·
|
our ability to convince the veterinary community of the clinical utility of our products and their
potential advantages over existing tests and devices;
|
|
|
·
|
the willingness or ability by pet owners to pay for our products and the willingness of veterinarians
to recommend our products;
|
|
|
·
|
the willingness of veterinarians to utilize our diagnostic tests and devices; and
|
|
|
·
|
where applicable, the willingness of testing labs to buy our assay equipment.
|
Our dependence
on suppliers could limit our ability to develop and commercialize certain products
We rely on third-party
suppliers to provide components in our products, manufacture products that we do not manufacture ourselves and perform services
that we do not provide ourselves. Because these suppliers are independent third parties with their own financial objectives, actions
taken by them could have a materially negative effect on our results of operations. The risks of relying on suppliers include our
inability to enter into contracts with third-party suppliers on reasonable terms, inconsistent or inadequate quality control, relocation
of supplier facilities, supplier work stoppages and suppliers’ failure to comply with applicable regulations or their contractual
obligations. Problems with suppliers could materially negatively impact our ability to complete development, supply the market,
lead to higher costs or damage our reputation with our customers.
In addition, we currently
purchase many products and materials from sole or single sources. Some of the products that we purchase from these sources are
proprietary and, therefore, cannot be readily or easily replaced by alternative sources. To mitigate risks associated with sole
and single source suppliers, we will seek when possible to enter into long-term contracts that provide for an uninterrupted supply
of products at predictable prices. However, some suppliers may decline to enter into long-term contracts, and we are required to
purchase products with short term contracts or on a purchase order basis. There can be no assurance that suppliers with which we
do not have contracts will continue to supply our requirements for products, that suppliers with which we do have contracts will
always fulfill their obligations under these contracts, or that any of our suppliers will not experience disruptions in their ability
to supply our requirements for products. In cases where we purchase sole and single source products or components under purchase
orders, we are more susceptible to unanticipated cost increases or changes in other terms of supply. In addition, under some contracts
with suppliers we have minimum purchase obligations, and our failure to satisfy those obligations may result in loss of some or
all of our rights under these contracts or require us to compensate the supplier. If we are unable to obtain adequate quantities
of products in the future from sole and single source suppliers, we may be unable to supply the market, which could have a material
adverse effect on our results of operations.
The commercial
potential of our products is difficult to predict. The market for any product, or for companion animal diagnostics and medical
devices overall, is uncertain and may be smaller than we anticipate, which could significantly and negatively impact our revenue,
results of operations and financial condition.
We believe that the
emerging nature of our industry and our unproven business plan make it difficult to estimate the commercial potential of any of
our proposed or future products. The market for any product that we seek to commercialize will depend on important factors such
as the cost, utility and ease of use of our products, changing standards of care, preferences of veterinarians, the willingness
of pet owners to pay for such products, and the availability of competitive alternatives that may emerge either during the product
development process or after commercial introduction. If the market potential for our proposed and future products is less than
we anticipate due to one or more of these factors, it could negatively impact our business, financial condition and results of
operations. Further, the willingness of pet owners to pay for the use of our products may be less than we anticipate, and may be
negatively affected by overall economic conditions. Because relatively few pet owners purchase insurance for their companion animals,
pet owners are more likely to have to pay for the use of our products directly and may be unwilling or unable to pay for any such
use.
Our proposed
and future products will face significant competition and may be unable to compete effectively.
The development and
commercialization of veterinary diagnostics and medical devices is highly competitive, and our success depends on our ability to
compete effectively with other products in the market and identify potential partners for continued development and commercialization.
There are a number
of competitors in the diagnostic market that have substantially greater financial and operational resources and established marketing,
sales and service organizations. We expect to compete primarily with commercial clinical laboratories, hospitals’ clinical
laboratories and other veterinary diagnostic equipment manufacturers. Our principal competitors in the veterinary diagnostic market
are IDEXX Laboratories, Inc., Antech Diagnostics, a unit of VCA Inc., Abaxis, Inc., a wholly-owned subsidiary of Zoetis Inc., Heska
Corporation and Zoetis Inc. We must develop our distribution channels and build our direct sales force in order to compete effectively
in these markets. If we are unable to effectively manage our distribution channels in our highly competitive industry, we may fail
to retain customers or obtain new customers and our business will suffer.
Many of our competitors
and potential competitors have substantially more financial, technical and human resources than we do. Many also have far more
experience than we have in the development, manufacture, regulation and worldwide commercialization of animal health diagnostics
and medical devices. We also expect to compete with academic institutions, governmental agencies and private organizations that
are conducting research in the fields of animal diagnostics and medical devices. If such competing products are commercialized
prior to our products, or if our intellectual property protection fail to provide us with exclusive marketing rights for our products,
we may be unable to compete effectively in the markets in which we participate. Contractual agreements between clinics and from
competitors may limit practices’ ability to use other tests and technologies due to predetermined minimums in those agreements.
The research,
testing, manufacturing, labeling, approval, sale, marketing and distribution of our proposed and future products may be subject
to extensive regulation. We may be unable to obtain regulatory approval for our proposed or future diagnostic or medical device
products under applicable regulatory requirements or maintain any regulatory approval obtained. The denial, delay or loss of any
regulatory approval would prevent or delay our commercialization efforts and adversely affect our financial condition and results
of operations.
The research, testing,
manufacturing, labeling, approval, sale, marketing and distribution of our product candidates may be subject to extensive regulation.
We may not be able to market and sell any point-of-care diagnostic products or medical devices without pre-marketing approval from
the USDA-CVB and/or FDA-CVM. To gain approval to market a pet point-of-care diagnostic product kit or a medical device, we must
provide the results of specific tests required to be conducted in accordance with USDA-CVB and/or FDA-CVM’s guidance demonstrating
data from Assay Validation Studies that demonstrate the diagnostic accuracy, analytical sensitivity, analytical specificity and
ruggedness, and stability. In addition, we must provide manufacturing data meeting Good Manufacturing Procedures (“GMP”).
The USDA-CVB and/or FDA-CVM may also require us to conduct costly post-approval testing and/or collect post-approval safety data
to maintain our approval for any diagnostic or medical device. The results of our development activities, and the results of any
previous studies conducted by us or third parties, may not be predictive of future results of future studies, and failure can occur
at any time during or after the completion of development activities by us or our contract research organizations or CROs.
The USDA-CVB and/or
FDA-CVM can delay, limit, deny or revoke approval of any of our product candidates for many reasons, including:
|
|
·
|
if they disagree with our interpretation of data from our studies or other development efforts;
|
|
|
·
|
if they require additional studies or changes its approval policies or regulations;
|
|
|
·
|
if they do not approve of the specifications of our proposed and future products;
|
|
|
·
|
if they fail to approve the manufacturing processes of our third-party contract manufacturers;
and
|
|
|
·
|
if any approved product subsequently fails post-approval testing required by them.
|
Further, even if we
receive approval of our products, such approval may be for a more limited claim than we originally requested, the USDA-CVB may
not approve the labeling that we believe is necessary or desirable for the successful commercialization of our products and we
may be required to conduct costly post-approval testing. Any delay or failure in obtaining applicable regulatory approval for the
intended claims of our product candidates would delay or prevent commercialization of such products and would materially adversely
impact our business and prospects.
Our strategic
partnerships are important to our business. If we are unable to maintain any of these partnerships, or if these partnerships are
not successful, our business could be adversely affected.
We have entered into
a number of strategic partnerships that are important to our business and we expect to enter into similar partnerships as part
of our growth strategy. These partnerships may pose a number of risks, including:
|
|
·
|
partners may have significant discretion in determining the efforts and resources that they will
apply to these partnerships;
|
|
|
·
|
partners may not perform their obligations as expected;
|
|
|
·
|
partners may not pursue development of our product candidates or may elect not to continue or renew
development based on development results, changes in the partners’ strategic focus or available funding, or external factors,
such as an acquisition, that divert resources or create competing priorities;
|
|
|
·
|
partners could independently develop, or develop with third parties, products that compete directly
or indirectly with our products or product candidates if the partners believe that competitive products are more likely to be successfully
developed or can be commercialized under terms that are more economically attractive than ours, which may cause partners to cease
to devote resources to the development of our product candidates;
|
|
|
·
|
disagreements with partners, including disagreements over proprietary rights, contract interpretation
or the preferred course of development, might cause delays or termination of the research and development of product candidates,
might lead to additional responsibilities for us with respect to product candidates, or might result in litigation or arbitration,
any of which would be time-consuming and expensive;
|
|
|
·
|
partners may not properly maintain or defend their intellectual property rights or may use proprietary
information in such a way as to invite litigation that could jeopardize or invalidate the intellectual property or proprietary
information or expose us to potential litigation;
|
|
|
·
|
partners may infringe the intellectual property rights of third parties, which may expose us to
litigation and potential liability;
|
|
|
·
|
partners may learn about our technology and use this knowledge to compete with us in the future;
|
|
|
·
|
there may be conflicts between different partners that could negatively affect those partnerships
and potentially others; and
|
|
|
·
|
the number and type of our partnerships could adversely affect our attractiveness to future partners
or acquirers.
|
If any partnerships
we enter into do not result in the successful development of our product candidates or if one of our partners terminates its agreement
with us, we may not be able to successfully develop our product candidates, our continued development of our product candidates
could be delayed and we may need additional resources to develop additional product candidates. All of the risks relating to our
product development, regulatory approval and commercialization also apply to the activities of our partners and there can be no
assurance that our partnerships will produce positive results or successful products on a timely basis or at all.
Additionally, subject
to its contractual obligations to us, if a partner of ours is involved in a business combination or otherwise changes its business
priorities, the partner might deemphasize or terminate the development of any technology licensed to it by us. If one of our partners
terminates its agreement with us, we may find it more difficult to attract new partners and our perception in the business and
financial communities and our stock price could be adversely affected.
We may in the future
determine to partner with additional life science and technology companies for development of additional products. We face significant
competition in seeking appropriate partners. Our ability to reach a definitive agreement for partnership will depend, among other
things, upon our assessment of the partner’s resources and expertise, the terms and conditions of the proposed partnership
and the proposed partner’s evaluation of a number of factors. If we are unable to reach agreements with suitable partners
on a timely basis, on acceptable terms, or at all, we may not be able to access technologies that are important for the future
development of our business. If we elect to fund and undertake development activities on our own, we may need to obtain additional
expertise and additional capital, which may not be available to us on acceptable terms or at all. If we fail to enter into partnerships
and do not have sufficient funds or expertise to undertake the necessary development activities, we may not be able to further
develop our product candidates and our business may be materially and adversely affected.
Under the
terms of our partnership arrangements, we are required to make significant milestone and other payments to our strategic partners.
The timing of any such payments is uncertain and could adversely affect our cash flows and results of operations. If we are not
able to make such payments when due, our business could be materially and adversely affected.
In November 2018, we
entered into a development and supply agreement with Qorvo Biotechnologies, LLC, or Qorvo, a wholly-owned subsidiary of Qorvo,
Inc. Under this agreement, Qorvo is responsible for the development of certain assay cartridges and the related instrument. We
agreed to pay the associated non-recurring engineering costs of up to $500,000 per assay cartridge and the instrument and are responsible
for the validation of the assay cartridges and the instrument. Under the terms of this agreement, we were required to pay Qorvo
additional milestone payments in cash or, if elected by Qorvo, additional unregistered common shares having a value calculated
as specified in the agreement. All of the milestones under this agreement have been met and we paid Qorvo a total of $10.0 in cash
in connection therewith. Under the terms of the agreement, we will be responsible for the cost of additional development work undertaken
by Qorvo on our behalf.
In May 2018, we entered
into a development, commercialization and exclusive distribution agreement with Seraph Biosciences, Inc., or Seraph. Under this
agreement, we are responsible for development and validation, and their associated costs. Seraph is entitled to additional payments
for development costs. Seraph will be entitled to receive up to an additional $7,000,000, payable 50 percent in cash and 50 percent
in additional unregistered common shares, upon the achievement of a series of staged, specified milestones, including completion
of laboratory studies and field studies, production and commercial shipment of products. In addition, we have agreed to pay Seraph
license fees based on a percentage of gross profit from commercial sales of ZM-020. At June 30, 2020, all milestone payments remain
unpaid.
We will rely
on third parties to conduct certain portions of our development activities. If these third parties do not successfully carry out
their contractual duties or meet expected deadlines, we may be unable to obtain regulatory approval for or commercialize our product
candidates.
We have used contract
manufacturing organizations (“CMOs”) and contract research organizations (“CROs”) to conduct our manufacturing
and research and development activities. We expect to continue to do so, including with respect to our manufacturing, clinical
validation, verification and beta testing of our proposed and future diagnostic and medical device products. These CMOs and CROs
are not our employees, and except for contractual duties and obligations, we have limited ability to control the amount or timing
of resources that they devote to our programs or manage the risks associated with their activities on our behalf. We are responsible
to regulatory authorities for ensuring that products subject to regulatory authority are manufactured using good manufacturing
practices and studies are conducted in accordance with the development plans and trial protocols, and any failure by our CMOs and
CROs to do so may adversely affect our ability to obtain regulatory approvals, subject us to penalties, or harm our credibility
with regulators.
Our agreements with
our CMOs and CROs may allow termination by the CMOs and CROs in certain circumstances with little or no advance notice to us. These
agreements generally will require our CMOs and CROs to reasonably cooperate with us at our expense for an orderly winding down
of the CMOs’ and CROs’ services under the agreements. If the CMOs and CROs conducting our manufacturing and studies
do not comply with their contractual duties or obligations to us, or if they experience work stoppages, do not meet expected deadlines,
terminate their agreements with us or need to be replaced, or if the quality or accuracy of the data they obtain is compromised
due to the failure to adhere to our development protocols or quality expectations or for any other reason, we may need to secure
new arrangements with alternative CMOs and CROs, which could be difficult and costly. In such event, our studies also may need
to be extended, delayed or terminated as a result, or may need to be repeated. If any of the foregoing were to occur, regulatory
approval and commercialization of our product candidates may be delayed, and we may be required to expend substantial additional
resources.
The failure of any
CMO and CRO to perform adequately or the termination of any arrangements with any of them may adversely affect our business.
We will rely
on third-party manufacturers to produce our products. If we experience problems with any of these suppliers, the manufacturing
of our product candidates or products could be delayed.
We do not have the
capability to manufacture our proposed and future products and do not intend to develop that capability. As a result, we will rely
on CMOs to produce our proposed and future products. We expect to enter into contracts with CMOs for the commercial scale production
of the products we intend to commercialize. Reliance on CMOs involves risks, including:
|
|
·
|
the inability to meet our product specifications and quality requirements consistently;
|
|
|
·
|
inability to access production facilities on a timely basis;
|
|
|
·
|
inability or delay in increasing manufacturing capacity;
|
|
|
·
|
manufacturing and product quality issues related to the scale-up of manufacturing;
|
|
|
·
|
costs and validation of new equipment and facilities required for commercial level activity;
|
|
|
·
|
a failure to satisfy any applicable regulatory requirements on a consistent basis;
|
|
|
·
|
the inability to negotiate manufacturing agreements with third parties under commercially reasonable
terms;
|
|
|
·
|
termination or nonrenewal of manufacturing agreements with third parties in a manner or at a time
that is costly or damaging to us;
|
|
|
·
|
the reliance on a single source of supply which, if unavailable, would delay our ability to complete
the development and testing and commercialization of our products;
|
|
|
·
|
the lack of qualified backup suppliers for supplies that are currently purchased from a single
source supplier;
|
|
|
·
|
operations of our CMOs or suppliers could be disrupted by conditions unrelated to our business
or operations, including the bankruptcy of the CMO or supplier;
|
|
|
·
|
carrier disruptions or increased costs that are beyond our control; and
|
|
|
·
|
the failure to deliver products under specified storage conditions and in a timely manner.
|
Any of these risks
could cause the delay of validation studies, clinical trials, regulatory submissions, the receipt of any required approvals or
the commercialization of our products, cause us to incur higher costs and prevent us from commercializing our product candidates
successfully. Furthermore, if our CMOs fail to deliver the required commercial quantities of finished product on a timely basis
and at commercially reasonable prices and we are unable to find one or more replacement manufacturers capable of production at
a substantially equivalent cost, in substantially equivalent volumes and quality and on a timely basis, we would likely be unable
to meet demand for our products and could lose potential revenue.
Even if a
product receives regulatory approval, it may never achieve market acceptance or commercial success.
Even if we obtain USDA-CVB
or other regulatory approvals for a specific product, that product may not achieve market acceptance among veterinarians and pet
owners and may not be commercially successful. Market acceptance of our products depends on a number of factors, including:
|
|
·
|
the claims for which our products are approved or intended;
|
|
|
·
|
the acceptance by veterinarians and pet owners of the product as safe and effective;
|
|
|
·
|
the proper training and use of our products by veterinarians;
|
|
|
·
|
the potential and perceived advantages of our products over alternative diagnostics or medical
devices;
|
|
|
·
|
the cost of our products in relation to alternative diagnostics and willingness to pay for our
products, if approved, on the part of veterinarians and pet owners;
|
|
|
·
|
the willingness of pet owners to pay for the use of our products, relative to other discretionary
items, especially during economically challenging times;
|
|
|
·
|
the relative convenience and ease of use; and
|
|
|
·
|
the effectiveness of our sales and marketing efforts.
|
If our products fail
to achieve market acceptance or commercial success, our business could fail and you could lose your entire investment.
If we are
unable to establish sales capabilities on our own or through third parties, we may not be able to market and sell our existing
or future products or generate product revenue.
We do not currently
have a fully staffed sales organization. We intend to commercialize our products with a direct sales force and through third-party
distributors. To achieve this, we will be required to build a direct sales organization and to establish relationships with distributors
of veterinary products. We also will have to build our marketing, sales, managerial and other non-technical capabilities and make
arrangements with third parties for distribution and to perform certain of these other services, and we may not be successful in
doing so. Building an internal sales organization is time consuming and expensive and will significantly increase our compensation
expense. We may be unable to secure third-party distribution contracts with distributors on favorable terms or at all. We have
no prior experience in the marketing, sale and distribution of diagnostic products or medical devices for companion animals and
there are significant risks involved in building and managing a sales organization, including our ability to hire, retain and motivate
qualified individuals, generate sufficient sales leads, provide adequate training to sales and marketing personnel, and effectively
oversee a geographically dispersed sales and marketing team. If we are unable to build an effective sales organization and/or if
we are unable to secure relationships with third-party distributors for our products, we will not be able to successfully commercialize
our products, our future product revenue will suffer and we would incur significant additional losses.
In jurisdictions outside
of the United States we intend to utilize companies with an established commercial presence to market our products in those jurisdictions,
but we may be unable to enter into such arrangements on acceptable terms, it at all.
If we fail
to attract and keep senior management and key scientific personnel, we may be unable to successfully develop any of our existing
or future product candidates, conduct our in-licensing and development efforts and commercialize any of our existing or future
products.
Our success depends
in part on our continued ability to attract, retain and motivate highly qualified management and scientific personnel. We are highly
dependent upon our senior management, particularly Robert Cohen, our interim Chief Executive Officer, Ann Cotter, our Chief Financial
Officer, Stephanie Morley, DVM, our President and Chief Medical Officer, and Bruk Herbst, our Chief Commercial Officer. The loss
of services of any of these individuals could delay or prevent the successful development of our existing or future product pipeline,
completion of our planned development efforts or the commercialization of our product candidates. Although we have entered employment
agreements with Dr. Morley and Mr. Herbst for one-year terms (automatically extending for one-year terms thereafter) there can
be no assurance that either of Dr. Morley or Mr. Herbst will extend their terms of service. We have also entered into an employment
agreement with Mr. Cohen without a fixed term of service.
Consolidation
of our customers could negatively affect the pricing of our products.
Veterinarians will
be our primary customers for our proposed and future products. In recent years, there has been a trend towards the consolidation
of veterinary clinics and animal hospitals. If this trend continues, these large clinics and hospitals could attempt to leverage
their buying power to obtain favorable pricing from us and other similar companies. Any resulting downward pressure on the prices
of any of our products could have a material adverse effect on our results of operations and financial condition.
We will need
to increase the size of our organization and may not successfully manage our growth.
We will need to significantly
expand our organization and systems to support our future expected growth. If we fail to manage our growth effectively, we will
not be successful, and our business could fail.
We may seek
to raise additional funds in the future through debt financing which may impose operational restrictions on our business and may
result in dilution to existing or future holders of our common shares.
We expect that we will
need to raise additional capital in the future to help fund our business operations. Debt financing, if available, may require
restrictive covenants, which may limit our operating flexibility and may restrict or prohibit us from:
|
|
·
|
paying dividends and/or making certain distributions, investments and other restricted payments;
|
|
|
·
|
incurring additional indebtedness or issuing certain preferred shares;
|
|
|
·
|
selling some or all of our assets;
|
|
|
·
|
entering into transactions with affiliates;
|
|
|
·
|
creating certain liens or encumbrances;
|
|
|
·
|
merging, consolidating, selling or otherwise disposing of all or substantially all of our assets;
and
|
|
|
·
|
designating our subsidiaries as unrestricted subsidiaries.
|
Debt financing may
also involve debt instruments that are convertible into or exercisable for our common shares. The conversion of the debt to equity
financing may dilute the equity position of our existing shareholders.
We may not
be able to obtain or maintain sufficient insurance on commercially reasonable terms or with adequate coverage against potential
liabilities in order to protect ourselves against product liability claims.
Our business exposes
us to potential product liability risks that are inherent in the testing, manufacturing and marketing of diagnostic products and
medical devices. We may become subject to product liability claims resulting from the use of our product candidates. We do not
currently have product liability insurance and we may not be able to obtain or maintain this type of insurance for any future trials
or product candidates. In addition, product liability insurance is becoming increasingly expensive. Being unable to obtain or maintain
product liability insurance in the future on acceptable terms or with adequate coverage against potential liabilities could have
a material adverse effect on our business.
We may acquire
other businesses or form joint ventures that may be unsuccessful and could adversely dilute your ownership of our company.
As part of our business
strategy, we may pursue in-licenses or acquisitions of other complementary assets and businesses and may also pursue strategic
alliances. We have no experience in acquiring other assets or businesses and have limited experience in forming such alliances.
We may not be able to successfully integrate any acquisitions into our existing business, and we could assume unknown or contingent
liabilities or become subject to possible stockholder claims in connection with any related-party or third-party acquisitions or
other transactions. We also could experience adverse effects on our reported results of operations from acquisition-related charges,
amortization of acquired technology and other intangibles and impairment charges relating to write-offs of goodwill and other intangible
assets from time to time following an acquisition. Integration of an acquired company requires management resources that otherwise
would be available for ongoing development of our existing business. We may not realize the anticipated benefits of any acquisition,
technology license or strategic alliance.
To finance future acquisitions,
we may choose to issue shares of our common stock as consideration, which would dilute your ownership interest in us. Alternatively,
it may be necessary for us to raise additional funds through public or private financings. Additional funds may not be available
on terms that are favorable to us and, in the case of equity financings, may result in dilution to our stockholders.
Risks Related to Government Regulation
Various government
regulations could limit or delay our ability to develop and commercialize our products or otherwise negatively impact our business.
In the U.S., the manufacture
and sale of certain diagnostic products are regulated by agencies such as the USDA, the FDA or the EPA. While our point-of-care
Bulk Acoustic Wave sensor-based diagnostic platform and Raman spectroscopy-based diagnostic platform and our reference lab-based
diagnostic test for canine cancer do not require approval by the USDA-CVB prior to sale in the U.S., these diagnostic solutions
will be subject to post-marketing oversight by the FDA-CVM. In addition, delays in obtaining regulatory approvals for new products
or product upgrades could have a negative impact on our growth and profitability.
The manufacture and
sale of our products, as well as our research and development processes, are subject to similar and potentially more stringent
laws in foreign countries.
We are also subject
to a variety of federal, state, local and international laws and regulations that govern, among other things, the importation and
exportation of products; our business practices in the U.S. and abroad, such as anti-corruption and anti-competition laws; and
immigration and travel restrictions. These legal and regulatory requirements differ among jurisdictions around the world and are
rapidly changing and increasingly complex. The costs associated with compliance with these legal and regulatory requirements are
significant and likely to increase in the future.
Any failure to comply
with applicable legal and regulatory requirements could result in fines, penalties and sanctions; product recalls; suspensions
or discontinuations of, or limitations or restrictions on, our ability to design, manufacture, market, import, export or sell our
products; and damage to our reputation.
Even if we
receive regulatory approval for a product candidate, we will be subject to ongoing FDA-CVM or USDA-CVB obligations and continued
regulatory oversight, which may result in significant additional expense. Additionally, any product candidates, if approved, will
be subject to labeling and manufacturing requirements and could be subject to other restrictions. Failure to comply with these
regulatory requirements or the occurrence of unanticipated problems with our products could result in significant penalties.
If the FDA-CVM or USDA-CVB
approves any of our existing or future diagnostic product candidates, the manufacturing processes, labeling, packaging, distribution,
adverse event reporting, storage, advertising, promotion and recordkeeping for the product will be subject to extensive and ongoing
regulatory requirements. These requirements include post-marketing information and reports, establishment registration, and product
listing, as well as continued compliance with GMP, GLP and GCP for any studies that we conduct post-approval. Later discovery of
previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with our third-party
manufacturers or manufacturing processes, or failure to comply with regulatory requirements, may result in, among other things:
|
|
·
|
restrictions on the marketing or manufacturing of the product, withdrawal of the product from the
market, or voluntary product recalls;
|
|
|
·
|
fines, warning letters or holds on promotional materials and claims;
|
|
|
·
|
refusal by the FDA-CVM or USDA-CVB to approve pending applications or supplements to approved applications
filed by us or our strategic collaborators, or suspension or revocation of product license approvals;
|
|
|
·
|
product seizure or detention, or refusal to permit the import or export of products; and
|
|
|
·
|
injunctions or the imposition of civil or criminal penalties.
|
The FDA-CVM’s
or USDA-CVB’s policies may change and additional government regulations may be enacted that could prevent, limit or delay
regulatory approval of our product candidates. We cannot predict the likelihood, nature or extent of government regulation that
may arise from future legislation or administrative action in the United States or abroad. If we are slow or unable to adapt to
changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory
compliance, we may lose any marketing approval that we may have obtained and we may not achieve or sustain profitability, which
would adversely affect our business.
Legislative
or regulatory reforms with respect to veterinary diagnostics, medical devices and test kits may make it more difficult and costly
for us to obtain regulatory clearance or approval of any of our existing or future product candidates and to produce, market, and
distribute our products after clearance or approval is obtained.
From time to time,
legislation is drafted and introduced in the U.S. Congress that could significantly change the statutory provisions governing the
testing, regulatory clearance or approval, manufacture, and marketing of regulated and/or licensed products. In addition, FDA-CVM
and USDA-CVB regulations and guidance are often revised or reinterpreted by the FDA-CVM and USDA-CVB in ways that may significantly
affect our business and our products. Similar changes in laws or regulations can occur in other countries. Any new regulations
or revisions or reinterpretations of existing regulations in the United States may impose additional costs or lengthen review times
of any of our existing or future product candidates. We cannot determine what effect changes in regulations, statutes, legal interpretation
or policies, when and if promulgated, enacted or adopted may have on our business in the future. Such changes could, among other
things, require:
|
|
·
|
changes to manufacturing methods;
|
|
|
·
|
recall, replacement or discontinuance of certain products; and
|
|
|
·
|
additional record-keeping.
|
Each of these would
likely entail substantial time and cost and could materially harm our financial results. In addition, delays in receipt of or failure
to receive regulatory clearances or approvals for any future products would harm our business, financial condition, and results
of operations.
Risks Related to Intellectual Property
Our ability
to obtain intellectual property protection for our products is limited.
Our diagnostic technologies
are dependent on intellectual property developed by our strategic partners and licensed to us. We do not own the intellectual property
rights that underlie these technology licenses. Our rights to use the technology we license are subject to the negotiation of,
continuation of and compliance with the terms of our licenses. However, we have filed four U.S. patent applications and two Patent
Cooperation Treaty (PCT) applications for U.S. and international protection of our diagnostic tests. These applications cover tests
developed for our ZM-017, ZM-022 and ZM-020 technology platforms. Even if such patents are issued, we do not expect that all of
the patents will provide significant protection for our intellectual property.
Some of our
products may or may not be covered by a patent. Further if an application is filed, it is not certain that a patent will be granted
or if granted whether it will be held to be valid. All of which may impact our market share and ability to prevent others (competitor
third parties) from making, selling, or using our products.
We intend to rely upon
a combination of patents, trade secret protection, confidentiality agreements, and license agreements to protect the intellectual
property related to our proposed and future products. We may not be successful in protecting our intellectual property rights,
including our unpatented proprietary know-how and trade secrets, or in avoiding claims that we infringed on the intellectual property
rights of others. In addition to relying on patent and trademark rights, we rely on unpatented proprietary know-how and trade secrets,
and employ various methods, including confidentiality agreements with employees and consultants, customers and suppliers to protect
our know-how and trade secrets. However, these methods and our patents and trademarks may not afford complete protection and there
can be no assurance that others will not independently develop the know-how and trade secrets or develop better production methods
than us. Further, we may not be able to deter current and former employees, contractors and other parties from breaching confidentiality
agreements and misappropriating proprietary information and it is possible that third parties may copy or otherwise obtain and
use our information and proprietary technology without authorization or otherwise infringe on our intellectual property rights.
In the future, we may also rely on litigation to enforce our intellectual property rights and contractual rights, and, if not successful,
we may not be able to protect the value of our intellectual property. Any litigation could be protracted and costly and could have
a material adverse effect on our business and results of operations regardless of its outcome.
If we are
unable to obtain trademark registrations for our products our business could be adversely affected.
We have pending trademark
applications for our company name and composite marks comprised of our company name, logo and/or slogan in the U.S., Canada, European
Union, the United Kingdom, and Mexico. In addition, we have approved pending trademark applications for our “Voice of the
Vet” mark in the U.S. and Canada. We have secured two registrations in the European Union for our company name, company name
and logo, and for the mark “Voice of the Vet powered by Zomedica” (and Design). We also have secured registrations
in Brazil for our company name and logo. While we cannot make assurances that any pending trademark applications will mature to
registration, most of these applications are now poised to mature to registration.
We have also filed
for protection of several product names in the U.S., Canada and European Union. Currently, no significant hurdles have been encountered
in the registration process. Moreover, any name we propose to use with our product candidates in the United States must be approved
by the FDA-CVM or the USDA-CVB if it is a regulated product. The FDA-CVM typically conducts a review of proposed product names,
including an evaluation of potential for confusion with other product names. If the FDA-CVM or the USDA-CVB object to any of our
proposed proprietary product names, we may be required to expend significant additional resources in an effort to identify a suitable
substitute name that would qualify under applicable trademark laws, not infringe the existing rights of third parties and be acceptable
to the FDA-CVM and the USDA-CVB.
Third parties
may have intellectual property rights, which may require us to obtain a license or other applicable rights to make, sell or use
our products. If such rights are not granted or obtained, I could have a material adverse effect on our business, financial condition
and results of operations.
Our success depends
in part on our ability to obtain, or license from third parties, patents, trademarks, trade secrets and similar proprietary rights
without infringing on the proprietary rights of third parties. Although we believe our intellectual property rights are sufficient
to allow us to conduct our business without incurring liability to third parties, our products may infringe on the intellectual
property rights of such persons. Furthermore, no assurance can be given that we will not be subject to claims asserting the infringement
of the intellectual property rights of third parties seeking damages, the payment of royalties or licensing fees and/or injunctions
against the sale of our products. Any such litigation could be protracted and costly and could have a material adverse effect on
our business, financial condition and results of operations.
Our diagnostic
technologies depend on certain technologies that are licensed to us. We do not control these technologies and any loss of our rights
to them could prevent us from marketing our diagnostic product candidates.
Our diagnostic technologies
are dependent on intellectual property developed by our strategic partners and licensed to us. We do not own the intellectual property
rights that underlie these licenses. Our rights to use the technology we license are subject to the negotiation of, continuation
of and compliance with the terms of our licenses. We do not control the prosecution, maintenance or filing of the patents and other
intellectual property licensed to us, or the enforcement of these intellectual property rights against third parties. The patents
and patent applications underlying our licenses were not written by us or our attorneys, and we do not have control over the drafting
and prosecution of such rights. Our partners might not have given the same attention to the drafting and prosecution of patents
and patent applications as we would have if we had been the owners of the intellectual property rights and had control over such
drafting and prosecution. We cannot be certain that drafting and/or prosecution of the licensed patents and patent applications
has been or will be conducted in compliance with applicable laws and regulations or will result in valid and enforceable patents
and other intellectual property rights.
Our intellectual
property agreements with third parties may be subject to disagreements over contract interpretation, which could narrow the scope
of our rights to the relevant intellectual property or technology or increase our financial or other obligations to our licensors.
Certain provisions
in our intellectual property agreements may be susceptible to multiple interpretations. The resolution of any contract interpretation
disagreement that may arise could affect the scope of our rights to the relevant intellectual property or technology, or affect
financial or other obligations under the relevant agreement, either of which could have a material adverse effect on our business,
financial condition, results of operations and prospects.
In addition, while
it is our policy to require our employees and contractors who may be involved in the conception or development of intellectual
property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement
with each party who in fact conceives or develops intellectual property that we regard as our own. Our assignment agreements may
not be self-executing or may be breached, and we may be forced to bring claims against third parties, or defend claims they may
bring against us, to determine the ownership of what we regard as our intellectual property.
We may be
subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information
of third parties.
We have received confidential
and proprietary information from third parties. In addition, we employ individuals who were previously employed at other pharmaceutical
or animal health companies. We may be subject to claims that we or our employees, consultants or independent contractors have inadvertently
or otherwise improperly used or disclosed confidential information of these third parties or our employees’ former employers.
Litigation may be necessary to defend against any such claims. Even if we are successful in defending against any such claims,
such litigation could result in substantial cost and be a distraction to our management and employees.
Risks Related to Our Preferred Shares
We will be
obligated to pay a significant portion of our net sales to the holders of our Series 1 Preferred Shares. This payment obligation
will materially and adversely affect our liquidity and capital resources, may adversely impact our ability to raise additional
capital, and could adversely affect the trading price of our common shares.
We are obligated to
make annual payments to the holders of our Series 1 Preferred Shares in an amount equal to nine percent of the net sales (as defined
in the Series 1 Preferred Shares), if any, of our company and our affiliates (the “Net Sales Payments”) until such
time as the holders have received total Net Sales Payments equal to nine times the aggregate stated value of the outstanding Series
1 Preferred Shares. Such payments will materially and adversely affect our liquidity and capital resources which could result in
a shortage of capital necessary to fund our operations or to take advantage of business opportunities as they arise. Our obligation
to make these payments may make it more difficult for us to raise additional capital on terms acceptable to us, or at all. This
payment obligation also may adversely affect investor perceptions of our company which could adversely affect the trading price
of our common shares.
In the event
of a sale of our company, holders of our Series 1 Preferred Shares will be entitled to a substantial premium on the purchase price
they paid for their Series 1 Preferred Shares, which will reduce the sale proceeds to be received by holders of our common shares.
In the event that our
company is the subject of a “fundamental transaction” (defined in the Series 1 Preferred Shares to include an amalgamation,
merger or other business combination transaction involving our company in which our shareholders do not have the right to cast
more than 50% of the votes that may be cast for the election of directors, or a sale, lease or other disposition of the properties
and/or assets of our company as an entirety or substantially as an entirety to a third party) the holders of the Series 1 Preferred
Shares will have the right, in preference to the holders of our common shares, to receive a portion of the aggregate consideration
paid in the fundamental transaction that will represent a substantial premium on the purchase price they paid for their Series
1 Preferred Shares. Such premium will reduce the proceeds of any such fundamental transaction that would be received by holders
of our common shares.
In the event
of the liquidation, dissolution or winding up of our company, holders of the Series 1 Preferred Shares will have a liquidation
preference over holders of our common shares and if the net assets of our company available for distribution to holders of our
equity securities is not sufficient to pay this liquidation preference in full, holders of our common shares would receive no liquidating
distribution in respect of their common shares.
In the event of the
liquidation, dissolution or winding up of our company, holders of the Series 1 Preferred Shares will have a liquidation preference
equal to the stated value of the Series 1 Preferred Shares less the Net Sales Returns (as defined in the Series 1 Preferred Shares)
paid on the Series 1 Preferred Shares before holders of our common shares would be entitled to any proceeds of such liquidation,
dissolution or winding up. If the net assets of our company available for distribution to holders of our equity securities is not
sufficient to pay this liquidation preference in full, holders of our common shares would receive no liquidating distribution in
respect of their common shares.
Our Series
1 Preferred Shares will be reclassified as a liability on our consolidated balance sheet once we begin to recognize revenues which
may cause us to fail to meet the NYSE American’s continued listing requirements.
Because we are obligated
to make annual payments to the holders of our Series 1 Preferred Shares in an amount equal to nine percent of the Net Sales (as
defined in the Series 1 Preferred Shares), if any, of our company and our affiliates, once we begin to recognize revenues from
our commercial activities, we will be required under United States general accounting principles to reclassify the Series 1 Preferred
Shares as a liability on our consolidated balance sheet. The reclassification will significantly increase our total liabilities
and significantly reduce our shareholders’ equity. Under the NYSE American’s continued listing requirements, we are
required to maintain shareholders’ equity of at least $4.0 million, which will increase to $6.0 million after December 31,
2020. As a result of the reclassification, we may fail to meet this continued listing requirement. If we are unable to satisfy
the NYSE American’s continued listing requirements, our common shares could be delisted from the NYSE American which could
adversely affect the liquidity and market price of our common shares.
Risks Related to Our Common Shares
If securities
or industry analysts do not publish research or reports about our company, or if they issue adverse or misleading opinions regarding
us or our stock, our stock price and trading volume could decline.
Although we have research
coverage by securities and industry analysts, if coverage is not maintained, the market price for our stock may be adversely affected.
Our stock price also may decline if any analyst who covers us issues an adverse or erroneous opinion regarding us, our business
model, our intellectual property or our stock performance, or if our product validations and operating results fail to meet analysts’
expectations. If one or more analysts cease coverage of us or fail to regularly publish reports on us, we could lose visibility
in the financial markets, which could cause our stock price or trading volume to decline and possibly adversely affect our ability
to engage in future financings.
We expect
that the price of our common shares will fluctuate substantially.
You should consider
an investment in our common shares risky and invest only if you can withstand a significant loss and wide fluctuations in the market
value of your investment. The price of our common shares that will prevail in the market after the sale of our common shares by
a selling shareholder may be higher or lower than the price you have paid. Numerous factors, including many over which we have
no control, may have a significant impact on the market price of our common shares. These risks include those described or referred
to in this “Risk Factors” section and elsewhere in this report as well as, among other things:
|
|
·
|
any delays in, or suspension or failure of, our existing and future studies;
|
|
|
·
|
announcements of regulatory approval or disapproval of any of our existing or future product candidates
or of regulatory actions affecting us or our industry;
|
|
|
·
|
delays in the commercialization of our existing or future product candidates;
|
|
|
·
|
manufacturing and supply issues related to our development programs and commercialization of our
existing or future product candidates;
|
|
|
·
|
quarterly variations in our results of operations or those of our competitors;
|
|
|
·
|
changes in our earnings estimates or recommendations by securities analysts or adverse publicity
about us or our product candidates;
|
|
|
·
|
announcements by us or our competitors of new product candidates, significant contracts, commercial
relationships, acquisitions or capital commitments;
|
|
|
·
|
announcements relating to future development or license agreements including termination of such
agreements;
|
|
|
·
|
adverse developments with respect to our intellectual property rights or those of our principal
collaborators;
|
|
|
·
|
commencement of litigation involving us or our competitors;
|
|
|
·
|
any major changes in our board of directors or management;
|
|
|
·
|
new legislation in the United States relating to the prescription, sale, distribution or pricing
of pet pharmaceuticals or diagnostic products;
|
|
|
·
|
product liability claims, other litigation or public concern about the safety of our product candidates
or future products;
|
|
|
·
|
market conditions in the animal health industry, in general, or in the pet therapeutics sector,
in particular, including performance of our competitors; and
|
|
|
·
|
general economic conditions in the United States and abroad.
|
In addition, the stock
market, in general, or the market for stocks in our industry, in particular, may experience broad market fluctuations, which may
adversely affect the market price or liquidity of our common shares. Any sudden decline in the market price of our common shares
could trigger securities class-action lawsuits against us. If any of our shareholders were to bring such a lawsuit against us,
we could incur substantial costs defending the lawsuit and the time and attention of our management would be diverted from our
business and operations. We also could be subject to damages claims if we are found to be at fault in connection with a decline
in our stock price.
We are an
“emerging growth company,” as defined under the JOBS Act and if we take advantage of reduced disclosure requirements
applicable to “emerging growth companies,” our common shares could be less attractive to investors.
We are an “emerging
growth company,” as defined in the Jumpstart Our Business Startups Act of 2012, as amended, or the JOBS Act, and, for as
long as we continue to be an “emerging growth company,” we may choose to take advantage of certain exemptions from
various reporting requirements applicable to other public companies but not to “emerging growth companies,” including,
but not limited to, not being required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley
Act of 2002, as amended, or SOX, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy
statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder
approval of any golden parachute payments not previously approved. We could be an “emerging growth company” for up
to five years, or until the earliest of (i) the last day of the first fiscal year in which our annual gross revenues exceed $1.07
billion, (ii) the date that we become a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act,
which would occur if the market value of our common shares that is held by non-affiliates exceeds $700 million as of the last business
day of our most recently completed second fiscal quarter, or (iii) the date on which we have issued more than $1 billion in non-convertible
debt during the preceding three year period. We cannot predict if investors will find our common shares less attractive if we choose
to continue to rely on these exemptions. If some investors find our common shares less attractive as a result of any choices to
reduce future disclosure, there may be a less active trading market for our common shares and our stock price may be more volatile.
In addition, Section
107 of the JOBS Act provides that an “emerging growth company” can take advantage of the extended transition period
provided in Section 7(a)(2)(B) of the Securities Act for complying with new or revised accounting standards. An “emerging
growth company” can delay the adoption of certain accounting standards until those standards would otherwise apply to private
companies. We have chosen to “opt out” of such extended transition period, however, and, as a result, we are required
to comply with new or revised accounting standards on the relevant dates on which adoption of such standards is required for non-emerging
growth companies. Section 107 of the JOBS Act provides that our decision to opt out of the extended transition period for complying
with new or revised accounting standards is irrevocable.
Our Articles
of Amalgamation (as amended) authorize us to issue an unlimited number of common shares and preferred shares without shareholder
approval and we may issue additional equity securities, or engage in other transactions that could dilute your ownership interest,
which may adversely affect the market price of our common shares
Our Articles of Amalgamation
(as amended) authorize our Board of Directors, subject to the provisions of the ABCA, to issue an unlimited number of common shares
and preferred shares without shareholder approval. Our Board of Directors may determine from time to time to raise additional capital
by issuing common shares, preferred shares or other equity securities. We are not restricted from issuing additional securities,
including securities that are convertible into or exchangeable for, or that represent the right to receive, common shares or preferred
shares. Because our decision to issue securities in any future offering will depend on market conditions and other factors beyond
our control, we cannot predict or estimate the amount, timing, or nature of any future offerings, or the prices at which such offerings
may be affected. Additional equity offerings may dilute the holdings of our existing shareholders or reduce the market price of
our common shares, or both. Holders of our common shares are not entitled to pre-emptive rights or other protections against dilution.
New investors also may have rights, preferences and privileges that are senior to, and that adversely affect, the then-current
holders of our common shares. Additionally, if we raise additional capital by making offerings of debt or preference shares, upon
our liquidation, holders of our debt securities and preferred shares, and lenders with respect to other borrowings, may receive
distributions of our available assets before the holders of our common shares.
We have incurred
significant costs as a result of operating as a U.S. public company, and our management will continue to devote substantial time
to new compliance initiatives.
As a U.S. publicly
traded company, we will incur additional significant legal, accounting and other expenses that we have not incurred in the past,
particularly after we are no longer an “emerging growth company” as defined under the JOBS Act. In addition, new and
changing laws, regulations and standards relating to corporate governance and public disclosure, including the Dodd-Frank Wall
Street Reform and Consumer Protection Act and the rules and regulations promulgated and to be promulgated thereunder, as well as
under the Sarbanes-Oxley Act, the JOBS Act, and the rules and regulations of the U.S. Securities and Exchange Commission, or SEC,
have created uncertainty for U.S. public companies and increased our costs and time that our board of directors and management
must devote to complying with these rules and regulations. We expect these rules and regulations to increase our legal and financial
compliance costs and lead to a diversion of management time and attention from revenue generating activities.
For as long as we remain
an “emerging growth company” as defined in the JOBS Act, we may choose to take advantage of certain exemptions from
various reporting requirements that are applicable to other U.S. public companies that are not “emerging growth companies.”
These exceptions provide for, but are not limited to, relief from the auditor attestation requirements of Section 404 of the Sarbanes-Oxley
Act, less extensive disclosure obligations regarding executive compensation in our periodic reports and proxy statements, exemptions
from the requirements to hold a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute
payments not previously approved and an extended transition period for complying with new or revised accounting standards. We may
take advantage of these reporting exemptions until we are no longer an “emerging growth company.” We may remain an
“emerging growth company” for up to five years. See “JOBS Act” in this report. To the extent we are no
longer eligible to use exemptions from various reporting requirements under the JOBS Act, we may be unable to realize our anticipated
cost savings from those exemptions.
Failure to
maintain effective internal control over financial reporting in accordance with Section 404 of the Sarbanes-Oxley Act could have
a material adverse effect on our business and share price.
As a Canadian public
company, we were not required to evaluate our internal control over financial reporting in a manner that meets the standards of
U.S. public companies required by Section 404 of the Sarbanes-Oxley Act, or Section 404. We were required to meet these standards
in the course of preparing our financial statements as of and for the year ended December 31, 2019, and our management has reported
on the effectiveness of our internal control over financial reporting for such year. Additionally, under the JOBS Act, our independent
registered public accounting firm is not required to attest to the effectiveness of our internal control over financial reporting
pursuant to Section 404 of the Sarbanes-Oxley Act until we are no longer an “emerging growth company.” The rules governing
the standards that must be met for our management to assess our internal control over financial reporting are complex and require
significant documentation, testing and possible remediation.
In connection with
the implementation of the necessary procedures and practices related to internal control over financial reporting, we may identify
deficiencies that we may not be able to remediate in time to meet the deadline imposed by the Sarbanes-Oxley Act for compliance
with the requirements of Section 404. In addition, we may encounter problems or delays in completing the implementation of any
requested improvements and receiving a favorable attestation in connection with the attestation provided by our independent registered
public accounting firm. We will be unable to issue securities in the public markets through the use of a shelf registration statement
if we are not in compliance with Section 404. Furthermore, failure to achieve and maintain an effective internal control environment
could have a material adverse effect on our business and share price and could limit our ability to report our financial results
accurately and timely.
If we sell
common shares in future financings, shareholders may experience immediate dilution and, as a result, our share price may decline.
We may from time to
time issue additional common shares at a discount from the existing trading price of our common shares. As a result, our shareholders
would experience immediate dilution upon the sale of any shares of our common shares at such discount. In addition, as opportunities
present themselves, we may enter into financing or similar arrangements in the future, including the issuance of debt securities,
preferred shares or common shares. If we issue common shares or securities convertible into common shares, our common shareholders
would experience additional dilution and, as a result, our share price may decline.
Future sales
of our common shares by our shareholders or the perception that these sales may occur could cause our stock price to decline.
As of the Record Date, we had 564,051,438 common shares outstanding. Substantially all of
our outstanding common shares have been registered for resale or other disposition by the holders thereof or are otherwise freely
tradable by the holders thereof.
Sales of a substantial
number of our common shares by our shareholders or the perception that these sales may occur, could depress the market price of
our common shares and could impair our ability to raise capital through the sale of additional equity securities, even if there
is no relationship between such sales and the performance of our business.
We have never
and do not, in the future, intend to pay dividends on our common shares, and your ability to achieve a return on your investment
will depend on appreciation in the market price of our common shares.
We have never paid
and do not expect to pay dividends on our common shares in the future. We intend to invest our future earnings, if any, to fund
our growth and not to pay any cash dividends on our common shares. Since we do not intend to pay dividends, your ability to receive
a return on your investment will depend on any future appreciation in the market price of our common shares. There is no assurance
that our common shares will appreciate in price.
An active,
liquid and orderly market for our common shares may not develop or be sustained, and you may not be able to sell your common shares.
Our common shares trade
on the NYSE American exchange. We cannot assure you that an active trading market for our common shares will develop or be sustained.
The lack of an active market may impair your ability to sell the common shares at the time you wish to sell them or at a price
that you consider reasonable. An inactive market may also impair our ability to raise capital by selling common shares and may
impair our ability to acquire other businesses, applications or technologies using our common shares as consideration, which, in
turn, could materially adversely affect our business.
We are subject
to the continued listing requirements of the NYSE American. If we are unable to comply with such requirements, our common shares
would be delisted from the NYSE American, which would limit investors’ ability to effect transactions in our common shares
and subject us to additional trading restrictions.
Our common shares are currently listed on the NYSE American. In order to maintain our listing,
we must maintain certain share prices, financial and share distribution targets, including maintaining a minimum amount of shareholders’
equity and a minimum number of public shareholders. In addition to these objective standards, the NYSE American may delist the
securities of any issuer if, in its opinion, the issuer’s financial condition and/or operating results appear unsatisfactory;
if it appears that the extent of public distribution or the aggregate market value of the security has become so reduced as to
make continued listing on the NYSE American inadvisable; if the issuer sells or disposes of principal operating assets or ceases
to be an operating company; if an issuer fails to comply with the NYSE American’s listing requirements; if an issuer’s
common stock sells at what the NYSE American considers a “low selling price” (generally trading below $0.20 per share
for an extended period of time); or if any other event occurs or any condition exists which makes continued listing on the NYSE
American, in its opinion, inadvisable. On April 10, 2020, we received a deficiency letter from the NYSE American indicating that
the we are not compliance with Section 1003(f)(v) of the NYSE American Company Guide, because our common shares have been selling
for a low price per share for a substantial period time. In addition, the NYSE American has advised us that if the trading price
of our common shares falls below $0.06 per share, our common shares may be immediately suspended from further trading on the exchange.
Our shareholders failed
to approve a proposal to effect a reverse split of our common shares at our annual and special meeting of shareholders held on
September 25, 2020. If shareholders approve the domestication and the domestication is effected, we intend to seek stockholder
approval for a reverse split of our common stock promptly thereafter to regain compliance with the NYSE American’s continued
listing standards, although no assurance can be given that stockholders will approve a reverse stock split.
If the NYSE American
delists our common shares from trading on its exchange and we are not able to list our securities on another national securities
exchange, we expect our common shares would qualify to be quoted on an over-the-counter market. If this were to occur, we could
face significant material adverse consequences, including:
|
|
·
|
a limited availability of market quotations for our securities;
|
|
|
·
|
reduced liquidity for our securities;
|
|
|
·
|
a determination that our common shares are a “penny stock” which will require brokers
trading in our common shares to adhere to more stringent rules and possibly result in a reduced level of trading activity in the
secondary trading market for our securities;
|
|
|
·
|
a limited amount of news and analyst coverage; and
|
|
|
·
|
a decreased ability to issue additional securities or obtain additional financing in the future.
|
CAUTIONARY
NOTE REGARDING Forward-Looking Statements
This proxy circular/prospectus
contains forward-looking statements, or forward-looking information, as defined under applicable securities laws (collectively,
“forward-looking statements”). In some cases, you can identify these statements by forward-looking words such as “may,”
“might,” “will,” “should,” “expects,” “plans,” “anticipates,”
“believes,” “estimates,” “predicts,” “potential” or “continue,” the
negative or plural of these words and other comparable terminology. Forward-looking statements in this prospectus include, but
are not limited to, statements about:
|
|
·
|
the success, cost and timing of our research and development activities, validation studies and
beta testing, including with respect to our lead product, TRUFORMA™;
|
|
|
·
|
our ability to obtain and maintain any required approvals from the USDA Center for Veterinary Biologics
for our proposed and future diagnostic products, to the extent applicable;
|
|
|
·
|
our ability to obtain funding for our operations;
|
|
|
·
|
the ability of our contract research organizations to appropriately conduct our safety studies
and certain development activities;
|
|
|
·
|
the ability of our contract manufacturing organizations to manufacture and supply our products;
|
|
|
·
|
our plans to develop and commercialize our planned and future products;
|
|
|
·
|
the expected impact of the novel coronavirus pandemic on our operations, including the development
and commercialization of our TRUFORMA™ platform and the five initial assays;
|
|
|
·
|
our ability to develop and commercialize products that can compete effectively;
|
|
|
·
|
the size and growth of the veterinary diagnostics and medical device markets;
|
|
|
·
|
our ability to obtain and maintain intellectual property protection for our planned and future
products candidates;
|
|
|
·
|
regulatory developments in the United States;
|
|
|
·
|
the loss of key scientific or management personnel;
|
|
|
·
|
our expectations regarding the period during which we will be an “emerging growth company”
under the JOBS Act;
|
|
|
·
|
the accuracy of our estimates regarding expenses, future revenues, capital requirements and needs
for additional financing;
|
|
|
·
|
our status as a “passive foreign investment company” for U.S. federal income tax purposes;
and
|
|
|
·
|
the anticipated U.S. and Canadian federal income tax consequences of the domestication.
|
These forward-looking
statements, which are subject to risks, uncertainties and assumptions about us, may include projections of our future financial
performance, our anticipated growth strategies and anticipated trends in our business. These statements are only predictions based
on our current expectations and projections about future events. There are important factors that could cause our actual results,
level of activity, performance or achievements to differ materially from the results, level of activity, performance or achievements
expressed or implied by the forward-looking statements, including those factors discussed under the caption entitled “Risk
Factors” above.
Although we believe
the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, level of activity,
performance or achievements. Moreover, neither we nor any other person assumes responsibility for the accuracy and completeness
of any of these forward-looking statements. We are under no duty to update any of these forward-looking statements after the date
of this proxy circular/prospectus to conform our prior statements to actual results or revised expectations.
Business
Overview
We are a veterinary
health company creating products for companion animals by focusing on the unmet needs of clinical veterinarians. We expect that
our product portfolio will include innovative diagnostics and medical devices that emphasize patient health and practice health.
With a team that includes clinical veterinary professionals, our goal is to provide veterinarians the opportunity to increase productivity
and grow revenue while better serving the animals in their care.
Market Opportunity
U.S. consumers are
expected to spend an estimated $99 billion on their pets in 2020, according to the American Pet Products Association, or APPA,
an increase of approximately 4% from 2019 ($95.7 billion). According to a 2019 pet owner survey conducted by American Pet Products
Association (APPA), 67% of U.S. households have at least one pet. This equates to nearly 85 million homes. Eighty-three percent
of dog-owning households visit the vet every year and it is estimated that there were 170 million clinical visits in the U.S in
2018. The veterinary care segment is expected to account for an estimated $30.2 billion in revenue in 2020, an increase of approximately
3% ($29.3 billion) from 2019. According to dvm360 Magazine’s State of the Profession survey for 2015, diagnostics comprise
18%, and vaccinations, pharmaceuticals and biologicals comprise 25% of gross revenue at the veterinary practice level.
Veterinary diagnostics
is a growing market. The dvm360 Magazine survey also states that 61% of respondents indicated that they were providing more diagnostic
services than the prior year. Similarly, a 2016 Credit Suisse survey of veterinarians found that 73% of respondents expected their
diagnostic testing to increase over the next 12 months. According to MarketsandMarkets, the companion animal diagnostics market
is projected to reach $2.8 billion by 2024 from $1.7 billion in 2019, at a Compounded Annual Growth Rate, or CAGR of 9.8%. The
growth in this market is driven by the rising companion animal population, increasing utilization of pet insurance, and the growth
in the number of veterinary practitioners in developed countries. The growing demand for rapid tests and portable instruments for
point-of-care services is expected to offer potential growth opportunities for market players in the coming years. Furthermore,
in 2020, IHS Markit reported that the global veterinary immunodiagnostic market was expected to grow at a CAGR of 9.6% and reach
$2.1 billion by 2022.
We believe that several
factors have contributed and will continue to contribute to an increase in spending on pet healthcare. Companion animals are generally
living longer, with the average lifespan for dogs increasing to 15.4 years, according to the University of Washington’s Dog
Aging Project. In 2015, the American Animal Hospital Association estimated that the average dog will need approximately $3,600
in veterinary care over its lifespan. We believe humanization of pets, longer pet lifespan, and the physical and emotional benefits
of support animals will bolster growth of the veterinary health care market.
COVID-19 restrictions
appear to have positively affected the relationship people have with their pets. The American Pet Products Association reports
greater than 70% of pet owners surveyed in its COVID-19 Pulse Survey indicated they were spending more time with pets, which alleviated
the stress of social distancing and isolation. New pet adoptions and fostering appear to have risen at least early in the pandemic
with USA Today reporting many shelters experiencing a significant reduction in their shelter populations. While it is unknown if
a permanent increase in pet placement has occurred, demand for veterinary services is rebounding as restrictions ease and veterinary
services are deemed essential. VetSuccess tracks weekly financial performance for veterinary practices across the United States.
As of September 30, 2020, it reports financial performance for 3,040 clinics. While year over year revenue and invoices for these
practices were down by 2.3% and 5.4% respectively for the period beginning February 1 and ending April 30, there has been steady
improvement each month with VetSuccess reporting year over year increase in revenue by 18.5% and invoices by 8.2% for the month
of September. The market for companion animal veterinary services has long been considered recession proof by some as pet care
expenditures grew during the 2001 and 2008-2009 recessions.
Development of Companion Animal Diagnostics
The development of
companion animal diagnostics continues to evolve with the addition of new technologies to diagnostic portfolios. We believe that
these new technologies may allow for the following:
|
|
·
|
Enhanced capability to detect the frequency of occurrence and severity of diseases and conditions
that impact companion animals;
|
|
|
·
|
Increased accuracy and faster means to obtain test results;
|
|
|
·
|
Wider availability of new diagnostic tools; and
|
|
|
·
|
Enhanced economic benefits for veterinarians.
|
Compared to human diagnostic
development, the development of companion animal diagnostics is generally faster and less expensive since it typically requires
smaller clinical studies, with fewer subjects. We believe that the lower cost of developing companion animal diagnostics enables
us to pursue multiple diagnostic candidates simultaneously and to spread the risk of failure across a number of candidates, rather
than concentrating all of our resources on one diagnostic candidate that may ultimately fail to achieve regulatory approval or
market acceptance.
Unmet Medical Needs
We believe that there
is a significant unmet medical need for faster, more efficient and accurate disease/condition detection solutions for veterinarians.
We have identified diagnostic assay candidates that we believe have the potential to satisfy unmet needs or improve upon existing
diagnostic processes frequently used by companion animal veterinarians.
Product Pipeline
TRUFORMA™
Platform
Our strategic focus
is on the final development and commercialization of our TRUFORMA™ diagnostic biosensor platform and the first five assays
for the detection of thyroid disorders in dogs and cats and adrenal disorders in dogs. The TRUFORMA™ platform uses Bulk Acoustic
Wave (BAW) technology to provide a non-optical and fluorescence free detection system for use at the point-of-care. We believe
that BAW technology will enable precise and repeatable test results at the point-of-care during a typical veterinary appointment.
The TRUFORMATM platform is being developed together with Qorvo Biotechnologies, LLC., or Qorvo Biotech.
Dogs commonly suffer
from thyroid and adrenal disorders, while cats commonly suffer from thyroid disease. Diagnostics are a vital part of identifying
these disorders in sick patients exhibiting signs of illness as well as screening apparently healthy patients at risk for disease
to detect disease before significant illness occurs. Multiple assays must be performed to reach a definitive diagnosis. The ability
to run all the necessary tests at the point-of- care does not currently exist and tests must often be sent to a reference lab.
The three thyroid assays we are developing are: total T4 (thyroxine) validated for dogs and cats; free T4 validated for dogs and
cats; and TSH (thyroid stimulating hormone) validated for dogs and cats. The two adrenal assays we are developing are cortisol
and endogenous ACTH (adrenocorticotropic hormone) validated for dogs.
Current methods for
diagnosing the two most common canine adrenal diseases, Cushing’s and Addison’s, may require multiple visits, potential
hospitalization of dogs for injections and multiple blood draws over time which is expensive, stresses patients, and requires significant
veterinary staff time. The availability of canine eACTH at point-of-care has the potential to change the way veterinarians manage
two chronic and potentially life-threatening diseases in dogs by providing accurate results more quickly.
Hyperthyroidism is
a common and chronic metabolic disorder in cats, requiring life-long treatment. It is estimated that over 10 percent of all senior
cats will develop hyperthyroidism. (Feline hyperthyroidism - J Feline Med Surg. 2012 Nov;14(11):804-18. doi: 10.1177/1098612X12464462.
Hyperthyroidism in cats: what's causing this epidemic of thyroid disease and can we prevent it? Peterson M.)
Verification of TRUFORMA’s™
three initial assays was completed in the second quarter of 2020. Assuming successful completion of verification of the remaining
two assays we intend to commence a pilot program in parallel to the validation work to optimize the customer experience. We believe
our initial assays do not require premarket regulatory approval by U.S. regulators. Following commercial launch of TRUFORMA™,
we will proceed with the development of the platform’s next three assays for non-infectious gastrointestinal disease.
Pathogen
Detection Program
Following the commercial
launch of TRUFORMA™, we expect to continue the development of another point-of-care diagnostic platform, which is based on
miniaturized laser-based Raman spectroscopy technology and is designed to detect pathogens in companion animals. We believe this
platform will enable the identification of biological and biochemical signatures in complex biological samples and has the potential
to achieve reference lab sensitivity/specificity to screen for a wide range of pathogens in companion animal feces, urine, respiratory,
and dermatological samples in minutes without the need for extensive sample prep or the use of reagents. The diagnostic platform
requires a small fecal sample preparation. Additionally, the platform has automated analysis and does not require specialized staff
training. We believe that this diagnostic platform does not require pre-market regulatory approval for use with companion animals
in the United States. This platform is being developed together with Seraph Biosciences, Inc., or Seraph.
Veterinarians must
use labor intensive, unreliable manual processes to identify fecal parasitic infections at the point-of-care. Consequently, veterinarians
frequently send samples out to reference labs to test for and identify parasitic infections, which is relatively expensive and
may take several days, delaying diagnosis and treatment. We expect that our diagnostic platform will deliver multiple benefits
over existing technology, including speed of results with minimal sample preparation time to achieve real-time analysis of a sample
in a disposable cuvette. We believe testing with our platform can result in a reliable identification of fecal and urine infections
within minutes. In addition to faster results and treatment, an added benefit to pet owners is expected to be the small sample
size requirements, which are expected to reduce the burden of collection by the owner prior to the visit and stress associated
with direct collection during the patient visit.
We have filed a U.S.
patent application covering compositions and methods for five fecal parasite detection assays for detecting parasitic infections
in cats and dogs and submitted a U.S. provisional patent application covering compositions and methods for detecting urinary tract
infections for use in our pathogen detection platform.
“Liquid
Biopsy” Platform
We have performed initial
development work on a circulating tumor cell (CTC) “liquid biopsy” platform for use in a reference lab setting as a
canine cancer diagnostic. This platform is intended for use to detect canine cancers faster, more affordably and less invasively
compared to existing methods, which can be expensive and cost prohibitive for pet owners. We have worked on the development of
an assay for use with this platform that targets hard-to-diagnose canine cancers, such as hemangiosarcoma and osteosarcoma. This
platform is being developed together with Celsee, Inc., a Bio-Rad Laboratories company, or Celsee.
The liquid biopsy is
a blood test that we believe has the potential to detect the presence of CTCs, which are cells that have shed from a primary tumor
into neighboring blood vessels and are transported throughout the body’s circulatory system. Diagnosing certain cancers in
canines is difficult because the location of the tumor may make it difficult or risky to obtain cell material through a biopsy.
In addition, the overall health of a canine may increase the risk of performing a biopsy. Our initial development work focused
on testing for difficult to biopsy cancers such as hemangiosarcoma and osteosarcoma in canines. Other diagnostic technologies,
such as advanced imaging, are expensive while others, such as histopathologic examination, may take several days or more to provide
a definitive diagnosis. We believe that the detection of CTCs in the blood could provide strong clinical support for a cancer diagnosis
without the need for an invasive tissue biopsy or other expensive or time-consuming diagnostic test.
We have done development
work on an assay for the detection of a blood-borne lymphoma cancer intended for use with this liquid biopsy platform. Lymphomas
represent approximately 10-25% of all cancers diagnosed in dogs.
The lymphoma assay
is designed to identify specific genetic abnormalities using fluorescence in situ hybridization (FISH). FISH tests are regularly
used for cancers in human medicine, such as the HER2 breast cancer test.
In early 2020, we successfully
completed the development and manufacturing milestones for our liquid biopsy platform.
Consistent with our
focus on the development of point-of-care diagnostic platforms, we intend to seek one or more partners for the further development
and commercialization of the liquid biopsy platform.
License
Agreements
In November 2018, we
entered into a development and supply agreement with Qorvo focused on bringing Qorvo’s piezo-electric BAW sensor to the veterinary
health sector. Under the terms of this agreement, we have exclusive, global rights to develop and market Qorvo's investigational
point-of-care diagnostic platform for veterinary use. Under the agreement, Qorvo and we will collaborate on the development of
veterinary diagnostic assays. The joint development work initially targets five assay cartridge candidates to detect the thyroid
and adrenal disorders in dogs and cats. Qorvo is responsible for the development of the assay cartridges and the instrument. We
have agreed to pay for the associated non-recurring engineering costs of up to $500,000 per assay cartridge and the instrument
and are responsible for the validation of the assay cartridges and the instrument. Qorvo will supply us, on an exclusive basis,
with the instruments and the related assay cartridges to be developed under the agreement pursuant to a rolling forecast, subject
to specified minimum purchase requirements, at prices specified in the agreement. We will be responsible for the marketing and
sale of the disposable assay cartridges and instruments.
The agreement, which
is exclusive worldwide in the practice of veterinary medicine for the health and wellbeing of any non-human animal, has an initial
term of ten years (subject to early termination and extension in certain circumstances).
We paid Qorvo $1.0
million and issued to Qorvo unregistered common shares having a value of $4.4 million, consisting of an aggregate of 2,565,789
common shares with an ascribed price of $1.52 per share. We paid an additional $5 million of milestone payments in the first quarter
of 2019. In the second and third quarters of 2020, we paid our final two milestone payments to Qorvo for $3 million and $2 million,
respectively. In connection with the agreement, we entered into a registration rights agreement providing Qorvo with certain registration
rights with respect to the common shares to be issued by us under the agreement.
In May 2018, we entered
into a development, commercialization and exclusive distribution agreement with Seraph, a human biomedical device company. Under
the terms of this agreement, we have exclusive global veterinary industry rights to develop and market a novel pathogen detection
system in the form of a point-of-care diagnostic instrument. The agreement covers development and validation of our fecal/urine
pathogen detection assays. We are responsible for development and validation, and their associated costs. Seraph will supply us,
on an exclusive basis, with the hardware platform, associated software and the consumables to be developed under the agreement,
pursuant to a rolling forecast, at prices specified in the agreement. We will be responsible for the marketing and sale of the
hardware platform, associated software and the consumables. The agreement, which is exclusive to the field of global veterinary
diagnostic applications, has a term of seven years (subject to adjustment in certain circumstances) and automatically renews for
additional one-year terms thereafter.
We paid Seraph up-front
fees of $500,000 and issued to Seraph unregistered common shares having a value of $1,238,513, consisting of an aggregate of 641,717
common shares at an ascribed price of $1.9479 per share. Seraph is entitled to additional payments for development costs. Seraph
will be entitled to receive up to an additional $7,000,000, payable 50 percent in cash and 50 percent in additional unregistered
common shares, upon the achievement of a series of staged, specified milestones, including completion of laboratory studies and
field studies, production and commercial shipment of products. Seraph is entitled to certain registration rights with respect to
the common shares to be issued by us under the agreement. In addition, we have agreed to pay Seraph license fees based on a percentage
of gross profit from commercial sales of the product.
In January 2017, we
entered into a collaborative research agreement with Celsee, a developer of diagnostics for the detection and quantification of
cells and other markers. Subsequent to this agreement, in December 2017, we entered into a license and supply agreement with Celsee
for exclusive global rights to develop and market Celsee’s liquid biopsy platform. The agreement with Celsee covers the development
and commercialization of liquid biopsy assays and related consumables for the detection of cancer in companion animals.
We paid Celsee up-front
fees of $500,000 and issued to Celsee unregistered common shares having a value of $230,131, consisting of an aggregate of 112,314
common shares at an ascribed price of $2.2259 per share. We issued Celsee an additional 657,894 unregistered common shares having
a value of $1 million at an ascribed price of $1.52. upon the achievement of specified milestones-namely, completion of product
development and upon successful completion of manufacturing milestones. All milestone payments to Celsee were satisfied as of December
31, 2019.
In January 2020, we
amended and restated the Celsee agreement to acknowledge the completion of the initial development work and to provide for definitive
supply and pricing terms for the liquid biopsy instrument and related consumables.
Under the terms of
the restated agreement, we continue to have veterinary oncology care exclusive global rights to develop and market Celsee’s
liquid biopsy platform for use by veterinarians as a cancer diagnostic.
Celsee will supply
us on an exclusive basis with the assays and the consumables for the products to be developed under the agreement pursuant to a
rolling forecast to be provided by us at prices specified in the agreement. We will be responsible for the marketing and sale of
the assays and the related consumables. The agreement, which is exclusive in the field of veterinary cancer diagnostic applications,
has a term of five years (subject to termination in certain circumstances) and automatically renews for additional two-year terms
thereafter (subject to either party determining not to renew).
Research and Development
We engage in development
work on our diagnostic platforms in conjunction with our strategic partners. We also engage in research and development activities
relating to our drug product candidates. We use various contract research organizations, or CROs, to assist in performing our research
and development activities.
In connection with
these activities, we have incurred and will continue to incur significant research and development expenses. Our research and development
expenses were $10,345,291 for the year ended December 31, 2019 and $10,317,153 for the year ended December 31, 2018.
Sales and Marketing
We intend to commercialize
our diagnostic products with a direct sales force and industry partners. We intend to market these products directly to veterinarians
whom we believe are self-motivated to utilize advanced diagnostics in order to improve the health of companion animals, while also
generating additional revenue.
We believe our strategy
of marketing directly to veterinarians is consistent with the current practice of veterinarians who perform some of their own diagnostic
tests and send other diagnostic samples to reference labs for analysis. We also intend to selectively utilize distributors,
which we believe will enable us to expand our commercial reach to a majority of all veterinarians in our chosen markets. We believe
that we can compete effectively with a combination of our own direct sales force and complementary distributors.
To support our marketing
efforts, we introduced a unique “Voice of the Vet” program in the fourth quarter of 2016 to gather insights and better
understand the needs of veterinarians and their practices, and to gauge interest for potential future product offerings, while
building brand awareness as a valued veterinary partner. Our Voice of the Vet program allows veterinarians, practice managers and
veterinary technicians to participate in conversations where they can share ideas and experiences with each other, as well as with
us through an interactive platform.
During 2019 and 2020,
we have increased our investment in building brand and product awareness as a valued veterinary partner with clinical practitioners.
Additionally, we are
continuing to conduct comprehensive market research across the United States with private, corporate and institutional clinics
along with key opinion leaders and academia to obtain feedback on our product development efforts and to build relationships with
key market influencers.
Manufacturing
We have no internal manufacturing capabilities
for our proposed and future products.
Under our license and
supply agreements, Qorvo, Seraph and Celsee are responsible for the manufacture and supply of the equipment and consumables to
us. These strategic partners have primary responsibility for assuring that all products will be manufactured in accordance with
applicable laws and meet all agreed upon specifications.
Intellectual Property
We intend to rely primarily
upon a combination of in-licensing exclusive rights, regulatory exclusivity, proprietary know-how, and confidentiality agreements
to protect our diagnostic assays, product formulations, processes, methods and other technologies and to preserve any trade secrets
and operate without infringing on the proprietary rights of other parties, both in the United States and in other countries. We
currently do not own any issued patents, although we intend to apply for patent protection where feasible
Our diagnostic technologies
are dependent on intellectual property developed by our strategic partners and licensed to us. We do not own the intellectual property
rights that underlie these technology licenses. Our rights to use the technology we license are subject to the negotiation of,
continuation of and compliance with the terms of our licenses. However, we have filed one provisional patent, four U.S. patent
applications and four Patent Cooperation Treaty (PCT) applications for U.S. and international protection of our diagnostic tests.
Three of these applications cover tests developed for our liquid biopsy platforms, and the remainder cover pathogen detection.
We depend upon the
skills, knowledge and experience of our management personnel, as well as that of our other employees, advisors, consultants and
contractors, none of which are patentable. To help protect our know-how, and any inventions for which patents may be difficult
to obtain or enforce, we require all of our employees, consultants, advisors and other contractors to enter into customary confidentiality
and inventions agreements that prohibit the disclosure of confidential information and, where applicable, require disclosure and
assignment to us of the ideas, developments, discoveries and inventions important to our business.
Competition
Our potential competitors
include large human pharmaceutical and medical diagnostics companies, small businesses focused on animal health, and reference
laboratory services provided by academic institutions and in-clinic product providers. These competitors include Idexx Laboratories,
Inc., Antech Diagnostics, a unit of VCA Inc., Abaxis, Inc., a wholly-owned subsidiary of Zoetis Inc., Heska Corporation and Zoetis
Inc.
Many of our competitors
and potential competitors have substantially more financial, technical, and human resources than we do. Many also have far more
experience in the development, manufacture, regulation and worldwide commercialization of animal diagnostics and medical devices.
We also expect to compete with academic institutions, governmental agencies and private organizations that are conducting research
in the fields of animal diagnostics and animal medical devices. If such competing products are successfully commercialized before
our products, or if our intellectual property protection fails to provide us with exclusive marketing rights for some of our products,
we may be unable to effectively compete in the markets in which we participate.
Government Regulation
Diagnostic
Product Candidates
Our diagnostic product
candidates may be subject to regulatory review by the USDA-CVB and/or post-marketing oversight by the USDA-CVB or FDA-CVM. Generally
speaking, full diagnostic kits aimed at the detection of an infectious disease in animals, including the materials required for
testing along with instructions for use and interpretation of results, used at the point-of-care, including in-office diagnostic
tests, may be subject to pre-market regulatory review and approval by the USDA-CVB. The USDA-CVB’s review process for diagnostics
is subject to some variability based on the type of diagnostic kit being reviewed, however, the USDA-CVB will generally review
the results of specific tests that are required to be conducted in accordance with the USDA-CVB’s testing criteria. These
include diagnostic sensitivity/specificity studies, conducted using a large number of samples of U.S. origin, reproducibility/repeatability/suitability
studies used to evaluate test kits under field conditions in participating laboratories and ruggedness studies in which manufacturers
measure the ruggedness or robustness of the diagnostic test kits based on the capacity of the assay to remain unaffected by small
variations in or deviations from the instructions for use (for example, not allowing the samples to reach the designated temperature)
and stability studies. Diagnostic products and testing kits that do not claim to detect or diagnose an infectious disease and that
are not designed for use at the point-of-care are generally subject only to post-marketing oversight by the FDA-CVM or the USDA-CVB.
While the sale of these products does not require premarket approval by the FDA-CVM and does not subject us to the FDA-CVM’s
cGMP requirements, these products must not be adulterated, mislabeled or misbranded under the Federal Food, Drug and Cosmetic Act,
or the FDC Act, and are subject to post-marketing review.
Other Regulatory
Considerations
Regulatory rules relating
to human food safety, food additives, or drug residues in food will not apply to our product candidates because our product candidates
are not intended for use in food animals or food production animals.
Advertising and promotion
of animal health products is controlled by regulations in the United States. These rules generally restrict advertising and promotion
to those claims and uses that have been reviewed and authorized by the FDA-CVM.
We will be required
to conduct post-approval monitoring of any approved product and to submit reports of product quality defects, adverse events or
unexpected results, and be subject to regulatory inspection from time to time. Safety, quality, or efficacy concerns can lead to
product recalls, withdrawals or suspended or declining sales, as well as product liability and other claims.
Employees
As of October 1, 2020, we had 17 employees.
Of our employees, three are engaged in research and development activities, seven are engaged in business development and marketing
activities, and seven are engaged in corporate and administrative activities. None of our employees are represented by labor unions
or covered by collective bargaining agreements.
Properties
Our corporate headquarters
and research and development laboratory are located in Ann Arbor, Michigan where we lease and occupy approximately 16,226 feet
pursuant to a lease that expires January 31, 2025. In February 2020 we entered into an amended lease agreement whereby our original
lease for approximately 26,540 square feet of space was bought out and a new lease was issued for 16, 226 square feet of office
space
Legal Proceedings
On November 1,
2019, Heska Corporation (“Heska”) filed a complaint for damages and injunctive relief (the
“Complaint”) in the United States District Court for the Middle District of North Carolina, Case
1:19-cv-01108-LCB-JLW, against Qorvo US, Inc. (“Qorvo US”), Qorvo Biotechnologies, LLC (“Qorvo
Biotech” and, together with Qorvo US, “Qorvo”) and us (collectively with Qorvo, the
“Defendants”) which was amended on November 22, 2019. The amended Complaint alleges, among other things, that the
Defendants improperly obtained Heska’s trade secrets and confidential information and/or conspired to use improper
means to misappropriate Heska’s trade secrets related to an instrument and related consumable products for performing
immunoassay analysis of biomarkers and other substances. The amended Complaint seeks compensatory and exemplary damages, as
well as preliminary and permanent injunctive relief to prevent the Defendants from commercializing our
TRUFORMATM diagnostic instrument. On January 21, 2020, the Defendants filed a motion seeking dismissal of the
Complaint. On February 11, 2020, Heska filed its response to the Defendants’ motion to dismiss to which the Defendants
responded on February 25, 2020. Heska subsequently moved to strike a portion of the Defendants’ response. On September
30, 2020, the court denied the Defendants’ motion to dismiss and granted Heska’s motion to strike. On October 14,
2020 the Defendants filed their answer to the amended Complaint. We believe that the allegations in the amended Complaint
have no merit and will not have a material adverse effect on our business, results of operations or financial condition.
Under the terms of
the Development and Supply Agreement, dated November 26, 2018, by and between Qorvo Biotech and the Company (as amended, the “Qorvo
Agreement”), Qorvo Biotech agreed to indemnify us and certain related parties against claims alleging infringement or misappropriation
of third-party intellectual property rights, subject to certain limitations and exceptions. Qorvo Biotech has notified us that
Qorvo Biotech has assumed the defense of the amended Complaint and will indemnify us for losses arising from the amended Complaint
in accordance with the terms of the Qorvo Agreement. Qorvo Biotech has further advised us that it intends to mount a vigorous defense
to the claims in the amended Complaint, and that it believes the allegations contained in the amended Complaint are without merit.
Corporate Information
Zomedica Corp. (formerly,
Wise Oakwood Ventures Inc.) was originally incorporated as Wise Oakwood Ventures Inc. on January 7, 2013 under the Business
Corporations Act (Alberta). On October 28, 2013, we completed our initial public offering in Canada and became classified
as a Capital Pool Company, as defined under the rules of the TSX Venture Exchange, or TSX-V. On April 21, 2016, we changed our
name to Zomedica Pharmaceuticals Corp. and consolidated our common shares on a one-for-two and one-half (2½) basis. ZoMedica
Pharmaceuticals Inc., or ZoMedica Inc., was incorporated on May 14, 2015 under the Canada Business Corporations Act.
On April 21, 2016, we completed a qualifying transaction, or the Qualifying Transaction, under TSX-V Policy 2.4 - Capital
Pool Companies, consisting of a three-cornered amalgamation among our company, ZoMedica Inc. and our wholly-owned subsidiary.
Under the Qualifying Transaction, ZoMedica Inc. and our subsidiary were amalgamated to form Zomedica Pharmaceuticals Ltd., or Zomedica
Ltd. As consideration for the amalgamation, shareholders of ZoMedica Inc. became the owners of 97.6% (non-diluted) of our common
shares, and ZoMedica Ltd. became our wholly-owned subsidiary. Subsequent to the Qualifying Transaction, Zomedica Ltd. was vertically
amalgamated into our company. We entered into the Qualifying Transaction in order to accomplish the following:
|
|
·
|
Enable our shareholders to own shares in a company that was publicly traded on the TSX-V;
|
|
|
·
|
Expand our shareholder base to include the public shareholders of Wise Oakwood; and
|
|
|
·
|
Obtain access to the cash resources raised by Wise Oakwood in its initial public offering.
|
On November 10, 2017,
our shares were approved for listing on the NYSE American under the symbol “ZOM”. On November 20, 2017 the U.S. Securities
and Exchange Commission declared our registration statement on Form S-1 effective. Our common shares commenced trading on the NYSE
American on November 21, 2017.
On February 10, 2020,
we effected the voluntary withdrawal of our common shares from listing on the TSX-V.
On October 2, 2020,
we changed our name to Zomedica Corp.
Our principal executive
offices are located at 100 Phoenix Drive, Suite 180, Ann Arbor, MI 48108, and our telephone number is (734) 369-2555. Our website
address is www.zomedica.com. The information contained in, or accessible through, our website is not part of the registration statement
of which this prospectus forms a part.
Market
PRICE OF OUR Common SHARES and Related SHAREholder Matters
Our common shares are
listed on the NYSE American under the symbol “ZOM.” On October 21, 2020, the last reported sale price of our common
shares on the NYSE American was $0.0755 per share.
Holders
As of the Record Date, we had approximately
200 record holders of our common shares. The number of record holders is based on the actual number of holders registered on the
books of our transfer agent and does not reflect holders of shares in “street name” or persons, partnerships, associations,
corporations or other entities identified in security position listings maintained by depository trust companies.
Management’s
Discussion and Analysis of
Financial Condition and Results of Operation
Management’s Discussion and Analysis
of Financial Condition and Results of Operations is intended to help the reader understand the results of operations and financial
condition of our company. The Management’s Discussion and Analysis of Financial Condition and Results of Operations should
be read in conjunction with our audited consolidated financial statements and notes thereto for the years ended December 31, 2019
and December 31, 2018 and our unaudited consolidated financial statements and notes thereto for the three- and six-month periods
ended June 30, 2020 and June 30, 2019 included elsewhere herein.
Overview
We are a veterinary health company creating
products for companion animals by focusing on the unmet needs of clinical veterinarians. We expect that our product portfolio will
include innovative diagnostics and medical devices that emphasize patient health and practice health. With a team that includes
clinical veterinary professionals, our goal is to provide veterinarians the opportunity to increase productivity and grow revenue
while better serving the animals in their care.
Our strategic focus is on the final development
and commercialization of our TRUFORMA™ diagnostic biosensor platform and the first five assays for the detection of adrenal
and thyroid disorders in cats and dogs. The TRUFORMA™ platform uses Bulk Acoustic Wave (BAW) technology to provide a non-optical
and fluorescence free detection system for use at the point-of-care. We believe that BAW technology will enable precise and repeatable
test results at the point-of-care during a typical veterinary appointment. We believe that the TRUFORMA™ diagnostic platform
does not require pre-market regulatory approval for use with companion animals in the United States.
Following the commercial launch of TRUFORMA™,
we expect to continue the development of another point-of-care diagnostic platform, which is based on miniaturized laser-based
Raman spectroscopy technology and is designed to detect pathogens in companion animals. We believe this platform will enable the
identification of biological and biochemical signatures in complex biological samples and has the potential to achieve reference
lab sensitivity/specificity to screen for a wide range of pathogens in companion animal feces, urine, respiratory, and dermatological
samples in minutes without the need for extensive sample prep or the use of reagents. The diagnostic platform requires a small
fecal sample preparation. Additionally, the platform has automated analysis and does not require specialized staff training. Assuming
development work is successfully completed we expect the commercial launch of our fecal test to occur by 2022 and urine tests by
2023. We believe that this diagnostic platform does not require pre-market regulatory approval for use with companion animals in
the United States.
We have performed initial development work on
a circulating tumor cell (CTC) “liquid biopsy” platform for use in a reference lab setting as a canine cancer diagnostic.
This platform is intended for use to detect canine cancers faster, more affordably and less invasively compared to existing methods,
which can be expensive and cost-prohibitive for pet owners. We have worked on the development of an assay for use with this platform
that targets hard-to-diagnose canine cancers, such as hemangiosarcoma and osteosarcoma.
Consistent with our focus on the development
of point-of-care diagnostic platforms, we intend to seek one or more partners for the further development and commercialization
of the liquid biopsy platform.
We are a development-stage company with no products
approved for marketing and sale, and we have not generated any revenue. We have incurred significant net losses since our inception.
We incurred net losses of $5,307,990 and $7,758,607 for the three and six months ended June 30, 2020 and net losses of $2,404,427
and $14,081,337 for the three and six months ended June 30, 2019$19,784,054 and $16,647,687 for the years ended December 31, 2019
and December 31, 2018, respectively. These losses have resulted principally from costs incurred in connection with investigating
and developing our product candidates, research and development activities and general and administrative costs associated with
our operations. As of June 30, 2020, we had an accumulated deficit of $59,816,448 and cash and cash equivalents of $29,103,049.
For the foreseeable future, we expect to continue
to incur losses, which will increase significantly from historical levels as we expand our product development activities, commercialize
products, seek regulatory approvals for our planned and future products to the extent required, and expand our sales and marketing
activities.
For further information on the regulatory, business
and product pipeline, please see “Business” above. For further information on the risk factors, please see “Risk
Factors” above.
Revenue
We do not have any products approved for sale,
have not generated any revenue from product sales since our inception and do not expect to generate any revenue from the sale of
products in the near future. If our development efforts result in clinical success or collaboration agreements with third parties
for any of our product candidates, we may generate revenue from those product candidates.
Operating Expenses
The majority of our operating expenses to date
have been for the general and administrative activities related to general business activities, capital market activities and stock-based
compensation, and research and development activities related to the development of our product candidates.
Research and Development Expense
All costs of research and development are expensed
in the period in which they are incurred. Research and development costs primarily consist of salaries and related expenses for
personnel, fees paid to consultants, outside service providers, professional services, travel costs and materials used in clinical
trials and research and development.
General and Administrative Expense
General and administrative expense consists
primarily of personnel costs, including salaries, related benefits and stock-based compensation for employees, consultants and
directors. General and administrative expenses also include rent and other facilities costs and professional and consulting fees
for legal, accounting, tax services and other general business services.
Professional Fees
Professional fees include attorney’s fees,
accounting fees and consulting fees incurred in connection with product investigation and analysis, regulatory analysis, government
relations, audit, securities offerings, investor relations, and general corporate and intellectual property advice.
Income Taxes
As of December 31, 2019, we had net operating
loss carryforwards for federal and state income tax purposes of $16,140,344 and non-capital loss carryforwards for Canadian income
tax purposes of approximately $20,366,610, which will begin to expire in fiscal year 2035. We have evaluated the factors bearing
upon the realizability of our deferred tax assets, which are comprised principally of net operating loss carryforwards and non-capital
loss carryforwards. We concluded that, due to the uncertainty of realizing any tax benefits as of December 31, 2019, a valuation
allowance was necessary to fully offset our deferred tax assets.
Critical Accounting Policies and Significant Judgments
and Estimates
Our management’s discussion and analysis
of financial condition and results of operations is based on our consolidated financial statements, which have been prepared in
accordance with accounting principles generally accepted in the United States, or U.S. GAAP. The preparation of our consolidated
financial statements and related disclosures requires us to make estimates and assumptions that affect the reported amounts of
assets and liabilities, and revenue, costs and expenses and related disclosures during the reporting periods. On an ongoing basis,
we evaluate our estimates and judgments, including those described below. We base our estimates on historical experience and on
various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments
about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ
from these estimates under different assumptions or conditions.
While our significant accounting policies are
more fully described in Note 3 of the notes to our consolidated financial statements appearing elsewhere in this report, we believe
that the estimates and assumptions involved in the following accounting policies may have the greatest potential impact on our
financial statements.
JOBS Act
The Jumpstart Our Business Startups Act, or
the JOBS Act, contains provisions that, among other things, reduce certain reporting requirements for an “emerging growth
company.” We have irrevocably elected not to avail ourselves of the JOBS Act provision that an emerging growth company may
delay adopting new or revised accounting standards until such times as those standards apply to private companies.
In addition, we are in the process of evaluating
the benefits of relying on the other exemptions and reduced reporting requirements provided by the JOBS Act. Subject to certain
conditions set forth in the JOBS Act, if as an “emerging growth company” we choose to rely on such exemptions, we may
not be required to, among other things, (i) provide an auditor’s attestation report on our system of internal controls over
financial reporting pursuant to Section 404, and (ii) comply with any requirement that may be adopted by the Public Company Accounting
Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information
about the audit and the financial statements (auditor discussion and analysis). These exemptions will apply until December 31,
2022 or until we no longer meet the requirements of being an “emerging growth company,” whichever is earlier.
Use of
Estimates
The preparation of consolidated financial statements
in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and
liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts
of expenses during the year. Actual results could differ from those estimates.
Areas where significant judgment is involved
in making estimates are, the determination of fair value of stock-based compensation, the useful lives of property and equipment,
allocation of proceeds from financings to shares and warrants, fair value of placement agent warrants and forecasting future cash
flows for assessing the going concern assumption.
Research
and Development Costs
Research and development expenses comprise costs
incurred in performing research and development activities, including salaries and benefits, safety and efficacy studies and contract
manufacturing costs, contract research costs, patent procurement costs, materials and supplies, and occupancy costs. Research and
development activities include internal and external activities associated with research and development studies of current product
candidates and advancing product candidates towards a goal of obtaining regulatory approval to manufacture and market the product
candidate.
Research and development costs related to continued
research and development programs are expensed as incurred in accordance with ASC topic 730.
Translation
of Foreign Currencies
The functional currency, as determined by management,
is U.S. dollars, which is also our reporting currency. Transactions denominated in currencies other than U.S. dollars and the monetary
value of assets and liabilities are remeasured at the period-end exchange rates. Revenue and expenses are measured at rates of
exchange prevailing on the transaction dates. All of the exchange gains or losses resulting from these other transactions are recognized
in the consolidated statements of operations and comprehensive loss.
Stock-Based
Compensation
We measure the cost of equity-settled transactions
by reference to the fair value of the equity instruments at the date at which they are granted if the fair value of the goods or
services received by us cannot be reliably estimated.
We calculate stock-based compensation using
the fair value method, under which the fair value of the options at the grant date is calculated using the Black-Scholes Option
Pricing Model, and subsequently expensed over the vesting period of the option using the graded vesting method. The provisions
of our stock-based compensation plans do not require us to settle any options by transferring cash or other assets, and therefore
we classify the awards as equity. Stock-based compensation expense recognized during the period is based on the value of stock-based
payment awards that ultimately are expected to vest. We estimate forfeitures at the time of grant and revise these estimates, if
necessary, in subsequent periods if actual forfeitures differ from those estimates. The expected term, which represents the period
of time that options granted are expected to be outstanding, is estimated based on an average of the term of the options. The risk-free
rate assumed in valuing the options is based on the Canadian treasury yield curve in effect at the time of grant for the expected
term of the option. The expected dividend yield percentage at the date of grant is nil as we are not expected to pay dividends
in the foreseeable future.
Loss Per
Share
Basic loss per share, or EPS, is computed by
dividing the loss attributable to common shareholders by the weighted average number of common shares outstanding. Diluted EPS
reflects the potential dilution that could occur from common shares issuable through the exercise or conversion of stock options,
restricted stock awards, warrants, and convertible securities. In certain circumstances, the conversion of options, warrants, and
convertible securities are excluded from diluted EPS if the effect of such inclusion would be anti-dilutive.
The dilutive effect of stock options is determined
using the treasury stock method. Stock options and warrants to purchase our common shares issued during the period were not included
in the computation of diluted EPS, as the effect would be anti-dilutive.
Comprehensive
Loss
We follow ASC topic 220. This statement establishes
standards for reporting and display of comprehensive (loss) income and its components. Comprehensive loss is net loss plus certain
items that are recorded directly to shareholders' equity. We currently have no other comprehensive loss items.
Results of Operations
Three and six months ended June 30, 2020 compared to three
and six months ended June 30, 2019
Our results of operations for the three and six months ended June
30, 2020 and June 30, 2020 are as follows:
|
|
|
Three Months Ended June 30,
|
|
|
Six Months Ended June 30,
|
|
|
|
|
2020
|
|
|
|
2019
|
|
|
Change
|
|
|
2020
|
|
|
|
2019
|
|
|
Change
|
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
%
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
%
|
|
|
Expenses
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
3,908,171
|
|
|
|
1,061,507
|
|
|
|
2,846,664
|
|
|
|
268
|
%
|
|
|
4,503,570
|
|
|
|
8,592,882
|
|
|
|
(4,089,312
|
)
|
|
|
-48
|
%
|
|
General and administrative
|
|
|
988,734
|
|
|
|
901,319
|
|
|
|
87,415
|
|
|
|
10
|
%
|
|
|
2,272,261
|
|
|
|
4,113,677
|
|
|
|
(1,841,416
|
)
|
|
|
-45
|
%
|
|
Professional fees
|
|
|
282,791
|
|
|
|
231,647
|
|
|
|
51,144
|
|
|
|
22
|
%
|
|
|
573,472
|
|
|
|
989,946
|
|
|
|
(416,474
|
)
|
|
|
-42
|
%
|
|
Amortization - right-of-use asset
|
|
|
–
|
|
|
|
127,345
|
|
|
|
(127,345
|
)
|
|
|
N/A
|
|
|
|
42,448
|
|
|
|
254,690
|
|
|
|
(212,242
|
)
|
|
|
N/A
|
|
|
Amortization - intangible
|
|
|
44,990
|
|
|
|
270
|
|
|
|
44,720
|
|
|
|
16563
|
%
|
|
|
90,025
|
|
|
|
537
|
|
|
|
89,488
|
|
|
|
16664
|
%
|
|
Depreciation
|
|
|
77,859
|
|
|
|
68,925
|
|
|
|
8,934
|
|
|
|
13
|
%
|
|
|
154,275
|
|
|
|
130,979
|
|
|
|
23,296
|
|
|
|
18
|
%
|
|
Loss from operations
|
|
|
5,302,545
|
|
|
|
2,391,013
|
|
|
|
2,911,532
|
|
|
|
122
|
%
|
|
|
7,636,051
|
|
|
|
14,082,711
|
|
|
|
(6,446,660
|
)
|
|
|
-46
|
%
|
|
Interest income
|
|
|
(247
|
)
|
|
|
–
|
|
|
|
(247
|
)
|
|
|
NA
|
|
|
|
(328
|
)
|
|
|
–
|
|
|
|
(328
|
)
|
|
|
NA
|
|
|
Interest expense
|
|
|
–
|
|
|
|
12,164
|
|
|
|
(12,164
|
)
|
|
|
-100
|
%
|
|
|
732
|
|
|
|
18,338
|
|
|
|
(17,606
|
)
|
|
|
-96
|
%
|
|
Loss on property and equipment
|
|
|
–
|
|
|
|
1,308
|
|
|
|
(1,308
|
)
|
|
|
-100
|
%
|
|
|
69,834
|
|
|
|
1,308
|
|
|
|
68,526
|
|
|
|
N/A
|
|
|
Loss on right-of-use asset
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
N/A
|
|
|
|
59,097
|
|
|
|
–
|
|
|
|
59,097
|
|
|
|
N/A
|
|
|
Gain on settlement of liabilities
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
N/A
|
|
|
|
–
|
|
|
|
(19,737
|
)
|
|
|
19,737
|
|
|
|
N/A
|
|
|
Other income
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
N/A
|
|
|
|
(5,500
|
)
|
|
|
–
|
|
|
|
(5,500
|
)
|
|
|
N/A
|
|
|
Foreign exchange gain
|
|
|
5,692
|
|
|
|
(58
|
)
|
|
|
5,750
|
|
|
|
-9914
|
%
|
|
|
(1,279
|
)
|
|
|
(1,283
|
)
|
|
|
4
|
|
|
|
0
|
%
|
|
Loss before income taxes
|
|
|
5,307,990
|
|
|
|
2,404,427
|
|
|
|
2,903,563
|
|
|
|
121
|
%
|
|
|
7,758,607
|
|
|
|
14,081,337
|
|
|
|
(6,322,730
|
)
|
|
|
-45
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income tax expense
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
N/A
|
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
N/A
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss and comprehensive loss
|
|
|
5,307,990
|
|
|
|
2,404,427
|
|
|
|
2,903,563
|
|
|
|
121
|
%
|
|
|
7,758,607
|
|
|
|
14,081,337
|
|
|
|
(6,322,730
|
)
|
|
|
-45
|
%
|
Revenue
We did not generate any revenue during the three
and six months ended June 30, 2020 and June 30, 2019.
Research and Development
Research and development expense for the three
months ended June 30, 2020 was $3,908,171 compared to $1,061,507 for the three months ended June 30, 2019, an increase of $ 2,846,664
or 268%. The increase primarily resulted from a milestone expense of $3,000,000 recognized in accordance with our development and
supply agreement with Qorvo Biotechnologies, LLC. (“Qorvo”) and increased regulatory fees. This increase partially
was offset by a reduction in salaries and bonuses of $129,234 and supplies of $39,730.
Research and development expense for the six
months ended June 30, 2020 was $4,503,570 compared to $8,592,882 for the six months ended June 30, 2019, a reduction of $4,089,312
or 48%. The decrease primarily was due to a reduction of $2,000,000 in milestone expenses relating to TRUFORMA™ under our
development and supply agreement with Qorvo, a reduction of $736,841 in milestone expenses relating to our development of ZM-017
under our license and supply agreement with Celsee, Inc., a reduction of $933,754 in consulting expenses, a reduction of $188,157
in salaries, bonus and benefits, and a reduction of $73,882 in supplies.
General and Administrative
General and administrative expense for the three
months ended June 30, 2020 was $988,734, compared to $901,319 for the three months ended June 30, 2019, an increase of $87,415
or 10%. The increase primarily was due to an increase in salaries, bonuses and benefits of $52,202, inclusive of $135,844 in cost
associated with options granted to employees in the period. After adjusting for the stock option compensation expense, salaries
decreased by $53,075. This decrease was due to no bonuses being earned in the current period versus the prior period, offset by
increases in salaries. Other increases include rent expense of $75,672 relating to the reclassification of right-of-use asset amortization,
$26,999 in insurance expense, and $14,192 in office expense. These increases were offset by reductions in travel and accommodation
expense of $62,080, marketing and investor relations expense of $11,078, office supplies of $4,314 and $4,178 for regulatory fees.
General and administrative expense for the six
months ended June 30, 2020 was $2,272,261, compared to $4,113,677 for the six months ended June 30, 2019, a decrease of $1,841,416
or 45%. The decrease primarily was due to a reduction in salaries, bonuses and benefits of $2,142,824 as a result of a reduction
in stock option compensation expense compared to the prior period. After adjusting for the stock option compensation expense, salaries
decreased $45,105. This decrease was due to no bonuses being earned in the current period versus the prior period, offset in part
by an increase in salary expense. This decrease was partially offset by an increase in office expense associated with the expensing
of furniture in the office space completed in the first quarter.
Professional Fees
Professional fees for the three months ended
June 30, 2020 were $282,791 compared to $231,647 for the three months ended June 30, 2019, an increase of $51,144 or 22%. The increase
was due to additional legal fees associated with the COVID-19 pandemic disclosures, our offering activity and the appointment of
our new Interim Chief Executive Officer.
Professional fees for the six months ended June
30, 2020 were $573,472 compared to $989,946 for the six months ended June 30, 2019, a decrease of $416,474 or 42%. The decrease
was primarily due to expenses incurred in the prior period related to the preparation of our S-3 resale registration statement
and our S-8 registration statement.
Net Loss
Our net loss for the three months ended June
30, 2020 was $5,307,990 or $0.02 per share, compared with a net loss of $2,404,427 or $0.02 per share, for the three months ended
June 30, 2019, an increase of $2,903,563 or 121%. The net loss in each period was attributed to the matters described above.
Our net loss for the six months ended June 30,
2020 was $7,758,607 or $0.05 per share, compared with a net loss of $14,081,337 or $0.13 per share for the six months ended June
30, 2019, a decrease of $6,322,730 or 45%. The net loss in each period was attributed to the matters described above. We expect
to continue to record net losses in future periods until such time, if ever, as we have sufficient revenue from our product candidates
to offset our operating expenses.
Cash Flows
Three and six months ended June 30, 2020 compared to three
and six months ended June 30, 2019
The following table shows a summary of our cash
flows for the periods set forth below:
|
|
|
Three months ended June 30,
|
|
Six months ended June 30,
|
|
|
|
|
2020
|
|
|
|
2019
|
|
|
Change
|
|
|
2020
|
|
|
|
2019
|
|
|
Change
|
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
%
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
%
|
|
|
Cash flows used in operating activities
|
|
|
(5,720,528
|
)
|
|
|
(8,436,011
|
)
|
|
|
2,715,483
|
|
|
|
-32
|
%
|
|
|
(7,893,653
|
)
|
|
|
(11,017,287
|
)
|
|
|
3,123,634
|
|
|
|
-28
|
%
|
|
Cash flows from financing activities
|
|
|
33,326,825
|
|
|
|
11,966,905
|
|
|
|
21,359,920
|
|
|
|
178
|
%
|
|
|
35,478,603
|
|
|
|
14,973,733
|
|
|
|
20,504,870
|
|
|
|
137
|
%
|
|
Cash flows from (used) in investing activities
|
|
|
-
|
|
|
|
(5,477
|
)
|
|
|
5,477
|
|
|
|
-100
|
%
|
|
|
1,007,513
|
|
|
|
(74,563
|
)
|
|
|
1,082,076
|
|
|
|
-1451
|
%
|
|
Increase in cash
|
|
|
27,606,297
|
|
|
|
3,525,417
|
|
|
|
24,080,880
|
|
|
|
683
|
%
|
|
|
28,592,463
|
|
|
|
3,881,883
|
|
|
|
24,710,580
|
|
|
|
637
|
%
|
|
Cash and cash equivalents, beginning of period
|
|
|
1,496,752
|
|
|
|
2,296,731
|
|
|
|
(799,979
|
)
|
|
|
-35
|
%
|
|
|
510,586
|
|
|
|
1,940,265
|
|
|
|
(1,429,679
|
)
|
|
|
-74
|
%
|
|
Cash and cash equivalents, end of period
|
|
|
29,103,049
|
|
|
|
5,822,148
|
|
|
|
23,280,901
|
|
|
|
400
|
%
|
|
|
29,103,049
|
|
|
|
5,822,148
|
|
|
|
23,280,901
|
|
|
|
400
|
%
|
Operating Activities
Net cash used in operating activities for the
three months ended June 30, 2020 was $5,720,528, compared to $8,436,011 for the three months ended June 30, 2019, a decrease of
$2,715,483 or 32%. The decrease resulted primarily from a lower net loss in the second quarter of 2020 compared to the second quarter
of 2019, offset in part by a reduction of current liabilities.
Net cash used in operating activities for the
six months ended June 30, 2020 was $7,893,653, compared to $11,017,287 for the six months ended June 30, 2019, a decrease of $3,123,634
or 28%. The decrease resulted primarily from a lower net loss in the first half of 2020 compared to the first half of 2019, offset
in part by a reduction in current liabilities and an increase in current assets.
Financing Activities
Net cash from financing activities for the three
months ended June 30, 2020 was $33,326,825, compared to $11,966,905 for the three months ended June 30, 2019, an increase of $21,359,920
or 178%. The increase resulted primarily from the sale of our equity securities for total gross proceeds of approximately $23,998,783
as well as $9,826,359 cash received from warrant exercises, $1,465,500 cash received from pending warrant exercises, and cash received
of $527,360 from the SBA’s Paycheck Protection Program, offset in part by increased stock issuance costs of $2,491,177.
Net cash from financing activities for the six
months ended June 30, 2020 was $35,478,603, compared to $14,973,733 for the six months ended June 30, 2019 an increase of $20,504,870
or 137%. The increase resulted primarily from the sale of our equity securities for total gross proceeds of approximately $26,498,783,
cash received of $9,826,359 from warrant exercises, $1,465,500 cash received from pending warrant exercises and cash received of
$527,360 from the SBA’s Paycheck Protection Program offset in part by stock issuance costs of $2,839,399.
Investing Activities
Net cash used in investing activities for the
three months ended June 30, 2020 was nil, compared to $5,477 for the three months ended June 30, 2019, a decrease of $5,477 or
100%.
Net cash from investing activities for the six
months ended June 30, 2020 was $1,007,513, compared to net cash used of 74,563 for the six months ended June 30, 2019, an increase
of $1,082,076 or 1,451%. The increase in net cash from investing activities related to the cancellation and buyout of our office
lease.
Year ended December 31, 2019 compared
to year ended December 31, 2018
Our results of operations for the year ended December 31, 2019 and December
31, 2018 are as follows:
|
|
|
|
Year ended
|
|
Year Ended
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2019
|
|
|
|
December 31, 2018
|
|
|
|
Change
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Expenses
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
|
|
Research and development
|
|
|
10,345,291
|
|
|
|
10,317,153
|
|
|
|
28,138
|
|
|
0
|
%
|
|
General and administrative
|
|
|
7,114,777
|
|
|
|
4,521,349
|
|
|
|
2,593,428
|
|
|
57
|
%
|
|
Professional fees
|
|
|
1,536,646
|
|
|
|
1,534,977
|
|
|
|
1,669
|
|
|
0
|
%
|
|
Amortization - right-of-use asset
|
|
|
509,381
|
|
|
|
–
|
|
|
|
509,381
|
|
|
N/A
|
|
|
Amortization - intangible
|
|
|
1,082
|
|
|
|
2,083
|
|
|
|
(1,001
|
)
|
|
-48
|
%
|
|
Depreciation
|
|
|
277,150
|
|
|
|
203,684
|
|
|
|
73,466
|
|
|
36
|
%
|
|
Loss from operations
|
|
|
19,784,327
|
|
|
|
16,579,246
|
|
|
|
3,204,081
|
|
|
19
|
%
|
|
Interest Expense
|
|
|
18,338
|
|
|
|
–
|
|
|
|
18,338
|
|
|
N/A
|
|
|
Gain on settlement of liabilities
|
|
|
(19,737
|
)
|
|
|
–
|
|
|
|
(19,737
|
)
|
|
N/A
|
|
|
Loss on sale of fixed assets
|
|
|
1,308
|
|
|
|
69,382
|
|
|
|
(68,074
|
)
|
|
-98
|
%
|
|
Foreign exchange gain
|
|
|
(182
|
)
|
|
|
(941
|
|
|
|
759
|
|
|
-81
|
%
|
|
Loss before income taxes
|
|
|
19,784,054
|
|
|
|
16,647,687
|
|
|
|
3,135,367
|
|
|
19
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income tax expense
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
N/A
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss and comprehensive loss
|
|
|
19,784,054
|
|
|
|
16,647,687
|
|
|
|
3,135,367
|
|
|
19
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Revenue
We did not generate any revenue during the years
ended December 31, 2019 and December 31, 2018.
Research and Development
Research and development expense for the year
ended December 31, 2019 was $10,345,291 compared to $10,317,153 for the year ended December 31, 2018, an increase of $28,138 or
less than 1%. The increase was primarily due to an increase in third-party expenses relating to the development of our product
candidate and the addition of full-time employees. Contracted expenditures increased $1,098,987 and salaries increased $96,934.
These costs were partially offset by a reduction in licensing fees. In 2019, we paid licensing fees of $5,000,000 related to the
development of our TRUFORMATM platform, and licensing fees of $736,841 related to the development of our liquid
biopsy platform. In 2018, we paid licensing fees of $5,413,158 related to the development of our TRUFORMATM platform
and $1,738,513 related to the development of our pathogen detection platform.
General and Administrative
General and administrative expense for the year
ended December 31, 2019 was $7,114,777, compared to $4,521,349 for the year ended December 31, 2018, an increase of $2,593,428
or 57%. The increase was due to an increase in salaries, bonus and benefits of $2,853,769, which included share-based compensation
expense of $2,539,092 resulting from the granting of options to purchase an aggregate of 7,495,000 common shares, all of which
vested upon the dates of grant. Other increases in salaries, bonus and benefits were due to increases in sales, marketing and other
administrative salaries and benefits. Marketing and investor relations expense increased by $245,997, and travel and accommodation
expense increased by $169,383, which were partially offset by a reduction in regulatory costs of $329,666 and a decrease in rent
of $263,279 which was reclassified to amortization of right-of-use asset.
Professional Fees
Professional fees for the year ended December
31, 2019 were $1,536,646 compared to $1,534,977 for the year ended December 31, 2018, an increase of $1,669 or less than 1%. The
increase was primarily due to increased expenses resulting from an increase in our SEC reporting and registration activities.
Net Loss
Our net loss for the year ended December 31,
2019 was $19,784,054, or $0.19 per share, compared with a net loss of $16,647,687, or $0.18 per share, for the year ended December
31, 2018, an increase of $3,135,167 or 19%. The net loss in each period was attributed to the matters described above. We expect
to continue to record net losses in future periods until such time as we have sufficient revenue from our product candidates to
offset our operating expenses.
Cash Flows
Year ended December 31, 2019 compared to year ended December
31, 2018
The following table shows a summary of our cash flows for
the periods set forth below:
|
|
|
|
Year ended
|
|
|
|
Year ended
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2019
|
|
|
|
December 31, 2018
|
|
|
|
Change
|
|
|
|
|
|
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
%
|
|
|
Cash flows used in operating activities
|
|
|
(15,634,064
|
)
|
|
|
(11,147,528
|
)
|
|
|
(4,486,536
|
)
|
|
|
40
|
%
|
|
Cash flows provided by financing activities
|
|
|
14,891,317
|
|
|
|
10,258,643
|
|
|
|
4,632,674
|
|
|
|
45
|
%
|
|
Cash flows used in investing activities
|
|
|
(686,932
|
)
|
|
|
(618,997
|
)
|
|
|
(67,935
|
)
|
|
|
11
|
%
|
|
Decrease in cash
|
|
|
(1,429,679
|
)
|
|
|
(1,507,882
|
)
|
|
|
78,203
|
|
|
|
-5
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents, beginning of year
|
|
|
1,940,265
|
|
|
|
3,448,147
|
|
|
|
(1,507,882
|
)
|
|
|
-44
|
%
|
|
Cash and cash equivalents, end of year
|
|
|
510,586
|
|
|
|
1,940,265
|
|
|
|
(1,429,679
|
)
|
|
|
-74
|
%
|
Operating Activities
Net cash used in operating activities for the
year ended December 31, 2019 was $15,634,064, compared to $11,147,528 for the year ended December 31, 2018, an increase of $4,486,536
or 40%. The increase in net cash used in operating activities resulted primarily from the increase in our net loss, as well as
a decrease in accounts payable and accrued liabilities of $288,994, partially offset by an increase in non-cash expenses including
stock-based compensation of $2,539,092, amortization of right-of-use asset of $509,381, utilization of deposits and prepaid expenses
of $332,826 and depreciation of $277,150.
Financing Activities
Net cash provided by financing activities for
the year ended December 31, 2019 was $14,891,317, compared to net cash provided by financing activities of $10,258,643 for the
year ended December 31, 2018, an increase of $4,632,674 or 45%. The increase in cash from financing activities resulted from $12,000,000
in proceeds from the private sale of preferred shares, $3,000,000 in proceeds from our underwritten public offering of common stock,
net of financing costs, and $600,000 in proceeds from the exercise of stock options, partially offset by $4,002,496 from the private
sale of our common shares, proceeds of $2,034,307 from the exercise of stock options and common stock subscriptions of $4,280,000,
partially offset by stock issuance costs of $58 in 2018.
Investing Activities
Net cash used in investing activities for the
year ended December 31, 2019 was $686,932, compared to $618,997 for the year ended December 31, 2018, an increase of $67,935 or
11%. The increase in net cash used in investing activities resulted primarily from costs associated with the digital data platform,
the construction of marketing assets, and the capitalization of integration costs associated with the implementation of an ERP
system, partially offset by investments in additional leasehold improvements, equipment and furniture for additional office and
lab space in Ann Arbor during 2018.
Liquidity and Capital Resources
We have incurred losses and negative cash flows
from operations and have not generated any revenue since our inception in May 2015. As of June 30, 2020, we had an accumulated
deficit of $59,816,448. We have funded our working capital requirements primarily through the sale of our securities and the exercise
of stock options and warrants.
As of June 30, 2020, the Company had cash of
$29,103,049, prepaid expenses and deposits of $782,647, and accounts receivable of $182,496. Current assets amounted to $30,068,192,
with current liabilities of $1,963,965, resulting in working capital (defined as current assets minus current liabilities) of $28,104,227.
As of June 30, 2020, we had shareholders’
equity of $29,578,961.
In July 2020, we sold an aggregate of (i) 162,500,000
common shares, (ii) pre-funded warrants to purchase up to 25,000,000 common shares and (iii) Series D warrants to purchase an aggregate
of 187,500,000 common shares for aggregate gross proceeds of $30,000,000. The pre-funded warrants sold in the July offering have
been exercised in full.
Since June 30, 2020, we have received an aggregate
of $863,550 from the exercise of outstanding warrants.
As of August 10, 2020, we had cash and cash equivalents of approximately $55,000,000.
On October 17, 2017 we entered into a five-year
$5,000,000 unsecured working capital facility with Equidebt LLC, one of our shareholders (the “Equidebt Facility”).
Amounts borrowed under the Equidebt Facility bear interest at a rate of 14% per annum payable at maturity. All amounts borrowed
under the Equidebt Facility become due and payable on October 17, 2022. We can make two borrowings per month under the Equidebt
Facility, each of which must be for a minimum of $250,000. The Equidebt Facility is unsecured. As of June 30, 2020, no amounts
have been borrowed against this facility.
We believe that our existing cash resources
will be sufficient to fund our expected working capital needs through December 2022. If we raise additional funds by issuing equity
securities, our existing security holders will likely experience dilution, and the incurring of indebtedness would result in increased
debt service obligations and could require us to agree to operating and financial covenants that could restrict operations. In
the event that we are unable to obtain sufficient capital to meet our working capital requirements, we may be required to change
or curtail current or planned operations in order to conserve cash until such time, if ever, that sufficient proceeds from operations
are generated. In such an event, we may not be able to take advantage of business opportunities and may have to terminate or delay
safety and efficacy studies, curtail our product development programs, or sell or assign rights to our product candidates, products
and technologies.
Our future capital requirements depend on many
factors, including, but not limited to:
|
|
·
|
the scope, progress, results and costs of researching and developing our current or future product
candidates;
|
|
|
·
|
the timing of, and the costs involved in, obtaining regulatory approvals for any of our current
or future product candidates;
|
|
|
·
|
the number and characteristics of the product candidates we pursue;
|
|
|
·
|
the cost of manufacturing our current and future product candidates and any products we successfully
commercialize;
|
|
|
·
|
the cost of commercialization activities if any of our current or future product candidates are
approved for sale, including marketing, sales, service, customer support and distribution costs;
|
|
|
·
|
the expenses needed to attract and retain skilled personnel;
|
|
|
·
|
the costs associated with being a public company;
|
|
|
·
|
our ability to establish and maintain strategic collaborations, licensing or other arrangements
and the financial terms of such agreements; and,
|
|
|
·
|
the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing possible
patent claims, including litigation costs and the outcome of any such litigation.
|
Off Balance Sheet Arrangements
Since inception, we have not engaged in the
use of any off-balance sheet arrangements, such as structured finance entities, special purpose entities or variable interest entities.
Contingencies and Legal Proceedings
On November 1, 2019, Heska Corporation
(“Heska”) filed a complaint for damages and injunctive relief (the “Complaint”) in the United States
District Court for the Middle District of North Carolina, Case 1:19-cv-01108-LCB-JLW, against Qorvo US, Inc. (“Qorvo
US”), Qorvo Biotechnologies, LLC (“Qorvo Biotech” and, together with Qorvo US, “Qorvo”) and us
(collectively with Qorvo, the “Defendants”) which was amended on November 22, 2019. The amended Complaint
alleges, among other things, that the Defendants improperly obtained Heska’s trade secrets and confidential information
and/or conspired to use improper means to misappropriate Heska’s trade secrets related to an instrument and related
consumable products for performing immunoassay analysis of biomarkers and other substances. The amended Complaint seeks
compensatory and exemplary damages, as well as preliminary and permanent injunctive relief to prevent the Defendants from
commercializing our TRUFORMATM diagnostic instrument. On January 21, 2020, the Defendants filed a motion
seeking dismissal of the Complaint. On February 11, 2020, Heska filed its response to the Defendants’ motion to dismiss
to which the Defendants responded on February 25, 2020. Heska subsequently moved to strike a portion of the Defendants’
response. On September 30, 2020, the court denied the Defendants’ motion to dismiss and granted Heska’s motion to
strike. On October 14, 2020 the Defendants filed their answer to the amended Complaint. We believe that the allegations in
the amended Complaint have no merit and will not have a material adverse effect on our business, results of operations or
financial condition.
Under the terms of the Development and Supply
Agreement, dated November 26, 2018, by and between Qorvo Biotech and the Company (as amended, the “Qorvo Agreement”),
Qorvo Biotech agreed to indemnify us and certain related parties against claims alleging infringement or misappropriation of third-party
intellectual property rights, subject to certain limitations and exceptions. Qorvo Biotech has notified us that Qorvo Biotech has
assumed the defense of the amended Complaint and will indemnify us for losses arising from the amended Complaint in accordance
with the terms of the Qorvo Agreement. Qorvo Biotech has further advised us that it intends to mount a vigorous defense to the
claims in the amended Complaint, and that it believes the allegations contained in the amended Complaint are without merit.
Quantitative and Qualitative Disclosures about Liquidity
and Market Risk
Liquidity risk is the risk that we will encounter
difficulty raising liquid funds to meet our commitments as they fall due. In meeting our liquidity requirements, we closely monitor
our forecasted cash requirements with expected cash drawdown.
We are exposed to interest rate risk, which
is affected by changes in the general level of interest rates. Due to the fact that our cash is deposited with major financial
institutions in an interest-bearing savings account, we do not believe that the results of operations or cash flows would be affected
to any significant degree by a sudden change in market interest rates given their relative short-term nature.
We are also exposed to credit risk at period
end from the carrying value of our cash. We manage this risk by maintaining bank accounts with a Canadian Chartered Bank and a
U.S. bank that is a member of the Federal Deposit Insurance Corporation. Our cash is not subject to any external restrictions.
We are exposed to changes in foreign exchange
rates between the Canadian and United States dollar which could affect the value of our cash. We had no foreign currency hedges
or other derivative financial instruments as of December 31, 2019. We do not enter into financial instruments for trading or speculative
purposes and do not currently utilize derivative financial instruments.
We have balances denominated in Canadian dollars
that give rise to exposure to foreign exchange (“FX”) risk relating to the impact of translating certain non-U.S. dollar
balance sheet accounts as these statements are presented in U.S. dollars. A strengthening U.S. dollar will lead to a FX loss, while
a weakening U.S. dollar will lead to a FX gain. For each Canadian dollar balance of $1.0 million, a +/- 10% movement in the Canadian
currency held by us versus the U.S. dollar would affect our loss and other comprehensive loss by $100,000.
Recently adopted accounting pronouncements
In February 2016, the FASB issued new guidance,
ASU No. 2016-02, Leases (Topic 842). The new standard establishes a right-of-use model (“ROU”) that requires a lessee
to recognize a ROU asset and lease liability on the balance sheet for all leases with a term longer than 12 months. Leases will
be classified as finance or operating, with classification affecting the pattern and classification of expense recognition in the
income statement. Additional qualitative and quantitative disclosures are also required by the new guidance.
A modified retrospective transition approach
is required, applying the new standard to all leases existing at the date of initial application. We adopted the new standard with
an initial application date of January 1, 2019 and used the effective date as its date of initial application. Consequently, financial
information was not updated, and the disclosures required under the new standard were not provided for dates and periods before
January 1, 2019.
The new standard provides a number of optional
practical expedients in transition. We have elected the ‘package of practical expedients’, which permits us not to
reassess under the new standard prior conclusions about lease identification, lease classification and initial direct costs. We
have not elected the use-of-hindsight or the practical expedient pertaining to land easements; the latter not being applicable
to the Company.
On August 29, 2018, the FASB issued ASU 2018-15,
which amends ASC 350-40 to address a customer’s accounting for implementation costs incurred in a cloud computing arrangement
(CCA) that is a service contract. ASU 2018-15 aligns the accounting for costs incurred to implement a CCA that is a service arrangement
with the guidance on capitalizing costs associated with developing or obtaining internal-use software. Specifically, the ASU amends
ASC 350 to include in its scope implementation costs of a CCA that is a service contract and clarifies that a customer should apply
ASC 350-40 to determine which implementation costs should be capitalized in a CCA that is considered a service contract. The amendments
in this update are effective for public business entities for fiscal years beginning after December 15, 2019. Early adoption is
permitted.
We chose to adopt this guidance on July 1, 2019
using the prospective transition method.
MANAGEMENT
Executive Officers and Directors
The following table sets forth the name, age
and position of each of our directors and executive officers as of the Record Date:
|
Name
|
Age
|
Position
|
|
Robert Cohen
|
63
|
Interim Chief Executive Officer and Director
|
|
Ann Marie Cotter
|
50
|
Chief Financial Officer
|
|
Stephanie Morley
|
45
|
President and Chief Medical Officer
|
|
Bruk Herbst
|
51
|
Chief Commercial Officer
|
|
Jeffrey Rowe (1)(2)(3)
|
64
|
Chairman of the Board
|
|
Rodney Williams (1)(2)(3)
|
59
|
Director
|
|
Johnny D. Powers (1)(2)(3)
|
59
|
Director
|
|
Chris MacLeod (1)(2)(3)
|
51
|
Director
|
|
Christopher Wolfenberg
|
47
|
Director
|
Notes:
|
(1)
|
|
Member of the Audit Committee.
|
|
(2)
|
|
Member of the Compensation Committee.
|
|
(3)
|
|
Member of the Nominating and Corporate Governance Committee.
|
Management
Robert Cohen, has been our Interim
Chief Executive Officer of the Corporation since June 2020. From April 2017 to May 2019, he was President and Chief Executive Officer
of EmboMedics, Inc., an early stage medical device company. From November 2009 to February 2017 he was a founder and President
and Chief Executive Officer of Miromatrix Medical Inc., an early stage biotechnology company. Mr. Cohen also served as President
and Chief Executive Officer of Travanti Pharma Inc., Advanced Circulatory Systems Inc., and GCI Medical. In addition, he served
in senior management positions at St. Jude Medical, Inc, Sulzermedica, and Pfizer Inc. Mr. Cohen has a Bachelor of Arts degree
from Bates College and has a J.D. degree from the University of Maine School of Law.
Ann Cotter, CPA has been our Chief
Financial Officer and Corporate Secretary since October 2020. From August 2020 until October 2020 she served as our Interim Chief
Financial Officer and Corporate Secretary. From August 2018 to August 2020, she served as our Vice President, Finance. From April
2018 until August 2018, Ms. Cotter served as the chief financial officer of American Fence & Supply Co., Inc. From January
2018 until March 2018, she was a consultant to Aston Carter, a professional placement services company. From August 2014 until
September 2017, she served as the corporate controller to Promotion Technology Group, an audio visual solutions company. Ms. Cotter
holds a Master of Taxation from Capital University Law School and a Bachelor of Arts in Economics and Spanish from Capital University.
She is a Certified Public Accountant, licensed in Ohio since 1996.
Stephanie Morley, DVM has been our
President and Chief Medical Officer since September 2020. From September 2019 until September 2020 she served as our President
and Chief Operations Officer. From October 2015 until September of 2019, she served as our Chief Operating Officer and Vice President
of Product Development. Prior thereto, from July 2015 until October 2015, Dr. Morley was a consultant for us providing strategic
and tactical support. From December 2013 to August 2015 Dr. Morley served as Associate Director of Business Development with the
University of Michigan Medical School. From April 2006 to August 2013 Dr. Morley held several positions of increasing responsibility
with MPI Research, a contract research organization, including Vice President of Operations. Dr. Morley is a trained veterinarian,
having earned her DVM degree from Michigan State University. After earning her DVM degree, Dr. Morley was a practicing veterinarian
with Oakwood Animal Hospital in Kalamazoo, MI and Adobe Animal Medical Center in Albuquerque, NM where she assumed dual roles of
both clinical practitioner and operations management.
Bruk Herbst has been our Chief Commercial
Officer since July 2017. From October 2015 to December 2016 Mr. Herbst was the Executive Senior Vice President of Sales and Marketing
at i4C Innovations Inc. d/b/a Voyce, an animal health and wellness company. From October 2007 to September 2015, he served as Executive
Senior Director and Head of U.S. Sales at IDEXX Laboratories, Inc., a developer, manufacturer and distributor of products and services
for the companion animal veterinary and other markets, where he was responsible for in-clinic and reference lab diagnostics, point
of care, information technologies and digital radiology. From January 1999 to October 2007 Mr. Herbst also held commercial leadership
roles in patient monitoring, pharmacy and diagnostics with Omnicare Specialty Care Group and Life Systems. He holds a Bachelor
of Science degree in business from the University of Arizona.
Non-Management
Directors
Rodney Williams, MBA has
served as a director and the Chair of our corporate governance committee (now called the nominating and corporate governance committee
(the “Nominating and Corporate Governance Committee”)) since April 2016. He is currently employed as President and
CEO for PaceMate as of April 27, 2019. Previously, Mr. Williams served as an independent director on the board for PaceMate as
well as Corporate Global Vice President Portfolio and Services for publicly-traded Align Technologies (ALGN). Mr. Williams was
an entrepreneur-in-residence with PTV Healthcare Capital, a private equity investment firm and he has been with PTV since October
2015. Prior to PTV, he was President and CEO of Heart Rhythm Society Consulting Services from January 2013 through August 2015.
From January 2008 through January 2013, Mr. Williams served as Senior Vice President of Global Product Planning and Marketing at
St. Jude Medical Inc. Mr. Williams also served in commercial leadership roles in sales and marketing at GE Healthcare, Johnson
and Johnson, and Bausch & Lomb. Mr. Williams earned both his MBA and Bachelor of Science degrees from the University of Southern
California and attended the General Management Executive Leadership Program at The Wharton School of Business. We selected Mr.
Williams to serve on our Board of Directors due to his experience with both large and small-cap medical technology and related
health care companies and his global commercialization expertise.
Jeffrey Rowe has been Chairman of the
Board and Chairman of our compensation committee (the “Compensation Committee”) since December 2019, and has served
as a Director and the Chairman of our audit committee since April 2016. Until his retirement in October 2015, Mr. Rowe served as
Executive Vice President and a Director of Diplomat Pharmacy, Inc., the largest independent specialty pharmacy company in the U.S.
During his tenure with Diplomat, the company grew from a single location with less than $5 million in revenue, to sixteen locations
and $3 billion in sales, and became publicly traded on the New York Stock Exchange. Prior to his career with Diplomat, Mr. Rowe
owned two successful community pharmacies in Genesee County, Michigan. He holds a Bachelor of Pharmacy degree from Ferris State
University. We selected Mr. Rowe to serve on our Board of Directors due to his financial expertise and his extensive experience
in pharmaceutical operations, the specialty pharmacy industry and fundamental business strategies involving accreditation, contracting,
cybersecurity and regulation, combined with an expertise in compounding and integrative medicine.
Johnny D. Powers has been a director
since August 2019. Dr. Powers has over 30 years of experience in the medical diagnostics industry, including over seven years of
experience in veterinary healthcare as a senior executive at IDEXX Laboratories. Dr. Powers was Executive Vice President of IDEXX
from 2012 until 2016, overseeing multiple business units, including IDEXX Reference Labs, Telemedicine Services, Rapid Assay Point-of-Care
Products, Bioresearch and Worldwide Operations. He joined IDEXX as Corporate Vice President in February 2009, where he led IDEXX
Reference Labs to a global leadership position. Dr. Powers holds a bachelor's degree in chemistry from Wake Forest University,
an M.S. in chemical engineering from Clemson University, an M.B.A. from the Duke University Fuqua School of Business and a Ph.D.
in biochemical engineering from North Carolina State University. Dr. Powers has an extensive and proven track record of product
innovation, commercial execution and operational excellence in the medical diagnostics industry. He has led the development and
commercialization of hundreds of innovative diagnostic platforms, products and services in early-stage businesses as well as global,
multi-billion-dollar companies. We selected Mr. Powers to serve on our Board of Directors due to his background and experience
in the veterinary healthcare field and his proven capabilities in commercial operations.
Chris MacLeod has been a director since
July 2020. Mr. MacLeod is an attorney whose practice is focused on complex business litigation. Since January 2010, Mr. MacLeod
has been a founding partner of Cambridge LLP, a Canadian law firm. Mr. MacLeod holds a bachelor’s degree in political science
and religious studies from the University of Regina and an LLB from the University of Saskatchewan. We selected Mr. MacLeod to
serve on the Board of Directors due to his experience in representing companies in international matters.
Christopher Wolfenberg has been a director
since August 2020. Mr. Wolfenberg is a partner with Fasken Martineau DuMoulin LLP. He has practiced business law for
almost 20 years and has acted as a director and officer of public, private and not for profit corporations. Mr. Wolfenberg holds
an LL.M. from Cornell Law School, an LL.B. from Queen’s University and a Bachelor of Social Sciences from the University
of Ottawa. He has been awarded recognition for his career in law and his community service.
Board Composition
At
our 2020 annual shareholders’ meeting, the size of the board was fixed at six directors. Our existing bylaws provide that
our directors will hold office until the close of the first annual meeting of shareholders following his or her election unless
the director is elected for a stated term. Our board of directors is responsible for the business and affairs of our company and
considers various matters that require its approval.
Our board of directors is comprised of a majority
of directors who are “independent” (as discussed below), and the Board has established an Audit Committee, a Compensation
Committee and a Nominating and Corporate Governance Committee. We have adopted charters for our each of these committees and a
code of ethics and business conduct, or Code of Ethics. Our Code of Ethics is available on our website at www.zomedica.com. The
committee charters are also available for review on our website.
Under the Alberta Business Corporations Act,
(“ABCA”), at least 25% of our directors must be “resident Canadians” as defined in the ABCA. Messrs. MacLeod
and Wolfenberg meet this requirement.
Director Independence
Our board of directors has determined that all
of our directors, other than Mr. Cohen and Mr. Wolfenberg, are “independent,” as defined under the NYSE American. For
purposes of the NYSE American rules, an independent director means a person other than an executive officer or employee of our
company or any other individual having a relationship which, in the opinion of our board of directors, would interfere with the
exercise of independent judgment in carrying out the responsibilities of a director, subject to certain additional limitations.
Such directors are also deemed to be “independent” under applicable Canadian securities laws.
Code of Ethics
Our board of directors has adopted the Code
of Ethics, which applies to all officers, directors and employees. Our Code of Ethics is available on our website at www.zomedica.com.
Information contained in, or accessible through, our website does not constitute part of this proxy circular/prospectus. We intend
to disclose any amendments to our Code of Ethics, or waivers of its requirements, on our website or in our filings under the Exchange
Act.
Board Committees
Our board of directors has three standing committees:
The Audit Committee, the Compensation Committee and the Nominating and Corporate Governance Committee. All of our committee members
are “independent,” as defined under the NYSE American rules and for purposes of Canadian securities laws. Dr. Powers
was an “independent” director until December 2, 2019, when he was named as a Strategic Advisor to provide day-to-day
strategic oversight and management guidance to Mr. Rampertab, our former Interim Chief Executive Officer. Upon the expiration of
our agreement with Dr. Powers, he became an “independent” director.
Each of our committee charters is available
on our website at www.zomedica.com.
Audit Committee
Our audit committee is currently comprised of
four members, Mr. Rowe (Chairman), Mr. Williams, Dr. Powers and Mr. MacLeod. Each member of our audit committee is a non-employee
member of our board of directors. We have designated Mr. Rowe as our “audit committee financial expert,” as defined
under Item 407 of Regulation S-K. All of the members of our audit committee are “independent” members of our board
of directors, as required by the NYSE American rules and Canadian securities laws.
The purpose of our audit committee of our board
of directors is to oversee (i) the integrity of our company’s financial statements, our company’s accounting and financial
reporting processes and financial statement audits; (ii) our company’s compliance with applicable legal and regulatory requirements;
(iii) our company’s systems of internal control over financial reporting and disclosure controls and procedures; (iv) the
independent auditor’s engagement, qualifications, performance, compensation and independence; (v) review of related party
transactions; and (vi) compliance with the company’s corporate policies. The audit committee’s function is one of oversight,
whereas the planning and conduct of the audit is the responsibility of the independent auditor, and the financial statements are
the responsibility of the company’s management.
Each member of the audit committee has experience
reviewing financial statements and dealing with related accounting and auditing issues and is “financially literate”
within the meaning of applicable securities laws.
The audit committee has the sole authority to
pre-approve all audit and permitted non-audit services provided by the independent auditor.
Compensation
Committee
Our compensation committee is currently comprised
of four members, Mr. Rowe (Chairman), Mr. Williams, Dr. Powers and Mr. MacLeod. All of the members of our compensation committee
are “independent”, as defined under the NYSE American rules and for purposes of Canadian securities laws.
The purpose of our compensation committee is
to (i) make recommendations to our board of directors relating to evaluation and compensation of our executives, (ii) oversee incentive,
equity-based and other compensatory plans in which executive officers and key employees of our company participate, (iii) review
and participate in determining director compensation and (iv) prepare any report on executive compensation required by the rules
and regulations of the Commission and the listing standards of NYSE American.
Nominating
and Corporate Governance Committee
Our nominating and corporate governance committee
is currently comprised of four members, Mr. Williams (Chairman), Mr. Rowe, Dr. Powers and Mr. MacLeod. All of the members of our
nominating and corporate governance committee are “independent” as defined under the NYSE American rules and for the
purposes of Canadian securities laws.
The purpose of our nominating and corporate
governance committee of our board of directors is to carry out the responsibilities delegated by the board of directors relating
to our director nominations process, developing and maintaining our company’s corporate governance policies, and any related
matters required by the federal securities laws or by the applicable listing rules of the NYSE American.
Board Leadership
Structure and Role in Risk Oversight
Although we have not adopted a formal policy
on whether the Chairman and Chief Executive Officer positions should be separate or combined, we have determined that it is in
our best interests and the best interests of our shareholders to separate these roles at this time. Mr. Cohen currently serves
as our Interim Chief Executive Officer and Mr. Rowe serves as Chairman of our board of directors.
Our board of directors is primarily responsible
for overseeing our risk management processes. The board of directors receives and reviews periodic reports from management, auditors,
legal counsel, and others, as considered appropriate regarding our assessment of risks. The board of directors focuses on the most
significant risks facing our general risk management strategy, and us and also ensures that risks undertaken by us are consistent
with the board’s appetite for risk. While the board oversees our risk management, management is responsible for day-to-day
risk management processes. We believe that this division of responsibilities is the most effective approach for addressing the
risks facing us and that our board leadership structure supports this approach.
EXECUTIVE
AND DIRECTOR COMPENSATION
The following table shows the compensation for
each of the years ended December 31, 2019 and December 31, 2018 awarded to or earned by our principal executive officer and our
two other most highly compensated executive officers who were serving as executive officers as of December 31, 2019. The persons
listed in the following table are referred to herein as the “named executive officers.”
|
Name and Principal Position
|
|
|
|
($)
|
|
|
|
Bonus
($)
|
|
Option
Awards (4)
($)
|
|
|
|
All Other
Compensation (5)
($)
|
|
|
|
Total
($)
|
|
Gerald Solensky, Jr.(1)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2018
|
|
|
309,737
|
|
|
|
70,000
|
|
–
|
|
|
|
24,000
|
|
|
|
403,737
|
|
(Former) Chief Executive Officer and Director
|
2019
|
|
|
334,610
|
|
|
|
30,000
|
|
812,490
|
|
|
|
336,730
|
|
|
|
1,513,830
|
|
Shameze Rampertab(2)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(former) Interim Chief Executive Officer, Chief Financial
|
2018
|
|
|
242,128
|
|
|
|
72,625
|
|
–
|
|
|
|
7,390
|
|
|
|
322,143
|
|
Officer, Corporate Secretary and Director
|
2019
|
|
|
226,261
|
|
|
|
31,289
|
|
367,555
|
|
|
|
8,013
|
|
|
|
633,118
|
|
Stephanie Morley(3)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
President and Chief Operations Officer
|
2018
|
|
|
202,596
|
|
|
|
70,000
|
|
–
|
|
|
|
24,000
|
|
|
|
296,596
|
|
|
2019
|
|
|
207,292
|
|
|
|
30,000
|
|
448,212
|
|
|
|
26,082
|
|
|
|
711,586
|
__________________
(1) Mr. Solensky received no compensation for his services
as our President and Chief Executive Officer prior to December 31, 2015, other than a payment in the amount of $4,238, which he
subsequently gifted back to ZoMedica Pharmaceuticals Inc. Mr. Solensky entered into an employment agreement in December 2016
and amended in January 2017 pursuant to which he received an annual salary of $285,000 and a monthly car allowance of $2,000. Mr.
Solensky entered into an amended employment agreement in July 2018 pursuant to which he is entitled to receive an annual salary
of $325,000 and a monthly car allowance of $2,000. On December 2, 2019 Mr. Solensky resigned his positions of Chief Executive Officer
and Chairman of the Board. As part of his resignation Mr. Solensky received $338,286 in severance. No grant was issued to Mr. Solensky
in 2018. In 2019, Mr. Solensky received stock options of 2,100,000 shares valued at 812,490. Mr. Solensky resigned as an officer
and director of our company effective December 2, 2019.
(2) Pursuant to Mr. Rampertab’s amended employment
agreement in November 2016, he was entitled to receive an annual salary of $225,563. He also received a monthly car allowance of
$602. On December 2, 2019, Mr. Rampertab was named Interim Chief Executive Officer. No grant was issued to Mr. Rampertab in 2018;
in 2019 Mr. Rampertab received a stock option grant of 950,000 shares valued at $370,984. Mr. Rampertab ceased to be Interim Chief
Executive Officer upon the appointment of Mr. Cohen to that position. Mr. Rampertab resigned as a director on August 6, 2020 and
as Chief Financial Officer and Corporate Secretary effective August 14, 2020.
(3) Dr. Morley began serving as a consultant in July
2015 and received consulting fees of cash of $16,822 and 329,636 of common shares having a value of $22,600 at the times of issuance.
She was appointed Chief Operating Officer of ZoMedica Pharmaceuticals Inc. in October 2015. In connection with her appointment,
she received a signing bonus consisting of 485,944 common shares having a value of $87,400 at the time of issuance. Dr.
Morley entered into an employment agreement with ZoMedica Pharmaceuticals, Inc. in October 2015 pursuant to which she receives
an annual salary of $150,000 per annum, which was increased to $200,000 effective July 2017. Dr. Morley also received a monthly
allowance of $2,000 effective July 2017 for vehicle and tax preparation. Dr. Morley entered into an amended employment agreement
with Zomedica Pharmaceuticals, Inc. in September 16, 2019 which increased her salary from $200,000 to $225,000 per annum. There
was no change to the monthly car and tax preparation allowance of $2,000. No grant was issued to Dr. Morley in 2018; in 2019. Dr.
Morley received two stock option grants of 900,000 valued at $351,458 and 500,000 valued at $100,022.
(4) Represents the aggregate grant date fair value for
grants made in 2019 and 2018, respectively, computed in accordance with FASB ASC Topic 718. The assumptions we used in valuing
options are described in Note 11 to our financial statements included in this proxy circular/prospectus
(5) All Other Compensation represents monthly allowances
and severance amounts, net of vacation accrual.
In connection with Ms. Cotter’s appointment
as Chief Financial Officer, we have agreed to pay her a base salary of $200,000 per year. Ms. Cotter will be eligible to receive
an annual discretionary bonus of up to 25% of her base salary (pro-rated for 2020) based on the achievement of certain individual
and corporate performance objectives established by our chief executive officer and the Board.
If Ms. Cotter has a “separation of service”
within the meaning of Section 409A of the Code as a result of the Company terminating her employment without cause, then Ms. Cotter
shall be entitled to receive, in a lump sum and upon the execution of a standard release agreement, an amount equal to six month’s
base salary less applicable tax withholding.
In connection with her appointment, Ms. Cotter
received options to purchase an aggregate of 300,000 common shares at an exercise price of $0.11 per share. Options to purchase
75,000 common shares vested on the date of grant and the remaining options will vest ratably over a three-year period on the anniversary
of the date of grant.
Employment
and Consulting Agreements
Robert Cohen
In June 2020, we entered into a written employment
agreement with Mr. Cohen pursuant to which he serves as our Interim Chief Executive Officer. As amended, which does not have a
specified term, provides for Mr. Cohen to receive a base salary of $341,000 per year. Mr. Cohen will be eligible to receive a one-time
discretionary bonus of up to $81,500 at the end of Mr. Cohen’s employment based on the achievement of certain performance
and financial objectives established by our board of directors. Mr. Cohen is also eligible to participate in our employee benefit
plans, including health and 401(k) plans, subject to any qualification period therefor. He is also be entitled to the reimbursement
of reasonable business expenses. In the employment agreement, Mr. Cohen has agreed to customary confidentiality, non-competition
and intellectual property covenants.
In connection with his appointment, our board
granted to Mr. Cohen options to purchase an aggregate of 2,000,000 common shares at an exercise price of $0.19 per share under
our Stock Option Plan. One-third of such options are immediately exercisable, one-third vest three months from the date of grant
and the remaining one-third vest six months from the date of grant. The options will expire on the fifth anniversary of the date
of grant, subject to earlier termination upon the occurrence of certain circumstances.
Stephanie Morley
In connection with her appointment as President
Dr. Morley entered into a new employment agreement with us, which has an initial term of three years and automatically extends
for one-year terms unless either party elects to terminate it. Under the agreement, Dr. Morley will receive an annual base salary
of $225,000, subject to annual review and will be entitled to quarterly cash bonuses upon the achievement of certain specified
objectives established by the Board. Pursuant to the agreement, Dr. Morley will receive a $2,000 monthly allowance in respect of
the following items: (i) vehicle and (ii) tax preparation. Dr. Morley is entitled to four weeks paid vacation time. Pursuant to
the agreement, any options granted to Dr. Morley will be subject to accelerated vesting in the event that Dr. Morley’s employment
is terminated by us without cause. The agreement also includes customary non-solicitation, confidentiality and assignment of inventions
provisions. In the event that Dr. Morley has a “separation from service” within the meaning of Section 409A of the
Internal Revenue Code of 1986, as amended, or the Code, Dr. Morley would have the right to exercise all of her options, and we
would be required to pay her a lump sum equal to 12 months of her base salary and any quarterly bonus allocable or payable prior
to the date of termination.
Outstanding Equity Awards at Fiscal Year End
The following table sets forth certain information,
on an award-by-award basis, concerning outstanding equity awards for each named executive officer as of December 31, 2019.
|
Option awards
|
|
Stock awards
|
|
Name
|
|
Number of securities underlying unexercised options (#) exercisable
|
|
Number of securities underlying options (#) unexercisable
|
|
Equity incentive plan awards: number of securities underlying unexercised unearned options (#)
|
|
Option exercise price ($)
|
|
Option expiration date
|
|
Number of shares or units of stock that have not vested (#)
|
|
Market value of shares of units of stock that have not vested ($)
|
|
Equity incentive plan awards: number of unearned shares, units or other rights that have not vested (#)
|
Equity incentive plan awards: market or payout value of unearned shares, units or other rights that have not vested ($)
|
|
Gerald Solensky, Jr.(1)
|
|
|
1,705,265
|
|
|
–
|
|
|
–
|
|
|
1.52
|
|
1/10/2021
|
|
–
|
|
–
|
|
–
|
|
–
|
|
Shameze Rampertab (1)
|
|
|
950,000
|
|
|
–
|
|
|
–
|
|
|
1.52
|
|
1/10/2021
|
|
–
|
|
–
|
|
–
|
|
–
|
|
Stephanie Morley (1)
|
|
|
900,000
|
|
|
–
|
|
|
–
|
|
|
1.52
|
|
1/10/2021
|
|
–
|
|
–
|
|
–
|
|
–
|
|
Bruk Herbst (1)
|
|
|
300,000
|
|
|
–
|
|
|
–
|
|
|
1.52
|
|
1/10/2021
|
|
–
|
|
–
|
|
–
|
|
–
|
|
Stephanie Morley (2)
|
|
|
500,000
|
|
|
|
|
|
|
|
|
.43
|
|
9/16/2021
|
|
–
|
|
–
|
|
–
|
|
–
|
__________________
(1) Stock options vest
immediately upon issue, with an issue date of January 10, 2019, and expire on January 10, 2021.
(2) Stock options vest
immediately upon issue, with an issue date of September 16, 2019 and expire on September 16, 2021.
Equity Compensation Plan Information
The following table provides information, as
of December 31, 2019, with respect to all compensation arrangements maintained by us, including individual compensation arrangements,
under which shares are authorized for issuance.
|
Plan Category
|
|
|
Number of Securities to be issued upon outstanding options rights
|
|
|
|
Weighted-average exercise price outstanding options and rights
|
|
|
|
Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in columns (a))
|
|
|
|
|
(a)
|
|
|
|
(b)
|
|
|
|
(c)
|
|
Equity compensation plans approved by shareholders
|
|
|
7,040,265
|
|
|
$
|
1.29
|
|
|
|
0
|
|
Equity compensation plans not approved by shareholders
|
|
|
Nil
|
|
|
|
N/A
|
|
|
|
Nil
|
|
Total
|
|
|
7,040,265
|
|
|
$
|
1.29
|
|
|
|
0
|
Stock Option
Plans
As of December 31, 2015, Zomedica Pharmaceuticals
Corp (formerly, Wise Oakwood Ventures Inc.), had a shareholder-approved option plan, or the WOW Plan, pursuant to which options
to purchase 200,000 common shares were outstanding. The terms of the WOW Plan were substantially similar to those of our current
Stock Option Plan. In connection with the Qualifying Transaction, these options were consolidated into options to purchase 80,000
common shares of Zomedica Pharmaceuticals Corp. and fully exercised and the WOW Plan was terminated.
In April 2016, concurrent with the completion
of the Qualifying Transaction, we adopted a new equity stock option plan, the Stock Option Plan. The Stock Option Plan was approved
by our shareholders. The purpose of the Stock Option Plan is to attract and retain employees, consultants, officers and directors
to our company and to motivate them to advance the interests of our company by affording them with the opportunity, through share
options, to acquire an equity interest in our company and benefit from its growth.
Administration. The Stock Option Plan
is administered by our board of directors. Our board of directors may grant options to purchase shares of our common shares or
such other shares as may substitute therefore in the capital of Zomedica Pharmaceuticals Corp. Our board of directors also has
authority to determine the terms and conditions of each award, prescribe, amend and rescind rules and regulations relating to the
Stock Option Plan, and amend the terms of awards (provided that no amendment may materially prejudice the rights of a participant
without consent such participant’s consent). Our board of directors may delegate authority to a committee of our directors
or to an officer. Our board or directors may terminate the Stock Option Plan.
Eligibility. Persons eligible to receive
awards under the Stock Option Plan include any person who is an employee, officer, director or consultant provided that any consultant
has performed and/or continues to perform services for our company under a written agreement and on an ongoing basis or is expected
to provide a service to our company.
Shares Subject to the Stock Option Plan.
The aggregate number of shares of common shares available for issuance in connection with options and awards granted under the
Stock Option Plan is ten percent of the total number of issued and outstanding common shares calculated on a non-diluted basis.
If any award of options granted under the Stock Option Plan expires or terminates without having been fully exercised, that number
of common shares shall become available for the purpose of future grants under the Stock Option Plan.
Terms and Conditions of Options. Our
board of directors will determine the exercise price of options granted under the Stock Option Plan. The exercise price of stock
options may not be less than that from time to time permitted under the rules of any stock exchange on which the common shares
are then listed. In addition, the exercise price of an option must be paid in cash.
The number of common shares subject to each
option shall be determined by our board of directors with the following limitations. The number of common shares reserved for issuance
to any one individual, consultant, person conducting investor relations or insider (as defined in the Securities Act (Alberta))
in a 12-month period may not exceed 5%, 2%, 2% and 10%, respectively, of the issued and outstanding common shares at the time of
the grant.
No option may be exercisable for more than ten
years from the date of grant. Options granted under the Stock Option Plan will be exercisable at such time or times as our board
of directors prescribes at the time of grant. Options shall only be exercised by the participant as long as the optionee remains
or was within the last ninety days an employee, officer, director or consultant, if the optionee dies, within one year of the optionee's
death or if an optionee is engaged in investor relations activities, within 30 days of being so engaged by our company.
All benefits, rights and options accruing under
the Stock Option Plan are non-transferrable and non-assignable unless specifically provided in the grant. During the lifetime of
a participant, any options granted under the Stock Option Plan may only be exercised by the participant and in the event of the
death of a participant, by the person or persons to whom the participant's rights under the option pass by the participant's will
or applicable law.
Effect of Certain Corporate Transactions.
In the event of a sale by our company of all or substantially all of its assets or in the event of a change of control (as defined
in the Stock Option Plan) of our company, each participant shall be entitled to exercise, in whole or in part, the options granted
to such participant under the Stock Option Plan, either during the term of the option or within ninety days after the date of the
sale or change of control, whichever first occurs. Upon completion of the domestication, holders of our outstanding options will
continue to hold the same options, which will remain exercisable for an equivalent number of shares of the same class of common
stock of Zomedica Corp. (the Delaware corporation), for the equivalent exercise price per share, without any action by the holder,
in accordance with their terms.
Director Compensation
We have not established a formal compensation
policy for our outside directors. Set forth below is information concerning the compensation of directors, other than directors
who are our employees, paid during the year ended December 31, 2019:
On January 10, 2019, we granted the following
options to our non-employee directors:
|
|
·
|
Mr. LeBar - options to acquire 400,000 common shares,
|
|
|
·
|
Mr. Williams - options to acquire 400,000 common shares,
|
|
|
·
|
Mr. Rowe - options to acquire 350,000 common shares.
|
Each of these options had an exercise price $1.52 per common share,
were immediately exercisable and expire two years from the date of grant.
On August 16, 2019, we granted the following
options to Dr. Powers in connection with his appointment as a director of our company: - options to acquire 500,000 common shares
at an exercise price of $0.26 per common share; options to acquire 100,000 common shares at an exercise price of $0.35 per common
share; options to acquire 100,000 common shares at an exercise price of $0.45 per common share; options to acquire 100,000 common
shares at an exercise price of $0.55 per common share; options to acquire100,000 common shares at an exercise price of $0.65 per
common share; and options to acquire 100,000 common shares at an exercise price of $0.75 per common share. Each of these options
were immediately exercisable and expire two years from the date of grant.
The table below summarizes the compensation
we paid to our non-employee directors in 2019.
|
Name
|
|
Fees earned or
paid in cash ($)
|
|
|
|
Stock Awards
($)
|
|
|
|
Option Awards
($)
|
|
|
|
Total ($)
|
|
Jim Lebar
|
|
–
|
|
|
|
–
|
|
|
$
|
156,204
|
|
|
$
|
156,204
|
|
Rod Williams
|
|
–
|
|
|
|
–
|
|
|
$
|
156,204
|
|
|
$
|
156,204
|
|
Jeff Rowe
|
|
–
|
|
|
|
–
|
|
|
$
|
136,678
|
|
|
$
|
136,678
|
|
Johnny D. Powers
|
|
–
|
|
|
|
–
|
|
|
$
|
97,986
|
|
|
$
|
97,986
|
On December 3, 2019, we named Dr. Powers as
a Strategic Advisor to provide day-to-day strategic oversight and management guidance to Mr. Rampertab, our former Interim Chief
Executive Officer. In connection therewith, we entered into a strategic advisory agreement with Dr. Powers pursuant to which we
agreed to pay him the following compensation:
|
|
·
|
The sum of $190 per hour, billable in increments of one-quarter of an hour;
|
|
|
·
|
An option grant of 250,000 options to acquire common shares, with an exercise price at fair market
value, a two-year term and full vesting on date of grant, subject to establishment and approval by the Board of Directors accordance
with the stock option plan;
|
|
|
·
|
Reimbursement for travel expenses, food, lodging, any per diem allowance, equipment, supplies,
or similar items
|
As of December 31, 2019, Dr. Powers had not received any compensation
under this agreement. The agreement expired on April 30, 2020. We paid Dr. Powers a total of $15,533 under this agreement and reimbursed
him for $3,729 of related out-of-pocket expenses.
Securities Ownership
of Certain beneficIal owners and management
The table below sets forth certain information
with respect to beneficial ownership of our securities as of the Record Date by:
|
|
·
|
each person known by our directors and executive officers to beneficially own, or control or direct,
directly or indirectly, more than 5% of our issued and outstanding common shares;
|
|
|
·
|
each of our directors and each of our named executive officers; and
|
|
|
·
|
all our executive officers and directors, as a group.
|
The number of common
shares beneficially owned by each shareholder as indicated below has been determined in accordance with applicable United States
securities requirements. Under such rules, beneficial ownership includes any common shares as to which a person has sole or shared
voting power or investment power. Percentage ownership is based on 564,051,438 common shares outstanding on the Record Date. In
computing the number of common shares beneficially owned by a person and the percentage ownership of that person, common shares
subject to stock options, warrants or other rights held by such person that are currently convertible or exercisable or will become
convertible or exercisable within 60 days of the Record Date are considered outstanding, although these common shares are not considered
outstanding for purposes of computing the percentage ownership of any other person.
Unless otherwise stated, the address of each
beneficial holder is c/o Zomedica Pharmaceuticals Corp., 100 Phoenix Drive, Suite 180, Ann Arbor, Michigan 48108. We believe, based
on information provided to us, that each of the shareholders listed below has sole voting and investment power with respect to
the common shares beneficially owned by the shareholder unless noted otherwise, subject to community property laws where applicable.
|
|
Beneficial Ownership
|
|
Name of the Beneficial Owner
|
Number of Shares Beneficially Owned
|
Percentage of Total Outstanding common shares
|
|
Jeffrey Rowe (Chairman)(1)
|
12,302,980
|
2.2%
|
|
Robert Cohen (Interim Chief Executive Officer and Director)(2)
|
666,667
|
*
|
|
Ann Marie Cotter (Interim Chief Financial Officer) (3)
|
125,000
|
*
|
|
Stephanie Laine Morley (President & Chief Medical Officer)(4)
|
1,877,080
|
*
|
|
Bruk Herbst (Chief Commercial Officer)(5)
|
240,996
|
*
|
|
Chris Macleod (Director)(6)
|
50,150
|
*
|
|
Johnny D. Powers (Director)(7)
|
62,500
|
*
|
|
Rodney Williams (Director)(8)
|
251,900
|
*
|
|
Christopher Wolfenberg (Director)
|
N/A
|
*
|
|
All executive officers and directors as a group (9 persons)
|
15,577,273
|
2.8%
|
* Less than one percent
|
|
(1)
|
Includes 11,120,000 common shares held in the Rowe Family GST Trust, 664,480 common shares held
by the Jeffrey M. Rowe U/T/A dated November 5, 2004 (the “Jeffrey M. Rowe Living Trust”) and 181,000 common shares
held by Mr. Rowe through his IRA. Mr. Rowe's sister, Michele Ramo, serves as trustee to the Rowe Family GST Trust with Mr. Rowe's
oversight. Mr. Rowe has disclaimed all beneficial ownership of the common shares held in the Rowe Family GST Trust except to the
extent of his pecuniary interest therein. Mr. Rowe serves as trustee to the Jeffrey M. Rowe Living Trust and exclusively makes
all investment decisions on behalf of this trust. Also includes options to purchase 62,500 common shares exercisable within 60
days of the Record Date.
|
|
|
(2)
|
Consists of options to purchase 666,667 common shares exercisable within 60 days of the Record
Date.
|
|
|
(3)
|
Consists of options to purchase 125,000 common shares exercisable within 60 days of the Record
Date.
|
|
|
(4)
|
Includes 641,685 common shares held by The Dr. Stephanie Morley Revocable Living Trust and 5,000
common shares held by Dr. Morley's children. Also includes options to purchase 412,500 common shares exercisable within 60 days
of the Record Date
|
|
|
(5)
|
Includes 3,000 common shares held by Mr. Herbst’s children and options to purchase 175,000
common shares exercisable within 60 days of the Record Date.
|
|
|
(6)
|
Includes options to purchase 43,750 common shares exercisable within 60 days of the Record Date.
|
|
|
(7)
|
Consists of options to purchase 62,500 common shares exercisable within 60 days of the Record Date.
|
|
|
(8)
|
Includes 40,000 common shares held by Entrust Group Inc. FBO Rodney James Williams IRA and options
to purchase 50,000 common shares exercisable within 60 days of the Record Date.
|
Certain Relationships
and Related PARTY Transactions
Other than compensation arrangements for our
named executive officers and directors, we describe below each transaction or series of transactions, since January 1, 2019, to
which we were a party or will be a party, in which:
|
|
·
|
the amounts involved exceeded or will exceed $120,000; and
|
|
|
·
|
any of our directors, executive officers or holders of more than 5% of our common shares, or any
member of the immediate family of the foregoing persons, had or will have a direct or indirect material interest.
|
Compensation arrangements for our named executive
officers and directors are described in “Executive and Director Compensation”.
Equidebt Working
Capital Facility
On September 1, 2017, Equidebt LLC, or Equidebt,
one of our shareholders, which is controlled by Bradley J. Hayosh and Jeffrey S. Starman, entered into a Loan Agreement, or the
Loan Agreement, with Mr. Solensky pursuant to which Equidebt agreed to provide Mr. Solensky with an unsecured line of credit in
the amount of $5,000,000 for the purpose of enabling Mr. Solensky to exercise options to purchase up to 950,000 common shares expiring
on December 21, 2018 and to purchase additional common shares from us from time to time, or the line of credit. Amounts borrowed
under the line of credit were to bear interest at a rate of 14% per annum payable at maturity. In addition, Mr. Solensky was required
to pay Equidebt a monthly maintenance fee of $6,250 per month payable at maturity. All amounts borrowed under the line of credit
were to become due and payable on September 1, 2022. Upon the occurrence of an Event of Default (defined in the Loan Agreement
to include Mr. Solensky’s failure to make payments under the line of credit or his other indebtedness when due, the occurrence
of certain insolvency events relating to Mr. Solensky or the occurrence of a substantial change in the existing or prospective
financial condition or net worth of Mr. Solensky which Equidebt determines to be materially adverse), Equidebt had the right to
declare all amounts outstanding under the line of credit immediately due and payable. We were not a party to the line of credit,
which was full recourse against Mr. Solensky.
As a result of discussions with the NYSE American
in connection with our application to list our common shares, we restructured and replaced the line of credit. Accordingly, on
October 17, 2017, we entered into a Loan Agreement, or the Working Capital Loan Agreement, with Equidebt pursuant to which Equidebt
agreed to provide us with a five-year $5,000,000 unsecured working capital line of credit, or the working capital line of credit.
Amounts borrowed under the working capital line of credit bear interest at a rate of 14% per annum payable at maturity. All amounts
borrowed under the line of credit become due and payable on October 17, 2022. Upon the occurrence of an Event of Default (defined
in the Working Capital Loan Agreement to include our failure to make payments under the working capital line of credit or our other
indebtedness when due, the occurrence of certain insolvency events relating to us, Equidebt has the right to declare all amounts
outstanding under the working capital line of credit immediately due and payable. No amounts were outstanding under the Equidebt
Facility at December 31, 2019.
In May 2018, we announced that we had commenced
a private offering to accredited investors in the United States of up to 4,651,162 common shares at a price of $2.15 per share
for aggregate gross proceeds of up to $10.0 million (the “May 2018 Private Placement”). We sold an aggregate of 1,861,627
common shares in the May 2018 Private Placement for gross proceeds of approximately $4.0 million. The 1,861,627 common shares are
covered by the Company’s Registration Statement on Form S-3 filed with the Commission on February 7, 2019 (File No. 333-229014).
In connection with our May 2018 private placement, Equidebt LLC acquired 1,209,302 of our common shares at a price of $2.15 per
share for total proceeds of approximately $2.6 million.
Wickfield Phoenix
LLC Lease Agreement
Wickfield Phoenix LLC is an affiliate of Wickfield
Properties, LLC, which is controlled by Bradley J. Hayosh and Jeffrey S. Starman, who beneficially own over 5% of the common shares.
On February 1, 2020 we entered into a new lease with Wickfield Phoenix LLC for 16,226 square feet of office space and cancelled
our existing lease with Wickfield Phoenix LLC. The new lease period is for 60 months, commencing on February 1, 2020 and ending
on January 31, 2025 with a monthly rent payment of $32,452. Upon cancellation of our existing lease, we received a refund of prepaid
rent in the amount of $1,002,113.
Sale of Series 1
Preferred Shares
In May 2019, we
issued five Series 1 preferred shares to Wickfield Capital LLC for an aggregate purchase price of $5,000,000. In June 2019,
we issued an additional seven Series 1 preferred shares to Wickfield Bridge Fund LLC for an aggregate purchase price of
$7,000,000. Wickfield Capital LLC and Wickfield Bridge Fund LLC are affiliates of Jeffrey S. Starman.
Indemnification
of Officers and Directors
As permitted by Delaware law, our proposed certificate
of incorporation will provide that, to the fullest extent permitted by Delaware law, no director will be personally liable to us
or our stockholders for monetary damages for breach of fiduciary duty as a director. Pursuant to Delaware law such protection would
be not available for liability:
|
|
·
|
for any breach of a duty of loyalty to us or our stockholders;
|
|
|
·
|
for acts or omissions not in good faith or that involve intentional misconduct or a knowing violation
of law;
|
|
|
·
|
for any transaction from which the director derived an improper benefit; or
|
|
|
·
|
arising from unlawful payments of dividends or unlawful stock repurchases or redemptions as provided in Section 174 of the DGCL.
|
Our
proposed certificate of incorporation provides for exculpation to the fullest extent permitted by the DGCL as the same exists
or as may hereafter be amended. Therefore, if Delaware law is amended after the approval by our stockholders of the certificate
of incorporation to authorize corporate action further eliminating or limiting the personal liability of directors, then the liability
of our directors will be eliminated or limited to the fullest extent permitted by Delaware law. In addition, the proposed bylaws
of Zomedica Corp. will provide that we are required to indemnify and advance expenses to our current and former directors and
officers as incurred in connection with legal proceedings against them for which they may be indemnified and that the rights conferred
in the bylaws are not exclusive.
THE SPECIAL MEETING
Solicitation of Proxies
This proxy circular/prospectus
is furnished in connection with the solicitation of proxies by our management for use at our virtual-only Special Meeting to be
held at 8:30 a.m. (Toronto time) on December 15, 2020, via live audio online webcast at https://web.lumiagm.com/157200202, password
“zomedica2020”, and at any postponements or adjournments thereof for the purposes set forth in the Notice of Special
Meeting, which accompanies this proxy circular/prospectus. This proxy circular/prospectus is dated 2020, and is first being mailed
to shareholders on or about , 2020. Solicitation of proxies will be primarily by mail, but may also be undertaken by way of telephone,
facsimile or oral communication by our board of directors of (the “Board of Directors” or the “Board”),
and our officers and regular employees, at no additional compensation. We will bear all costs associated with the solicitation
of proxies.
We have retained Alliance Advisors to assist
us in soliciting votes for the Special Meeting. We have agreed to pay Alliance Advisors a fee of approximately U.S. $100,000 for
its services and to reimburse it for its reasonable out-of-pocket expenses.
We will also, upon request, reimburse brokers
and other persons holding Common Shares in their names, or in the names of nominees, for their reasonable out-of-pocket expenses
for forwarding proxy materials to the beneficial owners of the Common Shares and to obtain proxies or voting instructions.
The record date for
the determination of shareholders entitled to receive notice of, and to vote at, the Special Meeting is October 22, 2020. At such
date, 564,051,438 common shares and 12 Series 1 preferred shares were outstanding.
No person is authorized to give any information
or to make any representations other than those contained in this proxy circular/prospectus and, if given or made, such information
or representations should not be relied upon as having been authorized by us. The delivery of this proxy circular/prospectus shall
not, under any circumstances, create an implication that there has not been any change in the information set forth herein since
the date of this proxy circular/prospectus.
Virtual-Only Special Meeting
We are holding the Special Meeting as a virtual-only
meeting, which will be conducted by live audio online webcast, as a result of the extremely serious impact of the global coronavirus
pandemic, and in response to recent public health measures by applicable government authorities, and to prudently protect the health
and well-being of our communities and our shareholders, employees, services partners and other stakeholders who wish to participate
in the Special Meeting. Shareholders will have an opportunity to participate in the Special Meeting online regardless of their
physical location. Shareholders will not be able to attend the Special Meeting in person.
Registered Shareholders and duly appointed proxyholders
who participate in the Special Meeting online will be able to listen to the Special Meeting, ask questions and vote, all in real
time, provided they are connected to the internet and comply with all of the requirements set out below under “Participation
in the Special Meeting.” Non-registered shareholders who have not duly appointed themselves as proxyholders may still participate
in the Special Meeting as guests. Guests will be able to listen to the Special Meeting, but will not be able to vote at the Special
Meeting or ask questions.
Voting by Proxy
Registered Shareholders
A shareholder is a registered shareholder (sometimes
referred to as a “Registered Shareholder”) if the shareholder has a share certificate in the shareholder’s name.
Registered Shareholders can vote by proxy by completing the instrument of proxy (as defined below), then signing and dating it
and providing it to AST Trust Company (Canada), the transfer agent of the Corporation (“AST”), in the manner provided
below.
Accompanying this proxy circular/prospectus
is an Instrument of Proxy (the “instrument of proxy” or the “proxy”) for use at the Special Meeting. Registered
Shareholders who wish to be represented by proxy by a person other than those named in the instrument of proxy are required to
insert the name of the designated person (discussed further below), date and sign the enclosed proxy and provide it to AST and
register the proxyholder by contacting AST at 1-866-751-6315 (within North America) or 212-235-5754 (outside North America), and
provide AST with the required information for your proxyholder so that AST may provide the proxyholder with a control number. Registering
the proxyholder is an additional step to be completed after the Registered Shareholder has submitted the instrument of proxy. The
control number will allow your proxyholder to log in to and vote at the Special Meeting online. Without a control number, your
proxyholder will not be able to vote or ask questions at the Special Meeting . They will only be able to attend the Special Meeting
online as a guest.
All properly executed proxies for Registered
Shareholders must be mailed so as to reach or be deposited at the offices of AST Trust Company (Canada), Attention: Proxy Department,
P.O. Box 721, Agincourt, ON M1S 0A1. Registered Shareholders can also fax their proxy to 416-368-2502 or toll-free within North
America 1-866-781-3111 or scan and e-mail their proxy to proxyvote@astfinancial.com. In any event, AST must receive a Registered
Shareholder’s completed instrument of proxy not later than 48 hours (excluding Saturdays, Sundays and statutory holidays)
prior to the time set for the Special Meeting or any adjournment or postponement thereof. Registered Shareholders may also use the internet (at www.astvotemyproxy.com) or a touch-tone
telephone (call toll free 1-888-489-5760 (English only)) to submit their voting instructions.
The persons designated in the instrument of
proxy are officers of our company. A shareholder has the right to appoint a company or person (who need not be a shareholder) other
than the persons designated in the accompanying instrument of proxy, to attend at and represent the shareholder at the Special
Meeting. To exercise this right, a Registered Shareholder should insert the name of the designated representative in the blank
space provided on the instrument of proxy and strike out the names of management's nominees. Alternatively, a Registered Shareholder
may complete another appropriate instrument of proxy. As indicated above, the Registered Shareholder must also take the additional
step of registering the proxyholder as to obtain a control number from AST.
The instrument of proxy must be signed by the
Registered Shareholder or the Registered Shareholder's duly appointed attorney authorized in writing or, if the shareholder is
a corporation, by a duly authorized officer or attorney of such corporation. An instrument of proxy signed by a person acting as
attorney or in some other representative capacity (including a representative of a corporate shareholder) should indicate that
person's capacity (following his or her signature) and should be accompanied by the appropriate instrument evidencing qualification
and authority to act (unless such instrument has previously been filed with us).
Beneficial Shareholders
If your common shares are held in a stock brokerage
account or by a bank or other nominee, you are considered the beneficial owner of Common Shares (a “Beneficial Shareholder”),
and these proxy materials are being forwarded to you by your broker, bank or nominee (each, an “Intermediary”), which
is considered, with respect to those common shares, the shareholder of record. As a Beneficial Shareholder, you have the right
to direct your Intermediary as to how to vote and are also able to appoint a proxyholder to participate at the Special Meeting.
If you are a Beneficial Shareholder of common
shares registered in the name of your broker, bank or other Intermediary, your common shares are held by your broker, bank or other
Intermediary, or in “street name,” and you will need to obtain a voting instruction form the organization that holds
your common shares and follow the instructions included on that form regarding how to instruct the organization to vote your common
shares.
Under applicable Canadian laws, your common
shares cannot be voted by your bank, broker or other Intermediary except in accordance with voting instructions received from you
as the Beneficial Shareholder.
We have distributed copies of this proxy circular/prospectus
for distribution to Beneficial Shareholders. Intermediaries are required to forward this proxy circular/prospectus to Beneficial
Shareholders unless a Beneficial Shareholder has waived the right to receive them. Typically, Intermediaries will use a service
company (such as Broadridge Financial Solutions, Inc., or Broadridge) to forward these materials to Beneficial Shareholders. Further,
subject to United States federal securities law requirements applicable to Beneficial Shareholders in the United States, we do
not intend to pay for Intermediaries to deliver any proxy-related materials (including this proxy circular/prospectus) to “objecting
beneficial owners”, and as a consequence, such objecting beneficial owners may not receive these materials unless their Intermediary
assumes the cost of delivery.
Generally, Beneficial Shareholders who have
not waived the right to receive meeting materials and are not “objecting beneficial owners” will:
|
|
a)
|
have received, together with the proxy circular/prospectus, a voting instruction form which must be completed, signed and delivered by the Beneficial Shareholder in accordance with the directions on the voting instruction form; voting instruction forms sent by Broadridge permit the completion of the voting instruction form by telephone or through Broadridge's dedicated voting website; and
|
|
|
b)
|
less typically, be given an instrument of proxy which has already been signed by the Intermediary (typically by a facsimile, stamped signature) which is restricted as to the number of Common Shares beneficially owned by the Beneficial Shareholder but which is otherwise uncompleted. This instrument of proxy need not be signed by the Beneficial Shareholder. In this case, the Beneficial Shareholder who wishes to submit a proxy should otherwise properly complete the instrument of proxy and deposit it with AST as indicated below.
|
The purpose of these procedures is to permit
Beneficial Shareholders to direct the voting of the common shares they beneficially own. Should a Beneficial Shareholder wish to
participate at the Special Meeting and vote his or her common shares in real time (or have another person attend and vote on behalf
of the Beneficial Shareholder), the Beneficial Shareholder should strike out the names of the persons named in the instrument of
proxy or voting instruction form and insert the Beneficial Shareholder's (or such other person's) name in the blank space provided
or, in the case of a voting instruction form, follow the corresponding instructions on the form.
If you are a Beneficial Shareholder located
in the United States and wish to participate at the Special Meeting and vote your common shares in real time (or have another person
attend and vote on your behalf) we understand that the above process involving usage of a voting instruction form received from
your Intermediary may not be possible, and you may be required to obtain a valid legal proxy from your Intermediary. The Beneficial
Shareholder should follow the instructions from its Intermediary included with the legal proxy form or the voting information form
sent to the Beneficial Shareholder, or contact the applicable Intermediary to request a legal proxy form or a legal proxy if the
Beneficiary has not received one. After obtaining a valid legal proxy from the Intermediary, the Beneficial Shareholder must then
submit such legal proxy to AST. All properly executed legal proxies for Beneficial Shareholders must be mailed so as to reach
or be deposited at the offices of AST Trust Company (Canada), Attention: Proxy Department, P.O. Box 721, Agincourt, ON M1S 0A1.
Beneficial Shareholders can also fax their proxy to 416-368-2502 or toll-free within North America 1-866-781-3111 or scan and e-mail
their proxy to proxyvote@astfinancial.com. In any event, AST must receive a Beneficial Shareholder’s completed instrument
of proxy not later than 48 hours (excluding Saturdays, Sundays and statutory holidays) prior to the time set for the Special Meeting
or any adjournment or postponement thereof.
The Chairman of the Special Meeting may waive
or extend the proxy cut-off without notice.
Beneficial Shareholders should carefully
follow the instructions of their Intermediaries and their service companies and allow enough time for the Intermediaries to process
the instructions prior to the Special Meeting. If the Beneficial Shareholder desires to participate at the Special Meeting and
vote his or her Common Shares, or appoint a third party proxyholder, the Beneficial Shareholder will also need to register the
proxyholder as to permit the proxyholder to be able to participate at the Special Meeting. AST will provide registered proxyholders
with a control number after the proxy deadline as to enable them to participate in the Special Meeting (see “Voting by
Proxy - Registered Shareholders” and “Participation in the Special Meeting “). Without a control number,
the proxyholder will not be able to vote or ask questions at the Special Meeting. They will only be able to attend the Special
Meeting online as a guest.
Changing Your Vote
A Registered Shareholder who has submitted an
instrument of proxy may revoke it at any time prior to the exercise thereof. In addition to any manner permitted by law, a proxy
may be revoked by instrument in writing executed by the Registered Shareholder or by his or her duly authorized attorney or, if
the Registered Shareholder is a corporation, executed by a duly authorized officer or attorney of the corporation and deposited
either: (i) at the registered office of our company at any time up to and including the last business day preceding the day of
the Special Meeting, or any adjournments or postponements thereof, at which the instrument of proxy is to be used; or (ii) with
the Chairman of the Special Meeting on the day of the Special Meeting, or any adjournment or postponement thereof. In addition,
an instrument of proxy may be revoked in any other manner permitted by law.
A Beneficial Shareholder may also revoke voting
instructions previously submitted. If a Beneficial Shareholder wishes to revoke voting instructions, he or she should contact their
Intermediary to discuss what procedures need to be followed. The change or revocation of voting instructions by a Beneficial Shareholder
can take several days or longer to complete and accordingly, any such action should be completed well in advance of the deadline
prescribed in the instrument of proxy or voting instruction form by the Intermediary or its service company to ensure it is given
effect at the Special Meeting .
Voting of Proxies and Exercise of Discretion by
Proxyholders
All shares represented at the Special Meeting
by properly executed proxies will be voted on any ballot that may be called for and, where a choice with respect to any matter
to be acted upon has been specified in the instrument of proxy, the shares represented by the instrument of proxy will be voted
in accordance with such instructions. The management designees named in the accompanying instrument of proxy will vote or withhold
from voting the shares in respect of which they are appointed in accordance with the direction of the shareholder appointing him
or her on any ballot that may be called for at the Special Meeting. The persons named in the enclosed proxy will have discretionary
authority with respect to any amendments or variations of the matters of business to be acted on at the Special Meeting or any
other matters properly brought before the Special Meeting or any adjournment or postponement thereof, in each instance, to the
extent permitted by law, whether or not the amendment, variation or other matter that comes before the meeting is routine and whether
or not the amendment, variation or other matter that comes before the meeting is contested. In the event that amendments or variations
to matters identified in the Notice are properly brought before the Special Meeting or any further or other business is properly
brought before the Special Meeting, it is the intention of the management designees to vote in accordance with their best judgment
on such matters or business. At the time of printing this proxy circular/prospectus, our management knows of no such amendment,
variation or other matter to come before the Special Meeting other than the matters referred to in the accompanying Notice.
Participation in the Special Meeting
We are holding the Special Meeting as a virtual-only
meeting, which will be conducted by live audio online webcast. Shareholders will not be able to participate in the Special Meeting
in person.
Participating in the Special Meeting online
enables Registered Shareholders and duly appointed proxyholders, including Beneficial Shareholders who have appointed themselves
as proxyholders, to participate in the Special Meeting and ask questions, all in real time. Registered Shareholders and duly appointed
proxyholders can vote at the appropriate times during the Special Meeting. Guests, including Beneficial Shareholders who have not
appointed themselves as proxyholders, can log into the Special Meeting as set out below. Guests can listen to the Special Meeting
, but are not able to vote or ask questions.
|
|
·
|
Log in online at https://web.lumiagm.com/157200202. We recommend that you log in at least one hour before the Special Meeting starts;
|
|
|
·
|
Click “Login” and then enter your 13-digit control number (see below) and password
“zomedica2020” (case sensitive); OR
|
|
|
·
|
Click “Guest” and then complete the online form.
|
For any technical difficulties or trouble accessing
our virtual-only Special Meeting , please go to: http://go.lumiglobal.com/faq.
Registered Shareholders: The 13-digit control number is located
on the instrument of proxy.
Duly appointed proxyholders: AST will provide the proxyholder
with a 13-digit control number after the proxy voting deadline has passed and the proxyholder has been duly appointed and registered
as described above.
It is necessary for a proxyholder to obtain
a control number in order to participate at the Special Meeting. Without a control number, the proxyholder will not be able to
vote or ask questions and vote at the Special Meeting. The proxyholder will only be able to participate as a guest.
If you participate in the Special Meeting online,
it is important that you are connected to the internet at all times during the Special Meeting in order to vote when balloting
commences. It is your responsibility to ensure connectivity for the duration of the Special Meeting. You should allow ample time
to check into the Special Meeting online and complete the Special Meeting procedures.
VOTING
SECURITIES AND PRINCIPAL HOLDERS OF VOTING SECURITIES
Voting Shares and Record Date
Our authorized share
capital consists of an unlimited number of common shares without nominal or par value and an unlimited number of preferred shares
without nominal or par value, of which 20 shares have been designated as Series 1 preferred shares. The record date for the determination
of shareholders entitled to receive notice of and to vote at the Special Meeting is October 22, 2020 (the “Record Date”).
As at the Record Date, there were 564,051,438 common shares issued and outstanding as fully paid and non-assessable and 12 Series
1 preferred shares issued and outstanding as fully paid and non-assessable.
Common Shares
The holders of common shares are entitled to
notice of and to vote at all special meetings of shareholders and are entitled to one vote per common share. Subject to the rights
of holders of any preferred shares, the holders of common shares are entitled to receive such dividends as the Board of Directors
declares and, upon liquidation, dissolution or winding-up, subject to the rights of holders of any preferred shares, to receive
such assets of our company as are distributable to holders of common shares.
Series 1 Preferred Shares
The holders of the Series 1 preferred shares
are not entitled to receive notice of, attend or vote at any meeting of the shareholders of the Corporation, subject to applicable
law, including the provisions of the ABCA. However, in accordance with Section 189(3) of the ABCA, each share of our company carries
the right to vote in respect the domestication whether or not it otherwise carries the right to vote. The holder of the Series
1 preferred shares is entitled to one vote per share on the domestication.
Voting of Shares – General
Only Registered Shareholders whose names are
entered in our register of shareholders at the close of business on the Record Date will be entitled to receive notice of and to
vote at the Special Meeting, provided that, to the extent that: (i) a Registered Shareholder has transferred the ownership of any
shares subsequent to the Record Date; and (ii) the transferee of those shares produces properly endorsed share certificates, or
otherwise establishes that he or she owns the shares and demands, not later than ten days before the Special Meeting, that his
or her name be included on the shareholder list before the Special Meeting, in which case the transferee shall be entitled to vote
his or her shares at the Meeting.
Votes Required
The holders of at least two thirds of our common
shares and Series 1 preferred shares present at the Special Meeting virtually or by proxy (and assuming a quorum of our outstanding
shares are represented at the Special Meeting virtually or by proxy) must vote together as a single class to approve the domestication
proposal. The holders of a majority of our common shares present at the Special Meeting virtually or by proxy (and assuming a quorum
of our outstanding common shares are represented at the Special Meeting virtually or by proxy) must vote to approve the incentive
plan proposal. No cumulative voting rights are authorized.
Effect of Abstentions and Broker Non-Votes
Under applicable United States laws, banks and
brokers acting as nominees are permitted to use discretionary voting authority to vote proxies for proposals that are deemed “routine”
by the New York Stock Exchange (the exchange that makes such determinations), but are not permitted to use discretionary voting
authority to vote proxies for proposals that are deemed “non-routine” by the New York Stock Exchange. A broker
“non-vote” occurs when a proposal is deemed “non-routine” and a nominee holding shares for a beneficial
owner does not have discretionary voting authority with respect to the matter being considered and has not received instructions
from the beneficial owner. The determination of which proposals are deemed “routine” versus “non-routine”
may not be made by the New York Stock Exchange until after the date on which this proxy circular/prospectus has been mailed
to you. As such, it is important that you provide voting instructions to your bank, broker or other nominee, if you wish to determine
the voting of your common shares.
However, under applicable Canadian securities
laws, banks, brokers and other Intermediaries are not permitted to vote common shares held on behalf of Beneficial Shareholders
except in accordance with voting instructions received from the applicable Beneficial Shareholder. Therefore, Intermediaries in
Canada are not permitted to exercise discretionary voting authority in relation to “non-routine” business. Further,
the Business Corporations Act (Alberta) (the “ABCA”), the statute pursuant to which our company has been incorporated,
provides that shares of a corporation that are registered to a registrant or a registrant’s nominee and are not beneficially
owned by the registrant are not permitted to be voted without instructions from the beneficial owner, and the registrant is not
permitted to appoint a proxyholder in respect of those shares without voting instructions from the beneficial owner.
Pursuant to the ABCA and applicable Canadian
securities laws, in connection with shareholder votes to be conducted at the Special Meeting, it is only necessary to count votes
cast “FOR” or “AGAINST” the applicable item of business. As a result, abstentions and broker non-votes,
if any, will be treated as votes not cast at the Special Meeting in relation to the particular item of business, and therefore
will not affect the outcome of the vote on the any of such items of business.
To our knowledge, no person owned more than 10% of our outstanding
common shares as of the Record Date. Wickfield Capital LLC and its affiliate, Wickfield Bridge Fund LLC, were the record owners
of all of our outstanding Series 1 preferred shares as of the Record Date. For additional beneficial ownership information, see
“Securities Ownership of Certain Beneficial Owners and Management.”
The
Domestication
General
The Board of Directors is proposing to
change our jurisdiction of incorporation from the Province of Alberta in Canada to the State of Delaware in the United States
through a transaction called a ‘‘continuance’’ under Section 189 of the ABCA and a ‘‘domestication’’
under Section 388 of the DGCL (collectively referred to as a ‘‘domestication’’ herein),
and approve a new certificate of incorporation to be effective on the date of the domestication. Upon the effectiveness of the
domestication, we will become subject to the DGCL on the date of our domestication, but will be deemed for the purposes of the
DGCL to have commenced our existence in Delaware on the date we originally commenced our existence in Canada. Under the DGCL,
a corporation becomes domesticated in Delaware by filing a certificate of corporate domestication and a certificate of incorporation
for the corporation being domesticated. The Board of Directors has unanimously approved our domestication and the related certificate
of incorporation of Zomedica Corp., believes it to be in our best interests, and unanimously recommends approval of the domestication
and the approval of the certificate of incorporation of Zomedica Corp. to our shareholders.
The domestication will be effective on the date
set forth in the certificate of corporate domestication and the certificate of incorporation, as filed with the office of the Secretary
of State of the State of Delaware. A copy of the certificate of corporate domestication is attached to this proxy circular/prospectus
as Exhibit B. Thereafter, we will be subject to the certificate of incorporation filed in Delaware, a copy of which is attached
to this proxy circular/prospectus as Exhibit C. We will be discontinued in Alberta as of the date shown on the certificate
of discontinuance issued by the Registrar appointed under the ABCA, which we expect to be the date of domestication in Delaware.
The domestication will not interrupt our corporate
existence or operations or the trading markets of our common shares. Upon consummation of the domestication, (i) the property of
Zomedica Corp. (the Alberta corporation) will continue to be the property of Zomedica Corp. (the Delaware corporation); (ii) Zomedica
Corp. (the Delaware corporation) will continue to be liable for the obligations of Zomedica Corp. (the Alberta corporation); (iii)
any existing cause of action, claim or liability to prosecution in respect of Zomedica Corp. (the Alberta corporation) shall be
unaffected; (iv) any civil, criminal or administrative action or proceeding pending by or against Zomedica Corp. (the Alberta corporation)
may be continued to be prosecuted by or against Zomedica Corp. (the Delaware corporation); and (v) any conviction against, or ruling,
order or judgment in favor of or against Zomedica Corp. (the Alberta corporation) may be enforced by or against Zomedica Corp (the
Delaware corporation). Each outstanding common share at the time of the domestication will remain issued and outstanding as a share
of common stock of Zomedica Corp. (the Delaware corporation) after our corporate existence is continued out of the Province of
Alberta in Canada under the ABCA and into the State of Delaware in the United States under the domestication procedures of the
DGCL. Our common shares are currently traded and will continue to be traded on the NYSE American exchange under the symbol ‘‘ZOM.’’
We will continue to be subject to the rules
and regulations of the NYSE American exchange. In addition, we will continue to file periodic reports with the SEC pursuant to
the Exchange Act. We will continue to make filings on SEDAR but, except in limited circumstances, those filings will consist of
the filings we make with the SEC. Upon our domestication, our Board of Directors intends to adopt bylaws, copies of which are attached
to this proxy circular/prospectus as Exhibit D. A copy of Section 191 of the ABCA addressing dissenters’ rights in
connection with the continuance is attached to this proxy circular/prospectus as Exhibit E.
Principal Reasons for the Domestication
We are pursuing the
domestication for a number of reasons. Our domestication is intended to reduce the regulatory burden and cost of being subject
to the laws and regulations of both the United States and Canada and to enhance shareholder value over the long term by, among
other things, reducing our operating costs and enhancing the marketability of our capital stock by becoming a Delaware corporation.
In addition, our corporate offices and operations are located in the United States and a large percentage of our shareholders are
located there. We believe that the domestication will save approximately $500,000 per year in duplicative, unnecessary compliance
costs, which will directly benefit shareholders.
Our Board of Directors chose the State of Delaware
to be our domicile because the DGCL expressly accommodates continuances under Section 189 of the ABCA and because the provisions
of the DGCL, specifically those dealing with the duties and obligations of directors of a Delaware corporation, have been interpreted
by a substantial body of case law in the Delaware courts.
In considering its recommendation in favor of
the domestication, our Board of Directors weighed the estimated Canadian income tax liability to us arising from this transaction.
See ‘‘The Domestication—United States and Canadian Income Tax Considerations.’’
Effects of the Domestication
There are material differences between Alberta
corporate law and Delaware corporate law with respect to shareholders rights, and Delaware law may offer shareholders more or less
protection depending on the particular matter. A detailed overview of the material differences is set forth below.
Applicable Law. As of the effective date
of the domestication, our legal jurisdiction of incorporation will be Delaware, and we will no longer be subject to the provisions
of the ABCA. All matters of corporate law will be determined under the DGCL. We will retain our original incorporation date in
Alberta as our date of incorporation for purposes of the DGCL.
Assets, Liabilities, Obligations, Etc.
Under Delaware law, as of the effective date of the domestication, all of our assets, property, rights, liabilities and obligations
immediately prior to the domestication will continue to be our assets, property, rights, liabilities and obligations. Alberta
corporate law will cease to apply to us on the date shown on the certificate of discontinuance to be issued by the Registrar appointed
under the ABCA, which we expect to be the date of domestication in Delaware. We will thereafter be subject to the obligations
imposed under Delaware corporate law.
Business and Operations. The domestication,
if approved, will effect a change in the legal jurisdiction of incorporation as of the effective date of the domestication, but
our business and operations will remain the same.
Officers and Directors. Our Board currently
consists of six members: Robert Cohen, Jeffrey Rowe, Rodney Williams, Johnny D. Powers, Chris MacLeod and Christopher Wolfenberg.
These directors will continue to serve as directors from and after the effective date of the domestication. Immediately following
the domestication, our officers will also be unchanged. Our officers are Robert Cohen, Interim Chief Executive Officer, Ann Cotter,
Chief Financial Officer, Stephanie Morley, President and Chief Medical Officer, and Bruk Herbst, Chief Commercial Officer.
As of the completion of the domestication,
the board of directors of Zomedica Corp. will be divided into three classes of directors, each serving staggered three-year terms.
Upon expiration of the term of a class of directors, directors for that class will be elected for three-year terms at the annual
meeting of stockholders in the year in which that term expires. Each director’s term continues until the election and qualification
of his successor, or his earlier death, resignation or removal. Subject to the rights of holders of any series of preferred stock
to elect directors, the authorized number of the directors will be fixed from time to time by resolution of the board of directors.
No decrease in the authorized number of directors will have the effect of removing an incumbent director from the board of directors
until the incumbent director’s term expires. Any increase or decrease in the number of directors will be distributed among
the three classes so that, as nearly as possible, each class will consist of one-third of the directors.
Treatment of Outstanding Capital Stock, Warrants
and Options
The existing share certificates representing
our common shares and Series 1 preferred shares will continue to represent the same number of the same class of our common stock
and Series 1 Preferred Stock after the domestication without any action on your part. You will not be required to exchange any
share certificates. We will only issue new certificates to you representing shares of capital stock of Zomedica Corp. upon a transfer
of your shares or at your request. Holders of our outstanding warrants and options will continue to hold the same securities, which
will remain exercisable for an equivalent number of shares of the same class of common stock of Zomedica Corp., for the equivalent
exercise price per share, without any action by the holder, in accordance with their terms.
Shareholder Approval
The domestication is subject to various conditions,
including approval of the special resolution authorizing the domestication and the approval of the certificate of incorporation
of Zomedica Corp. by our shareholders at the Special Meeting. A copy of the special resolution is attached to this proxy circular/prospectus
as Exhibit A. Under the ABCA, the change of jurisdiction requires affirmative votes, whether virtually or by proxy, of at
least two thirds of the votes cast by the holders of all of our shares whether or not such shares otherwise carry the right to
vote, consisting of the sole holder of our Series 1 preferred shares and the holders of our common shares at the Special Meeting
where a quorum is present, being at least two persons present and holding or representing by proxy not less than 25% of the outstanding
shares entitled to be voted at the Special Meeting. Assuming we receive the requisite shareholder approval for the domestication,
our Board of Directors will retain the right to terminate or abandon the domestication if it determines that consummating the domestication
would be inadvisable or not in our best interests, or if all of the respective conditions to consummation of the domestication
have not occurred, without further approval of the shareholders. There are no time limits on the duration of the authorization
resulting from a favorable shareholder vote.
Regulatory and Other Approvals
The change of jurisdiction is subject to the
authorization of the Registrar of Corporations appointed under the ABCA, or the Registrar. The Registrar is empowered to authorize
the change of jurisdiction if, among other things, the Registrar is satisfied that the change of jurisdiction will not adversely
affect our creditors or shareholders.
Subject to the authorization of the continuance
by the Registrar appointed under the ABCA and the approval of our shareholders, we anticipate that we will file a certificate of
corporate domestication and a certificate of incorporation with the Secretary of State of the State of Delaware pursuant to the
DGCL promptly following the Special Meeting, and that we will be domesticated in Delaware on the effective date of such filings.
Promptly thereafter, we intend to give notice to the Registrar appointed under the ABCA that we have been domesticated under the
laws of the State of Delaware and request that the Registrar of Corporations appointed under the ABCA issue us a certificate of
discontinuance bearing the same date as the date of effectiveness of our certificate of corporate domestication and certificate
of incorporation by the Secretary of State of the State of Delaware.
Comparison of Shareholder Rights
The principal attributes of our capital stock
before and after domestication are comparable; however, there will be material differences in the rights of our shareholders under
Delaware law.
On the effective date of the domestication,
we will be deemed, for purposes of the DGCL, to have been incorporated under the laws of the State of Delaware from our inception
and we will be governed by the DGCL and the certificate of incorporation filed with the certificate of corporate domestication.
Differences between Alberta corporate law and Delaware corporate law and between our current articles of incorporation and bylaws
and the proposed Delaware certificate of incorporation and bylaws will result in various changes in the rights of our shareholders.
The following summary comparison highlights provisions of applicable Alberta corporate law and our current Alberta articles of
incorporation and bylaws and Delaware corporate law and the proposed certificate of incorporation and bylaws of Zomedica Corp.
The proposed certificate of incorporation and bylaws of Zomedica Corp. are attached to this proxy circular/prospectus as Exhibit
C and Exhibit D, respectively. This summary is qualified in its entirety by our governing corporate instruments.
Capital Structure. Under our current
Alberta articles of incorporation, we have the authority to issue an unlimited number of common shares and an unlimited number
of preferred shares. Under Alberta law, there is no franchise tax on our issued and outstanding common shares and preferred shares.
Under our proposed Delaware certificate of incorporation, Zomedica Corp. will have the authority to issue a total of 2,010,000,000
shares of capital stock, consisting of 2,000,000,000 shares of common stock, $0.0001 par value per share, and 10,000,000 shares
of preferred stock, $0.0001 par value per share. Pursuant to Delaware law, Zomedica Corp. will initially be required to pay an
annual corporate franchise tax of approximately $50,000 in order to maintain its good standing as a Delaware corporation. The amount
of that annual franchise tax could change if Zomedica Corp.’s gross assets or capital structure changes.
Shareholder Approval; Vote on Extraordinary
Corporate Transactions. Under the ABCA, certain extraordinary corporate actions, such as a name change, amalgamations (other
than with certain affiliated corporations), continuances to another jurisdiction and sales, leases or exchanges of all, or substantially
all, of the property of a corporation (other than in the ordinary course of business), and other extraordinary corporate actions
such as liquidations, dissolutions and arrangements (if ordered by a court), among others, are required to be approved by a ‘‘special
resolution’’ of shareholders.
A ‘‘special resolution’’
is a resolution (1) passed by not less than two-thirds of the votes cast by the shareholders who voted in respect of the resolution
at a meeting duly called and held for that purpose or (2) signed by all shareholders entitled to vote on the resolution. In specified
cases, a special resolution to approve an extraordinary corporate action is also required to be approved separately by the holders
of a class or series of shares, including in certain cases a class or series of shares not otherwise carrying voting rights (unless
in certain cases the share provisions with respect to such class or series of shares provide otherwise).
The DGCL generally requires the affirmative vote of a majority of the
outstanding stock entitled to vote thereon to authorize any merger, consolidation, dissolution or sale of substantially all of
the assets of a corporation, except that no authorizing stockholder vote is required of a corporation surviving a merger if (a)
such corporation’s certificate of incorporation is not amended in any respect by the merger, (b) each share of stock of such
corporation outstanding immediately prior to the effective date of the merger will be an identical outstanding or treasury share
of the surviving corporation after the effective date of the merger and (c) the number of shares to be issued in the merger, together
with the number of shares initially issuable upon conversion of any other shares, securities or obligations issued or delivered
in the merger, does not exceed 20 percent of such corporation’s outstanding common stock immediately prior to the effective
date of the merger. Stockholder approval is also not required under the DGCL in certain circumstances in which a parent corporation
merges or consolidates with a subsidiary of which it owns at least 90 percent of the outstanding shares of each class of stock.
Finally, unless required by its certificate of incorporation, stockholder approval is not required under the DGCL for a corporation
to merge with or into a direct or indirect wholly owned subsidiary of a holding company (as defined in the DGCL) in certain circumstances.
Stockholder approval is also not required under certain circumstances in connection with a merger agreement involving a corporation
listed on a national securities exchange or held of record by more than 2,000 holders where an acquirer consummates a tender offer
that would result in the acquirer owning sufficient shares to approve the merger and certain other requirements are met.
Amendments to the Governing Documents.
Under the ABCA, amendments to the articles of incorporation generally requires approval by special resolution of the shareholders
entitled to vote thereon. If the proposed amendment would affect a particular class of securities in certain specified ways, the
holders of shares of that class would be entitled to vote separately as a class on the proposed amendment, whether or not the shares
otherwise carry the right to vote.
Unless the articles, bylaws or a unanimous
shareholders agreement otherwise provides, the ABCA allows the directors, by resolution, to make, amend or repeal any bylaws that
regulate the business or affairs of the corporation. The bylaw amendment must be ratified by the shareholders at the next meeting
of shareholders. At the meeting, the shareholders may confirm, reject or amend the bylaw amendment or repeal, as applicable.
If a bylaw, or an amendment or a repeal of a
bylaw, is rejected by the shareholders, or if the directors do not submit a bylaw, or an amendment or a repeal of a bylaw, to the
shareholders, the bylaw, amendment or repeal ceases to be effective and no subsequent resolution of the directors to make, amend
or repeal a bylaw having substantially the same purpose or effect is effective until it is approved by the shareholders.
The DGCL requires a vote of the corporation’s
board of directors followed by the affirmative vote of a majority of the outstanding stock of entitled to vote thereon for any
amendment to the certificate of incorporation. If an amendment increases or decreases the aggregate number of authorized shares
of a class, increases or decreases the par value of the shares of such class, or alters the powers, preferences or special rights
of a particular class of stock so as to affect them adversely, that class shall be given the power to vote as a class on the amendment,
whether or not entitled to vote thereon by the certificate of incorporation. If an amendment would alter or change the powers,
preferences or special rights of one or more series of any class of stock so as to affect them adversely, but shall not so affect
the entire class, then only the shares of the series so affected must approve the amendment as a class even if the certificate
of incorporation does not provide that right. The DGCL also states that the power to adopt, amend or repeal the by-laws of a corporation
shall be in the stockholders entitled to vote, provided that the corporation in its certificate of incorporation may confer such
power on the board of directors in addition to the stockholders. The proposed certificate of incorporation will confer such power
on Zomedica Corp.’s board of directors.
Place of Meetings. Pursuant to the ABCA,
if the articles of the corporation so provide, meetings of shareholders may be held outside of Alberta. Our current articles provide
that meetings of shareholders may be held outside of Alberta as the Board so determines.
The DGCL provides that meetings of the
stockholders be held at any place in or outside of Delaware designated by, or in the manner provided in, the certificate of incorporation,
bylaws or, if not so designated, as may be determined by the board of directors. The proposed bylaws of Zomedica Corp. provide
that meetings of the stockholders may be held at any place designated by the Board of Directors and may also be held solely by
means of electronic communication.
Quorum of Shareholders. The ABCA provides
that, unless the bylaws provide otherwise, a quorum of shareholders is present at a meeting of shareholders (irrespective of the
number of persons actually present at the meeting) if holders of a majority of the shares entitled to vote at the meeting are present
in person or represented by proxy. The current bylaws provide that a quorum is present if there are at least two persons present
and holding or representing by proxy not less than 25% of the outstanding common shares entitled to be voted at the meeting.
Under the DGCL, the certificate of incorporation
or bylaws may specify the number of shares the holders of which shall be present or represented by proxy at any meeting in order
to constitute a quorum, but in no event shall a quorum consist of less than one-third of the shares of such class or series or
classes or series. In the absence of such specification in the certificate of incorporation or bylaws, a majority of the shares
entitled to vote, present in person or represented by proxy, shall constitute a quorum. The proposed bylaws of Zomedica Corp.
provide that the holders of not less than one-third of the voting power of the outstanding shares entitled to vote at the meeting
shall constitute a quorum at a meeting of stockholders.
Calling Meetings. The ABCA provides that
the directors shall call an annual meeting of shareholders not later than 15 months after the last preceding annual meeting, and
may at any time call a special meeting of shareholders. The registered holders or beneficial owners of not less than 5% of the
issued shares of a corporation that carry the right to vote at a meeting sought to be held may requisition the directors to call
a meeting of shareholders for the purposes stated in the requisition, but the beneficial owners of shares do not hereby acquire
the direct right to vote at the meeting that is the subject of the requisition.
The DGCL provides that a special meeting of
the stockholders may be called by the board of directors of the corporation or by any person or persons as may be authorized by
the certificate of incorporation or bylaws of the corporation. The certificate of incorporation of Zomedica Corp. will provide
that a special meeting of stockholders may be called by the board of directors, by the chairman of the board of directors or by
the chief executive officer and may not be called by another person or persons.
Shareholder Consent in Lieu of Meeting.
Under the ABCA, a resolution in writing signed by all of the shareholders entitled to vote on that resolution is as valid as if
it had been passed at a meeting of shareholders. Under the DGCL, unless otherwise limited by the certificate of incorporation,
stockholders may act by written consent without a meeting if holders of outstanding stock representing not less than the minimum
number of votes that would be necessary to authorize or take the action at an annual or special meeting at which all shares entitled
to vote thereon were present and voted execute and deliver a written consent providing for the action. The certificate of incorporation
of Zomedica Corp. will specifically require stockholders to take action only at a meeting of stockholders.
Advance Notice Provisions for Shareholder
Nominations. The ABCA does not require advance notice for shareholder nominations, but an Alberta corporation may require such
advance notice pursuant to its by-laws if approved by shareholders.
Our bylaws provide that any shareholder wishing
to nominate a person for election to our board of directors must provide notice to our secretary, in the case of an annual meeting
of shareholders, not less than 30 nor more than 65 days prior to the date of the annual meeting of shareholders; provided, however,
that in the event that the annual meeting of shareholders is to be held on a date that is less than 50 days after the date (the
“Notice Date”) on which the first public announcement of the date of the annual meeting was made, notice by the nominating
shareholder may be made not later than the close of business on the tenth day following the Notice Date; and (ii) in the case of
a special meeting (which is not also an annual meeting) of shareholders called for the purpose of electing directors (whether or
not called for other purposes), not later than the close of business on the 15th day following the day on which the first public
announcement of the date of the special meeting of shareholders was made. Any such notice must also contain the information and
comply with the other procedural requirements of the bylaws.
The DGCL does not require advance notice
for stockholder nominations or other business, but a Delaware corporation may require such advance notice pursuant to its
by-laws.
Under the proposed bylaws of Zomedica Corp.,
any stockholder nominations or proposals may only be made at the annual meeting of stockholders if, among other things, timely
notice thereof is provided in writing to the secretary of Zomedica Corp. To be timely, a stockholder’s notice shall be delivered
to the secretary of Zomedica Corp. at the principal executive offices of Zomedica Corp. not less than ninety (90) days nor more
than one hundred and twenty (120) days prior to the first anniversary of the preceding year’s annual meeting; provided,
however, that in the event that the date of the annual meeting is advanced by more than thirty (30) days, or delayed by more than
seventy (70) days, from the anniversary date of the previous year’s meeting, notice by the stockholder to be timely must
be so delivered not earlier than one hundred and twenty (120) days prior to such annual meeting and not later than the close of
business on the later of the ninetieth (90th) day prior to such annual meeting or the tenth (10th) day following the day on which
public announcement of the date of such meeting is first made. Any such notice must also contain the information and comply with
the other procedural requirements of the bylaws.
Director Election, Qualification and Number.
The ABCA provides for the election of directors by a majority of votes cast at an annual meeting of shareholders. The ABCA states
that a corporation shall have one or more directors but a distributing corporation whose shares are held by more than one person
shall have not fewer than 3 directors, at least 2 of whom are not officers or employees of the corporation or its affiliates. Additionally,
at least one fourth of the directors must be Canadian residents unless the corporation has fewer than four directors, in which
case at least one director must be a Canadian resident.
As of the completion of the domestication, the
certificate of incorporation of Zomedica Corp. will provide that the board of directors will be divided into three classes, Class
I, Class II and Class III. Our Class I directors, Mr. MacLeod and Mr. Wolfenberg, will stand for reelection at our 2021 annual
meeting of stockholders, our Class II directors, Mr. Williams and Dr. Powers, will stand for reelection at our 2022 annual meeting
of stockholders and our Class III directors, Mr. Rowe and Mr. Cohen, will stand for reelection at our 2023 annual meeting of stockholders.
The DGCL has no requirement that directors have
specific qualifications or be independent of the corporation; however, the governance standards of the NYSE American require a
majority of a listed company’s directors to be independent.
Vacancies on Board of Directors. Under
the ABCA, a vacancy among the directors created by the removal of a director may be filled at a meeting of shareholders at which
the director is removed. The ABCA also allows a vacancy on the board to be filled by a quorum of directors, except when the vacancy
results from an increase in the number of minimum number of directors or the vacancy is a result of a failure to elect the number
or minimum number of directors required by the articles. In addition, the ABCA authorizes the directors to, if the articles so
provide, between annual general meetings, appoint one or more additional directors of the corporation to serve until the next annual
general meeting, so long as the number of additional directors shall not at any time exceed 1/3 of the number of directors who
held office at the expiration of the last annual meeting of the corporation.
Delaware law provides that a vacancy or a newly
created directorship may be filled by a majority of the directors then in office, although less than a quorum, or by the sole remaining
director, unless otherwise provided in the certificate of incorporation or bylaws. The proposed certificate of incorporation of
Zomedica Corp. will provide that, subject to the rights of holders of any series of preferred stock, any newly created directorship
or any vacancy on the board of directors may be filled solely by the affirmative vote of a majority of the total number of directors
then in office, even if less than a quorum, or by a sole remaining director and may not be filled by the stockholders. Any director
so elected will, in the case of a newly created directorship, hold office for the full term of the class in which the newly created
directorship was created or, in the case of a vacancy, hold office for the remaining term of his or her predecessor and in each
case until his or her successor is elected and qualified, subject to his or her earlier death, disqualification, resignation or
removal.
Removal of Directors; Terms of Directors.
Under the ABCA, provided that the articles of a corporation do not provide for cumulative voting, and subject to a unanimous shareholder
agreement, shareholders of the corporation may, by ordinary resolution passed at a special meeting, remove any director or directors
from office. If holders of a class or series of shares have the exclusive right to elect one or more directors, a director elected
by them may only be removed by an ‘‘ordinary resolution’’ at a meeting of the shareholders of that class
or series.
An ‘‘ordinary resolution’’
means a resolution (1) passed by a majority of the votes cast by the shareholders who voted in respect of that resolution, or (2)
signed by all the shareholders entitled to vote on that resolution.
As of the completion of the domestication, the
certificate of incorporation of Zomedica Corp. will provide that the board of directors will be divided into three classes, Class
I, Class II and Class III. Class I directors will stand for reelection at our 2021 annual meeting of stockholders, Class II directors
will stand for reelection at our 2022 annual meeting of stockholders and Class III directors will stand for reelection at our 2023
annual meeting of stockholders. Each Class of directors will thereafter be elected for a three-year term.
Under the proposed certificate of incorporation
of Zomedica Corp., directors may only be removed for cause Delaware by the holders of a majority of the shares entitled to vote
at an election of directors.
Fiduciary Duty of Directors. Directors
of a corporation incorporated under the ABCA have fiduciary obligations to the corporation. The ABCA requires directors and officers
of an Alberta corporation, in exercising their powers and discharging their duties, to act honestly and in good faith with a view
to the best interests of the corporation and exercise the care, diligence and skill that a reasonably prudent person would exercise
in comparable circumstances.
The fiduciary obligations of directors
of corporations incorporated or organized under the DGCL are more expansive and run not just to the corporation but to the corporation’s
stockholders. These obligations fall into two broad categories: a duty of care and a duty of loyalty. The duty of care requires
a director to act with the care an ordinarily prudent person in a like position would exercise under similar circumstances and
requires that directors inform themselves prior to making a business decision, of all material information reasonably available
to them. The duty of loyalty requires directors to act in good faith and to exercise their powers in the interests of the corporation
and all of its stockholders and not in the directors’ own interests or in the interest of another person (including a family
member) or organization. Stated more simply, the duty of loyalty precludes directors from using their corporate position to make
a personal profit or gain, or for other personal advantage. The business judgment rule protects decisions made by disinterested
directors and presumes that in making a business decision the directors of a corporation acted on an informed basis, in good faith,
and in the honest belief that the action taken was in the best interests of the corporation. Where the business judgment rule
applies a court will not second guess the decision by a board of directors.
The personal liability of a director
under the DGCL for breach of his or her fiduciary duty may be expansive and can be established by the corporation, through a derivative
action brought on behalf of the stockholders, or by an aggrieved stockholder, in a separate action. The proposed certificate of
incorporation of Zomedica Corp. seeks to limit directors’ personal liability for actions that may be judged to be in violation
of their fiduciary duties; however, these limitations are not effective with respect to the following proscribed conduct under
the DGCL:
|
|
·
|
any breach of the director’s duty of loyalty to the corporation or its stockholders;
|
|
|
·
|
acts or omissions not in good faith or that involve intentional misconduct or a knowing violation
of law;
|
|
|
·
|
an unlawful payment of a dividend or an unlawful stock purchase or redemption as provided in Section 174 of the DGCL; and
|
|
|
·
|
any transaction from which the director derived an improper personal benefit.
|
Indemnification of Officers and Directors.
Under the ABCA and pursuant to our current bylaws, we will indemnify present or former directors or officers against all costs,
charges and expenses, including an amount paid to settle an action or satisfy a judgment that is reasonably incurred by the individual
in relation to any civil, criminal, administrative, investigative or other proceeding in which the individual is involved because
of his or her association with us. In order to qualify for indemnification such directors or officers must:
|
|
1)
|
have acted honestly and in good faith with a view to the best interests of the corporation; and
|
|
|
2)
|
in the case of a criminal or administrative action or proceeding enforced by a monetary penalty,
have had reasonable grounds for believing that his conduct was lawful.
|
We currently carry liability insurance for our
company’s and its subsidiaries’ officers and directors. This will continue following the domestication.
The ABCA also provides that such persons are
entitled to indemnity from the corporation in respect of all costs, charges and expenses reasonably incurred in connection with
the defense of any such proceeding if the person seeking the indemnity was substantially successful on the merits in the person’s
defense of the action or proceeding, is fairly and reasonably entitled to the indemnity, and otherwise meets the qualifications
for indemnity described in 1) and 2) above. The corporation may advance funds to such persons in order to defray the costs, charges
and expenses of a proceeding, but if the person does not meet the conditions described in the preceding sentence he or she shall
repay the funds advanced.
The DGCL permits a corporation to indemnify
any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action, suit or proceeding,
whether civil, criminal, administrative or investigative (other than an action by or in the right of the corporation) by reason
of the fact that the person is or was a director, officer, employee or agent of the corporation, or is or was serving at the request
of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other
enterprise, against expenses (including attorneys’ fees), judgments, fines and amounts paid in settlement actually and reasonably
incurred by the person in connection with such action, suit or proceeding if the person acted in good faith and in a manner he
or she reasonably believed to be in (or not opposed to) the best interests of the corporation and, with respect to any criminal
action, had no reasonable cause to believe the conduct was unlawful. In derivative actions, however, the DGCL only permits indemnification
against expenses (including attorneys’ fees) actually and reasonably incurred by the person, except that no indemnification
may be made with respect to any claim, issue or matter as to which such person is adjudged to be liable to the corporation (unless
otherwise determined by the court in which such action was brought). A corporation must indemnify an officer or director “to
the extent” the person is successful on the merits or otherwise in defense of an action, suit or proceeding. The proposed
bylaws of Zomedica Corp. will generally provide for the indemnification of the corporation’s officers and directors and
former officers and directors to the fullest extent permitted by Delaware law, as the same may be amended from time to time.
The DGCL allows the corporation to advance
expenses before the resolution of an action, if an undertaking is provided by or on behalf of the person seeking advancement to
repay any such amount advanced if they are later determined not to be entitled to indemnification. In addition, Delaware law authorizes
a corporation to purchase insurance for the benefit of its officers and directors whether or not the corporation would have the
power to indemnify against the liability covered by the policy.
Dissent or Dissenters’ Appraisal Rights.
The ABCA provides that shareholders of a corporation entitled to vote on certain matters are entitled to exercise dissent rights
and demand payment for the fair value of their shares, determined as of the close of business on the last business day on which
the resolution from which the shareholder dissents was adopted, in connection with specified matters, including, among others:
|
|
·
|
an amendment to our articles of incorporation to add, change or remove any provisions restricting
the issue or transfer of shares;
|
|
|
·
|
amend our articles to add, change or remove any restrictions on the business or businesses that
the corporation may carry on;
|
|
|
·
|
amend our articles to add or remove an express statement establishing the unlimited liability of the shareholders;
|
|
|
·
|
any amalgamation with another corporation (other than with certain affiliated corporations);
|
|
|
·
|
a continuance under the laws of another jurisdiction; and
|
|
|
·
|
a sale, lease or exchange of all or substantially all the property of the corporation other than
in the ordinary course of business.
|
However, a shareholder is not entitled to dissent
if an amendment to the articles is effected by a court order approving a reorganization or by a court order made in connection
with an action for an oppression remedy.
Under the DGCL, record holders of shares
of any class or series have the right, in certain circumstances, to be paid in cash the fair value of their shares as appraised
by the Delaware Court of Chancery as of the effective time of a merger or consolidation exclusive of any element of value arising
from the accomplishment or expectation of the merger or consolidation. The DGCL grants appraisal rights only in the case of mergers
or consolidations and not in the case of a sale or transfer of assets or a purchase of assets for stock regardless of the number
of shares being issued, and does not grant dissenters’ rights in connection with amendments to the certificate of incorporation.
Further, no appraisal rights are available for (i) shares of any class or series listed on a national securities exchange or held
of record by more than 2,000 shareholders or (ii) any shares of stock of a surviving corporation if the merger did not require
for its approval the vote of the stockholders of the surviving corporation as provided in Section 251(f) of the DGCL, provided,
however, that notwithstanding the foregoing, appraisal rights are available for the shares of any class or series of stock if
the holders thereof are required by the terms of the agreement of merger or consolidation to accept for such stock anything other
than (a) stock of the surviving corporation (or depositary receipts in respect thereof), (b) stock of another corporation which
is either listed on a national securities exchange or held of record by more than 2,000 shareholders, (c) cash in lieu of fractional
shares, or (d) some combination of the above. The DGCL does not provide stockholders of a corporation with appraisal rights when
the corporation acquires another business through the issuance of its stock (i) in exchange for the assets of the business to
be acquired, (ii) in exchange for the outstanding stock of the corporation to be acquired, or (iii) in a merger of the corporation
to be acquired with a subsidiary of the acquiring corporation.
Oppression
Remedy.
The ABCA provides an oppression remedy that
enables a court to make any order, whether interim or final, to rectify matters that are oppressive or unfairly prejudicial to
or that unfairly disregard the interests of any security holder, creditor, director or officer of the corporation if an application
is made to a court by a ‘‘complainant.’’
A ‘‘complainant’’ with
respect to a corporation means any of the following:
|
|
·
|
a present or former registered holder or beneficial owner of a security of the corporation or any
of its affiliates,
|
|
|
·
|
a present or former director or officer of the corporation or of any of its affiliates,
|
|
|
·
|
a creditor in respect of an application under a derivative action; or
|
|
|
·
|
any other person who, in the discretion of the court, is a proper person to make the application.
|
The oppression remedy provides the court with
very broad and flexible powers to intervene in corporate affairs to protect shareholders and other complainants. While conduct
that is in breach of fiduciary duties of directors or that is contrary to the legal right of a complainant will normally trigger
the court’s jurisdiction under the oppression remedy, the exercise of that jurisdiction does not depend on a finding of a
breach of those legal and equitable rights.
The DGCL does not provide for an oppression
remedy. However, the DGCL provides a variety of legal and equitable remedies to a corporation’s stockholders for improper
acts or omissions of a corporation, its officers and directors. Under the DGCL, generally, only stockholders can bring an action
alleging a breach of fiduciary duty by the directors of a corporation.
Derivative Actions. Under the ABCA, a
complainant may also apply to the court for permission to bring an action in the name of, and on behalf of, the corporation or
any of its subsidiaries, or to intervene in an existing action to which the corporation or its subsidiary is a party, for the purpose
of prosecuting, defending or discontinuing an action on the corporation’s behalf or on behalf of its subsidiary. Under the
ABCA, no action may be brought and no intervention in an action may be made unless a court is satisfied that:
|
|
(1)
|
the complainant has given reasonable notice to the directors of the corporation or its subsidiary
of the complainant’s intention to apply to the court if the directors of the corporation or its subsidiary do not bring,
diligently prosecute, defend or discontinue the action,
|
|
|
(2)
|
the complainant is acting in good faith, and
|
|
|
(3)
|
it appears to be in the interests of the corporation or its subsidiary that the action be brought,
prosecuted, defended or discontinued.
|
Under the ABCA, the court in a derivative action
may make any order it sees fit including orders pertaining to the control or conduct of the lawsuit by the complainant or the making
of payments to former and present shareholders and payment of reasonable legal fees incurred by the complainant.
Similarly, under Delaware law a stockholder
may bring a derivative action on behalf of the corporation to enforce a corporate right, including the breach of a director’s
duty to the corporation. Delaware law requires, among other things, that the plaintiff in a derivative suit be a stockholder of
the corporation at the time of the wrong complained of and remain so throughout the duration of the suit; and that the plaintiff
make a demand on the directors of the corporation to assert the corporate claim or assert that demand would be futile.
Business Combinations. Section 203 of
the DGCL provides, with some exceptions, that a Delaware corporation may not engage in any business combination with a person,
or an affiliate or associate of such person, who is an interested stockholder for three years from the time that person became
an interested stockholder unless:
|
|
·
|
prior to such time, the board of directors approved either the business combination or transaction which resulted in the stockholder
becoming an “interested stockholder”;
|
|
|
·
|
upon consummation of the transaction that resulted in the stockholder becoming an ‘‘interested
stockholder,’’ the ‘‘interested stockholder’’ owned at least 85% of a Delaware corporation’s
outstanding voting stock at the time the transaction commenced, excluding for purposes of determining the number of shares outstanding
those shares owned (i) by persons who are directors and are also officers and (ii) employee stock plans in which the participants
do not have the right to determine confidentially whether shares held subject to the plans will be tendered in the tender or exchange
offer; or
|
|
|
·
|
at or subsequent to such time, the business combination is approved by the board of directors and authorized at an annual or special
meeting of stockholders, and not by written consent, by the affirmative vote of at least two-thirds of the outstanding voting stock not
owned by the ‘‘interested stockholder.’’
|
A ‘‘business combination’’
is defined to include mergers, asset sales and other transactions resulting in financial benefit to a stockholder. In general,
an ‘‘interested stockholder’’ is a person who, together with affiliates and associates, owns 15% or more
of a corporation’s voting stock or within three years did own 15% or more of a corporation’s voting stock.
A corporation may, at its option, exclude itself
from the coverage of Section 203 by an appropriate provision in its certificate of incorporation. The proposed certificate of incorporation
of Zomedica Corp. does not contain such an exclusion.
Although there is no comparable provision relating
to business combinations under the ABCA, rules prescribing required approval thresholds and disclosure in respect of transaction
that constitute a “business combination” or a “related party transaction” do exist under applicable Canadian
securities laws.
Specifically, Multilateral Instrument 61-101—Protection
of Minority Security Holders in Special Transactions (‘‘MI 61-101’’) which is applicable to reporting
issuers in Canada, including our company, contains detailed requirements in connection with transactions that constitute a “business
combination” or a “related party transaction” (among others). A ‘‘business combination” as
defined under MI 61-101 means, generally, any amalgamation, arrangement, consolidation, amendment to the terms of a class of equity
securities, or any other transaction by an issuer as a consequence of which the interest of a holder of an equity security of an
issuer may be terminated without the holder’s consent, regardless of whether the equity security is replaced with another
security, with some exceptions. A ‘‘related party transaction’’ as defined under MI 61-101 means, generally,
any transaction by which an issuer, directly or indirectly, consummates one or more specified transactions with a related party,
including purchasing or disposing of an asset, issuing securities or assuming liabilities. A ‘‘related party’’
as defined in MI 61-101 generally includes (1) directors and senior officers of the issuer; (2) holders of voting securities of
the issuer carrying more than 10% of the voting rights attached to all the issuer’s outstanding voting securities; and (3)
holders of a sufficient number of any securities of the issuer to materially affect control of the issuer.
MI 61-101 requires, subject to certain exceptions,
specific detailed disclosure in the proxy circular sent to securityholders in connection with the consideration of a business combination
or a related party transaction and, subject to certain exceptions, the preparation of a formal valuation of the subject matter
of the transaction, and the inclusion of a summary of the valuation in the proxy circular. MI 61-101 also requires, subject to
certain exceptions, that an issuer not engage in a business combination or a related party transaction unless the disinterested
shareholders of the issuer have approved the transaction by a simple majority of the votes cast.
Examination of Corporate Records. Under
the ABCA, upon payment of a reasonable fee, a person is entitled during usual business hours to examine certain corporate records,
such as the securities register and a list of shareholders, and to make copies of or extracts from such documents.
Under Delaware law, for any proper purpose,
stockholders have the right to inspect, upon written demand under oath stating the purpose for such inspection, the corporation’s
stock ledger, list of stockholders and other specified books and records, among other things, and to make copies or extracts of
the same. A proper purpose means a purpose reasonably related to a person’s interest as a stockholder.
Anti-Takeover Effects. Some powers granted
to companies under Delaware law may allow a Delaware corporation to make itself potentially less vulnerable to hostile takeover
attempts. These powers include the ability to:
|
|
·
|
implement a classified board of directors, which prevents an immediate change in control of the
board;
|
|
|
·
|
require that notice of nominations for directors be given to the corporation prior to a meeting
where directors will be elected, which may give management an opportunity to make a greater effort to solicit its own proxies;
|
|
|
·
|
only allow the board of directors to call a special meeting of stockholders, which may deny an activist stockholder the ability to call a meeting
to make disruptive changes;
|
|
|
·
|
eliminate stockholders’ ability to act by written consent, which would require prior notice of any action proposed to be taken by the stockholders
of the corporation;
|
|
|
·
|
remove a director from a classified board only for cause, which gives some protection to directors
on a staggered board from arbitrary removal;
|
|
|
·
|
provide that the power to determine the number of directors and to fill vacancies be vested solely
in the board, so that the incumbent board, not a raider, would control vacant board positions;
|
|
|
·
|
provide for supermajority voting in some circumstances, including mergers and amendments to the certificate of incorporation
and bylaws; and
|
|
|
·
|
issue ‘‘blank check’’ preferred stock, which may be used to make a corporation
less attractive to a raider.
|
The proposed certificate of incorporation and
bylaws of Zomedica Corp. include the following provisions, which may discourage a hostile takeover attempt:
|
|
·
|
a requirement that stockholders provide prior notice to nominate directors or propose other action
at the annual meeting of stockholders;
|
|
|
·
|
a prohibition on the ability of stockholders to call a special meeting of stockholders;
|
|
|
·
|
an elimination of the ability of stockholders to take action by written consent;
|
|
|
·
|
provisions that give the board of directors the sole authority to determine the number of directors
and fill newly created directorships and vacancies on the board of directors; and
|
|
|
·
|
provisions that give the board of directors the authority to issue preferred stock having rights, privileges, limitations and other
characteristics that are established by the board of directors without stockholder approval.
|
Related Party Transactions. The ABCA
does not restrict related party transactions; however, in Canada, takeovers and other related party transactions are addressed
in provincial securities legislation and policies which may apply to us, including MI 61-101, as discussed above. In addition,
neither the ABCA nor our articles restrict us from adopting a shareholder rights plan or a ‘‘poison pill’’
but the application and enforceability of such measures for Canadian issuers differ from those in effect for their Delaware counterparts.
Under the DGCL, certain contracts or
transactions in which one or more of a corporation’s directors or officers has an interest are not void or voidable simply
because of such interest, provided that certain conditions are met, such as obtaining required disinterested board approval, fully
disclosing to stockholders the material facts regarding the director’s or officer’s relationship or interest and as
to the contract or transaction and receiving good faith stockholder approval thereof, or the contract or transaction is fair to
the corporation.
Exclusive Forum Provision. Under our
current Alberta articles of incorporation, we have not placed any restrictions on the forum in which certain actions may be brought.
The proposed certificate of incorporation of
Zomedica Corp. provides that, unless Zomedica Corp. consents in writing to the selection of an alternative forum, the Court of
Chancery of the State of Delaware will, to the fullest extent permitted by applicable law, be the sole and exclusive forum for
(i) any derivative action or proceeding brought on its behalf, (ii) any action asserting a claim of breach of a fiduciary duty
owed by any of its current or former directors, officers, other employees or stockholders to it or its stockholders, (iii) any
action asserting a claim arising pursuant to any provision of the DGCL, its proposed certificate of incorporation or proposed
bylaws or as to which the DGCL confers jurisdiction on the Court of Chancery of the State of Delaware, or (iv) any action asserting
a claim that is governed by the internal affairs doctrine, in each such case subject to such Court of Chancery of the State of
Delaware having personal jurisdiction over the indispensable parties named as defendants therein.
Furthermore, Section 22 of the Securities Act
creates concurrent jurisdiction for federal and state courts over all such Securities Act actions. Accordingly, both state and
federal courts have jurisdiction to entertain such claims. To prevent having to litigate claims in multiple jurisdictions and
the threat of inconsistent or contrary rulings by different courts, among other considerations, Zomedica Corp.’s certificate
of incorporation will provide that the federal district courts of the United States of America will be the exclusive forum for
resolving any complaint asserting a cause of action arising under the Securities Act.
The exclusive forum provisions described
above will not apply to claims arising under the Exchange Act.
Other Important Ownership and Exchange Controls
There is no limitation imposed by applicable
Alberta law or by our articles on the right of a non-resident to hold or vote our common shares, other than as discussed herein.
Competition Act. Limitations on the ability
to acquire and hold our common shares may be imposed by the Competition Act (Canada). This legislation permits the Commissioner
of Competition, or Commissioner, to review any acquisition or establishment, directly or indirectly, including through the acquisition
of shares, of control over or of a significant interest in us. This legislation grants the Commissioner jurisdiction, for up to
one year after the acquisition has been substantially completed, to seek a remedial order, including an order to prohibit the acquisition
or require divestitures, from the Canadian Competition Tribunal, which order may be granted where the Competition Tribunal finds
that the acquisition substantially prevents or lessens, or is likely to substantially prevent or lessen, competition.
This legislation also requires any person or
persons who intend to acquire more than 20% of our voting shares or, if such person or persons already own more than 20% of our
voting shares prior to the acquisition, more than 50% of voting our shares, to file a notification with the Canadian Competition
Bureau if certain financial thresholds are exceeded. Where a notification is required, unless an exemption is available, the legislation
prohibits completion of the acquisition until the expiration of the applicable statutory waiting period, unless the Commissioner
either waives or terminates such waiting period.
Investment Canada Act. The Investment
Canada Act requires each ‘‘non-Canadian’’ (as defined in the Investment Canada Act) who acquires
‘‘control’’ of an existing ‘‘Canadian business’’, where the acquisition of control
is not a reviewable transaction, to file a notification in prescribed form with the responsible federal government department or
departments not later than 30 days after closing. Subject to certain exemptions, a transaction that is reviewable under the Investment
Canada Act may not be implemented until an application for review has been filed and the responsible Minister of the federal
cabinet has determined that the investment is likely to be of ‘‘net benefit to Canada’’ taking into account
certain factors set out in the Investment Canada Act.
Under the Investment Canada Act, an investment
in our common shares by a non-Canadian would be reviewable only if it were an investment to acquire control of us pursuant to the
Investment Canada Act and the applicable review threshold were exceeded. The applicable threshold varies based on the country
of origin of the investor and whether they could be considered to be a “state-owned enterprise.” For private sector
investors from countries that are members of the World Trade Organization, or the WTO, a review would only be required if our enterprise
value (as determined pursuant to the Investment Canada Act) exceeded C$1.075 billion. A higher threshold, C$1.613 billion,
applies to investors from certain countries (Trade Agreement Investors, as determined pursuant to the Investment Canada Act). Control
acquisitions by investors who are state-owned enterprises (as determined pursuant to the Investment Canada Act) from WTO member
nations are reviewable where the book value of our assets exceeded C$428 million. Control acquisitions by investors from non-WTO
countries are reviewable where the book value of our assets exceeds C$5 million. These thresholds may be adjusted annually.
The Investment Canada Act contains various rules
to determine if there has been an acquisition of control. For example, for purposes of determining whether an investor has acquired
control of a corporation by acquiring shares, the following general rules apply, subject to certain exceptions: the acquisition
of a majority of the undivided ownership interests in the voting shares of the corporation is deemed to be acquisition of control
of that corporation; the acquisition of less than a majority, but one-third or more, of the voting shares of a corporation or of
an equivalent undivided ownership interest in the voting shares of the corporation is presumed to be acquisition of control of
that corporation unless it can be established that, on the acquisition, the corporation is not controlled in fact by the acquirer
through the ownership of voting shares; and the acquisition of less than one third of the voting shares of a corporation or of
an equivalent undivided ownership interest in the voting shares of the corporation is deemed not to be acquisition of control of
that corporation.
Under the Investment Canada Act, review
on a discretionary basis may also be undertaken by the federal government in respect of a much broader range of investments by
a non-Canadian to ‘‘acquire, in whole or part, or to establish an entity carrying on all or any part of its operations
in Canada.’’ No financial threshold applies to a national security review. The relevant test is whether such investment
by a non-Canadian could be ‘‘injurious to national security.’’ The federal government has broad discretion
to determine whether an investor is a non-Canadian and therefore subject to national security review. Review on national security
grounds is at the discretion of the Canadian government, and may occur on a pre- or post-closing basis.
Certain transactions relating to our common
shares will generally be exempt from the Investment Canada Act, subject to the federal government’s prerogative to
conduct a national security review, including:
|
|
(1)
|
the acquisition of our common shares by a person in the ordinary course of that person’s
business as a trader or dealer in securities;
|
|
|
(2)
|
the acquisition of control of us in connection with the realization of security granted for a loan
or other financial assistance and not for any purpose related to the provisions of the Investment Canada Act; and
|
|
|
(3)
|
the acquisition of control of us by reason of an amalgamation, merger, consolidation or corporate
reorganization following which the ultimate direct or indirect control in fact of us, through ownership of our common shares, remains
unchanged.
|
Other. There is no law, governmental
decree or regulation in Alberta that restricts the export or import of capital, or that would affect the remittance of dividends
(if any) or other payments by us to non-resident holders of our common shares, other than withholding tax requirements.
Dissenting Rights of Shareholders
A registered shareholder has the right to dissent
(a ‘‘Dissenting Shareholder’’) to the domestication pursuant to Section 191 of the ABCA. The following
description of the right to dissent and appraisal to which Dissenting Shareholders are entitled is not a comprehensive statement
of the procedures to be followed by a Dissenting Shareholder who seeks payment of the fair value of such Dissenting Shareholder’s
shares, and is qualified in its entirety by the reference to the full text of Section 191 of the ABCA, the text of which is attached
at Exhibit E. A Dissenting Shareholder who intends to exercise the right to dissent and appraisal should carefully consider
and comply with the provisions of Section 191 of the ABCA. Failure to strictly comply with the provisions of Section 191 of the
ABCA and to adhere to the procedures established therein may result in the loss of all rights thereunder.
Under the ABCA, a registered holder of shares
is entitled, in addition to any other right such holder may have, to dissent and to be paid by us the fair value of the shares
held by such Dissenting Shareholder in respect of which such Dissenting Shareholder dissents, determined as of the close of business
on the last business day before the day on which the special resolution approving the domestication was adopted. A registered shareholder
may dissent only with respect to all of the shares held by such registered holder or on behalf of any one beneficial owner and
registered in the Dissenting Shareholder’s name. Only registered shareholders may dissent. Persons who are beneficial
owners of shares registered in the name of a broker, custodian, nominee or other intermediary who wish to dissent should be aware
that they may only do so through the registered owner of such securities. A registered holder, such as a broker, who holds common
shares as nominee for beneficial holders, some of whom wish to dissent, must exercise dissent rights on behalf of such beneficial
owners with respect to the common shares held for such beneficial owners. In such case, the demand for dissent should set forth
the number of common shares covered by it.
A Dissenting Shareholder must send to us a written
objection to the special resolution to approve the domestication prior to the Special Meeting, or a Dissent Notice.
A registered shareholder wishing to exercise
the right to dissent with respect to such holder’s shares must not vote such shares at the Special Meeting, either by the
submission of a proxy or by personally voting, in favor of the special resolution approving the domestication. The ABCA does not
provide, and we will not assume, that a vote against the domestication constitutes a Dissent Notice.
An application may be made to the Alberta Court
of Queen’s Bench, or the Court, by us or by a Dissenting Shareholder after the adoption of the special resolution approving
the domestication to fix the fair value of the Dissenting Shareholder’s shares. If such an application to the Court is made
by us or a Dissenting Shareholder, we must, unless the Court otherwise orders, send to each Dissenting Shareholder a written offer
to pay the Dissenting Shareholder an amount considered by our Board of Directors to be the fair value of the shares. The offer,
unless the Court otherwise orders, will be sent to each Dissenting Shareholder at least 10 days before the date on which the application
is returnable, if we are the applicant, or within 10 days after we are served with notice of the application, if a Dissenting Shareholder
is the applicant. The offer will be made on the same terms to each Dissenting Shareholder and will be accompanied by a statement
showing how the fair value was determined.
A Dissenting Shareholder may make an agreement
with us for the purchase of such holder’s shares in the amount of the offer made by us (or otherwise) at any time before
the Court pronounces an order fixing the fair value of the shares.
A Dissenting Shareholder is not required to
give security for costs in respect of an application and, except in special circumstances, will not be required to pay the costs
of the application or appraisal. On the application, the Court will make an order fixing the fair value of the shares of all
Dissenting Shareholders who are parties to the
application, giving judgment in that amount against us and in favor of each of those Dissenting Shareholders, and fixing the time
within which we must pay that amount payable to the Dissenting Shareholders. The Court may, in its discretion, allow a reasonable
rate of interest on the amount payable to each Dissenting Shareholder calculated from the date on which the Dissenting Shareholder
ceases to have any rights as a shareholder of our company, until the date of payment.
On the domestication becoming effective, or
upon the making of an agreement between us and the Dissenting Shareholder as to the payment to be made by us to the Dissenting
Shareholder, or upon the pronouncement of a Court order, whichever first occurs, the Dissenting Shareholder will cease to have
any rights as a shareholder of our company other than the right to be paid the fair value of such holder’s shares, in the
amount agreed to between us and the Dissenting Shareholder or in the amount of the judgment, as the case may be. Until one of these
events occurs, the Dissenting Shareholder may withdraw the Dissenting Shareholder’s dissent, or if the domestication has
not yet become effective we may rescind the domestication resolution, and, in either event, the dissent and appraisal proceedings
in respect of that Dissenting Shareholder will be discontinued.
We will not make a payment to a Dissenting Shareholder
under Section 191 if there are reasonable grounds for believing that we are or would after the payment be unable to pay its liabilities
as they become due, or that the realizable value of our assets would thereby be less than the aggregate of our liabilities. In
such event, we will notify each Dissenting Shareholder that we are lawfully unable to pay the Dissenting Shareholders for their
shares in which case the Dissenting Shareholder may, be written notice to us within 30 days after receipt of such notice, withdraw
such holder’s written objection, in which case such Dissenting Shareholder shall be deemed to have participated in the domestication
as a shareholder. If the Dissenting Shareholder does not withdraw such holder’s written objection such Dissenting Shareholder
retains status as a claimant against us to be paid as soon as we are lawfully entitled to do so or, in a liquidation, to be ranked
subordinate to creditors but prior to its shareholders.
All shares held by Dissenting Shareholders who
exercise their right to dissent will, if the holders are ultimately paid the fair value thereof, be deemed to be transferred to
us on the effective date of the domestication in exchange for such fair value as of the close of business on the last business
day before the domestication is approved the shareholders. If such Dissenting Shareholders ultimately are not entitled to be paid
the fair value of the shares, such Dissenting Shareholders will be deemed to have participated in the domestication on the same
basis as a non-dissenting holder notwithstanding the provisions of Section 191 of the ABCA.
THE ABOVE SUMMARY DOES NOT PURPORT TO PROVIDE
A COMPREHENSIVE STATEMENT OF THE PROCEDURES TO BE FOLLOWED BY A DISSENTING SHAREHOLDER WHO SEEKS PAYMENT OF THE FAIR VALUE OF THEIR
SHARES. SECTION 191 OF THE ABCA REQUIRES ADHERENCE TO THE PROCEDURES ESTABLISHED THEREIN AND FAILURE TO DO SO MAY RESULT IN THE
LOSS OF ALL RIGHTS THEREUNDER. IT IS SUGGESTED THAT IF YOU WANT TO AVAIL YOURSELF OF YOUR RIGHTS THAT YOU SEEK YOUR OWN LEGAL ADVICE.
FAILURE TO COMPLY STRICTLY WITH THE PROVISIONS OF THE ABCA MAY PREJUDICE YOUR RIGHT OF DISSENT. SECTION 191 OF THE ABCA IS ATTACHED
HEREIN AS EXHIBIT E AND IS INCORPORATED HEREIN BY REFERENCE.
Accounting Treatment of the Domestication
Our domestication as a Delaware corporation
represents a transaction between entities under common control. Assets and liabilities transferred between entities under common
control are accounted for at carrying value. Accordingly, the assets and liabilities of Zomedica Corp. will be reflected at their
carrying value to us.
United States and Canadian Income Tax Considerations
The domestication may have income tax consequences
in both the United States and Canada. The material tax consequences of the domestication to us and our current shareholders are
summarized below. As used herein, the term the “Corporation” refers to Zomedica Corp., an Alberta corporation.
United
States Federal Income Tax Consequences.
The following discussion sets forth certain
material U.S. federal income tax consequences of the domestication to the Corporation, Zomedica Corp., a Delaware corporation
and the U.S. Holders (as defined below) and Non-U.S. Holders (as defined below) of the Corporation’s shares or warrants,
as well as certain of the expected material U.S. federal income consequences to Non-U.S. Holders of the ownership and disposition
of shares of common stock and warrants of Zomedica Corp.
This discussion does not address all aspects of taxation that may be relevant
to particular holders in light of their personal investment or tax circumstances or to persons that are subject to special tax
rules. In particular, this description of U.S. federal income tax consequences does not address the tax treatment of special classes
of holders, such as banks, insurance companies, tax-exempt entities, financial institutions, broker-dealers, regulated investment
companies, controlled foreign corporations, passive foreign investment companies, investors in pass-through entities and the applicable
pass-through entity, including partnerships and Subchapter S corporations holding shares of stock or warrants, persons holding
shares of stock or warrants as part of a hedging or conversion transaction or as part of a ‘‘straddle,’’
U.S. expatriates, holders who acquired their shares or warrants pursuant to the exercise of employee stock options or otherwise
as compensation or through a tax-qualified retirement plan and holders who exercise dissent rights. We assume in this discussion
that all holders hold shares and warrants of the Corporation and stock and warrants of Zomedica Corp. as capital assets within
the meaning of the Code. This discussion is based on current provisions of the Code, U.S. Treasury Regulations, judicial opinions,
published positions of the U.S. Internal Revenue Service (the “IRS”), and other applicable authorities, all as in
effect on the date of this Circular and all of which are subject to differing interpretations or change, possibly with retroactive
effect. Neither the Corporation nor Zomedica Corp. has sought, or will seek, a ruling from the IRS as to any U.S. federal income
tax consequence described herein. The IRS may disagree with the description herein, and its determination may be upheld by a court.
This discussion does not give a detailed discussion of any state, local or foreign tax considerations. We urge all holders to
consult their own tax advisors about the U.S. federal tax consequences of acquiring, holding, and disposing of the shares and
warrants of the Corporation and stock and warrants of Zomedica Corp., as well as any tax consequences that may arise under the
laws of any foreign, state, local or other taxing jurisdiction or under any applicable tax treaty.
As used in this summary, the term ‘‘U.S.
Holder’’ means a beneficial owner of shares or warrants of the Corporation or stock or warrants of Zomedica Corp.
that is for U.S. federal income tax purposes:
|
|
·
|
a citizen or resident of the United States;
|
|
|
·
|
a corporation (including any entity treated as a corporation for U.S. federal income tax purposes)
created or organized under the laws of the United States, any state thereof, or the District of Columbia;
|
|
|
·
|
an estate the income of which is taxable in the United States regardless of its source; or
|
|
|
·
|
a trust, the administration of which is subject to the primary supervision of a U.S. Court and
one or more United States persons (within the meaning of Section 7701(a)(30)) have the authority to control all substantial decisions
of the trust, or that has a valid election in effect under applicable Treasury Regulations to be treated as a United States person.
|
A beneficial owner of shares or warrants of
the Corporation or stock or warrants of Zomedica Corp. that is not a U.S. Holder is referred to below as a ‘‘Non-U.S.
Holder.’’
If a partnership (including for this purpose
any other entity, either organized within or without the United States, that is treated as a partnership for U.S. federal income
tax purposes) holds shares or warrants of the Corporation or stock or warrants of Zomedica Corp., the tax treatment of a partner
as a beneficial owner of the shares, stock or warrants generally will depend upon the status of the partner and the activities
of the partnership. Foreign partnerships also generally are subject to special U.S. federal income tax documentation requirements.
For purposes of the below discussion, because
any unit consisting of (i) one common share and one Series A warrant to acquire one common share, (ii) one common share and one-half
of one Series B warrant to acquire one common share, (iii) one common share and one Series C warrant to acquire one common share,
or (iv) one common share and one Series D warrant to acquire one common share, is separable at the option of the holder, the Corporation
is treating any common share and warrant to acquire common shares held by a holder in the form of a single unit as separate instruments
and is assuming that such unit itself will not be treated as an integrated instrument. Accordingly, any cancellation or separation
of the units in connection with the consummation of the domestication generally should not be a taxable event for U.S. federal
income tax purposes. This position is not free from doubt, and no assurance can be given that the IRS would not assert, or that
a court would not sustain, a contrary position.
Code Section 368 Reorganization Provisions. The change in the Corporation’s
place of incorporation pursuant to the domestication should constitute a reorganization within the meaning of Code Section 368(a)(1)(F)
(an “F Reorganization”), and generally should not represent a taxable transaction to the Corporation for U.S. federal
income tax purposes, provided that holders of not more than one percent (1%) of the Corporation’s shares entitled to vote
on the transaction elect to exercise their dissenters’ rights (“Dissenting Shareholders”). Under IRS guidance,
if holders of less than one percent (1%) of the Corporation’s shares entitled to vote on the domestication exercise their
dissenters’ rights, the domestication should qualify as an F Reorganization.
If the domestication does not qualify as
an F Reorganization for the reason stated above, it should qualify as a tax-free transaction to the Corporation under Code
Section 368(a)(1)(D) (a “D Reorganization”), unless the Corporation is required to use an amount of its assets to
satisfy claims of Dissenting Shareholders which would prevent the Corporation from transferring substantially all of its
assets to Zomedica Corp. Historically, for advance ruling purposes, the IRS defines “substantially all” to mean
at least seventy percent (70%) of the fair market value of gross assets, and at least ninety percent (90%) of the fair market
value of net assets, of an entity. In determining if Zomedica Corp. acquires “substantially all” of the assets of
the Corporation, payments of cash by the Corporation to Dissenting Shareholders will not be considered assets acquired by
Zomedica Corp. The Corporation will satisfy the ‘‘substantially all’’ test unless it is required to
pay to Dissenting Shareholders more than thirty percent (30%) of the fair market value of its gross assets or more than ten
percent (10%) of the fair market value of its net assets. The Corporation believes that the amount it may be required to pay
to Dissenting Shareholders will not prevent it from transferring ‘‘substantially all’’ of its assets
to Zomedica Corp. within the meaning of the IRS ruling guidelines.
The Corporation believes based on all
relevant facts that the domestication qualifies as a tax-free reorganization. If the domestication is a tax-free
reorganization within the meaning of Code Section 368, the tax basis of Zomedica Corp. stock and warrants received by a U.S.
Holder in the exchange will equal his or her tax basis in the Corporation’s shares and warrants surrendered in the
exchange increased by any gain recognized by such holder in the exchange or the “all earnings and profits amount”
included in such U.S. Holder’s income, as discussed under the headings “Effects of Code Section 367(b)” and
“Passive Foreign Investment Company Considerations” below. The holding period for the Zomedica Corp. stock and
warrants received in the exchange will be the same as the U.S. Holder’s holding period for the shares and warrants of
the Corporation surrendered in the exchange.
In the event the domestication does not
qualify as a tax-free reorganization, a U.S. Holder would recognize gain or loss with respect to such holder’s shares
and warrants of the Corporation equal to the difference between the holder’s basis in such shares and warrants and the
fair market value, as of the effective time of the domestication, of the Zomedica Corp. stock and warrants received in the
exchange. In such event, a U.S. Holder’s aggregate basis in the Zomedica Corp. stock and warrants received in the
exchange would equal its fair market value and such U.S. Holder’s holding period would begin the day after the
domestication.
Effects of Code Section 367(b).
Code Section 367(b) applies to certain non-recognition transactions involving foreign corporations. When it applies, Code
Section 367 (b) can have the effect of imposing U.S. federal income tax on U.S. Holders in connection with transactions that
would otherwise be tax free. The domestication should be considered an inbound reorganization subject to the rules of Code
Section 367(b).
Under Code Section 367(b) and the Treasury
Regulations promulgated thereunder, U.S. Holders who own, directly or by attribution, ten percent (10%) or more of the total
voting power of the Corporation’s all classes of shares or ten percent (10%) or more of the total value of shares of
all classes of stock of the Corporation (“10% Shareholders”) would be required to recognize a deemed dividend on
the domestication equal to the “all earnings and profits amount,” as determined under Section 1.367(b)-2 of the
Treasury Regulations, attributable to their shares in the Corporation.
A U.S. Holder that is not a 10% Shareholder is not
required to include the “all earnings and profits amount” attributable to such U.S. Holder’s shares in the
Corporation in income as a deemed dividend. Instead, absent making the Deemed Dividend Election discussed below, such U.S.
Holder must recognize gain (but not loss) on such holder’s shares if such shares have a fair market value of $50,000 or
more on the date of the exchange and the fair market value of Zomedica Corp. stock received in the exchange exceeds the U.S.
Holder’s tax basis of the shares of the Corporation surrendered in the exchange. However, such U.S. Holder can make an
election to include in income as a deemed dividend on the domestication an amount equal to the “all earnings and
profits amount” attributable to the shares owned by such U.S. Holder in the Corporation (the “Deemed Dividend
Election”). If a U.S. Holder makes a Deemed Dividend Election, then such holder does not recognize any gain on the
exchange under Code Section 367(b).
A Deemed Dividend Election can be made only
if the Corporation provides the U.S. Holder the information to substantiate the “all earnings and profits amount” attributable
to such U.S. Holder’s shares in the Corporation and the U.S. Holder elects and files certain notices with such holder’s
U.S. federal income tax return for the year in which the domestication occurs. U.S. Holders should consult their own tax advisors
regarding whether to make the Deemed Dividend Election and, if advisable, the appropriate filing requirements with respect to this
election.
The term “all earnings and profits amount,”
as defined under Treasury Regulations Section 1.367(b)-2(d), means the net positive earnings (if any) of a foreign (i.e., non-U.S.)
corporation that are determined according to principles substantially similar to those applicable to domestic corporations, but
taking certain modifications into account. Based on the Corporation’s limited activity at the holding company level and the
size of the Corporation’s existing earnings and profits deficit, the Corporation believes that no U.S. Holder should have
a positive “all earnings and profits amount” attributable to such holder’s shares in the Corporation, and accordingly
no 10% Shareholder or U.S. Holder who makes a Deemed Dividend Election should be required to include any such amount in income
on the domestication. The Corporation’s belief with respect to the “all earnings and profits amount” results
from calculations performed by the Corporation’s accounting firm based on information provided to them by the Corporation.
However, no assurance can be given that the IRS will agree with such calculations. If it does not agree, then a U.S. Holder may
be subject to the adverse U.S. federal income tax consequences discussed above.
A U.S. Holder that is not a 10% Shareholder
and owns shares in the Corporation with a fair market value of less than $50,000 on the day of the exchange should not be subject
to U.S. federal income tax on the domestication as a result of Code Section 367(b).
Passive Foreign Investment Company Considerations.
In addition to the discussion under the heading “Effects of Code Section 367(b)” above, the domestication might be
a taxable event to U.S. Holders under the passive foreign investment company (“PFIC”) rules of the Code, to the extent
that Code Section 1291(f) applies.
Generally, a foreign corporation will be a PFIC
if either (a) at least seventy-five percent (75%) of its gross income in a taxable year of the foreign corporation, including its
pro rata share of the gross income of any corporation in which it is considered to own at least twenty-five percent (25%) of the
shares by value, is passive income or (b) at least fifty percent (50%) of its assets in a taxable year of the foreign corporation,
ordinarily determined based on fair market value and averaged quarterly over the year, including its pro rata share of the assets
of any corporation in which it is considered to own at least twenty-five percent (25%) of the shares by value, are held for the
production of, or produce, passive income. Passive income generally includes dividends, interest, rents and royalties (other than
certain rents or royalties derived from the active conduct of a trade or business) and gains from the disposition of passive assets.
The Corporation believes that it was classified
as a PFIC during the Corporation’s taxable year ended December 31, 2019, and based on current business plans and financial
expectations, the Corporation believes it will continue to be a PFIC for the current year. However, the determination of whether
a foreign corporation is a PFIC is primarily factual and depends, in part, on the application of complex U.S. federal income tax
rules, which are subject to differing interpretations. Hence, the IRS might not agree that the Corporation is a PFIC.
Code Section 1291(f) generally requires
that, to the extent provided in Treasury Regulations, a United States person who disposes of stock of a PFIC recognizes gain
notwithstanding any other provision of the Code. No final Treasury Regulations have been promulgated under Code Section
1291(f). Proposed Treasury Regulations were promulgated in 1992 with a retroactive effective date. If finalized in their
current form, these Treasury Regulations would generally require gain recognition by a U.S. Holder exchanging equity
securities of the Corporation for equity securities of Zomedica Corp., if the Corporation were classified as a PFIC at any
time during such U.S. Holder’s holding period in such equity securities and such person had not made either a
“qualified electing fund” election under Code Section 1295 (as described below), for the first taxable year in
which such U.S. Holder owned shares of the Corporation or in which the Corporation was a PFIC, whichever is later, or a
‘‘mark-to-market’’ election under Code Section 1296 (as described below). As discussed below, a QEF
election and mark-to-market election generally cannot be made with respect to the Corporation’s warrants or preferred
shares . The tax on any such gain so recognized would be imposed at the rate applicable to ordinary income and an interest
charge would apply based on a complex set of computational rules designed to offset the tax deferral to such U.S. Holder on
the Corporation’s undistributed earnings. Under these rules:
|
|
·
|
the gain is allocated ratably to each day that the U.S. Holder owned such equity securities;
|
|
|
·
|
the amount of gain allocated to the period that predates the first day of the first taxable year
in which the Corporation was a PFIC is included in the U.S. Holder’s income as ordinary income and taxed at ordinary rates
prevailing during such time period;
|
|
|
·
|
the amount of gain allocated to the “current shareholder year” (i.e., the taxable year
in which the U.S. Holder recognizes the gain) is also included in the U.S. Holder’s income as ordinary income and taxed at
ordinary rates prevailing during such time period;
|
|
|
·
|
the amount of gain allocated to other taxable years or portions thereof (the “prior PFC years”)
is taxed at the highest tax rate in effect for that year applicable to such U.S. Holder; and
|
|
|
·
|
the interest charge amount is the interest imposed on the underpayments of tax for each prior PFIC
year of the U.S. Holder.
|
It is difficult to predict whether, in what
form, and with what effective date final Treasury Regulations under Code Section 1291(f) will be adopted.
The impact of the PFIC rules on a U.S. Holder
under the proposed Treasury Regulations would depend on whether the U.S. Holder has made a timely and effective election to treat
the Corporation as a “qualified electing fund” under Code Section 1295 (a “QEF election”) for the first
taxable year in the U.S. Holder’s holding period during which the Corporation qualified as a PFIC.
Under proposed Treasury Regulations,
if a U.S. Holder has an option, warrant, or other right to acquire stock of a PFIC, such option, warrant or right is considered
to be PFIC stock subject to the PFIC rules. However, a U.S. Holder of an option, warrant or right to acquire stock of a PFIC may
not make a QEF Election that will apply to the option, warrant or other right to acquire PFIC stock. In addition, under proposed
Treasury Regulations, if a U.S. Holder holds an option, warrant or other right to acquire stock of a PFIC, the holding period
with respect to shares of stock of the PFIC acquired on exercise of such option, warrant or other right will include the period
that the option, warrant or other right was held. Consequently, under the proposed Treasury Regulations, if a U.S. Holder of shares
made a QEF Election, such election generally would not have been treated as a timely QEF Election with respect to shares acquired
upon exercise of warrants of the Corporation and the PFIC rules discussed above will continue to apply with respect to such shares.
However, a U.S. Holder of such shares should have been eligible to make a timely QEF Election if such U.S. Holder met certain
requirements and made a “purging election,” electing in the year in which such shares were received to recognize gain
(which would have been taxed under the PFIC rules discussed above) as if such shares were sold for fair market value on the date
such U.S. Holder acquired them by exercising the corresponding warrants. Each U.S. Holder should consult its own tax advisor regarding
the application of the PFIC rules to warrants of the Corporation. A U.S. Holder that has made a valid QEF Election effective for
the first taxable year in the U.S. Holder’s holding period will not be subject to the general PFIC rules. As a result, such
U.S. Holder would not recognize gain or loss as a result of the PFIC rules under the proposed Treasury Regulations as a result
of the domestication.
The impact of the PFIC rules on a U.S.
Holder would also depend on whether the U.S. Holder has made an election under Code Section 1296. U.S. Holders who hold
(actually or constructively) stock of a foreign corporation that is classified as a PFIC may annually elect to mark such
stock to its market value if such stock is regularly traded on an established exchange (a “mark-to-market
election”). No assurance can be given that the Corporation’s common shares are considered to be regularly traded
for purposes of the mark-to-market election or whether the other requirements of this election are satisfied. If such an
election is available and has been made, such U.S. Holders will generally not be subject to the PFIC rules discussed herein.
However, if the mark-to-market election is made by a U.S. Holder after the beginning of the holding period for the PFIC
stock, then the PFIC rules will apply to certain dispositions of, distributions on and other amounts taxable with respect to
the Corporation’s ordinary shares. A mark-to- market election is not available with respect to the Corporation’s
warrants or preferred shares.
The PFIC rules are complex, and the implementation of certain aspects of the PFIC rules
requires the issuance of Treasury Regulations which in many instances have not been promulgated and which, when promulgated,
may have retroactive effect. Pursuant to the proposed Treasury Regulations discussed above, U.S. Holders that have not made a
timely QEF Election or a mark-to-market election might be subject to U.S. federal income taxation on the domestication to the
extent their shares or warrants have a fair market value in excess of their tax basis. U.S. Holders should consult their tax
advisors about the potential applicability of the proposed Treasury Regulations.
A U.S. Holder that owns (or is deemed to own)
shares in a PFIC during any taxable year of the U.S. Holder generally is required to file an IRS Form 8621 (or whether or not a
QEF Election or mark-to-market election is made) and such other information as may be required under the Treasury Regulations and
other IRS guidance. In addition to penalties, a failure to satisfy such reporting requirements may result in an extension of the
period during which the IRS can assess a tax. U.S. Holders should consult their own tax advisors regarding these filing requirements.
Information Reporting: Backup Withholding
Tax. Taxable payments made pursuant to the domestication (if any) generally will be subject to information reporting and backup
withholding tax, at the rate of twenty-four percent (24%), if a U.S. Holder (a) fails to furnish such U.S. Holder’s correct
U.S. taxpayer identification number, (b) furnishes an incorrect U.S. taxpayer identification number, (c) is notified by the IRS
that such U.S. Holder has previously failed to properly report items subject to backup withholding tax, or (d) fails to certify,
under penalty of perjury, that such U.S. Holder has furnished its correct U.S. taxpayer identification number and that the IRS
has not notified such U.S. Holder that it is subject to backup withholding tax (generally on IRS Form W-9). However, U.S. Holders
that are corporations generally are excluded from these information reporting and backup withholding tax rules. Any amounts withheld
under the U.S. backup withholding tax rules will be allowed as a credit against a U.S. Holder's U.S. federal income tax liability,
if any, or will be refunded, if such U.S. Holder furnishes required information to the IRS. Each U.S. Holder should consult its
own tax advisor regarding the information reporting and backup withholding tax rules.
Consequences of the Domestication to
Non-U.S. Holders. The exchange of equity securities of the Corporation for securities of Zomedica Corp. by a Non-U.S.
Holder should not be a taxable transaction for such holder for U.S. federal income tax purposes.
Distributions on Common
Stock of Zomedica Corp. to Non-U.S. Holders. The Corporation has not declared or paid any cash dividends on its common
shares since its inception and Zomedica Corp. does not anticipate paying cash dividends on its common stock. Subject to the
discussion below under “Payments to Foreign Financial Institutions and Non-financial Foreign Entities” and
“Information Reporting and Backup Withholding with Respect to Non-U.S. Holders,” if Zomedica Corp. makes a
distribution of cash or other property (other than certain pro rata distributions of common stock) in respect of its common
stock, the distribution will be treated as a dividend to the extent it is paid from Zomedica Corp.’s current or
accumulated earnings and profits (as determined under U.S. federal income tax principles). If the amount of a distribution
exceeds Zomedica Corp.’s current and accumulated earnings and profits, such excess generally will be treated first as a
tax-free return of capital to the extent of the Non-U.S. Holder’s tax basis in such common stock, and then as gain
realized on the sale or other disposition of the common stock that will be treated as described under the section entitled
“Sale or Other Taxable Disposition of Common Stock or Warrants” below.
Distributions treated as dividends on Zomedica
Corp.’s common stock that are paid to or for the account of a Non-U.S. Holder and are not effectively connected with a trade
or business in the United States conducted by such Non-U.S. Holder generally will be subject to U.S. federal withholding tax at
a rate of thirty percent (30%), or at a lower rate if provided by an applicable tax treaty and the Non-U.S. Holder provides the
documentation (generally, IRS Form W-8BEN) required to claim benefits under such tax treaty to the applicable withholding agent
prior to the payment of the dividends. Non-U.S. Holders that do not timely provide the applicable withholding agent with the required
certification, but that qualify for a reduced treaty rate, may obtain a refund of any excess amounts withheld by timely filing
an appropriate claim for refund with the IRS.
Pursuant to the income tax treaty between the
United States and Canada (the “Treaty”), dividends paid by a U.S. corporation to a Canadian resident where each qualifies
for benefits under the Treaty are generally subject to U.S. federal withholding tax at a maximum rate of fifteen percent (15%).
Non-U.S. Holders should consult their tax advisors regarding their eligibility for claiming benefits under the Treaty and regarding
their particular circumstances to claim a tax credit or tax deduction against their Canadian (or other) tax liability for any U.S.
federal withholding tax.
If, however, a dividend is effectively connected
with the conduct of a trade or business in the United States by a Non-U.S. Holder (and, if required by an applicable tax treaty
that a Non-U.S. Holder relies upon, is attributable to a permanent establishment maintained by the Non-U.S. Holder in the United
States), such dividend generally will not be subject to the thirty percent (30%) U.S. federal withholding tax if such Non-U.S.
Holder provides the appropriate documentation (generally, IRS Form W-8ECI) to the applicable withholding agent. Instead, such Non-U.S.
Holder generally will be subject to U.S. federal income tax on such dividend in substantially the same manner as a U.S. Holder
(except as provided by an applicable tax treaty). In addition, a Non-U.S. Holder that is a corporation may be subject to a branch
profits tax at the rate of thirty percent (30%) (or a lower rate if provided by an applicable tax treaty) on its effectively connected
earnings and profits for the taxable year, subject to certain adjustments.
Sale or Other Taxable Disposition
of Common Stock or Warrants. Subject to the discussion below under “Payments to Foreign Financial Institutions and Non-
financial Foreign Entities with Respect to Non-U.S. Holders” and “Information Reporting and Backup Withholding with
Respect to Non-U.S. Holders,” a Non-U.S. Holder generally will not be subject to U.S. federal income tax, including by way
of withholding, on gain recognized on a sale or other disposition of common stock or warrants of Zomedica Corp., unless any one
of the following is true:
|
|
·
|
the gain is effectively connected with the
Non-U.S. Holder’s conduct of a trade or business in the United States and, if an applicable tax treaty requires, attributable
to a U.S. permanent establishment maintained by such Non- U.S. Holder, in which event such Non-U.S. Holder generally will be subject
to U.S. federal income tax on such gain in substantially the same manner as a U.S. Holder (except as provided by an applicable
tax treaty) and, if it is a corporation, may also be subject to a branch profits tax at the rate of thirty percent (30%) (or a
lower rate if provided by an applicable tax treaty) on all or a portion of its effectively connected earnings and profits for the
taxable year, subject to certain adjustments;
|
|
|
·
|
the Non-U.S. Holder is an individual who is
present in the United States for 183 or more days in the taxable year of the sale, exchange or other disposition and certain other
requirements are met; or
|
|
|
·
|
Zomedica Corp. is a “United States real property holding corporation”
(a “USRPHC”) for U.S. federal income tax purposes at any time during the shorter of the period during which such Non-U.S.
Holder holds the equity securities of Zomedica Corp. disposed of, or the five-year period ending on the date such Non-U.S. Holder
disposes of the equity securities of Zomedica Corp. and, in the event of equity securities that are regularly traded on an established
securities market for tax purposes, the Non-U.S. Holder held, directly or indirectly, at any time within the five-year period
preceding such disposition more than five percent (5%) of such regularly traded equity securities.
|
Generally, a corporation is a USRPHC
if the fair market value of its U.S. real property interests equals or exceeds fifty percent (50%) of the sum of the fair market
value of its worldwide real property interests and its other assets used or held for use in a trade or business (all as determined
for U.S. federal income tax purposes). It is not believed that Zomedica Corp. will be or will become a USRPHC. However, since
the determination of USRPHC status in the future will be based upon the composition of its assets from time to time and there
are uncertainties in the application of certain relevant rules, there can be no assurance that it will or will not be a USRPHC
in the future.
Payments to Foreign Financial Institutions
and Non-financial Foreign Entities. Withholding taxes may be imposed under the Code provisions commonly known as the Foreign
Account Tax Compliance Act (“FATCA”) on certain types of payments made to non-U.S. financial institutions and certain
other non-U.S. entities. Specifically, a thirty percent (30%) withholding tax may be imposed on dividends on common stock of Zomedica
Corp. paid to a “foreign financial institution” or a “non-financial foreign entity” (each as defined in
the Code), unless (1) the foreign financial institution undertakes certain diligence and reporting obligations, (2) the non-financial
foreign entity either certifies it does not have any ‘‘substantial United States owners’’ (as defined in
the Code) or furnishes identifying information regarding each substantial United States owner, or (3) the foreign financial institution
or non-financial foreign entity otherwise qualifies for an exemption from these rules. If the payee is a foreign financial institution
and is subject to the diligence and reporting requirements in (1) above, it must enter into an agreement with the IRS requiring,
among other things, that it undertake to identify accounts held by certain “specified United States persons” or “U.S.
owned foreign entities” (each as defined in the Code), annually report certain information about such accounts, and withhold
thirty percent (30%) on certain payments to non-compliant foreign financial institutions and certain other account holders. An
intergovernmental agreement governing FATCA between the United States and an applicable foreign country may modify the requirements
described in this paragraph. Non-U.S. Holders should consult their own tax advisors regarding the potential application of withholding
under FATCA to their investment in common stock of Zomedica Corp.
Information Reporting and Backup Withholding
with Respect to Non-U.S. Holders. A Non-U.S. Holder may have to comply with specific certification procedures to establish
that the holder is not a United States person (generally on IRS Form W-8BEN), or otherwise establish an exemption, in order to
avoid backup withholding and information reporting tax requirements with respect to payments of dividends on the stock of Zomedica
Corp.
The payment of the proceeds of a disposition
of Zomedica Corp. equity securities by a Non-U.S. Holder to or through the U.S. office of a broker generally will be reported
to the IRS and reduced by backup withholding unless the Non-U.S. Holder either certifies its status as a Non-U.S. Holder under
penalties of perjury or otherwise establishes an exemption and the broker has no actual knowledge to the contrary. Information
reporting requirements, but not backup withholding, will also apply to payments of the proceeds from sales of Zomedica Corp. equity
securities by foreign offices of U.S. brokers or foreign brokers with certain types of relationships to the United States, unless
the broker has documentary evidence in its records that the holder is a Non-U.S. Holder and certain other conditions are met,
or the holder otherwise establishes an exemption.
Backup withholding is not an additional tax.
Any amounts that are withheld under the backup withholding rules will be refunded or credited against the Non-U.S. Holder’s
U.S. federal income tax liability if certain required information is furnished to the IRS. Non-U.S. Holders should consult their
own tax advisors regarding application of backup withholding in their particular circumstance and the availability of and procedure
for obtaining an exemption from backup withholding under current Treasury Regulations.
Material Canadian Federal Income Tax Considerations
The following is a summary of the principal
Canadian federal income tax considerations under the Tax Act generally applicable to the Corporation and to shareholders who,
for the purposes of the Tax Act and at all relevant times, (i) deal at arm’s length and are not affiliated with the Corporation
and (ii) hold their common shares or Series 1 preferred shares (collectively referred to in this summary as “Shares”)
as capital property. Shares of the Corporation will generally be capital property of a shareholder unless they are held in the
course of carrying on a business of trading or dealing in securities or have been acquired in a transaction or transactions considered
to be an adventure or concern in the nature of trade.
This summary assumes that the Corporation will
cease to be resident in Canada for purposes of the Tax Act at the time of the Domestication and that from the time of the Domestication
and at all relevant times thereafter, the Corporation will be a resident of the United States for purposes of the Canada-U.S. Tax
Convention (1980), as amended (the “Treaty”) and will be entitled to the benefits of the Treaty.
This
summary does not apply to a shareholder (i) an interest in which would be a “tax shelter investment” (as defined in
the Tax Act), (ii) that is a “financial institution” (as defined in the Tax Act) for purposes of the mark-to-market
rules, (iii) that is a “specified financial institution” or “restricted financial institution” (each as
defined in the Tax Act), (iv) that has elected to determine its Canadian tax results in a foreign currency pursuant to the functional
currency reporting rules in the Tax Act, (v) in respect of whom the Corporation will be a “foreign affiliate” for purposes
of the Tax Act at any time after the Domestication, (vi) that is a corporation resident in Canada that is, or becomes, controlled
by a non-resident for the purposes of the "foreign affiliate dumping" rules in the Tax Act, or (vii) that has or will
enter into a “derivative forward agreement” or “synthetic disposition agreement” (each as defined in the
Tax Act) with respect to the Shares.
This summary does not describe the tax considerations
with respect to holding or disposing of options or warrants of the Corporation. Holders of such options or warrants should consult
their own tax advisors.
This summary is based upon the current provisions
of the Tax Act and the regulations thereunder (the “Regulations”) in force as of the date hereof, all specific proposal
(the “Proposed Amendments”) to amend the Tax Act or the Regulations that have been publicly announced by, or on behalf
of, the Minister of Finance (Canada) prior to the date hereof and counsel’s understanding of the current published administrative
and assessing practices of the Canada Revenue Agency (“CRA”). No assurance can be given that any Proposed Amendments
will be enacted in their current proposed form, or at all. This summary does not take into account or anticipate any other changes
to the law, whether by legislative, governmental or judicial decision or action, nor does it take into account provincial, territorial
or foreign income tax legislation or considerations, which may differ from the Canadian federal income tax considerations.
For the purposes of the Tax Act, all amounts
must be determined in Canadian dollars based on an appropriate exchange rate as determined in accordance with the provisions of
the Tax Act.
This summary is of a general nature only,
is not exhaustive of all possible Canadian federal income tax considerations and is not intended to be, nor should it be construed
to be, legal or tax advice to any particular holder. Therefore, holders should consult their own tax advisors with respect to their
particular circumstances.
The Corporation. Upon the domestication,
the Corporation will cease to be resident in Canada for purposes of the Tax Act and will thereafter no longer be subject to Canadian
income tax on its worldwide income (but will be subject to U.S. federal and state tax). However, if the Corporation carries on
business in Canada, or if it realizes gains from the disposition of “taxable Canadian property” (as defined in the
Tax Act), it will be subject to Canadian income tax in respect of such Canadian source income, subject to relief under the Treaty.
Management of the Corporation does not expect that the Corporation will carry on business in Canada or hold any taxable Canadian
property following the domestication.
For purposes of the Tax Act, the Corporation’s
taxation year will be deemed to have ended immediately before it ceases to be a resident of Canada and a new taxation year will
be deemed to have begun at that time. Immediately before its deemed year end, the Corporation will be deemed to have disposed of
each of its properties for proceeds of disposition equal to the fair market value of such properties and to have reacquired such
properties immediately thereafter at a cost amount equal to fair market value. The Corporation will be subject to income tax under
Part I of the Tax Act on any income and net taxable capital gains realized as a result of the deemed dispositions of its properties.
The Corporation will also be subject to an additional
“emigration tax” under Part XIV of the Tax Act on the amount by which the fair market value, immediately before its
deemed year end resulting from the Domestication, of all of the property owned by the Corporation exceeds the total of certain
of its liabilities and the paid-up capital of all the issued and outstanding shares of the Corporation immediately before the deemed
year end. This additional tax is generally payable at the rate of 25 percent, but will be reduced to 5 percent under the Treaty
unless it can reasonably be concluded that one of the main reasons for the Corporation becoming resident in the United States was
to reduce the amount of emigration tax or Canadian withholding tax under Part XIII of the Tax Act.
Management of the Corporation has advised that
as of the date hereof, the Corporation does not expect to pay income tax as a result of the deemed disposition of its properties
on the domestication. Management of the Corporation has also advised that as of the date hereof, the aggregate of the paid-up capital
of the shares and the liabilities of the Corporation is not less than the current fair market value of all of the property of the
Corporation and accordingly that the domestication will not result in any liability for emigration tax.
Shareholders are cautioned that the CRA may
not agree with the Corporation’s determination of the fair market value of its properties at the relevant time. It is also
possible that the fair market value of the Corporation’s properties may change between the date hereof and the time of the
Domestication. Should unforeseen events lead to a potential for greater tax liability than currently expected, the Board has the
right to not proceed with the domestication.
Shareholders Resident in Canada. The
following portion of this summary is applicable to shareholders who are resident in Canada for purposes of the Tax Act (“Canadian
Resident Holders”).
Canadian Resident Holders will not be
considered to have disposed of their Shares of the Corporation as a result of the domestication. If a Canadian Resident
Holder sells or otherwise disposes of Shares following the domestication (other than to the Corporation on redemption,
purchase for cancellation or otherwise), such Canadian Resident Holder will realize a capital gain (or a capital loss) equal
to the amount by which the proceeds of disposition for the Shares exceed (or are exceeded by) the aggregate of the adjusted
cost base of such Shares and any reasonable costs of disposition. One-half of any capital gain will be included in income as
a taxable capital gain and one-half of any capital loss will be deducted as an allowable capital loss against taxable capital
gains realized in the year of disposition. Any unused allowable capital losses may be applied to reduce net taxable capital
gains realized in the three preceding taxation years or any subsequent taxation year, subject to the detailed provisions of
the Tax Act in that regard.
Dividends received or deemed to be received by a Canadian Resident Holder on the Shares of the
Corporation will be included in computing the Canadian Resident Holder’s income for tax purposes. In the case of a
Canadian Resident Holder that is an individual, such dividends will not be subject to the gross-up and dividend tax credit
rules normally applicable in respect of taxable dividends received from taxable Canadian corporations. In the case of a
Canadian Resident Holder that is a corporation, such Canadian Resident Holder will not be able to deduct the amount of
dividends in computing its taxable income. A “Canadian-controlled private corporation” (as defined in the Tax
Act) may be liable to pay an additional refundable tax of 10 2/3% on its “aggregate investment income” which is
defined to include amounts in respect of taxable capital gains and certain dividends. To the extent that U.S. withholding
taxes are imposed on dividends paid by the Corporation following the domestication, the amount of such tax will generally be
eligible for a Canadian foreign tax credit or tax deduction, subject to the detailed rules and limitations under the Tax Act.
Canadian Resident Holders are advised to consult their own tax advisors with respect to the availability of a Canadian
foreign tax credit or deduction having regard to their particular circumstances.
A Canadian Resident Holder that validly
exercises Dissent Rights (a “Resident Dissenter”) will be entitled to be paid by the Corporation the fair value
of their Shares in respect of which they dissent. Although the matter is not free from doubt, a Resident Dissenter will
generally be deemed to have received a dividend on their Shares equal to the amount, if any, by which the fair value of their
Shares exceeds the paid-up capital of such Shares for purposes of the Tax Act. The amount of this deemed dividend could, in
some circumstances, be treated as proceeds of disposition in the case of a Resident Dissenter that is a corporation. The
difference between the fair value of the Shares and the amount of any deemed dividend would be treated as proceeds of
disposition of the Shares for the purposes of computing any capital gain or capital loss realized by a Resident Dissenter on
the disposition of the Shares. Any interest awarded to a Resident Dissenter by a court will be included in the Resident
Dissenter’s income for Canadian income tax purposes. Canadian Resident Holders who are considering exercising Dissent
Rights in connection with the domestication are urged to consult with their tax advisors with respect to the tax consequences
to them of dissenting. The Corporation may be liable to pay tax under Part VI.1 of the Tax Act on any amount that is deemed
to be a dividend paid to a Resident Dissenter that holds Series 1 preferred shares.
A Canadian Resident Holder that is a
“specified Canadian entity” for a taxation year or a fiscal period and whose total “cost amount” of
“specified foreign property” (as such terms are defined in the Tax Act) at any time in the year or fiscal period
exceeds $100,000 will be required to file an information return for the year or period disclosing prescribed information.
Shares will be “specified foreign property” for these purposes and Canadian Resident Holders should consult their
own tax advisors to determine whether these rules are applicable in their particular case.
Pursuant to the offshore investment fund
property rules in section 94.1 of the Tax Act (the “OIFP Rules”), if in a particular year a Canadian Resident
Holder holds or has an interest in Shares, and the Shares may reasonably be considered to derive their value, directly or
indirectly, primarily from portfolio investments in (i) shares of the capital stock of one or more corporations, (ii)
indebtedness or annuities, (iii) interests in one or more corporations, trusts, partnerships, organizations, funds or
entities, (iv) commodities, (v) real estate, (vi) Canadian or foreign resource properties, (vii) currency of a country other
than Canada, (viii) rights or options to acquire or dispose of any of the foregoing, or (ix) any combination of the
foregoing, and one of the main reasons for holding an interest in the Shares is to reduce or defer the Canadian tax liability
that would have applied to the income, profits and gains generated by the portfolio investments if such income, profits and
gains had been earned directly by the holder, the Canadian Resident Holder will generally be required to include in computing
income for the year an amount equal to the amount, if any, by which (i) an imputed return for the taxation year computed on a
monthly basis and calculated as the product obtained when the Canadian Resident Holder’s “designated cost”
(within the meaning of the Tax Act) of the Shares at the end of the month, is multiplied by one-twelfth of the total of (A)
the applicable prescribed rate for the period that includes such month, and (B) two percent, exceeds (ii) the Canadian
Resident Holder’s income for the year (other than a capital gain) in respect of the Shares determined without reference
to these rules. The OIFP Rules are complex and their application depends, to a large extent, on the reasons for a Canadian
Resident Holder acquiring or holding the Shares. Canadian Resident Holders are urged to consult their own tax advisors
regarding the application and consequences of the OIFP Rules in their own particular circumstances.
U.S. Resident
Holders. The following portion of this summary is applicable to holders of Shares who are resident in the United States
for purposes of the Tax Act and the Treaty and are entitled to the benefits of the Treaty, and who do not use or hold their
Shares and will not use their Shares in the course of carrying on a business in Canada (“U.S. Resident Holders”).
Special rules, which are not discussed in this summary, may apply to a holder of Shares that is a non-resident insurer that
carries on an insurance business in Canada and elsewhere. Such holders should consult their own tax advisors.
U.S. Resident Holders will not be
considered to have disposed of their Shares as a result of the domestication. After the domestication, U.S. Resident Holders
will not be subject to Canadian withholding tax on dividends received from the Corporation.
A U.S. Resident Holder will not be subject
to tax under the Tax Act on any capital gain realized on the disposition of Shares after the domestication unless such Shares
are “taxable Canadian property” of the U.S. Resident Holder for purposes of the Tax Act.
Provided that the common shares of the
Corporation are listed on a designated stock exchange (which includes the NYSE) at a particular time, the common shares will
generally not be taxable Canadian property of a U.S. Resident Holder at that time unless at any time during the 60-month
period that ends at that time (i) 25% or more of the issued shares of any class of the capital stock of the Corporation were
owned by or belonged to one or any combination of the U.S. Resident Holder, persons with whom the U.S. Resident Holder did
not deal at arm’s length, and partnerships in which the U.S. Resident Holder or a person who did not deal at
arm’s length with the U.S. Resident Holder holds a membership interest directly or indirectly through one or more
partnerships, and (ii) more than 50% of the fair market value of the common shares was derived directly or indirectly from
one or any combination of real or immovable properties situated in Canada, Canadian resource properties, timber resource
properties and options in respect of, or interests in, or for civil law rights in, any such properties whether or not such
properties exist.
The Series 1 preferred shares will
generally not be taxable Canadian property of a U.S. Resident Holder any particular time if, during the 60-month period that
ends at that time, more than 50% of the fair market value of the Series 1 preferred shares was derived directly or indirectly
(otherwise than through a corporation, partnership or trust the shares or interests in which were not themselves table
Canadian property at the particular time) from one or any combination of real or immovable properties situated in Canada,
Canadian resource properties, timber resource properties and options in respect of, or interests in, or for civil law rights
in, any such properties whether or not such properties exist.
However, in certain circumstances, the
Shares may be deemed to be taxable Canadian property of a U.S. Resident Holder for purposes of the Tax Act.
A U.S. Resident Holder that validly
exercises Dissent Rights (a “U.S. Resident Dissenter”) will be entitled to receive from the Corporation the fair
value of the Shares in respect of which they dissent. Although the matter is not free from doubt, a U.S. Resident Dissenter
will generally be deemed to have received a dividend on the Shares equal to the amount, if any, by which the fair value of
the Shares exceeds the paid-up capital of such Shares for purposes of the Tax Act. Any such deemed dividend will be subject
to Canadian withholding tax at a rate of 25% of the gross amount of the dividend, but will be reduced to the rate of 15%
under the provisions of the Treaty, other than for U.S. Resident Dissenters that are U.S. corporations owning at least 10% of
the voting stock of the Corporation, in which case the rate of withholding on dividends under the Treaty would be 5%. A U.S.
Resident Dissenter will also be considered to have disposed of the Shares for proceeds of disposition equal to the amount
paid to such U.S. Resident Dissenter. Any interest awarded to a U.S. Resident Dissenter by a court will not be subject to
withholding tax under the Tax Act, provided such interest is not “participating debt interest” for purposes of
the Tax Act. U.S. Resident Holders who are considering exercising Dissent Rights in connection with the Domestication are
urged to consult with their tax advisors with respect to the tax consequences of such action. A U.S. Resident Dissenter will
not be subject to tax under the Tax Act on any capital gain realized on the disposition of Shares unless the Shares are
“taxable Canadian property” for purposes of the Tax Act, as discussed above. . The Corporation may be liable to
pay tax under Part VI.1 of the Tax Act on any amount that is deemed to be a dividend paid to a U.S. Resident Dissenter that
holds Series 1 preferred shares.
Eligibility for Investment.
Following the domestication, the Corporation will cease to be a “public corporation” for purposes of the Tax Act.
However, management of the Corporation has advised that it intends that the common shares continue to be listed on the NYSE.
Provided that the common shares of the Corporation are listed on a “designated stock exchange” such as the NYSE
at the time of the domestication and thereafter, the common shares will continue to be a qualified investment for trusts
governed by a registered retirement savings plan (“RRSP”), registered retirement income fund
(“RRIF”), deferred profit sharing plan, registered disability savings plan (“RDSP”), registered
education savings plan (“RESP”) and tax-free savings account (“TFSA”) (collectively,
“Registered Plans”).
Notwithstanding that the common shares of the Corporation may be a qualified investment for
a trust governed by a RRSP, RRIF, RESP, RDSP or TFSA, the annuitant of a RRSP or RRIF, the subscriber of a RESP or the holder
of a RDSP or TFSA, as the case may be, will be subject to a penalty tax if such common shares are a “prohibited
investment” (as defined in the Tax Act). The common shares will generally not be a “prohibited investment”
for a trust governed by a RRSP, RRIF, RESP, RDSP or TFSA provided that (i) the annuitant of the RRSP or the RRIF, the
subscriber of the RESP or the holder of the RDSP or TFSA, as the case may be, deals at “arm’s length” (as
defined in the Tax Act) with the Corporation and does not have a “significant interest” (as defined in the Tax
Act) in the Corporation or (ii) the common shares are “excluded property” (as defined in subsection 207.01(1) of
the Tax Act) for the RRSP, RRIF, RESP, RDSP or TFSA. Shareholders holding common shares through such plans should consult
their own tax advisors as to whether their common shares would be a prohibited investment in their particular
circumstances.
Description
of Capital Stock
Unless the context provides otherwise, the following
description of our capital stock assumes the consummation of the domestication has already occurred. The following description
of Zomedica Corp.’s capital stock is not complete and is subject to and qualified in its entirety by its proposed certificate
of incorporation and bylaws, which are attached as Exhibit C and Exhibit D, respectively, to this Circular.
Zomedica Corp’s authorized capital stock
consists of 2,000,000,000 shares of common stock, $0.0001 par value per share, and 10,000,000 shares of preferred stock, $0.0001
par value per share. Assuming the domestication had occurred on the Record Date, shares of common stock would have been issued
and outstanding and 12 shares of Series 1 preferred stock would have been issued and outstanding.
Common Stock
Holders of common stock are entitled
to one vote for each share held of record on all matters on which stockholders are permitted to vote. The proposed certificate
of incorporation of Zomedica Corp.. provides that, except as otherwise provided by law, the affirmative vote of a majority in
voting power of the shares of common stock, present in person or represented by proxy at a meeting at which a quorum is present
shall be the act of the stockholders. Delaware law requires the affirmative vote of a majority in voting power of the outstanding
shares to authorize certain extraordinary actions, such as mergers, consolidations, dissolutions or an amendment to the certification
of incorporation of Zomedica Corp. There is no cumulative voting for the election of directors. Upon a liquidation, Zomedica Corp.’s
creditors and any holders of preferred stock with preferential liquidation rights will be paid before a distribution to holders
of its common stock. The holders of the common stock of Zomedica Corp. would be entitled to receive a pro rata amount per share
of any excess distribution. Holders of common stock have no preemptive or subscription rights. There are no conversion rights,
redemption rights, sinking fund provisions or fixed dividend rights with respect to the common stock. All outstanding shares of
the common stock are fully paid and nonassessable.
Preferred Stock
The proposed certificate of incorporation
of Zomedica Corp. empowers the Board of Directors to issue up to 10,000,000 shares of preferred stock from time to time, in one
or more series. The Board of Directors also may fix the designation, powers, preferences and rights and the qualifications, limitations
and restrictions of those shares, including dividend rights, conversion rights, voting rights, redemption rights, terms of sinking
funds, liquidation preferences and the number of shares constituting any series or the designation, of the series. Terms selected
could decrease the amount of earnings and assets available for distribution to holders of common stock. The rights of holders
of the common stock will be subject to the rights of the holders of any preferred shares that may be issued in the future. Additionally,
the issuance of preferred stock may have the effect of decreasing the market price of the common stock. Although Zomedica Corp.
has no present intention to issue any shares of preferred stock, any issuance could have the effect of making it more difficult
for a third party to acquire a majority of Zomedica Corp.’s outstanding voting stock.
Series
1 Preferred Shares
Zomedica Corp. has authorized a series of 20
shares of preferred stock designated as Series 1 preferred stock. 12 shares of Series 1 preferred stock are issued and outstanding.
Each share of Series preferred stock has a “stated value” of $1,000,000 per share.
Holders of the Series 1 preferred stock
are not entitled to dividends but, in lieu thereof, are entitled to a return on their investment in the Series 1 preferred stock
in lieu of the payment of ordinary dividends defined as “Net Sales Returns”. Net Sales Returns are to equal 9% of
“Net Sales” made by Zomedica Corp. or its affiliates for the sale of its products. Holders of Series 1 preferred stock
are entitled to receive Net Sales Returns out of lawfully available funds therefor until such time as the holders have received
total Net Sales Returns in respect of each share of Series 1 preferred stock equal to 9 times the Stated Value of each shares
of Series 1 preferred stock. Subject to applicable law and the presence of lawfully available funds therefor, Zomedica Corp. will
have the right to redeem the outstanding shares of Series 1 preferred stock at any time at a redemption price equal to 9 times
the aggregate Stated Value of each outstanding share of Series 1 preferred stock, less the aggregate amount of the Net Sales Returns
paid in respect of each share, or the redemption amount. Upon any dissolution, liquidation or winding up, or other distribution
of the assets of Zomedica Corp. for the purposes of winding up its affairs (other than a Fundamental Transaction, as defined below),
the holders of Series 1 preferred stock will be entitled to a receive the Stated Value of each share of Series 1 preferred stock
less the Net Sales Returns paid on each share. Such amount shall be paid before any distribution is made to the holders of the
common stock. A “Fundamental Transaction” is defined in the Series 1 preferred stock to mean: (a) an amalgamation,
merger or other business combination transaction involving Zomedica Corp. whereby all or substantially all of the outstanding
shares of common stock are sold, transferred or exchanged pursuant to which our stockholders before the transaction do not have
the right after the transaction to cast more than 50% of the votes that may be cast for the election of directors of the successor
or continuing corporation; or (b) a sale, lease or disposition of the properties and/or assets of Zomedica Corp. as an entirety
or substantially as an entirety to another person and the subsequent distribution of the consideration received to holders of
common stock. In the event of a Fundamental Transaction, the holders of Series 1 preferred stock will be entitled to receive consideration
for each share of Series 1 preferred stock equal to a multiple of the Stated Value of the Series 1 preferred stock ranging from
5.0 to 9.0 depending on the date of completion of the Fundamental Transaction and the date of the original issuance of the Series
1 preferred shares by us, subject to a cap equal to the Redemption Amount. The entitlement of holders of Series 1 preferred stock
in the event of a Fundamental Transaction ranks prior in right of payment to the rights of holders of common stock. In connection
with any proposed amendment to the rights, privileges, restrictions or conditions attaching to the Series 1 preferred stock, there
is a requirement for such holders to approve the amendment at a meeting by the affirmative vote of not less than two-thirds, with
holders entitled to one vote in respect of each share of Series 1 preferred stock held. This approval requirement is in addition
to any other approval requirement under applicable law.
Warrants
As of the Record Date,
we had 280,005,613 common shares issuable upon the exercise of outstanding warrants with a weighted average exercise price of approximately
$0.16 per share. Such warrants will remain outstanding after the domestication and will be exercisable for shares of Zomedica Corp.
common stock on the same terms, without any action by the holders thereof.
Potential Anti-takeover Effect of Delaware Law,
Our Certificate of Incorporation and Bylaws
The proposed certificate of incorporation and
bylaws of Zomedica Corp. include the following provisions, which may discourage a hostile takeover attempt:
|
|
·
|
a requirement that stockholders provide prior notice to nominate directors or propose other action
at the annual meeting of stockholders;
|
|
|
·
|
a prohibition on the ability of stockholders to call a special meeting of stockholders;
|
|
|
·
|
an elimination of the ability of stockholders to take action by written consent;
|
|
|
·
|
provisions that give the board of directors the sole authority to determine the number of directors
and fill newly created directorships and vacancies on the board of directors; and
|
|
|
·
|
provisions that give the board of directors the authority to issue preferred stock having rights,
privileges, limitations and other characteristics that are established by the board of directors without stockholder approval.
|
In addition, Section 203 of the DGCL
prohibits a publicly held Delaware corporation from engaging in various “business combination” transactions with
any “interested stockholder” for a period of three years after the time of the transaction on which the person
became an “interested stockholder,: unless:
|
|
·
|
the corporation's Board of Directors approved the transaction before the “interested stockholder”
obtained such status;
|
|
|
·
|
upon consummation of the transaction that resulted in the stockholder becoming an “interested
stockholder,” the “interested stockholder” owned at least 85% of the voting stock of the corporation outstanding
at the time the transaction commenced, excluding for purposes of determining the number of shares outstanding those shares owned
(i) by persons who are directors and are also officers and (ii) employee stock plans in which the participants do not have the
right to determine confidentially whether shares held subject to the plans will be tendered in the tender or exchange offer; or
|
|
|
·
|
on or subsequent to such time, the business combination is approved by the corporation's Board of Directors and authorized at an annual or
special meeting of stockholders, and not by written consent, by two thirds of the holders of the outstanding common stock not owned by
the “interested stockholder.”
|
A “business combination” is defined
to include certain mergers, asset sales and other transactions resulting in financial benefit to a stockholder. In general, an
“interested stockholder” is a person who, together with affiliates and associates, owns 15% or more of a corporation's
voting stock or within three years owned 15% or more of a corporation's voting stock. The statute could prohibit or delay mergers
or other takeover or change in control attempts.
Exclusive Forum Provision
The certificate of incorporation of Zomedica
Corp. will require that, unless we consent in writing to the selection of an alternative forum:
|
|
·
|
any derivative action or proceeding brought on our behalf;
|
|
|
·
|
any action asserting a claim of breach of any fiduciary duty owed by any current or former director,
officer other employee or stockholder of Zomedica Corp. to the corporation or our stockholders;
|
|
|
·
|
any action asserting a claim arising pursuant to any provision of the DGCL, our certificate of
incorporation or bylaws or as to which the DGCL confers jurisdiction on the Court of Chancery of the State of Delaware; or
|
|
|
·
|
any action asserting a claim governed by the internal affairs doctrine;
|
the Court of Chancery of the State of Delaware will, to the fullest
extent permitted by law, be the exclusive forum or if the Court of Chancery of the State of Delaware does not have subject matter
jurisdiction thereof, the federal district court of the State of Delaware.
Furthermore, Zomedica Corp.’s certificate
of incorporation will provide that the federal district courts of the United States of America will be the exclusive forum for
resolving any complaint asserting a cause of action arising under the Securities Act.
The exclusive forum provisions described above
will not apply to claims arising under the Securities Exchange Act of 1934, as amended, or the Exchange Act.
Listing
The common stock of Zomedica Corp. will continue
to be listed on the NYSE American under the symbol “ZOM.”
Transfer Agent and Registrar
The transfer agent and registrar for our common
shares is AST Trust Company (Canada). Our transfer agent’s address is 1 Toronto Street, Suite 1200, Toronto, Ontario M5C
2VC and its telephone number is (416) 682-3844. Our co-transfer agent is American Stock Transfer & Trust Company.
Interest
of CERTAIN PERSONS OR COMPANIES in Matters to be Acted on
Except as disclosed in this proxy circular/prospectus,
none of the directors or executive officers of our company at any time since the beginning of our last financial year, nor any
associate or affiliate of any of the foregoing persons, has any material interest, direct or indirect, by way of beneficial ownership
of securities or otherwise, in any matter to be acted on at the Special Meeting.
INTEREST OF INFORMED PERSONS IN MATERIAL
TRANSACTIONS
None of the informed persons of our company
(as defined in National Instrument 51-102 - Continuous Disclosure Obligations), nor any associate or affiliate of the foregoing
persons has any material interest, direct or indirect, in any transaction since the commencement of our most recently completed
financial year or in any proposed transaction which, in either case, has or will materially affect our company.
Legal
Matters
Certain legal matters relating to the domestication
under United States law will be passed upon by Lowenstein Sandler LLP, New York, New York. Certain legal matters relating to the
United States and Canadian tax consequences of the domestication will be passed upon respectively by Lowenstein Sandler LLP, New
York, New York, and by Norton Rose Fulbright Canada LLP.
Experts
The consolidated financial statements of Zomedica
Corp. as of December 31, 2019 and December 21, 2018 and for each of the years in the period ended December 31, 2019 included in
this proxy circular/prospectus have been audited by MNP LLP, an independent registered public accounting firm, as stated in their
report. Such consolidated financial statements have been included herein in reliance on the report of such firm given upon their
authority as experts in accounting and auditing.
other
business
A shareholder or proxyholder will vote on any
other items of business that may be properly brought before the Special Meeting. As of the date of this proxy circular/prospectus,
we are not aware of any other matters to be brought before the Special Meeting.
WHERE
YOU CAN FIND MORE INFORMATION
This proxy circular/prospectus constitutes part
of a registration statement on Form S-4 that we have filed with the SEC. You may read and copy this proxy circular/prospectus
at the SEC's Public Reference Room at 100 F Street, N.E., Washington, D.C. 20549. You may also obtain copies of this proxy
circular/prospectus by mail from the Public Reference Section of the SEC at prescribed rates. To obtain information on the operation
of the Public Reference Room, you can call the SEC at 1-800-SEC-0330. The SEC also maintains an Internet website that contains
reports, proxy and information statements and other information regarding issuers that file electronically with the SEC. The address
of the SEC's Internet website is http://www.sec.gov.
Financial information on our company is provided
in our comparative annual financial statements and management discussion and analysis for the year ended December 31, 2019 which
can also be accessed at www.sedar.com.
We file periodic reports, proxy statements and
other documents with the SEC. We will provide you without charge, upon your oral or written request, with a copy of any or all
reports, proxy statements and other documents we file with the SEC (other than exhibits to such documents). Requests for such copies
should be directed to:
Zomedica Pharmaceuticals Corp.
100 Phoenix Drive, Suite 180
Ann Arbor, Michigan 48108
(734) 369-2555
INDEX TO FINANCIAL STATEMENTS
Zomedica Pharmaceuticals Corp.
Consolidated financial statements
For the years ended December 31, 2019
and 2018
(Expressed in United States Dollars, except
as otherwise noted)
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Shareholders
of Zomedica Pharmaceuticals Corp.:
Opinion on the Consolidated Financial
Statements
We have audited the accompanying consolidated
financial statements of Zomedica Pharmaceuticals Corp. and its subsidiaries (the “Company”), which comprise the consolidated
balance sheets as at December 31, 2019 and 2018, and the consolidated statements of operations and comprehensive loss, shareholders’
equity and cash flows for each of the years in the two-year period ended December 31, 2019, and the related notes, comprising a
summary of significant accounting policies and other explanatory information (collectively referred to as the consolidated financial
statements). In our opinion, the consolidated financial statements present fairly, in all material respects, the consolidated financial
position of the Company as at December 31, 2019 and 2018, and its consolidated results of operations and its consolidated cash
flows for each of the years in the two-year period ended December 31, 2019, in conformity with accounting principles generally
accepted in the United States of America (US GAAP).
Material Uncertainty Related to Going
Concern
The accompanying consolidated financial
statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the consolidated
financial statements, the Company’s recurring losses from operations raise substantial doubt about its ability to continue
as a going concern. Management’s plans concerning these matters are also discussed in Note 1 to the consolidated financial
statements. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements
are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated
financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight
Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal
securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with
the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether
the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required
to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are
required to obtain an understanding of internal control over financial reporting, but not for the purpose of expressing an opinion
on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures
to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing
procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and
disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant
estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe
that our audits provide a reasonable basis for our opinion.
|
/s/ MNP LLP
|
|
|
|
Chartered Professional Accountants
Licensed Professional Accountants
We have served as the Company's auditor
since 2015.
Toronto, Canada
|
|
February 26, 2020
|
|
Zomedica Pharmaceuticals Corp.
|
|
Consolidated balance sheets
|
|
As at December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
|
|
|
|
December 31,
|
|
|
|
December 31,
|
|
|
|
|
Note
|
|
|
2019
|
|
|
|
2018
|
|
|
|
|
|
|
|
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current assets:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
|
|
|
|
$
|
510,586
|
|
|
$
|
1,940,265
|
|
|
Prepaid expenses, deposits and deferred financing costs
|
|
|
5
|
|
|
|
1,228,585
|
|
|
|
1,867,034
|
|
|
Tax credits
receivable
|
|
|
|
|
|
|
67,618
|
|
|
|
53,659
|
|
|
|
|
|
|
|
|
|
1,806,789
|
|
|
|
3,860,958
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Prepaid expenses, deposits and deferred financing costs
|
|
|
5
|
|
|
|
-
|
|
|
|
1,442,415
|
|
|
Property and equipment
|
|
|
6
|
|
|
|
729,142
|
|
|
|
717,088
|
|
|
Right-of-use asset
|
|
|
8
|
|
|
|
1,103,658
|
|
|
|
-
|
|
|
Intangible assets
|
|
|
7
|
|
|
|
543,395
|
|
|
|
13,058
|
|
|
|
|
|
|
|
|
$
|
4,182,984
|
|
|
$
|
6,033,519
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and shareholders' equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current liabilities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued liabilities
|
|
|
|
|
|
$
|
2,087,525
|
|
|
$
|
2,376,519
|
|
|
|
|
|
|
|
|
|
2,087,525
|
|
|
|
2,376,519
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shareholders' equity:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Capital stock
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Series 1 preferred shares, without par value; 20 shares authorized (2018 - nil) Issued and outstanding 12 series 1 preferred shares (2018 - nil)
|
|
|
10
|
|
|
$
|
11,961,397
|
|
|
$
|
-
|
|
|
Unlimited common shares without par value; Issued and outstanding 108,038,398 common shares (2018 - 97,598,898)
|
|
|
11
|
|
|
|
38,566,820
|
|
|
|
30,410,648
|
|
|
Common stock subscribed
|
|
|
|
|
|
|
-
|
|
|
|
4,280,000
|
|
|
Additional paid-in capital
|
|
|
12
|
|
|
|
3,625,083
|
|
|
|
1,240,139
|
|
|
Accumulated deficit
|
|
|
|
|
|
|
(52,057,841
|
)
|
|
|
(32,273,787
|
)
|
|
|
|
|
|
|
|
|
2,095,459
|
|
|
|
3,657,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
4,182,984
|
|
|
$
|
6,033,519
|
|
Signed on behalf of the Board:
|
“Jeff Rowe”
|
“ Rod Williams”
|
|
Chairman of the Board
|
Director
|
Nature of operations and going concern (Note 1)
Commitments and contingencies (Note 15)
The accompanying notes are an integral part of these consolidated financial statements.
|
Zomedica Pharmaceuticals Corp.
|
|
Consolidated statements of operations and comprehensive loss
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
|
|
|
|
December 31,
|
|
|
|
December 31,
|
|
|
|
|
Note
|
|
|
2019
|
|
|
|
2018
|
|
|
|
|
|
|
|
|
|
|
Expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
18
|
|
|
$
|
10,345,291
|
|
|
$
|
10,317,153
|
|
|
General and administrative
|
|
|
18
|
|
|
|
7,114,777
|
|
|
|
4,521,349
|
|
|
Professional fees
|
|
|
18
|
|
|
|
1,536,646
|
|
|
|
1,534,977
|
|
|
Amortization - right-of-use asset
|
|
|
7
|
|
|
|
509,381
|
|
|
|
-
|
|
|
Amortization - intangible asset
|
|
|
7
|
|
|
|
1,082
|
|
|
|
2,083
|
|
|
Depreciation
|
|
|
6
|
|
|
|
277,150
|
|
|
|
203,684
|
|
|
Loss from operations
|
|
|
|
|
|
|
19,784,327
|
|
|
|
16,579,246
|
|
|
Loss on fixed assets
|
|
|
6
|
|
|
|
1,308
|
|
|
|
69,382
|
|
|
Interest expense
|
|
|
|
|
|
|
18,338
|
|
|
|
-
|
|
|
Gain on settlement of liabilities
|
|
|
|
|
|
|
(19,737
|
)
|
|
|
-
|
|
|
Foreign exchange gain
|
|
|
|
|
|
|
(182
|
)
|
|
|
(941
|
)
|
|
Loss before income taxes
|
|
|
|
|
|
|
19,784,054
|
|
|
|
16,647,687
|
|
|
Income tax expense
|
|
|
14
|
|
|
|
-
|
|
|
|
-
|
|
|
Net loss and comprehensive loss
|
|
|
|
|
|
$
|
19,784,054
|
|
|
$
|
16,647,687
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of common shares - basic and diluted
|
|
|
|
|
|
|
106,297,975
|
|
|
|
93,440,341
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss per share - basic and diluted
|
|
|
|
|
|
$
|
(0.19
|
)
|
|
$
|
(0.18
|
)
|
The accompanying notes are an integral part of these consolidated financial statements.
|
Zomedica Pharmaceuticals Corp.
|
|
Consolidated statements of shareholders’ equity
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
|
|
|
Series 1 preferred stock
|
|
Common stock
|
|
|
|
|
|
|
|
|
|
|
|
Note
|
|
|
Shares
|
|
|
|
Amount
|
|
|
|
Shares
|
|
|
|
Amount
|
|
|
|
Common stock subscribed
|
|
|
|
Additional paid-in capital
|
|
|
|
Accumulated deficit
|
|
|
|
Total
|
|
|
Balance at December 31, 2017
|
|
|
|
|
|
|
-
|
|
|
$
|
-
|
|
|
|
90,225,869
|
|
|
$
|
18,244,659
|
|
|
$
|
-
|
|
|
$
|
1,768,526
|
|
|
$
|
(15,626,100
|
)
|
|
$
|
4,387,085
|
|
|
Stock issuance for services
|
|
|
|
|
|
|
-
|
|
|
|
-
|
|
|
|
3,207,506
|
|
|
|
5,651,671
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
5,651,671
|
|
|
Stock issuance for financing, net of cost
|
|
|
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,861,627
|
|
|
|
3,944,336
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
3,944,336
|
|
|
Stock-based compensation
|
|
|
13
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
7,288
|
|
|
|
-
|
|
|
|
7,288
|
|
|
Stock issuance due to exercise of options
|
|
|
11,13
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,303,896
|
|
|
|
2,569,982
|
|
|
|
-
|
|
|
|
(535,675
|
)
|
|
|
-
|
|
|
|
2,034,307
|
|
|
Common stock subscribed
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4,280,000
|
|
|
|
|
|
|
|
|
|
|
|
4,280,000
|
|
|
Net loss
|
|
|
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(16,647,687
|
)
|
|
|
(16,647,687
|
)
|
|
Balance at December 31, 2018
|
|
|
|
|
|
|
-
|
|
|
$
|
-
|
|
|
|
97,598,898
|
|
|
$
|
30,410,648
|
|
|
$
|
4,280,000
|
|
|
$
|
1,240,139
|
|
|
$
|
(32,273,787
|
)
|
|
$
|
3,657,000
|
|
|
Stock issuance for services
|
|
|
11
|
|
|
|
-
|
|
|
|
-
|
|
|
|
707,236
|
|
|
|
792,104
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
792,104
|
|
|
Stock issuance for financing, net of cost
|
|
|
10,11
|
|
|
|
12
|
|
|
|
11,961,397
|
|
|
|
9,337,529
|
|
|
|
6,609,920
|
|
|
|
(4,280,000
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
14,291,317
|
|
|
Stock-based compensation
|
|
|
13
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,539,092
|
|
|
|
-
|
|
|
|
2,539,092
|
|
|
Stock issuance due to exercise of options
|
|
|
11,13
|
|
|
|
-
|
|
|
|
-
|
|
|
|
394,735
|
|
|
|
754,148
|
|
|
|
-
|
|
|
|
(154,148
|
)
|
|
|
-
|
|
|
|
600,000
|
|
|
Net loss
|
|
|
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(19,784,054
|
)
|
|
|
(19,784,054
|
)
|
|
Balance at December 31, 2019
|
|
|
|
|
|
|
12
|
|
|
$
|
11,961,397
|
|
|
|
108,038,398
|
|
|
$
|
38,566,820
|
|
|
$
|
-
|
|
|
$
|
3,625,083
|
|
|
$
|
(52,057,841
|
)
|
|
$
|
2,095,459
|
|
The accompanying notes are an integral part of these consolidated financial statements.
|
Zomedica Pharmaceuticals Corp.
|
|
Consolidated statements of cash flows
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
|
Note
|
|
|
2019
|
|
|
|
2018
|
|
|
|
|
|
|
|
|
|
|
Cash flows used in operating activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
|
|
|
|
$
|
(19,784,054
|
)
|
|
$
|
(16,647,687
|
)
|
|
Adjustments for
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation
|
|
|
6
|
|
|
|
277,150
|
|
|
|
203,684
|
|
|
Amortization - intangible assets
|
|
|
7
|
|
|
|
1,082
|
|
|
|
2,083
|
|
|
Amortization - right-of-use asset
|
|
|
|
|
|
|
509,381
|
|
|
|
-
|
|
|
Loss on fixed asets
|
|
|
|
|
|
|
1,308
|
|
|
|
69,382
|
|
|
Stock issued for services
|
|
|
11
|
|
|
|
792,104
|
|
|
|
5,651,671
|
|
|
Stock-based compensation
|
|
|
13
|
|
|
|
2,539,092
|
|
|
|
7,288
|
|
|
Change in non-cash operating working capital
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Trade and other receivable
|
|
|
|
|
|
|
(13,959
|
)
|
|
|
(25,387
|
)
|
|
Prepaid expenses
|
|
|
|
|
|
|
239,953
|
|
|
|
(124,230
|
)
|
|
Deposits
|
|
|
|
|
|
|
92,873
|
|
|
|
(1,832,114
|
)
|
|
Accounts payable and accrued liabilities
|
|
|
|
|
|
|
(288,994
|
)
|
|
|
1,547,782
|
|
|
|
|
|
|
|
|
|
(15,634,064
|
)
|
|
|
(11,147,528
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from financing activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash proceeds from issuance of preferred shares
|
|
|
10
|
|
|
|
12,000,000
|
|
|
|
-
|
|
|
Cash proceeds from issuance of common shares
|
|
|
11
|
|
|
|
3,000,000
|
|
|
|
8,282,496
|
|
|
Cash received from stock option exercises
|
|
|
|
|
|
|
600,000
|
|
|
|
2,034,307
|
|
|
Cash paid on stock issuance costs
|
|
|
|
|
|
|
(708,683
|
)
|
|
|
(58,160
|
)
|
|
|
|
|
|
|
|
|
14,891,317
|
|
|
|
10,258,643
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows used in investing activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash received from sale of fixed assets
|
|
|
6
|
|
|
|
-
|
|
|
|
9,000
|
|
|
Investment in intangibles
|
|
|
7
|
|
|
|
(531,419
|
)
|
|
|
-
|
|
|
Investment in property and equipment
|
|
|
6
|
|
|
|
(155,513
|
)
|
|
|
(627,997
|
)
|
|
|
|
|
|
|
|
|
(686,932
|
)
|
|
|
(618,997
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Decrease in cash and cash equivalents
|
|
|
|
|
|
|
(1,429,679
|
)
|
|
|
(1,507,882
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents, beginning of year
|
|
|
|
|
|
|
1,940,265
|
|
|
|
3,448,147
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents, end of year
|
|
|
|
|
|
$
|
510,586
|
|
|
$
|
1,940,265
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental cash flow information:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest Paid
|
|
|
|
|
|
$
|
18,338
|
|
|
$
|
-
|
|
The accompanying notes are an integral part of these consolidated financial statements.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
1.
|
Nature of operations and going concern
|
Zomedica Pharmaceuticals Corp. ("Zomedica"
or the “Company”) was incorporated on January 7, 2013 under the Business Corporations Act (Alberta) as Wise
Oakwood Ventures Inc. (“WOW”) and was classified as a capital pool company, as defined in Policy 2.4 of the TSX Venture
Exchange. ZoMedica Pharmaceuticals Inc. was incorporated on May 14, 2015 under the Canada Business Corporations Act.
On April 21, 2016, the Company closed its
qualifying transaction (“Transaction”), consisting of the acquisition of ZoMedica Pharmaceuticals Inc. (“ZoMedica”)
pursuant to a three-cornered amalgamation, whereby ZoMedica was amalgamated with 9674128 Canada Inc. (which was wholly-owned by
WOW) and common shares and options of the Company were issued to former holders of ZoMedica securities as consideration. The amalgamated
company changed its name to Zomedica Pharmaceuticals Ltd. and WOW subsequently changed its name to Zomedica Pharmaceuticals Corp.
Prior to completion of the Transaction, WOW consolidated its common shares on the basis of the one post-consolidation common share
for every 2.5 pre-consolidation common shares. The Transaction constituted WOW’s qualifying transaction under TSX Venture
Exchange Policy 2.4 – Capital Pool Companies. The shares of Zomedica Pharmaceuticals Corp. began trading on the TSX
Venture Exchange under the new symbol “ZOM” on Monday, May 2, 2016. On June 21, 2016, the Company filed Articles of
Amalgamation and vertically amalgamated with its wholly-owned subsidiary, Zomedica Pharmaceuticals Ltd.
Zomedica has one corporate subsidiary,
Zomedica Pharmaceuticals, Inc., a Delaware company whose results and operations are included in these consolidated financial statements.
The Company is a biopharmaceutical company targeting health and wellness solutions for the companion pet through a ground-breaking
approach that focuses on the needs of the veterinarians themselves. Zomedica's head office is located at 100 Phoenix Drive, Suite
180, Ann Arbor, MI 48108 and its registered office is located at 3400, 350-7th Ave SW, Calgary, AB, T2P 3N9.
On November 20, 2017, Zomedica announced
that its registration statement on Form S-1 was declared effective by the U.S. Securities and Exchange Commission (SEC) and on
November 21, 2017, the Company’s common shares began trading on the NYSE under the symbol “ZOM”.
Going concern
The consolidated financial statements are prepared on a going concern
basis, which assumes that the Company will be able to realize its assets and meet its obligations in the normal course of operations
for the foreseeable future. The Company has incurred losses from operations since inception and has an accumulated deficit of
$52,057,841 as at December 31, 2019 (December 31, 2018 - $32,273,787). The Company has funded its research and development (“R&D”)
activities principally through the issuance of securities and loans from related parties. There is no certainty that such funding
will be available going forward. These conditions raise substantial doubt about its ability to continue as a going concern and
realize its assets and pay its liabilities as they become due.
In order for the Company to continue as a going concern and fund
any significant expansion of its operations or R&D activities, the Company will require significant additional capital. The
Company’s ultimate success will depend on whether its future product candidates receive the necessary regulatory approval
and it is able to successfully market approved products. The Company cannot be certain that it will be able to receive regulatory
approval for any of its future product candidates, or that it will reach the level of sales and revenues necessary to achieve
and sustain profitability.
The availability of equity or debt financing
will be affected by, among other things, the results of the Company’s research and development, its ability to obtain regulatory
approvals, the market acceptance of its products, the state of the capital markets generally, strategic alliance agreements, and
other relevant commercial considerations. In addition, if the Company raises additional funds by issuing equity securities,
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
1.
|
Nature of operations and going concern
(continued)
|
Going concern (continued)
its then existing security holders will likely experience dilution,
and the incurring of indebtedness would result in increased debt service obligations and could require the Company to agree to
operating and financial covenants that would restrict its operations. Any failure on its part to raise additional funds on terms
favorable to the Company or at all, may require the Company to significantly change or curtail its current or planned operations
in order to conserve cash until such time, if ever, that sufficient proceeds from operations are generated, and could result in
the Company not taking advantage of business opportunities. There is no certainty the Company will be able to raise the necessary
funds to continue operations as a going concern. These financial statements do not reflect adjustments, if any, which would be
required to the carrying amounts or classification of assets and liabilities, or the amounts of reported expenses, should the
use of the going concern assumption be determined not be appropriate. Such adjustments, if any, could be material.
The accounting policies set out below have been applied consistently
in the consolidated financial statements. The consolidated financial statements have been prepared in conformity with accounting
principles generally accepted in the United States of America (“U.S. GAAP”).
Basis of consolidation
These consolidated financial statements
include the accounts of the Company and its wholly owned operating subsidiary, Zomedica Pharmaceuticals, Inc.
All inter-company accounts and transactions have been eliminated
on consolidation.
|
|
3.
|
Significant accounting policies
|
Use of estimates
The preparation of the consolidated financial statements in conformity
with U.S GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities
and disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts
of revenue and expenses during the period. Actual results could differ from those estimates.
Areas where significant judgment is involved in making estimates
are: the determination of fair value of stock-based compensation; the useful lives and recoverability of property and equipment;
and forecasting future cash flows for assessing the going concern assumption.
Basis of measurement
The consolidated financial statements have
been prepared on the historical cost basis except as otherwise noted.
Functional and reporting currencies
The Company’s and subsidiary’s
functional currency, as determined by management, is US dollars, which is also the Company’s reporting currency.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
3.
|
Significant accounting policies (continued)
|
Cash and cash equivalents
The Company considers all highly liquid
securities with an original maturity of three months or less to be cash equivalents. Cash and cash equivalents are comprised of
cash on hand and cash held in trust related to share issuances. The cash held in trust is readily available to the Company and
is classified as current.
The financial risks associated with these instruments are minimal
and the Company has not experienced any losses from investments in these securities.
Property and equipment
Property and equipment are carried at historical cost less accumulated
depreciation and any accumulated impairment losses. Maintenance and repair expenditures that do not improve or extend the life
are expensed in the period incurred.
Depreciation is recognized so as to write off the cost less their
residual values over their useful lives, using the straight-line method. The estimated useful lives, residual values and depreciation
methods are reviewed at the end of each year, with the effect of any changes in estimate accounted for on a prospective basis.
An item of property and equipment is derecognized
upon disposal or when no future economic benefits are expected to arise from the continued use of the asset. Any gain or loss arising
on the disposal or retirement of an item of property, plant and equipment is determined as the difference between the sales proceeds
and the carrying amount of the asset and is recognized in profit or loss.
Estimated useful lives for the principal
asset categories are as follows:
|
Computer equipment (years)
|
|
|
3
|
|
|
Furniture and equipment (years)
|
|
5
|
-
|
7
|
|
Laboratory equipment (years)
|
|
5
|
-
|
7
|
|
Leasehold improvements
|
|
Over shorter of estimated useful life and lease term
|
Impairment of long-lived assets
Long-lived assets are reviewed for impairment when events or circumstances
indicate that the carrying value of an asset may not be recoverable. For assets that are to be held and used, impairment is recognized
when the sum of estimated undiscounted future cash flows associated with the asset or group of assets is less than its carrying
value. If impairment exists, an adjustment is made to write the asset down to its fair value, and a loss is recorded as the difference
between the carrying value and fair value.
Research and development
Research and development costs related
to continued research and development programs are expensed as incurred in accordance with ASC topic 730.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
3.
|
Significant accounting policies (continued)
|
Share issue costs
Share issue costs are recorded as a reduction
of the proceeds from the issuance of capital stock.
Foreign currency
In respect of transactions denominated in currencies other than
the Company and its wholly owned operating subsidiaries’ functional currencies, the monetary assets and liabilities are remeasured
at the period end rates. Revenue and expenses are remeasured at rates of exchange prevailing on the transaction dates. All of the
exchange gains or losses resulting from these transactions are recognized in the consolidated statements of operations and comprehensive
loss.
Stock-based compensation
The Company measures the cost of equity-settled
transactions by reference to the fair value of the equity instruments at the date at which they are granted if the fair value of
the goods or services received by the Company cannot be reliably estimated.
The Company calculates stock-based compensation using the fair value
method, under which the fair value of the options at the grant date is calculated using the Black-Scholes Option Pricing Model,
and subsequently expensed over the vesting period of the option using the graded vesting method. The provisions of the Company's
stock-based compensation plans do not require the Company to settle any options by transferring cash or other assets, and therefore
the Company classifies the awards as equity. Stock-based compensation expense recognized during the period is based on the value
of stock-based payment awards that are ultimately expected to vest.
The Company estimates forfeitures at the
time of grant and revised, if necessary, in subsequent periods if actual forfeitures differ from those estimates.
Loss per share
Basic loss per share (“EPS”) is computed by dividing
the loss attributable to common shareholders by the weighted average number of common shares outstanding. Diluted EPS reflects
the potential dilution that could occur from common shares issuable through the exercise or conversion of stock options, restricted
stock awards, warrants and convertible securities. In certain circumstances, the conversion of options is excluded from diluted
EPS if the effect of such inclusion would be anti-dilutive.
The dilutive effect of stock options is determined using the treasury
stock method. Stock options to purchase common shares of the Company during fiscal 2019 and 2018 were not included in the computation
of diluted EPS because the Company has incurred a loss for the year ended December 31, 2019 and 2018 and the effect would be anti-dilutive.
Comprehensive loss
The Company follows ASC topic 220. This
statement establishes standards for reporting and display of comprehensive (loss) income and its components. Comprehensive loss
is net loss plus certain items that are recorded directly to shareholders' equity. The Company has no other comprehensive loss
items.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
3.
|
Significant accounting policies (continued)
|
Intangible assets
Intangible assets with finite useful lives
that are acquired separately are carried at cost less accumulated amortization and accumulated impairment losses. Amortization
is recognized on a straight-line basis over their estimated useful lives. The estimated useful lives and amortization methods are
reviewed at the end of each year, with the effect of any changes in estimate being accounted for on a prospective basis. Intangible
assets with indefinite useful lives that are acquired separately are carried at cost less accumulated impairment losses.
|
Computer software and website (years)
|
|
3
|
|
Trademarks (years)
|
|
15
|
The Company has not yet commenced the amortization
of the website as it is still in development.
Fair value measurement
Under ASC topic 820, fair value is defined
as the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants
at the measurement date (i.e., an exit price). ASC topic 820 establishes a hierarchy for inputs to valuation techniques used in
measuring fair value that maximizes the use of observable inputs and minimizes the use of unobservable inputs by requiring that
the most observable inputs be used when available. Observable inputs are inputs that reflect assumptions market participants would
use in pricing the asset or liability developed based on market data obtained from sources independent of the Company. Unobservable
inputs are inputs that reflect the Company's own assumptions about the assumptions market participants would use in pricing the
asset or liability developed based on the best information available in the circumstances. There are three levels to the hierarchy
based on the reliability of inputs, as follows:
|
|
l
|
Level 1 - Observable
inputs that reflect quoted prices (unadjusted) for identical assets or liabilities in active markets.
|
|
|
|
|
|
|
l
|
Level 2 - Inputs
other than quoted prices included in Level 1 that are observable for the asset or liability, either directly or indirectly. Level
2 inputs include quoted prices for similar assets or liabilities in active markets, or quoted prices for identical or similar assets
and liabilities in markets that are not active.
|
|
|
|
|
|
|
l
|
Level 3 - Unobservable
inputs for the asset or liability.
|
The degree of judgment exercised by the
Company in determining fair value is greatest for instruments categorized in Level 3.
Income taxes
The Company accounts for income taxes in
accordance with Accounting Standard Codification 740, Income Taxes ("ASC 740"), on a tax jurisdictional basis. The Company
files income tax returns in Canada and the province of Alberta and its subsidiary files income tax returns in the United States
and various states, including the headquarters in Michigan.
Deferred tax assets and liabilities are
recognized for the expected future tax consequences of temporary differences between the tax bases of assets and liabilities and
their financial statement reported amounts using enacted tax rates and laws in effect in the year in which the differences are
expected to reverse. A valuation allowance is provided against deferred tax assets when it is determined to be more likely than
not that the deferred tax asset will not be realized.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
3.
|
Significant accounting policies (continued)
|
Income taxes (continued)
The Company assesses the likelihood of the financial statement effect
of an uncertain tax position that should be recognized when it is more likely than not that the position will be sustained upon
examination by a taxing authority based on the technical merits of the tax position, circumstances, and information available as
of the reporting date. The Company is subject to examination by taxing authorities in jurisdictions such as the United States and
Canada. Management does not believe that there are any uncertain tax positions that would result in an asset or liability for taxes
being recognized in the accompanying consolidated financial statements. The Company recognizes tax-related interest and penalties,
if any, as a component of income tax expense.
ASC 740 prescribes recognition threshold
and measurement attributes for the financial statement recognition and measurement of a tax position taken or expected to be taken
in a tax return. ASC 740 also provides guidance on de-recognition, classification, interest and penalties, accounting in periods,
disclosure and transition. At December 31, 2019 and 2018, the Company has not taken any tax positions that would require disclosure
under ASC 740.
Segmented reporting
The Company currently operates as a single
segment. Its principal business relates to the discovery, development and commercialization of innovative pharmaceuticals for the
companion pet.
Recently adopted accounting pronouncements
In February 2016, the FASB issued new guidance, ASU No. 2016-02,
Leases (Topic 842). The new standard establishes a right-of-use model (“ROU”) that requires a lessee to recognize a
ROU asset and lease liability on the balance sheet for all leases with a term longer than 12 months. Leases will be classified
as finance or operating, with classification affecting the pattern and classification of expense recognition in the income statement.
Additional qualitative and quantitative disclosures are also required by the new guidance.
A modified retrospective transition approach
is required, applying the new standard to all leases existing at the date of initial application. The Company adopted the new standard
with an initial application date of January 1, 2019 and used the effective date as its date of initial application. Consequently,
financial information was not updated, and the disclosures required under the new standard were not provided for dates and periods
before January 1, 2019.
The new standard provides a number of optional
practical expedients in transition. The Company has elected the ‘package of practical expedients’, which permits the
Company not to reassess under the new standard prior conclusions about lease identification, lease classification and initial direct
costs. The Company has not elected the use-of-hindsight or the practical expedient pertaining to land easements; the latter not
being applicable to the Company.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
3.
|
Significant accounting policies (continued)
|
Recently adopted accounting pronouncements (continued)
On August 29, 2018, the FASB issued ASU 2018-15, which amends ASC
350-40 to address a customer’s accounting for implementation costs incurred in a cloud computing arrangement (CCA) that
is a service contract. ASU 2018-15 aligns the accounting for costs incurred to implement a CCA that is a service arrangement with
the guidance on capitalizing costs associated with developing or obtaining internal-use software. Specifically, the ASU amends
ASC 350 to include in its scope implementation costs of a CCA that is a service contract and clarifies that a customer should
apply ASC 350-40 to determine which implementation costs should be capitalized in a CCA that is considered a service contract.
The amendments in this update are effective for public business entities for fiscal years beginning after December 15, 2019. Early
adoption is permitted. The Company chose to early adopt this guidance on July 1, 2019 using the prospective transition method.
|
|
4.
|
Critical accounting judgments and key sources of estimation uncertainty
|
The preparation of financial statements
requires management to make judgments, estimates and assumptions that affect the application of policies and reported amounts of
assets and liabilities, and revenue and expenses. The estimates and associated assumptions are based on historical experience and
various other factors that are believed to be reasonable under the circumstances, the results of which form the basis of making
the judgments about carrying values of assets and liabilities that are not readily apparent from other sources. Actual results
may differ from these estimates.
The estimates and underlying assumptions
are reviewed on an ongoing basis. Revisions to accounting estimates are recognized in the period in which the estimate is revised
if the revision affects only that period or in the period of the revision and further periods if the review affects both current
and future periods.
Critical areas of estimation and judgements
in applying accounting policies include the following:
Going concern
These consolidated financial statements
have been prepared in accordance with U.S GAAP on a going concern basis, which assumes the realization of assets and discharge
of liabilities in the normal course of business within the foreseeable future. Management uses judgment in determining assumptions
for cash flow projections, such as anticipated financing, anticipated sales and future commitments to assess the Company’s
ability to continue as a going concern. A critical judgment is that the Company continues to raise funds going forward and satisfy
their obligations as they become due.
Useful lives and recoverability of property
and equipment
As described in Note 3 above, the Company
reviews the estimated useful lives of property and equipment with definite useful lives at the end of each year and assesses whether
the useful lives of certain items should be shortened or extended, due to various factors including technology, competition and
revised service offerings. During the year ended December 31, 2019 and 2018, the Company was not required to adjust the useful
lives of any assets based on the factors described above. Long-lived assets are reviewed for impairment when events or circumstances
indicate that the carrying value of an asset may not be recoverable. During the year ended December 31, 2019 and 2018, The Company
did not identify any events or circumstances to indicate that the carrying value of property and equipment was not recoverable.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
4.
|
Critical accounting judgments and key sources of estimation uncertainty (continued)
|
Stock-based payments
The Company estimates the fair value of convertible securities such
as options using the Black-Scholes option-pricing model which requires significant estimation around assumptions and inputs such
as expected life, expected volatility, estimated forfeiture rates and expected dividends.
|
|
5.
|
Prepaid expenses, deposits and deferred financing costs
|
|
|
|
|
2019
|
|
|
|
2018
|
|
|
Prepaid rent (i)
|
|
$
|
-
|
|
|
$
|
1,613,038
|
|
|
Deposits (ii)
|
|
|
1,033,231
|
|
|
|
1,596,104
|
|
|
Prepaid marketing (iii)
|
|
|
19,829
|
|
|
|
37,465
|
|
|
Prepaid insurance (iii)
|
|
|
110,636
|
|
|
|
33,372
|
|
|
Other (iv)
|
|
|
64,889
|
|
|
|
29,470
|
|
|
Total
|
|
$
|
1,228,585
|
|
|
$
|
3,309,449
|
|
|
|
(i)
|
On July 31, 2018 the Company entered into an amended lease agreement with Wickfield Phoenix, LLC
for an additional 18,640 square feet of office space. The Company prepaid the full outstanding balance of $1,269,073. As of January
1, 2019, the balance of the prepaid rent, inclusive of the original and amended lease amounts was $1,613,038. In accordance with
the Company’s adoption of ASC 842 on January 1, 2019, this amount was reclassified as a right-of-use asset in the consolidated
balance sheet. As of December 31, 2018, the Company classified $509,380 as a current asset in the consolidated balance sheet.;
|
|
|
(ii)
|
Deposits include payments made to vendors in advance and are primarily associated with research
activity, design fees for additional office space and equipment purchases. As of December 31, 2019, the Company classified all
amounts as a current asset. As of December 31, 2018, $1,257,347 was classified as a current asset in the consolidated balance sheet;
|
|
|
(iii)
|
As of December 31, 2019, and 2018, all amounts were classified as a current asset in the consolidated
balance sheet;
|
|
|
(iv)
|
Other is comprised of deferred financing costs, subscription payments
and software licensing. As of December 31, 2019, and 2018, the Company classified all amounts as a current asset in the consolidated
balance sheet.
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
6.
|
Property and equipment
|
|
|
|
|
Computer equipment
|
|
|
|
Furniture and equipment
|
|
|
|
Laboratory equipment
|
|
|
|
Leasehold improvements
|
|
|
|
Total
|
|
|
Cost
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2017
|
|
$
|
151,155
|
|
|
$
|
76,058
|
|
|
$
|
245,729
|
|
|
$
|
36,957
|
|
|
$
|
509,899
|
|
|
Additions
|
|
|
18,847
|
|
|
|
105,821
|
|
|
|
246,375
|
|
|
|
256,954
|
|
|
|
627,997
|
|
|
Disposals
|
|
|
-
|
|
|
|
-
|
|
|
|
(139,467
|
)
|
|
|
(10,936
|
)
|
|
|
(150,403
|
)
|
|
Balance at December 31, 2018
|
|
|
170,002
|
|
|
|
181,879
|
|
|
|
352,637
|
|
|
|
282,975
|
|
|
|
987,493
|
|
|
Additions
|
|
|
218,076
|
|
|
|
3,415
|
|
|
|
3,350
|
|
|
|
65,672
|
|
|
|
290,513
|
|
|
Disposals
|
|
|
(2,210
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(2,210
|
)
|
|
Balance at December 31, 2019
|
|
|
385,868
|
|
|
|
185,294
|
|
|
|
355,987
|
|
|
|
348,647
|
|
|
|
1,275,796
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated depreciation
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2017
|
|
|
42,802
|
|
|
|
11,845
|
|
|
|
74,875
|
|
|
|
9,220
|
|
|
|
138,742
|
|
|
Depreciation
|
|
|
62,116
|
|
|
|
17,740
|
|
|
|
86,368
|
|
|
|
37,460
|
|
|
|
203,684
|
|
|
Disposals
|
|
|
-
|
|
|
|
-
|
|
|
|
(61,547
|
)
|
|
|
(10,474
|
)
|
|
|
(72,021
|
)
|
|
Balance at December 31, 2018
|
|
|
104,918
|
|
|
|
29,585
|
|
|
|
99,696
|
|
|
|
36,206
|
|
|
|
270,405
|
|
|
Depreciation
|
|
|
88,417
|
|
|
|
26,617
|
|
|
|
68,519
|
|
|
|
93,597
|
|
|
|
277,150
|
|
|
Disposals
|
|
|
(901
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(901
|
)
|
|
Balance at December 31, 2018
|
|
|
192,434
|
|
|
|
56,202
|
|
|
|
168,215
|
|
|
|
129,803
|
|
|
|
546,653
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net book value as at:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2018
|
|
$
|
65,084
|
|
|
$
|
152,294
|
|
|
$
|
252,941
|
|
|
$
|
246,769
|
|
|
$
|
717,088
|
|
|
December 31, 2019
|
|
$
|
193,434
|
|
|
$
|
129,092
|
|
|
$
|
187,772
|
|
|
$
|
218,844
|
|
|
$
|
729,142
|
|
In August 2018, the Company relocated
part of its operations to a new building. Due to the relocation, leasehold improvements with a net book value of $462 were written
off and equipment with a net book value of $77,920 was sold for $9,000. The net loss on disposal recorded was $69,382.
In February 2019, the Company reclassified
$135,000 out of prepaid assets into property and equipment.
In May 2019, the Company disposed of assets
with a net book value of $1,308. A net loss on disposal was recorded in the consolidated statement of loss and comprehensive loss.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
|
|
Computer software
|
|
|
|
Trademarks
|
|
|
|
Website
|
|
|
|
Total intangible assets
|
|
|
Cost
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2017
|
|
$
|
5,143
|
|
|
$
|
16,236
|
|
|
$
|
-
|
|
|
$
|
21,379
|
|
|
Additions
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Balance at December 31, 2018
|
|
|
5,143
|
|
|
|
16,236
|
|
|
|
-
|
|
|
|
21,379
|
|
|
Additions
|
|
|
-
|
|
|
|
-
|
|
|
|
531,419
|
|
|
|
531,419
|
|
|
Balance at December 31, 2019
|
|
|
5,143
|
|
|
|
16,236
|
|
|
|
531,419
|
|
|
|
552,798
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated amortization
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2017
|
|
|
4,143
|
|
|
|
2,095
|
|
|
|
-
|
|
|
|
6,238
|
|
|
Amortization
|
|
|
1,000
|
|
|
|
1,083
|
|
|
|
-
|
|
|
|
2,083
|
|
|
Balance at December 31, 2018
|
|
|
5,143
|
|
|
|
3,178
|
|
|
|
-
|
|
|
|
8,321
|
|
|
Amortization
|
|
|
-
|
|
|
|
1,082
|
|
|
|
-
|
|
|
|
1,082
|
|
|
Balance at December 31, 2019
|
|
|
5,143
|
|
|
|
4,260
|
|
|
|
-
|
|
|
|
9,403
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net book value as at:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2018
|
|
$
|
-
|
|
|
$
|
13,058
|
|
|
$
|
-
|
|
|
$
|
13,058
|
|
|
December 31, 2019
|
|
$
|
-
|
|
|
$
|
11,975
|
|
|
$
|
531,419
|
|
|
$
|
543,395
|
|
The estimated future amortization of intangible is as follows:
|
2020
|
$
|
178,229
|
|
2021
|
|
178,229
|
|
2022
|
|
178,229
|
|
2023
|
|
1,089
|
|
2024
|
|
1,089
|
|
Total
|
$
|
536,865
|
As discussed in Note 3, the Company adopted ASC 842 with an initial
application date of January 1, 2019. The Company is party to two lease agreements under which it rents office and laboratory space.
The rent for both of these leases was prepaid upon inception and therefore at adoption the Company reclassified its prepaid lease
balances of $1,613,039 to a right-of-use asset.
The Company amortizes the asset on a straight-line basis over the
remaining life of the lease and records the expense in the consolidated statement of operations and comprehensive loss. During
the year ended 2019 and 2018, the Company recognized $509,381 and nil, respectively, in amortization expense in the consolidated
statements of operations and comprehensive loss.
On October 18, 2017, the Company entered into a loan arrangement
with a shareholder of the Company, pursuant to which such shareholder has agreed to provide a loan facility to the Company, whereby
the Company may borrow up to $5,000,000, with the proceeds to be used for working capital and general corporate purposes. The term
of the loan facility is five (5) years, with principal and interest payments being due only at the time of maturity. Under the
loan agreement, the Company may borrow in one or more advances, provided however that a minimum amount of $250,000 must be borrowed
at any one time and not more than two advances may occur per month. Interest shall accrue at a rate of fourteen percent (14%) per
annum, payable upon maturity. As of December 31, 2019, no amounts have been borrowed.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
The Company is authorized to issue up
to 20 shares of our Series 1 Preferred Shares, all without par value, and each having a stated value of $1,000,000. The Series
1 Preferred Shares do not have voting rights except to the extent required by applicable law and are not convertible into the
Company’s common shares. Holders of the Series 1 Preferred Shares will not be entitled to dividends but, in lieu thereof,
will receive Net Sales Returns (“Net Sales Returns” is defined as annual payments equal to 9 percent of Net Sales)
until such time as the holders have received total Net Sales Returns equal to 9 times the aggregate stated value of the outstanding
Series 1 Preferred Shares. The Company will have the right to redeem the outstanding Series 1 Preferred Shares at any time at
a redemption price equal to 9 times the aggregate stated value of the Series 1 Preferred Shares outstanding less the aggregate
amount of the Net Sales Returns paid (the “Redemption Amount”).
Upon any dissolution, liquidation or winding
up, whether voluntary or involuntary, holders of Series 1 Preferred Shares will be entitled to a liquidation preference equal to
the stated value of the Series 1 Preferred Shares less the Net Sales Returns paid on the Series 1 Preferred Shares.
In the event of a fundamental transaction
(defined to include an amalgamation, merger or other business combination transaction involving our company in which our shareholders
do not have the right to cast more than 50% of the votes that may be cast for the election of directors, or a sale, lease or other
disposition of the properties and/or assets of our company as an entirety or substantially as an entirety to a third party), the
holders of the Series 1 Preferred Shares will be entitled to receive consideration for their Series 1 Preferred Shares equal to
a multiple of the stated value of the Series 1 Preferred Shares ranging from 5.0 to 9.0 depending on the timing of the fundamental
transaction, subject to a cap equal to the redemption amount. The Company has assessed the likelihood of any Net Sales Payments
to the Series 1 Preferred shareholders to be remote.
Issued and outstanding preferred stock:
|
|
|
|
Number of preferred stock
|
|
|
|
Preferred stock amount
|
|
|
Balance at December 31, 2018
|
|
|
-
|
|
|
$
|
-
|
|
|
Stock issued from financing (i)
|
|
|
12
|
|
|
|
11,961,397
|
|
|
Balance at December 31, 2019
|
|
|
12
|
|
|
$
|
11,961,397
|
|
|
|
(i)
|
On May 9, 2019, the Company entered into subscription agreements to sell $12,000,000 of its Series
1 Preferred Shares to an accredited investor in a private placement at a purchase price of $1,000,000 per Series 1 Preferred Share;
$5,000,000 of the purchase price was paid on May 9, 2019 and the remaining $7,000,000 was paid on June 7, 2019. The Company recorded
$38,603 of share issuance costs as an offset to preferred stock in the year ended December 31, 2019.
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
The Company
is authorized to issue an unlimited number of common shares, all without par value.
Issued and outstanding common
stock:
|
|
|
|
Number of common stock
|
|
|
|
Common stock amount
|
|
|
Balance at December 31, 2017
|
|
|
90,225,869
|
|
|
$
|
18,244,659
|
|
|
Stock issuance for services (i and iii)
|
|
|
3,207,506
|
|
|
|
5,651,671
|
|
|
Stock issued from financing, net of costs (ii)
|
|
|
1,861,627
|
|
|
|
3,944,336
|
|
|
Stock issuance due to exercise of options
|
|
|
2,303,896
|
|
|
|
2,569,982
|
|
|
Balance at December 31, 2018
|
|
|
97,598,898
|
|
|
$
|
30,410,648
|
|
|
Stock issuance for services (iv and v)
|
|
|
707,236
|
|
|
|
792,104
|
|
|
Stock issuance from financing (vi and vii)
|
|
|
9,337,529
|
|
|
|
6,609,920
|
|
|
Stock issuance due to exercise of options
|
|
|
394,735
|
|
|
|
754,148
|
|
|
Balance at December 31, 2019
|
|
|
108,038,398
|
|
|
$
|
38,566,820
|
|
|
|
i)
|
On May 10, 2018, the Company issued 641,717 common shares in accordance with a Development, Commercialization
and Exclusive Distribution Agreement with a third party and recognized $1,238,513 as a research and development expense in the
consolidated statements of operations and comprehensive loss.
|
|
|
ii)
|
On May 15, 2018, the Company issued 255,815 common shares for gross proceeds of $550,000. On June 28, 2018, the Company issued
1,605,812 common shares for gross proceeds of $3,452,496. The Company recorded $56,160 of share issuance costs as an offset to
capital stock.
|
|
|
iii)
|
On November 26, 2018, the Company issued 2,565,789 common shares in accordance with a Development
and Supply Agreement with a third party and recognized $4,413,158 as a research and development expense in the consolidated statements
of operations and comprehensive loss.
|
|
|
iv)
|
On January 14, 2019, the Company settled $75,000 of amounts due to a vendor by issuing 49,342 common
shares valued at $55,263 at the date of issuance. The Company recorded a $19,737 gain on the settlement of liabilities in the consolidated
statement of loss and comprehensive loss;
|
|
|
v)
|
On January 14, 2019, the Company issued 657,894 common shares in satisfaction of $1,000,000 of
all remaining milestones under a License and Supply Agreement with a third party. The Company recognized $736,841 as research and
development expense, based on the value of the common stock on the date of issuance;
|
|
|
vi)
|
On January 14, 2019, the Company completed a non-brokered private placement, and issued 2,815,789
common shares. Gross proceeds of $4,280,000 were received prior to December 31, 2018.The Company recorded $465 of share issuance
costs as an offset to common stock;
|
|
|
vii)
|
On March 28, 2019, the Company completed an underwritten public offering of its common stock pursuant
to which the Company sold an aggregate 6,521,740 common shares for gross proceeds of $3,000,000. The Company recorded $669,615
of share issuance costs as an offset to common stock in the year ended December 31, 2019.
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
12.
|
Common stock subscribed
|
During December 2018, the Company offered
its common shares in a private offering of 6,578,947 common shares at a price of $1.52 per share, for aggregate gross proceeds
of up to $10,000,000. The Company received subscription funds in the aggregate amount of $4,280,000 from investors. These
common shares were not issued until after December 31, 2018.
|
|
13.
|
Stock-based compensation
|
During the year ended December 31, 2019,
the Company issued 7,495,000 stock options, each option entitling the holder to purchase on common share of the Company. During
the year ended December 31, 2019, 394,735 options were exercised.
During the year ended December 31, 2018,
the Company issued nil stock options. During the year ended December 31, 2018, an aggregate of 2,303,896 options were exercised.
The continuity of stock options are as
follows:
|
|
|
|
Number of
Options
|
|
|
|
Weighted
Avg Exercise Price
|
|
|
Balance at December 31, 2017
|
|
|
8,080,000
|
|
|
$
|
1.21
|
|
|
Stock options exercised January 8, 2018
|
|
|
(124,000
|
)
|
|
|
0.20
|
|
|
Stock options exercised January 26, 2018
|
|
|
(100,000
|
)
|
|
|
0.20
|
|
|
Stock options exercised March 8, 2018
|
|
|
(50,000
|
)
|
|
|
0.19
|
|
|
Stock options exercised March 13, 2018
|
|
|
(176,000
|
)
|
|
|
0.19
|
|
|
Stock options exercised March 22, 2018
|
|
|
(50,000
|
)
|
|
|
0.19
|
|
|
Stock options exercised March 26, 2018
|
|
|
(240,000
|
)
|
|
|
0.19
|
|
|
Stock options exercised March 28, 2018
|
|
|
(325,000
|
)
|
|
|
0.19
|
|
|
Stock options exercised March 29, 2018
|
|
|
(562,996
|
)
|
|
|
2.13
|
|
|
Stock options exercised April 20, 2018
|
|
|
(154,000
|
)
|
|
|
0.20
|
|
|
Stock options expired April 21, 2018
|
|
|
(1,946,000
|
)
|
|
|
0.20
|
|
|
Stock options expired June 9, 2018
|
|
|
(100,000
|
)
|
|
|
1.16
|
|
|
Stock options expired June 21, 2018
|
|
|
(400,000
|
)
|
|
|
1.13
|
|
|
Stock options expired August 14, 2018
|
|
|
(75,000
|
)
|
|
|
2.10
|
|
|
Stock options exercised September 27, 2018
|
|
|
(85,000
|
)
|
|
|
1.45
|
|
|
Stock options expired September 28, 2018
|
|
|
(5,000
|
)
|
|
|
2.12
|
|
|
Stock options exercised on October 11, 2018
|
|
|
(200,000
|
)
|
|
|
1.15
|
|
|
Stock options expired November 12, 2018
|
|
|
(250,000
|
)
|
|
|
2.08
|
|
|
Stock options expired November 12, 2018
|
|
|
(600,000
|
)
|
|
|
1.14
|
|
|
Stock options exercised on November 29, 2018
|
|
|
(175,000
|
)
|
|
|
1.13
|
|
|
Stock options exercised on December 20, 2018
|
|
|
(26,900
|
)
|
|
|
1.11
|
|
|
Stock options expired December 21, 2018
|
|
|
(1,978,100
|
)
|
|
|
1.11
|
|
|
Stock options exercised on December 21, 2018
|
|
|
(35,000
|
)
|
|
|
1.11
|
|
|
Balance at December 31, 2018
|
|
|
422,004
|
|
|
$
|
1.95
|
|
|
Stock options granted January 10, 2019
|
|
|
5,995,000
|
|
|
|
1.52
|
|
|
Stock options expired February 24, 2019
|
|
|
(35,000
|
)
|
|
|
1.15
|
|
|
Stock options exercised March 8, 2019
|
|
|
(164,473
|
)
|
|
|
1.52
|
|
|
Stock options exercised March 15, 2019
|
|
|
(164,473
|
)
|
|
|
1.52
|
|
|
Stock options exercised March 29, 2019
|
|
|
(65,789
|
)
|
|
|
1.52
|
|
|
Stock options expired May 23, 2019
|
|
|
(10,000
|
)
|
|
|
1.52
|
|
|
Stock options expired June 16, 2019
|
|
|
(40,000
|
)
|
|
|
1.52
|
|
|
Stock options cancelled July 14, 2019
|
|
|
(5,000
|
)
|
|
|
1.52
|
|
|
Stock options cancelled August 13, 2019
|
|
|
(5,000
|
)
|
|
|
1.52
|
|
|
Stock options expired August 14, 2019
|
|
|
(387,004
|
)
|
|
|
2.11
|
|
|
Stock options granted August 19, 2019
|
|
|
500,000
|
|
|
|
0.26
|
|
|
Stock options granted August 19, 2019
|
|
|
100,000
|
|
|
|
0.35
|
|
|
Stock options granted August 19, 2019
|
|
|
100,000
|
|
|
|
0.45
|
|
|
Stock options granted August 19, 2019
|
|
|
100,000
|
|
|
|
0.55
|
|
|
Stock options granted August 19, 2019
|
|
|
100,000
|
|
|
|
0.65
|
|
|
Stock options granted August 19, 2019
|
|
|
100,000
|
|
|
|
0.75
|
|
|
Stock options granted September 16, 2019
|
|
|
500,000
|
|
|
|
0.43
|
|
|
Balance at December 31, 2019
|
|
|
7,040,265
|
|
|
$
|
1.28
|
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
13.
|
Stock-based compensation (continued)
|
As at December 31, 2018, details of the issued and outstanding stock
options are as follows:
|
Grant date
|
|
|
Exercise price (CDN$)
|
|
|
|
Number of options issued and outstanding
|
|
|
|
Number of vested options outstanding
|
|
|
|
Weighted Avg Remaining Life (years)
|
|
|
February 24, 2017
|
|
$
|
1.50
|
|
|
|
35,000
|
|
|
|
35,000
|
|
|
|
0.15
|
|
|
August 14, 2017
|
|
$
|
2.75
|
|
|
|
387,004
|
|
|
|
387,004
|
|
|
|
0.62
|
|
|
Balance at December 31, 2018
|
|
|
|
|
|
|
422,004
|
|
|
|
422,004
|
|
|
|
|
|
As at December 31, 2019, details of the
issued and outstanding stock options are as follows:
|
Grant date
|
|
|
Exercise price
(USD$)
|
|
|
|
Number of
options
|
|
|
|
Number of
vested options
|
|
|
|
Weighted Avg
Remaining Life
(years)
|
|
|
January 10, 2019
|
|
$
|
1.52
|
|
|
|
5,540,265
|
|
|
|
5,540,265
|
|
|
|
1.03
|
|
|
August 19, 2019
|
|
|
0.26
|
|
|
|
500,000
|
|
|
|
500,000
|
|
|
|
1.64
|
|
|
August 19, 2019
|
|
|
0.35
|
|
|
|
100,000
|
|
|
|
100,000
|
|
|
|
1.64
|
|
|
August 19, 2019
|
|
|
0.45
|
|
|
|
100,000
|
|
|
|
100,000
|
|
|
|
1.64
|
|
|
August 19, 2019
|
|
|
0.55
|
|
|
|
100,000
|
|
|
|
100,000
|
|
|
|
1.64
|
|
|
August 19, 2019
|
|
|
0.65
|
|
|
|
100,000
|
|
|
|
100,000
|
|
|
|
1.64
|
|
|
August 19, 2019
|
|
|
0.75
|
|
|
|
100,000
|
|
|
|
100,000
|
|
|
|
1.64
|
|
|
September 16, 2019
|
|
|
0.43
|
|
|
|
500,000
|
|
|
|
500,000
|
|
|
|
1.71
|
|
|
Balance at December 31, 2019
|
|
|
|
|
|
|
7,040,265
|
|
|
|
7,040,265
|
|
|
|
|
|
The Company granted 7,495,000 stock options during the
year ended December 2019 (December 31, 2018 - $nil).. The fair value of options granted during the year ended December 31, 2019
was estimated using the Black-Scholes option pricing model to determine the fair value of options granted using the following
assumptions:
|
|
|
January 10, 2019
|
|
August 19, 2019
|
|
September 16, 2019
|
|
Volatility
|
|
|
68%
|
|
|
|
|
87%
|
|
|
|
|
89%
|
|
|
Risk-free interest rate
|
|
|
2.56%
|
|
|
|
|
1.48%
|
|
|
|
|
1.74%
|
|
|
Expected life (years)
|
|
|
2
|
|
|
|
|
2
|
|
|
|
|
2
|
|
|
Dividend yield
|
|
|
0
|
|
|
|
|
0
|
|
|
|
|
0
|
|
|
Common share price
|
|
|
$1.23
|
|
|
$
|
|
0.26
|
|
|
|
|
$0.42
|
|
|
Strike price
|
|
|
$1.52
|
|
|
|
$ 0.26
|
-
|
$ 0.75
|
|
|
|
$0.43
|
|
|
Forfeiture rate
|
|
|
nil
|
|
|
|
|
nil
|
|
|
|
|
nil
|
|
The
Company recorded $2,539,092 stock-based compensation for the year ended December 31, 2019 and $7,288 of stock-based compensation
for the year ended December 31, 2018. During the year ended December 31, 2019, the Company recorded the cash receipt of $600,000
as common stock and reclassified $154,148 of stock-based compensation to common stock due to the exercise of 394,735 options disclosed
above. During the year ended December 31, 2018, the Company recorded the cash receipt of $2,034,307 as common stock and reclassified
$535,675 of stock-based compensation to common stock due to the exercise of 2,303,896 options disclosed above.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
The reconciliation of the combined
Canadian federal and provincial statutory income tax rate of 27% (2018- 27%) to the effective tax rate is as follows:
|
|
|
|
For the year ended December 31, 2019
|
|
|
|
For the year ended December 31, 2018
|
|
|
Loss before income taxes
|
|
$
|
(19,784,054
|
)
|
|
$
|
(16,647,687
|
)
|
|
Expected income tax expense (recovery)
|
|
|
(5,341,690
|
)
|
|
|
(4,494,880
|
)
|
|
Difference in foreign tax rates
|
|
|
54,660
|
|
|
|
53,280
|
|
|
Tax rate changes and other adjustments
|
|
|
-
|
|
|
|
(850,310
|
)
|
|
Stock based compensation and non-deductible expenses
|
|
|
771,640
|
|
|
|
(312,810
|
)
|
|
Prior Period Adjustments
|
|
|
261,160
|
|
|
|
-
|
|
|
Share issuance costs recorded in equity
|
|
|
(198,930
|
)
|
|
|
-
|
|
|
Change in valuation allowance
|
|
|
4,453,160
|
|
|
|
5,604,720
|
|
|
Total income tax expense
|
|
$
|
-
|
|
|
$
|
-
|
|
The following
table summarizes the components of deferred tax:
|
Deferred Tax Assets
|
|
|
2019
|
|
|
|
2018
|
|
|
Intangible assets - licenses
|
|
$
|
3,622,890
|
|
|
$
|
2,105,660
|
|
|
Share issuance costs
|
|
|
301,180
|
|
|
|
171,590
|
|
|
Reserves
|
|
|
20,410
|
|
|
|
18,650
|
|
|
Non-capital losses carried forward - Canada
|
|
|
5,498,910
|
|
|
|
3,605,540
|
|
|
Net operating losses carried forward - US
|
|
|
4,154,520
|
|
|
|
2,965,930
|
|
|
Investment Tax Credits
|
|
|
27,330
|
|
|
|
192,760
|
|
|
Operating leases
|
|
|
6,560
|
|
|
|
-
|
|
|
Donations
|
|
|
350
|
|
|
|
-
|
|
|
Total deferred tax assets
|
|
$
|
13,632,310
|
|
|
$
|
9,060,130
|
|
|
|
|
|
|
|
|
|
|
|
|
Deferred Tax Liabilities
|
|
|
|
|
|
|
|
|
|
Property and equipment
|
|
$
|
(231,240
|
)
|
|
$
|
(112,220
|
)
|
|
Total deferred tax liabilities
|
|
$
|
(231,240
|
)
|
|
$
|
(112,220
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Valuation allowance
|
|
$
|
13,401,070
|
|
|
$
|
8,947,910
|
|
|
Net deferred tax asset
|
|
$
|
-
|
|
|
$
|
-
|
|
No deferred
tax asset has been recognized, as it is not more likely than not to be realized. Consequently, a valuation allowance has been applied
against the net deferred tax asset. The Canadian non-capital loss carry forwards expire as noted in the table below.
|
2036
|
|
$
|
3,758,060
|
|
|
2037
|
|
|
4,278,990
|
|
|
2038
|
|
|
5,421,880
|
|
|
2039
|
|
|
6,907,680
|
|
|
Total
|
|
$
|
20,366,610
|
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
14. Income taxes (continued)
The
Company’s US non-operating income tax losses expire as follows:
|
2035
|
|
$
|
856,301
|
|
|
2036
|
|
|
1,484,636
|
|
|
2037
|
|
|
3,831,764
|
|
|
indefinitely
|
|
|
9,967,643
|
|
|
Total
|
|
$
|
16,140,344
|
|
|
15.
|
Commitments and contingencies
|
On October 1, 2018 the Company entered
into a one-year rental agreement. The Company elected not to account for these leases in accordance with ASC 842 as they are for
a one-year term. The total future annual lease payments for the premises are as follows:
|
2020
|
|
$
|
33,280
|
|
Total
|
|
$
|
33,280
|
On November 26, 2018, the Company entered
into a Development and Supply Agreement and as part of this agreement, the Company has contingent future outflows as follows:
|
|
o
|
1st payment: At the later of the achievement of a future milestone event or March 15,
2019 - $2,000,000 in cash
|
|
|
·
|
2nd payment: At the later of the achievement of a future milestone event or March 15,
2019, can decide to receive payment as follows:
|
|
|
o
|
$1,500,000 in cash and $1.95 million in equity
|
|
|
o
|
3rd payment: At the later of the achievement of a future milestone event or September
12, 2019, can decide to receive payment as follows:
|
|
|
o
|
$1,500,000 in cash and $1.95 million in equity
|
|
|
o
|
4th payment: At the later of the achievement of a future milestone or February 19, 2020
- $2,000,000 in cash.
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
15.
|
Commitments and contingencies (continued)
|
As at December
31, 2019, the first and second milestone payments have been made. Neither the 3rd nor 4th events related to the above agreement
have been met as of December 31, 2019.
On May 10,
2018, the Company entered into a Development, Commercialization and Exclusive Distribution Agreement. As part of the agreement,
the Company is required to make the following future milestone payments:
|
|
·
|
$3,500,000 in cash payment upon the achievement of future development milestones
|
|
|
·
|
$3,500,000 in equity based on the number of the Company’s common stock determined by dividing
the amount due by the volume-weighted average price of the Company’s common stock on the NYSE American exchange over the
10 trading days prior to the achievement of the milestone event.
|
As
at December 31, 2019, none of the future development milestones related to the above agreement have been met.
From time to time, the Company may be exposed
to claims and legal actions in the normal course of business. As at September 30, 2019, and continuing as at November 12, 2019,
the Company is not aware of any pending or threatened material litigation claims against the Company, other than as described below.
On November 1, 2019, Heska Corporation
(“Heska”) filed a complaint for damages and injunctive relief (the “Complaint”) in the United States District
Court for the Middle District of North Carolina, Case 1:19-cv-01108-LCB-JLW, against Qorvo US, Inc. (“Qorvo US”),
Qorvo Biotechnologies, LLC (“Qorvo Biotech” and, together with Qorvo US, “Qorvo”) and the Company (collectively
with Qorvo, the “Defendants”). The Complaint alleges, among other things, that the Defendants improperly obtained
Heska’s trade secrets and confidential information and/or conspired to use improper means to misappropriate Heska’s
trade secrets related to an instrument and related consumable products for performing immunoassay analysis of biomarkers and other
substances. The Complaint seeks compensatory and exemplary damages, as well as preliminary and permanent injunctive relief to
prevent the Defendants from commercializing the Company’s TRUFORMATM diagnostic instrument. On January 21, 2020,
the Defendants filed a motion seeking dismissal of the Complaint. On February 11, 2020, Heska filed its response to the Defendants’
motion to dismiss to which the Defendants responded on February 25, 2020. The Company believes that the allegations in the Complaint
have no merit and will not have a material adverse effect on the Company’s business, results of operations or financial
condition, and the Company reaffirms its intention to commence the commercialization of its TRUFORMATM platform by
the end of 2020.
Under the terms of the Development and
Supply Agreement, dated November 26, 2018, by and between Qorvo Biotech and the Company (the “Qorvo Agreement”), Qorvo
Biotech agreed to indemnify the Company and certain related parties against claims alleging infringement or misappropriation of
third-party intellectual property rights, subject to certain limitations and exceptions. Qorvo Biotech has notified the Company
that Qorvo Biotech has assumed the defense of the Complaint and will indemnify the Company for losses arising from the Complaint
in accordance with the terms of the Qorvo Agreement. Qorvo Biotech has further advised the Company that it intends to mount a
vigorous defense to the claims in the Complaint, and that it believes the allegations contained in the Complaint are without merit.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
16.
|
Financial instruments
|
The Company follows ASC topic 820, “Fair
Value Measurements” which defines fair value, establishes a framework for measuring fair value, and expands disclosures about
fair value measurements. The provisions of ASC topic 820 apply to other accounting pronouncements that require or permit fair value
measurements. ASC topic 820 defines fair value as the exchange price that would be received for an asset or paid to transfer a
liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between
market participants at the measurement date; and establishes a three level hierarchy for fair value measurements based upon the
transparency of inputs to the valuation of an asset or liability as of the measurement date. Inputs refers broadly to the assumptions
that market participants would use in pricing the asset or liability, including assumptions about risk. To increase consistency
and comparability in fair value measurements and related disclosures, the fair value hierarchy prioritizes the inputs to valuation
techniques used to measure fair value into three broad levels. The three levels of the hierarchy are defined as follows:
Level 1 inputs are quoted prices (unadjusted)
in active markets for identical assets or liabilities.
Level 2 inputs are inputs other than quoted
prices included within Level 1 that are observable for the asset or liability, either directly or indirectly for substantially
the full term of the financial instrument.
Level 3 inputs are unobservable inputs
for asset or liabilities.
The categorization within the valuation
hierarchy is based upon the lowest level of input that is significant to the fair value measurement.
|
|
(i)
|
The Company calculates expected volatility based on historical volatility of the Company’s
stock price. When there is insufficient data available, The Company uses a peer group that is publicly traded to calculate expected
volatility.
|
An increase/decrease in the volatility
would have resulted in an increase/decrease in the fair value of the options.
The carrying values of cash, trade and
other receivable, accounts payable and accrued liabilities and shareholder loans payable approximates their fair values because
of the short-term nature of these instruments.
|
|
(b)
|
Interest rate and credit risk
|
Interest rate risk is the risk that
the value of a financial instrument might be adversely affected by a change in interest rates. The Company does not believe that
the results of operations or cash flows would be affected to any significant degree by a sudden change in market interest rates,
relative to interest rates on cash and cash equivalents due to the short-term nature of these balances.
The Company is also exposed to credit risk
at period end from the carrying value of its cash. The Company manages this risk by maintaining bank accounts with a Canadian Chartered
Bank. The Company’s cash is not subject to any external restrictions.
|
|
(c)
|
Foreign exchange risk
|
The Company has balances in Canadian dollars
that give rise to exposure to foreign exchange (“FX”) risk relating to the impact of translating certain non-U.S. dollar
balance sheet accounts as these statements are presented in U.S. dollars. A strengthening U.S. dollar will lead to a FX loss while
a weakening U.S. dollar will lead to a FX gain. For each Canadian dollar balance of $1.0 million, a +/- 10% movement in the
Canadian currency held by the Company versus the U.S. dollar would affect the Company’s loss and other comprehensive
loss by $0.1 million.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
16.
|
Financial instruments (continued)
|
(d) Liquidity risk
Liquidity risk is the risk that the Company
will encounter difficulty raising liquid funds to meet commitments as they fall due. In meeting its liquidity requirements, the
Company closely monitors its forecasted cash requirements with expected cash drawdown.
The following are the contractual maturities
of the undiscounted cash flows of financial liabilities as at December 31, 2019 and 2018:
|
|
|
|
|
|
|
|
|
|
|
December 31, 2019
|
|
|
|
Less than
3 months
|
|
|
|
3 to 6
months
|
|
|
|
6 to 9
months
|
|
|
|
9 months
1 year
|
|
|
|
Greater than
1 year
|
|
|
|
Total
|
|
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
Third parties
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued liabilities
|
|
|
2,087,525
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,087,525
|
|
|
Related parties
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shareholder's loan payable
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
2,087,525
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,087,525
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2018
|
|
|
|
|
Less than
3 months
|
|
|
|
3 to 6
months
|
|
|
|
6 to 9
months
|
|
|
|
9 months
1 year
|
|
|
|
Greater than
1 year
|
|
|
|
Total
|
|
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
Third parties
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued liabilities
|
|
|
2,376,519
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,376,519
|
|
|
Related parties
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shareholder's loan payable
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
2,376,519
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,376,519
|
|
|
17.
|
Segmented information
|
The Company's operations comprise a single
reportable segment engaged in the research, development targeting health and wellness solutions for the companion pet. As the operations
comprise a single reportable segment, amounts disclosed in the financial statements for loss for the period, depreciation and total
assets also represent segmented amounts. In addition, all of the Company's long-lived assets are in the United States of America
(“US”).
|
|
|
|
|
|
December 31,
|
|
|
|
December 31,
|
|
|
|
|
|
|
|
2019
|
|
|
|
2018
|
|
|
|
|
|
|
|
$
|
|
|
|
$
|
|
|
Total assets
|
|
|
|
|
|
|
|
|
|
|
|
Canada
|
|
|
249,929
|
|
|
383,567
|
|
|
|
US
|
|
|
3,933,055
|
|
|
5,649,952
|
|
|
|
|
|
|
|
|
|
Total US property and equipment
|
|
|
|
|
729,142
|
|
|
717,088
|
|
Total US right-of-use asset
|
|
|
|
|
1,103,658
|
|
|
-
|
|
|
|
|
|
|
1,832,800
|
|
|
717,088
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
|
For the year ended December 31,
|
|
|
|
2019
|
|
|
|
|
Research and
|
|
|
|
Professional
|
|
|
|
General and
|
|
|
|
|
|
Development
|
|
|
|
Fees
|
|
|
|
Administrative
|
|
|
|
|
|
|
|
|
|
|
Salaries, bonus and benefits
|
|
$
|
789,848
|
|
|
$
|
-
|
|
|
$
|
5,447,455
|
|
|
Contracted expenditures
|
|
|
2,843,998
|
|
|
|
-
|
|
|
|
-
|
|
|
Marketing and investor relations
|
|
|
2,303
|
|
|
|
-
|
|
|
|
471,887
|
|
|
Travel and accommodation
|
|
|
37,181
|
|
|
|
-
|
|
|
|
435,530
|
|
|
Insurance
|
|
|
96,623
|
|
|
|
-
|
|
|
|
259,791
|
|
|
License fees
|
|
|
5,936,841
|
|
|
|
-
|
|
|
|
-
|
|
|
Office
|
|
|
50,599
|
|
|
|
-
|
|
|
|
327,258
|
|
|
Consultants
|
|
|
251,096
|
|
|
|
1,536,646
|
|
|
|
-
|
|
|
Regulatory
|
|
|
127,190
|
|
|
|
-
|
|
|
|
106,230
|
|
|
Rent
|
|
|
-
|
|
|
|
-
|
|
|
|
32,473
|
|
|
Supplies
|
|
|
209,612
|
|
|
|
-
|
|
|
|
34,153
|
|
|
Total
|
|
$
|
10,345,291
|
|
|
$
|
1,536,646
|
|
|
$
|
7,114,777
|
|
|
|
|
For the year ended December 31,
|
|
|
|
2018
|
|
|
|
|
Research and
|
|
|
|
Professional
|
|
|
|
General and
|
|
|
|
|
|
Development
|
|
|
|
Fees
|
|
|
|
Administrative
|
|
|
|
|
|
|
|
|
|
|
Salaries, bonus and benefits
|
|
$
|
692,913
|
|
|
$
|
-
|
|
|
$
|
2,593,686
|
|
|
Contracted expenditures
|
|
|
1,745,011
|
|
|
|
-
|
|
|
|
-
|
|
|
Marketing and investor relations
|
|
|
-
|
|
|
|
-
|
|
|
|
225,890
|
|
|
Travel and accommodation
|
|
|
21,251
|
|
|
|
-
|
|
|
|
266,147
|
|
|
Insurance
|
|
|
82,469
|
|
|
|
-
|
|
|
|
307,544
|
|
|
License fees
|
|
|
7,151,671
|
|
|
|
-
|
|
|
|
-
|
|
|
Office
|
|
|
69,299
|
|
|
|
-
|
|
|
|
365,395
|
|
|
Consultants
|
|
|
214,013
|
|
|
|
1,534,977
|
|
|
|
-
|
|
|
Regulatory
|
|
|
76,210
|
|
|
|
-
|
|
|
|
435,896
|
|
|
Rent
|
|
|
45,081
|
|
|
|
-
|
|
|
|
295,752
|
|
|
Supplies
|
|
|
219,235
|
|
|
|
-
|
|
|
|
31,039
|
|
|
Total
|
|
$
|
10,317,153
|
|
|
$
|
1,534,977
|
|
|
$
|
4,521,349
|
|
|
|
19.
|
Capital risk management
|
The capital of the Company includes equity,
which is comprised of issued common capital stock, common stock subscribed, additional paid-in capital, and accumulated deficit.
The Company's objective when managing its capital is to safeguard the ability to continue as a going concern in order to provide
returns for its shareholders, and other stakeholders and to maintain a strong capital base to support the Company's core activities.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the consolidated financial statements
|
|
For the years ended December 31, 2019 and 2018
|
|
(Stated in United States dollars)
|
|
|
|
|
For the year ended December 31, 2019
|
|
|
|
For the year ended December 31, 2018
|
|
|
|
|
|
|
|
|
Numerator
|
|
|
|
|
|
|
|
|
|
Net loss for the year
|
|
$
|
19,784,054
|
|
|
$
|
16,647,687
|
|
|
Denominator
|
|
|
|
|
|
|
|
|
|
Weighted average shares - basic
|
|
|
106,297,975
|
|
|
|
93,440,341
|
|
|
Stock options
|
|
|
-
|
|
|
|
-
|
|
|
Denominator for diluted loss per share
|
|
|
106,297,975
|
|
|
|
93,440,341
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss per share - basic and diluted
|
|
$
|
(0.19
|
)
|
|
$
|
(0.18
|
)
|
For the
above-mentioned periods, the Company had securities outstanding which could potentially dilute basic earnings per share in
the future but were excluded from the computation of diluted loss per share in the periods presented, as their effect would
have been anti-dilutive. The Company excluded 7,040,265 from the calculation of diluted earnings per share as their effect
would have been anti-dilutive.
|
|
21.
|
Related party transactions and key management compensation
|
As of the year ended December 31, 2019, the Company had outstanding
severance payments due to former Chairman of the board and former CEO, Gerald Solensky, Jr. for $169,143. As of the year ended
December 31, 2018, the Company had nil related party balances outstanding.
Key management personnel are comprised
of the Company’s directors and executive officers. In addition to their salaries, key management personnel also receive share-based
compensation. Key management personnel compensation is as follows:
|
|
|
|
For the year ended December 31, 2019
|
|
|
|
For the year ended December 31, 2018
|
|
|
Salaries and benefits, including bonuses
|
|
$
|
1,463,830
|
|
|
$
|
1,428,036
|
|
|
Stock-based compensation
|
|
|
1,842,313
|
|
|
|
-
|
|
|
Total
|
|
$
|
3,306,143
|
|
|
$
|
1,428,036
|
|
On February 14, 2020 the Company completed
a registered direct offering of 20,833,334 of its common shares at a purchase price of $0.12 per share, for gross proceeds of $2,500,000.
In addition, in a concurrent private placement, the Company issued to the investors warrants to purchase up to 20,833,334 common
shares, which represent 100% of the number of common shares issued in the registered direct offering, with an exercise price $0.20
per share and a five-year exercise period commencing six (6) months of the issuance date. The gross proceeds to from the offering,
before deducting the placement agent’s fees and other estimated offering expenses payable by the Company, are $2,500,000.
Zomedica Pharmaceuticals Corp.
Condensed unaudited interim consolidated financial statements
For the three and six months ended June 30, 2020 and 2019
(Expressed in United States Dollars, except as otherwise noted)
Zomedica Pharmaceuticals Corp.
Condensed unaudited interim consolidated balance sheets
As at June 30, 2020 and December 31, 2019
(Stated in United States dollars)
|
|
|
|
|
|
|
|
June 30,
|
|
|
|
December 31,
|
|
|
|
|
|
Note
|
|
|
|
2020
|
|
|
|
2019
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current assets:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
|
|
|
|
$
|
29,103,049
|
|
|
$
|
510,586
|
|
|
Prepaid expenses and deposits
|
|
|
5
|
|
|
|
782,647
|
|
|
|
1,228,585
|
|
|
Tax credits and other receivables
|
|
|
|
|
|
|
182,496
|
|
|
|
67,618
|
|
|
|
|
|
|
|
|
|
30,068,192
|
|
|
|
1,806,789
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Property and equipment
|
|
|
6
|
|
|
|
798,901
|
|
|
|
729,142
|
|
|
Right-of-use asset
|
|
|
8
|
|
|
|
1,441,124
|
|
|
|
1,103,658
|
|
|
Intangible assets
|
|
|
7
|
|
|
|
453,370
|
|
|
|
543,395
|
|
|
|
|
|
|
|
|
$
|
32,761,587
|
|
|
$
|
4,182,984
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and shareholders' equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current liabilities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued liabilities
|
|
|
|
|
|
$
|
1,731,469
|
|
|
$
|
2,087,525
|
|
|
Current portion of lease obligations
|
|
|
8
|
|
|
|
232,496
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
1,963,965
|
|
|
|
2,087,525
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Lease obligations
|
|
|
8
|
|
|
|
1,218,661
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
3,182,626
|
|
|
|
2,087,525
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shareholders' equity:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Capital stock
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Series 1 preferred shares, without par value;
|
|
|
|
|
|
|
|
|
|
|
|
|
|
20 shares authorized (2019 - 20)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issued and outstanding
|
|
|
|
|
|
|
|
|
|
|
|
|
|
12 series 1 preferred shares (2019 - 12)
|
|
|
10
|
|
|
|
11,961,397
|
|
|
|
11,961,397
|
|
|
Unlimited common shares without par value;
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issued and outstanding
|
|
|
|
|
|
|
|
|
|
|
|
|
|
361,039,946 common shares (2019 - 108,038,398)
|
|
|
11
|
|
|
|
67,328,922
|
|
|
|
38,566,820
|
|
|
Shares to be issued
|
|
|
12
|
|
|
|
1,465,500
|
|
|
|
-
|
|
|
Additional paid-in capital
|
|
|
13,14
|
|
|
|
8,639,590
|
|
|
|
3,625,083
|
|
|
Accumulated deficit
|
|
|
|
|
|
|
(59,816,448
|
)
|
|
|
(52,057,841
|
)
|
|
|
|
|
|
|
|
|
29,578,961
|
|
|
|
2,095,459
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
32,761,587
|
|
|
$
|
4,182,984
|
|
Commitments and contingencies (Note 15)
The accompanying notes are an integral part of these condensed unaudited
interim consolidated financial statements.
Zomedica Pharmaceuticals Corp.
Condensed unaudited interim consolidated statements of operations and comprehensive loss
For the three and six months ended June 30, 2020 and 2019
(Stated in United States dollars)
|
|
|
|
|
|
|
|
Three months ended June 30,
|
|
|
|
Six months ended June 30,
|
|
|
|
|
|
Note
|
|
|
|
2020
|
|
|
|
2019
|
|
|
|
2020
|
|
|
|
2019
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
18
|
|
|
$
|
3,908,171
|
|
|
$
|
1,061,507
|
|
|
$
|
4,503,570
|
|
|
$
|
8,592,882
|
|
|
General and administrative
|
|
|
18
|
|
|
|
988,734
|
|
|
|
901,319
|
|
|
|
2,272,261
|
|
|
|
4,113,677
|
|
|
Professional fees
|
|
|
18
|
|
|
|
282,791
|
|
|
|
231,647
|
|
|
|
573,472
|
|
|
|
989,946
|
|
|
Amortization - right-of-use asset
|
|
|
8
|
|
|
|
-
|
|
|
|
127,345
|
|
|
|
42,448
|
|
|
|
254,690
|
|
|
Amortization - intangible assets
|
|
|
7
|
|
|
|
44,990
|
|
|
|
270
|
|
|
|
90,025
|
|
|
|
537
|
|
|
Depreciation
|
|
|
6
|
|
|
|
77,859
|
|
|
|
68,925
|
|
|
|
154,275
|
|
|
|
130,979
|
|
|
Loss from operations
|
|
|
|
|
|
|
5,302,545
|
|
|
|
2,391,013
|
|
|
|
7,636,051
|
|
|
|
14,082,711
|
|
|
Interest income
|
|
|
|
|
|
|
(247
|
)
|
|
|
-
|
|
|
|
(328
|
)
|
|
|
-
|
|
|
Interest expense
|
|
|
|
|
|
|
-
|
|
|
|
12,164
|
|
|
|
732
|
|
|
|
18,338
|
|
|
Loss on disposal of property and equipment
|
|
|
6
|
|
|
|
-
|
|
|
|
1,308
|
|
|
|
69,834
|
|
|
|
1,308
|
|
|
Loss on right-of-use-asset
|
|
|
8
|
|
|
|
-
|
|
|
|
-
|
|
|
|
59,097
|
|
|
|
-
|
|
|
Gain on settlement of liabilities
|
|
|
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(19,737
|
)
|
|
Other income
|
|
|
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(5,500
|
)
|
|
|
-
|
|
|
Foreign exchange loss (gain)
|
|
|
|
|
|
|
5,692
|
|
|
|
(58
|
)
|
|
|
(1,279
|
)
|
|
|
(1,283
|
)
|
|
Loss before income taxes
|
|
|
|
|
|
|
5,307,990
|
|
|
|
2,404,427
|
|
|
|
7,758,607
|
|
|
|
14,081,337
|
|
|
Income tax expense
|
|
|
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Net loss and comprehensive loss
|
|
|
|
|
|
$
|
5,307,990
|
|
|
$
|
2,404,427
|
|
|
$
|
7,758,607
|
|
|
$
|
14,081,337
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of common shares - basic and diluted
|
|
|
|
|
|
|
214,830,818
|
|
|
|
108,038,398
|
|
|
|
166,814,645
|
|
|
|
104,528,705
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss per share - basic and diluted
|
|
|
|
|
|
$
|
(0.02
|
)
|
|
$
|
(0.02
|
)
|
|
$
|
(0.05
|
)
|
|
$
|
(0.13
|
)
|
The accompanying notes are an integral part of these condensed unaudited
interim consolidated financial statements.
Zomedica Pharmaceuticals Corp.
Condensed unaudited interim consolidated statements of shareholders’ equity
For the three and six months ended June 30, 2020 and 2019
(Stated in United States dollars)
|
|
|
|
|
Series 1 preferred stock
|
|
|
Common stock
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Note
|
|
Shares
|
|
|
Amount
|
|
|
Shares
|
|
|
Amount
|
|
|
Common
stock
subscribed
|
|
|
Shares to be
issued
|
|
|
Additional
paid-in
capital
|
|
|
Accumulated
deficit
|
|
|
Total
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2018
|
|
|
|
|
-
|
|
|
$
|
-
|
|
|
|
97,598,898
|
|
|
$
|
30,410,648
|
|
|
$
|
4,280,000
|
|
|
$
|
-
|
|
|
$
|
1,240,139
|
|
|
$
|
(32,273,787
|
)
|
|
$
|
3,657,000
|
|
|
Stock issuance for services
|
|
11
|
|
|
-
|
|
|
|
-
|
|
|
|
707,236
|
|
|
|
792,104
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
792,104
|
|
|
Stock-based compensation
|
|
13
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,341,104
|
|
|
|
-
|
|
|
|
2,341,104
|
|
|
Stock issuance for financing, net of cost
|
|
10,11
|
|
|
12
|
|
|
|
11,962,811
|
|
|
|
9,337,529
|
|
|
|
6,690,922
|
|
|
|
(4,280,000
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
14,373,733
|
|
|
Stock issued due to exercise of options
|
|
11,13
|
|
|
-
|
|
|
|
-
|
|
|
|
394,735
|
|
|
|
754,148
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(154,148
|
)
|
|
|
-
|
|
|
|
600,000
|
|
|
Net loss
|
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(14,081,337
|
)
|
|
|
(14,081,337
|
)
|
|
Balance at June 30, 2019
|
|
|
|
|
12
|
|
|
$
|
11,962,811
|
|
|
|
108,038,398
|
|
|
$
|
38,647,822
|
|
|
$
|
-
|
|
|
|
|
|
|
$
|
3,427,095
|
|
|
$
|
(46,355,124
|
)
|
|
$
|
7,682,604
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2019
|
|
|
|
|
12
|
|
|
$
|
11,961,397
|
|
|
|
108,038,398
|
|
|
$
|
38,566,820
|
|
|
$
|
-
|
|
|
$
|
-
|
|
|
$
|
3,625,083
|
|
|
$
|
(52,057,841
|
)
|
|
$
|
2,095,459
|
|
|
Stock, warrants and pre-funded warrants issuance for financing
|
|
11
|
|
|
-
|
|
|
|
-
|
|
|
|
175,330,001
|
|
|
|
15,984,325
|
|
|
|
-
|
|
|
|
1,465,500
|
|
|
|
10,514,458
|
|
|
|
-
|
|
|
|
27,964,283
|
|
|
Stock issuance costs
|
|
11
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(1,755,376
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
(1,084,024
|
)
|
|
|
-
|
|
|
|
(2,839,400
|
)
|
|
Placement agent warrants
|
|
11
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(154,767
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
154,767
|
|
|
|
-
|
|
|
|
-
|
|
|
Stock-based compensation
|
|
13
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
290,866
|
|
|
|
-
|
|
|
|
290,866
|
|
|
Stock issued due to exercise of warrants and pre-funded warrants
|
|
11
|
|
|
-
|
|
|
|
-
|
|
|
|
77,671,547
|
|
|
|
14,687,920
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(4,861,560
|
)
|
|
|
-
|
|
|
|
9,826,360
|
|
|
Net loss
|
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(7,758,607
|
)
|
|
|
(7,758,607
|
)
|
|
Balance at June 30, 2020
|
|
|
|
|
12
|
|
|
$
|
11,961,397
|
|
|
|
361,039,946
|
|
|
$
|
67,328,922
|
|
|
$
|
-
|
|
|
$
|
1,465,500
|
|
|
$
|
8,639,590
|
|
|
$
|
(59,816,448
|
)
|
|
$
|
29,578,961
|
|
The accompanying notes are an integral part of these condensed unaudited
interim consolidated financial statements.
Zomedica Pharmaceuticals Corp.
Condensed unaudited interim consolidated statements of cash flows
For the three and six months ended June 30, 2020 and 2019
(Stated in United States dollars)
|
|
|
|
|
|
|
|
Three months ended June 30,
|
|
|
|
Six months ended June 30,
|
|
|
|
|
|
Note
|
|
|
|
2020
|
|
|
|
2019
|
|
|
|
2020
|
|
|
|
2019
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows used in operating activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss for the period
|
|
|
|
|
|
$
|
(5,307,990
|
)
|
|
$
|
(2,404,427
|
)
|
|
$
|
(7,758,607
|
)
|
|
$
|
(14,081,337
|
)
|
|
Adjustments for
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation
|
|
|
6
|
|
|
|
77,859
|
|
|
|
68,925
|
|
|
|
154,275
|
|
|
|
130,979
|
|
|
Amortization - intangible assets
|
|
|
7
|
|
|
|
44,990
|
|
|
|
270
|
|
|
|
90,025
|
|
|
|
537
|
|
|
Amortization - right-of-use-asset
|
|
|
8
|
|
|
|
-
|
|
|
|
127,345
|
|
|
|
42,448
|
|
|
|
254,690
|
|
|
Loss on disposal of property and equipment
|
|
|
6
|
|
|
|
-
|
|
|
|
1,308
|
|
|
|
69,834
|
|
|
|
1,308
|
|
|
Loss on right-of-use asset
|
|
|
8
|
|
|
|
-
|
|
|
|
-
|
|
|
|
59,097
|
|
|
|
-
|
|
|
Non-cash portion of rent expense
|
|
|
8
|
|
|
|
6,019
|
|
|
|
-
|
|
|
|
10,032
|
|
|
|
-
|
|
|
Stock issued for services
|
|
|
11
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
792,104
|
|
|
Stock-based compensation
|
|
|
13
|
|
|
|
135,844
|
|
|
|
-
|
|
|
|
290,866
|
|
|
|
2,341,104
|
|
|
Change in non-cash operating working capital
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Tax credits and other receivables
|
|
|
|
|
|
|
(40,032
|
)
|
|
|
(17,578
|
)
|
|
|
(114,878
|
)
|
|
|
(41,891
|
)
|
|
Prepaid expenses
|
|
|
|
|
|
|
56,284
|
|
|
|
92,418
|
|
|
|
67,908
|
|
|
|
263,010
|
|
|
Deposits
|
|
|
|
|
|
|
(318,643
|
)
|
|
|
(327,138
|
)
|
|
|
78,762
|
|
|
|
(98,075
|
)
|
|
Accounts payable and accrued liabilities
|
|
|
|
|
|
|
(374,859
|
)
|
|
|
(5,977,134
|
)
|
|
|
(883,414
|
)
|
|
|
(579,716
|
)
|
|
|
|
|
|
|
|
|
(5,720,528
|
)
|
|
|
(8,436,011
|
)
|
|
|
(7,893,652
|
)
|
|
|
(11,017,287
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from financing activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds from financing of preferred shares
|
|
|
10
|
|
|
|
-
|
|
|
|
12,000,000
|
|
|
|
-
|
|
|
|
12,000,000
|
|
|
Proceeds from issuance of common shares, warrants and pre-funded warrants
|
|
|
11,14
|
|
|
|
23,998,783
|
|
|
|
-
|
|
|
|
26,498,783
|
|
|
|
3,000,000
|
|
|
Cash received from warrant exercises
|
|
|
13
|
|
|
|
9,826,359
|
|
|
|
-
|
|
|
|
9,826,359
|
|
|
|
600,000
|
|
|
Cash received from shares to be issued
|
|
|
12
|
|
|
|
1,465,500
|
|
|
|
-
|
|
|
|
1,465,500
|
|
|
|
-
|
|
|
Cash paid on stock issuance costs
|
|
|
11,14
|
|
|
|
(2,491,177
|
)
|
|
|
(33,095
|
)
|
|
|
(2,839,400
|
)
|
|
|
(626,267
|
)
|
|
Cash received for government loan
|
|
|
9
|
|
|
|
527,360
|
|
|
|
-
|
|
|
|
527,360
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
33,326,825
|
|
|
|
11,966,905
|
|
|
|
35,478,602
|
|
|
|
14,973,733
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows (used in) from investing activities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash from sale of property and equipment
|
|
|
6
|
|
|
|
-
|
|
|
|
-
|
|
|
|
5,400
|
|
|
|
-
|
|
|
Investment in property and equipment
|
|
|
6
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Cash from lease repurchase
|
|
|
8
|
|
|
|
-
|
|
|
|
(5,477
|
)
|
|
|
1,002,113
|
|
|
|
(74,563
|
)
|
|
|
|
|
|
|
|
|
-
|
|
|
|
(5,477
|
)
|
|
|
1,007,513
|
|
|
|
(74,563
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Increase in cash and cash equivalents
|
|
|
|
|
|
|
27,606,297
|
|
|
|
3,525,417
|
|
|
|
28,592,463
|
|
|
|
3,881,883
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents, beginning of period
|
|
|
|
|
|
|
1,496,752
|
|
|
|
2,296,731
|
|
|
|
510,586
|
|
|
|
1,940,265
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents, end of period
|
|
|
|
|
|
$
|
29,103,049
|
|
|
$
|
5,822,148
|
|
|
$
|
29,103,049
|
|
|
$
|
5,822,148
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental cash flow information:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest paid
|
|
|
|
|
|
$
|
-
|
|
|
$
|
12,164
|
|
|
$
|
651
|
|
|
$
|
18,338
|
|
The accompanying notes are an integral part of these condensed unaudited
interim consolidated financial statements.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
1. Nature of operations and going concern
Zomedica Pharmaceuticals Corp. ("Zomedica" or the “Company”)
was incorporated on January 7, 2013 under the Business Corporations Act (Alberta) as Wise Oakwood Ventures Inc. (“WOW”)
and was classified as a capital pool company, as defined in Policy 2.4 of the TSX Venture Exchange. ZoMedica Pharmaceuticals Inc.
was incorporated on May 14, 2015 under the Canada Business Corporations Act.
On April 21, 2016, the Company closed its qualifying transaction
(“Transaction”), consisting of the acquisition of ZoMedica Pharmaceuticals Inc. (“ZoMedica”) pursuant to
a three-cornered amalgamation, whereby ZoMedica was amalgamated with 9674128 Canada Inc. (which was wholly-owned by WOW) and common
shares and options of the Company were issued to former holders of ZoMedica securities as consideration. The amalgamated company
changed its name to Zomedica Pharmaceuticals Ltd. and WOW subsequently changed its name to Zomedica Pharmaceuticals Corp. Prior
to completion of the Transaction, WOW consolidated its common shares on the basis of the one post-consolidation common share for
every 2.5 pre-consolidation common shares. The Transaction constituted WOW’s qualifying transaction under TSX Venture Exchange
Policy 2.4 – Capital Pool Companies. The shares of Zomedica Pharmaceuticals Corp. began trading on the TSX Venture
Exchange under the new symbol “ZOM” on Monday, May 2, 2016. On June 21, 2016, the Company filed Articles of Amalgamation
and vertically amalgamated with its wholly-owned subsidiary, Zomedica Pharmaceuticals Ltd. On February 10, 2020 the Company voluntarily
delisted from the TSX-V.
Zomedica has one corporate subsidiary, Zomedica Pharmaceuticals,
Inc., a Delaware company whose results and operations are included in these consolidated financial statements. We are a development
stage veterinary health company focused on creating point-of-care diagnostic platforms for use by veterinarians treating companion
animals by focusing on the unmet needs of clinical veterinarians. Zomedica's head office is located at 100 Phoenix Drive, Suite
180, Ann Arbor, MI 48108 and its registered office is located at 3400, 350-7th Ave SW, Calgary, AB, T2P 3N9.
The accounting policies set out below have been applied consistently
in the condensed unaudited interim consolidated financial statements. The condensed unaudited interim consolidated financial statements
do not include all of the information required for annual consolidated financial statements and should be read in conjunction with
the Company’s audited consolidated financial statements for the year ended December 31, 2019. In the opinion of management,
all adjustments (consisting only of normal recurring adjustments) considered necessary for fair presentation have been included.
Operating results for the three and six months ended June 30, 2020 are not necessarily indicative of the results that may be expected
for the year ending December 31, 2020.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
|
2.
|
Basis of preparation (continued)
|
These condensed unaudited interim consolidated financial statements
were prepared using the same basis of presentation, accounting policies and methods of computation as those of the audited consolidated
financial statements for the year ended December 31, 2019.
Basis of consolidation
These condensed unaudited interim consolidated financial statements
include the accounts of the Company and its wholly owned operating subsidiary, Zomedica Pharmaceuticals, Inc.
All inter-company accounts and transactions have been eliminated
on consolidation.
|
3.
|
Significant accounting policies
|
Use of estimates
The preparation of the condensed unaudited interim consolidated
financial statements in conformity with accounting principles generally accepted in the United States of America (“U.S. GAAP”)
requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure
of contingent assets and liabilities at the date of the condensed unaudited interim consolidated financial statements and the reported
amounts of revenue and expenses during the period. Actual results could differ from those estimates.
Areas where significant judgment is involved
in making estimates are, the determination of fair value of stock-based compensation, the useful lives of property and equipment,
allocation of proceeds from financings to shares and warrants, fair value of placement agent warrants and forecasting future cash
flows for assessing the going concern assumption.
Basis of measurement
The condensed unaudited interim consolidated financial statements
have been prepared on the historical cost basis except as otherwise noted.
Functional and reporting currencies
The Company’s and subsidiary’s functional currency,
as determined by management, is US dollars, which is also the Company’s reporting currency.
The accounting policies set out below have been applied consistently
to all periods and companies presented in the condensed unaudited interim consolidated financial statements.
Research and development
Research and development costs related to continued research and
development programs are expensed as incurred in accordance with ASC topic 730.
Share issue costs
Share issue costs are recorded as a reduction of the proceeds from
the issuance of capital stock.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
|
3.
|
Significant accounting policies (continued)
|
Translation of foreign currencies
In respect of other transactions denominated
in currencies other than the Company and its wholly owned operating subsidiaries’ functional currencies, the monetary assets
and liabilities are remeasured at the period end rates. Revenue and expenses are measured at rates of exchange prevailing on the
transaction dates. All of the exchange gains or losses resulting from these other transactions are recognized in the condensed
unaudited interim consolidated statements of operations and comprehensive loss.
Stock-based compensation
The Company measures the cost of equity-settled transactions by
reference to the fair value of the equity instruments at the date at which they are granted if the fair value of the goods or services
received by the Company cannot be reliably estimated.
The Company calculates stock-based compensation using the fair value
method, under which the fair value of the options at the grant date is calculated using the Black-Scholes Option Pricing Model,
and subsequently expensed over the vesting period of the option using the graded vesting method. The provisions of the Company's
stock-based compensation plans do not require the Company to settle any options by transferring cash or other assets, and therefore
the Company classifies the awards as equity. Stock-based compensation expense recognized during the period is based on the value
of stock-based payment awards that are ultimately expected to vest.
The Company estimates forfeitures at the time of grant and revised,
if necessary, in subsequent periods if actual forfeitures differ from those estimates.
Loss per share
Basic loss per share (“EPS”)
is computed by dividing the loss attributable to common shareholders by the weighted average number of common shares outstanding.
Diluted EPS reflects the potential dilution that could occur from common shares issuable through the exercise or conversion of
stock options, restricted stock awards, warrants and convertible securities. In certain circumstances, the conversion of options
is excluded from diluted EPS if the effect of such inclusion would be anti-dilutive.
The dilutive effect of stock options is determined using the treasury
stock method. Stock options to purchase common shares of the Company during the period were not included in the computation of
diluted EPS because the Company has incurred a loss for the three and six months ended June 30, 2020 as the effect would be anti-dilutive.
Comprehensive loss
The Company follows ASC topic 220. This statement establishes standards
for reporting and display of comprehensive (loss) income and its components. Comprehensive loss is net loss plus certain items
that are recorded directly to shareholders' equity. The Company has no other comprehensive loss items.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
4.
|
Critical accounting judgments and key sources of estimation uncertainty
|
The preparation of financial statements in accordance with U.S.
GAAP requires management to make judgments, estimates and assumptions that affect the application of policies and reported amounts
of assets and liabilities, and revenue and expenses. The estimates and associated assumptions are based on historical experience
and various other factors that are believed to be reasonable under the circumstances, the results of which form the basis of making
the judgments about carrying values of assets and liabilities that are not readily apparent from other sources. Actual results
may differ from these estimates.
The estimates and underlying assumptions are reviewed on an ongoing
basis. Revisions to accounting estimates are recognized in the period in which the estimate is revised if the revision affects
only that period or in the period of the revision and further periods if the review affects both current and future periods.
Critical areas of estimation and judgements in applying accounting
policies include the following:
Going concern
These condensed unaudited interim consolidated financial statements
have been prepared in accordance with U.S. GAAP on a going concern basis, which assumes the realization of assets and discharge
of liabilities in the normal course of business within the foreseeable future. Management uses judgment in determining assumptions
for cash flow projections, such as anticipated financing, anticipated sales and future commitments to assess the Company’s
ability to continue as a going concern.
Stock-based payments
The Company estimates the fair value of convertible securities such
as options using the Black-Scholes option-pricing model which requires significant estimation around assumptions and inputs such
as expected term to maturity, expected volatility and expected dividends.
Useful lives of property and equipment
The Company reviews the estimated useful lives of property and equipment
with definite useful lives at the end of each year and assesses whether the useful lives of certain items should be shortened or
extended, due to various factors including technology, competition and revised service offerings. During the three and six months
ended June 30, 2020 and 2019, the Company was not required to adjust the useful lives of any assets based on the factors described
above. Long-lived assets are reviewed for impairment when events or circumstances indicate that the carrying value of an asset
may not be recoverable.
The impact of the novel strain of coronavirus (“COVID-19”)
Since December 31, 2019, the outbreak of the novel strain of coronavirus,
specifically identified as “COVID-19”, has resulted in the World Health Organization declaring this virus a global
pandemic in March 2020. Governments around the world have enacted emergency measures to combat the spread of the virus. These measures,
which include the implementation of travel bans, self-imposed quarantine periods and social distancing and closure of businesses
have caused material disruption to businesses resulting in an economic slowdown. Governments and central banks have responded with
significant monetary and fiscal interventions designed to stabilize the financial markets. A critical estimate for the Company
is to assess the impact of the pandemic on the recoverability of long-lived assets as well as the availability of future financing
in assessing the going concern assumption.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
5.
|
Prepaid expenses and deposits
|
|
|
|
|
June 30,
|
|
|
|
December 31,
|
|
|
|
|
|
2020
|
|
|
|
2019
|
|
|
Deposits (i)
|
|
$
|
655,203
|
|
|
$
|
1,033,231
|
|
|
Prepaid marketing (ii)
|
|
|
33,505
|
|
|
|
19,829
|
|
|
Prepaid insurance (ii)
|
|
|
19,860
|
|
|
|
110,636
|
|
|
Other (iii)
|
|
|
74,079
|
|
|
|
64,889
|
|
|
Total
|
|
$
|
782,647
|
|
|
$
|
1,228,585
|
|
|
|
(i)
|
Deposits include payments made to vendors in advance and are primarily associated with research activity, leasing deposits
and costs for additional office space. As of June 30, 2020, and December 31, 2019, the Company classified all amounts as a current
asset in the consolidated balance sheet, respectively;
|
|
|
(ii)
|
As of June 30, 2020, and December 31, 2019, all amounts were classified as a current asset in the consolidated balance sheet;
|
|
|
(iii)
|
Other is comprised of deferred financing costs, subscription payments, utilities, travel costs and software licensing. As of
June 30, 2020, and December 31, 2019, the Company classified all amounts as a current asset in the consolidated balance sheet.
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
|
6.
|
Property and equipment
|
|
|
|
|
Computer equipment
|
|
|
|
Furniture and equipment
|
|
|
|
Laboratory equipment
|
|
|
|
Leasehold improvements
|
|
|
|
Total
|
|
|
Cost
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2018
|
|
$
|
170,002
|
|
|
$
|
181,879
|
|
|
$
|
352,637
|
|
|
$
|
282,975
|
|
|
$
|
987,493
|
|
|
Additions
|
|
|
218,076
|
|
|
|
3,415
|
|
|
|
3,350
|
|
|
|
65,672
|
|
|
|
290,513
|
|
|
Disposals
|
|
|
(2,210
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(2,210
|
)
|
|
Balance at December 31, 2019
|
|
|
385,868
|
|
|
|
185,294
|
|
|
|
355,987
|
|
|
|
348,647
|
|
|
|
1,275,796
|
|
|
Additions
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
299,268
|
|
|
|
299,268
|
|
|
Disposals
|
|
|
(9,933
|
)
|
|
|
(64,018
|
)
|
|
|
(13,712
|
)
|
|
|
(76,455
|
)
|
|
|
(164,117
|
)
|
|
Balance at June 30, 2020
|
|
|
375,935
|
|
|
|
121,276
|
|
|
|
342,275
|
|
|
|
571,460
|
|
|
|
1,410,947
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated depreciation
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2018
|
|
|
104,918
|
|
|
|
29,585
|
|
|
|
99,696
|
|
|
|
36,206
|
|
|
|
270,405
|
|
|
Depreciation
|
|
|
88,417
|
|
|
|
26,617
|
|
|
|
68,519
|
|
|
|
93,597
|
|
|
|
277,150
|
|
|
Disposals
|
|
|
(901
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(901
|
)
|
|
Balance at December 31, 2019
|
|
|
192,434
|
|
|
|
56,202
|
|
|
|
168,215
|
|
|
|
129,803
|
|
|
|
546,654
|
|
|
Depreciation
|
|
|
44,102
|
|
|
|
8,857
|
|
|
|
34,705
|
|
|
|
66,611
|
|
|
|
154,275
|
|
|
Disposals
|
|
|
(2,849
|
)
|
|
|
(28,505
|
)
|
|
|
(30,843
|
)
|
|
|
(26,686
|
)
|
|
|
(88,883
|
)
|
|
Balance at June 30, 2020
|
|
|
233,687
|
|
|
|
36,554
|
|
|
|
172,077
|
|
|
|
169,728
|
|
|
|
612,046
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net book value as at:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2019
|
|
$
|
193,434
|
|
|
$
|
129,092
|
|
|
$
|
187,772
|
|
|
$
|
218,844
|
|
|
$
|
729,142
|
|
|
June 30, 2020
|
|
$
|
142,248
|
|
|
$
|
84,722
|
|
|
$
|
170,198
|
|
|
$
|
401,732
|
|
|
$
|
798,901
|
|
In February 2020, the Company disposed
of assets with a net book value of $75,234. The Company received proceeds of $5,400 and recorded a loss of $69,834 in the consolidated
statement of loss and comprehensive loss for the three months ended March 31, 2020 and six months ended June 30, 2020.
In February 2020, the Company reclassified $299,268 of prepaid expenses
to property and equipment for leasehold improvements that became ready for use in February 2020 but were paid for in 2019.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
|
|
|
Computer software
|
|
|
|
Trademarks
|
|
|
|
Website
|
|
|
|
Total intangible assets
|
|
|
Cost
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2018
|
|
$
|
5,143
|
|
|
$
|
16,236
|
|
|
$
|
-
|
|
|
$
|
21,379
|
|
|
Additions
|
|
|
-
|
|
|
|
-
|
|
|
|
531,419
|
|
|
|
531,419
|
|
|
Balance at December 31, 2019
|
|
|
5,143
|
|
|
|
16,236
|
|
|
|
531,419
|
|
|
|
552,798
|
|
|
Additions
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Balance at June 30, 2020
|
|
|
5,143
|
|
|
|
16,236
|
|
|
|
531,419
|
|
|
|
552,798
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated amortization
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2018
|
|
|
5,143
|
|
|
|
3,178
|
|
|
|
|
|
|
|
8,321
|
|
|
Amortization
|
|
|
-
|
|
|
|
1,082
|
|
|
|
|
|
|
|
1,082
|
|
|
Balance at December 31, 2019
|
|
|
5,143
|
|
|
|
4,260
|
|
|
|
|
|
|
|
9,403
|
|
|
Amortization
|
|
|
-
|
|
|
|
544
|
|
|
|
89,481
|
|
|
|
90,025
|
|
|
Balance at June 30, 2020
|
|
|
5,143
|
|
|
|
4,804
|
|
|
|
89,481
|
|
|
|
99,428
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net book value as at:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2019
|
|
$
|
-
|
|
|
$
|
11,976
|
|
|
$
|
531,419
|
|
|
$
|
543,395
|
|
|
June 30, 2020
|
|
$
|
-
|
|
|
$
|
11,432
|
|
|
$
|
441,938
|
|
|
$
|
453,370
|
|
Total estimated future amortization of intangible assets for
each fiscal year is as follows:
|
|
2020
|
|
|
$
|
90,796
|
|
|
|
2021
|
|
|
|
180,144
|
|
|
|
2022
|
|
|
|
180,144
|
|
|
|
2023
|
|
|
|
1,089
|
|
|
|
2024
|
|
|
|
1,089
|
|
|
|
2025
|
|
|
|
108
|
|
|
|
Total
|
|
|
$
|
453,370
|
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
The Company adopted ASC 842 with an initial
application date of January 1, 2019. The Company was party to two lease agreements under which it rented office and laboratory
space. The rent for both leases was prepaid upon inception and therefore at January 1, 2019, the Company reclassified its prepaid
lease balances of $1,613,038 to a right-of-use asset. The Company recorded nil and $42,448 of amortization on the right-of-use
asset for the three and six months ended June 30, 2020 (June 30, 2019 - $254,690).
On February 1, 2020 the Company cancelled its existing lease with
Wickfield Phoenix LLC and entered into a new lease. The new lease period is for 60 months, commencing on February 1, 2020 and ending
on January 31, 2025 with a monthly rent payment of $32,452 escalating to $36,525 over the lease period. Upon cancellation of the
existing lease, the Company received a refund of prepaid rent in the amount of $1,002,113. The carrying value of the right of use
asset was $1,061,210 upon cancellation. In February 2020, the Company recorded a loss on right-of-use asset of $59,097 in the consolidated
statements of operations and comprehensive loss.
On February 1, 2020, the Company recorded
a right-of-use asset and a corresponding lease liability in the amount of $1,553,611 using the Company’s incremental borrowing
rate of 12%. During the three and six months ended June 30, 2020, the Company recognized $103,375 and $172,291 in rent expense
with $21,763 and $38,992 recorded in research and development expenses and $81,612 and $137,630 recorded in general and administrative
expense in the consolidated statements of operations and comprehensive loss. During the three and six months ended June 30, 2019,
the Company recognized $5,940 and $11,880 in rent expense with nil recorded in research and development expenses and $5,940 and
$11,880 recorded in general and administrative expense in the consolidated statements of operations and comprehensive loss. During
the three and six months ended June 30, 2020, the Company also recorded $4,331 in rent expense related to month to month leases
with the entirety in general and administrative expense in the consolidated statements of operations and comprehensive loss.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
Right-of-use asset
|
|
Premise lease
|
|
Cost
|
|
|
|
|
|
Aggregate lease commitments
|
|
$
|
2,067,505
|
|
|
Less: impact of present value
|
|
|
(513,894
|
)
|
|
Balance at June 30, 2020
|
|
|
1,553,611
|
|
|
|
|
|
|
|
|
Reduction in right-of-use asset
|
|
|
|
|
|
Straight line amortization
|
|
|
172,292
|
|
|
Interest
|
|
|
(59,805
|
)
|
|
Balance at June 30, 2020
|
|
|
112,487
|
|
|
|
|
|
|
|
|
Net book value as at:
|
|
|
|
|
|
June 30, 2020
|
|
$
|
1,441,124
|
|
|
|
|
|
|
|
|
Lease liabilities
|
|
|
Premise lease
|
|
|
|
|
|
|
|
|
Additions
|
|
$
|
1,553,612
|
|
|
Payments
|
|
|
(162,260
|
)
|
|
Interest
|
|
|
59,805
|
|
|
Total lease liabilities at June 30, 2020
|
|
|
1,451,157
|
|
|
|
|
|
|
|
|
Current portion of lease liabilities
|
|
|
232,496
|
|
|
Long term portion of lease liabilities
|
|
|
1,218,661
|
|
|
Total lease liabilities at June 30, 2020
|
|
$
|
1,451,157
|
|
|
Total remaining undiscounted lease liabilities related to the above lease are as follows:
|
|
|
|
|
|
|
|
2020
|
|
$
|
194,712
|
|
|
2021
|
|
|
400,133
|
|
|
2022
|
|
|
412,137
|
|
|
2023
|
|
|
424,501
|
|
|
2024
|
|
|
437,236
|
|
|
2025
|
|
|
36,326
|
|
|
Total
|
|
$
|
1,905,045
|
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
On October 17, 2017, the Company entered into a loan arrangement
with a shareholder of the Company, pursuant to which such shareholder has agreed to provide a loan facility to the Company, whereby
the Company may borrow up to $5,000,000, with the proceeds to be used for working capital and general corporate purposes. The term
of the loan facility is five (5) years, with principal and interest payments being due only at the time of maturity. Under the
loan agreement, the Company may borrow in one or more advances, provided however that a minimum amount of $250,000 must be borrowed
at any one time and not more than two advances may occur per month. Interest shall accrue at a rate of fourteen percent (14%) per
annum, payable upon maturity. As of June 30, 2020, no amounts have been borrowed.
In April of 2020, the Company received $527,360 from the SBA’s
Paycheck Protection Program. The receipt is currently reported in accounts payable and accrued liabilities. If the loan is required
to be repaid it will be granted a two-year term at 1% interest.
The Company is authorized to issue 20 shares of Series 1 Preferred
Shares, all without par value, and each having a stated value of $1,000,000. The Series 1 Preferred Shares do not have voting rights
except to the extent required by applicable law and are not convertible into the Company’s common shares. Holders of the
Series 1 Preferred Shares will not be entitled to dividends but, in lieu thereof, will receive Net Sales Payments (“Net Sales
Payments” is defined as annual payments equal to 9 percent of sales) until such time as the holders have received total Net
Sales Payments equal to 9 times the aggregate stated value of the outstanding Series 1 Preferred Shares. The Company will have
the right to redeem the outstanding Series 1 Preferred Shares at any time at a redemption price equal to 9 times the aggregate
stated value of the Series 1 Preferred Shares outstanding less the aggregate amount of the Net Sales Payments paid (the “Redemption
Amount”).
Upon any dissolution, liquidation or winding up, whether voluntary
or involuntary, holders of Series 1 Preferred Shares will be entitled to a liquidation preference equal to the stated value of
the Series 1 Preferred Shares less the Net Sales Payments paid on the Series 1 Preferred Shares.
In the event of a fundamental transaction (defined to include an
amalgamation, merger or other business combination transaction involving our company in which our shareholders do not have the
right to cast more than 50% of the votes that may be cast for the election of directors, or a sale, lease or other disposition
of the properties and/or assets of our company as an entirety or substantially as an entirety to a third party), the holders of
the Series 1 Preferred Shares will be entitled to receive consideration for their Series 1 Preferred Shares equal to a multiple
of the stated value of the Series 1 Preferred Shares ranging from 5.0 to 9.0 depending on the timing of the fundamental transaction,
subject to a cap equal to the redemption amount. The Company has assessed the likelihood of any Net Sales Payments to the Series
1 Preferred shareholders to be remote.
Issued and outstanding preferred stock:
|
|
|
|
Number of
|
|
|
|
|
|
|
|
|
|
preferred
|
|
|
|
Preferred
|
|
|
|
|
|
stock
|
|
|
|
stock amount
|
|
|
Balance at December 31, 2018
|
|
|
-
|
|
|
$
|
-
|
|
|
Stock issued from financing (i)
|
|
|
12
|
|
|
|
11,961,397
|
|
|
Balance at December 31, 2019
|
|
|
12
|
|
|
$
|
11,961,397
|
|
|
Balance at June 30, 2020
|
|
|
12
|
|
|
$
|
11,961,397
|
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
The Company is authorized to issue
an unlimited number of common shares, all without par value.
Issued and outstanding common stock:
|
|
|
Number of
|
|
|
|
|
|
|
|
common
|
|
|
Common
|
|
|
|
|
stock
|
|
|
stock amount
|
|
|
Balance at December 31, 2018
|
|
|
97,598,898
|
|
|
$
|
30,410,648
|
|
|
Stock issuance for services (i and ii)
|
|
|
707,236
|
|
|
|
792,104
|
|
|
Stock issued from financing (iii and iv)
|
|
|
9,337,529
|
|
|
|
6,690,922
|
|
|
Stock issued due to exercise of options
|
|
|
394,735
|
|
|
|
754,148
|
|
|
Balance at June 30, 2019
|
|
|
108,038,398
|
|
|
$
|
38,647,822
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2019
|
|
|
108,038,398
|
|
|
$
|
38,566,820
|
|
|
Stock issued from financing (v,vi,vii)
|
|
|
175,330,001
|
|
|
|
15,984,325
|
|
|
Stock issuance costs
|
|
|
-
|
|
|
|
(1,755,376
|
)
|
|
Placement agent costs
|
|
|
-
|
|
|
|
(154,767
|
)
|
|
Stock issued from the financing and exercise of pre-funded warrants (viii)
|
|
|
12,162,492
|
|
|
|
1,080,289
|
|
|
Stock issued from the exercise of warrants (ix)
|
|
|
65,509,055
|
|
|
|
13,607,631
|
|
|
Balance at June 30, 2020
|
|
|
361,039,946
|
|
|
$
|
67,328,922
|
|
|
(i)
|
On January 14, 2019, the Company settled $75,000 of amounts due to a vendor by issuing
49,342 common shares valued at $55,263 at the date of issuance. The Company recorded a $19,737 gain on the settlement of liabilities.
|
|
(ii)
|
On January 14, 2019, the Company issued 657,894 common shares in satisfaction of $1,000,000
of all remaining milestones under a License and Supply Agreement with a third party. The Company recognized $736,841 as research
and development expense, based on the value of the common stock on the date of issuance.
|
|
(iii)
|
On January 14, 2019, the Company completed a non-brokered private placement, and issued
2,815,789 common shares. Gross proceeds of $4,280,000 were received prior to December 31, 2018.The Company recorded $465 of share
issuance costs as an offset to common stock.
|
|
(iv)
|
On March 28, 2019, the Company completed an underwritten public offering of its common
stock pursuant to which the Company sold an aggregate 6,521,740 common shares for gross proceeds of $3,000,000. The Company recorded
$592,707 of share issuance costs as an offset to common stock.
|
|
(v)
|
On February 14, 2020, the Company completed a registered direct offering (“RDO”)
of its common shares and a simultaneous private placement of its warrants (“Series A Warrants”) in a fixed combination
of one common share and a Series A Warrant to purchase one common share, resulting in the sale of 20,833,334 common shares and
Series A Warrants to purchase 20,833,334 common shares at a combined offering price of $0.12 per share and related Series A Warrant.
Each Series A Warrant has an exercise price of $0.20 per share, is exercisable six months after issuance and has a term of 5.5
years. The Company also issued warrants to the placement agents to purchase 1,041,667 common shares at an exercise price of $0.15
per share (“Series A Placement Agent Warrants”), which were exercisable immediately upon issuance and have a term
of 5 years. In aggregate, the Company issued 20,833,334 common shares, 20,833,334 Series A Warrants in addition to 1,041,667 Series
A Placement Agent Warrants.
|
The Company raised $2,500,000 in gross proceeds as part of the RDO. The Company
recorded $1,705,655 as the value of common shares under common stock and $794,345 as the value of Series A Warrants under additional
paid-in-capital in the consolidated statements of shareholders’ equity.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
|
11.
|
Common stock (continued)
|
The direct cash costs related to the
issuance of the common shares and warrants issued in February were $348,220. These direct costs were recorded as an offset against
the statement of shareholders’ equity with $238,217 being recorded under capital stock and $110,003 being recorded under
additional paid-in-capital. The Company also recorded the value of the Series A Placement Agent Warrants in the amount of $52,496
as an offset against the statement of shareholders’ equity with $35,816 being recorded under capital stock and $16,680 being
recorded under additional paid-in-capital.
|
(vi)
|
On April 9, 2020 the Company completed a confidentially marketed public offering (“CMPO”)
of its common shares and warrants (“Series B Warrants”) of 33,333,334 common shares and warrants to purchase up to
16,666,667 common shares. The securities were sold in a fixed combination of one common share and 0.5 of a Series B Warrant at
a combined offering price of $0.12 per share and accompanying warrant. Each whole warrant is exercisable immediately for one common
share after issuance, at an exercise price of $0.15 per share and has a term of 5 years. The Company also issued warrants to the
placement agents to purchase 1,666,667 common shares at an exercise price of $0.15 per share (“Series B Placement Agent Warrants”),
which were exercisable immediately upon issuance and have a term of 5 years. In aggregate, the Company issued 33,333,334 common
shares,16,666,667 Series B Warrants in addition to 1,666,667 Series B Placement Agent Warrants.
|
The Company raised $4,000,000 in gross
proceeds in the CMPO. The Company recorded $2,942,248 as the value of common shares under common stock and $1,057,752 as the value
of Series B Warrants under additional paid-in-capital in the consolidated statements of shareholders’ equity.
The direct
cash costs related to the issuance of the common shares and warrants issued in April were $582,977. These direct costs were recorded
as an offset against the statement of shareholders’ equity with $428,283 being recorded under capital stock and $154,694
being recorded under additional paid-in-capital. The Company also recorded the value of the Series B Placement Agent Warrants in
the amount of $161,714 as an offset against the statement of shareholders’ equity with $118,951 being recorded under capital
stock and $42,763 being recorded under additional paid-in-capital.
|
(vii)
|
On May 29, 2020 the Company completed a public offering of its common shares or common share equivalents
(“Series C pre-funded warrants”), and warrants (“Series C Warrants”) in a fixed combination of one common
share or Series C pre-funded warrant, and a Series C Warrant to purchase one common share, resulting in the sale of 121,163,333
common shares, 12,170,000 pre-funded warrants, and Series C Warrants to purchase 133,333,333 common shares at a combined offering
price of $0.15 per share for the common shares and related Series C Warrant, or a combined offering price of $0.1499 per pre-funded
warrant and related Series C warrant. Each Series C pre-funded warrant has an exercise price of $0.0001 per share, is exercisable
immediately after issuance, is exercisable only on a cashless exercise basis, and will not expire prior to exercise. Each Series
C Warrant has an exercise price of $0.15 per share, is exercisable immediately after issuance and has a term of 2 years.
|
The Company
raised $19,998,783 in gross proceeds as part of the public offering.
The Company recorded $11,336,422 as the value of common shares under common stock, $1,080,289 as the value of the pre-funded warrants
and $7,582,072 as the value of Series C Warrants under additional paid-in-capital in the consolidated statements of shareholders’
equity.
The direct
cash costs related to the issuance of the common shares, Series C pre-funded warrants and Series C Warrants issued in May were
$1,908,202. These direct costs were recorded as an offset against the statement of shareholders’ equity with $1,088,876 being
recorded under capital stock and $819,327 being recorded under additional paid-in-capital.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
|
11.
|
Common stock (continued)
|
|
(viii)
|
All Series C pre-funded warrants were exercised in June 2020. Upon exercise
the value of the warrant exercise was based on the one-day VWAP of the Company stock the day before the exercise request date.
The cashless exercise option resulted in the issuance of 12,162,492 shares.
|
|
(ix)
|
As of June 30, 2020, 11,602,084 Series B Warrants have been exercised, resulting in additional
cash proceeds of $1,740,313 and 53,906,971 Series C Warrants have been exercised, resulting in additional cash proceeds of $8,086,046.
All warrants were exercised during the three months ended June 30, 2020.
|
The Company received cash on Series C Warrant
exercises in for 9,770,000 shares at an exercise price of $0.15 per share, for aggregate gross proceeds of $1,465,500. The Company
issued the shares in the subsequent period.
|
13.
|
Stock-based compensation
|
During the three months ended June 30, 2020 and 2019, nil options
were exercised. During the six months ended June 30, 2020 and 2019, nil and 394,735 options were exercised, respectively. During
the three months ended June 30, 2020 and 2019, the Company issued 2,000,000 and nil stock options, respectively. During the six
months ended June 30, 2020 and 2019, the Company issued 7,056,000 and 5,995,000 stock options, respectively, each option entitling
the holder to purchase one common share of the Company.
The continuity of stock options are as follows:
|
|
|
|
Number of
options
|
|
|
|
Weighted avg
exercise price
|
|
|
Balance at December 31, 2019
|
|
|
7,040,265
|
|
|
$
|
1.28
|
|
|
Stock options forfeited January 23, 2020
|
|
|
(50,000
|
)
|
|
|
1.52
|
|
|
Stock options forfeited February 25, 2020
|
|
|
(5,000
|
)
|
|
|
1.12
|
|
|
Stock options forfeited March 1, 2020
|
|
|
(50,000
|
)
|
|
|
1.52
|
|
|
Stock options granted March 14, 2020
|
|
|
5,056,000
|
|
|
|
0.19
|
|
|
Stock options forfeited April 21, 2020
|
|
|
(150,000
|
)
|
|
|
0.19
|
|
|
Stock options forfeited May 4, 2019
|
|
|
(15,000
|
)
|
|
|
0.19
|
|
|
Stock options forfeited May 5, 2020
|
|
|
(30,000
|
)
|
|
|
1.52
|
|
|
Stock options forfeited May 7, 2020
|
|
|
(15,000
|
)
|
|
|
1.52
|
|
|
Stock options forfeited June 11, 2020
|
|
|
(15,000
|
)
|
|
|
1.52
|
|
|
Stock options granted June 16, 2020
|
|
|
2,000,000
|
|
|
|
0.19
|
|
|
Balance at June 30, 2020
|
|
|
13,766,265
|
|
|
$
|
0.73
|
|
|
Vested at June 30, 2020
|
|
|
8,805,932
|
|
|
$
|
1.04
|
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
|
13.
|
Stock-based compensation (continued)
|
As at June 30, 2020, details of the issued and outstanding stock
options were as follows:
|
Grant date
|
|
|
Exercise price
|
|
|
|
Number of options issued and outstanding
|
|
|
|
Number of vested options outstanding
|
|
|
|
Number of unvested options outstanding
|
|
|
|
Weighted Avg Remaining Life outstanding (years)
|
|
|
January 10, 2019
|
|
$
|
1.52
|
|
|
|
5,375,265
|
|
|
|
5,375,265
|
|
|
|
-
|
|
|
|
0.53
|
|
|
August 19, 2019
|
|
|
0.26
|
|
|
|
500,000
|
|
|
|
500,000
|
|
|
|
-
|
|
|
|
1.14
|
|
|
August 19, 2019
|
|
|
0.35
|
|
|
|
100,000
|
|
|
|
100,000
|
|
|
|
-
|
|
|
|
1.14
|
|
|
August 19, 2019
|
|
|
0.45
|
|
|
|
100,000
|
|
|
|
100,000
|
|
|
|
-
|
|
|
|
1.14
|
|
|
August 19, 2019
|
|
|
0.55
|
|
|
|
100,000
|
|
|
|
100,000
|
|
|
|
-
|
|
|
|
1.14
|
|
|
August 19, 2019
|
|
|
0.65
|
|
|
|
100,000
|
|
|
|
100,000
|
|
|
|
-
|
|
|
|
1.14
|
|
|
August 19, 2019
|
|
|
0.75
|
|
|
|
100,000
|
|
|
|
100,000
|
|
|
|
-
|
|
|
|
1.14
|
|
|
September 16, 2019
|
|
|
0.43
|
|
|
|
500,000
|
|
|
|
500,000
|
|
|
|
-
|
|
|
|
1.21
|
|
|
March 14, 2020
|
|
|
0.19
|
|
|
|
4,891,000
|
|
|
|
1,264,000
|
|
|
|
3,627,000
|
|
|
|
4.71
|
|
|
June 16, 2020
|
|
|
0.19
|
|
|
|
2,000,000
|
|
|
|
666,667
|
|
|
|
1,333,333
|
|
|
|
4.96
|
|
|
Balance at June 30, 2020
|
|
|
|
|
|
|
13,766,265
|
|
|
|
8,805,932
|
|
|
|
4,960,333
|
|
|
|
|
|
The fair value of options granted during the three and six months
ended June 30, 2020 was estimated using the Black-Scholes option pricing model to determine the fair value of options granted using
the following assumptions:
|
|
June 16, 2020
|
|
March 14, 2020
|
|
January 10, 2019
|
|
Volatility
|
100%
|
|
87%
|
|
68%
|
|
Risk-free interest rate
|
0.21%
|
|
0.49%
|
|
2.56%
|
|
Expected life (years)
|
5
|
|
5
|
|
2
|
|
Divedend yield
|
0%
|
|
0%
|
|
0%
|
|
Common share price
|
$0.19
|
|
$0.18
|
|
$1.23
|
|
Strike price
|
$0.19
|
|
$0.19
|
|
$1.52
|
|
Forfeiture rate
|
nil
|
|
nil
|
|
nil
|
The
Company recorded $135,844 and $290,866 of stock-based compensation for the three and six months ended June 30, 2020 (2019 –
nil and $2,341,104). For the three and six months ended June 30, 2020 the Company recorded nil cash receipts due to the exercise
of options. The Company recorded nil in cash receipts due to the exercise of options during the three months ended June 30, 2019.
The Company recorded the cash receipt of $600,000 and reclassified $154,148 of stock-based compensation to common stock due to
the exercise of options during the six months ended June 30, 2019.
The Company has estimated its stock option forfeitures to be nil
for the three and six months ended June 30, 2020 (three and six months ended June 30, 2019 - nil).
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
14. Warrants
In connection with the February 14, 2020 RDO, the Company issued
20,833,334 five and one half-year Series A Warrants to purchase one common share at an exercise price of $.20. The Company also
issued 1,041,667 placement agent warrants to purchase one common share at an exercise price of $0.15 per share.
In connection with the April 9, 2020 CMPO, the Company issued 16,666,667
five-year Series B Warrants to purchase one common share at an exercise price of $0.15. The Company also issued 1,666,667 placement
agent warrants to purchase one common at an exercise price of $0.15 per share.
In connection with the May 29, 2020 public
offering, the Company issued 133,333,333 two-year Series C Warrants to purchase on share of common stock at an exercise price of
$0.15. The Company also issued 12,170,000 Series C pre-funded warrants to purchase common shares at an exercise price of $0.0001
on a cashless exercise basis. As of June 30, 2020, the Series C pre-funded warrants have all been exercised.
As at June 30, 2020, details of the outstanding warrants were as follows:
|
Original Issue date
|
|
|
Exercise
Price
|
|
|
|
Warrants
Outstanding
|
|
|
|
Weighted Average Remaining Life
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
February 14, 2020
|
|
|
0.20
|
|
|
|
20,833,334
|
|
|
|
5.13
|
|
|
February 14, 2020
|
|
|
0.15
|
|
|
|
1,041,667
|
|
|
|
4.62
|
|
|
April 9, 2020
|
|
|
0.15
|
|
|
|
6,731,250
|
|
|
|
4.77
|
|
|
May 29, 2020
|
|
|
0.15
|
|
|
|
79,426,362
|
|
|
|
1.91
|
|
|
Balance at June 30, 2020
|
|
|
|
|
|
|
108,032,613
|
|
|
|
|
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
14. Warrants (continued)
The fair value of warrants issued the three
and six months ended June 30, 2020 was estimated using the Black-Scholes option pricing model to determine the fair value of warrants
granted using the following assumptions:
|
|
|
Series A Warrants
February 14, 2020
|
|
Placement Agent Warrants
February 14, 2020
|
|
Volatility
|
|
87%
|
|
87%
|
|
Risk-free interest rate
|
|
1.42%
|
|
1.42%
|
|
Expected life (years)
|
|
5.5
|
|
5
|
|
Dividend yield
|
|
0%
|
|
0%
|
|
Common share price
|
|
$0.12
|
|
$0.12
|
|
Strike price
|
|
$0.20
|
|
$0.15
|
|
Forfeiture rate
|
|
nil
|
|
nil
|
|
|
|
|
|
|
|
|
|
Series B Warrants
April 9, 2020
|
|
Placement Agent Warrants
April 9, 2020
|
|
Volatility
|
|
99%
|
|
99%
|
|
Risk-free interest rate
|
|
0.41%
|
|
0.41%
|
|
Expected life (years)
|
|
5
|
|
5
|
|
Dividend yield
|
|
0%
|
|
0%
|
|
Common share price
|
|
$0.14
|
|
$0.14
|
|
Strike price
|
|
$0.15
|
|
$0.15
|
|
Forfeiture rate
|
|
nil
|
|
nil
|
|
|
|
|
|
|
|
|
|
Series C Warrants
May 29, 2020
|
|
|
|
Volatility
|
|
118%
|
|
|
|
Risk-free interest rate
|
|
0.16%
|
|
|
|
Expected life (years)
|
|
2
|
|
|
|
Dividend yield
|
|
0%
|
|
|
|
Common share price
|
|
$0.16
|
|
|
|
Strike price
|
|
$0.15
|
|
|
|
Forfeiture rate
|
|
nil
|
|
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
15.
|
Commitments and contingencies
|
On November 26, 2018, the Company entered into a Development and
Supply Agreement and as part of this agreement, the Company has contingent future outflows as follows:
|
|
•
|
1st payment: At the later of the achievement of a future milestone event or September 12, 2019, can decide to receive payment as follows:
|
|
|
|
|
|
|
|
|
°
|
$3,000,000 in cash or
|
|
|
|
|
|
|
|
|
°
|
$1,500,000 in cash and $1.95 million in equity
|
|
|
|
|
|
|
|
•
|
2nd payment: At the later of the achievement of a future milestone or February 19, 2020 - $2,000,000 in cash.
|
|
|
|
|
|
|
|
•
|
3rd payment: At the later of the achievement of a future
milestone event or September 12, 2019, can decide to receive payment as follows:
|
|
|
|
|
|
|
|
|
°
|
$3,000,000 in cash or
|
|
|
|
|
|
|
|
|
°
|
$1,500,000 in cash and $1.95 million in equity
|
|
|
|
|
|
|
|
•
|
4th payment: At the later of the achievement of a future milestone or February 19, 2020 - $2,000,000 in cash.
|
As of June 30, 2020, the first and second milestones have been met
and paid. The third milestone was met and paid in April 2020. Per the terms of the agreement the cash option of $3,000,000 was
chosen. The fourth milestone payment has not been met.
On May 10, 2018, the Company entered into a Development, Commercialization
and Exclusive Distribution Agreement. As part of the agreement, the Company is required to make the following future milestone
payments:
|
|
•
|
1st payment: $3,500,000 in cash payment upon the achievement
of future development milestones
|
|
|
|
|
|
|
•
|
2nd payment: $3,500,000 in equity based on the number of the Company’s common stock determined by dividing the amount due by the
volume-weighted average price of the Company’s common stock on the NYSE American exchange over the 10 trading days prior to the
achievement of the milestone event.
|
|
|
|
|
|
As of June 30, 2020, neither of the future
development milestones related to the above agreement have been met.
On November 1, 2019, Heska Corporation
(“Heska”) filed a complaint for damages and injunctive relief (the “Complaint”) in the United States District
Court for the Middle District of North Carolina, Case 1:19-cv-01108-LCB-JLW, against Qorvo US, Inc. (“Qorvo US”), Qorvo
Biotechnologies, LLC (“Qorvo Biotech” and, together with Qorvo US, “Qorvo”) and the Company (collectively
with Qorvo, the “Defendants”). The Complaint alleges, among other things, that the Defendants improperly obtained Heska’s
trade secrets and confidential information and/or conspired to use improper means to misappropriate Heska’s trade secrets
related to an instrument and related consumable products for performing immunoassay analysis of biomarkers and other substances.
The Complaint seeks compensatory and exemplary damages, as well as preliminary and permanent injunctive relief to prevent the Defendants
from commercializing the Company’s TRUFORMATM diagnostic instrument. On January 21, 2020, the Defendants filed a motion seeking
dismissal of the Complaint. On February 11, 2020, Heska filed its response to the Defendants’ motion to dismiss to which
the Defendants responded on February 25, 2020. The Court has not yet ruled on Defendants’ motion to dismiss, and the litigation
remains stayed pending a ruling on that motion. The Company believes that the allegations in the Complaint have no merit and will
not have a material adverse effect on the Company’s business, results of operations or financial condition. Under the terms
of the Development and Supply Agreement, dated November 26, 2018, by and between Qorvo Biotech and the Company (the “Qorvo
Agreement”), Qorvo Biotech agreed to indemnify the Company and certain related parties against claims alleging infringement
or misappropriation of third-party intellectual property rights, subject to certain limitations and exceptions. Qorvo Biotech has
notified the Company that Qorvo Biotech has assumed the defense of the Complaint and will indemnify the Company for losses arising
from the Complaint in accordance with the terms of the Qorvo Agreement. Qorvo Biotech has further advised the Company that it intends
to mount a vigorous defense to the claims in the Complaint, and that it believes the allegations contained in the Complaint are
without merit.
|
16.
|
Financial instruments
|
The Company follows ASC topic 820, “Fair Value Measurements”
which defines fair value, establishes a framework for measuring fair value, and expands disclosures about fair value measurements.
The provisions of ASC topic 820 apply to other accounting pronouncements that require or permit fair value measurements. ASC topic
820 defines fair value as the exchange price that would be received for an asset or paid to transfer a liability (an exit price)
in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants at
the measurement date; and establishes a three level hierarchy for fair value measurements based upon the transparency of inputs
to the valuation of an asset or liability as of the measurement date. Inputs refers broadly to the assumptions that market participants
would use in pricing the asset or liability, including assumptions about risk. To increase consistency and comparability in fair
value measurements and related disclosures, the fair value hierarchy prioritizes the inputs to valuation techniques used to measure
fair value into three broad levels. The three levels of the hierarchy are defined as follows:
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
|
16.
|
Financial instruments (continued)
|
Level 1 inputs are quoted prices (unadjusted) in active markets
for identical assets or liabilities.
Level 2 inputs are inputs other than quoted prices included within
Level 1 that are observable for the asset or liability, either directly or indirectly for substantially the full term of the financial
instrument.
Level 3 inputs are unobservable inputs for asset or liabilities.
The categorization within the valuation hierarchy is based upon
the lowest level of input that is significant to the fair value measurement.
|
|
(i)
|
The Company calculates expected volatility based on historical volatility of the Company’s peer group that is publicly
traded for options.
|
An increase/decrease in the volatility would have resulted in an
increase/decrease in the fair value of the options.
The carrying values of cash, tax credit and other receivables, accounts
payable and accrued liabilities and shareholder loans payable approximates their fair values because of the short-term nature of
these instruments.
|
|
(b)
|
Interest rate and credit risk
|
Interest rate risk is the risk that the
value of a financial instrument might be adversely affected by a change in interest rates. The Company does not believe that the
results of operations or cash flows would be affected to any significant degree by a sudden change in market interest rates, relative
to interest rates on cash and cash equivalents, due to the short-term nature of these balances.
The Company is also exposed to credit risk at period end from the
carrying value of its cash. The Company manages this risk by maintaining bank accounts with a Canadian Chartered Bank. The Company’s
cash is not subject to any external restrictions.
|
|
(c)
|
Foreign exchange risk
|
The Company has balances in Canadian dollars that give rise to exposure
to foreign exchange (“FX”) risk relating to the impact of translating certain non-U.S. dollar balance sheet accounts
as these statements are presented in U.S. dollars. A strengthening U.S. dollar will lead to a FX loss while a weakening U.S. dollar
will lead to a FX gain. For each Canadian dollar balance of $1.0 million, a +/- 10% movement in the Canadian currency held
by the Company versus the U.S. dollar would affect the Company’s loss and other comprehensive loss by $0.1 million.
Liquidity risk is the risk that the Company will encounter difficulty
raising liquid funds to meet commitments as they fall due. In meeting its liquidity requirements, the Company closely monitors
its forecasted cash requirements with expected cash drawdown.
The following are the contractual maturities of the undiscounted
cash flows of financial liabilities as at June 30, 2020 and December 31, 2019:
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
|
16.
|
Financial instruments (continued)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
June 30, 2020
|
|
|
|
|
|
Less than
|
|
|
|
3 to 6
|
|
|
|
6 to 9
|
|
|
|
9 months
|
|
|
|
Greater than
|
|
|
|
|
|
|
|
|
|
3 months
|
|
|
|
months
|
|
|
|
months
|
|
|
|
1 year
|
|
|
|
1 year
|
|
|
|
Total
|
|
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
Third parties
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued liabilities
|
|
|
1,731,469
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,731,469
|
|
|
|
|
|
1,731,469
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,731,469
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, 2019
|
|
|
|
|
|
Less than
|
|
|
|
3 to 6
|
|
|
|
6 to 9
|
|
|
|
9 months
|
|
|
|
Greater than
|
|
|
|
|
|
|
|
|
|
3 months
|
|
|
|
months
|
|
|
|
months
|
|
|
|
1 year
|
|
|
|
1 year
|
|
|
|
Total
|
|
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
Third parties
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued liabilities
|
|
|
2,087,525
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,087,525
|
|
|
|
|
|
2,087,525
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,087,525
|
|
|
|
17.
|
Segmented information
|
The Company's operations comprise a single reportable segment engaged
in the research, development targeting health and wellness solutions for the companion animal. As the operations comprise a single
reportable segment, amounts disclosed in the financial statements for loss for the period, depreciation and total assets also represent
segmented amounts. In addition, all of the Company's long-lived assets are in the United States of America (“US”).
|
|
|
|
June 30,
|
|
|
|
December 31,
|
|
|
|
|
|
2020
|
|
|
|
2019
|
|
|
|
|
|
$
|
|
|
|
$
|
|
|
Total assets
|
|
|
|
|
|
|
|
|
|
Canada
|
|
|
27,291,934
|
|
|
|
249,929
|
|
|
US
|
|
|
5,469,653
|
|
|
|
3,933,055
|
|
|
|
|
|
|
|
|
|
|
|
|
Total US property and equipment
|
|
|
798,901
|
|
|
|
729,142
|
|
|
Tota US right-of-use asset
|
|
|
1,441,124
|
|
|
|
1,103,658
|
|
|
|
|
|
2,240,025
|
|
|
|
1,832,800
|
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
|
|
For the three months ended June 30,
|
|
For the three months ended June 30,
|
|
|
|
2020
|
|
2019
|
|
|
|
Research and
|
|
Professional
|
|
General and
|
|
Research and
|
|
Professional
|
|
General and
|
|
|
|
Development
|
|
Fees
|
|
Administrative
|
|
Development
|
|
Fees
|
|
Administrative
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Salaries, bonus and benefits
|
|
$
|
129,259
|
|
|
$
|
-
|
|
|
$
|
726,290
|
|
|
$
|
258,493
|
|
|
$
|
-
|
|
|
$
|
674,088
|
|
|
Contracted expenditures
|
|
|
515,944
|
|
|
|
-
|
|
|
|
-
|
|
|
|
557,719
|
|
|
|
-
|
|
|
|
-
|
|
|
Marketing and investor relations
|
|
|
-
|
|
|
|
-
|
|
|
|
67,827
|
|
|
|
-
|
|
|
|
-
|
|
|
|
78,905
|
|
|
Travel and accommodation
|
|
|
-
|
|
|
|
-
|
|
|
|
393
|
|
|
|
2,727
|
|
|
|
-
|
|
|
|
62,473
|
|
|
Insurance
|
|
|
202
|
|
|
|
-
|
|
|
|
46,318
|
|
|
|
1,430
|
|
|
|
-
|
|
|
|
19,319
|
|
|
License fees
|
|
|
3,000,000
|
|
|
|
-
|
|
|
|
-
|
|
|
|
50,000
|
|
|
|
-
|
|
|
|
-
|
|
|
Office
|
|
|
8,282
|
|
|
|
-
|
|
|
|
44,406
|
|
|
|
13,169
|
|
|
|
-
|
|
|
|
30,214
|
|
|
Consultants
|
|
|
75,150
|
|
|
|
282,791
|
|
|
|
-
|
|
|
|
89,094
|
|
|
|
231,647
|
|
|
|
-
|
|
|
Regulatory
|
|
|
151,073
|
|
|
|
-
|
|
|
|
21,888
|
|
|
|
42,647
|
|
|
|
-
|
|
|
|
26,066
|
|
|
Rent
|
|
|
21,763
|
|
|
|
-
|
|
|
|
81,612
|
|
|
|
-
|
|
|
|
-
|
|
|
|
5,940
|
|
|
Supplies
|
|
|
6,498
|
|
|
|
-
|
|
|
|
-
|
|
|
|
46,228
|
|
|
|
-
|
|
|
|
4,314
|
|
|
Total
|
|
$
|
3,908,171
|
|
|
$
|
282,791
|
|
|
$
|
988,734
|
|
|
$
|
1,061,507
|
|
|
$
|
231,647
|
|
|
$
|
901,319
|
|
|
|
|
For the six months ended June 30,
|
|
For the six months ended June 30,
|
|
|
|
2020
|
|
2019
|
|
|
|
Research and
|
|
Professional
|
|
General and
|
|
Research and
|
|
Professional
|
|
General and
|
|
|
|
Development
|
|
Fees
|
|
Administrative
|
|
Development
|
|
Fees
|
|
Administrative
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Salaries, bonus and benefits
|
|
$
|
315,643
|
|
|
$
|
-
|
|
|
$
|
1,503,436
|
|
|
$
|
503,800
|
|
|
$
|
-
|
|
|
$
|
3,646,260
|
|
|
Contracted expenditures
|
|
|
878,812
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,812,566
|
|
|
|
-
|
|
|
|
-
|
|
|
Marketing and investor relations
|
|
|
-
|
|
|
|
-
|
|
|
|
117,379
|
|
|
|
-
|
|
|
|
-
|
|
|
|
131,415
|
|
|
Travel and accommodation
|
|
|
407
|
|
|
|
-
|
|
|
|
12,915
|
|
|
|
12,776
|
|
|
|
-
|
|
|
|
119,737
|
|
|
Insurance
|
|
|
441
|
|
|
|
-
|
|
|
|
92,349
|
|
|
|
2,896
|
|
|
|
-
|
|
|
|
40,100
|
|
|
License fees
|
|
|
3,000,000
|
|
|
|
-
|
|
|
|
-
|
|
|
|
5,936,841
|
|
|
|
-
|
|
|
|
-
|
|
|
Office
|
|
|
22,077
|
|
|
|
-
|
|
|
|
298,196
|
|
|
|
19,597
|
|
|
|
-
|
|
|
|
97,457
|
|
|
Consultants
|
|
|
78,126
|
|
|
|
573,472
|
|
|
|
-
|
|
|
|
148,880
|
|
|
|
989,946
|
|
|
|
-
|
|
|
Regulatory
|
|
|
151,073
|
|
|
|
-
|
|
|
|
110,356
|
|
|
|
63,645
|
|
|
|
-
|
|
|
|
50,170
|
|
|
Rent
|
|
|
38,992
|
|
|
|
-
|
|
|
|
137,630
|
|
|
|
-
|
|
|
|
-
|
|
|
|
11,880
|
|
|
Supplies
|
|
|
17,999
|
|
|
|
-
|
|
|
|
-
|
|
|
|
91,881
|
|
|
|
-
|
|
|
|
16,658
|
|
|
Total
|
|
$
|
4,503,570
|
|
|
$
|
573,472
|
|
|
$
|
2,272,261
|
|
|
$
|
8,592,882
|
|
|
$
|
989,946
|
|
|
$
|
4,113,677
|
|
|
|
19.
|
Capital risk management
|
The capital of the Company includes equity, which is comprised of
issued common shares, additional paid-in capital, and accumulated deficit. The Company's objective when managing its capital is
to safeguard the ability to continue as a going concern in order to provide returns for its shareholders, and other stakeholders
and to maintain a strong capital base to support the Company's core activities.
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
|
|
|
|
For the three months ended June 30,
|
|
|
|
For the six months ended June 30,
|
|
|
|
|
|
2020
|
|
|
|
2019
|
|
|
|
2020
|
|
|
|
2019
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Numerator
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss for the period
|
|
$
|
5,307,990
|
|
|
$
|
2,404,427
|
|
|
$
|
7,758,607
|
|
|
$
|
14,081,337
|
|
|
Denominator
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average shares - basic
|
|
|
214,830,818
|
|
|
|
108,038,398
|
|
|
|
166,814,645
|
|
|
|
104,528,705
|
|
|
Warrants
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Stock options
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Denominator for diluted loss per share
|
|
|
214,830,818
|
|
|
|
108,038,398
|
|
|
|
166,814,645
|
|
|
|
104,528,705
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss per share - basic and diluted
|
|
$
|
(0.02
|
)
|
|
$
|
(0.02
|
)
|
|
$
|
(0.05
|
)
|
|
$
|
(0.13
|
)
|
For the above-mentioned
periods, the Company had securities outstanding which could potentially dilute basic earnings per share in the future but were
excluded from the computation of diluted loss per share in the periods presented, as their effect would have been anti-dilutive.
|
21.
|
Related party transactions and key management compensation
|
Key management personnel are comprised of the Company’s directors
and executive officers. In addition to their salaries, key management personnel also receive share-based compensation. Key management
personnel compensation is as follows:
|
|
|
For the three months ended June 30,
|
|
|
For the six months ended June 30,
|
|
|
|
|
2020
|
|
|
2019
|
|
|
2020
|
|
|
2019
|
|
|
Salaries and benefits, including bonuses
|
|
$
|
183,831
|
|
|
$
|
375,177
|
|
|
$
|
340,366
|
|
|
$
|
635,898
|
|
|
Stock-based compensation
|
|
|
279,944
|
|
|
|
-
|
|
|
|
379,557
|
|
|
|
1,644,325
|
|
|
Total
|
|
$
|
463,775
|
|
|
$
|
375,177
|
|
|
$
|
719,923
|
|
|
$
|
2,280,223
|
|
|
Zomedica Pharmaceuticals Corp.
|
|
Notes to the condensed unaudited interim consolidated financial statements
|
|
For the three and six months ended June 30, 2020 and 2019
|
|
(Stated in United States dollars)
|
On July 7, 2020 the Company completed a $30,000,000 public offering
of its common shares or common share equivalent (“Series D pre-funded warrants”), and warrants (“Series D Warrants”)
in a fixed combination of one common share or pre-funded warrant and a Series D Warrant to purchase one common share, resulting
in the sale of 162,500,000 common shares, 25,000,000 Series D pre-funded warrants, and Series D Warrants to purchase 187,500,000
common shares at a combined offering price of $0.16 per share for the common shares and related Series D warrants or a combined
offering price of $0.1599 per pre-funded warrant and related Series D warrant. Each Series D pre-funded warrant has an exercise
price of $0.0001 per share, is exercisable immediately after issuance, is exercisable only on a cashless exercise basis, and will
not expire prior to exercise. Each Series D Warrant has an exercise price of $0.16 per share, is exercisable immediately after
issuance and has a term of 2 years.
As part of this transaction, one of the
Company’s directors purchased 625,000 shares for $100,000 and received Series D Warrants to purchase an additional 625,000
common shares.
As of July 20, 2020, all the Series D pre-funded warrants have
all been exercised.
23. Comparative figures
Certain prior year amounts have been reclassified for consistency with the current year
presentation. These reclassifications had no effect on the reported results of operations. Adjustments have been made to the consolidated
schedule of expenses for the three and six months ended June 30, 2019, to classify health insurance benefits as part of salaries,
wages and bonuses and audit fees to professional fees. This change in classification does not affect previously reported cash flows
from operating activities in the consolidated statements of cash flows.
EXHIBIT “A” to proxy circular/prospectus
(Special Resolution)
RESOLVED AS A SPECIAL RESOLUTION THAT:
|
|
1.
|
the Corporation be authorized to make application to the Registrar of Corporations
of Alberta for approval to file a certificate of corporate domestication and a certificate of incorporation under the Delaware
General Corporation Law (‘‘DGCL’’) to continue the Corporation from the Business Corporations
Act (Alberta) (the ‘‘ABCA’’) to the DGCL as if the Corporation had been incorporated
under the DGCL, and to make application to the Registrar of Corporations of Alberta for the issuance of a certificate of discontinuance;
|
|
|
2.
|
the Corporation is authorized to file with the Secretary of State of the
State of Delaware the certificate of corporate domestication and a certificate of incorporation pursuant to, and in accordance
with the DGCL as if it has been incorporated thereunder (the ‘‘Domestication’’);
|
|
|
3.
|
effective on the date of the Domestication, the Corporation
shall file a certificate of corporate domestication and certificate of incorporation and bylaws in the forms as set out in Exhibits
B, C, and D to the proxy circular/prospectus dated as of October , 2020 (the ‘‘proxy circular/prospectus’’),
each of which is hereby approved in all respects;
|
|
|
4.
|
subject to the issuance of such certificate of discontinuance under the ABCA and a certificate of corporate domestication under the DGCL, and without affecting the validity of the Corporation and existence of the Corporation by or under its articles of incorporation under the ABCA, and of any act done thereunder, the Corporation’s certificate of incorporation as set out in Exhibit C to the proxy circular/prospectus shall be adopted by the Corporation in accordance with the requirements of the DGCL upon the issuance of a certificate of corporate domestication under the DGCL;
|
|
|
5.
|
notwithstanding that this special resolution shall have been duly passed
by the shareholders of the Corporation, the directors of the Corporation are authorized, in their sole discretion, to abandon the
application for a certificate of corporate domestication and a certificate of incorporation under the DGCL at any time prior to
the filing or issue thereof without further approval of the shareholders of the Corporation.
|
|
|
6.
|
any one (or more) director or officer of the Corporation is authorized and
directed, on behalf of the Corporation, to take all necessary steps and proceedings and to execute, deliver and file any and all
declarations, agreements, documents and other instruments and do all such other acts and things (whether under corporate seal of
the Corporation or otherwise) that may be necessary or desirable to give effect to this special resolution.
|
EXHIBIT “B” to proxy circular/prospectus
(Form of Certificate of Corporate Domestication)
CERTIFICATE OF CORPORATE DOMESTICATION
OF ZOMEDICA CORP.
The
undersigned, currently a corporation organized and existing under the laws of Canada, for the purposes of domesticating under the
Delaware General Corporation Law (the "DGCL"), does certify that:
|
|
1.
|
The corporation (hereinafter called the "corporation") was first formed, incorporated, or otherwise
came into being on March 14, 2005 under the laws of the Province of Alberta, Canada.
|
|
|
2.
|
The name of the corporation immediately prior to the filing of this certificate of corporate domestication was Zomedica Corp.
|
|
|
3.
|
The name of the corporation as set forth in its certificate of incorporation filed or to be filed in accordance with the DGCL
is Zomedica Corp.
|
|
|
4.
|
The jurisdiction that constituted the seat, siège social, or principal place of business or central administration
of the corporation, or other equivalent thereto under applicable law immediately prior to the filing of this certificate of corporate
domestication pursuant to the provisions of the DGCL is Alberta, Canada.
|
|
|
5.
|
The domestication has been approved in the manner provided for by the document, instrument, agreement or other writing, as
the case may be, governing the internal affairs of the corporation and the conduct of its business or by applicable non-Delaware
law, as appropriate.
|
|
|
6.
|
The effective time of this certificate of corporate domestication shall be [DATE], 2020.
|
IN
WITNESS WHEREOF, the corporation has caused this Certificate to be executed by its duly authorized officer on this [ ]
day of [MONTH], 2020.
|
|
|
ZOMEDICA CORP.,
|
|
|
|
an Alberta corporation
|
|
|
|
By:
|
|
|
|
|
|
Robert Cohen
Interim Chief Executive Officer
|
EXHIBIT “C” to proxy circular/prospectus
(Form of Certificate of Incorporation of Zomedica)
CERTIFICATE OF INCORPORATION OF
ZOMEDICA CORP.
I, the undersigned,
for the purpose of creating and organizing a corporation under the provisions of and subject to the requirements of the General
Corporation Law of the State of Delaware (the “DGCL”), certify as follows:
ARTICLE
I
The name of the corporation
is “Zomedica Corp.” (hereinafter called the “Corporation”).
ARTICLE
II
The address of the
Corporation’s registered office in the State of Delaware is 251 Little Falls Drive, Wilmington, New Castle County, Delaware
19808. The name of its registered agent at such address is Corporation Service Company.
ARTICLE
III
The purpose of the
Corporation is to engage in any lawful act or activity for which corporations may be organized under the DGCL.
The Corporation is
being incorporated in connection with the domestication of Zomedica Corp., corporation incorporated under the laws of the Province
of Alberta, Canada (the “Alberta Corporation”), as a Delaware corporation (the “Domestication”), and this
Certificate of Incorporation is being filed simultaneously with the Certificate of Domestication of the Alberta Corporation.
ARTICLE
IV
(A)
Classes of Stock. The total number of shares of all classes of capital stock that the Corporation is authorized to
issue is 2,010,000,000 shares which shall be divided into two classes of stock to be designated “Common Stock”
and “Preferred Stock”. The total number of shares of Common Stock that the Corporation is authorized to issue
is 2,000,000,000 shares, par value $0.0001 per share. The total number of shares of Preferred Stock that the Corporation is authorized
to issue is 10,000,000 shares, par value $0.0001 per share. Subject to the rights of the holders of any series of Preferred Stock,
the number of authorized shares of any of the Common Stock or Preferred Stock may be increased or decreased (but not below the
number of shares thereof then outstanding) by the affirmative vote of the holders of a majority in voting power of the stock of
the Corporation entitled to vote thereon irrespective of the provisions of Section 242(b)(2) of the DGCL, and no vote of the holders
of any of the Common Stock or Preferred Stock voting separately as a class shall be required therefor.
Upon the filing of the Certificate of Domestication
of the Alberta Corporation and this Certificate of Incorporation (the “Effective Time”), (i) each common share, no
par value, of the Alberta Corporation (“Common Shares”) issued and outstanding immediately prior to the Effective Time
will for all purposes be deemed to be one issued and outstanding, fully paid and nonassessable share of Common Stock, and (ii)
each Series 1 Preferred Share of the Alberta Corporation (“Old Series 1 Preferred Shares”) issued and outstanding immediately
prior to the Effective Time will for all purposes be deemed to be one issued and outstanding, fully paid and nonassessable Series
1 Preferred Share (as defined below), in each case without any action required on the part of the Corporation or the holders thereof.
Any share certificate that, immediately prior to the Effective Time, represented Common Shares or Old Series 1 Shares will, from
and after the Effective Time, automatically and without the necessity of presenting the same for exchange, also represent the same
number of shares of the Common Stock or Series 1 Preferred Shares, respectively.
(B)
Common Stock. The powers, preferences and relative participating, optional or other special rights, and the qualifications,
limitations and restrictions of the Common Stock are as follows:
1.
Ranking. The voting, dividend and liquidation rights of the holders of the Common Stock are subject to and qualified
by the rights of the holders of the Preferred Stock of any series as may be set forth herein or designated by the Board of Directors
of the Corporation (the “Board”) upon any issuance of the Preferred Stock of any series.
2.
Voting. Except as otherwise provided by law or by the resolution or resolutions providing for the issue of any series
of Preferred Stock, the holders of outstanding shares of Common Stock shall have the exclusive right to vote for the election and
removal of directors and for all other purposes. Notwithstanding any other provision of this Certificate of Incorporation (as the
same may be amended and/or restated from time to time, including the terms of any Preferred Stock Designation (as defined below),
this “Certificate of Incorporation”) to the contrary, the holders of Common Stock shall not be entitled to vote
on any amendment to this Certificate of Incorporation (including any Preferred Stock Designation) that relates solely to the terms
of one or more outstanding series of Preferred Stock if the holders of such affected series are entitled, either separately or
together as a class with the holders of one or more other such series, to vote thereon pursuant to this Certificate of Incorporation
(including any Preferred Stock Designation) or the DGCL.
3.
Dividends. Subject to the rights of the holders of Preferred Stock, holders of shares of Common Stock shall be entitled
to receive such dividends and distributions and other distributions in cash, stock or property of the Corporation when, as and
if declared thereon by the Board from time to time out of assets or funds of the Corporation legally available therefor.
4.
Liquidation. Subject to the rights of the holders of Preferred Stock, shares of Common Stock shall be entitled to
receive the assets and funds of the Corporation available for distribution in the event of any liquidation, dissolution or winding
up of the affairs of the Corporation, whether voluntary or involuntary. A liquidation, dissolution or winding up of the affairs
of the Corporation, as such terms are used in this Section B(4), shall not be deemed to be occasioned by or to include any consolidation
or merger of the Corporation with or into any other person or a sale, lease, exchange or conveyance of all or a part of its assets.
(C)
Preferred Stock.
Shares of Preferred
Stock may be issued from time to time in one or more series. The Board is hereby authorized to provide by resolution or resolutions
from time to time for the issuance, out of the unissued shares of Preferred Stock, of one or more series of Preferred Stock, without
stockholder approval, by filing a certificate pursuant to the applicable law of the State of Delaware (the “Preferred
Stock Designation”), setting forth such resolution and, with respect to each such series, establishing the number of
shares to be included in such series, and fixing the voting powers, full or limited, or no voting power of the shares of such series,
and the designation, preferences and relative, participating, optional or other special rights, if any, of the shares of each such
series and any qualifications, limitations or restrictions thereof. The powers, designation, preferences and relative, participating,
optional and other special rights of each series of Preferred Stock, and the qualifications, limitations and restrictions thereof,
if any, may differ from those of any and all other series at any time outstanding. The authority of the Board with respect to each
series of Preferred Stock shall include, but not be limited to, the determination of the following:
(a)
the designation of the series, which may be by distinguishing number, letter or title;
(b)
the number of shares of the series, which number the Board may thereafter (except where otherwise provided in the Preferred
Stock Designation) increase or decrease (but not below the number of shares thereof then outstanding);
(c)
the amounts or rates at which dividends will be payable on, and the preferences, if any, of shares of the series in respect
of dividends, and whether such dividends, if any, shall be cumulative or noncumulative;
(d)
the dates on which dividends, if any, shall be payable;
(e)
the redemption rights and price or prices, if any, for shares of the series;
(f)
the terms and amount of any sinking fund, if any, provided for the purchase or redemption of shares of the series;
(g)
the amounts payable on, and the preferences, if any, of shares of the series in the event of any voluntary or involuntary
liquidation, dissolution or winding up of the affairs of the Corporation;
(h)
whether the shares of the series shall be convertible into or exchangeable for, shares of any other class or series, or
any other security, of the Corporation or any other corporation, and, if so, the specification of such other class or series or
such other security, the conversion or exchange price or prices or rate or rates, any adjustments thereof, the date or dates at
which such shares shall be convertible or exchangeable and all other terms and conditions upon which such conversion or exchange
may be made;
(i)
restrictions on the issuance of shares of the same series or any other class or series;
(j)
the voting rights, if any, of the holders of shares of the series generally or upon specified events; and
(k)
any other powers, preferences and relative, participating, optional or other special rights of each series of Preferred
Stock, and any qualifications, limitations or restrictions thereof, all as may be determined from time to time by the Board and
stated in the resolution or resolutions providing for the issuance of such Preferred Stock.
Without limiting the generality of the
foregoing, the resolutions providing for issuance of any series of Preferred Stock may provide that such series shall be superior
or rank equally or be junior to any other series of Preferred Stock to the extent permitted by law.
(D)
Series 1 Preferred Shares
20 shares of Preferred
Stock of the Corporation are designated “Series 1 Preferred Shares.” Unless otherwise indicated, references to “Sections”
in this Part (D) of Article IV refer to sections of Part (D) of this Article IV. The rights, privileges, restrictions and conditions
attaching to the Series 1 Preferred Shares are as follows:
1.
Authorization
1.1
The Corporation is authorized to issue up to 20 Series 1 Preferred Shares (each a “Preferred Share”).
2.
Stated Value
2.1
The stated value of the Preferred Shares (the “Stated Value”) shall be U.S.$1,000,000 per Preferred Share.
3.
Voting Rights
3.1
Subject to the requirements of the DGCL, the holders of the Preferred Shares shall not, as such, be entitled to receive
notice of, to attend or to vote at any meeting of the stockholders of the Corporation.
4.
Dividends
4.1
The holders of the Preferred Shares shall not be entitled to receive any dividends that are declared by the Board.
4.2
Notwithstanding the foregoing, the directors may declare dividends on any class or series of share other than the Preferred
Shares.
5.
Net Sales Return
5.1
The holders of the Preferred Shares shall have the right to receive a return on their investment in the Preferred Shares
in lieu of the payment of ordinary dividends, with such return being in the aggregate amount of nine percent (9%) of
Net Sales (the “Net Sales Return”), subject to the terms and conditions set forth herein and the presence of
lawfully available funds therefor. Such return shall be payable out of lawfully available funds therefor until such time as the
aggregate Net Sales Returns paid to the holders of the Preferred Shares equals the Redemption Amount (as hereinafter defined),
at which time such right shall terminate and no further Net Sales Returns shall be paid or payable to the holders of Preferred
Shares.
5.2
Definitions. For purposes of this Section 5, the following terms have the following meanings:
a.
“Affiliate” means any person controlled directly or indirectly through one or more intermediaries,
by the Corporation. A Person shall be regarded as in control of the Corporation if the Corporation owns or directly or indirectly
controls more than fifty percent (50%) of the voting shares or other ownership interest of the other person, or if it possesses,
directly or indirectly, the power to direct or cause the direction of the management and policies of such person;
b.
“Licensing Proceeds” means all cash received by the Corporation and its Affiliates from third
party licensees or partners with respect to licensing or partnering arrangements with respect to a Product, including, without
limitation: (i) royalties based on sales of Products by third party licensees or their sublicensees; (ii) any licensing fees (including,
without limitation, upfront fees) for rights to develop or commercialize Products, or other payments in connection with the licensing
of rights with respect to Products; (iii) milestone payments (including without limitation, those based on development, regulatory
or commercialization milestones for Products); and (iv) research and development funding;
c.
“Net Sales” means for any period, the gross amount invoiced by the Corporation and its Affiliates
for the sale of Products, (including, without limitation, third party agents, distributors and wholesalers), less the total of
the following, to the extent applicable:
(i)
trade, cash and/or quantity discounts not already reflected in the amount invoiced;
(ii)
all excise, sales and other consumption taxes (including VAT) and custom duties, whether or not specifically identified
as such in the invoice to the third party;
(iii)
freight, distribution, insurance and other transportation charges, whether or not specifically identified as such in the
invoice to the third party;
(iv)
amounts repaid or credited by reason of rejections, defects or returns or because of chargebacks, retroactive price reductions,
refunds or billing errors;
(v)
any royalty amounts or license fees payable by the Corporation to a non-Affiliate third party for access to, or licensing
in of, such non-Affiliate third party’s intellectual property rights for use or exploitation of the Products; and
(vi)
rebates and similar payments made with respect to sales paid for or reimbursed by any governmental or regulatory authority
such as, by way of illustration, United States Federal or state Medicaid, Medicare or similar state program or equivalent foreign
governmental program.
For purposes of determining
Net Sales, “sale” will not include transfers or dispositions for charitable, promotional, pre-clinical, clinical, regulatory
or governmental purposes; and
d.
“Product” means any product produced or sold by the Corporation.
5.3
Timing of Net Sales Return Payments. With respect to Products that the Corporation commercializes directly, Net Sales
Returns, if any, will, subject to the presence of lawfully available funds therefor, be paid annually 15 business days after the
issuance of the Corporation’s audited financial statements for the prior year. With respect to Products that the Corporation
sublicenses or otherwise disposes of to a third-party, Net Sales Returns, if any, will, subject to the presence of lawfully available
funds therefor, be paid 7 business days after the end of the applicable quarter in which such Licensing Proceeds were received
by the Corporation (each of the foregoing dates upon which Net Sales Returns are to be calculated are hereinafter referred to as
a “Net Sales Return Record Date”, as applicable).
5.4
Allocation of Net Sales Returns. Once the Net Sales Returns have been calculated in accordance with this Section,
the Board shall declare a dividend in the amount of the Net Sales Net Sales Return which shall be allocated and payable in cash
out of funds legally available therefor on a pro rata basis to the holders of the Preferred Shares as of the applicable
Net Sales Return Record Date.
5.5
Termination upon Redemption. Upon the exercise by the Corporation of its right of Redemption pursuant to Section
6 herein, the right of the holders of the Preferred Shares to receive the Net Sales Returns shall terminate.
5.6
Unsecured Obligations. The Net Sales Returns are unsecured obligations of the Corporation.
5.7
Amendments, Modifications and Waivers. All modifications, amendments or waivers to the Net Sales Returns shall require
the written consent of the Corporation and the Holders of the majority of the issued and outstanding Preferred Shares.
6.
Redemption
6.1
The redemption amount of the Preferred Shares (the “Redemption Amount”) shall be equal to the following:
(Stated Value multiplied by nine (9))
less (cumulative aggregate Net Sales Returns that have been paid to holders (as of the Redemption Date)).
6.2
Subject to applicable law and the presence of lawfully available funds therefor, the Corporation shall have the right to
redeem, at any time following the payment to the holders of the Preferred Shares of the Redemption Amount, all, or from time to
time any part of, the then outstanding Preferred Shares via the delivery to such holders of a Redemption Notice in accordance with
Section 6.4 herein, together with all accrued and unpaid Net Sales Returns in respect thereof up to the most recent Net Sales Return
Record Date (the foregoing is herein referred to as the “Redemption”).
6.3
In case only a part of the then outstanding Preferred Shares is at any time to be redeemed, the Preferred Shares so to be
redeemed shall be redeemed pro rata, excluding fractions, from the holdings of all shareholders of Preferred Shares
or in such other manner as the Board deems reasonable.
6.4
On any Redemption of Preferred Shares under this Section 6, the Corporation shall, subject to the unanimous waiver of notice
by the registered holders thereof, give at least 10 days before the date fixed for redemption (the “Redemption Date”),
a notice in writing of the intention of the Corporation to redeem Preferred Shares (the “Redemption Notice”)
to each person who at the date of giving of such notice is a registered holder of Preferred Shares to be redeemed. The Redemption
Notice shall set out the calculation of the Redemption Amount, the Redemption Date and the number of Preferred Shares so held that
are to be redeemed.
6.5
The delivery of the Redemption Amount by such reasonable means as the Corporation deems desirable, on or before the Redemption
Date shall be deemed to give effect to the Redemption on the Redemption Date. Notwithstanding the foregoing, the Corporation shall
be entitled to require at any time, and from time to time, that the Redemption Amount be delivered to holders of Preferred Shares
only upon presentation and surrender at the registered office of the Corporation or at any other place or places designated by
the Redemption Notice of the certificate or certificates for such Preferred Shares to be redeemed.
6.6
From and after the Redemption Date, the Preferred Shares shall no longer be deemed to be outstanding for any purpose and
shall represent only the right to receive the Redemption Amount.
6.7
Preferred Shares that are redeemed or deemed to be redeemed in accordance with this Section 6 shall, subject to applicable
law, be and be deemed to be retired and returned to the authorized but unissued capital of the Corporation.
7.
Liquidation and Fundamental Transactions
7.1
In the event of the liquidation, dissolution or winding up of the Corporation or other distribution of the assets of the
Corporation among its shareholders for the purpose of winding up its affairs (other than in the case of a Fundamental Transaction),
the holders of Preferred Shares shall be entitled to receive the Stated Value per share (on a pro rata basis)
less Net Sales Returns paid or payable up to the most recent Net Sales Return Record Date prior to the commencement of any such
liquidation, dissolution, winding up or other distribution of the assets of the Corporation, to be paid all such money before any
money shall be paid or property or assets distributed to the holders of any Common Stock or other shares in the capital of the
Corporation ranking junior to the Preferred Shares with respect to return of capital.
7.2
A “Fundamental Transaction” means a transaction or series of transactions that result in any of the following:
a.
an amalgamation, arrangement, merger, consolidation, takeover, reverse takeover, reorganization or other business combination
or other similar transaction of the Corporation with or into any other person (provided that the other person is not an affiliate
of the Corporation) whereby all or substantially all of the issued and outstanding shares of Common Stock are sold, transferred
or exchanged for cash and/or securities pursuant to which the shareholders of the Corporation immediately prior to such Fundamental
Transaction do not immediately thereafter own shares of a successor continuing corporation that entitle them to cast more than
50% of the votes attaching to all shares in the capital of the successor or continuing corporation that may be cast to elect directors
of that corporation;
b.
a sale, lease, disposition or conveyance of the property and/or assets of the Corporation as an entirety or substantially
as an entirety to any other person (provided that the other person is not an affiliate of the Corporation) for consideration consisting
of cash and/or securities and the subsequent distribution of all of such consideration to all of the holders of Common Stock, on
a pro rata basis;
7.3
In the event of a Fundamental Transaction, the holders of the Preferred Shares shall be entitled to receive in exchange
for their Preferred Shares the following amount (such amount, the “Fundamental Transaction Amount”):
a.
Fundamental Transaction completed within the first year from issuance of the Old Series 1 Preferred Shares:
5 x Stated Value;
b.
Fundamental Transaction completed within the second year from issuance of the Old Series 1 Preferred Shares:
5.5 x Stated Value;
c.
Fundamental Transaction completed within the third or fourth year from issuance of the Old Series 1 Preferred Shares:
6 x Stated Value;
d.
Fundamental Transaction completed within the fifth year from issuance of the Old Series 1 Preferred Shares:
7 x Stated Value; and
e.
Fundamental Transaction completed more than six (6) years from issuance of the Old Series 1 Preferred Shares:
9 x Stated Capital.
provided however that in each case, the
maximum aggregate amount payable under this Section 7.3 shall be the lesser of: (a) the aggregate consideration payable under the
Fundamental Transaction plus any Net Sales Returns paid to date; and (b) the Redemption Amount.
7.4
The Fundamental Transaction Amount shall be paid in cash or, if the consideration payable under the Fundamental Transaction
consists of securities or other assets (other than cash), the value attributed to such consideration in the Fundamental Transaction.
The acquirer under the Fundamental Transaction (whether that be the Corporation, any successor entity or any other third party
offeror) shall pay to each holder of Preferred Shares, such holder’s pro rata portion of the Fundamental
Transaction Amount in cash, securities or other assets (as applicable under the Fundamental Transaction) no later than 30 days
after the consummation of the Fundamental Transaction upon surrender of the Preferred Shares for cancellation.
7.5
The right of the holders to receive the Fundamental Transaction Amount shall be prior in right of payment to the right of
the holders of shares of Common Stock or other shares in the capital of the Corporation ranking junior to the Preferred Shares
with respect to return of capital to receive any payment in connection with a Fundamental Transaction.
7.6
From and after the effective date of a Fundamental Transaction, the Preferred Shares shall no longer be deemed to be outstanding
for any purpose and shall represent only the right to receive the Fundamental Transaction Amount.
7.7
In the event of any voluntary or involuntary liquidation, dissolution or winding up of the Corporation, after the payment
of all preferential amounts required to be paid to the holders of Preferred Shares, the remaining assets of the Corporation available
for distribution to its shareholders shall be distributed among the holders of the Common Stock, pro rata based
on the number of shares held by each holder.
8.
Amendments
8.1
At any time that the Preferred Shares are outstanding, the rights, privileges, restrictions and conditions attached to the
Preferred Shares may be amended, modified, suspended, altered or repealed but only if consented to, or approved by, the holders
of the Preferred Shares in the manner hereinafter specified and in accordance with any requirements of applicable law.
9.
Approval by Holders of Preferred Shares
9.1
For the purpose of Section 8, any consent or approval given by the holders of Preferred Shares shall be deemed to have been
sufficiently given if it shall have been given in writing by all the holders of the outstanding Preferred Shares or by a resolution
passed at a meeting of holders of Preferred Shares duly called and held upon not less than 21, nor more than 60, days’ notice
in writing to the holders at which the holders of at least 50% of the outstanding Preferred Shares are present or are represented
by proxy and carried by the affirmative vote of not less than two-thirds of the votes cast at such meeting. On every ballot cast
at every meeting of the holders of the Preferred Shares, every holder of a Preferred Share shall be entitled to one (1) vote in
respect of each Preferred Share held. Subject to the foregoing, the formalities to be observed in respect of the giving or waiving
of notice of any such meeting or adjourned meeting and the conduct thereof shall be those from time to time prescribed in the Bylaws
(as defined below).
ARTICLE
V
This Article V is inserted
for the management of the business and for the conduct of the affairs of the Corporation.
(A)
General Powers. The business and affairs of the Corporation shall be managed by or under the direction of the Board,
except as otherwise provided by this Certificate of Incorporation or the DGCL.
(B)
Number of Directors; Election of Directors. Subject to the rights of holders of any series of Preferred Stock to
elect directors, the number of the directors of the Corporation shall be fixed from time to time by resolution of the Board.
(C)
(C) Classes of Directors. Subject to the rights of holders of any series of Preferred Stock to elect directors, the
Board shall be divided into three classes, designated Class I, Class II and Class III. Each class shall consist, as nearly as may
be possible, of one third of the total number of directors constituting the entire Board. The Board is authorized to assign members
of the Board already in office to Class I, Class II or Class III at the time such classification becomes effective.
(D)
Terms of Office. Subject to the rights of holders of any series of Preferred Stock to elect directors, each director
shall serve for a term ending on the date of the third annual meeting of stockholders following the annual meeting of stockholders
at which such director was elected; provided that each director initially assigned to Class I shall serve for a term expiring
at the Corporation’s first annual meeting of stockholders held following the time at which the initial classification of
the Board becomes effective; each director initially assigned to Class II shall serve for a term expiring at the Corporation’s
second annual meeting of stockholders held following the time at which the initial classification of the Board becomes effective;
and each director initially assigned to Class III shall serve for a term expiring at the Corporation’s third annual meeting
of stockholders held following the time at which the initial classification of the Board becomes effective; provided further, that
the term of each director shall continue until the election and qualification of his or her successor and be subject to his or
her earlier death, disqualification, resignation or removal.
(E)
Vacancies. Subject to the rights of holders of any series of Preferred Stock, any newly created directorship that
results from an increase in the number of directors or any vacancy on the Board that results from the death, disability, resignation,
disqualification or removal of any director or from any other cause shall be filled solely by the affirmative vote of a majority
of the total number of directors then in office, even if less than a quorum, or by a sole remaining director and shall not be filled
by the stockholders. Any director elected in accordance with the preceding sentence shall, in the case of a newly created directorship,
hold office for the full term of the class in which the newly created directorship was created or, in the case of a vacancy, hold
office for the remaining term of his or her predecessor and in each case until his or her successor shall be elected and qualified,
subject to his or her earlier death, disqualification, resignation or removal.
(F)
Removal. Subject to the rights of the holders of any series of Preferred Stock, any director or the entire Board
may be removed from office at any time, but only for cause.
(G)
Committees. Pursuant to the Bylaws of the Corporation (as the same may be amended and/or restated from time to time,
the “Bylaws”), the Board may establish one or more committees to which may be delegated any or all of the powers
and duties of the Board to the full extent permitted by law.
(H)
Stockholder Nominations and Introduction of Business. Advance notice of stockholder nominations for election of directors
and other business to be brought by stockholders before a meeting of stockholders shall be given in the manner provided by the
Bylaws.
(I)
Preferred Stock Directors. During any period when the holders of any series of Preferred Stock have the right to
elect additional directors as provided for or fixed pursuant to the provisions of Article IV hereof or any Preferred Stock Designation,
then upon commencement and for the duration of the period during which such right continues: (i) the then otherwise total number
of authorized directors of the Corporation shall automatically be increased by such specified number of directors, and the holders
of such Preferred Stock shall be entitled to elect the additional directors so provided for or fixed pursuant to said provisions,
and (ii) each such additional director shall serve until such director’s successor shall have been duly elected and qualified,
or until such director’s right to hold such office terminates pursuant to said provisions, whichever occurs earlier, subject
to his earlier death, disqualification, resignation or removal. Except as otherwise provided for or fixed pursuant to the provisions
of Article IV hereof or any Preferred Stock Designation, whenever the holders of any series of Preferred Stock having such right
to elect additional directors are divested of such right pursuant to the provisions of such stock, the terms of office of all such
additional directors elected by the holders of such stock, or elected to fill any vacancies resulting from the death, resignation,
disqualification or removal of such additional directors, shall forthwith terminate and the total authorized number of directors
of the Corporation shall be reduced accordingly.
ARTICLE
VI
Unless and except to
the extent that the Bylaws shall so require, the election of directors of the Corporation need not be by written ballot.
ARTICLE
VII
To the fullest extent
permitted by the DGCL as the same exists or as may hereafter be amended, a director of the Corporation shall not be personally
liable to the Corporation or its stockholders for monetary damages for breach of fiduciary duty as a director; provided,
however, that nothing contained in this Article VII shall eliminate or limit the liability of a director (i) for any breach
of the director’s duty of loyalty to the Corporation or its stockholders, (ii) for acts or omissions not in good faith or
which involve intentional misconduct or a knowing violation of law, (iii) pursuant to the provisions of Section 174 of the DGCL,
or (iv) for any transaction from which the director derived an improper personal benefit. No repeal or modification of this Article
VII shall apply to or have any adverse effect on any right or protection of, or any limitation of the liability of, a director
of the Corporation existing at the time of such repeal or modification with respect to acts or omissions occurring prior to such
repeal or modification.
ARTICLE
VIII
The Corporation may
indemnify, and advance expenses to, to the fullest extent permitted by law, any person who was or is a party to or is threatened
to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative
or investigative by reason of the fact that the person is or was a director, officer, employee or agent of the Corporation, or
is or was serving at the request of the Corporation as a director, officer, employee or agent of another corporation, partnership,
joint venture, trust or other enterprise.
ARTICLE
IX
Except as otherwise
provided in the terms of any series of Preferred Stock, any action required or permitted to be taken by the stockholders of the
Corporation must be effected at an annual or special meeting of the stockholders and may not be effected by written consent in
lieu of a meeting.
ARTICLE
X
Special meetings of
stockholders for any purpose or purposes may be called at any time by the Board, the Chairman of the Board or the Chief Executive
Officer of the Corporation, and may not be called by another other person or persons. Business transacted at any special meeting
of stockholders shall be limited to matters relating to the purpose or purposes stated in the notice of meeting.
ARTICLE
XI
The Corporation reserves
the right at any time from time to time to amend, alter, change or repeal any provision contained in this Certificate of Incorporation,
and any other provisions authorized by the DGCL may be added or inserted, in the manner now or hereafter prescribed by law; and
all rights, preferences and privileges of whatsoever nature conferred upon stockholders, directors or any other persons whomsoever
by and pursuant to this Certificate of Incorporation in its present form or as hereafter amended are granted subject to the right
reserved in this Article XI. Notwithstanding any other provision of this Certificate of Incorporation or any provision of law that
might otherwise permit a lesser vote or no vote, but in addition to any affirmative vote of the holders of any series of Preferred
Stock required by law, by this Certificate of Incorporation or by any Preferred Stock Designation, the affirmative vote of the
holders of at least sixty-six and two-thirds percent (66 2/3%) in voting power of the stock of the Corporation entitled to vote
thereon shall be required to amend, alter, change or repeal, or adopt any provision inconsistent with, any of Article V, Article
VII, Article VIII, Article IX, Article X, Article XII, Article XIII, and this sentence of this Certificate of Incorporation, or
in each case, the definition of any capitalized terms used therein or any successor provision (including, without limitation, any
such article or section as renumbered as a result of any amendment, alteration, change, repeal or adoption of any other provision
of this Certificate of Incorporation). Any amendment, repeal or modification of any of Article VII, Article VIII and this sentence
shall not adversely affect any right or protection of any person existing thereunder with respect to any act or omission occurring
prior to such repeal or modification.
ARTICLE
XII
In furtherance and
not in limitation of the powers conferred upon it by law, the Board is expressly authorized and empowered to adopt, amend and repeal
the Bylaws. Notwithstanding any other provision of this Certificate of Incorporation or any provision of law that might otherwise
permit a lesser vote, but in addition to any affirmative vote of the holders of any series of Preferred Stock required by law,
by this Certificate of Incorporation or by any Preferred Stock Designation, the Bylaws may also be amended, altered or repealed
by the stockholders of the Corporation and new Bylaws may be adopted by the stockholders of the Corporation by the affirmative
vote of the holders of at least sixty-six and two-thirds percent (66 2/3%) in voting power of the outstanding stock of the Corporation
entitled to vote thereon.
ARTICLE
XIII
Unless the Corporation
consents in writing to the selection of an alternative forum, (A) (i) any derivative action or proceeding brought on behalf of
the Corporation, (ii) any action asserting a claim of breach of a fiduciary duty owed by any current or former director, officer,
other employee or stockholder of the Corporation to the Corporation or the Corporation’s stockholders, (iii) any action asserting
a claim arising pursuant to any provision of the DGCL, this Certificate of Incorporation or the Bylaws (as either may be amended
or restated) or as to which the DGCL confers jurisdiction on the Court of Chancery of the State of Delaware or (iv) any action
asserting a claim governed by the internal affairs doctrine of the law of the State of Delaware shall, to the fullest extent permitted
by law, be exclusively brought in the Court of Chancery of the State of Delaware or, if such court does not have subject matter
jurisdiction thereof, the federal district court of the State of Delaware; and (B) the federal district courts of the United States
shall be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities Act of
1933, as amended. Notwithstanding the foregoing, this Article XIII shall not apply to claims seeking to enforce any liability or
duty created by the Securities Exchange Act of 1934, as amended. To the fullest extent permitted by law, any person or entity purchasing
or otherwise acquiring or holding any interest in shares of capital stock of the Corporation shall be deemed to have notice of
and consented to the provisions of this Article XIII.
ARTICLE
XIV
The incorporator is , whose mailing address
is .
[Remainder of Page
Intentionally Left Blank]
I, THE UNDERSIGNED,
being the incorporator, for the purpose of forming a corporation pursuant to the DGCL, do make this Certificate of Incorporation,
hereby acknowledging, declaring, and certifying that the foregoing Certificate of Incorporation is my act and deed and that the
facts herein stated are true, and have accordingly hereunto set my hand this [DATE][st/th] day of [MONTH], 2020.
|
|
By:
|
|
|
|
|
[name], sole incorporator
|
|
|
|
|
EXHIBIT “D” to proxy circular/prospectus
(Form of Bylaws of Zomedica)
BYLAWS
OF
ZOMEDICA CORP.
ARTICLE
I
STOCKHOLDERS
1.1
Place of Meetings. All meetings of stockholders shall be held at such place, if any, as may be designated from time
to time by the Board of Directors (the “Board”) of Zomedica Corp. (the “Corporation”), the
Chairman of the Board or the Chief Executive Officer or, if not so designated, at the principal office of the Corporation. The
Board may, in its sole discretion, determine that a meeting shall not be held at any place, but may instead be held solely by means
of remote communication in accordance with Section 211(a)(2) of the General Corporation Law of the State of Delaware (the “DGCL”).
1.2
Annual Meeting. The annual meeting of stockholders for the election of directors to succeed those whose terms expire
and for the transaction of such other business as may properly be brought before the meeting shall be held on a date and at a time
designated by the Board, the Chairman of the Board or the Chief Executive Officer. The Board may postpone, recess, reschedule or
cancel any previously scheduled annual meeting of stockholders.
1.3
Special Meetings. Special meetings of stockholders for any purpose or purposes may be called in the manner set forth
in the Certificate of Incorporation. The Board may postpone, recess, reschedule or cancel any previously scheduled special meeting
of stockholders. Business transacted at any special meeting of stockholders shall be limited to matters relating to the purpose
or purposes stated in the notice of meeting.
1.4
Notice of Meetings. Except as otherwise provided by law, notice of each meeting of stockholders, whether annual or
special, shall be given not less than ten (10) nor more than sixty (60) days before the date of the meeting to each stockholder
entitled to vote at such meeting as of the record date for determining the stockholders entitled to notice of the meeting. The
notices of all meetings shall state the place, if any, date and time of the meeting, the means of remote communications, if any,
by which stockholders and proxyholders may be deemed to be present in person and vote at such meeting, and the record date for
determining the stockholders entitled to vote at the meeting (if such date is different from the record date for stockholders entitled
to notice of the meeting). The notice of a special meeting shall state, in addition, the purpose or purposes for which the meeting
is called.
1.5
Voting List. The Corporation shall prepare, at least ten (10) days before every meeting of stockholders, a complete
list of the stockholders entitled to vote at the meeting (provided, however, if the record date for determining the
stockholders entitled to vote is less than ten (10) days before the date of the meeting, the list shall reflect the stockholders
entitled to vote as of the tenth day before the meeting date), arranged in alphabetical order, and showing the address of each
stockholder and the number of shares registered in the name of each stockholder. Such list shall be open to the examination of
any stockholder, for any purpose germane to the meeting, for a period of at least ten (10) days prior to the meeting: (a) on a
reasonably accessible electronic network, provided that the information required to gain access to such list is provided
with the notice of the meeting, or (b) during ordinary business hours, at the principal place of business of the Corporation. If
the meeting is to be held at a place, then the list shall also be produced and kept at the time and place of the meeting during
the whole time thereof, and may be inspected by any stockholder who is present. If the meeting is to be held solely by means of
remote communication, then the list shall also be open to the examination of any stockholder during the whole time of the meeting
on a reasonably accessible electronic network, and the information required to access such list shall be provided with the notice
of the meeting. Except as otherwise provided by law, the list shall presumptively determine the identity of the stockholders entitled
to vote at the meeting and the number of shares held by each of them.
1.6
Quorum. Except as otherwise provided by law, the Certificate of Incorporation or these Bylaws,
the holders of not less than one-third of the voting power of the shares of the capital stock of the Corporation issued and outstanding
and entitled to vote at the meeting, present in person, present by means of remote communication in a manner, if any, authorized
by the Board in its sole discretion, or represented by proxy, shall constitute a quorum for the transaction of business; provided,
however, that where a separate vote by a class or classes or series of capital stock is required by law or the Certificate
of Incorporation, the holders of not less than one-third of the voting power of the shares of such class or classes or series of
the capital stock of the Corporation issued and outstanding and entitled to vote on such matter, present in person, present by
means of remote communication in a manner, if any, authorized by the Board in its sole discretion, or represented by proxy, shall
constitute a quorum entitled to take action with respect to the vote on such matter. A quorum, once established at a meeting, shall
not be broken by the withdrawal of enough votes to leave less than a quorum.
1.7
Adjournments. Any meeting of stockholders, annual or special, may be adjourned from time to time to any other time
and to any other place at which a meeting of stockholders may be held under these Bylaws by the Board, the chairman of the meeting
or, if directed to be voted on by the chairman of the meeting, by the stockholders present or represented at the meeting and entitled
to vote thereon, although less than a quorum. If the adjournment is for more than thirty (30) days, a notice of the adjourned meeting
shall be given to each stockholder of record entitled to vote at the meeting. If after the adjournment a new record date for determination
of stockholders entitled to vote is fixed for the adjourned meeting, the Board shall fix as the record date for determining stockholders
entitled to notice of such adjourned meeting the same or an earlier date as that fixed for determination of stockholders entitled
to vote at the adjourned meeting, and shall give notice of the adjourned meeting to each stockholder of record as of the record
date so fixed for notice of such adjourned meeting. At the adjourned meeting, the Corporation may transact any business which might
have been transacted at the original meeting.
1.8
Proxies. Each stockholder of record entitled to vote at a meeting of stockholders may vote in person (including by
means of remote communications, if any, by which stockholders may be deemed to be present in person and vote at such meeting) or
may authorize another person or persons to vote for such stockholder by a proxy executed or transmitted in a manner permitted by
applicable law. No such proxy shall be voted upon after three years from the date of its execution, unless the proxy expressly
provides for a longer period. A proxy shall be irrevocable if it states that it is irrevocable and if, and only as long as, it
is coupled with an interest sufficient in law to support an irrevocable power. A stockholder may revoke any proxy which is not
irrevocable by attending the meeting and voting in person or by delivering to the Secretary of the Corporation a revocation of
the proxy or a new proxy bearing a later date.
1.9
Action at Meeting. When a quorum is present at any meeting, any matter other than the election of directors to be
voted upon by the stockholders at such meeting shall be decided by the vote of the holders of shares of stock having a majority
in voting power of the votes cast by the holders of all of the shares of stock present or represented at the meeting and voting
affirmatively or negatively on such matter (or if there are two or more classes or series of stock entitled to vote as separate
classes, then in the case of each such class or series, the holders of a majority in voting power of the shares of stock of that
class or series present or represented at the meeting and voting affirmatively or negatively on such matter), except when a different
vote is required by express provision of applicable law, regulation applicable to the Corporation or its securities, the rules
or regulations of any stock exchange applicable to the Corporation, the Certificate of Incorporation or these Bylaws, in which
case such express provisions shall govern. Voting at meetings of stockholders need not be by written ballot. At all meetings of
stockholders for the election of directors at which a quorum is present a plurality of the votes cast shall be sufficient to elect.
1.10
Notice of Stockholder Business and Nominations.
(A) Annual
Meetings of Stockholders. (1) Nominations of persons for election to the Board and the proposal of other business to be considered
by the stockholders may be made at an annual meeting of stockholders only (a) pursuant to the Corporation’s notice of meeting
(or any supplement thereto), (b) by or at the direction of the Board or any committee thereof or (c) by any stockholder of the
Corporation who was a stockholder of record of the Corporation at the time the notice provided for in this Section 1.10 is delivered
to the Secretary of the Corporation, who is entitled to vote at the meeting upon such election of directors or upon such other
business, as the case may be, and who complies with the notice procedures set forth in this Section 1.10.
(2) For
any nominations or other business to be properly brought before an annual meeting by a stockholder pursuant to clause (c) of paragraph
(A)(1) of this Section 1.10, the stockholder must have given timely notice thereof in writing to the Secretary of the Corporation
(and must timely provide any updates or supplements to such notice at such times and in such forms provided by this Section 1.10)
and any such proposed business (other than the nominations of persons for election to the Board) must constitute a proper matter
for stockholder action. To be timely, a stockholder’s notice shall be delivered to the Secretary at the principal
executive
offices of the Corporation not later than the close of business on the ninetieth (90th) day, nor earlier than the close
of business on the one hundred twentieth (120th) day, prior to the first anniversary of the preceding year’s annual
meeting (provided, however, that in the event that the date of the annual meeting is more than thirty (30) days before or more
than seventy (70) days after such anniversary date, notice by the stockholder must be so delivered not earlier than the close of
business on the one hundred twentieth (120th) day prior to such annual meeting and not later than the close of business
on the later of the ninetieth (90th) day prior to such annual meeting or the tenth (10th) day following the
day on which public announcement of the date of such meeting is first made by the Corporation). In no event shall the public announcement
of an adjournment, postponement or recess of an annual meeting commence a new time period (or extend any time period) for the giving
of a stockholder’s notice as described above. The number of nominees a stockholder may nominate for election at the annual
meeting (or in the case of a stockholder giving the notice on behalf of a beneficial owner, the number of nominees a stockholder
may nominate for election at the annual meeting on behalf of such beneficial owner) shall not exceed the number of directors to
be elected at such annual meeting. To be in proper form for purposes of this Section 1.10, such stockholder’s notice shall
set forth: (a) as to each person whom the stockholder proposes to nominate for election as a director (i) the name, age, business
and residence address, and principal occupation or employment of the nominee, (ii) and all other information relating to such nominee
that is required to be disclosed in solicitations of proxies for election of directors in an election contest, or is otherwise
required, in each case pursuant to and in accordance with Section 14(a) of the Securities Exchange Act of 1934, as amended (the
“Exchange Act”), and the rules and regulations promulgated thereunder, (iii) a reasonably detailed description of any
compensatory, payment or other financial agreement, arrangement or understanding that such nominee has with any other person or
entity other than the Corporation including the amount of any payment or payments received or receivable thereunder, in each case
in connection with candidacy or service as a director of the Corporation, (iv) such person’s written consent to being named
in the Corporation’s proxy statement and associated proxy card as a nominee of the stockholder and to serving as a director
if elected and (v) all information with respect to such nominee that would be required to be set forth in a stockholder’s
notice pursuant to this Section 1.10 if such nominee were the stockholder giving notice hereunder; (b) as to any other business
that the stockholder proposes to bring before the
meeting, (i) a brief description of the business desired to be brought before
the meeting, (ii) the text of the proposal or business (including the text of any resolutions proposed for consideration and in
the event that such business includes a proposal to amend the Bylaws of the Corporation, the language of the proposed amendment),
(iii) the reasons for conducting such business at the meeting, (iv) any direct or indirect material interest in such business of
such stockholder and the beneficial owner, if any, on whose behalf the proposal is made and any other person or persons with whom
such stockholder or beneficial owner, if any, has any agreement, arrangement or understanding in connection with such proposal
and (v) such other information relating to any proposed item of business as the Corporation may reasonably require to determine
whether such proposed item of business is a proper matter for stockholder action; and (c) as to the stockholder giving the notice
and the beneficial owner, if any, on whose behalf the nomination or proposal is made (i) the name and address of such stockholder,
as they appear on the Corporation’s books, and of such beneficial owner, (ii) the class or series and number of shares of
capital stock of the Corporation which are, directly or indirectly, owned beneficially (within the meaning of Rule 13d-3 under
the Exchange Act) or of record by such stockholder and such beneficial owner (provided, that such stockholder and the beneficial
owner, if any, on whose behalf the nomination or proposal is made shall in all events be deemed to beneficially own any shares
of any class or series and number of shares of capital stock of the Corporation as to which such stockholder or beneficial owner,
if any, has a right to acquire beneficial ownership at any time in the future), (iii) a description of any agreement, arrangement
or understanding with respect to the nomination or proposal between or among such stockholder and/or such beneficial owner, any
of their respective affiliates or associates, and any others acting in concert with any of the foregoing (including their names),
including, in the case of a nomination, the nominee, (iv) a description of any agreement, arrangement or understanding (including
any derivative or short positions, profit interests, options, warrants, convertible securities, stock appreciation or similar rights,
hedging transactions, and borrowed or loaned shares) that has been entered into as of the date of the stockholder’s notice
by, or on behalf of, such stockholder and such beneficial owners, whether or not such instrument or right shall be subject to settlement
in underlying shares of capital stock of the Corporation, the effect or intent of which is to mitigate loss to, manage risk or
benefit of share price changes for, or increase or decrease the voting power of, such stockholder or such beneficial owner, with
respect to securities of the Corporation, (v) a representation that the
stockholder is a holder of record of stock of the Corporation
entitled to vote at such meeting upon such business or nomination, as the case may be, and intends to appear in person or by proxy
at the meeting to propose such business or nomination, (vi) a representation whether the stockholder or the beneficial owner, if
any, intends or is part of a group which intends (a) to deliver a proxy statement and/or form of proxy to holders of at least the
percentage of the Corporation’s outstanding capital stock required to approve or adopt the proposal or elect the nominee
and/or (b) otherwise to solicit proxies or votes from stockholders in support of such proposal or nomination, and (vii) any other
information relating to such stockholder and beneficial owner, if any, required to be disclosed in a proxy statement or other filings
required to be made in connection with solicitations of proxies for, as applicable, the proposal and/or for the election of directors
in an election contest pursuant to and in accordance with Section 14(a) of the Exchange Act and the rules and regulations promulgated
thereunder. The foregoing notice requirements of this paragraph (A) of this Section 1.10 shall be deemed satisfied by a stockholder
with respect to business other than a nomination if the stockholder has notified the Corporation of his, her or its intention to
present a proposal at an annual meeting in compliance with applicable rules and regulations promulgated under the Exchange Act
and such stockholder’s proposal has been included in a proxy statement that has been prepared by the Corporation to solicit
proxies for such annual meeting. The Corporation may require any proposed nominee to furnish such other information as the Corporation
may reasonably require to determine the eligibility of such proposed nominee to serve as a director of the Corporation. If requested
by the Corporation, the information required by clause (c) of this paragraph (A)(2) shall be supplemented by such stockholder and
any such beneficial owner not later than ten (10) days after the record date for the meeting to disclose such information as of
the record date. In addition, a stockholder seeking to nominate a director candidate or bring other business before the annual
meeting shall promptly provide any other information reasonably requested by the Corporation.
(3) Notwithstanding
anything in the second sentence of paragraph (A)(2) of this Section 1.10 to the contrary, in the event that the number of directors
to be elected to the Board at the annual meeting is increased effective after the time period for which nominations would otherwise
be due under paragraph (A)(2) of this Section 1.10 and there is no public announcement by the Corporation naming the nominees for
the additional directorships at least one hundred (100) days prior to the first anniversary of the preceding year’s annual
meeting, a stockholder’s notice required by this Section 1.10 shall also be considered timely, but only with respect to nominees
for the additional directorships, if it shall be delivered to the Secretary at the principal executive offices of the Corporation
not later than the close of business on the tenth (10th) day following the day on which such public announcement is
first made by the Corporation.
(B) Special
Meetings of Stockholders. Only such business shall be conducted at a special meeting of stockholders as shall have been brought
before the meeting pursuant to the Corporation’s notice of meeting. Nominations of persons for election to the Board may
be made at a special meeting of stockholders at which directors are to be elected pursuant to the Corporation’s notice of
meeting (1) by or at the direction of the Board or any committee thereof or (2) provided that the Board has determined that directors
shall be elected at such meeting, by any stockholder of the Corporation who is a stockholder of record at the time the notice provided
for in this Section 1.10 is delivered to the Secretary of the Corporation, who is entitled to vote at the meeting and upon such
election and who complies with the notice procedures set forth in this Section 1.10. The number of nominees a stockholder may nominate
for election at the special meeting (or in the case of a stockholder giving the notice on behalf of a beneficial owner, the number
of nominees a stockholder may nominate for election at the special meeting on behalf of such beneficial owner) shall not exceed
the number of directors to be elected at such special meeting. In the event the Corporation calls a special meeting of stockholders
for the purpose of electing one or more directors to the Board, any such stockholder entitled to vote in such election of directors
may nominate a person or persons (as the case may be) for election to such position(s) as specified in the Corporation’s
notice of meeting, if the stockholder’s notice required by paragraph (A)(2) of this Section 1.10 shall be delivered to the
Secretary at the principal executive offices of the Corporation not earlier than the close of business on the one hundred twentieth
(120th) day prior to such special meeting and not later than the close of business on the later of the ninetieth (90th)
day prior to such special meeting or the tenth (10th) day following the day on which the public announcement is first
made of the date of the special meeting and of the nominees proposed by the Board to be elected at such meeting. In no event shall
the public announcement of an adjournment, postponement or recess of a special meeting commence a new time period (or extend any
time period) for the giving of a stockholder’s notice as described above.
(C) General.
(1) Except as otherwise expressly provided in any applicable rule or regulation promulgated under the Exchange Act, only such persons
who are nominated in accordance with the procedures set forth in this Section 1.10 shall be eligible to be elected at an annual
or special meeting of stockholders of the Corporation to serve as directors and only such business shall be conducted at a meeting
of stockholders as shall have been brought before the meeting in accordance with the procedures set forth in this Section 1.10.
Except as otherwise provided by law, the chairman of the meeting shall have the power and duty (a) to determine whether a nomination
or any business proposed to be brought before the meeting was made or proposed, as the case may be, in accordance with the procedures
set forth in this Section 1.10 (including whether the stockholder or beneficial owner, if any, on whose behalf the nomination or
proposal is made, solicited (or is part of a group which solicited) or did not so solicit, as the case may be, proxies or votes
in support of such stockholder’s nominee or proposal in compliance with such stockholder’s representation as required
by clause (A)(2)(c)(vi) of this Section 1.10) and (b) if any proposed nomination or business was not made or proposed in compliance
with this Section 1.10, to declare that such nomination shall be disregarded or that such proposed business shall not be transacted.
Notwithstanding the foregoing provisions of this Section 1.10, unless otherwise required by law, if the stockholder (or a qualified
representative of the stockholder) does not appear at the annual or special meeting of stockholders of the Corporation to present
a nomination or proposed business, such nomination shall be disregarded and such proposed business shall not be transacted, notwithstanding
that proxies in respect of such vote may have been received by the Corporation. For purposes of this Section 1.10, to be considered
a qualified representative of the stockholder, a person must be a duly authorized officer, manager or partner of such stockholder
or must be authorized by a writing executed by such stockholder or an electronic transmission delivered by such stockholder to
act for such stockholder as proxy at the meeting of stockholders and such person must produce such writing or electronic transmission,
or a reliable reproduction of the writing or electronic transmission, at the meeting of stockholders.
(2) For
purposes of this Section 1.10, “public announcement” shall include disclosure in a press release reported by the Dow
Jones News Service, Associated Press or other national news service or in a document publicly filed by the Corporation with the
Securities and Exchange Commission pursuant to Section 13, 14 or 15(d) of the Exchange Act and the rules and regulations promulgated
thereunder.
(3) Notwithstanding
the foregoing provisions of this Section 1.10, a stockholder shall also comply with all applicable requirements of the Exchange
Act and the rules and regulations promulgated thereunder with respect to the matters set forth in this Section 1.10; provided however,
that any references in these Bylaws to the Exchange Act or the rules and regulations promulgated thereunder are not intended to
and shall not limit any requirements applicable to nominations or proposals as to any other business to be considered pursuant
to this Section 1.10 (including paragraphs (A)(1)(c) and (B) hereof), and compliance with paragraphs (A)(1)(c) and (B) of this
Section 1.10 shall be the exclusive means for a stockholder to make nominations or submit other business (other than, as provided
in the penultimate sentence of (A)(2), business other than nominations brought properly under and in compliance with Rule 14a-8
of the Exchange Act, as may be amended from time to time). Nothing in this Section 1.10 shall be deemed to affect any rights (a)
of stockholders to request inclusion of proposals or nominations in the Corporation’s proxy statement pursuant to applicable
rules and regulations promulgated under the Exchange Act or (b) of the holders of any series of Preferred Stock to elect directors
pursuant to any applicable provisions of the Certificate of Incorporation.
1.11
Conduct of Meetings; Inspectors of Election.
(A)
Meetings of stockholders shall be presided over by the Chairman of the Board, if any, or in the Chairman’s absence
by the Vice Chairman of the Board, if any, or in the Vice Chairman’s absence by the Chief Executive Officer, or in the Chief
Executive Officer’s absence, by the President, or in the President’s absence by a Vice President, or in the absence
of all of the foregoing persons by a chairman designated by the Board. The Secretary shall act as secretary of the meeting, but
in the Secretary’s absence the chairman of the meeting may appoint any person to act as secretary of the meeting.
(B)
The Board may adopt by resolution such rules, regulations and procedures for the conduct of any meeting of stockholders
of the Corporation as it shall deem appropriate including, without limitation, such guidelines and procedures as it may deem appropriate
regarding the participation by means of remote communication of stockholders and proxyholders not physically present at a meeting.
Except to the extent inconsistent with such rules, regulations and procedures as adopted by the Board, the chairman of any meeting
of stockholders shall have the right and authority to convene and (for any or no reason) to recess and/or adjourn the meeting,
to prescribe such rules, regulations and procedures and to do all such acts as, in the judgment of such chairman, are appropriate
for the proper conduct of the meeting. Such rules, regulations or procedures, whether adopted by the Board or prescribed by the
chairman of the meeting, may include, without limitation, the following: (i) the establishment of an agenda or order of business
for the meeting; (ii) rules and procedures for maintaining order at the meeting and the safety of those present; (iii) limitations
on attendance at or participation in the meeting to stockholders of record of the Corporation, their duly authorized and constituted
proxies or such other persons as shall be determined; (iv) restrictions on entry to the meeting after the time fixed for the commencement
thereof; and (v) limitations on the time allotted to questions or comments by participants. Unless and to the extent determined
by the Board or the chairman of the meeting, meetings of stockholders shall not be required to be held in accordance with the rules
of parliamentary procedure.
(C)
The chairman of the meeting shall announce at the meeting when the polls for each matter to be voted upon at the meeting
will be opened and closed. After the polls close, no ballots, proxies or votes or any revocations or changes thereto may be accepted.
(D)
The Corporation may, and if required by law, shall, in advance of any meeting of stockholders, appoint one or more inspectors
of election to act at the meeting and make a written report thereof. One or more other persons may be designated as alternate inspectors
to replace any inspector who fails to act. If no inspector or alternate is able to act at a meeting of stockholders, the chairman
of the meeting shall appoint one or more inspectors to act at the meeting. Unless otherwise required by law, inspectors may be
officers, employees, agents or representatives of the Corporation. Each inspector, before entering upon the discharge of such inspector’s
duties, shall take and sign an oath faithfully to execute the duties of inspector with strict impartiality and according to the
best of such inspector’s ability. The inspector shall have the duties prescribed by law and, when the vote is completed,
shall certify their determination of the result of the vote taken and of such other facts as may be required by law. Every vote
taken by ballots shall be counted by a duly appointed inspector or duly appointed inspectors.
ARTICLE
II
DIRECTORS
2.1
General Powers. The business and affairs of the Corporation shall be managed by or under the direction of the Board,
who may exercise all of the powers of the Corporation except as otherwise provided by law or the Certificate of Incorporation.
2.2
Number, Election; Term and Qualification. The total number of directors constituting the Board shall be as fixed
in, or in the manner provided by, the Certificate of Incorporation. Election of directors need not be by written ballot. The term
of office of each director shall be as specified in the Certificate of Incorporation. Directors need not be stockholders of the
Corporation.
2.3
Chairman of the Board; Vice Chairman of the Board. The Board may appoint from its members a Chairman of the Board
and a Vice Chairman of the Board, neither of whom need be an employee or officer of the Corporation. If the Board appoints a Chairman
of the Board, such Chairman shall perform such duties and possess such powers as are assigned by the Board and, if the Chairman
of the Board is also designated as the Corporation’s Chief Executive Officer, shall have the powers and duties of the Chief
Executive Officer prescribed in Section 3.7 of these Bylaws. If the Board appoints a Vice Chairman of the Board, such Vice Chairman
shall perform such duties and possess such powers as are assigned by the Board. Unless otherwise provided by the Board, the Chairman
of the Board or, in the Chairman’s absence, the Vice Chairman of the Board, if any, shall preside at all meetings of the
Board.
2.4
Quorum. The greater of (a) a majority of the directors at any time in office and (b) one-third of the whole Board
shall constitute a quorum of the Board. If at any meeting of the Board there shall be less than a quorum, a majority of the directors
present may adjourn the meeting from time to time without further notice other than announcement at the meeting, until a quorum
shall be present.
2.5
Action at Meeting. Every act or decision done or made by a majority of the directors present at a meeting duly held
at which a quorum is present shall be regarded as the act of the Board, unless a greater number is required by law or by the Certificate
of Incorporation.
2.6
Removal. Subject to the rights of holders of any series of Preferred Stock, directors of the Corporation may be removed
only as expressly provided in the Certificate of Incorporation.
2.7
Newly Created Directorships; Vacancies. Any newly created directorship that results from an increase in the number
of directors or any vacancy on the Board that results from the death, disability, resignation, disqualification or removal of any
director or from any other cause shall be filled in accordance with the Certificate of Incorporation.
2.8
Resignation. Any director may resign by delivering a resignation in writing or by electronic transmission to the
Corporation at its principal office or to the Chairman of the Board, the Chief Executive Officer, the President or the Secretary.
Such resignation shall be effective upon delivery unless it is specified to be effective at some later time or upon the happening
of some later event.
2.9
Regular Meetings. Regular meetings of the Board may be held without notice at such time and place as shall be determined
from time to time by the Board; provided that any director who is absent when such a determination is made shall be given
notice of the determination. A regular meeting of the Board may be held without notice immediately after and at the same place
as the annual meeting of stockholders.
2.10
Special Meetings. Special meetings of the Board may be called by the Chairman of the Board, the Chief Executive Officer,
the affirmative vote of a majority of the directors then in office, or by one director in the event that there is only a single
director in office.
2.11
Notice of Special Meetings. Notice of the date, place and time of any special meeting of the Board shall be given
to each director (a) in person or by telephone at least twenty-four (24) hours in advance of the meeting, (b) by
sending written notice by reputable overnight courier, telecopy, electronic mail, facsimile or other means of electronic transmission,
or delivering written notice by hand, to such director’s last known business, home or means of electronic transmission address
at least twenty-four (24) hours in advance of the meeting, or (c) by sending written notice by first-class mail to such
director’s last known business or home address at least seventy-two (72) hours in advance of the meeting. Such notice
may be given by the Secretary or by the Chairman of the Board, the Chief Executive Officer or one of the directors calling the
meeting. A notice or waiver of notice of a meeting of the Board need not specify the purposes of the meeting.
2.12
Meetings by Conference Communications Equipment. Directors may participate in meetings of the Board or any committee
thereof by means of conference telephone or other communications equipment by means of which all persons participating in the meeting
can hear each other, and participation by such means shall constitute presence in person at such meeting.
2.13
Action by Consent. Any action required or permitted to be taken at any meeting of the Board or of any committee thereof
may be taken without a meeting, if all members of the Board or committee, as the case may be, consent to the action in writing
or by electronic transmission. After an action is taken, the consent or consents relating thereto shall be filed with the minutes
of proceedings of the Board, or the committee thereof, in the same paper or electronic form as the minutes are maintained.
2.14
Committees. The Board may designate one or more committees, each committee to consist of one or more of the directors
of the Corporation with such lawfully delegable powers and duties as the Board thereby confers, to serve at the pleasure of the
Board. The Board may designate one or more directors as alternate members of any committee, who may replace any absent or disqualified
member at any meeting of the committee. In the absence or disqualification of a member of a committee, the member or members of
the committee present at any meeting and not disqualified from voting, whether or not such member or members constitute a quorum,
may unanimously appoint another member of the Board to act at the meeting in the place of any such absent or disqualified member.
Any such committee, to the extent provided in the resolution of the Board and subject to the provisions of law, shall have and
may exercise all the powers and authority of the Board in the management of the business and affairs of the Corporation and may
authorize the seal of the Corporation to be affixed to all papers which may require it. Each such committee shall keep minutes
and make such reports as the Board may from time to time request. Except as otherwise provided in the Certificate of Incorporation,
these Bylaws, or the resolution of the Board designating the committee, a committee may create one or more subcommittees, each
subcommittee to consist of one or more members of the committee, and delegate to a subcommittee any or all of the powers and authority
of the committee.
2.15
Compensation of Directors. Directors may be paid such compensation for their services and such reimbursement for
expenses of attendance at meetings as the Board may from time to time determine. No such payment shall preclude any director from
serving the Corporation or any of its parent or subsidiary entities in any other capacity and receiving compensation for such service.
ARTICLE
III
OFFICERS
3.1
Titles. The officers of the Corporation may consist of a Chief Executive Officer, a President, a Chief Financial
Officer, a Treasurer and a Secretary and such other officers with such other titles as the Board shall from time to time determine.
The Board may appoint such other officers, including one or more Vice Presidents and one or more Assistant Treasurers or Assistant
Secretaries, as it may deem appropriate from time to time.
3.2
Election. The officers of the Corporation shall be elected by the Board.
3.3
Qualification. No officer need be a stockholder. Any two or more offices may be held by the same person.
3.4
Tenure. Except as otherwise provided by law, by the Certificate of Incorporation or by these Bylaws, each officer
shall hold office until such officer’s successor is duly elected and qualified, unless a different term is specified in the
resolution electing or appointing such officer, or until such officer’s earlier death, resignation, disqualification or removal.
3.5
Resignation and Removal. Any officer may resign by delivering a resignation in writing or by electronic transmission
to the Corporation at its principal office or to the Board, the Chief Executive Officer, the President or the Secretary. Such resignation
shall be effective upon receipt unless it is specified to be effective at some later time or upon the happening of some later event.
Any officer may be removed at any time, with or without cause, by the affirmative vote of a majority of the directors then in office.
Except as the Board may otherwise determine, no officer who resigns or is removed shall have any right to any compensation as an
officer for any period following such officer’s resignation or removal, or any right to damages on account of such removal,
whether such officer’s compensation be by the month or by the year or otherwise, unless such compensation is expressly provided
for in a duly authorized written agreement with the Corporation
3.6
Vacancies. The Board may fill any vacancy occurring in any office for any reason and may, in its discretion, leave
unfilled, for such period as it may determine, any offices. Each such successor shall hold office for the unexpired term of such
officer’s predecessor and until a successor is duly elected and qualified, or until such officer’s earlier death, resignation,
disqualification or removal.
3.7
President; Chief Executive Officer. Unless the Board has designated another person as the Corporation’s Chief
Executive Officer, the President shall be the Chief Executive Officer of the Corporation. The Chief Executive Officer shall have
general charge and supervision of the business of the Corporation subject to the direction of the Board, and shall perform all
duties and have all powers that are commonly incident to the office of chief executive or that are delegated to such officer by
the Board. The President shall perform such other duties and shall have such other powers as the Board or the Chief Executive Officer
(if the President is not the Chief Executive Officer) may from time to time prescribe. In the event of the absence, inability or
refusal to act of the Chief Executive Officer or the President (if the President is not the Chief Executive Officer), the Vice
President (or if there shall be more than one, the Vice Presidents in the order determined by the Board) shall perform the duties
of the Chief Executive Officer and when so performing such duties shall have all the powers of and be subject to all the restrictions
upon the Chief Executive Officer.
3.8
Vice Presidents/Other Officers. Each Vice President and any other officer designated by the Board shall perform such
duties and possess such powers as the Board or the Chief Executive Officer may from time to time prescribe. The Board may assign
to any Vice President the title of Executive Vice President or Senior Vice President, and may assign to any Vice President or other
officer any other title selected by the Board.
3.9
Secretary and Assistant Secretaries. The Secretary shall perform such duties and shall have such powers as the Board
or the Chief Executive Officer may from time to time prescribe. In addition, the Secretary shall perform such duties and have such
powers as are incident to the office of the secretary, including without limitation the duty and power to give notices of all meetings
of stockholders and special meetings of the Board, to attend all meetings of stockholders and the Board and keep a record of the
proceedings, to maintain a stock ledger and prepare lists of stockholders and their addresses as required, to be custodian of corporate
records and the corporate seal and to affix and attest to the same on documents.
Any Assistant Secretary shall perform such duties
and possess such powers as the Board, the Chief Executive Officer or the Secretary may from time to time prescribe. In the event
of the absence, inability or refusal to act of the Secretary, the Assistant Secretary (or if there shall be more than one, the
Assistant Secretaries in the order determined by the Board) shall perform the duties and exercise the powers of the Secretary.
The chairman of any meeting of the Board or
of stockholders may designate a temporary secretary to keep a record of any meeting.
3.10
Treasurer and Assistant Treasurers. The Treasurer shall perform such duties and shall have such powers as may from
time to time be assigned by the Board or the Chief Executive Officer. In addition, the Treasurer shall perform such duties and
have such powers as are incident to the office of treasurer, including without limitation the duty and power to keep and be responsible
for all funds and securities of the Corporation, to deposit funds of the Corporation in depositories selected in accordance with
these Bylaws, to disburse such funds as ordered by the Board, to make proper accounts of such funds, and to render as required
by the Board statements of all such transactions and of the financial condition of the Corporation.
The Assistant Treasurers shall perform such
duties and possess such powers as the Board, the Chief Executive Officer or the Treasurer may from time to time prescribe. In the
event of the absence, inability or refusal to act of the Treasurer, the Assistant Treasurer (or if there shall be more than one,
the Assistant Treasurers in the order determined by the Board) shall perform the duties and exercise the powers of the Treasurer.
3.11
Salaries. Officers of the Corporation shall be entitled to such salaries, compensation or reimbursement as shall
be fixed or allowed from time to time by the Board.
3.12
Delegation of Authority. The Board may from time to time delegate the powers or duties of any officer to any other
officer or agent, notwithstanding any provision hereof.
ARTICLE
IV
CAPITAL STOCK
4.1
Issuance of Stock. Subject to the provisions of the Certificate of Incorporation, the whole or any part of any unissued
balance of the authorized capital stock of the Corporation or the whole or any part of any shares of the authorized capital stock
of the Corporation held in the Corporation’s treasury may be issued, sold, transferred or otherwise disposed of by vote of
the Board in such manner, for such lawful consideration and on such terms as the Board may determine.
4.2
Stock Certificates; Uncertificated Shares. The shares of the Corporation shall be represented by certificates, provided
that the Board may provide by resolution or resolutions that some or all of any or all classes or series of the Corporation’s
stock shall be uncertificated shares. Every holder of stock of the Corporation represented by certificates shall be entitled to
have a certificate, in such form as may be prescribed by law and by the Board, representing the number of shares held by such holder
registered in certificate form. Each such certificate shall be signed in a manner that complies with Section 158 of the DGCL and
each of the Chief Executive Officer, the President, a Vice President, the Secretary, an Assistant Secretary, the Treasurer or an
Assistant Treasurer are duly authorized to sign such certificates by, or in the name of, the Corporation, unless otherwise expressly
provided in the resolution of the Board electing such officer.
Each certificate for shares of stock which are
subject to any restriction on transfer pursuant to the Certificate of Incorporation, these Bylaws, applicable securities laws or
any agreement among any number of stockholders or among such holders and the Corporation shall have conspicuously noted on the
face or back of the certificate either the full text of the restriction or a statement of the existence of such restriction.
If the Corporation shall be authorized to issue
more than one class of stock or more than one series of any class, the powers, designations, preferences and relative participating,
optional or other special rights of each class of stock or series thereof and the qualifications, limitations or restrictions of
such preferences and/or rights shall be set forth in full or summarized on the face or back of each certificate representing shares
of such class or series of stock, provided that in lieu of the foregoing requirements there may be set forth on the face
or back of each certificate representing shares of such class or series of stock a statement that the Corporation will furnish
without charge to each stockholder who so requests the powers, designations, preferences and relative, participating, optional
or other special rights of each class of stock or series thereof and the qualifications, limitations or restrictions of such preferences
and/or rights.
Within a reasonable time after the issuance
or transfer of uncertificated shares, the registered owner thereof shall be given a notice, in writing or by electronic transmission,
containing the information required to be set forth or stated on certificates pursuant to Sections 151, 156, 202(a) or 218(a) of
the DGCL or, with respect to Section 151 of DGCL, a statement that the Corporation will furnish without charge to each stockholder
who so requests the powers, designations, preferences and relative participating, optional or other special rights of each class
of stock or series thereof and the qualifications, limitations or restrictions of such preferences and/or rights.
4.3
Transfers. Shares of stock of the Corporation shall be transferable in the manner prescribed by law, the Certificate
of Incorporation and in these Bylaws. Transfers of shares of stock of the Corporation shall be made only on the books of the Corporation
or by transfer agents designated to transfer shares of stock of the Corporation. Subject to applicable law, shares of stock represented
by certificates shall be transferred only on the books of the Corporation by the surrender to the Corporation or its transfer agent
of the certificate representing such shares properly endorsed or accompanied by a written assignment or power of attorney properly
executed, and with such proof of authority or the authenticity of signature as the Corporation or its transfer agent may reasonably
require. Except as may be otherwise required by law, by the Certificate of Incorporation or by these Bylaws, the Corporation shall
be entitled to treat the record holder of stock as shown on its books as the owner of such stock for all purposes, including the
payment of dividends and the right to vote with respect to such stock, regardless of any transfer, pledge or other disposition
of such stock until the shares have been transferred on the books of the Corporation in accordance with the requirements of these
Bylaws.
4.4
Lost, Stolen or Destroyed Certificates. The Corporation may issue a new certificate or uncertificated shares in place
of any previously issued certificate alleged to have been lost, stolen or destroyed, upon such terms and conditions as the Board
may prescribe, including the presentation of reasonable evidence of such loss, theft or destruction and the giving of such indemnity
and posting of such bond sufficient to indemnify the Corporation against any claim that may be made against it on account of the
alleged loss, theft or destruction of any such certificate or the issuance of such new certificate or uncertificated shares.
4.5
Record Date. In order that the Corporation may determine the stockholders entitled to notice of any meeting of stockholders
or any adjournment thereof, the Board may fix a record date, which record date shall not precede the date upon which the resolution
fixing the record date is adopted by the Board, and which record date shall, unless otherwise required by law, not be more than
sixty (60) nor less than ten (10) days before the date of such meeting. If the Board so fixes a date, such date shall also be the
record date for determining the stockholders entitled to vote at such meeting unless the Board determines, at the time it fixes
such record date, that a later date on or before the date of the meeting shall be the date for making such determination. If no
record date is fixed by the Board, the record date for determining stockholders entitled to notice of or to vote at a meeting of
stockholders shall be at the close of business on the day next preceding the day on which notice is given, or, if notice is waived,
at the close of business on the day next preceding the day on which the meeting is held. A determination of stockholders of record
entitled to notice of or to vote at a meeting of stockholders shall apply to any adjournment of the meeting; provided, however,
that the Board may fix a new record date for determination of stockholders entitled to vote at the adjourned meeting, and in such
case shall also fix as the record date for stockholders entitled to notice of such adjourned meeting the same or an earlier date
as that fixed for determination of stockholders entitled to vote in accordance herewith at the adjourned meeting.
In order that the Corporation may determine
the stockholders entitled to receive payment of any dividend or other distribution or allotment of any rights, or entitled to exercise
any rights in respect of any change, conversion or exchange of stock or for the purpose of any other lawful action, the Board may
fix a record date, which shall not be more than sixty (60) days prior to such action. If no such record date is fixed, the record
date for determining stockholders for any such purpose shall be at the close of business on the day on which the Board adopts the
resolution relating thereto.
4.6
Regulations. The issue and registration of shares of stock of the Corporation shall be governed by such other regulations
as the Board may establish.
ARTICLE
V
GENERAL PROVISIONS
5.1
Fiscal Year. Except as from time to time otherwise designated by the Board, the fiscal year of the Corporation shall
begin on the first day of January of each year and end on the last day of December in each year.
5.2
Corporate Seal. The corporate seal shall be in such form as shall be approved by the Board.
5.3
Waiver of Notice. Whenever notice is required to be given by law, by the Certificate of Incorporation or by these
Bylaws, a written waiver signed by the person entitled to notice, or a waiver by electronic transmission by the person entitled
to notice, whether before, at or after the time of the event for which notice is to be given, shall be deemed equivalent to notice
required to be given to such person. Neither the business nor the purpose of any meeting need be specified in any such waiver.
Attendance of a person at a meeting shall constitute a waiver of notice of such meeting, except when the person attends a meeting
for the express purpose of objecting at the beginning of the meeting, to the transaction of any business because the meeting is
not lawfully called or convened.
5.4
Voting of Securities. Except as the Board may otherwise designate, the Chief Executive Officer, the President, the
Chief Financial Officer or the Treasurer may waive notice, vote, consent, or appoint any person or persons to waive notice, vote
or consent, on behalf of the Corporation, and act as, or appoint any person or persons to act as, proxy or attorney-in-fact for
the Corporation (with or without power of substitution and re-substitution), with respect to the securities of any other entity
which may be held by this Corporation.
5.5
Evidence of Authority. A certificate by the Secretary, or an Assistant Secretary, or a temporary Secretary, as to
any action taken by the stockholders, directors, a committee or any officer or representative of the Corporation shall as to all
persons who rely on the certificate in good faith be conclusive evidence of such action.
5.6
Certificate of Incorporation. All references in these Bylaws to the Certificate of Incorporation shall be deemed
to refer to the Certificate of Incorporation of the Corporation, as amended and/or restated and in effect from time to time.
5.7
Severability. Any determination that any provision of these Bylaws is for any reason inapplicable, illegal or ineffective
shall not affect or invalidate any other provision of these Bylaws.
5.8
Pronouns. All pronouns used in these Bylaws shall be deemed to refer to the masculine, feminine or neuter, singular
or plural, as the identity of the person or persons may require.
5.9
Manner of Notice. Without limiting the manner by which notice otherwise may be given effectively to stockholders,
any notice to stockholders given by the Corporation under any provision of the DGCL, the Certificate of Incorporation or these
Bylaws may be given in writing directed to the stockholder’s mailing address (or by electronic transmission directed to the
stockholder’s electronic mail address, as applicable) as it appears on the records of the Corporation. Notice shall be given
(i) if mailed, when deposited in the United States mail, (ii) if delivered by courier service, the earlier of when the notice is
received or left at the stockholder’s address, or (iii) if given by electronic mail, when directed at to such stockholder’s
electronic mail address (unless the stockholder has notified the Corporation in writing or by electronic transmission of an objection
to receiving notice by electronic mail or such notice is prohibited by the DGCL to be given by electronic transmission). A notice
by electronic mail must include a prominent legend that the communication is an important notice regarding the Corporation. A notice
by electronic mail shall be deemed to include any files attached thereto and any information hyperlinked to a website if such electronic
mail includes the contact information of an officer or agent of the Corporation who is available to assist with accessing such
files or information. Any notice to stockholders under any provision of the DGCL, the Certificate of Incorporation or these Bylaws
provided by electronic transmission (other than any such notice given by electronic mail) may only be given in a form consented
to by such stockholder, and any such notice by electronic transmission shall be deemed to be given as provided by the DGCL.
5.10
Electronic Transmission. For purposes of these Bylaws, “electronic transmission” means any form of communication,
not directly involving the physical transmission of paper, including the use of, or participation in, one or more electronic networks
or databases (including one or more distributed electronic networks or databases), that creates a record that may be retained,
retrieved, and reviewed by a recipient thereof, and that may be directly reproduced in paper form by such a recipient through an
automated process.
ARTICLE
VI
AMENDMENTS
These Bylaws may be altered, amended or repealed,
in whole or in part, or new Bylaws may be adopted by the Board or by the stockholders as expressly provided in the Certificate
of Incorporation.
ARTICLE
VII
INDEMNIFICATION AND ADVANCEMENT
7.1
Power to Indemnify in Actions, Suits or Proceedings other than Those by or in the Right of the Corporation. Subject
to Section 7.3, the Corporation shall indemnify any person who was or is a party or is threatened to be made a party to any threatened,
pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative (other than an action
by or in the right of the Corporation) by reason of the fact that such person is or was a director or officer of the Corporation,
or, while a director or officer of the Corporation, is or was serving at the request of the Corporation as a director, officer,
employee or agent of another Corporation, partnership, joint venture, trust, employee benefit plan or other enterprise, against
expenses (including attorneys’ fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by
such person in connection with such action, suit or proceeding if such person acted in good faith and in a manner such person reasonably
believed to be in or not opposed to the best interests of the Corporation, and, with respect to any criminal action or proceeding,
had no reasonable cause to believe such person’s conduct was unlawful. The termination of any action, suit or proceeding
by judgment, order, settlement, conviction, or upon a plea of nolo contendere or its equivalent, shall not, of itself, create a
presumption that the person did not act in good faith and in a manner which such person reasonably believed to be in or not opposed
to the best interests of the Corporation, and, with respect to any criminal action or proceeding, had reasonable cause to believe
that such person’s conduct was unlawful.
7.2
Power to Indemnify in Actions, Suits or Proceedings by or in the Right of the Corporation. Subject to Section 7.3,
the Corporation shall indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending
or completed action or suit by or in the right of the Corporation to procure a judgment in its favor by reason of the fact that
such person is or was a director or officer of the Corporation, or, while a director or officer of the Corporation, is or was serving
at the request of the Corporation as a director, officer, employee or agent of another Corporation, partnership, joint venture,
trust, employee benefit plan or other enterprise against expenses (including attorneys’ fees) actually and reasonably incurred
by such person in connection with the defense or settlement of such action or suit if such person acted in good faith and in a
manner such person reasonably believed to be in or not opposed to the best interests of the Corporation; except that no indemnification
shall be made in respect of any claim, issue or matter as to which such person shall have been adjudged to be liable to the Corporation
unless and only to the extent that the Court of Chancery or the court in which such action or suit was brought shall determine
upon application that, despite the adjudication of liability but in view of all the circumstances of the case, such person is fairly
and reasonably entitled to indemnity for such expenses which the Court of Chancery or such other court shall deem proper.
7.3
Authorization of Indemnification. Any indemnification under this Article VII (unless ordered by a court) shall be
made by the Corporation only as authorized in the specific case upon a determination that indemnification of the director or officer
is proper in the circumstances because such person has met the applicable standard of conduct set forth in Section 7.1 or Section
7.2, as the case may be. Such determination shall be made, with respect to a person who is a director or officer at the time of
such determination, (i) by a majority vote of the directors who are not parties to such action, suit or proceeding, even though
less than a quorum, or (ii) by a committee of such directors designated by a majority vote of such directors, even though less
than a quorum, or (iii) if there are no such directors, or if such directors so direct, by independent legal counsel in a written
opinion or (iv) by the stockholders. Such determination shall be made, with respect to former directors and officers, by any person
or persons having the authority to act on the matter on behalf of the Corporation. To the extent, however, that a present or former
director or officer of the Corporation has been successful on the merits or otherwise in defense of any action, suit or proceeding
set forth in Section 7.1 or Section 7.2 or in defense of any claim, issue or matter therein, such person shall be indemnified against
expenses (including attorneys’ fees) actually and reasonably incurred by such person in connection therewith, without the
necessity of authorization in the specific case.
7.4
Good Faith Defined. For purposes of any determination under Section 7.3, a person shall be deemed to have acted in
good faith and in a manner such person reasonably believed to be in or not opposed to the best interests of the Corporation, or,
with respect to any criminal action or proceeding, to have had no reasonable cause to believe such person’s conduct was unlawful,
if such person’s action is based on good faith reliance on the records or books of account of the Corporation or another
enterprise, or on information supplied to such person by the officers of the Corporation or another enterprise in the course of
their duties, or on the advice of legal counsel for the Corporation or another enterprise or on information or records given or
reports made to the Corporation or another enterprise by an independent certified public accountant or by an appraiser or other
expert selected with reasonable care by the Corporation or another enterprise. The term “another enterprise” as used
in this Section 7.4 shall mean any other corporation or any partnership, joint venture, trust, employee benefit plan or other enterprise
of which such person is or was serving at the request of the Corporation as a director, officer, employee or agent. The provisions
of this Section 7.4 shall not be deemed to be exclusive or to limit in any way the circumstances in which a person may be deemed
to have met the applicable standard of conduct set forth in Section 7.1 or 7.2, as the case may be.
7.5
Right of Claimant to Bring Suit. Notwithstanding any contrary determination in the specific case under Section 7.3,
and notwithstanding the absence of any determination thereunder, if a claim under Sections 7.1 or 7.2 of the Article VII is not
paid in full by the Corporation within (i) ninety (90) days after a written claim for indemnification has been received by the
Corporation, or (ii) thirty (30) days after a written claim for an advancement of expenses has been received by the Corporation,
the claimant may at any time thereafter (but not before) bring suit against the Corporation in the Court of Chancery in the State
of Delaware to recover the unpaid amount of the claim, together with interest thereon, or to obtain advancement of expenses, as
applicable. It shall be a defense to any such action brought to enforce a right to indemnification (but not in an action brought
to enforce a right to an advancement of expenses) that the claimant has not met the standards of conduct which make it permissible
under the DGCL (or other applicable law) for the Corporation to indemnify the claimant for the amount claimed, but the burden of
proving such defense shall be on the Corporation. Neither a contrary determination in the specific case under Section 7.3 nor the
absence of any determination thereunder shall be a defense to such application or create a presumption that the claimant has not
met any applicable standard of conduct. If successful, in whole or in part, the claimant shall also be entitled to be paid the
expense of prosecuting such claim, including reasonable attorneys’ fees incurred in connection therewith, to the fullest
extent permitted by applicable law.
7.6
Expenses Payable in Advance. Expenses, including without limitation attorneys’ fees, incurred by a current
or former director or officer in defending any civil, criminal, administrative or investigative action, suit or proceeding shall
be paid by the Corporation in advance of the final disposition of such action, suit or proceeding upon receipt of an undertaking
by or on behalf of such current or former director or officer to repay such amount if it shall ultimately be determined that such
person is not entitled to be indemnified by the Corporation as authorized in this Article VII.
7.7
Nonexclusivity of Indemnification and Advancement of Expenses. The rights to indemnification and advancement of expenses
provided by or granted pursuant to this Article VII shall not be deemed exclusive of any other rights to which those seeking indemnification
or advancement of expenses may be entitled under the Certificate of Incorporation, any agreement, vote of stockholders or disinterested
directors or otherwise, both as to action in such person’s official capacity and as to action in another capacity while holding
such office, it being the policy of the Corporation that, subject to Section 7.11, indemnification of the persons specified in
Sections 7.1 and 7.2 shall be made to the fullest extent permitted by law. The provisions of this Article VII shall not be deemed
to preclude the indemnification of any person who is not specified in Section 7.1 or 7.2 but whom the Corporation has the power
or obligation to indemnify under the provisions of the DGCL, or otherwise.
7.8
Insurance. The Corporation may purchase and maintain insurance on behalf of any person who is or was a director,
officer, employee or agent of the Corporation, or is or was serving at the request of the Corporation as a director, officer, employee
or agent of another corporation, partnership, joint venture, trust, employee benefit plan or other enterprise against any liability
asserted against such person and incurred by such person in any such capacity, or arising out of such person’s status as
such, whether or not the Corporation would have the power or the obligation to indemnify such person against such liability under
the provisions of this Article VII.
7.9
Certain Definitions. For purposes of this Article VII, references to “the Corporation” shall include,
in addition to the resulting corporation, any constituent corporation (including any constituent of a constituent) absorbed in
a consolidation or merger which, if its separate existence had continued, would have had power and authority to indemnify its directors,
officers, employees or agents so that any person who is or was a director, officer, employee or agent of such constituent corporation,
or is or was serving at the request of such constituent corporation as a director, officer, employee or agent of another corporation,
partnership, joint venture, trust, employee benefit plan or other enterprise, shall stand in the same position under the provisions
of this Article VII with respect to the resulting or surviving corporation as such person would have with respect to such constituent
corporation if its separate existence had continued. For purposes of this Article VII, references to “fines” shall
include any excise taxes assessed on a person with respect of any employee benefit plan; and references to “serving at the
request of the Corporation” shall include any service as a director, officer, employee or agent of the Corporation which
imposes duties on, or involves services by, such director, officer, employee or agent with respect to an employee benefit plan,
its participants or beneficiaries; and a person who acted in good faith and in a manner such person reasonably believed to be in
the interest of the participants and beneficiaries of an employee benefit plan shall be deemed to have acted in a manner “not
opposed to the best interests of the Corporation” as referred to in this Article VII.
7.10
Survival of Indemnification and Advancement of Expenses. The indemnification and advancement of expenses provided
by, or granted pursuant to, this Article VII shall, unless otherwise provided when authorized or ratified, continue as to a person
who has ceased to be a director or officer and shall inure to the benefit of the heirs, executors and administrators of such a
person.
7.11
Limitation on Indemnification. Notwithstanding anything contained in this Article VII to the contrary, except for
proceedings to enforce rights to indemnification (which shall be governed by Section 7.5), the Corporation shall not be obligated
to indemnify any director, officer, employee or agent in connection with an action, suit proceeding (or part thereof) initiated
by such person unless such action, suit or proceeding (or part thereof) was authorized by the Board.
7.12
Contract Rights. The obligations of the Corporation under this Article VII to indemnify, and advance expenses to,
a person who is or was a director or officer of the Corporation shall be considered a contract between the Corporation and such
person, and no modification or repeal of any provision of this Article VII shall affect, to the detriment of such person, such
obligations of the Corporation in connection with a claim based on any act or failure to act occurring before such modification
or repeal.
EXHIBIT “E” to proxy circular/prospectus
(Section 191 of the ABCA)
SECTION 191 OF THE BUSINESS CORPORATIONS ACT (ALBERTA)
Shareholder’s Right to Dissent
191(1) Subject to sections 192 and
242, a holder of shares of any class of a corporation may dissent
if the corporation resolves to
|
|
(a)
|
amend its articles under section 173 or 174 to add, change or remove any
provisions restricting or constraining the issue or transfer of shares of that class,
|
|
|
(b)
|
amend its articles under section 173 to add, change or remove any restrictions
on the business or businesses that the corporation may carry on,
|
(b.1) amend its articles under section
173 to add or remove an express statement establishing the unlimited liability of shareholders as set out in section 15.2(1),
|
|
(c)
|
amalgamate with another corporation, otherwise than under section 184 or 187,
|
|
|
(d)
|
be continued under the laws of another jurisdiction under section 189, or
|
|
|
(e)
|
sell, lease or exchange all or substantially all its property under section 190.
|
|
|
(2)
|
A holder of shares of any class or series of shares entitled to vote under
section 176, other than section 176(1)(a), may dissent if the corporation resolves to amend its articles in a manner described
in that section.
|
|
|
(3)
|
In addition to any other right the shareholder may have, but subject to subsection
(20), a shareholder entitled to dissent under this section and who complies with this section is entitled to be paid by the corporation
the fair value of the shares held by the shareholder in respect of which the shareholder dissents, determined as of the close of
business on the last business day before the day on which the resolution from which the shareholder dissents was adopted.
|
|
|
(4)
|
A dissenting shareholder may only claim under this section with respect
to all the shares of a class held by the shareholder or on behalf of any one beneficial owner and registered in the name of the
dissenting shareholder.
|
|
|
(5)
|
A dissenting shareholder shall send to the corporation a written objection
to a resolution referred to in subsection (1) or (2)
|
|
|
(a)
|
at or before any meeting of shareholders at which the resolution is to be voted on, or
|
|
|
(b)
|
if the corporation did not send notice to the shareholder of the purpose of the meeting or of the
shareholder’s right to dissent, within a reasonable time after the shareholder learns that the resolution was adopted and
of the shareholder’s right to dissent.
|
|
|
(6)
|
An application may be made to the Court after the adoption of a resolution
referred to in subsection (1) or (2),
|
|
|
(a)
|
by the corporation, or
|
|
|
(b)
|
by a shareholder if the shareholder has sent an objection to the corporation under subsection (5),
|
to fix the fair value in accordance with subsection (3) of the shares
of a shareholder who dissents under this section, or to fix the time at which a shareholder of an unlimited liability corporation
who dissents under this section ceases to become liable for any new liability, act or default of the unlimited liability corporation.
|
|
(7)
|
If an application is made under subsection (6), the corporation shall, unless
the Court otherwise orders, send to each dissenting shareholder a written offer to pay the shareholder an amount considered by
the Directors to be the fair value of the shares.
|
|
|
(8)
|
Unless the Court otherwise orders, an offer referred to in subsection (7)
shall be sent to each dissenting shareholder
|
|
|
(a)
|
at least 10 days before the date on which the application is returnable,
if the corporation is the applicant, or
|
|
|
(b)
|
within 10 days after the corporation is served with a copy of the application,
if a shareholder is the applicant.
|
|
|
(9)
|
Every offer made under subsection (7) shall
|
|
|
(a)
|
be made on the same terms, and
|
|
|
(b)
|
contain or be accompanied with a statement showing how the fair value was determined.
|
|
|
(10)
|
A dissenting shareholder may make an agreement with the corporation for
the purchase of the shareholder’s shares by the corporation, in the amount of the corporation’s offer under
|
subsection (7) or otherwise, at any time
before the Court pronounces an order fixing the fair value of the shares.
|
|
(11)
|
A dissenting shareholder
|
|
|
(a)
|
is not required to give security for costs in respect of an application under subsection (6), and
|
|
|
(b)
|
except in special circumstances must not be required to pay the costs of
the application or appraisal.
|
|
|
(12)
|
In connection with an application under subsection (6), the Court may give directions for
|
|
|
(a)
|
joining as parties all dissenting shareholders whose shares have not been
purchased by the corporation and for the representation of dissenting shareholders who, in the opinion of the Court, are in need
of representation,
|
|
|
(b)
|
the trial of issues and interlocutory matters, including pleadings and questioning
under Part 5 of the Alberta Rules of Court,
|
|
|
(c)
|
the payment to the shareholder of all or part of the sum offered by the
corporation for the shares,
|
|
|
(d)
|
the deposit of the share certificates with the Court or with the corporation
or its transfer agent,
|
|
|
(e)
|
the appointment and payment of independent appraisers, and the procedures
to be followed by them,
|
|
|
(f)
|
the service of documents, and
|
|
|
(g)
|
the burden of proof on the parties.
|
|
|
(13)
|
On an application under subsection (6), the Court shall make an order
|
|
|
(a)
|
fixing the fair value of the shares in accordance with subsection (3) of
all dissenting shareholders who are parties to the application,
|
|
|
(b)
|
giving judgment in that amount against the corporation and in favour of
each of those dissenting shareholders,
|
|
|
(c)
|
fixing the time within which the corporation must pay that amount to a shareholder, and
|
|
|
(d)
|
fixing the time at which a dissenting shareholder of an unlimited liability
corporation ceases to become liable for any new liability, act or default of the unlimited liability corporation.
|
|
|
(a)
|
the action approved by the resolution from which the shareholder dissents becoming effective,
|
|
|
(b)
|
the making of an agreement under subsection (10) between the corporation
and the dissenting shareholder as to the payment to be made by the corporation for the shareholder’s shares, whether by the
acceptance of the corporation’s offer under subsection (7) or otherwise, or
|
|
|
(c)
|
the pronouncement of an order under subsection (13),
|
whichever first occurs, the shareholder
ceases to have any rights as a shareholder other than the right to be paid the fair value of the shareholder’s shares in
the amount agreed to between the corporation and the shareholder or in the amount of the judgment, as the case may be.
|
|
(15)
|
Subsection (14)(a) does not apply to a shareholder referred to in subsection (5)(b).
|
|
|
(16)
|
Until one of the events mentioned in subsection (14) occurs,
|
|
|
(a)
|
the shareholder may withdraw the shareholder’s dissent, or
|
|
|
(b)
|
the corporation may rescind the resolution,
|
and in either event proceedings under this
section shall be discontinued.
|
|
(17)
|
The Court may in its discretion allow a reasonable rate of interest on
the amount payable to each dissenting shareholder, from the date on which the shareholder ceases to have any rights as a shareholder
by reason of subsection (14) until the date of payment.
|
|
|
(18)
|
If subsection (20) applies, the corporation shall, within 10 days after
|
|
|
(a)
|
the pronouncement of an order under subsection (13), or
|
|
|
(b)
|
the making of an agreement between the shareholder and the corporation
as to the payment to be made for the shareholder’s shares,
|
notify each dissenting shareholder
that it is unable lawfully to pay dissenting shareholders for their shares.
|
|
(19)
|
Notwithstanding that a judgment has been given in favour of a dissenting
shareholder under subsection (13)(b), if subsection (20) applies, the dissenting shareholder, by written notice delivered to the
corporation within 30 days after receiving the notice under subsection (18), may withdraw the shareholder’s notice of objection,
in which case the corporation is deemed to consent to the withdrawal and the shareholder is reinstated to the shareholder’s
full rights as a shareholder, failing which the shareholder retains a status as a claimant against the corporation, to be paid
as soon as the corporation is lawfully able to do so or, in a liquidation, to be ranked subordinate to the rights of creditors
of the corporation but in priority to its shareholders.
|
|
|
(20)
|
A corporation shall not make a payment to a dissenting shareholder under
this section if there are reasonable grounds for believing that
|
|
|
(a)
|
the corporation is or would after the payment be unable to pay its liabilities
as they become due, or
|
|
|
(b)
|
the realizable value of the corporation’s assets would by reason of
the payment be less than the aggregate of its liabilities.
|
EXHIBIT “F” to proxy circular/prospectus
(Form of Proxy Card)
iNSTRUMENT
OF pROXY
I/We, being holder(s) of common shares
(the “Common Shares”) in the capital of Zomedica Pharmaceuticals Corp. (the “Corporation”), hereby appoint:
Robert Cohen, Interim Chief Executive Officer or failing him, Ann Marie Cotter, Chief Financial Officer and Corporate Secretary,
OR
|
|
|
|
To appoint a proxyholder other than the management nominees indicated above, print that name here
|
|
as proxy of the undersigned, to attend,
act and vote on behalf of the undersigned in accordance with the below directions (or if no directions have been given, FOR
each of the matters listed below) on all the following matters and any other matter that may properly come before the virtual-only
special meeting (the “Special Meeting”) of shareholders of the Corporation to be held virtually at 8:30 a.m. (Toronto
time) on December 15, 2020, and at any and all adjournments or postponements thereof in the same manner, to the same extent and
with the same powers as if the undersigned were personally present, with full power of substitution. The Corporation is holding
the meeting as a completely virtual meeting, which will be conducted via live audio cast, online at https://web.lumiagm.com/157200202,
password “zomedica2020”.
Management recommends voting FOR Resolutions
1 to 2. Please use a dark black pencil or pen.
|
1.
|
Proposal 1: Domestication from Alberta to Delaware
|
|
FOR
|
AGAINST
|
|
|
|
Approval of a special resolution as set
out at Exhibit A of the accompanying proxy circular/prospectus authorizing the Corporation to change its jurisdiction of incorporation from the
Province of Alberta, Canada to the State of Delaware, United States of America pursuant to section 189 of the Business Corporations
Act (Alberta), as more particularly described in the accompanying proxy circular/prospectus
|
|
☐
|
☐
|
|
|
|
|
|
|
|
|
|
2.
|
Proposal 2: Approval of the 2020 Stock Incentive Plan
|
|
FOR
|
AGAINST
|
|
|
|
Approval of, assuming the domestication proposal is approved, the Zomedica Corp. 2020 Stock Incentive Plan, or the 2020 Plan, as more particularly described in the accompanying proxy circular/prospectus
|
|
☐
|
☐
|
|
|
|
|
|
|
|
|
I/We authorize you to act in accordance
with my/our instructions set out above. I/We hereby revoke any proxy previously given with respect to the Special Meeting. If
no voting instructions are indicated above, this proxy will be voted FOR each of the matters described above. I/We authorize the
appointee to vote as they see fit with respect to any amendments or variations of the matters of business to be acted on at the
Special Meeting or any other matters properly brought before the Special Meeting or any adjournment or postponement thereof, in
each instance, to the extent permitted by law, whether or not the amendment, variation or other matter that comes before the Special
Meeting is routine and whether or not the amendment, variation or other matter that comes before the Special Meeting is contested.
Please sign exactly as your name(s) appear
on this proxy. Please see instructions below. All proxies must be received by 8:30 a.m. (Toronto time) on December 13, 2020.
Notes to Proxy
1. This proxy
is solicited by and on behalf of management of the Corporation for use at the Special Meeting.
2. This
proxy must be signed by a holder or his or her attorney duly authorized in writing. If you are an individual, please sign exactly
as your name appears on this proxy. If the holder is a corporation, a duly authorized officer or attorney of the corporation must
sign this proxy indicating your signing capacity, and you may be required to provide documentation evidencing your power to sign
this proxy.
3. If
the securities are registered in the name of an executor, administrator or trustee, please sign exactly as your name appears on
this proxy. If the securities are registered in the name of a deceased or other holder, the proxy must be signed by the legal representative
with his or her name printed below his or her signature, and evidence of authority to sign on behalf of the deceased or other holder
must be attached to this proxy.
4. If
a security is held by two or more individuals, then one of these individuals, in the absence of the others, may sign the proxy.
5. Every
shareholder has the right to appoint some other person or company of the shareholder’s choice, who need not be a shareholder
of the Corporation, to attend at and represent the shareholder at the Special Meeting or any adjournment or postponement thereof.
If you wish to appoint a person or company other than the persons who names are printed above, please insert the name of your chosen
proxyholder in the space provided above and return your proxy by mail, fax or email as provided below. The proxy must be received
by AST Trust Company (Canada) (“AST”), on behalf of the Corporation, by 8:30 a.m. (Toronto time) on December 13, 2020.
In addition, you must contact AST at 1-866-751-6315 (within North America) or 212-235-5754 (outside North America) and provide
AST with the required information for your proxyholder so that AST may provide the proxyholder with a control number. This also
must occur by 8:30 a.m. (Toronto time) on December 13, 2020. This control number will allow your proxyholder to log into and vote
at the Special Meeting. Without a control number, your proxyholder will only be able to log into the Special Meeting online as
a guest, and will not be able to vote or ask questions at the Special Meeting.
6. The
Common Shares of the Corporation represented by this proxy will be voted as directed by the holder. However, if such a direction
is not made in respect of any matter, then the Common Shares will be voted FOR each matter. This proxy confers discretionary authority
with respect to any amendments or variations of the matters of business to be acted on at the Special Meeting or any other matters
properly brought before the Special Meeting or any adjournment or postponement thereof, in each instance, to the extent permitted
by law, whether or not the amendment, variation or other matter that comes before the Special Meeting is routine and whether or
not the amendment, variation or other matter that comes before the Special Meeting is contested.
All holders should refer to the Corporation’s proxy circular/prospectus
dated October , 2020 for further information regarding completion and use of this proxy and other information pertaining to the
Special Meeting.
|
MAIL, FAX, EMAIL, INTERNET
or TELEPHONE
Complete and return your signed proxy in
the envelope provided or send to:
AST Trust Company (Canada)
P.O. Box 721
Agincourt, ON M1S 0A1
You may alternatively fax your
proxy to 416-368-2502 or toll free in Canada and United States to 1-866-781-3111 or scan and email to proxyvote@astfinancial.com.
When submitting your proxy please be sure to submit both sides
in order to capture the voting control number.
By Internet: You may alternatively vote on the Internet
by going to www.astvotemyproxy.com and enter your control number.
By Telephone: Using a touch-tone telephone, call toll
free 1-888-489-5760 (English only) and enter your control number.
If you vote by Internet or Telephone, DO NOT return the proxy
by mail, fax or email.
An undated proxy is deemed to be dated on the day it was received by AST Trust Company (Canada).
|
iNSTRUMENT
OF pROXY
I/We, being holder(s) of Series 1 preferred
shares (the “Preferred Shares”) in the capital of Zomedica Pharmaceuticals Corp. (the “Corporation”), hereby
appoint: Robert Cohen, Interim Chief Executive Officer or failing him, Ann Marie Cotter, Chief Financial Officer and Corporate
Secretary, OR
|
|
|
|
To appoint a proxyholder other than the management nominees indicated above, print that name here
|
|
as proxy of the undersigned, to attend,
act and vote on behalf of the undersigned in accordance with the below directions (or if no directions have been given, FOR
each of the matters listed below) on all the following matters and any other matter that may properly come before the virtual-only
special meeting (the “Special Meeting”) of shareholders of the Corporation to be held virtually at 8:30 a.m. (Toronto
time) on December 15, 2020, and at any and all adjournments or postponements thereof in the same manner, to the same extent and
with the same powers as if the undersigned were personally present, with full power of substitution. The Corporation is holding
the meeting as a completely virtual meeting, which will be conducted via live audio cast, online at https://web.lumiagm.com/157200202,
password “zomedica2020”.
Management recommends voting FOR Resolution
1. Please use a dark black pencil or pen.
|
1.
|
Proposal 1: Domestication from Alberta to Delaware
|
|
FOR
|
AGAINST
|
|
|
|
Approval of a special resolution as set
out at Exhibit A
of the accompanying proxy circular/prospectus authorizing the Corporation to change its jurisdiction of incorporation from the
Province of Alberta, Canada to the State of Delaware, United States of America pursuant to section 189 of the Business Corporations
Act (Alberta), as more particularly described in the accompanying proxy circular/prospectus
|
|
☐
|
☐
|
|
|
|
|
|
|
|
|
I/We authorize you to act in accordance
with my/our instructions set out above. I/We hereby revoke any proxy previously given with respect to the Special Meeting. If
no voting instructions are indicated above, this proxy will be voted FOR each of the matters described above. I/We authorize the
appointee to vote as they see fit with respect to any amendments or variations of the matters of business to be acted on at the
Special Meeting or any other matters properly brought before the Special Meeting or any adjournment or postponement thereof, in
each instance, to the extent permitted by law, whether or not the amendment, variation or other matter that comes before the Special
Meeting is routine and whether or not the amendment, variation or other matter that comes before the Special Meeting is contested.
Please sign exactly as your name(s) appear
on this proxy. Please see instructions below. All proxies must be received by 8:30 a.m. (Toronto time) on December 13, 2020.
Notes to Proxy
1. This
proxy is solicited by and on behalf of management of the Corporation for use at the Special Meeting.
2. This
proxy must be signed by a holder or his or her attorney duly authorized in writing. If you are an individual, please sign exactly
as your name appears on this proxy. If the holder is a corporation, a duly authorized officer or attorney of the corporation must
sign this proxy indicating your signing capacity, and you may be required to provide documentation evidencing your power to sign
this proxy.
3. If
the securities are registered in the name of an executor, administrator or trustee, please sign exactly as your name appears on
this proxy. If the securities are registered in the name of a deceased or other holder, the proxy must be signed by the legal representative
with his or her name printed below his or her signature, and evidence of authority to sign on behalf of the deceased or other holder
must be attached to this proxy.
4. If
a security is held by two or more individuals, then one of these individuals, in the absence of the others, may sign the proxy.
5. Every
shareholder has the right to appoint some other person or company of the shareholder’s choice, who need not be a shareholder
of the Corporation, to attend at and represent the shareholder at the Special Meeting or any adjournment or postponement thereof.
If you wish to appoint a person or company other than the persons who names are printed above, please insert the name of your chosen
proxyholder in the space provided above and return your proxy by mail, fax or email as provided below. The proxy must be received
by AST Trust Company (Canada) (“AST”), on behalf of the Corporation, by 8:30 a.m. (Toronto time) on December 13, 2020.
In addition, you must contact AST at 1-866-751-6315 (within North America) or 212-235-5754 (outside North America) and provide
AST with the required information for your proxyholder so that AST may provide the proxyholder with a control number. This also
must occur by 8:30 a.m. (Toronto time) on December 13, 2020. This control number will allow your proxyholder to log into and vote
at the Special Meeting. Without a control number, your proxyholder will only be able to log into the Special Meeting online as
a guest, and will not be able to vote or ask questions at the Special Meeting.
6. The
Preferred Shares of the Corporation represented by this proxy will be voted as directed by the holder. However, if such a direction
is not made in respect of any matter, then the Preferred Shares will be voted FOR each matter. This proxy confers discretionary
authority with respect to any amendments or variations of the matters of business to be acted on at the Special Meeting or any
other matters properly brought before the Special Meeting or any adjournment or postponement thereof, in each instance, to the
extent permitted by law, whether or not the amendment, variation or other matter that comes before the Special Meeting is routine
and whether or not the amendment, variation or other matter that comes before the Special Meeting is contested.
All holders should refer to the Corporation’s proxy circular/prospectus
dated October , 2020 for further information regarding completion and use of this proxy and other information pertaining to the
Special Meeting.
|
MAIL, FAX, EMAIL, INTERNET
or TELEPHONE
Complete and return your signed proxy in
the envelope provided or send to:
AST Trust Company (Canada)
P.O. Box 721
Agincourt, ON M1S 0A1
You may alternatively fax your
proxy to 416-368-2502 or toll free in Canada and United States to 1-866-781-3111 or scan and email to proxyvote@astfinancial.com.
When submitting your proxy please be sure to submit both sides
in order to capture the voting control number.
By Internet: You may alternatively vote on the Internet
by going to www.astvotemyproxy.com and enter your control number.
By Telephone: Using a touch-tone telephone, call toll
free 1-888-489-5760 (English only) and enter your control number.
If you vote by Internet or Telephone, DO NOT return this proxy
by mail, fax or email.
An undated proxy is deemed to be dated on the day it was received by AST Trust Company (Canada).
|
EXHIBIT “G” to proxy circular/prospectus
(2020 Stock Incentive Plan)
ZOMEDICA CORP.
2020 STOCK INCENTIVE PLAN
Section 1. Establishment and Purpose
The purpose of the
Zomedica Corp. 2020 Stock Incentive Plan (the “Plan”) is to provide a means whereby eligible employees, officers,
non-employee directors and other individual service providers of Zomedica Corp. (the “Company”) and its subsidiaries
may develop a sense of proprietorship and personal involvement in the development and financial success of the Company and to encourage
them to devote their best efforts to the business of the Company, thereby advancing the interests of the Company and its stockholders.
The Company, by means of the Plan, seeks to retain the services of such eligible persons and to provide incentives for such persons
to exert maximum efforts for the success of the Company and its subsidiaries.
The Plan permits the
grant of Nonqualified Stock Options, Incentive Stock Options, Stock Appreciation Rights, Restricted Stock, Stock Units, Performance
Shares, Performance Units, Incentive Bonus Awards, Other Cash-Based Awards and Other Stock-Based Awards. This Plan, as amended
and restated, shall become effective upon the date set forth in Section 17.1 hereof.
Section 2. Definitions
Wherever the following
capitalized terms are used in the Plan, they shall have the meanings specified below:
2.1 “Affiliate”
means, with respect to a Person, a Person that directly or indirectly Controls, or is Controlled by, or is under common Control
with, such Person.
2.2 “Applicable
Law” means the requirements relating to the administration of equity-based awards or equity compensation plans under
U.S. state corporate laws, U.S. federal and state securities laws, the Code, any stock exchange or quotation system on which the
Common Stock is listed or quoted and the applicable laws of any foreign country or jurisdiction where Awards are, or will be, granted
under the Plan.
2.3 “Award”
means an award of a Stock Option, Stock Appreciation Right, Restricted Stock, Stock Unit, Performance Share, Performance Unit,
Incentive Bonus Award, Other Cash-Based Award and/or Other Stock-Based Award granted under the Plan.
2.4 “Award
Agreement” means either (i) a written or electronic agreement entered into between the Company and a Participant
setting forth the terms and conditions of an Award including any amendment or modification thereof, or (ii) a written or electronic
statement issued by the Company to a Participant describing the terms and provisions of such Award, including any amendment or
modification thereof. The Committee may provide for the use of electronic, internet or other non-paper Award Agreements, and the
use of electronic, internet or other non-paper means for the acceptance thereof and actions thereunder by a Participant. Each Award
Agreement shall be subject to the terms and conditions of the Plan and need not be identical.
2.5 “Board”
means the Board of Directors of the Company.
2.6 “Cause”
means: (a) conviction of any crime (whether or not involving the Company) constituting a felony in the jurisdiction involved; (b)
engaging in any substantiated act involving moral turpitude; (c) engaging in any act which, in each case, subjects, or if generally
known would subject, the Company to public ridicule or embarrassment; (d) material violation of the Company’s policies, including,
without limitation, those relating to sexual harassment or the disclosure or misuse of confidential information; (e) serious neglect
or misconduct in the performance of the grantee’s duties for the Company or a subsidiary or willful or repeated failure or
refusal to perform such duties; in each case as determined by the Committee, which determination shall be final, binding and conclusive.
Notwithstanding the foregoing, if a Participant and the Company (or any of its Affiliates) have entered into an employment agreement,
consulting agreement or other similar agreement that specifically defines “cause,” then with respect to such Participant,
“Cause” shall have the meaning defined in that employment agreement, consulting agreement or other agreement.
2.7 “Change
in Control” shall be deemed to have occurred if any one of the following events shall occur:
(i) Any
Person becomes the beneficial owner (as defined in Rule 13d-3 under the Exchange Act) of shares of Common Stock representing more
than 50% of the total number of votes that may be cast for the election of directors of the Company;
(ii) The
consummation of any merger or other business combination of the Company, sale of all or substantially all of the Company’s
assets or combination of the foregoing transactions (a “Transaction”), other than a Transaction involving only
the Company and one or more of its subsidiaries, or a Transaction immediately following which the stockholders of the Company immediately
prior to the Transaction continue to have a majority of the voting power in the resulting entity;
(iii) Within
any 12-month period beginning on or after the Effective Date, the persons who were directors of the Company immediately before
the beginning of such period (the “Incumbent Directors”) shall cease (for any reason other than death) to constitute
at least a majority of the Board (or the board of directors of any successor to the Company); provided that any director who was
not a director as of the date hereof shall be deemed to be an Incumbent Director if such director was elected to the Board by,
or on the recommendation of or with the approval of, at least two-thirds of the directors who then qualified as Incumbent Directors
either actually or by prior operation of the foregoing unless such election, recommendation or approval was the result of an actual
or threatened election contest of the type contemplated by Rule 14a-11 promulgated under the Exchange Act or any successor provision;
or
(iv) the
stockholders of the Company approve a plan of complete liquidation or dissolution of the Company.
Notwithstanding
the foregoing, no event or condition shall constitute a Change in Control to the extent that, if it were, a penalty tax would be
imposed under Section 409A of the Code; provided that, in such a case, the event or condition shall continue to constitute a Change
in Control to the maximum extent possible (e.g., if applicable, in respect of vesting without an acceleration of distribution)
without causing the imposition of such penalty tax.
2.8 “Code”
means the Internal Revenue Code of 1986, as amended. For purposes of this Plan, references to sections of the Code shall be deemed
to include references to any applicable regulations thereunder and any successor or similar provision.
2.9 “Committee”
means the committee of the Board delegated with the authority to administer the Plan, or the full Board, as provided in Section 3
of the Plan. With respect to any decision relating to a Reporting Person, the Committee shall consist solely of two or more directors
who are disinterested within the meaning of Rule 16b-3 promulgated under the Exchange Act, as amended from time to time, or any
successor provision. The fact that a Committee member shall fail to qualify under any of these requirements shall not invalidate
an Award if the Award is otherwise validly made under the Plan. The Board may at any time appoint additional members to the Committee,
remove and replace members of the Committee with or without Cause, and fill vacancies on the Committee however caused.
2.10 “Common
Stock” means the Company’s Common Stock, par value $0.0001 per share.
2.11 “Company”
means Zomedica Corp., a Delaware corporation, and any successor thereto as provided in Section 15.8.
2.12 “Control”
means, as to any Person, the power to direct or cause the direction of the management and policies of such Person, or the power
to appoint directors of the Company, whether through the ownership of voting securities, by contract or otherwise (the terms “Controlled
by” and “under common Control with” shall have correlative meanings).
2.13 “Date
of Grant” means the date on which an Award under the Plan is granted by the Committee, or such later date as the Committee
may specify to be the effective date of an Award.
2.14 “Disability”
means a Participant being considered “disabled” within the meaning of Section 409A of the Code and Treasury Regulation
1.409A-3(i)(4), as well as any successor regulation or interpretation.
2.15 “Effective
Date” means the date set forth in Section 17.1 hereof.
2.16 “Eligible
Person” means any person who is an employee, officer, director, consultant, advisor or other individual service provider
of the Company or any Subsidiary, or any person who is determined by the Committee to be a prospective employee, officer, director,
consultant, advisor or other individual service provider of the Company or any Subsidiary; provided that the Award Agreement for
any grant of an Award to a prospective employee, officer, director, consultant, advisor or other individual service provider will
contain appropriate forfeiture provisions in the event such individual does not become employed or engaged by the Company or applicable
Subsidiary .
2.17 “Exchange
Act” means the Securities Exchange Act of 1934, as amended.
2.18 “Fair
Market Value” of a share of Common Stock shall be, as applied to a specific date (i) the closing price of a share
of Common Stock as of such date on the principal established stock exchange or national market system on which the Common Stock
is then traded (or, if there is no trading in the Common Stock as of such date, the closing price of a share of Common Stock on
the most recent date preceding such date on which trades of the Common Stock were recorded), or (ii) if the shares of Common
Stock are not then traded on an established stock exchange or national market system but are then traded in an over-the-counter
market, the average of the closing bid and asked prices for the shares of Common Stock in such over-the-counter market as of such
date (or, if there are no closing bid and asked prices for the shares of Common Stock as of such date, the average of the closing
bid and the asked prices for the shares of Common Stock on the most recent date preceding such date on which such closing bid and
asked prices are available on such over-the-counter market), or (iii) if the shares of Common Stock are not then listed on
a national securities exchange or national market system or traded in an over-the-counter market, the price of a share of Common
Stock as determined by the Committee in its discretion in a manner consistent with Section 409A of the Code and Treasury Regulation
1.409A-1(b)(5)(iv), as well as any successor regulation or interpretation.
2.19 “Incentive
Bonus Award” means an Award granted under Section 12 of the Plan.
2.20 “Incentive
Stock Option” means a Stock Option granted under Section 6 hereof that is intended to meet the requirements of Section
422 of the Code and the regulations promulgated thereunder.
2.21 “Nonqualified
Stock Option” means a Stock Option granted under Section 6 hereof that is not an Incentive Stock Option.
2.22 “Other
Cash-Based Award” means a contractual right granted to an Eligible Person under Section 13 hereof entitling such
Eligible Person to receive a cash payment at such times, and subject to such conditions, as are set forth in the Plan and the applicable
Award Agreement.
2.23 “Other
Stock-Based Award” means a contractual right granted to an Eligible Person under Section 13 representing a notional
unit interest equal in value to a share of Common Stock to be paid and distributed at such times, and subject to such conditions
as are set forth in the Plan and the applicable Award Agreement.
2.24 “Participant”
means any Eligible Person who holds an outstanding Award under the Plan.
2.25 “Performance
Shares” means a contractual right granted to an Eligible Person under Section 10 hereof representing a notional
unit interest equal in value to a share of Common Stock to be paid and distributed at such times, and subject to such conditions,
as are set forth in the Plan and the applicable Award Agreement.
2.26 “Performance
Unit” means a contractual right granted to an Eligible Person under Section 11 hereof representing a notional dollar
interest as determined by the Committee to be paid and distributed at such times, and subject to such conditions, as are set forth
in the Plan and the applicable Award Agreement.
2.27 “Person”
shall mean any individual, partnership, firm, trust, corporation, limited liability company or other similar entity. When two or
more Persons act as a partnership, limited partnership, syndicate or other group for the purpose of acquiring, holding or disposing
of Common Stock, such partnership, limited partnership, syndicate or group shall be deemed a “Person”.
2.28 “Plan”
means the Zomedica Corp. 2020 Stock Incentive Plan, as set forth herein and as may be amended from time to time.
2.29 “Reporting
Person” means an officer, director or greater than ten percent stockholder of the Company within the meaning of Rule
16a-2 under the Exchange Act, who is required to file reports pursuant to Rule 16a-3 under the Exchange Act.
2.30 “Restricted
Stock Award” means a grant of shares of Common Stock to an Eligible Person under Section 8 hereof that are issued
subject to such vesting and transfer restrictions and such other conditions as are set forth in the Plan and the applicable Award
Agreement.
2.31 “Securities
Act” means the Securities Act of 1933, as amended.
2.32 “Service”
means a Participant’s employment or other service relationship with the Company or any Subsidiary. A change in the capacity
in which a Participant renders service to the Company or a Subsidiary as an employee, director or consultant or a change in the
entity for which the Participant renders such service, provided that there is no interruption or termination of the Participant’s
Service with the Company or a Subsidiary, will not terminate a Participant’s Service; provided, however, that if the entity
for which a Participant is rendering services ceases to qualify as a Subsidiary, as determined by the Committee in its sole discretion,
such Participant’s Service will be considered to have terminated on the date such entity ceases to qualify as a Subsidiary.
For example, a change in status from an employee of the Company to a consultant to or director of the Company will not constitute
an interruption of Service. To the extent permitted by Applicable Law, the Committee or the chief executive officer of the Company,
in that party’s sole discretion, may determine whether a Participant’s Service will be considered interrupted in the
case of (i) any leave of absence approved by the Company or chief executive officer, including sick leave, military leave or any
other personal leave, or (ii) transfers between the Company, a Subsidiary, or their successors. Notwithstanding the foregoing,
a leave of absence will be treated as Service for purposes of vesting in an Award only to such extent as may be provided in the
Company’s (or a Subsidiary’s) leave of absence policy, in the written terms of any leave of absence agreement or policy
applicable to the Participant, or as otherwise required by Applicable Law. Unless the Committee provides otherwise, in its discretion,
or as otherwise required by Applicable Law, vesting of Options shall be tolled during any unpaid leave of absence by a Participant.
2.33 “Stock
Appreciation Right” means a contractual right granted to an Eligible Person under Section 7 hereof entitling such
Eligible Person to receive a payment, upon the exercise of such right, in such amount and at such time, and subject to such conditions,
as are set forth in the Plan and the applicable Award Agreement.
2.34 “Stock
Option” means a contractual right granted to an Eligible Person under Section 6 hereof to purchase shares of Common
Stock at such time and price, and subject to such conditions, as are set forth in the Plan and the applicable Award Agreement.
2.35 “Stock
Unit Award” means a contractual right granted to an Eligible Person under Section 9 hereof representing notional
unit interests equal in value to a share of Common Stock to be paid and distributed at such times, and subject to such conditions,
as are set forth in the Plan and the applicable Award Agreement.
2.36 “Subsidiary”
means an entity (whether or not a corporation) that is wholly or majority owned or Controlled, directly or indirectly, by the Company;
provided, however, that with respect to Incentive Stock Options, the term “Subsidiary” shall include only an entity
that qualifies under Section 424(f) of the Code as a “subsidiary corporation” with respect to the Company.
Section 3. Administration
3.1 Committee
Members. The Plan shall be administered by the Committee; provided that the entire Board may act in lieu of the Committee on
any matter, subject to the requirements of Section 2.9 of the Plan with respect to an Award to a Reporting Person. If and to the
extent permitted by Applicable Law, the Committee may authorize one or more Reporting Persons (or other officers) to make Awards
to Eligible Persons who are not Reporting Persons (or other officers whom the Committee has specifically authorized to make Awards).
Subject to Applicable Law and the restrictions set forth in the Plan, the Committee may delegate administrative functions to individuals
who are Reporting Persons, officers, or employees of the Company or its Subsidiaries.
3.2 Committee
Authority. The Committee shall have such powers and authority as may be necessary or appropriate for the Committee to carry
out its functions as described in the Plan. Subject to the express limitations of the Plan, the Committee shall have authority
in its discretion to determine the Eligible Persons to whom, and the time or times at which, Awards may be granted, the number
of shares, units or other rights subject to each Award, the exercise, base or purchase price of an Award (if any), the time or
times at which an Award will become vested, exercisable or payable, the performance criteria, performance goals and other conditions
of an Award, the duration of the Award, and all other terms of the Award. Subject to the terms of the Plan, the Committee shall
have the authority to amend the terms of an Award in any manner that is not inconsistent with the Plan (including to extend the
post-termination exercisability period of Stock Options and Stock Appreciation Rights), provided that no such action (except an
action relating to a Change in Control) shall materially impair the rights of a Participant with respect to an outstanding Award
without the Participant’s consent. For purposes of the foregoing, any action of the Committee that alters or affects the
tax treatment of any Award shall not be considered to materially impair any rights of any Participant. The Committee shall also
have discretionary authority to interpret the Plan, to make all factual determinations under the Plan, and to make all other determinations
necessary or advisable for Plan administration, including, without limitation, to correct any defect, to supply any omission or
to reconcile any inconsistency in the Plan or any Award Agreement hereunder. The Committee may prescribe, amend, and rescind rules
and regulations relating to the Plan. The Committee’s determinations under the Plan need not be uniform and may be made by
the Committee selectively among Participants and Eligible Persons, whether or not such persons are similarly situated. The Committee
shall, in its discretion, consider such factors as it deems relevant in making its interpretations, determinations and actions
under the Plan including, without limitation, the recommendations or advice of any officer or employee of the Company or such attorneys,
consultants, accountants or other advisors as it may select. All interpretations, determinations, and actions by the Committee
shall be final, conclusive, and binding upon all parties.
3.3 No
Liability; Indemnification. Neither the Board nor any Committee member, nor any Person acting at the direction of the Board
or the Committee, shall be liable for any act, omission, interpretation, construction or determination made in good faith with
respect to the Plan, any Award or any Award Agreement. The Company and its Subsidiaries shall pay or reimburse any member of the
Committee, as well as any other Person who takes action on behalf of the Plan, for all reasonable expenses incurred with respect
to the Plan, and to the full extent allowable under Applicable Law shall indemnify each and every one of them for any claims, liabilities,
and costs (including reasonable attorney’s fees) arising out of their good faith performance of duties on behalf of the Company
with respect to the Plan. The Company and its Subsidiaries may, but shall not be required to, obtain liability insurance for this
purpose.
Section 4. Shares Subject to the
Plan
4.1 Share
Limitation.
(a)
Subject to adjustment pursuant to Section 4.3 hereof, the maximum aggregate number of shares
of Common Stock which may be issued under all Awards granted to Participants under the Plan shall be 200,000,000 shares (the “Initial
Limit”), all of which may, but need not, be issued in respect of Incentive Stock
(b)
Shares of Common Stock issued under the Plan may be either authorized but unissued shares or shares held in the Company’s
treasury. Any shares of Common Stock subject to Awards that are settled in Common Stock shall be counted against the maximum share
limitations of this Section 4.1(a) as one share of Common Stock for every share of Common Stock subject thereto. To the extent
that any Award under the Plan payable in shares of Common Stock is forfeited, cancelled, returned to or repurchased by the Company
for failure to satisfy vesting requirements or upon the occurrence of other forfeiture events, or otherwise terminates without
payment being made thereunder, the shares of Common Stock covered thereby will no longer be counted against the foregoing maximum
share limitations and may again be made subject to Awards under the Plan pursuant to such limitations. Shares of Common Stock that
otherwise would have been issued upon the exercise of a Stock Option or Stock Appreciation Right or in payment with respect to
any other form of Award, that are surrendered in payment or partial payment of the exercise price thereof and/or taxes withheld
with respect to the exercise thereof or the making of such payment, will no longer be counted against the foregoing maximum share
limitations and may again be made subject to Awards under the Plan pursuant to such limitations.
4.2 Individual
Participant Limitations. Subject to adjustment as provided in Section 4.3, the number of shares of Common Stock with respect
to which Awards may be granted during any calendar year to any one Eligible Person who is a non-employee director of the Board
shall not exceed 10,000,000.
4.3 Adjustments.
If there shall occur any change with respect to the outstanding shares of Common Stock by reason of any recapitalization, reclassification,
stock dividend, extraordinary dividend, stock split, reverse stock split, or other distribution with respect to the shares of Common
Stock, or any merger, reorganization, consolidation, combination, spin-off or other similar corporate change, or any other change
affecting the Common Stock, the Committee shall, in the manner and to the extent that it deems appropriate and equitable to the
Participants and consistent with the terms of the Plan, cause an adjustment to be made in (i) the maximum numbers and kind
of shares provided in Sections 4.1 and 4.2 hereof, (ii) the numbers and kind of shares of Common Stock, units, or other
rights subject to then outstanding Awards, (iii) the price for each share or unit or other right subject to then outstanding
Awards, (iv) the performance measures or goals relating to the vesting of an Award, and (v) any other terms of an Award
that are affected by the event to prevent dilution or enlargement of a Participant’s rights under an Award. Notwithstanding
the foregoing, in the case of Incentive Stock Options, any such adjustments shall, to the extent practicable, be made in a manner
consistent with the requirements of Section 424(a) of the Code.
Section 5. Participation and Awards
5.1 Designation
of Participants. All Eligible Persons are eligible to be designated by the Committee to receive Awards and become Participants
under the Plan. The Committee has the authority, in its discretion, to determine and designate from time to time those Eligible
Persons who are to be granted Awards, the types of Awards to be granted and the number of shares of Common Stock, units or other
amounts subject to such Awards. In selecting Eligible Persons to be Participants and in determining the type and amount of Awards
to be granted under the Plan, the Committee shall consider any and all factors that it deems relevant or appropriate.
5.2 Determination
of Awards. The Committee shall determine the terms and conditions of all Awards granted to Participants in accordance with
its authority under Section 3.2 hereof. An Award may consist of one type of right or benefit hereunder or of two or more such
rights or benefits granted in tandem or in the alternative. To the extent deemed appropriate by the Committee, an Award shall be
evidenced by an Award Agreement as described in Section 15.1 hereof.
Section 6. Stock Options
6.1 Grants
of Stock Options. A Stock Option may be granted to any Eligible Person selected by the Committee. Subject to the provisions
of Section 6.7 hereof and Section 422 of the Code, each Stock Option shall be designated, in the discretion of the Committee,
as an Incentive Stock Option or as a Nonqualified Stock Option.
6.2 Exercise
Price. The exercise price per share of a Stock Option shall not be less than 100 percent of the Fair Market Value of a share
of Common Stock on the Date of Grant, subject to adjustments as provided for under Section 4.2, provided that the Committee
may in its discretion specify for any Stock Option an exercise price per share that is higher than the Fair Market Value on the
Date of Grant and may establish an exercise price that is below Fair Market Value on the Date of Grant for Stock Options granted
to Participants who are not residents of the U.S if permitted by applicable law and any applicable rules of the principal established
stock exchange or national market system on which the Common Stock is traded.
6.3 Vesting
of Stock Options. The Committee shall in its discretion prescribe the time or times at which, or the conditions upon which,
a Stock Option or portion thereof shall become vested and/or exercisable. The requirements for vesting and exercisability of a
Stock Option may be based on the continued Service of the Participant for a specified time period (or periods) and/or on the attainment
of a specified performance goal (or goals) established by the Committee in its discretion. The Committee may, in its discretion,
accelerate the vesting or exercisability of any Stock Option at any time. The Committee in its sole discretion may allow a Participant
to exercise unvested Nonqualified Stock Options, in which case the shares of Common Stock then issued shall be Restricted Stock
having analogous vesting restrictions to the unvested Nonqualified Stock Options.
6.4 Term
of Stock Options. The Committee shall in its discretion prescribe in an Award Agreement the period during which a vested Stock
Option may be exercised, provided that the maximum term of a Stock Option shall be ten (10) years from the Date of Grant.
A Stock Option may be earlier terminated as specified by the Committee and set forth in an Award Agreement upon or following the
termination of a Participant’s Service, including by reason of voluntary resignation, death, Disability, termination for
Cause or any other reason. Except as otherwise provided in this Section 6 or in an Award Agreement as such agreement may be
amended from time to time upon authorization of the Committee, no Stock Option may be exercised at any time during the term thereof
unless the Participant is then in Service. Notwithstanding the foregoing, unless an Award Agreement provides otherwise:
(a) If
a Participant’s Service terminates by reason of his or her death, any Stock Option held by such Participant may, to the extent
then exercisable, be exercised by such Participant’s estate or any person who acquires the right to exercise such Stock Option
by bequest or inheritance at any time in accordance with its terms for up to one year after the date of such Participant’s
death (but in no event after the earlier of the expiration of the term of such Stock Option or such time as the Stock Option is
otherwise canceled or terminated in accordance with its terms). Upon expiration of such one-year period, no portion of the Stock
Option held by such Participant shall be exercisable and the Stock Option shall be deemed to be canceled, forfeited and of no further
force or effect.
(b) If
a Participant’s Service terminates by reason of his or her Disability, any Stock Option held by such Participant may, to
the extent then exercisable, be exercised by the Participant or his or her personal representative at any time in accordance with
its terms for up to one year after the date of such Participant’s termination of Service (but in no event after the earlier
of the expiration of the term of such Stock Option or such time as the Stock Option is otherwise canceled or terminated in accordance
with its terms). Upon expiration of such one-year period, no portion of the Stock Option held by such Participant shall be exercisable
and the Stock Option shall be deemed to be canceled, forfeited and of no further force or effect.
(c) If
a Participant’s Service terminates for any reason other than death, Disability or Cause, any Stock Option held by such Participant
may, to the extent then exercisable, be exercised by the Participant up until ninety (90) days following such termination of Service
(but in no event after the earlier of the expiration of the term of such Stock Option or such time as the Stock Option is otherwise
canceled or terminated in accordance with its terms). Upon expiration of such 90-day period, no portion of the Stock Option held
by such Participant shall be exercisable and the Stock Option shall be deemed to be canceled, forfeited and of no further force
or effect.
(d) If
a Participant’s Service terminates for Cause, any Stock Option held by such Participant, whether vested or unvested, shall
be deemed forfeited and canceled on the date of such termination of Service.
(e) To
the extent that a Stock Option of a Participant whose Service terminates is not exercisable, such Stock Option shall be deemed
forfeited and canceled on the ninetieth (90th) day after such termination of Service or at such earlier time as the Committee may
determine.
6.5 Stock
Option Exercise. Subject to such terms and conditions as shall be specified in an Award Agreement, a Stock Option may be exercised
in whole or in part at any time during the term thereof by notice in the form required by the Company, and payment of the aggregate
exercise price by certified or bank check, or such other means as the Committee may accept. As set forth in an Award Agreement
or otherwise determined by the Committee, in its sole discretion, at or after grant, payment in full or in part of the exercise
price of an Option may be made: (i) in the form of shares of Common Stock that have been held by the Participant for such
period as the Committee may deem appropriate for accounting purposes or otherwise, valued at the Fair Market Value of such shares
on the date of exercise; (ii) by surrendering to the Company shares of Common Stock otherwise receivable on exercise of the
Option; (iii) by a cashless exercise program implemented by the Committee in connection with the Plan; and/or (iv) by
such other method as may be approved by the Committee and set forth in an Award Agreement. Subject to any governing rules or regulations,
as soon as practicable after receipt of written notification of exercise and full payment of the exercise price and satisfaction
of any applicable tax withholding pursuant to Section 16.5, the Company shall deliver to the Participant evidence of book
entry shares of Common Stock, or upon the Participant’s request, Common Stock certificates in an appropriate amount based
upon the number of shares of Common Stock purchased under the Option. Unless otherwise determined by the Committee, all payments
under all of the methods indicated above shall be paid in United States dollars or shares of Common Stock, as applicable.
6.6 Additional
Rules for Incentive Stock Options.
(a) Eligibility.
An Incentive Stock Option may only be granted to an Eligible Person who is considered an employee under Treasury Regulation §1.421-7(h)
of the Company or any Subsidiary.
(b) Annual
Limits. No Incentive Stock Option shall be granted to an Eligible Person as a result of which the aggregate Fair Market Value
(determined as of the Date of Grant) of the stock with respect to which Incentive Stock Options are exercisable for the first time
in any calendar year under the Plan and any other stock option plans of the Company or any Subsidiary would exceed $100,000, determined
in accordance with Section 422(d) of the Code. This limitation shall be applied by taking Incentive Stock Options into account
in the order in which granted.
(c) Ten
Percent Stockholders. If a Stock Option granted under the Plan is intended to be an Incentive Stock Option, and if the Participant,
at the time of grant, owns stock possessing ten percent or more of the total combined voting power of all classes of Common Stock
of the Company or any Subsidiary, then (A) the Stock Option exercise price per share shall in no event be less than 110 percent
of the Fair Market Value of the Common Stock on the date of such grant and (B) such Stock Option shall not be exercisable
after the expiration of five (5) years following the date such Stock Option is granted.
(d) Disqualifying
Dispositions. If shares of Common Stock acquired by exercise of an Incentive Stock Option are disposed of within two (2) years
following the Date of Grant or one (1) year following the transfer of such shares to the Participant upon exercise, the Participant
shall, promptly following such disposition, notify the Company in writing of the date and terms of such disposition and provide
such other information regarding the disposition as the Company may reasonably require.
Section 7. Stock Appreciation Rights
7.1 Grant
of Stock Appreciation Rights. A Stock Appreciation Right may be granted to any Eligible Person selected by the Committee. Stock
Appreciation Rights may be granted on a basis that allows for the exercise of the right by the Participant or that provides for
the automatic payment of the right upon a specified date or event.
7.2 Base
Price. The base price of a Stock Appreciation Right shall be determined by the Committee in its sole discretion; provided,
however, that the base price for any grant of a Stock Appreciation Right shall not be less than 100 percent of the Fair Market
Value of a share of Common Stock on the Date of Grant, subject to adjustments as provided for under Section 4.2.
7.3 Vesting
Stock Appreciation Rights. The Committee shall in its discretion prescribe the time or times at which, or the conditions upon
which, a Stock Appreciation Right or portion thereof shall become vested and/or exercisable. The requirements for vesting and exercisability
of a Stock Appreciation Right may be based on the continued Service of a Participant for a specified time period (or periods) or
on the attainment of a specified performance goal (or goals) established by the Committee in its discretion. The Committee may,
in its discretion, accelerate the vesting or exercisability of any Stock Appreciation Right at any time.
7.4 Term
of Stock Appreciation Rights. The Committee shall in its discretion prescribe in an Award Agreement the period during which
a vested Stock Appreciation Right may be exercised, provided that the maximum term of a Stock Appreciation Right shall be ten (10) years
from the Date of Grant. A Stock Appreciation Right may be earlier terminated as specified by the Committee and set forth in an
Award Agreement upon or following the termination of a Participant’s Service, including by reason of voluntary resignation,
death, Disability, termination for Cause or any other reason. Except as otherwise provided in this Section 7 or in an Award
Agreement as such agreement may be amended from time to time upon authorization of the Committee, no Stock Appreciation Right may
be exercised at any time during the term thereof unless the Participant is then in the Service of the Company or one of its Subsidiaries.
7.5 Payment
of Stock Appreciation Rights. Subject to such terms and conditions as shall be specified in an Award Agreement, a vested Stock
Appreciation Right may be exercised in whole or in part at any time during the term thereof by notice in the form required by the
Company and payment of any exercise price. Upon the exercise of a Stock Appreciation Right and payment of any applicable exercise
price, a Participant shall be entitled to receive an amount determined by multiplying: (i) the excess of the Fair Market Value
of a share of Common Stock on the date of exercise of the Stock Appreciation Right over the base price of such Stock Appreciation
Right, by (ii) the number of shares as to which such Stock Appreciation Right is exercised. Payment of the amount determined
under the immediately preceding sentence may be made, as approved by the Committee and set forth in the Award Agreement, in shares
of Common Stock valued at their Fair Market Value on the date of exercise, in cash, or in a combination of shares of Common Stock
and cash, subject to applicable tax withholding requirements set forth in Section 16.5. If Stock Appreciation Rights are settled
in shares of Common Stock, then as soon as practicable following the date of settlement the Company shall deliver to the Participant
evidence of book entry shares of Common Stock, or upon the Participant’s request, Common Stock certificates in an appropriate
amount.
Section 8. Restricted Stock Awards
8.1 Grant
of Restricted Stock Awards. A Restricted Stock Award may be granted to any Eligible Person selected by the Committee. The Committee
may require the payment by the Participant of a specified purchase price in connection with any Restricted Stock Award. The Committee
may provide in an Award Agreement for the payment of dividends and distributions to the Participant at such times as paid to stockholders
generally or at the times of vesting or other payment of the Restricted Stock Award. If any dividends or distributions are paid
in stock while a Restricted Stock Award is subject to restrictions under Section 8.3 of the Plan, the dividends or other distributions
shares shall be subject to the same restrictions on transferability as the shares of Common Stock to which they were paid unless
otherwise set forth in the Award Agreement. The Committee may also subject the grant of any Restricted Stock Award to the execution
of a voting agreement with the Company or with any Affiliate of the Company.
8.2 Vesting
Requirements. The restrictions imposed on shares of Common Stock granted under a Restricted Stock Award shall lapse in accordance
with the vesting requirements specified by the Committee in the Award Agreement. Upon vesting of a Restricted Stock Award, such
Award shall be subject to the tax withholding requirement set forth in Section 14.5. The requirements for vesting of a Restricted
Stock Award may be based on the continued Service of the Participant for a specified time period (or periods) or on the attainment
of a specified performance goal (or goals) established by the Committee in its discretion. The Committee may, in its discretion,
accelerate the vesting of a Restricted Stock Award at any time. If the vesting requirements of a Restricted Stock Award shall not
be satisfied, the Award shall be forfeited and the shares of Common Stock subject to the Award shall be returned to the Company.
In the event that the Participant paid any purchase price with respect to such forfeited shares, unless otherwise provided by the
Committee in an Award Agreement, the Company will refund to the Participant the lesser of (i) such purchase price and (ii) the
Fair Market Value of such shares on the date of forfeiture.
8.3 Restrictions.
Shares granted under any Restricted Stock Award may not be transferred, assigned or subject to any encumbrance, pledge, or charge
until all applicable restrictions are removed or have expired, unless otherwise allowed by the Committee. The Committee may require
in an Award Agreement that certificates representing the shares granted under a Restricted Stock Award bear a legend making appropriate
reference to the restrictions imposed, and that certificates representing the shares granted or sold under a Restricted Stock Award
will remain in the physical custody of an escrow holder until all restrictions are removed or have expired.
8.4 Rights
as Stockholder. Subject to the foregoing provisions of this Section 8 and the applicable Award Agreement, the Participant
to whom a Restricted Stock Award is made shall have all rights of a stockholder with respect to the shares granted to the Participant
under the Restricted Stock Award, including the right to vote the shares and receive all dividends and other distributions paid
or made with respect thereto, unless the Committee determines otherwise at the time the Restricted Stock Award is granted.
8.5 Section 83(b)
Election. If a Participant makes an election pursuant to Section 83(b) of the Code with respect to a Restricted Stock Award,
the Participant shall file, within 30 days following the Date of Grant, a copy of such election with the Company (directed to the
Secretary thereof) and with the Internal Revenue Service, in accordance with the regulations under Section 83 of the Code. The
Committee may provide in an Award Agreement that the Restricted Stock Award is conditioned upon the Participant’s making
or refraining from making an election with respect to the Award under Section 83(b) of the Code.
Section 9. Stock Unit Awards
9.1 Grant
of Stock Unit Awards. A Stock Unit Award may be granted to any Eligible Person selected by the Committee. The value of each
Stock Unit under a Stock Unit Award is equal to the Fair Market Value of the Common Stock on the applicable date or time period
of determination, as specified by the Committee. A Stock Unit Award shall be subject to such restrictions and conditions as the
Committee shall determine. A Stock Unit Award may be granted together with a dividend equivalent right with respect to the shares
of Common Stock subject to the Award, which may be accumulated and may be deemed reinvested in additional Stock Units, as determined
by the Committee in its discretion. If any dividend equivalents are paid while a Stock Unit Award is subject to restrictions under
Section 9 of the Plan, the dividend equivalents shall be subject to the same restrictions on transferability as the Stock Units
to which they were paid, unless otherwise set forth in the Award Agreement.
9.2 Vesting
of Stock Unit Awards. On the Date of Grant, the Committee shall, in its discretion, determine any vesting requirements with
respect to a Stock Unit Award, which shall be set forth in the Award Agreement. The requirements for vesting of a Stock Unit Award
may be based on the continued Service of the Participant for a specified time period (or periods) or on the attainment of a specified
performance goal (or goals) established by the Committee in its discretion. The Committee may, in its discretion, accelerate the
vesting of a Stock Unit Award at any time. A Stock Unit Award may also be granted on a fully vested basis, with a deferred payment
date as may be determined by the Committee or elected by the Participant in accordance with rules established by the Committee.
9.3 Payment
of Stock Unit Awards. A Stock Unit Award shall become payable to a Participant at the time or times determined by the Committee
and set forth in the Award Agreement, which may be upon or following the vesting of the Award. Payment of a Stock Unit Award may
be made, at the discretion of the Committee, in cash or in shares of Common Stock, or in a combination thereof as described in
the Award Agreement, subject to applicable tax withholding requirements set forth in Section 16.5. Any cash payment of a Stock
Unit Award shall be made based upon the Fair Market Value of the Common Stock, determined on such date or over such time period
as determined by the Committee. Notwithstanding the foregoing, unless specified otherwise in the Award Agreement, any Stock Unit,
whether settled in Common Stock or cash, shall be paid no later than two and one-half months after the later of the calendar year
or fiscal year in which the Stock Units vest. If Stock Unit Awards are settled in shares of Common Stock, then as soon as practicable
following the date of settlement the Company shall deliver to the Participant evidence of book entry shares of Common Stock, or
upon the Participant’s request, Common Stock certificates in an appropriate amount.
Section 10. Performance Shares
10.1 Grant
of Performance Shares. Performance Shares may be granted to any Eligible Person selected by the Committee. A Performance Share
Award shall be subject to such restrictions and condition as the Committee shall specify. A Performance Share Award may be granted
with a dividend equivalent right with respect to the shares of Common Stock subject to the Award, which may be accumulated and
may be deemed reinvested in additional Stock Units, as determined by the Committee in its discretion.
10.2 Value
of Performance Shares. Each Performance Share shall have an initial value equal to the Fair Market Value of a Share on the
Grant Date. The Committee shall set performance goals in its discretion that, depending on the extent to which they are met over
a specified time period, shall determine the number of Performance Shares that shall be paid to a Participant.
10.3 Earning
of Performance Shares. After the applicable time period has ended, the number of Performance Shares earned by the Participant
over such time period shall be determined as a function of the extent to which the applicable corresponding performance goals have
been achieved. This determination shall be made solely by the Committee. The Committee may, in its discretion, waive any performance
or vesting conditions relating to a Performance Share Award.
10.4 Form
and Timing of Payment of Performance Shares. The Committee shall pay at the close of the applicable Performance Period, or
as soon as practicable thereafter, any earned Performance Shares in the form of cash or in shares of Common Stock or in a combination
thereof, as specified in a Participant’s Award Agreement, subject to applicable tax withholding requirements set forth in
Section 16.5. Notwithstanding the foregoing, unless specified otherwise in the Award Agreement, all Performance Shares shall
be paid no later than two and one-half months following the later of the calendar year or fiscal year in which such Performance
Shares vest. Any shares of Common Stock paid to a Participant under this Section 10.4 may be subject to any restrictions deemed
appropriate by the Committee. If Performance Shares are settled in shares of Common Stock, then as soon as practicable following
the date of settlement the Company shall deliver to the Participant evidence of book entry shares of Common Stock, or upon the
Participant’s request, Common Stock certificates in an appropriate amount.
Section 11. Performance Units
11.1 Grant
of Performance Units. Performance Units may be granted to any Eligible Person selected by the Committee. A Performance Unit
Award shall be subject to such restrictions and conditions as the Committee shall specify in a Participant’s Award Agreement.
11.2 Value
of Performance Units. Each Performance Unit shall have an initial notional value equal to a dollar amount determined by the
Committee, in its sole discretion. The Committee shall set performance goals in its discretion that, depending on the extent to
which they are met over a specified time period, will determine the number of Performance Units that shall be settled and paid
to the Participant.
11.3 Earning
of Performance Units. After the applicable time period has ended, the number of Performance Units earned by the Participant,
and the amount payable in cash, in shares or in a combination thereof, over such time period shall be determined as a function
of the extent to which the applicable corresponding performance goals have been achieved. This determination shall be made solely
by the Committee. The Committee may, in its discretion, waive any performance or vesting conditions relating to a Performance Unit
Award.
11.4 Form
and Timing of Payment of Performance Units. The Committee shall pay at the close of the applicable Performance Period, or as
soon as practicable thereafter, any earned Performance Units in the form of cash or in shares of Common Stock or in a combination
thereof, as specified in a Participant’s Award Agreement, subject to applicable tax withholding requirements set forth in
Section 16.5. Notwithstanding the foregoing, unless specified otherwise in the Award Agreement, all Performance Units shall
be paid no later than two and one-half months following the later of the calendar year or fiscal year in which such Performance
Units vest. Any shares of Common Stock paid to a Participant under this Section 11.4 may be subject to any restrictions deemed
appropriate by the Committee. If Performance Units are settled in shares of Common Stock, then as soon as practicable following
the date of settlement the Company shall deliver to the Participant evidence of book entry shares of Common Stock, or upon the
Participant’s request, Common Stock certificates in an appropriate amount.
Section 12. Incentive Bonus Awards
12.1 Incentive
Bonus Awards. The Committee, at its discretion, may grant Incentive Bonus Awards to such Participants as it may designate from
time to time. The terms of a Participant’s Incentive Bonus Award shall be set forth in the Participant’s Award Agreement.
Each Award Agreement shall specify such general terms and conditions as the Committee shall determine.
12.2 Incentive
Bonus Award Performance Criteria. The determination of Incentive Bonus Awards for a given year or years may be based upon the
attainment of specified levels of Company or Subsidiary performance as measured by pre-established, objective performance criteria
determined at the discretion of the Committee. The Committee shall (i) select those Participants who shall be eligible to receive
an Incentive Bonus Award, (ii) determine the performance period, (iii) determine target levels of performance, and (iv) determine
the level of Incentive Bonus Award to be paid to each selected Participant upon the achievement of each performance level. The
Committee generally shall make the foregoing determinations prior to the commencement of services to which an Incentive Bonus Award
relates, to the extent applicable, and while the outcome of the performance goals and targets is uncertain.
12.3 Payment
of Incentive Bonus Awards.
(a) Incentive
Bonus Awards shall be paid in cash or Common Stock, as set forth in a Participant’s Award Agreement. Payments shall be made
following a determination by the Committee that the performance targets were attained and shall be made within two and one-half
months after the later of the end of the fiscal or calendar year in which the Incentive Award is no longer subject to a substantial
risk of forfeiture.
(b) The
amount of an Incentive Bonus Award to be paid upon the attainment of each targeted level of performance shall equal a percentage
of a Participant’s base salary for the fiscal year, a fixed dollar amount, or such other formula, as determined by the Committee.
Section 13. Other Cash-Based Awards
and Other Stock-Based Awards
13.1 Other
Cash-Based and Stock-Based Awards. The Committee may grant other types of equity-based or equity-related Awards not otherwise
described by the terms of this Plan (including the grant or offer for sale of unrestricted Shares) in such amounts and subject
to such terms and conditions, as the Committee shall determine. Such Awards may involve the transfer of actual shares of Common
Stock to a Participant, or payment in cash or otherwise of amounts based on the value of shares of Common Stock. In addition, the
Committee, at any time and from time to time, may grant Cash-Based Awards to a Participant in such amounts and upon such terms
as the Committee shall determine, in its sole discretion.
13.2 Value
of Cash-Based Awards and Other Stock-Based Awards. Each Other Stock-Based Award shall be expressed in terms of shares of Common
Stock or units based on shares of Common Stock, as determined by the Committee, in its sole discretion. Each Other Cash-Based Award
shall specify a payment amount or payment range as determined by the Committee, in its sole discretion. If the Committee exercises
its discretion to establish performance goals, the value of Other Cash-Based Awards that shall be paid to the Participant will
depend on the extent to which such performance goals are met.
13.3 Payment
of Cash-Based Awards and Other Stock-Based Awards. Payment, if any, with respect to Other Cash-Based Awards and Other Stock-Based
Award shall be made in accordance with the terms of the Award, in cash or Shares as the Committee determines.
14. Change
in Control
14.1 Effect
of Change in Control.
(a) The
Committee may, at the time of the grant of an Award and as set forth in an Award Agreement, provide for the effect of a “Change
in Control” on an Award. Such provisions may include any one or more of the following: (i) the acceleration or extension
of time periods for purposes of exercising, vesting in, or realizing gain from any Award, (ii) the elimination or modification
of performance or other conditions related to the payment or other rights under an Award, (iii) provision for the cash settlement
of an Award for an equivalent cash value, as determined by the Committee, or (iv) such other modification or adjustment to
an Award as the Committee deems appropriate to maintain and protect the rights and interests of Participants upon or following
a Change in Control. To the extent necessary for compliance with Section 409A of the Code, an Award Agreement shall provide
that an Award subject to the requirements of Section 409A that would otherwise become payable upon a Change in Control shall
only become payable to the extent that the requirements for a “change in control” for purposes of Section 409A
have been satisfied.
(b) Notwithstanding
anything to the contrary set forth in the Plan, unless otherwise provided by an Award Agreement, upon or in anticipation of any
Change in Control, the Committee may, in its sole and absolute discretion and without the need for the consent of any Participant,
take one or more of the following actions contingent upon the occurrence of that Change in Control: (i) cause any or all outstanding
Stock Options and Stock Appreciation Rights held by Participants affected by the Change in Control to become vested and immediately
exercisable, in whole or in part; (ii) cause any or all outstanding Restricted Stock, Stock Units, Performance Shares, Performance
Units, Incentive Bonus Award and any other Award held by Participants affected by the Change in Control to become non-forfeitable,
in whole or in part; (iii) cancel any Stock Option or Stock Appreciation Right in exchange for a substitute option in a manner
consistent with the requirements of Treasury Regulation §1.424-1(a) or §1.409A-1(b)(5)(v)(D), as applicable (notwithstanding
the fact that the original Stock Option may never have been intended to satisfy the requirements for treatment as an Incentive
Stock Option); (iv) cancel any Restricted Stock, Stock Units, Performance Shares or Performance Units held by a Participant
in exchange for restricted stock or performance shares of or stock or performance units in respect of the capital stock of any
successor corporation; (v) redeem any Restricted Stock held by a Participant affected by the Change in Control for cash and/or
other substitute consideration with a value equal to the Fair Market Value of an unrestricted share of Common Stock on the date
of the Change in Control; (vi) terminate any Award in exchange for an amount of cash and/or property equal to the amount,
if any, that would have been attained upon the exercise of such Award or realization of the Participant’s rights as of the
date of the occurrence of the Change in Control (the “Change in Control Consideration”); provided, however that if
the Change in Control Consideration with respect to any Option or Stock Appreciation Right does not exceed the exercise price of
such Option or Stock Appreciation Right, the Committee may cancel the Option or Stock Appreciation Right without payment of any
consideration therefor. Any such Change in Control Consideration may be subject to any escrow, indemnification and similar obligations,
contingencies and encumbrances applicable in connection with the Change in Control to holders of Common Stock. Without limitation
of the foregoing, if as of the date of the occurrence of the Change in Control the Committee determines that no amount would have
been attained upon the realization of the Participant’s rights, then such Award may be terminated by the Company without
payment. The Committee may cause the Change in Control Consideration to be subject to vesting conditions (whether or not the same
as the vesting conditions applicable to the Award prior to the Change in Control) and/or make such other modifications, adjustments
or amendments to outstanding Awards or this Plan as the Committee deems necessary or appropriate.
(c) The
Committee may require a Participant to (i) represent and warrant as to the unencumbered title to the Participant’s Awards,
(ii) bear such Participant’s pro rata share of any post-closing indemnity obligations, and be subject to the same or similar
post-closing purchase price adjustments, escrow terms, offset rights, holdback terms and similar conditions as the other holders
of Common Stock, and (iii) execute and deliver such documents and instruments as the Committee may reasonably require for the Participant
to be bound by such obligations. The Committee will endeavor to take action under this Section 14 in a manner that does not cause
a violation of Section 409A of the Code with respect to an Award.
15. General
Provisions
15.1 Award
Agreement. To the extent deemed necessary by the Committee, an Award under the Plan shall be evidenced by an Award Agreement
in a written or electronic form approved by the Committee setting forth the number of shares of Common Stock or units subject to
the Award, the exercise price, base price, or purchase price of the Award, the time or times at which an Award will become vested,
exercisable or payable and the term of the Award. The Award Agreement may also set forth the effect on an Award of termination
of Service under certain circumstances. The Award Agreement shall be subject to and incorporate, by reference or otherwise, all
of the applicable terms and conditions of the Plan, and may also set forth other terms and conditions applicable to the Award as
determined by the Committee consistent with the limitations of the Plan. Award Agreements evidencing Incentive Stock Options shall
contain such terms and conditions as may be necessary to meet the applicable provisions of Section 422 of the Code. The grant of
an Award under the Plan shall not confer any rights upon the Participant holding such Award other than such terms, and subject
to such conditions, as are specified in the Plan as being applicable to such type of Award (or to all Awards) or as are expressly
set forth in the Award Agreement.
15.2 Forfeiture
Events/Representations. The Committee may specify in an Award Agreement at the time of the Award that the Participant’s
rights, payments and benefits with respect to an Award shall be subject to reduction, cancellation, forfeiture or recoupment upon
the occurrence of certain specified events, in addition to any otherwise applicable vesting or performance conditions of an Award.
Such events shall include, but shall not be limited to, termination of Service for Cause, violation of material Company policies,
breach of noncompetition, confidentiality or other restrictive covenants that may apply to the Participant, or other conduct by
the Participant that is detrimental to the business or reputation of the Company. The Committee may also specify in an Award Agreement
that the Participant’s rights, payments and benefits with respect to an Award shall be conditioned upon the Participant making
a representation regarding compliance with noncompetition, confidentiality or other restrictive covenants that may apply to the
Participant and providing that the Participant’s rights, payments and benefits with respect to an Award shall be subject
to reduction, cancellation, forfeiture or recoupment on account of a breach of such representation. Notwithstanding the foregoing,
the confidentiality restrictions set forth in an Award Agreement shall not, and shall not be interpreted to, impair a Participant
from exercising any legally protected whistleblower rights (including under Rule 21 of the Exchange Act). In addition and without
limitation of the foregoing, any amounts paid hereunder shall be subject to recoupment in accordance with The Dodd–Frank
Wall Street Reform and Consumer Protection Act and any implementing regulations thereunder, any “clawback” policy adopted
by the Company or as is otherwise required by applicable law or stock exchange listing condition.
15.3 No
Assignment or Transfer; Beneficiaries.
(a) Awards
under the Plan shall not be assignable or transferable by the Participant, except by will or by the laws of descent and distribution,
and shall not be subject in any manner to assignment, alienation, pledge, encumbrance or charge. Notwithstanding the foregoing,
the Committee may provide in an Award Agreement that the Participant shall have the right to designate a beneficiary or beneficiaries
who shall be entitled to any rights, payments or other benefits specified under an Award following the Participant’s death.
During the lifetime of a Participant, an Award shall be exercised only by such Participant or such Participant’s guardian
or legal representative. In the event of a Participant’s death, an Award may, to the extent permitted by the Award Agreement,
be exercised by the Participant’s beneficiary as designated by the Participant in the manner prescribed by the Committee
or, in the absence of an authorized beneficiary designation, by the legatee of such Award under the Participant’s will or
by the Participant’s estate in accordance with the Participant’s will or the laws of descent and distribution, in each
case in the same manner and to the same extent that such Award was exercisable by the Participant on the date of the Participant’s
death.
(b) Limited
Transferability Rights. Notwithstanding anything else in this Section 15.3 to the contrary, the Committee may in its discretion
provide in an Award Agreement that an Award in the form of a Nonqualified Stock Option, share-settled Stock Appreciation Right,
Restricted Stock, Performance Share or share-settled Other Stock-Based Award may be transferred, on such terms and conditions as
the Committee deems appropriate, either (i) by instrument to the Participant’s “Immediate Family” (as defined
below), (ii) by instrument to an inter vivos or testamentary trust (or other entity) in which the Award is to be passed to
the Participant’s designated beneficiaries, or (iii) by gift to charitable institutions. Any transferee of the Participant’s
rights shall succeed and be subject to all of the terms of the applicable Award Agreement and the Plan. “Immediate Family”
means any child, stepchild, grandchild, parent, stepparent, grandparent, spouse, former spouse, sibling, niece, nephew, mother-in-law,
father-in-law, son-in-law, daughter-in-law, brother-in-law, or sister-in-law, and shall include adoptive relationships.
15.4 Rights
as Stockholder. A Participant shall have no rights as a holder of shares of Common Stock with respect to any unissued securities
covered by an Award until the date the Participant becomes the holder of record of such securities. Except as provided in Section 4.2
hereof, no adjustment or other provision shall be made for dividends or other stockholder rights, except to the extent that the
Award Agreement provides for dividend payments or dividend equivalent rights.
15.5 Employment
or Service. Nothing in the Plan, in the grant of any Award or in any Award Agreement shall confer upon any Eligible Person
or Participant any right to continue in Service, or interfere in any way with the right of the Company or any of its Subsidiaries
to terminate the employment or other service relationship of an Eligible Person or Participant for any reason at any time.
15.6 Fractional
Shares. In the case of any fractional share or unit resulting from the grant, vesting, payment or crediting of dividends or
dividend equivalents under an Award, the Committee shall have the discretionary authority to (i) disregard such fractional
share or unit, (ii) round such fractional share or unit to the nearest lower or higher whole share or unit, or (iii) convert
such fractional share or unit into a right to receive a cash payment.
15.7 Other
Compensation and Benefit Plans. The amount of any compensation deemed to be received by a Participant pursuant to an Award
shall not constitute includable compensation for purposes of determining the amount of benefits to which a Participant is entitled
under any other compensation or benefit plan or program of the Company or any Subsidiary, including, without limitation, under
any bonus, pension, profit-sharing, life insurance, salary continuation or severance benefits plan, except to the extent specifically
provided by the terms of any such plan.
15.8 Plan
Binding on Transferees. The Plan shall be binding upon the Company, its transferees and assigns, and the Participant, the
Participant’s executor, administrator and permitted transferees and beneficiaries. In addition, all obligations of the Company
under this Plan with respect to Awards granted hereunder shall be binding on any successor to the Company, whether the existence
of such successor is the result of a direct or indirect purchase, merger, consolidation, or otherwise, of all or substantially
all of the business and/or assets of the Company.
15.9 Foreign
Jurisdictions. The Committee may adopt, amend and terminate such arrangements and grant such Awards, not inconsistent with
the intent of the Plan, as it may deem necessary or desirable to comply with any tax, securities, regulatory or other laws of other
jurisdictions with respect to Awards that may be subject to such laws. The terms and conditions of such Awards may vary from the
terms and conditions that would otherwise be required by the Plan solely to the extent the Committee deems necessary for such purpose.
Moreover, the Board may approve such supplements to or amendments, restatements or alternative versions of the Plan, not inconsistent
with the intent of the Plan, as it may consider necessary or appropriate for such purposes, without thereby affecting the terms
of the Plan as in effect for any other purpose.
15.10 Substitute
Awards in Corporate Transactions. Nothing contained in the Plan shall be construed to limit the right of the Committee to grant
Awards under the Plan in connection with the acquisition, whether by purchase, merger, consolidation or other corporate transaction,
of the business or assets of any corporation or other entity. Without limiting the foregoing, the Committee may grant Awards under
the Plan to an employee or director of another corporation who becomes an Eligible Person by reason of any such corporate transaction
in substitution for Awards previously granted by such corporation or entity to such person. The terms and conditions of the substitute
Awards may vary from the terms and conditions that would otherwise be required by the Plan solely to the extent the Committee deems
necessary for such purpose. Any shares of Common Stock subject to these substitute Awards shall not be counted against any of the
maximum share limitations set forth in the Plan.
Section 16. Legal Compliance
16.1 Securities
Laws. No shares of Common Stock will be issued or transferred pursuant to an Award unless and until all then applicable requirements
imposed by Federal and state securities and other laws, rules and regulations and by any regulatory agencies having jurisdiction,
and by any exchanges upon which the shares of Common Stock may be listed, have been fully met. As a condition precedent to the
issuance of shares pursuant to the grant or exercise of an Award, the Company may require the Participant to take any reasonable
action to meet such requirements. The Committee may impose such conditions on any shares of Common Stock issuable under the Plan
as it may deem advisable, including, without limitation, restrictions under the Securities Act, as amended, under the requirements
of any exchange upon which such shares of the same class are then listed, and under any blue sky or other securities laws applicable
to such shares. The Committee may also require the Participant to represent and warrant at the time of issuance or transfer that
the shares of Common Stock are being acquired only for investment purposes and without any current intention to sell or distribute
such shares. All Common Stock issued pursuant to the terms of this Plan shall constitute “restricted securities,” as
that term is defined in Rule 144 promulgated pursuant to the Securities Act, and may not be transferred except in compliance herewith
and with the registration requirements of the Securities Act or an exemption therefrom. Certificates representing Common Stock
acquired pursuant to an Award may bear such legend as the Company may consider appropriate under the circumstances.
16.2 Incentive
Arrangement. The Plan is designed to provide an ongoing, pecuniary incentive for Participants to produce their best efforts
to increase the value of the Company. The Plan is not intended to provide retirement income or to defer the receipt of payments
hereunder to the termination of a Participant’s employment or beyond. The Plan is thus intended not to be a pension or welfare
benefit plan that is subject to Employee Retirement Income Security Act of 1974 (“ERISA”), and shall be construed accordingly.
All interpretations and determinations hereunder shall be made on a basis consistent with the Plan’s status as not an employee
benefit plan subject to ERISA.
16.3 Unfunded
Plan. The adoption of the Plan and any reservation of shares of Common Stock or cash amounts by the Company to discharge its
obligations hereunder shall not be deemed to create a trust or other funded arrangement. Except upon the issuance of Common Stock
pursuant to an Award, any rights of a Participant under the Plan shall be those of a general unsecured creditor of the Company,
and neither a Participant nor the Participant’s permitted transferees or estate shall have any other interest in any assets
of the Company by virtue of the Plan. Notwithstanding the foregoing, the Company shall have the right to implement or set aside
funds in a grantor trust, subject to the claims of the Company’s creditors or otherwise, to discharge its obligations under
the Plan.
16.4 Section 409A
Compliance. To the extent applicable, it is intended that the Plan and all Awards hereunder comply with the requirements of
Section 409A of the Code or an exemption thereto, and the Plan and all Award Agreements shall be interpreted and applied by
the Committee in a manner consistent with this intent in order to avoid the imposition of any additional tax under Section 409A
of the Code. Notwithstanding anything in the Plan to the contrary, in the event that any provision of the Plan or an Award Agreement
is determined by the Committee, in its sole discretion, to not comply with the requirements of Section 409A of the Code or
an exemption thereto, the Committee shall, in its sole discretion, have the authority to take such actions and to make such interpretations
or changes to the Plan or an Award Agreement as the Committee deems necessary, regardless of whether such actions, interpretations
or changes shall adversely affect a Participant, subject to the limitations, if any, of applicable law. If an Award is subject
to Section 409A of the Code, any payment made to a Participant who is a “specified employee” of the Company or any
Subsidiary shall not be made before the date that is six months after the Participant’s “separation from service”
to the extent required to avoid the adverse consequences of Section 409A of the Code. For purposes of this Section 16.4, the terms
“separation from service” and “specified employee” shall have the meanings set forth in Section 409A of
the Code. In no event whatsoever shall the Company be liable for any additional tax, interest or penalties that may be imposed
on any Participant by Section 409A of the Code or any damages for failing to comply with Section 409A of the Code.
16.5 Tax
Withholding.
(a) The
Company shall have the power and the right to deduct or withhold, or require a participant to remit to the Company, the minimum
statutory amount to satisfy federal, state, and local taxes, domestic or foreign, required by law or regulation to be withheld
with respect to any taxable event arising as a result of this Plan, but in no event shall such deduction or withholding or remittance
exceed the minimum statutory withholding requirements unless permitted by the Company and such additional withholding amount will
not cause adverse accounting consequences and is permitted under Applicable Law. Notwithstanding the foregoing, if a minimum statutory
amount of withholding does not apply under the laws of any foreign jurisdiction, the Company may withhold such amount for remittance
to the applicable taxing authority of such jurisdiction as the Company determines in its discretion, uniformly applied, to be appropriate.
(b) A
Participant may, in order to fulfill the withholding obligation, tender previously-acquired shares of Common Stock or have shares
of stock withheld from the exercise, provided that the shares have an aggregate Fair Market Value sufficient to satisfy in whole
or in part the applicable withholding taxes. The broker-assisted exercise procedure described in Section 6.5 may also be utilized
to satisfy the withholding requirements related to the exercise of a Stock Option.
(c) Notwithstanding
the foregoing, a Participant may not use shares of Common Stock to satisfy the withholding requirements to the extent that (i)
there is a substantial likelihood that the use of such form of payment or the timing of such form of payment would subject the
Participant to a substantial risk of liability under Section 16 of the Exchange Act; (ii) such withholding would constitute a violation
of the provisions of any law or regulation (including the Sarbanes-Oxley Act of 2002); or (iii) such withholding would cause adverse
accounting consequences for the Company.
16.6 No
Guarantee of Tax Consequences. Neither the Company, the Board, the Committee nor any other Person make any commitment or guarantee
that any federal, state, local or foreign tax treatment will apply or be available to any Participant or any other person hereunder.
16.7 Severability.
If any provision of the Plan or any Award Agreement shall be determined to be illegal or unenforceable by any court of law in any
jurisdiction, the remaining provisions hereof and thereof shall be severable and enforceable in accordance with their terms, and
all provisions shall remain enforceable in any other jurisdiction.
16.8 Stock
Certificates; Book Entry Form. Notwithstanding any provision of the Plan to the contrary,
unless otherwise determined by the Committee or required by any applicable law, rule or regulation, any obligation set forth in
the Plan pertaining to the delivery or issuance of stock certificates evidencing shares of Common Stock may be satisfied by having
issuance and/or ownership of such shares recorded on the books and records of the Company (or,
as applicable, its transfer agent or stock plan administrator).
16.9 Governing
Law. The Plan and all rights hereunder shall be subject to and interpreted in accordance with the laws of the State of Delaware,
without reference to the principles of conflicts of laws, and to applicable Federal securities laws.
Section 17. Effective Date, Amendment
and Termination
17.1 Effective
Date. The effective date of the Plan shall be the date on which the Plan is approved by the Board; provided, however, that
Awards granted under the Plan subsequent to the approval of the Plan by the Board shall be valid only if the Plan is approved by
the requisite percentage of the holders of the Common Stock of the Company within one year of the date on which such Board approval
occurs. If such stockholder approval is not obtained within one year after the date of the Board’s approval of the Plan,
then all Awards previously granted under the Plan shall terminate and cease to be outstanding, and no further Awards shall be granted
under the Plan.
17.2 Amendment;
Termination. The Board may suspend or terminate the Plan (or any portion thereof) at any time and may amend the Plan at any
time and from time to time in such respects as the Board may deem advisable or in the best interests of the Company or any Subsidiary;
provided, however, that (a) no such amendment, suspension or termination shall materially impair any rights or materially increase
any obligations under any Award theretofore made under the Plan without the consent of the Participant affected thereby (or, after
the Participant’s death, the person having the right to exercise the Award), (b) to the extent necessary and desirable to
comply with any applicable law, regulation, or stock exchange rule, the Company shall obtain stockholder approval of any Plan amendment
in such a manner and to such a degree as required, and (c) stockholder approval is required for any amendment to the Plan that
(i) increases the number of shares of Common Stock available for issuance under the Plan, or (ii) changes the persons or class
of persons eligible to receive Awards. For purposes of the foregoing, any action of the Board or the Committee that alters or affects
the tax treatment of any Award shall not be considered to materially impair any rights of any Participant. The Plan will continue
in effect until terminated in accordance with this Section 17.2; provided, however, that no Award will be granted hereunder
on or after the 10th anniversary of the date of the adoption of the Plan by the Board (the “Expiration Date”);
but provided further, that Awards granted prior to such Expiration Date may extend beyond that date.
. . .
ADOPTION AND APPROVAL OF PLAN
Date Plan initially adopted by Board: October 7,
2020
Date Plan approved by Shareholders: December
15, 2020
Effective Date of Plan: October 7,
2020
INFORMATION NOT REQUIRED IN PROSPECTUS
|
|
Item 20.
|
Indemnification of Directors and Officers
|
Under the Business Corporations Act
(Alberta), or ABCA, except in respect of an action by or on behalf of the company to procure a judgment in its favor, we may
indemnify a director or officer of the company, a former director or officer of the company or a person who acts or acted at the
company’s request as a director or officer of a body corporate of which the company is or was a shareholder or creditor,
and the director’s or officer’s heirs and legal representatives, against all costs, charges and expenses, including
an amount paid to settle an action or satisfy a judgment, reasonably incurred by the director or officer in respect of any civil,
criminal or administrative action or proceeding to which the director or officer is made a party by reason of being or having been
a director or officer of that company or body corporate, if:
(a) the director or officer
acted honestly and in good faith with a view to the best interests of the company, and
(b) in the case of a criminal
or administrative action or proceeding that is enforced by a monetary penalty, the director or officer had reasonable grounds for
believing that the director’s or officer’s conduct was lawful.
Notwithstanding anything in the ABCA, a person referred to above
is entitled to indemnity from the company in respect of all costs, charges and expenses reasonably incurred by the person in connection
with the defense of any civil, criminal or administrative action or proceeding to which the person is made a party by reason of
being or having been a director or officer of the corporation or body corporate, if the person seeking indemnity
|
|
(i)
|
was substantially successful on the merits in the person’s defense of the action or
proceeding,
|
|
|
(ii)
|
fulfils the conditions set out in paragraphs (a) and (b) above, and
|
|
|
(iii)
|
is fairly and reasonably entitled to indemnity.
|
The ABCA provides that we may advance funds to a person in order
to defray the costs, charges and expenses of a proceeding referred to above, but if the person does not meet the conditions of
paragraphs (i), (ii) and (iii) above he or she shall repay the funds advanced.
In accordance with the ABCA, we may purchase and maintain insurance
for the benefit of any director or officer of the company, a former director or officer of the company or a person who acts or
acted at the company’s request as a director or officer of a body corporate of which the company is or was a shareholder
or creditor, and the director’s or officer’s heirs and legal representatives against any liability incurred by the
person:
(a) in the person’s
capacity as a director or officer of the company, except when the liability relates to the person’s failure to act honestly
and in good faith with a view to the best interests of the company; or
(b) in the person’s
capacity as a director or officer of another body corporate if the person acts or acted in that capacity at the company’s
request, except when the liability relates to the person’s failure to act honestly and in good faith with a view to the best
interests of the body corporate.
We currently maintain insurance policies in the amount of $3.75
million per covered person.
In addition to the foregoing provisions of the ABCA, our by-laws
require us to indemnify each of our directors, officers, former directors and officers and persons who act or acted at our request
as a director or officer, or in a similar capacity, of a body corporate of which the company is or was a shareholder or creditor,
and his heirs and legal representatives, against all costs, charges and expenses, including an amount paid to settle an action
or satisfy a judgment, reasonably incurred by him in respect of any civil, criminal or administrative action or proceeding to which
the individual is made a party by reason of being or having been a director of officer of us or such body corporate, provided that
he:
|
|
·
|
acted honestly and in good faith with a view to our best interests; and
|
|
|
·
|
in the case of a criminal or administrative action or proceeding that is enforced by a monetary
penalty, the individual had reasonable grounds for believing that his or her conduct was lawful.
|
The proposed certificate of incorporation of Zomedica Corp.
includes a provision that eliminates the personal liability of the directors for monetary damages to the fullest extent permitted
by Delaware law. Under current Delaware law, a director's liability to a corporation and its stockholders may not be limited for:
|
|
•
|
a breach of the director's duty of loyalty to the corporation or its stockholders;
|
|
|
•
|
acts or omissions not in good faith or which involve intentional misconduct or a knowing violation
of law; an unlawful payment of a dividend or an unlawful stock purchase or redemption; and
|
|
|
•
|
any transaction from which the director derived an improper personal benefit.
|
The proposed bylaws of Zomedica Corp. generally provide for mandatory
indemnification and advancement of expenses of our directors and officers to the fullest extent permitted under Delaware law.
|
|
Item 21.
|
Exhibits and Financial Statement Schedules
|
The following exhibits and financial statement schedules are filed
as part of this registration statement. Exhibits and financial statement schedules that have previously been filed are incorporated
by reference, as noted. Exhibits and financial statement schedules filed herewith appear beginning at page F-1.
|
4.3
|
|
Placement Agent Warrant issued in connection with February 2020 offering (incorporated by reference to Exhibit 4.2 to the Company’s Current Report on Form 8-K filed with the Commission on February 13, 2020)
|
|
4.4
|
|
Placement Agent Warrant issued in connection with April 2020 offering (incorporated by reference to Exhibit 4.2 to the Company’s Current Report on Form 8-K filed with the Commission on April 8, 2020)
|
|
4.5
|
|
Form of Series C Warrant (incorporated by reference to Exhibit 4.5 to the Company’s Registration Statement on Form S-1 (File No.333-238322) filed with the Commission on May 15, 2020)
|
|
4.6
|
|
Form of Series D Warrant (incorporated by reference to Exhibit 4.1 to the Company’s Current Report on Form 8-K filed with the Commission on July 6, 2020)
|
|
5.1*
|
|
Opinion of Lowenstein Sandler LLP
|
|
8.1*
|
|
Opinion of Norton Rose Fulbright Canada LLP as to certain Canadian federal income tax matters
|
|
8.2*
|
|
Opinion of Lowenstein Sandler LLP as to certain United States federal income tax matters
|
|
10.1+
|
|
Executive Employment Agreement, dated June 16, 2020, among Zomedica Pharmaceuticals, Inc., Zomedica Pharmaceuticals Corp. and Robert Cohen (incorporated by reference to Exhibit 10.1 to the Company’s Current Report on Form 8-K filed with the Commission on June 17, 2020)
|
|
10.2+
|
|
Executive Employment Agreement between Zomedica Pharmaceuticals Corp. and Stephanie Morley (incorporated by reference to Exhibit 10.30 to the Company’s Current Report on Form 8-K filed on September 17, 2019)
|
|
10.3+
|
|
Amended and Restated Stock Option Plan (incorporated by reference to Exhibit 99.1 to the Company's Current Report on Form 8-K filed with the Commission on June 17, 2020)
|
|
10.4+
|
|
Executive Employment Agreement between ZoMedica Pharmaceuticals Inc. and Bruk Herbst (incorporated by reference to Exhibit 10.16 to the Company's Registration Statement on Form S-1 filed with the Commission on November 20, 2017 (File No. 333-217409))
|
|
10.5
|
|
Loan Agreement, dated October 17, 2017, by and between Zomedica Pharmaceuticals Corp. and Equidebt LLC (incorporated by reference to Exhibit 10.20 to the Company's Registration Statement on Form S-1 filed with the Commission on November 20, 2017 (File No. 333-217409))
|
|
10.6
|
|
Line of Credit Promissory Note, dated October 17, 2017, from Zomedica Pharmaceuticals Corp. in favor of Equidebt LLC (incorporated by reference to Exhibit 10.21 to the Company's Registration Statement on Form S-1 filed with the Commission on November 20, 2017 (File No. 333-217409))
|
|
10.7#
|
|
Development, Commercialization and Exclusive Distribution Agreement, dated May 10, 2018, by and between Seraph Biosciences, Inc. and Zomedica Pharmaceuticals Corp. (incorporated by reference to Exhibit 10.24 to the Company’s Quarterly Report on Form 10-Q filed with the Commission on August 19, 2018 (File No. 001-38298))
|
|
10.8#
|
|
Development and Supply Agreement, dated November 26, 2018, with Qorvo Biotechnologies,
LLC (incorporated by reference to Exhibit 10.27 to the Company’s Annual Report on Form 10-K filed with the Commission on
February 26, 2019 (File No. 001-38298))
|
|
10.9^*
|
|
Amendment, dated August 14, 2020, to Development and Supply Agreement with Qorvo Biotechnologies, LLC
|
|
10.10*
|
|
Letter Agreement, dated September 3, 2020, with Qorvo Biotechnologies, LLC
|
|
10.11
|
|
Sales Agreement, dated December 7, 2018, by and between Zomedica Pharmaceuticals Corp. and Cantor Fitzgerald & Co. (incorporated by reference to Exhibit 1.1 to the Company’s Registrations Statement on Form S-3 filed with the Commission on December 20, 2018 (File No. 333-228926))
|
|
10.12
|
|
Form of Preferred Share Subscription Agreement for May 2019 Offering (incorporated by reference to Exhibit 10.29 to the Company’s Quarterly Report on Form 10-Q filed with the Commission on May 10, 2019 (File No. 001-38298))
|
|
10.13^
|
|
Amended and Restated Exclusive License and Supply Agreement, dated January 17, 2020, by and between Celsee, Inc. and Zomedica Pharmaceuticals Corp. (incorporated by reference to Exhibit 10.31 to the Company’s Annual Report on Form 10-K filed with the Commission on February 26, 2020 (File No. 001-38298))
|
|
10.14
|
|
Lease Agreement for 100 Phoenix Drive, Ann Arbor, 48108 (incorporated by reference to Exhibit 10.32 to the Company’s Annual Report on Form 10-K filed with the Commission on February 26, 2020 (File No. 001-38298))
|
|
10.15+
|
|
Consulting Agreement between Zomedica Pharmaceuticals Corp. and Johnny D. Powers (incorporated by reference to Exhibit 10.33 to the Company’s Annual Report on Form 10-K filed with the Commission on February 26, 2020 (File No. 001-38298))
|
|
10.16
|
|
Promissory Note, dated April 15, 2020, between Bank of America, NA and Zomedica Pharmaceuticals Corp. (incorporated by reference to Exhibit 10.1 to the Company’s Current Report on Form 8-K filed with the Commission on April 24, 2020 (File No. 001-38298))
|
|
10.16
|
|
Securities Purchase Agreement, dated February 12, 2020, among the Company and the investors named therein (incorporated by reference to Exhibit 10.1 to the Company’s Current Report on Form 8-K filed with the Commission on February 13, 2020)
|
|
10.17
|
|
Placement Agency Agreement, dated April 7, 2020, by and between the Company and H.C. Wainwright & Co., LLC (incorporated by reference to Exhibit 10.1 to the Company’s Current Report on Form 8-K filed with the Commission on April 8, 2020)
|
|
10.18
|
|
Securities Purchase Agreement, dated April 7, 2020, among the Company and the investors named therein (incorporated by reference to Exhibit 10.2 to the Company’s Current Report on Form 8-K filed with the Commission on April 8, 2020)
|
|
10.19
|
|
Form of Securities Purchase Agreement to be entered into among the Company and certain investors (incorporated by reference to Exhibit 10.23 to the Company’s Registration Statement on Form S-1 (File No.333-238322) filed with the Commission on May 15, 2020)
|
|
10.20
|
|
Form of Placement Agency Agreement by and between the Company and H.C. Wainwright & Co., LLC (incorporated by reference to Exhibit 1.1 to Amendment No. 2 to the Company’s Registration Statement on Form S-1 (File No.333-238322) filed with the Commission on May 22, 2020)
|
|
10.21
|
|
Placement Agency Agreement, dated July 1, 2020, by and between the Company and H.C. Wainwright & Co., LLC (incorporated by reference to Exhibit 10.1 to the Company’s Current Report on Form 8-K filed with the Commission on July 6, 2020)
|
|
10.22
|
|
Form of Securities Purchase Agreement, dated July 1, 2020, among the Company and the
purchasers party thereto (incorporated by reference to Exhibit 10.2 to the Company’s Current Report on Form 8-K filed with
the Commission on July 6, 2020)
|
|
10.23
|
|
Form of 2020 Equity Incentive Plan (Incorporated by reference from Exhibit G to the Proxy Circular/Prospectus)
|
|
21.1
|
|
List of Subsidiaries (incorporated by reference to Exhibit 21.1 to the Company's Registration Statement on Form S-1 filed with the Commission on April 21, 2017 (File No. 333-217409))
|
|
23.1**
|
|
Consent of MNP LLP
|
|
23.2*
|
|
Consents of Lowenstein Sandler LLP (included in its opinions filed as Exhibit 5.1 and Exhibit 8.2)
|
|
23.3*
|
|
Consent of Norton Rose Fulbright Canada LLP (included in its opinion filed as Exhibit 8.1)
|
|
24.1*
|
|
Power of Attorney
|
*
Previously filed.
**
Filed herewith.
# The registrant has received
confidential treatment for certain portions of this exhibit.
+ Indicates
management contract or compensatory plan.
^ Certain
identified information has been excluded from this exhibit because it is both (i) not material and (ii) would be competitively
harmful if publicly disclosed.
Item 22. Undertakings
(a) The undersigned registrant hereby undertakes:
(1) to file, during any period
in which offers or sales are being made, a post-effective amendment to this registration statement:
(i) to include
any prospectus required by section 10(a)(3) of the Securities Act of 1933;
(ii) to reflect
in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective
amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the
registration statement.
(iii) to
include any material information with respect to the plan of distribution not previously disclosed in the registration statement
or any material change to such information in the registration statement;
(2) that, for the purpose of
determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration
statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the
initial bona fide offering thereof.
(3) to remove from registration
by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering.
(4) that, for the purpose of
determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of the securities,
the undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration
statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or
sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser
and will be considered to offer or sell such securities to such purchaser:
(i) any preliminary
prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424;
(ii) any
free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to
by the undersigned registrant;
(iii) the
portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant
or its securities provided by or on behalf of the undersigned registrant; and
(iv) any
other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
(b) The undersigned registrant hereby undertakes
that:
(1) for purposes of determining
any liability under the Securities Act of 1933, the information omitted from the form of prospectus filed as part of this registration
statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b)
(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time
it was declared effective.
(2) for the purpose of determining
any liability under the Securities Act of 1933, each post-effective amendment that contains a form of prospectus shall be deemed
to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time
shall be deemed to be the initial bona fide offering thereof.
Insofar as indemnification for liabilities
arising under the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the registrant pursuant
to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission
such indemnification is against public policy as expressed in the Securities Act of 1933 and is, therefore, unenforceable. In the
event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred
or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding)
is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant
will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate
jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act of 1933
and will be governed by the final adjudication of such issue.
(c) The undersigned registrant hereby undertakes
to respond to requests for information that is incorporated by reference into the prospectus pursuant to Items 4, 10(b), 11,
or 13 of this Form, within one business day of receipt of such request, and to send the incorporated documents by first class mail
or other equally prompt means. This includes information contained in documents filed subsequent to the effective date of the registration
statement through the date of responding to the request.
(d) The undersigned registrant hereby undertakes
to supply by means of a post-effective amendment all information concerning a transaction, and the company being acquired involved
therein, that was not the subject of and included in the registration statement when it became effective.
SIGNATURES
Pursuant to the requirements of the Securities
Act of 1933, as amended, the registrant has duly caused this amendment to registration statement to be signed on its behalf by
the undersigned, thereunto duly authorized, in the City of Ann Arbor, State of Michigan, on October 22, 2020.
|
|
ZOMEDICA CORP.
|
|
|
|
|
|
|
|
By:
|
/s/ Robert Cohen
|
|
|
|
|
Robert Cohen
|
|
|
|
|
Interim Chief Executive Officer and Director
|
Pursuant to the requirements of the Securities Act of 1933, as amended, this registration
statement has been signed by the following persons in the capacities indicated on the 9th day of October, 2020.
|
Signature
|
|
Title
|
|
|
|
|
|
/s/ Robert Cohen
|
|
Interim Chief Executive Officer and Director
|
|
Robert Cohen
|
|
(Principal Executive Officer)
|
|
|
|
|
|
/s/ Ann Marie Cotter
|
|
Chief Financial Officer and Secretary
|
|
Ann Marie Cotter
|
|
(Principal Financial and Accounting Officer)
|
|
|
|
|
|
*
|
|
Director
|
|
Jeffrey Rowe
|
|
|
|
|
|
|
|
*
|
|
Director
|
|
Chris Macleod
|
|
|
|
|
|
|
|
*
|
|
Director
|
|
Johnny D. Powers
|
|
|
|
|
|
|
|
*
|
|
Director
|
|
Rodney Williams
|
|
|
|
|
|
|
|
*
|
|
Director
|
|
Christopher Wolfenberg
|
|
|
|
*By:
|
/s/ Robert Cohen
|
|
|
|
Robert Cohen
|
|
|
|
Attorney-in-fact
|
|
Zomedica (AMEX:ZOM)
Historical Stock Chart
From Apr 2024 to May 2024
Zomedica (AMEX:ZOM)
Historical Stock Chart
From May 2023 to May 2024