UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the Month of October 2023
Commission File Number: 001-38097
ARGENX SE
(Translation of registrant’s name into English)
Laarderhoogtweg 25
1101 EB Amsterdam, the Netherlands
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual
reports under cover of Form 20-F or Form 40-F.
Form 20-F x
Form 40-F ¨
Indicate
by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ¨
Indicate
by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ¨
argenx SE
On October 31, 2023, argenx SE (the “Company) issued
a press release and investor presentation, copies of which are attached hereto as Exhibits 99.1 and 99.2, respectively, and are incorporated
by reference herein.
The information contained in this Current Report on Form 6-K,
including the exhibits hereto, is incorporated by reference into the Company’s Registration Statements on Forms F-3 (File No. 333-258251
and S-8 (File Nos. 333-225375, 333-258253 and 333-274721).
EXHIBITS
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934,
the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| |
ARGENX SE |
| |
|
|
| Date: October 31, 2023 |
By: |
/s/ Hemamalini (Malini) Moorthy |
| |
|
Hemamalini (Malini) Moorthy |
| |
|
General Counsel |
Exhibit 99.1

argenx Reports Third
Quarter 2023 Financial Results and Provides Business Update
- $329
million in third quarter global net product sales
- On
track to submit VYVGART® Hytrulo sBLA for CIDP by year-end 2023
- Results
from the ADVANCE-IV study published in The Lancet
- Management
to host conference call today at 1:30 pm CET (8:30 am ET)
October 31,
2023
Amsterdam, the
Netherlands – argenx SE (Euronext & Nasdaq: ARGX), a global immunology company committed to improving the lives of
people suffering from severe autoimmune diseases, today announced its third quarter 2023 financial results and provided a business update
and outlook for the remainder of the year.
“We continue
to prioritize patient impact with VYVGART and VYVGART Hytrulo, broadening our two gMG products into earlier treatment lines and new geographies.
VYVGART has now been used in thousands of patients over multiple treatment years, and its unique clinical profile has built patient trust
and physician confidence in the brand,” said Tim Van Hauwermeiren, Chief Executive Officer of argenx. “There is a significant
opportunity before us to transform autoimmunity across multiple indications with VYVGART. Based on the successful ADHERE trial, we are
ready to file the sBLA by the end of 2023 to bring our first-in-class FcRn blocker to CIDP patients as quickly as possible. We are also
on track with two near-term pivotal readouts and an ambitious plan forward over the coming years as we continue to execute and drive
innovation within our FcRn portfolio and across immunology more broadly.”
THIRD QUARTER 2023
AND RECENT BUSINESS UPDATE
VYVGART Expansion
VYVGART®
is a first-in-class antibody fragment targeting the neonatal Fc receptor (FcRn) and is now approved globally in seven countries
or regions (U.S., Japan, EU, UK, Israel, China, Canada) for generalized myasthenia gravis (gMG). VYVGART Hytrulo (subcutaneous (SC)
injection) was approved in the U.S. in June 2023. argenx is planning for multi-dimensional expansion to reach more patients with
gMG and other severe autoimmune diseases through additional global regulatory approvals.
| · | Generated
global net product revenues (inclusive of both VYVGART and VYVGART Hytrulo) of $329 million
in the third quarter of 2023 |
| · | Health
Canada approved VYVGART on September 21, 2023, marking the seventh global approval for
gMG |

| · | European
Commission (EC) approval of SC efgartigimod for gMG expected in fourth quarter of 2023 following
positive recommendation from Committee for Medicinal Products for Human Use (CHMP) of the
European Medicines Agency (EMA) |
| · | Japan
approval decision regarding SC efgartigimod for gMG expected by first quarter of 2024 |
| · | Japan
marketing authorization application (MAA) filed for VYVGART for primary immune thrombocytopenia
(ITP); approval decision expected in first quarter of 2024 |
| · | U.S.
supplemental Biologics License Application (sBLA) for VYVGART Hytrulo in chronic inflammatory
demyelinating polyneuropathy (CIDP) on track to be filed by end of 2023 |
| · | China
approval decision regarding SC efgartigimod for gMG expected by end of 2024 through partnership
with Zai Lab |
Efgartigimod Research
and Development
argenx is solidifying
its leadership in FcRn blockade and demonstrating the broad potential of efgartigimod by advancing its clinical development programs
of IgG-mediated autoimmune diseases. By 2025, efgartigimod is expected to be approved, in regulatory review or in development in 15 severe
autoimmune diseases
| · | Topline
data from ADVANCE-SC (ITP) expected in fourth quarter of 2023; results from ADVANCE-IV study
were published in The Lancet in September 2023 |
| · | Topline
data from ADDRESS (pemphigus) and GO/NO GO decision from BALLAD (bullous pemphigoid) both
expected around year-end 2023 |
| · | GO/NO
GO decision expected from ALKIVIA (myositis) in second half of 2024 |
| · | Topline
data from ALPHA (post-COVID postural orthostatic tachycardia syndrome (PC-POTS)) expected
in first quarter of 2024 and RHO (Sjogren’s syndrome) in first half of 2024 |
Pipeline Progress
argenx is advancing
a robust portfolio of innovative clinical programs, including empasiprubart (C2 inhibitor) and ARGX-119 (muscle-specific kinase (MuSK)
agonist). Both programs have the potential to be first-in-class opportunities for multiple severe indications.
| · | Topline
data from Phase 2 ARDA study of empasiprubart (ARGX-117) in multifocal motor neuropathy (MMN)
expected in 2024 |
| · | Phase
1 study of ARGX-119 ongoing in healthy volunteers; subsequent Phase 1b trial planned to assess
early signal detection in patients with congenital myasthenic syndrome (CMS) and amyotrophic
lateral sclerosis (ALS) |
Immunology Innovation
Program
argenx continues to
invest in its discovery engine, the Immunology Innovation Program, to foster a robust innovation ecosystem and drive early-stage pipeline
growth. argenx expects to nominate one new pipeline candidate in 2023.

THIRD QUARTER 2023
FINANCIAL RESULTS
argenx SE
UNAUDITED CONDENSED
CONSOLIDATED INTERIM STATEMENTS OF PROFIT OR LOSS
| | |
Three
Months Ended
September 30,
| | |
Nine
Months Ended
September 30,
| |
| (in thousands of $ except
for shares and EPS) | |
2023 | | |
2022 | | |
2023 | | |
2022 | |
| Product net sales | |
$ | 329,097 | | |
$ | 131,329 | | |
$ | 816,432 | | |
$ | 227,325 | |
| Collaboration revenue | |
| 692 | | |
| 6,652 | | |
| 3,047 | | |
| 9,262 | |
| Other operating income | |
| 10,050 | | |
| 8,508 | | |
| 31,275 | | |
| 26,565 | |
| Total operating
income | |
$ | 339,839 | | |
$ | 146,489 | | |
$ | 850,754 | | |
$ | 263,152 | |
| | |
| | | |
| | | |
| | | |
| | |
| Cost of sales | |
$ | (35,999 | ) | |
$ | (10,264 | ) | |
$ | (78,358 | ) | |
$ | (16,646 | ) |
| Research and development expenses | |
| (191,755 | ) | |
| (236,681 | ) | |
| (553,119 | ) | |
| (515,568 | ) |
| Selling, general and administrative expenses | |
| (191,930 | ) | |
| (108,181 | ) | |
| (503,079 | ) | |
| (336,845 | ) |
| Loss from investment in joint venture | |
| (743 | ) | |
| - | | |
| (2,623 | ) | |
| - | |
| Total operating expenses | |
| (420,427 | ) | |
| (355,126 | ) | |
| (1,137,179 | ) | |
| (869,059 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Operating loss | |
$ | (80,588 | ) | |
$ | (208,637 | ) | |
$ | (286,425 | ) | |
$ | (605,907 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Financial income | |
$ | 30,049 | | |
$ | 8,007 | | |
$ | 67,078 | | |
$ | 13,740 | |
| Financial expense | |
| (231 | ) | |
| (785 | ) | |
| (626 | ) | |
| (2,916 | ) |
| Exchange gains/(losses) | |
| (32,509 | ) | |
| (39,609 | ) | |
| (23,345 | ) | |
| (92,991 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Loss for the period
before taxes | |
$ | (83,279 | ) | |
$ | (241,024 | ) | |
$ | (243,318 | ) | |
$ | (688,074 | ) |
| Income tax (expense)/benefit | |
$ | 10,637 | | |
$ | 5,982 | | |
$ | 47,437 | | |
$ | 17,096 | |
| Loss for the period | |
$ | (72,642 | ) | |
$ | (235,042 | ) | |
$ | (195,881 | ) | |
$ | (670,978 | ) |
| Loss for the year attributable to: | |
| | | |
| | | |
| | | |
| | |
| Owners of the parent | |
$ | (72,642 | ) | |
$ | (235,042 | ) | |
$ | (195,881 | ) | |
$ | (670,978 | ) |
| Weighted average number of shares outstanding | |
| 58,128,233 | | |
| 55,203,655 | | |
| 56,512,254 | | |
| 54,049,119 | |
| Basis and diluted loss per share (in $) | |
| (1.25 | ) | |
| (4.26 | ) | |
| (3.47 | ) | |
| (12.41 | ) |
| Net increase/(decrease) in cash, cash equivalents and current
financial assets compared to year-end 2022 and 2021 | |
| | | |
| | | |
$ | 993,035 | | |
$ | 48,813 | |
| Cash and cash equivalents and current financial assets at
the end of the period | |
| | | |
| | | |
$ | 3,185,583 | | |
$ | 2,385,541 | |

DETAILS OF THE
FINANCIAL RESULTS
Total operating
income for the third quarter and year-to-date in 2023 was $339.8 million and $850.8 million, respectively, compared to $146.5 million
and $263.2 million for the same periods in 2022, and mainly consists of:
| · | Product
net sales of VYVGART for the three months ended and nine months ended September 30,
2023, were $329.1 million and $816.4 million, compared to $131.3 million and $227.3 million
for the same periods in 2022. |
| · | Other
operating income for the third quarter and year-to-date in 2023 was $10.1 million and
$31.3 million, respectively, compared to $8.5 million, and $26.6 million for the same periods
in 2022. The other operating income for the three and nine months ended September 30,
2023, primarily relates to research and development tax incentives and payroll tax rebates.
Other income also includes $0.7 million in royalty revenue from VYVGART sales in China. |
Total operating
expenses for the third quarter and year-to-date in 2023 were $420.4 million and $1,137.2 million, respectively, compared to $335.1
million and $869.1 million for the same periods in 2022, and mainly consists of:
| · | Cost
of sales for the third quarter and year-to-date in 2023 was $36.0 million and $78.4 million,
respectively, compared to $10.3 million and $16.6 million for the same periods in 2022. The
cost of sales was recognized with respect to the sale of VYVGART and VYVGART Hytrulo. |
| · | Research
and development expenses for the third quarter and year-to-date in 2023 were $191.8 million
and $553.1 million, respectively, compared to $236.7 million and $515.6 million for the same
periods in 2022. The research and development expenses mainly relate to external research
and development expenses and personnel expenses incurred in the clinical development of efgartigimod
in various indications and the expansion of other clinical and preclinical pipeline candidates. |
| · | Selling,
general and administrative expenses for the third quarter and year-to-date in 2023 were
$191.9 million and $503.1 million, respectively, compared to $108.2 million and $336.8 million
for the same periods in 2022. The selling, general and administrative expenses mainly relate
to professional and marketing fees linked to the commercialization of VYVGART and VYVGART
Hytrulo in the U.S., EU and Japan, and personnel expenses. |
Financial income
for the third quarter and year-to-date in 2023 was $30.0 million and $67.1 million, respectively, compared to $8.0 million and $13.7
million for the same periods in 2022. The increase in financial income is mainly due to an increase in interest income on current financial
assets and cash and cash equivalents attributable to higher interest rates.
Exchange losses
for the third quarter and year-to-date in 2023 were $32.5 million and $23.3 million respectively, compared to $39.6 million and $93.0
million of exchange losses for the same periods in 2022. Exchange gains/losses are mainly attributable to unrealized exchange rate gains
or losses on the cash, cash equivalents and current financial assets position in Euro.
Income tax
for the third quarter and year-to-date in 2023 was $10.6 million and $47.4 million of tax benefit, respectively, compared to $6.0 million
and $17.1 million of tax benefit for the same periods in 2022. Tax benefit for the nine months ended September 30, 2023, consists
of $23.8 million of income tax expense and $71.3 million of deferred tax income, compared to $15.0 million of income tax expense and
$32.1 million of deferred tax income for the comparable prior period.

Net loss for
the three and nine-month periods ended September 30, 2023, was $72.6 million and $195.9 million, respectively, compared to $235.0
million and $671.0 million over the prior year periods. On a per weighted average share basis, the net loss was $3.47 and $12.41 for
the nine months ended September 30, 2023 and 2022, respectively.
Cash, cash equivalents
and current financial assets totalled $3.2 billion as of September 30, 2023, compared to $2.2 billion as of December 31,
2022. The increase in cash and cash equivalents and current financial assets resulted primarily from the closing of a global offering
of shares, including a U.S. offering, which resulted in the receipt of $1.2 billion in net proceeds in July 2023, partially offset
by net cash flows used in operating activities.
EXPECTED 2024 FINANCIAL
CALENDAR
| · | February 29,
2024: FY 2023 financial results and business update |
| · | May 9,
2024: Q1 2024 financial results and business update |
| · | July 25,
2024: Q2 2024 financial results and business update |
| · | October 24,
2024: Q3 2024 financial results and business update |
CONFERENCE CALL
DETAILS
The third quarter
2023 financial results and business update will be discussed during a conference call and webcast presentation today at 1:30 pm CET/8:30
am ET. A webcast of the live call may be accessed on the Investors section of the argenx website at argenx.com/investors. A replay of
the webcast will be available on the argenx website.
Dial-in numbers:
Please dial in
15 minutes prior to the live call.
| Belgium |
32 800 50 201 |
| France |
33 800 943355 |
| Netherlands |
31 20 795 1090 |
| United Kingdom |
44 800 358 0970 |
| United States |
1 888 415 4250 |
| Japan |
81 3 4578 9081 |
| Switzerland |
41 43 210 11 32 |
About argenx
argenx
is a global immunology company committed to improving the lives of people suffering from severe autoimmune diseases. Partnering with
leading academic researchers through its Immunology Innovation Program (IIP), argenx aims to translate immunology breakthroughs into
a world-class portfolio of novel antibody-based medicines. argenx developed and is commercializing the first approved neonatal Fc receptor
(FcRn) blocker in the U.S., Japan, Israel, the EU, the UK, China
and Canada. The Company is evaluating efgartigimod in multiple serious autoimmune diseases and advancing several earlier stage experimental
medicines within its therapeutic franchises. For more information, visit www.argenx.com and follow us on LinkedIn, Twitter, and Instagram.

For further information,
please contact:
Media:
Erin Murphy
emurphy@argenx.com
Investors:
Alexandra Roy (US)
aroy@argenx.com
Lynn Elton (EU)
lelton@argenx.com
Forward-looking Statements
The contents of
this announcement include statements that are, or may be deemed to be, “forward-looking statements.” These forward-looking
statements can be identified by the use of forward-looking terminology, including the terms “believes,” “hope,”
“estimates,” “anticipates,” “expects,” “intends,” “may,” “will,”
or “should” and include statements argenx makes regarding its plans to execute and drive innovation within its FcRn portfolio
and across immunology; its plans for multi-dimensional expansion to reach more patients with gMG and other autoimmune diseases through
additional global regulatory approvals; advancement of, and anticipated clinical development, data readouts and regulatory milestones
and plans, including the (1) expected EC approval of SC efgartigimod for gMG in the fourth quarter of 2023, (2) expected approval
decision regarding SC efgartigimod for gMG in Japan by the first quarter of 2024, (3) expected MAA for VYVGART for primary ITP approval
decision in Japan in the first quarter of 2024, (4) expected filing of the sBLA for VYVGART Hytrulo in CIDP by the end of 2023,
(5) expected approval decision regarding SC efgartigimod for gMG in China by end of 2024 through its partnership with Zai Lab; (6) expected
topline data from ITP in the fourth quarter of 2023, (7) expected topline data from ADDRESS and the GO/NO GO decision from BALLAD
around year-end 2023, (8) expected GO/NO GO decision from ALKIVIA in the second half of 2024, (9) expected topline dtaa from
ALPHA in the first quarter of 2024 and RHO in the first half of 2024, (10) expected topline data from Phase 2 ARDA study of ARGX-117
in MMN in 2024, (11) planned Phase 1b trial to assess early signal detection in patients with CMS and ALS and (12) planned nomination
of a new pipeline candidate in 2023; continued investment in its Immunology Innovation Program to foster a robust innovation ecosystem
and drive early-stage pipeline growth; and 2023 business and financial outlook and related plans, the timeline of future releases of
financial results and business updates. By their nature, forward-looking statements involve risks and uncertainties and readers are cautioned
that any such forward-looking statements are not guarantees of future performance. argenx’s actual results may differ materially
from those predicted by the forward-looking statements as a result of various important factors, including inflation and deflation and
the corresponding fluctuations in interest rate; regional instability and conflicts, such as the conflict between Russia and Ukraine,
argenx’s expectations regarding the inherent uncertainties associated with competitive developments, preclinical and clinical trial
and product development activities and regulatory approval requirements; argenx’s reliance on collaborations with third parties;
estimating the commercial potential of argenx’s product candidates; argenx’s ability to obtain and maintain protection of
intellectual property for its technologies and drugs; argenx’s limited operating history; and argenx’s ability to obtain
additional funding for operations and to complete the development and commercialization of its product candidates. A further list and
description of these risks, uncertainties and other risks can be found in argenx’s U.S. Securities and Exchange Commission (SEC)
filings and reports, including in argenx’s most recent annual report on Form 20-F filed with the SEC as well as subsequent
filings and reports filed by argenx with the SEC. Given these uncertainties, the reader is advised not to place any undue reliance on
such forward-looking statements. These forward-looking statements speak only as of the date of publication of this document. argenx undertakes
no obligation to publicly update or revise the information in this press release, including any forward-looking statements, except as
may be required by law.
Exhibit 99.2

| Third Quarter 2023 Financial Results and Business Update
October 31, 2023 |

| Forward Looking Statements
This presentation has been prepared by argenx se (“argenx” or the “company”) for informational purposes only and not for any other purpose. Nothing contained in this presentation is, or
should be construed as, a recommendation, promise or representation by the presenter or the company or any director, employee, agent, or adviser of the company. This presentation does
not purport to be all-inclusive or to contain all of the information you may desire. Certain information contained in this presentation relates to or is based on studies, publications, surveys and
other data obtained from third-party sources and the company’s own internal estimates and research. While argenx believes these third-party studies, publications, surveys and other data to
be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information
obtained from third-party sources. In addition, no independent source has evaluated the reasonableness or accuracy of argenx’s internal estimates or research and no reliance should be
made on any information or statements made in this presentation relating to or based on such internal estimates and research.
The contents of this presentation include statements that are, or may be deemed to be, “forward-looking statements.” These forward-looking statements can be identified by the use of
forward-looking terminology, including the terms “believes,” “hope,” “estimates,” “anticipates,” “expects,” “intends,” “may,” “will,” or “should” and include statements argenx makes regarding
its launch of VYVGART for generalized myasthenia gravis (gMG) and expansion strategy to reach more patients with VYVGART through additional regulatory approvals; the safety profile and
efficacy of VYVGART, the advancement of ARGX-117 and ARGX-119 to clinical proof-of-concept; the planned SC approval and June 20th PDUFA date; its ability to transform gMG treatment for
patients; its anticipated clinical data readouts, including in chronic inflammatory demyelinating polyneuropathy (CIDP), immune thrombocytopenia (ITP), pemphigus vulgaris (PV), postural
orthostatic tachycardia syndrome (POTS) and multifocal motor neuropathy (MMN); the therapeutic potential and patient treatment experience of its product candidates, its strategy to
expand access to treatments through engagement with physicians, payors, and patient communities; expected filing of the sBLA for VYVGART Hytrulo in CIDP by the end of 2023; pending
regulatory reviews of SC efgartigimod for gMG in China, Canada, Europe, and Japan; expectations about its pipeline progress; its collaborations and their potential benefits; the intended
results of its strategy and its collaboration partners', advancement of, and anticipated clinical development, data readouts and regulatory milestones and plans, including the timing of
planned clinical trials and expected data readouts; the design of future clinical trials and the timing and outcome of regulatory filings and regulatory approvals; and its 2023 business and
financial outlook and related plans. By their nature, forward-looking statements involve risks and uncertainties and readers are cautioned that any such forward-looking statements are not
guarantees of future performance. argenx's actual results may differ materially from those predicted by the forward-looking statements as a result of various important factors, including the
effects of the COVID-19 pandemic, inflation and deflation and the corresponding fluctuations in interest rates; regional instability and conflicts, such as the conflict between Russia and
Ukraine, argenx's expectations regarding the inherent uncertainties associated with competitive developments, preclinical and clinical trial and product development activities and regulatory
approval requirements; argenx's reliance on collaborations with third parties; estimating the commercial potential of argenx's product candidates; argenx's ability to obtain and maintain
protection of intellectual property for its technologies and drugs; argenx's limited operating history; and argenx's ability to obtain additional funding for operations and to complete the
development and commercialization of its product candidates. A further list and description of these risks, uncertainties and other risks can be found in argenx's U.S. Securities and Exchange
Commission (SEC) filings and reports, including in argenx's most recent annual report on Form 20-F filed with the SEC as well as subsequent filings and reports filed by argenx with the SEC.
Given these uncertainties, the reader is advised not to place any undue reliance on such forward-looking statements. These forward-looking statements speak only as of the date of
publication of this document. argenx undertakes no obligation to publicly update or revise the information in this press release, including any forward-looking statements, except as may be
required by law.
This presentation contains trademarks, trade names and service marks of other companies, which are the property of their respective owners.
2 |
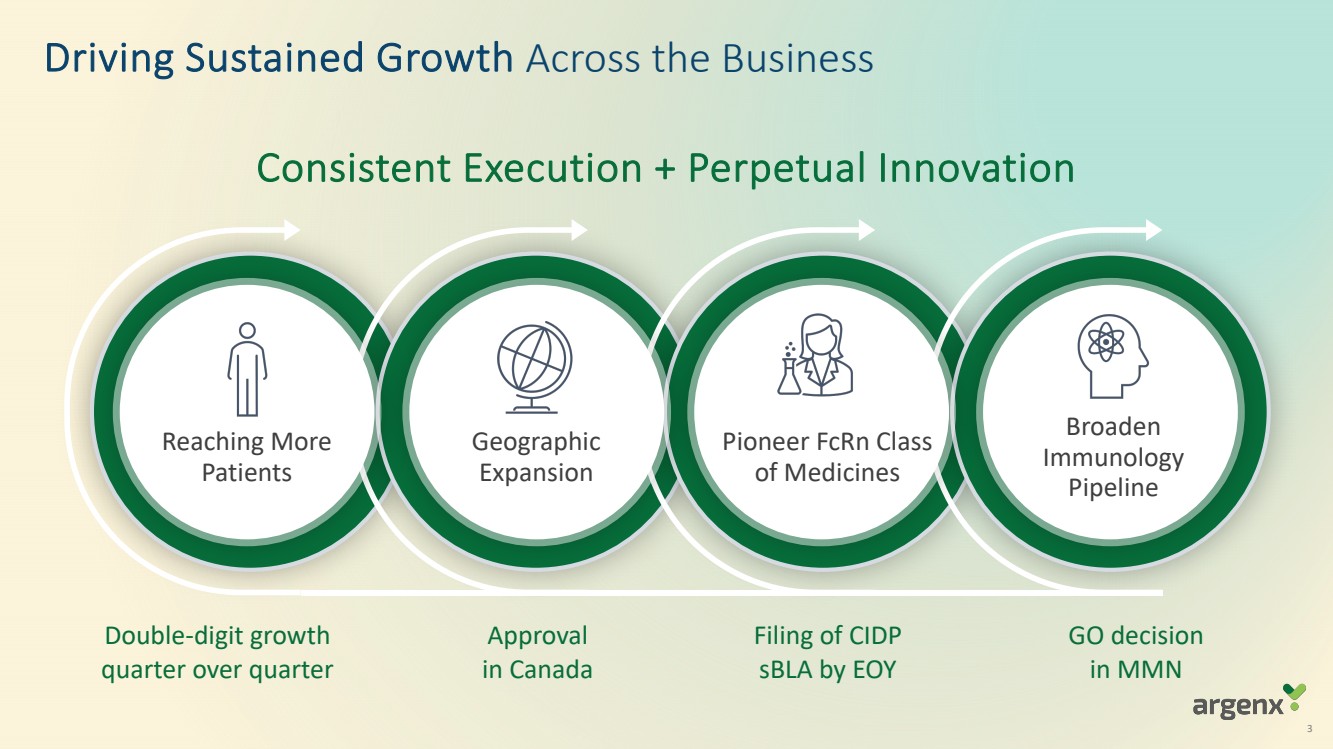
| Driving Sustained Growth Across the Business
Consistent Execution + Perpetual Innovation
Reaching More
Patients
Geographic
Expansion
Pioneer FcRn Class
of Medicines
Broaden
Immunology
Pipeline
Double-digit growth
quarter over quarter
Approval
in Canada
Filing of CIDP
sBLA by EOY
GO decision
in MMN
3 |

| What FcRn Leadership Looks Like Today
4
20 presentations demonstrating neuromuscular leadership
Consistent
Efficacy
Favorable
Safety
Deep FcRn
Expertise
MG-ADL, QMG,
QoL, MSE
TEAEs mild to
moderate
Unique target
modulation
>1,000
Patient Years
of safety data across indications
Real-world experience in
~6,000 patients |

| 5 q2w = every other week
Adult Chronic or Persistent
ITP patients
2 weeks screening
Mean platelet count <30x109/L
Stable concomitant ITP
therapy allowed
Stratification: splenectomy,
concomitant ITP therapy
0
Visit
Fixed weekly dosing Weekly or q2w dosing based on platelet count thresholds
Week 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24
Patients randomized 2:1 to receive VYVGART Hytrulo or placebo (24 weeks)
Fixed weekly or q2w dosing based on week 15
Primary endpoint: Sustained platelet count (≥50×109/L) in ≥4/6 visits between weeks 19 and 24
Stringent endpoint in line with regulatory feedback, addressing platelet count variability
Secondary and exploratory endpoints center around extent of disease control to illustrate real-world viability
Topline data expected 4Q 2023
ITP ADVANCE-SC Trial
Primary Endpoint |
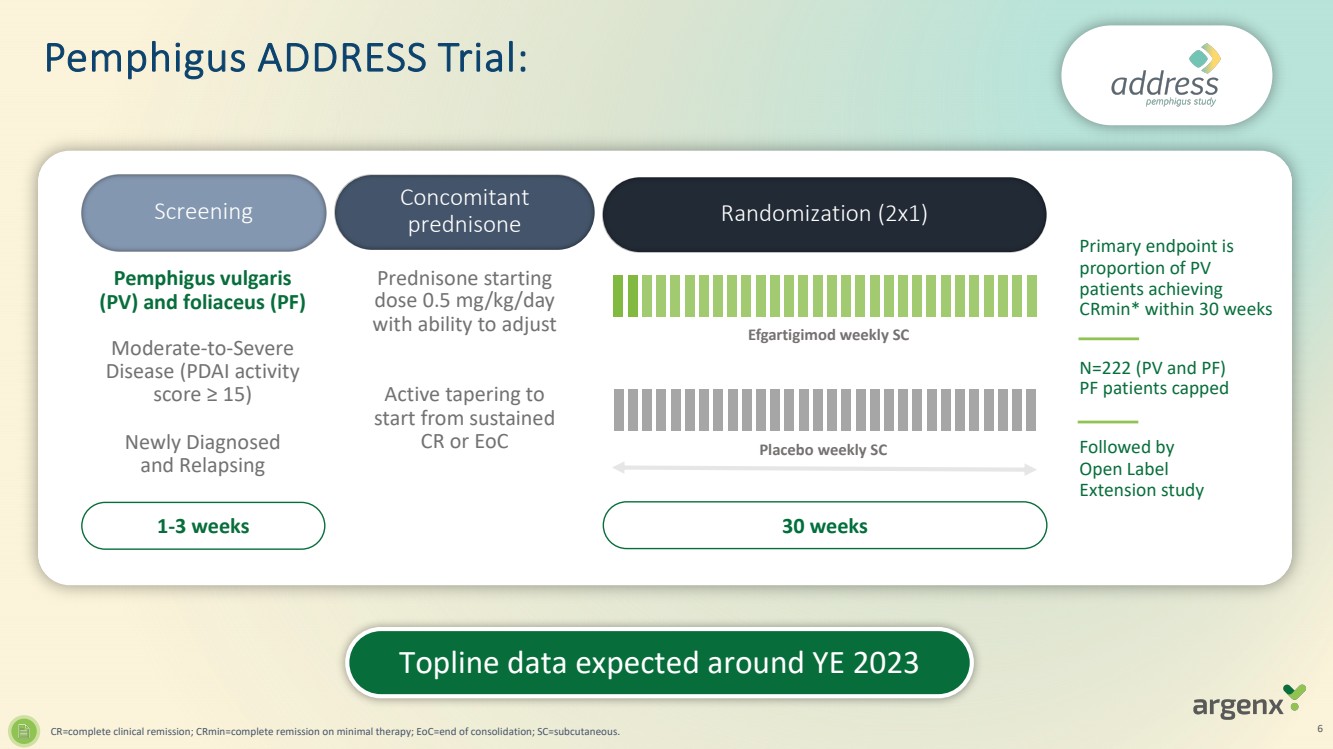
| 1-3 weeks
Screening
Efgartigimod weekly SC
Placebo weekly SC
Pemphigus vulgaris
(PV) and foliaceus (PF)
Moderate-to-Severe
Disease (PDAI activity
score ≥ 15)
Newly Diagnosed
and Relapsing
Prednisone starting
dose 0.5 mg/kg/day
with ability to adjust
Active tapering to
start from sustained
CR or EoC
Concomitant
prednisone Randomization (2x1)
30 weeks
Primary endpoint is
proportion of PV
patients achieving
CRmin* within 30 weeks
N=222 (PV and PF)
PF patients capped
Followed by
Open Label
Extension study
CR=complete clinical remission; CRmin=complete remission on minimal therapy; EoC=end of consolidation; SC=subcutaneous. 6
Pemphigus ADDRESS Trial:
Topline data expected around YE 2023 |

| 7
Steady Cadence of Upcoming Data Readouts
ITP-SC: Topline data expected in 4Q 2023
Pemphigus: Topline data expected around year-end 2023
Bullous Pemphigoid (BP): GO/NO GO decision expected around year-end 2023
Post-COVID Postural Orthostatic Tachycardia Syndrome (PC-POTS): Topline data expected in 1Q 2024
Sjogren’s Syndrome: Topline data expected in first half 2024
Myositis: GO/NO GO decision expected in second half of 2024
Multifocal Motor Neuropathy (MMN): Topline data from Phase 2 (empasiprubart) expected in
2024
clinical |
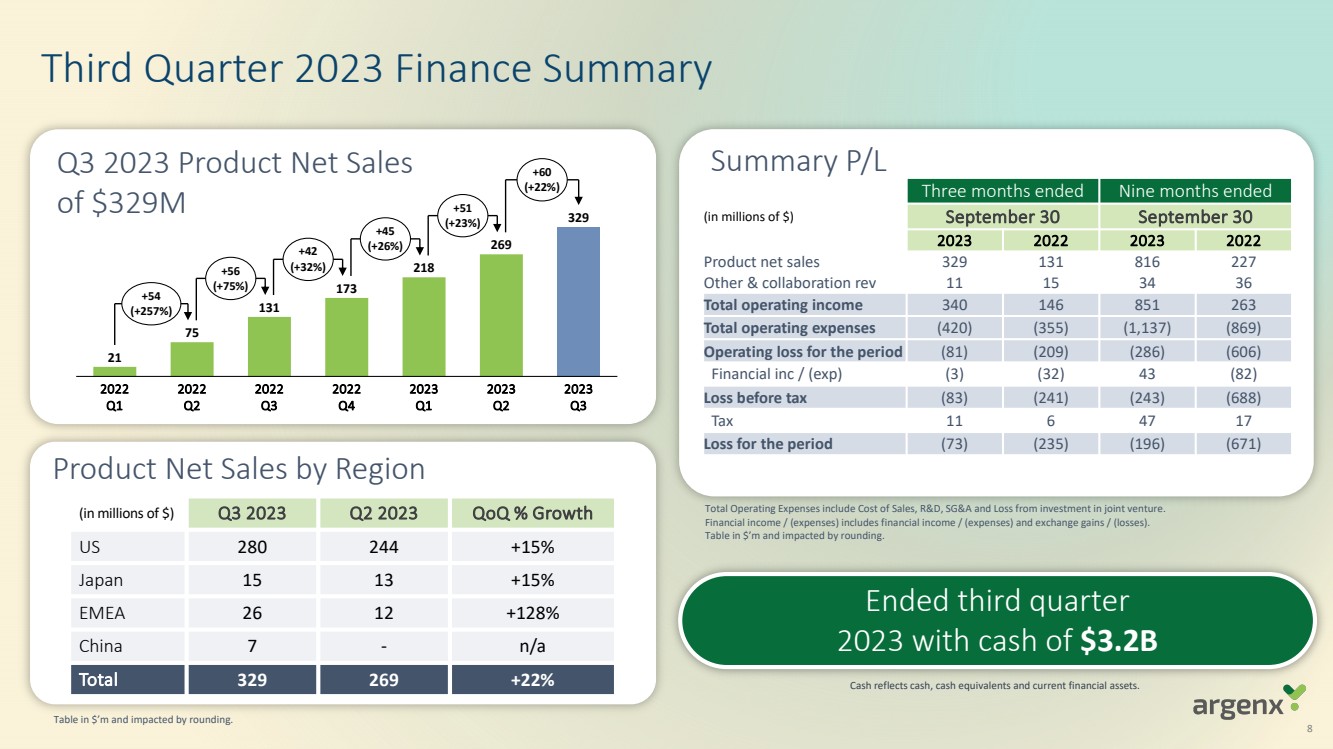
| Q3 2023 Product Net Sales
of $329M
Third Quarter 2023 Finance Summary
(in millions of $) Q3 2023 Q2 2023 QoQ % Growth
US 280 244 +15%
Japan 15 13 +15%
EMEA 26 12 +128%
China 7 - n/a
Total 329 269 +22%
Product Net Sales by Region
21
75
131
173
218
269
329
2022
Q1
2022
Q2
2023
Q2
2023
Q1
2022
Q3
2022
Q4
2023
Q3
+54
(+257%)
+56
(+75%)
+42
(+32%)
+45
(+26%)
+51
(+23%)
+60
(+22%) Three months ended Nine months ended
(in millions of $) September 30 September 30
2023 2022 2023 2022
Product net sales 329 131 816 227
Other & collaboration rev 11 15 34 36
Total operating income 340 146 851 263
Total operating expenses (420) (355) (1,137) (869)
Operating loss for the period (81) (209) (286) (606)
Financial inc / (exp) (3) (32) 43 (82)
Loss before tax (83) (241) (243) (688)
Tax 11 6 47 17
Loss for the period (73) (235) (196) (671)
Summary P/L
Cash reflects cash, cash equivalents and current financial assets.
Total Operating Expenses include Cost of Sales, R&D, SG&A and Loss from investment in joint venture.
Financial income / (expenses) includes financial income / (expenses) and exchange gains / (losses).
Table in $’m and impacted by rounding.
Ended third quarter
2023 with cash of $3.2B
8
Table in $’m and impacted by rounding. |
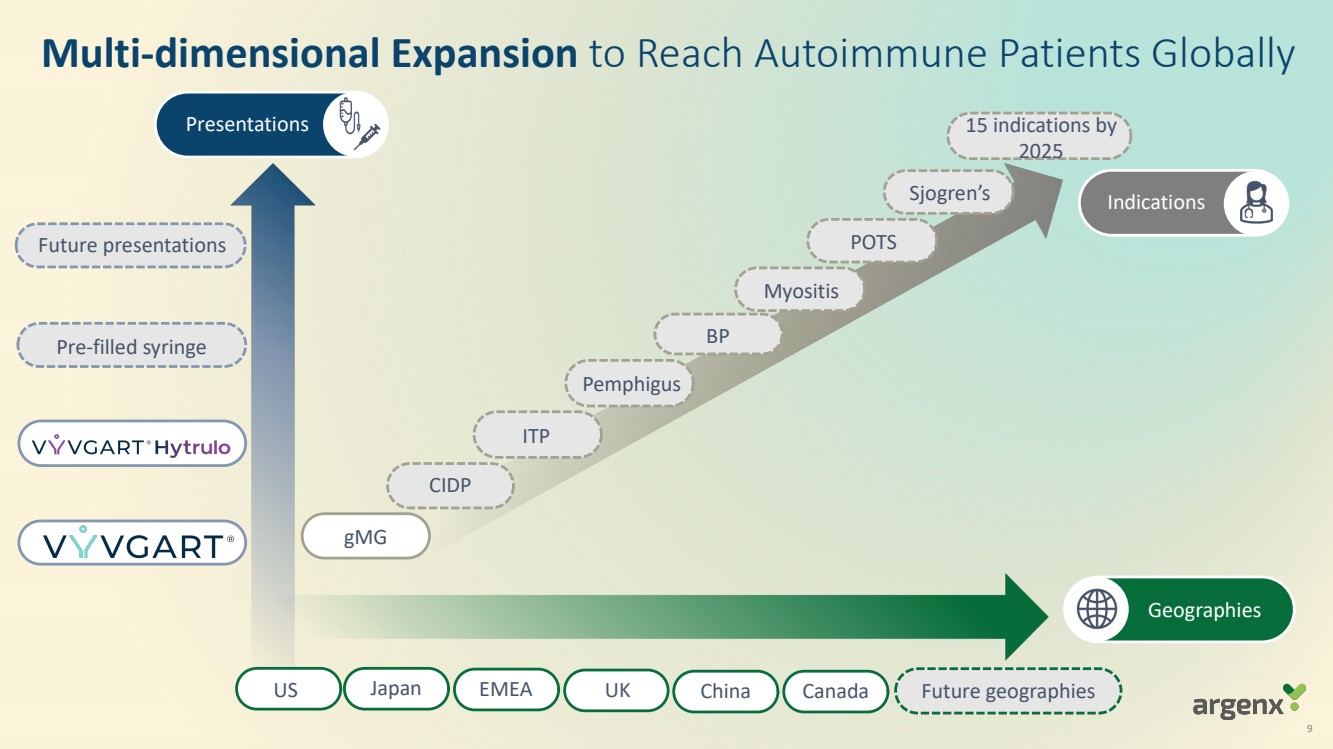
| 9
Geographies
Indications
Presentations
Pre-filled syringe
CIDP
BP
Pemphigus
ITP
gMG
Myositis
US Japan EMEA UK China Future geographies
Future presentations
Multi-dimensional Expansion to Reach Autoimmune Patients Globally
15 indications by
2025
POTS
Sjogren’s
Canada |

| 10
Optimizing Core Launch Strategies
Reaching Broader
gMG Population
Continued shift into
earlier lines
First Hytrulo
policies published
Driving brand loyalty
among prescribers
IN
$
COMBINED NET
816
VYVGART
M
& HYTRULO SALES YTD
Hytrulo contributing to expansion Hytrulo launch driven by
VYVGART-naïve patients
Policies in line with IV
88% of key target prescribers
reached since launch |
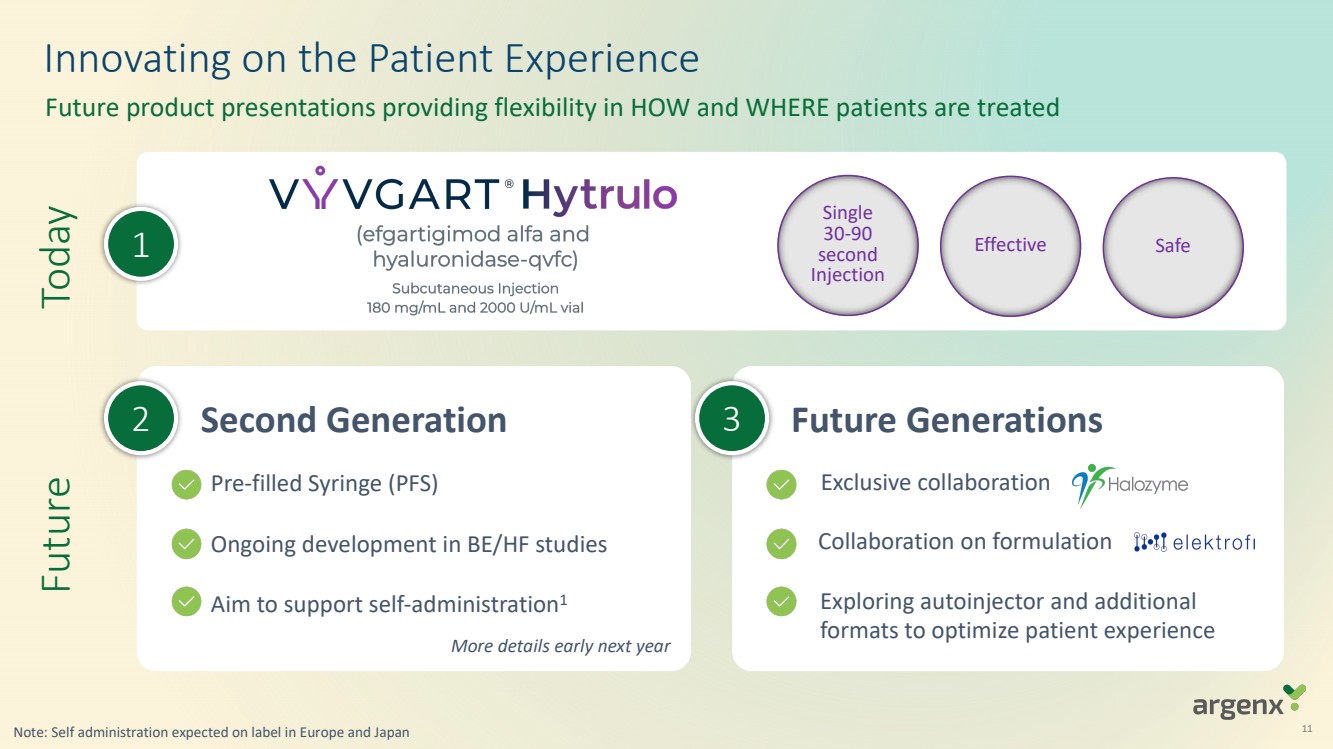
| 11
Today
1
Innovating on the Patient Experience
Future product presentations providing flexibility in HOW and WHERE patients are treated
Single
30-90
second
Injection
Effective Safe
Future
2 Second Generation
Future Generations
Exclusive collaborations with HALO and Elektrofi
on formulation
Exploring autoinjector and additional formats to
optimize patient experience More details early next year
3
Exploring autoinjector and additional
formats to optimize patient experience
Pre-filled Syringe (PFS)
Ongoing development in BE/HF studies
Aim to support self-administration1
Future Generations
Exclusive collaboration
Collaboration on formulation
Note: Self administration expected on label in Europe and Japan |
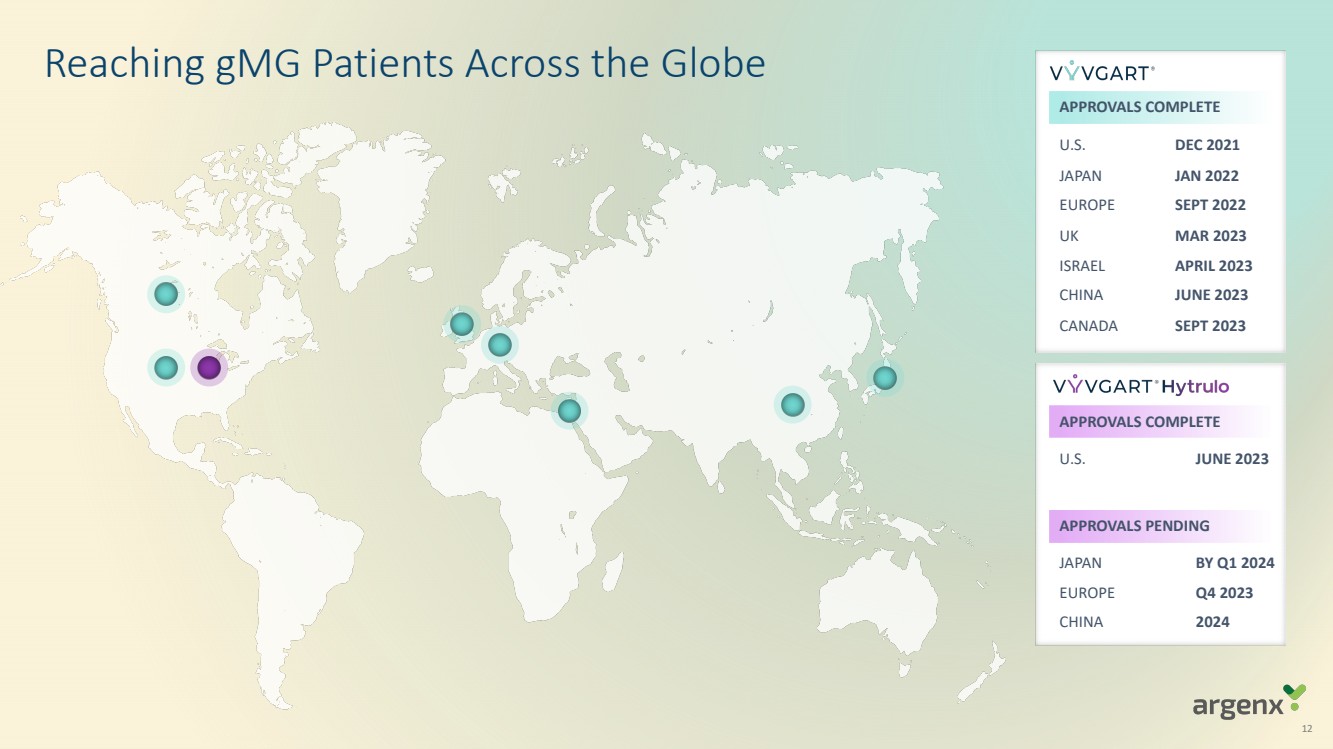
| 12
Reaching gMG Patients Across the Globe
APPROVALS COMPLETE
U.S.
JAPAN
EUROPE
UK
ISRAEL
CHINA
CANADA
DEC 2021
JAN 2022
SEPT 2022
MAR 2023
APRIL 2023
JUNE 2023
SEPT 2023
APPROVALS COMPLETE
U.S. JUNE 2023
APPROVALS PENDING
JAPAN
EUROPE
CHINA
BY Q1 2024
Q4 2023
2024 |

| Bringing Hope to Patients of
New Treatment Option
Advancing Hytrulo in CIDP
I was the type of woman that would run
first thing in the morning before work, and
then CIDP hit, and it was like hitting the
wall at a hundred miles an hour.
Crystal
Living With CIDP
Largest Global CIDP Trial
New standard set for
innovative trial design
Unlocking New Disease
Biology Insights
IgG shown to play significant role
in underlying biology of CIDP
Potential new
treatment modality
First Innovation
in 30+ Years
13
99% rollover into OLE
On track to file sBLA in 2023 with launch targeted in 2024 |

| 14
Our mission
continues…
Humility
Innovation Excellence
Co-Creation Empowerment |
argenx (NASDAQ:ARGX)
Historical Stock Chart
From May 2024 to Jun 2024
argenx (NASDAQ:ARGX)
Historical Stock Chart
From Jun 2023 to Jun 2024