Atossa Therapeutics Further Strengthens Intellectual Property Portfolio with Additional Broad Patent for Endoxifen
14 February 2023 - 1:15AM

Atossa Therapeutics, Inc. (Nasdaq: ATOS), a clinical stage
biopharmaceutical company developing innovative proprietary
medicines to address significant unmet need in cancer, today
announces that the United States Patent and Trademark Office
(USPTO) has granted a new patent (No. 11,572,334) directed to
(Z)-endoxifen encapsulated in an enteric capsule. This patent
further reinforces Atossa’s broad Intellectual Property portfolio
related to (Z)-endoxifen. A short 'explainer' video about the new
patent can be found here: Patent Update - Video Explainer.
Enteric capsules have an acid resistant coating to prevent them
from dissolving when they pass through the stomach. Enteric
capsules are dissolved when they pass through an alkaline
environment, which is usually when they reach the small intestine.
Delivering oral (Z)-endoxifen via an enteric capsule prevents
breakdown of the endoxifen in the stomach.
“We are very pleased with the scope and breadth of this new key
patent,” said Dr. Steven Quay, Atossa’s President and Chief
Executive Officer. “It ensures that in the U.S., Atossa will have
the only (Z)-endoxifen treatment delivered orally with an enteric
capsule, which we believe is the optimal way to deliver the drug.
This new patent, further strengthens our intellectual property
estate and should create long-term stockholder value.”
Atossa is developing its proprietary (Z)-endoxifen in both the
breast cancer treatment and prevention settings. Phase 2 trials are
ongoing with the goal of changing the treatment paradigm for
patients who are not benefiting from currently approved drugs and
helping reduce the incidence of breast cancer.
- Phase 2 “EVANGELINE” study – (Z)-endoxifen and exemestane +
goserelin as neoadjuvant treatment in premenopausal women with
ER+/HER2- breast cancer. The EVANGELINE trial is expected to enroll
approximately 175 patients at up to 25 sites across the United
States and builds on an earlier Phase 2 study that showed treatment
with (Z)-endoxifen reduced Ki-67, a commonly used measure of
cellular proliferation and growth in breast cancer tissue. The
EVANGELINE trial will evaluate safety and tolerability of treatment
with (Z)-endoxifen, reduction in Ki-67 and pathological response.
It will also study whether treatment with (Z)-endoxifen in
premenopausal women could provide clinical benefit without the need
for ovarian suppression.
- Phase 2 “Karisma-Endoxifen” study – (Z)-endoxifen in
premenopausal women with elevated mammographic breast density
(MBD). This randomized, double-blind, placebo-controlled trial of
healthy, pre-menopausal women with increased breast density is
expected to enroll 240 study participants. The treatment cohort
receives daily doses of (Z)-endoxifen for six months, over the
course of which mammograms will be conducted to measure reduction
in MBD. Patients will also be given a mammogram at 24 months to
assess the durability of the MBD changes.
Both trials seek to address significant unmet medical need in
breast cancer. While there are several FDA-approved neoadjuvant
therapies for ER- breast cancers, few good options exist for ER+
patients, which account for approximately 78% of breast cancers.
Current treatment approaches for premenopausal women diagnosed with
ER+ breast cancer include ovarian suppression, which can induce
menopause and dramatically impact a patient’s quality of life.
Additionally, MBD affects more than 10 million women in the United
States and many millions more worldwide. Increased MBD reduces the
ability of mammograms to detect cancer. Studies have also shown
that women with MBD have an increased risk of developing breast
cancer and that the higher the MBD, the higher the incidence of
breast cancer.
“There are a number of research-level medical applications of
endoxifen outside of breast cancer, including in bipolar disorder
disease in adults and in modulating genetically modified,
cell-based therapies for cancer and immune diseases, that may
require access to our endoxifen patents and technologies,”
continued Dr. Quay. “Atossa will monitor these activities carefully
as they approach commercialization, with an eye to establishing
appropriate licensing arrangements for the benefit of Atossa
shareholders.”
ABOUT (Z)-ENDOXIFEN(Z)-endoxifen is the most
active metabolite of the FDA approved Selective Estrogen Receptor
Modulator (SERM), tamoxifen. Studies by others have demonstrated
that the anti-estrogenic effects of tamoxifen are driven in a
concentration-dependent manner by (Z)-endoxifen. In addition to its
anti-estrogen effects, (Z)-endoxifen at higher concentrations has
been shown to target PKCβ1, a known oncogenic protein.
Atossa has developed a proprietary oral formulation of
(Z)-endoxifen that does not require liver metabolism to achieve
therapeutic concentrations and is encapsulated to bypass the
stomach as acidic conditions converts a greater proportion of
(Z)-endoxifen to the inactive (E)-endoxifen. Atossa’s (Z)-endoxifen
has been shown to be well tolerated in Phase 1 studies and in a
small Phase 2 study of women with breast cancer. We currently are
studying our (Z)-endoxifen in healthy women with measurable breast
density and premenopausal women with ER+/HER2- breast cancer.
ABOUT ATOSSA THERAPEUTICSAtossa Therapeutics,
Inc. is a clinical-stage biopharmaceutical company developing
innovative medicines in areas of significant unmet medical need in
oncology with a current focus on breast cancer and lung injury
caused by cancer treatments. For more information, please visit
www.atossatherapeutics.com
CONTACTS:Kyle GuseGeneral Counsel and Chief
Financial Officerkyle.guse@atossainc.com
Eric Van ZantenVP, Investor and Public
Relations610-529-6219eric.vanzanten@atossainc.com
FORWARD LOOKING STATEMENTSForward-looking
statements in this press release, which Atossa undertakes no
obligation to update, are subject to risks and uncertainties that
may cause actual results to differ materially from the anticipated
or estimated future results, including the risks and uncertainties
associated with any variation between interim and final clinical
results, actions and inactions by the FDA, the outcome or timing of
regulatory approvals needed by Atossa including those needed to
commence studies of AT-H201 and (Z)-endoxifen, lower than
anticipated rate of patient enrollment, estimated market size of
drugs under development, the safety and efficacy of Atossa’s
products, performance of clinical research organizations and
investigators, obstacles resulting from proprietary rights held by
others such as patent rights, whether reduction in breast density
or in Ki-67 or any other result from a neoadjuvant study is an
approvable endpoint for (Z)-endoxifen, whether Atossa can complete
acquisitions, and other risks detailed from time to time in
Atossa’s filings with the Securities and Exchange Commission,
including without limitation its periodic reports on Form 10-K and
10-Q, each as amended and supplemented from time to time.
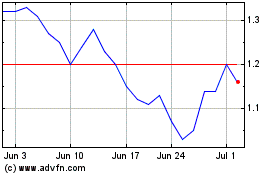
Atossa Therapeutics (NASDAQ:ATOS)
Historical Stock Chart
From Mar 2024 to Apr 2024
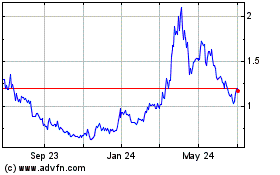
Atossa Therapeutics (NASDAQ:ATOS)
Historical Stock Chart
From Apr 2023 to Apr 2024